Potential of Biological Processes to Eliminate Antibiotics in Livestock Manure: An Overview
Abstract
:Simple Summary
Abstract
1. Introduction
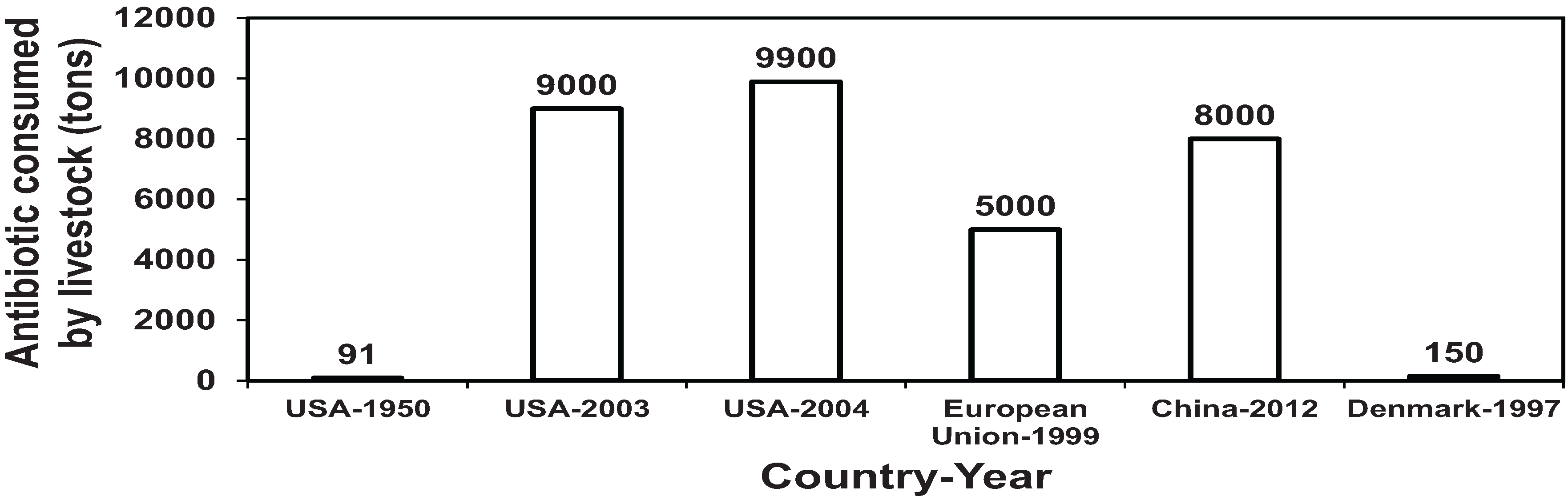
1.1. Antibiotic Consumption in Livestock Industry
1.2. Types of Antibiotics Used in Livestock
- b-Lactams: penicillins: amoxicillin, ampicillin, benzylpenicillin, cloxacilin, dicloxacilin, flucloxacillin, methicillin, mezlocillin, nafcillin, oxacillin, piperacillin, phenoxymethylcillin.
- Macrolides: azithromycin, clarithromycin, clindamycin, erythromycin, roxithromycin, spiramycin, tylosin, vancomycin.
- Sulphonamides: sulphadimidine, sulphamethoxazole.
- Trimethoprim.
- Fluorochinolones: ciprofloxacin, ofloxacin.
- Tetracyclines: chlortetracycline, doxycycline, oxytetracycline, tetracycline.
