Influence of Maternal Care on Behavioural Development of Domestic Dogs (Canis Familiaris) Living in a Home Environment
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Analysis of Maternal Care
2.3. Behavioural Tests for Puppies
2.4. Statistical Analysis
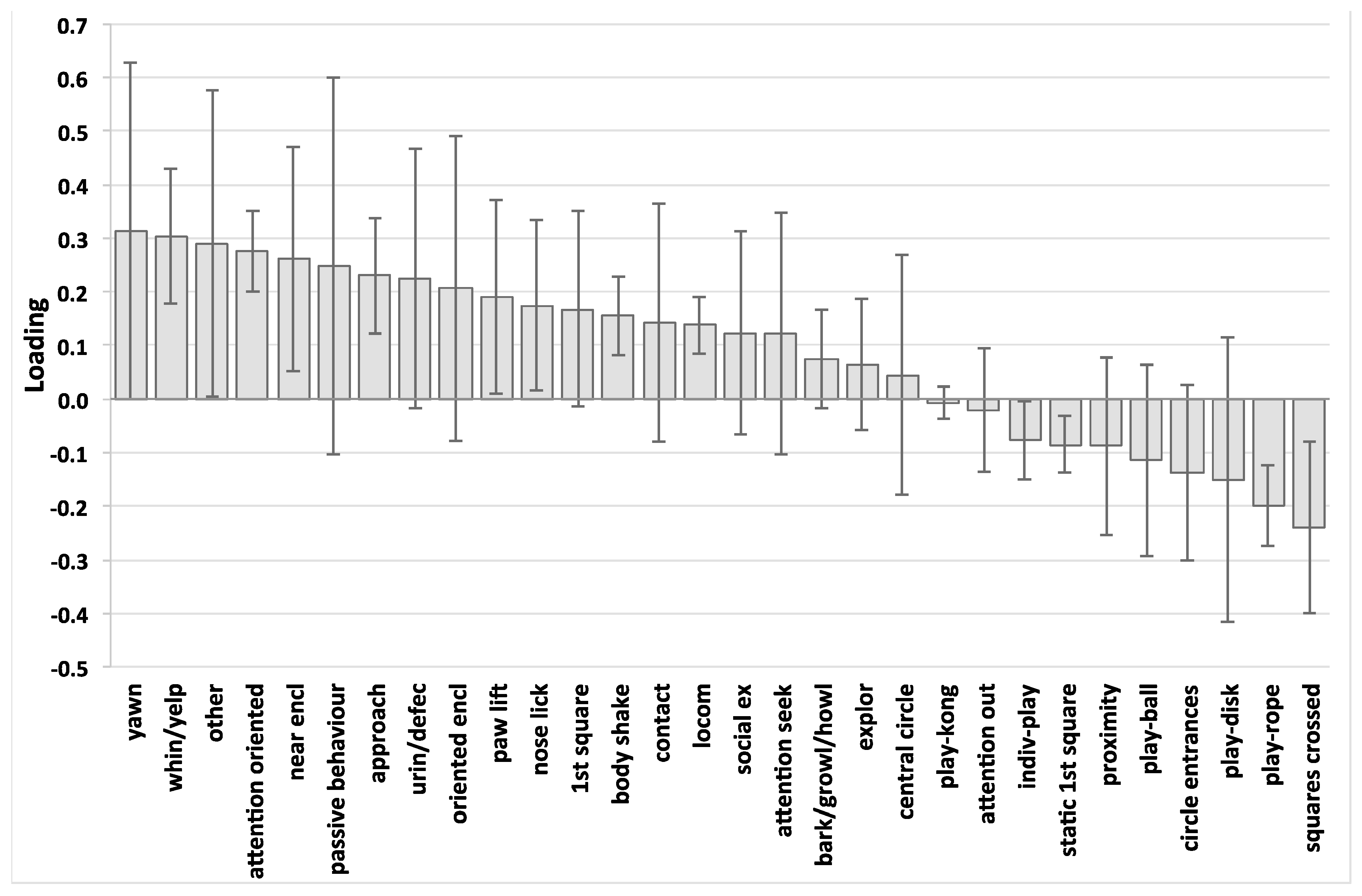
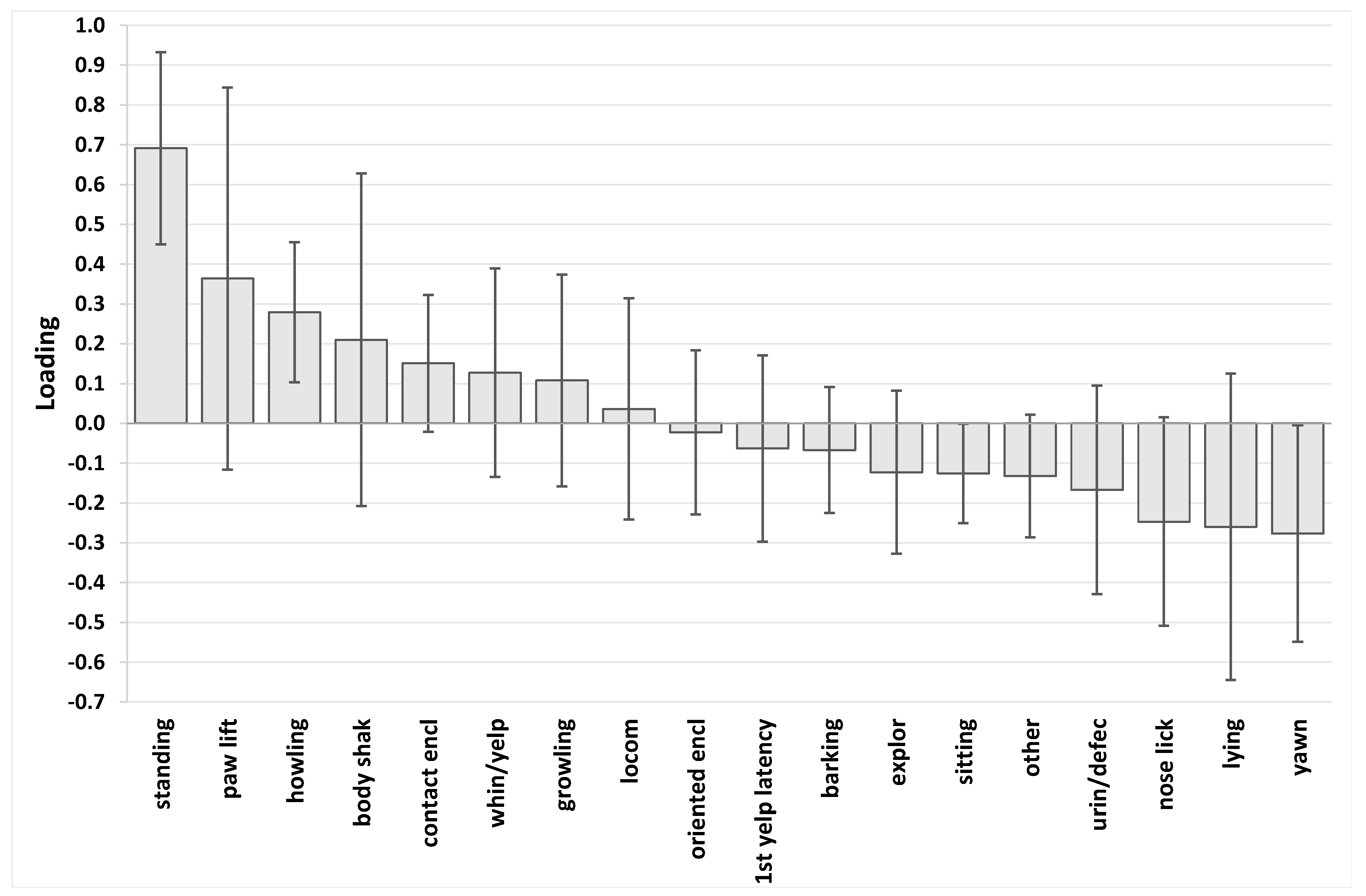
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Meaney, M.J. Maternal care, gene expression, and the transmission of individual differences in stress reactivity across generations. Annu. Rev. Neurosci. 2001, 24, 1161–1192. [Google Scholar] [CrossRef] [PubMed]
- Champagne, F.A.; Francis, D.D.; Mar, A.; Meaney, M.J. Variations in maternal care in the rat as a mediating influence for the effects of environment on development. Physiol. Behav. 2003, 79, 359–371. [Google Scholar] [CrossRef]
- Czerwinski, V.H.; Smith, B.P.; Hynd, P.I.; Hazel, S.J. The influence of maternal care on stress-related behaviors in domestic dogs: What can we learn from the rodent literature? J. Vet. Behav. Clin. Appl. Res. 2016, 14, 52–59. [Google Scholar] [CrossRef]
- Liu, D.; Diorio, J.; Tannenbaum, B.; Caldji, C.; Francis, D.; Freedman, A.; Sharma, S.; Pearson, D.; Plotsky, P.M.; Meaney, M.J. Maternal care, hippocampal glucocorticoid receptors, and hypothalamic-pituitary-adrenal responses to stress. Science 1997, 277, 1659–1662. [Google Scholar] [CrossRef] [PubMed]
- Caldji, C.; Tannenbaum, B.; Sharma, S.; Francis, D.; Plotsky, P.M.; Meaney, M.J. Maternal care during infancy regulates the development of neural systems mediating the expression of fearfulness in the rat. Proc. Natl. Acad. Sci. USA 1998, 95, 5335–5340. [Google Scholar] [CrossRef] [PubMed]
- Caldji, C.; Diorio, J.; Meaney, M.J. Variations in maternal care in infancy regulate the development of stress reactivity. Biol. Psychiatry 2000, 48, 1164–1174. [Google Scholar] [CrossRef]
- Caldji, C.; Francis, D.; Sharma, S.; Plotsky, P.M.; Meaney, M.J. The effects of early rearing environment on the development of GABAA and central benzodiazepine receptor levels and novelty-induced fearfulness in the rat. Neuropsychopharmacology 2000, 22, 219–229. [Google Scholar] [CrossRef]
- Johnson, E.O.; Kamilaris, T.C.; Calogero, A.E.; Gold, P.W.; Chrousos, G.P. Effects of early parenting on growth and development in a small primate. Pediatr. Res. 1996, 39, 999–1005. [Google Scholar] [CrossRef] [PubMed]
- Birnie, A.K.; Taylor, J.H.; Cavanaugh, J.; French, J.A. Quality of maternal and paternal care predicts later stress reactivity in the cooperatively-breeding marmoset (Callithrix geoffroyi). Psychoneuroendocrinology 2013, 38, 3003–3014. [Google Scholar] [CrossRef] [PubMed]