- Polyether antibiotic: monensin
1.3. Excretion of Antibiotics in Livestock Manure
| Antibiotic | Source of manure | Excretion level (%) | Status | Reference |
|---|---|---|---|---|
| Chlortetracycline | Steers feces | 75 | Not reported | [24] |
| Tetracycline | Animal feces | 25 | Not reported | [25] |
| Tylosin | Urine | 50–60 | Unchanged | [25] |
| Oxytetracycline | Castrate sheeps | 21 | Unchanged | [26] |
| Chlortetracycline | Young bulls | 17–75 | Unchanged | [26] |
| Tylosin | Pigs | 40 | Unaltered or as potent metabolites | [27] |
| Monensin | Beef cattle feces | 40% | Unchanged | [28] |
| Virginiamycin | Piggeries liquid manure | 20 | After several days of storage | [29] |
| Oxytetracycline | Calves manure (feces, urine, and bedding) | 23 | Not reported | [30] |
1.4. Concentration of Antibiotics in Livestock Manure
| Antibiotic | Matrix | Concentration | Reference |
|---|---|---|---|
| Oxytetracycline | Manure | 136 mg·L−1 | [14] |
| Chlortetracycline | 46 mg·L−1 | ||
| Tetracycline | Swine manure | 98 mg·L−1 | [11] |
| Oxytetracycline | 354 mg·L−1 | ||
| Chlortetracycline | 139 mg·L−1 | ||
| Doxycycline | 37 mg·L−1 | ||
| Sulfadiazine | 7.1 mg·L−1 | ||
| Tetracycline | Swine manure | 30 mg·kg−1 DM | [18] |
| Sulphonamides | 2 mg·kg−1 DM | ||
| Tylosin | Fresh calf manure | 0.11 mg·kg−1 | [35] |
| Oxytetracycline | 10 mg·kg−1 | ||
| Chlortetracycline, | Beef manure stockpile | 6.6 mg·kg−1 | [36] |
| Monensin | 120 mg·kg−1 | ||
| Tylosin | 8.1 mg·kg−1 | ||
| Oxytetracycline | Cow manure | 0.5–200 mg·L−1 | [38] |
| Chlortetracycline | Swine manure | 764.4 mg·L−1 | [12] |
| Chlortetracycline | Swine manure storage lagoon | 1 mg·L−1 | [37] |
| Oxytetracycline | 0.41 mg·L−1 |
1.5. Environmental Transport of Antibiotics from Livestock Manure
2. Persistence and Biodegradation of Antibiotics during Biological Processes of Manure Treatment
2.1. Persistence of Antibiotics in Manure
| Antibiotic | Medium matrix | Half-life (days unlessindicated otherwise) | Reference |
|---|---|---|---|
| Tetracycline | Biosolids storage | 37 to >77 | [49] |
| Tetracycline | Stored feedlot manure | 17.2 | [50] |
| Chlortetracycline | Composted manure | 3 | [51] |
| Chlortetracycline | Dairy manure | 6.8 | [50] |
| Chlortetracycline | Stored feedlot manure | 13.5 | [50] |
| Oxytetracycline | Stockpiled fresh manure (low-intensity composting) | 21 | [35] |
| Oxytetracycline | Dairy manure | 17.7 | [50] |
| Oxytetracycline | Stored feedlot manure | 31.1 | [50] |
| Oxytetracycline | Horse manure | 8.4 | [50] |
| Tylosin | Aerobic soil-manure slurry | 3.3–8.1 | [41] |
| Olaquindox | Aerobic soil-manure slurry | 5.8–8.8 | [41] |
| Metronidazole | Aerobic soil-manure slurry | 13.1–26.9 | [41] |
| Erythromycin | Storage of pig manure | 41 | [52] |
| Erythromycin | Biosolids storage | 7.0-17 | [49] |
| Roxithromycin | Storage of pig manure | 130 | [52] |
| Salinomycin | Storage of pig manure | 6 | [52] |
| Doxycycline | Biosolids storage | 53 to >77 | [49] |
| Clindamycin | Biosolids storage | 1.0–1.6 | [49] |
| Clarithromycin | Biosolids storage | 1.1–1.9 | [49] |
| Chemical group | Half-life (d) | Persistence class |
|---|---|---|
| Aminoglycosides | 30 | Moderately persistent |
| β-lactams | 5 | Slightly persistent |
| Macrolides | <2 to 21 | Impersistent to slightly persistent |
| Quinolones | 100 | Very persistent |
| Sulphonamides | <8 to 30 | Slightly to moderately persistent |
| Tetracyclines | 100 | Very persistent |
2.2. Biodegradation Level of Antibiotics in Manure Biological Treatment
| Treatment | Antibiotic | Concentration | Observed reduction | Reference |
|---|---|---|---|---|
| I. Anaerobic digestion | ||||
| Anaerobic digestion of swine manure 21 days | Chlortetracycline | [62] | ||
| 5.9 mg·L−1 | 98% (55 °C) | |||
| Anaerobic digestion of cattle manure (28 days) | Monensin | [62] | ||
| 0.30 mg·L−1 | 27% (55 °C) | |||
| Batch anaerobic digestion | Oxytetracycline | 20 mg·L−1 | 55%–73% at 37 °C | [63] |
| Anaerobic sequence batch reactor (ASBR) | Tylosin A | [1] | ||
| 5.8 mg·kg−1 | Decreased to 0.01 mg·L−1 in 48 h | |||
| Swine manure from lagoons | Tylosin | 0–400 mg·kg−1 | 95%–75% | [64] |
| II. Composting | ||||
| Composting (22–35 days) | Chlortetracycline | 1.5 mg·kg−1 | 99% | [65] |
| [65] | ||||
| Tylosin | 3.7 mg·kg−1 | 54% | ||
| Sulfamethazine | 10.8 mg·kg−1 | –76% | [65] | |
| Composting beef manure (35 days) abiotic removal | Oxytetracycline | 115 μg·g−1 DM | 99% (laboratory)25% (22 °C) | [51] |
| Composting | 20 mg·L−1 | [13] | ||
| Carbamazepine | 37% | |||
| III. Manure amended soil | ||||
| Soil | [66] | |||
| Chlortetracycline | 4.7 µg·kg−1 | 0%0% | ||
| Sulphanilamide | 0.25–1.0 mg·L−1 | 0% | [67] | |
| Tylosin | 5.6 µg·L−1 | 0% | [68] | |
| Erythromecin | 5.6 µg·L−1 | 25% | [68] | |
| Storage | [46] | |||
| Difloxacin | 17.6 mg·L−1 | 7% (10 °C and 20 °C) | ||
2.2.1. Tetracyclines
2.2.2. Tylosin
2.2.3. Other Antibiotics
2.4. Metabolites
3. Required Future Research
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Angenent, L.T.; Mau, M.; George, U.; Zahn, J.A.; Raskin, L. Effect of the presence of the antimicrobial tylosin in swine waste on anaerobic treatment. Water Res. 2008, 42, 2377–2384. [Google Scholar] [CrossRef]
- Kemper, N.; Färber, H.; Skutlarek, D.; Krieter, J. Analysis of antibiotic residues in liquid manure and leachate of dairy farms in northern germany. Agr. Water Manag. 2008, 95, 1288–1292. [Google Scholar] [CrossRef]
- Boxall, A.B.A.; Fogg, L.A.; Blackwell, P.A.; Blackwell, P.; Kay, P.; Pemberton, E.J.; Croxford, A. Veterinary medicines in the environment. Rev. Environ. Contam. Toxicol. 2004, 180, 1–91. [Google Scholar]
- Hamscher, G.; Pawelzick, H.; Sczesny, S.; Nau, H.; Hartung, J. Antibiotics in dust originating from a pig-fattening farm: A new source of health hazard for farmers? Environ. Health Perspect. 2003, 111, 1590–1594. [Google Scholar] [CrossRef]
- Dolliver, H.; Kumar, K.; Gupta, S. Sulfamethazine uptake by plants from manure-amended soil. J. Environ. Qual. 2007, 36, 1224–1230. [Google Scholar] [CrossRef]
- Mellon, M.; Benbrook, C.; Benbrook, K. Hogging it: Estimates of Antimicrobial Abuse in Livestock; Union of Concerned Scientists: Cambridge, MA, USA, 2001. [Google Scholar]
- Sarmah, A.K.; Meyer, M.T.; Boxall, A.B.A. A global perspective on the use, sales, exposure pathways, occurrence, fate and effects of veterinary antibiotics (VAs) in the environment. Chemosphere 2006, 65, 725–759. [Google Scholar] [CrossRef]
- Shea, K.M. Antibiotic resistance: What is the impact of agricultural uses of antibiotics on children's health? Pediatrics 2003, 112, 253–258. [Google Scholar]
- Arikan, O.A.; Mulbry, W.; Rice, C. Management of antibiotic residues from agricultural sources: Use of composting to reduce chlortetracycline residues in beef manure from treated animals. J. Hazard. Mater. 2009, 164, 483–489. [Google Scholar] [CrossRef]
- National Research Council. The Use of Drugs in Food Animals: Benefits and Risks; National Academy Press: Washington, DC, USA, 1999; p. 25. [Google Scholar]