- Ainsworth, M.D.S.; Blehar, M.C.; Waters, E.; Wall, S.N. Patterns of Attachment: A Psychological Study of the Strange Situation; Psychology Press: New York, NY, USA, 2015. [Google Scholar]
- Bowlby, J. A Secure Base: Clinical Applications of Attachment Theory; Taylor & Francis: Oxfordshire, UK, 2005. [Google Scholar]
- Jensen, C.L.; Champagne, F.A. Epigenetic and neurodevelopmental perspectives on variation in parenting behavior. Parent. Sci. Pract. 2012, 12, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Masís-Calvo, M.; Sequeira-Cordero, A.; Mora-Gallegos, A.; Fornaguera-Trías, J. Behavioral and neurochemical characterization of maternal care effects on juvenile Sprague-Dawley rats. Physiol. Behav. 2013, 118, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Gunnar, M.; Quevedo, K. The neurobiology of stress and development. Annu. Rev. Psychol. 2007, 58, 145–173. [Google Scholar] [CrossRef] [PubMed]
- Maestripieri, D. Effects of early experience on female behavioural and reproductive development in rhesus macaques. Proc. R. Soc. Lond. B Biol. Sci. 2005, 272, 1243–1248. [Google Scholar] [CrossRef] [PubMed]
- Tarullo, A.R.; Gunnar, M.R. Child maltreatment and the developing HPA axis. Horm. Behav. 2006, 50, 632–639. [Google Scholar] [CrossRef] [PubMed]
- Lupien, S.J.; McEwen, B.S.; Gunnar, M.R.; Heim, C. Effects of stress throughout the lifespan on the brain, behaviour and cognition. Nat. Rev. Neurosci. 2009, 10, 434–445. [Google Scholar] [CrossRef] [PubMed]
- Albers, E.M.; Marianne Riksen-Walraven, J.; Sweep, F.C.; de Weerth, C. Maternal behavior predicts infant cortisol recovery from a mild everyday stressor. J. Child Psychol. Psychiatry 2008, 49, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Rheingold, H.L. Maternal behavior in the dog. In Maternal Behavior in Mammals; Wiley: New York, NY, USA, 1963; pp. 169–202. [Google Scholar]
- Pal, S.K. Parental care in free-ranging dogs, Canis familiaris. Appl. Anim. Behav. Sci. 2005, 90, 31–47. [Google Scholar] [CrossRef]
- Piñol, M.J.; Cornelles, S.; Fatjó, J.; Ruiz de la Torre, J.L.; Amat, M.; Manteca, X. Effects of early separation and handling of puppies on maternal licking in the bitch. In Current Issues and Research in Veterinary Behavioral Medicine, Proceedings of the Fifth Veterinary Behavior Meeting, Minneapolis, MN, USA, 14–16 July 2005; Purdue University Press: West Lafayette, IN, USA, 2005. [Google Scholar]
- Arteaga, L.; Rödel, H.G.; Elizalde, M.T.; González, D.; Hudson, R. The pattern of nipple use before weaning among littermates of the domestic dog. Ethology 2013, 119, 12–19. [Google Scholar] [CrossRef]
- Guardini, G.; Bowen, J.; Raviglione, S.; Farina, R.; Gazzano, A. Maternal behaviour in domestic dogs: A comparison between primiparous and multiparous dogs. Dog Behav. 2015, 1, 22–33. [Google Scholar]
- Bray, E.E.; Sammel, M.D.; Cheney, D.L.; Serpell, J.A.; Seyfarth, R.M. Characterizing early maternal style in a population of guide dogs. Front. Psychol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Czerwinski, V.H.; Smith, B.P.; Hynd, P.I.; Hazel, S.J. Sampling maternal care behaviour in domestic dogs: What’s the best approach? Behav. Process. 2017, 140, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Foyer, P.; Wilsson, E.; Jensen, P. Levels of maternal care in dogs affect adult offspring temperament. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Guardini, G.; Mariti, C.; Bowen, J.; Fatjó, J.; Ruzzante, S.; Martorell, A.; Sighieri, C.; Gazzano, A. Influence of morning maternal care on the behavioural responses of 8-week-old Beagle puppies to new environmental and social stimuli. Appl. Anim. Behav. Sci. 2016, 181, 137–144. [Google Scholar] [CrossRef]
- Bray, E.E.; Sammel, M.D.; Cheney, D.L.; Serpell, J.A.; Seyfarth, R.M. Effects of maternal investment, temperament, and cognition on guide dog success. Proc. Natl. Acad. Sci. USA 2017, 114, 9128–9133. [Google Scholar] [CrossRef] [PubMed]
- Tiira, K.; Lohi, H. Early life experiences and exercise associate with canine anxieties. PLoS ONE 2015, 10, e0141907. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, L.; Trygg, J.; Wold, S. CV-ANOVA for significance testing of PLS and OPLS® models. J. Chemom. 2008, 22, 594–600. [Google Scholar] [CrossRef]
- Mogi, K.; Nagasawa, M.; Kikusui, T. Developmental consequences and biological significance of mother–infant bonding. Prog. Neuropsychopharmacol. Biol. Psychiatry 2011, 35, 1232–1241. [Google Scholar] [CrossRef] [PubMed]
- Coe, C.L.; Mendoza, S.P.; Smotherman, W.P.; Levine, S. Mother-infant attachment in the squirrel monkey: Adrenal response to separation. Behav. Biol. 1978, 22, 256–263. [Google Scholar] [CrossRef]
- Mineka, S.; Suomi, S.J. Social separation in monkeys. Psychol. Bull. 1978, 85, 1376–1400. [Google Scholar] [CrossRef] [PubMed]
- Nowak, R.; Boivin, X. Filial attachment in sheep: Similarities and differences between ewe-lamb and human-lamb relationships. Appl. Anim. Behav. Sci. 2015, 164, 12–28. [Google Scholar] [CrossRef]