- Chen, Y.; Zhang, H.; Luo, Y.; Song, J. Occurrence and assessment of veterinary antibiotics in swine manures: A case study in east china. Chin. Sci. Bull. 2012, 57, 606–614. [Google Scholar] [CrossRef]
- Pan, X.; Qiang, Z.; Ben, W.; Chen, M. Residual veterinary antibiotics in swine manure from concentrated animal feeding operations in shandong province, china. Chemosphere 2011, 84, 695–700. [Google Scholar] [CrossRef]
- Motoyama, M.; Nakagawa, S.; Tanoue, R.; Sato, Y.; Nomiyama, K.; Shinohara, R. Residues of pharmaceutical products in recycled organic manure produced from sewage sludge and solid waste from livestock and relationship to their fermentation level. Chemosphere 2011, 84, 432–438. [Google Scholar] [CrossRef]
- Martínez-Carballo, E.; González-Barreiro, C.; Scharf, S.; Gans, O. Environmental monitoring study of selected veterinary antibiotics in animal manure and soils in austria. Environ. Pollut. 2007, 148, 570–579. [Google Scholar] [CrossRef]
- Aust, M.O.; Godlinski, F.; Travis, G.R.; Hao, X.; McAllister, T.A.; Leinweber, P.; Thiele-Bruhn, S. Distribution of sulfamethazine, chlortetracycline and tylosin in manure and soil of canadian feedlots after subtherapeutic use in cattle. Environ. Pollut. 2008, 156, 1243–1251. [Google Scholar] [CrossRef]
- Christian, T.; Schneider, R.J.; Färber, H.A.; Skutlarek, D.; Meyer, M.T.; Goldbach, H.E. Determination of antibiotic residues in manure, soil, and surface waters. Acta Hydrochim. Hydrobiol. 2003, 31, 36–44. [Google Scholar] [CrossRef]
- Karci, A.; Balcioǧlu, I.A. Investigation of the tetracycline, sulfonamide, and fluoroquinolone antimicrobial compounds in animal manure and agricultural soils in turkey. Sci. Total Environ. 2009, 407, 4652–4664. [Google Scholar] [CrossRef]
- Jacobsen, A.M.; Halling-Sørensen, B. Multi-component analysis of tetracyclines, sulfonamides and tylosin in swine manure by liquid chromatography-tandem mass spectrometry. Anal. Bioanal. Chem. 2006, 384, 1164–1174. [Google Scholar] [CrossRef]
- Bound, J.P.; Voulvoulis, N. Pharmaceuticals in the aquatic environment—A comparison of risk assessment strategies. Chemosphere 2004, 56, 1143–1155. [Google Scholar] [CrossRef]
- Kumar, K.; Gupta, S.C.; Chander, Y.; Singh, A.K. Antibiotic use in agriculture and its impact on the terrestrial environment. Adv. Agron. 2005, 87, 1–54. [Google Scholar] [CrossRef]
- Antibiotics in Manure and Soil—A Grave Threat to Human and Animal Health; Policy Paper 43; National Academy of Agriculture Science: New Delhi, India, 2010; p. 20.
- Mackie, R.I.; Koike, S.; Krapac, I.; Chee-Sanford, J.; Maxwell, S.; Aminov, R.I. Tetracycline residues and tetracycline resistance genes in groundwater impacted by swine production facilities. Anim. Biotech. 2006, 17, 157–176. [Google Scholar] [CrossRef]
- Boxall, A.B.; Blackwell, P.; Cavallo, R.; Kay, P.; Tolls, J. The sorption and transport of a sulphonamide antibiotic in soil systems. Toxicol. Lett. 2002, 131, 19–28. [Google Scholar] [CrossRef]
- Elmund, G.K.; Morrison, S.M.; Grant, D.W.; Nevins, M.P. Role of excreted chlortetracycline in modifying the decomposition process in feedlot waste. Bull. Environ. Contam. Toxicol. 1971, 6, 129–132. [Google Scholar] [CrossRef]
- Feinman, S.E.; Matheson, J.C. Draft Environmental Impact Statement Subtherapeutic Antibacterial Agents in Animal Feeds; FDA: Rockville, MD, USA, 1978. [Google Scholar]