- Elliot, O.; Scott, J.P. The development of emotional distress reactions to separation, in puppies. J. Genet. Psychol. 1961, 99, 3–22. [Google Scholar] [CrossRef] [PubMed]
- Previde, E.P.; Ghirardelli, G.; Marshall-Pescini, S.; Valsecchi, P. Intraspecific attachment in domestic puppies (Canis familiaris). J. Vet. Behav. Clin. Appl. Res. 2009, 4, 89–90. [Google Scholar] [CrossRef]
- Nagasawa, M.; Shibata, Y.; Yonezawa, A.; Morita, T.; Kanai, M.; Mogi, K.; Kikusui, T. The behavioral and endocrinological development of stress response in dogs. Dev. Psychobiol. 2014, 56, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Mariti, C.; Ricci, E.; Zilocchi, M.; Gazzano, A. Owners as a secure base for their dogs. Behaviour 2013, 150, 1275–1294. [Google Scholar] [CrossRef]
- Seay, B.; Hansen, E.; Harlow, H.F. Mother-Infant separation in monkeys. J. Child Psychol. Psychiatry 1962, 3, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Mason, W.A. Chapter 9—Determinants of social behavior in young chimpanzees. In Behavior of Nonhuman Primates; Schrier, A.M., Harlow, H.F., Stollnitz, F., Eds.; Academic Press: New York, NY, USA, 1965; pp. 335–364. [Google Scholar]
- Mariti, C.; Carlone, B.; Ricci, E.; Sighieri, C.; Gazzano, A. Intraspecific attachment in adult domestic dogs (Canis familiaris): Preliminary results. Appl. Anim. Behav. Sci. 2014, 152, 64–72. [Google Scholar] [CrossRef]
- Mariti, C.; Carlone, B.; Votta, E.; Ricci, E.; Sighieri, C.; Gazzano, A. Intraspecific relationships in adult domestic dogs (Canis familiaris) living in the same household: A comparison of the relationship with the mother and an unrelated older female dog. Appl. Anim. Behav. Sci. 2017, 194, 62–66. [Google Scholar] [CrossRef]
- Pettijohn, T.F.; Wong, T.W.; Ebert, P.D.; Scott, J.P. Alleviation of separation distress in 3 breeds of young dogs. Dev. Psychobiol. 1977, 10, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Francis, D.D.; Young, L.J.; Meaney, M.J.; Insel, T.R. Naturally occurring differences in maternal care are associated with the expression of oxytocin and vasopressin (V1a) receptors: Gender differences. J. Neuroendocrinol. 2002, 14, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Starr-Phillips, E.J.; Beery, A.K. Natural variation in maternal care shapes adult social behavior in rats. Dev. Psychobiol. 2014, 56, 1017–1026. [Google Scholar] [CrossRef] [PubMed]
- Perkeybile, A.M.; Griffin, L.L.; Bales, K.L. Natural variation in early parental care correlates with social behaviors in adolescent prairie voles (Microtus ochrogaster). Front. Behav. Neurosci. 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Numan, M.; Insel, T.R. The Neurobiology of Parental Behavior; Springer: New York, NY, USA, 2003. [Google Scholar]
- Maestripieri, D.; Lindell, S.G.; Ayala, A.; Gold, P.W.; Higley, J.D. Neurobiological characteristics of rhesus macaque abusive mothers and their relation to social and maternal behavior. Neurosci. Biobehav. Rev. 2005, 29, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.R.; Park, J.S. Impact of attachment, temperament and parenting on human development. Korean J. Pediatr. 2012, 55, 449–454. [Google Scholar] [CrossRef] [PubMed]
- Feldman, R. Oxytocin and social affiliation in humans. Horm. Behav. 2012, 61, 380–391. [Google Scholar] [CrossRef] [PubMed]
- Winslow, J.T.; Noble, P.L.; Lyons, C.K.; Sterk, S.M.; Insel, T.R. Rearing effects on cerebrospinal fluid oxytocin concentration and social buffering in Rhesus monkeys—ProQuest. Neuropsychopharmacology 2003, 28, 910–918. [Google Scholar] [CrossRef] [PubMed]
- Lieberwirth, C.; Wang, Z. Social bonding: Regulation by neuropeptides. Front. Neurosci. 2014, 8. [Google Scholar] [CrossRef] [PubMed]
- Moore, C.L.; Power, K.L. Variation in maternal care and individual differences in play, exploration, and grooming of juvenile Norway rat offspring. Dev. Psychobiol. 1992, 25, 165–182. [Google Scholar] [CrossRef] [PubMed]
- Parent, C.I.; Meaney, M.J. The influence of natural variations in maternal care on play fighting in the rat. Dev. Psychobiol. 2008, 50, 767–776. [Google Scholar] [CrossRef] [PubMed]
- Fredericson, E. Distributed versus massed experience in a traumatic situation. J. Abnorm. Soc. Psychol. 1950, 45, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.; Scott, J.P.; Cherner, M.; Denenberg, V.H. Effects of restraint and isolation on yelping in puppies. Anim. Behav. 1960, 8, 1–5. [Google Scholar] [CrossRef]
- Mariti, C.; Gazzano, A.; Moore, J.L.; Baragli, P.; Chelli, L.; Sighieri, C. Perception of dogs’ stress by their owners. J. Vet. Behav. Clin. Appl. Res. 2012, 7, 213–219. [Google Scholar] [CrossRef]
- Francis, D.D.; Meaney, M.J. Maternal care and the development of stress responses. Curr. Opin. Neurobiol. 1999, 9, 128–134. [Google Scholar] [CrossRef]
- Francis, D.; Diorio, J.; Liu, D.; Meaney, M.J. Nongenomic transmission across generations of maternal behavior and stress responses in the rat. Science 1999, 286, 1155–1158. [Google Scholar] [CrossRef] [PubMed]