- Montforts, M.H.M.M.; Kalf, D.F.; Van Vlaardingen, P.L.A.; Linders, J.B.H.J. The exposure assessment for veterinary medicinal products. Sci. Total Environ. 1999, 225, 119–133. [Google Scholar] [CrossRef]
- Kolz, A.C.; Moorman, T.B.; Ong, S.K.; Scoggin, K.D.; Douglass, E.A. Degradation and metabolite production of tylosin in anaerobic and aerobic swine-manure lagoons. Water Environ. Res. 2005, 77, 49–56. [Google Scholar] [CrossRef]
- Donoho, A.; Manthey, J.; Occolowitz, J.; Zornes, L. Metabolism of monensin in the steer and rat. J. Agr. Food Chem. 1978, 26, 1090–1095. [Google Scholar] [CrossRef]
- Cocito, C. Antibiotics of the virginiamycin family, inhibitors which contain synergistic components. Microbiol. Rev. 1979, 43, 145–198. [Google Scholar]
- Arikan, O.A. Degradation and metabolization of chlortetracycline during the anaerobic digestion of manure from medicated calves. J. Hazard. Mater. 2008, 158, 485–490. [Google Scholar] [CrossRef]
- Hu, X.G.; Luo, Y.; Zhou, Q.X.; Xu, L. Determination of thirteen antibiotics residues in manure by solid phase extraction and high performance liquid chromatography. Chin. J. Anal. Chem. 2008, 36, 1162–1166. [Google Scholar] [CrossRef]
- Xian, Q.; Hu, L.; Chen, H.; Chang, Z.; Zou, H. Removal of nutrients and veterinary antibiotics from swine wastewater by a constructed macrophyte floating bed system. J. Environ. Manag. 2010, 91, 2657–2661. [Google Scholar] [CrossRef]
- Pruden, A. Production and transport of antibiotics from cafos. In Hormones and Pharmaceuticals Generated by Concentrated Animal Feeding Operations; Springer-Verlag: New York, NY, USA, 2009; pp. 63–70. [Google Scholar]
- Thiele-Bruhn, S. Pharmaceutical antibiotic compounds in soils–A review. J. Plant Nutr. Soil Sci. 2003, 166, 145–167. [Google Scholar] [CrossRef]
- De Liguoro, M.; Cibin, V.; Capolongo, F.; Halling-Sørensen, B.; Montesissa, C. Use of oxytetracycline and tylosin in intensive calf farming: Evaluation of transfer to manure and soil. Chemosphere 2003, 52, 203–212. [Google Scholar] [CrossRef]
- Dolliver, H.A.S.; Gupta, S.C. Antibiotic losses from unprotected manure stockpiles. J. Environ. Qual. 2008, 37, 1238–1244. [Google Scholar] [CrossRef]
- Campagnolo, E.R.; Johnson, K.R.; Karpati, A.; Rubin, C.S.; Kolpin, D.W.; Meyer, M.T.; Esteban, J.E.; Currier, R.W.; Smith, K.; Thu, K.M.; et al. Antimicrobial residues in animal waste and water resources proximal to large-scale swine and poultry feeding operations. Sci. Total Environ. 2002, 299, 89–95. [Google Scholar] [CrossRef]
- Ince, B.; Coban, H.; Turker, G.; Ertekin, E.; Ince, O. Effect of oxytetracycline on biogas production and active microbial populations during batch anaerobic digestion of cow manure. Bioprocess Biosyst. Eng. 2013, 36, 541–546. [Google Scholar]
- Wu, C.; Spongberg, A.L.; Witter, J.D. Sorption and biodegradation of selected antibiotics in biosolids. J. Environ. Sci. Health A 2009, 44, 454–461. [Google Scholar]
- Davis, J.G.; Truman, C.C.; Kim, S.C.; Ascough, J.C., II; Carlson, K. Antibiotic transport via runoff and soil loss. J. Environ. Qual. 2006, 35, 2250–2260. [Google Scholar] [CrossRef]
- Ingerslev, F.; Halling-Sørensen, B. Biodegradability of metronidazole, olaquindox, and tylosin and formation of tylosin degradation products in aerobic soil-manure slurries. Ecotoxicol. Environ. Safety 2001, 48, 311–320. [Google Scholar] [CrossRef]
- Hu, X.; Zhou, Q.; Luo, Y. Occurrence and source analysis of typical veterinary antibiotics in manure, soil, vegetables and groundwater from organic vegetable bases, northern china. Environ. Pollut. 2010, 158, 2992–2998. [Google Scholar] [CrossRef]