- Sequeira-Cordero, A.; Masís-Calvo, M.; Mora-Gallegos, A.; Fornaguera-Trías, J. Maternal behavior as an early modulator of neurobehavioral offspring responses by Sprague-Dawley rats. Behav. Brain Res. 2013, 237, 63–70. [Google Scholar] [CrossRef] [PubMed]
| Litter | Breed | Mother’s Age (Months) | Parity | N° Puppies | Males | Females | Provenance |
|---|---|---|---|---|---|---|---|
| 1 | Weimaraner | 18 | 1 | 5 | 3 | 2 | professional breeder |
| 2 | Belgian Shepherd Groenendal | 72 | 1 | 4 | 4 | 0 | professional breeder |
| 3 | Cross breed | 24 | 1 | 3 | 2 | 1 | not professional breeder |
| 4 | Belgian Shepherd Groenendal | 72 | 3 | 7 | 4 | 3 | professional breeder |
| 5 | Short Haired Dachshund | 60 | 3 | 5 | 2 | 3 | professional breeder |
| 6 | Belgian Shepherd Groenendal | 84 | 2 | 5 | 4 | 1 | professional breeder |
| 7 | German Shepherd | 72 | 3 | 7 | 2 | 5 | not professional breeder |
| 8 | Labrador Retriever | 24 | 1 | 7 | 5 | 2 | not professional breeder |
| 9 | Boxer | 84 | 3 | 9 | 1 | 8 | professional breeder |
| 10 | Border Collie | 24 | 1 | 6 | 4 | 2 | not professional breeder |
| 11 | Deutsch Drathaar | 18 | 1 | 5 | 3 | 2 | professional breeder |
| 12 | Boxer | 32 | 1 | 8 | 3 | 5 | not professional breeder |
| mean ± standard deviation | 48.7 ± 27.4 | 1.7 ± 1.0 | 5.9 ± 1.7 | 3.0 ± 1.2 | 2.8 ± 2.2 |
| Var ID (Primary) | Loading |
|---|---|
| MEAN__Puppy_contact | 0.635562 |
| MEAN__Puppy_nursing | 0.622256 |
| MEAN__Puppy_licking | −0.047369 |
| MEAN__Puppy_licking ag | 0.454549 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guardini, G.; Bowen, J.; Mariti, C.; Fatjó, J.; Sighieri, C.; Gazzano, A. Influence of Maternal Care on Behavioural Development of Domestic Dogs (Canis Familiaris) Living in a Home Environment. Animals 2017, 7, 93. https://doi.org/10.3390/ani7120093
Guardini G, Bowen J, Mariti C, Fatjó J, Sighieri C, Gazzano A. Influence of Maternal Care on Behavioural Development of Domestic Dogs (Canis Familiaris) Living in a Home Environment. Animals. 2017; 7(12):93. https://doi.org/10.3390/ani7120093
Chicago/Turabian StyleGuardini, Giovanna, Jon Bowen, Chiara Mariti, Jaume Fatjó, Claudio Sighieri, and Angelo Gazzano. 2017. "Influence of Maternal Care on Behavioural Development of Domestic Dogs (Canis Familiaris) Living in a Home Environment" Animals 7, no. 12: 93. https://doi.org/10.3390/ani7120093
APA StyleGuardini, G., Bowen, J., Mariti, C., Fatjó, J., Sighieri, C., & Gazzano, A. (2017). Influence of Maternal Care on Behavioural Development of Domestic Dogs (Canis Familiaris) Living in a Home Environment. Animals, 7(12), 93. https://doi.org/10.3390/ani7120093