- Chang, X.; Meyer, M.T.; Liu, X.; Zhao, Q.; Chen, H.; Chen, J.A.; Qiu, Z.; Yang, L.; Cao, J.; Shu, W. Determination of antibiotics in sewage from hospitals, nursery and slaughter house, wastewater treatment plant and source water in chongqing region of three gorge reservoir in china. Environ. Pollut. 2010, 158, 1444–1450. [Google Scholar] [CrossRef]
- Teeter, J.S.; Meyerhoff, R.D. Aerobic degradation of tylosin in cattle, chicken, and swine excreta. Environ. Res. 2003, 93, 45–51. [Google Scholar] [CrossRef]
- Winckler, C.; Grafe, A. Use of veterinary drugs in intensive animal production: Evidence for persistence of tetracycline in pig slurry. J. Soils Sediments 2001, 1, 66–70. [Google Scholar] [CrossRef]
- Lamshöft, M.; Sukul, P.; Zuhlke, S.; Spiteller, M. Behaviour of (14)c-sulfadiazine and (14)c-difloxacin during manure storage. Sci. Total Environ. 2010, 408, 1563–1568. [Google Scholar] [CrossRef]
- Ben, W.; Qiang, Z.; Adams, C.; Zhang, H.; Chen, L. Simultaneous determination of sulfonamides, tetracyclines and tiamulin in swine wastewater by solid-phase extraction and liquid chromatography-mass spectrometry. J. Chromatogr. A 2008, 1202, 173–180. [Google Scholar]
- Arikan, O.A.; Sikora, L.J.; Mulbry, W.; Khan, S.U.; Rice, C.; Foster, G.D. The fate and effect of oxytetracycline during the anaerobic digestion of manure from therapeutically treated calves. Process Biochem. 2006, 41, 1637–1643. [Google Scholar] [CrossRef]
- Wu, C.; Spongberg, A.L.; Witter, J.D. Determination of the persistence of pharmaceuticals in biosolids using liquid-chromatography tandem mass spectrometry. Chemosphere 2008, 73, 511–518. [Google Scholar] [CrossRef]
- Storteboom, H.N.; Kim, S.C.; Doesken, K.C.; Carlson, K.H.; Davis, J.G.; Pruden, A. Response of antibiotics and resistance genes to high-intensity and low-intensity manure management. J. Environ. Qual. 2007, 36, 1695–1703. [Google Scholar] [CrossRef]
- Arikan, O.A.; Sikora, L.J.; Mulbry, W.; Khan, S.U.; Foster, G.D. Composting rapidly reduces levels of extractable oxytetracycline in manure from therapeutically treated beef calves. Bioresour. Technol. 2007, 98, 169–176. [Google Scholar] [CrossRef]
- Schlusener, M.P.; Bester, K. Persistence of antibiotics such as macrolides, tiamulin and salinomycin in soil. Environ. Pollut. 2006, 143, 565–571. [Google Scholar] [CrossRef]
- Schlüsener, M.; von Arb, M.; Bester, K. Elimination of macrolides, tiamulin, and salinomycin during manure storage. Arch. Environ. Contam. Toxicol. 2006, 51, 21–28. [Google Scholar] [CrossRef]
- Ingerslev, F.; Halling-Sorensen, B. Biodegradability properties of sulfonamides in activated sludge. Environ. Toxicol. Chem. 2000, 19, 2467–2473. [Google Scholar] [CrossRef]
- Büyüksönmez, F.; Rynk, R.; Hess, T.F.; Bechinski, E. Occurrence, degradation and fate of pesticides during composting: Part II: Occurrence and fate of pesticides in compost and composting systems. Compost Sci. Utilization 2000, 8, 61–81. [Google Scholar] [CrossRef]
- 301D Closed Bottle Test. In Guidelines for Testing of Chemicals; Adopted by the Council on 17th July 1992; OECD: Paris, France, 1992.
- Al-Ahmad, A.; Daschner, F.D.; Kümmerer, K. Biodegradability of cefotiam, ciprofloxacin, meropenem, penicillin g, and sulfamethoxazole and inhibition of waste water bacteria. Arch. Environ. Contam. Toxicol. 1999, 37, 158–163. [Google Scholar] [CrossRef]
- Kümmerer, K.; Al-Ahmad, A.; Mersch-Sundermann, V. Biodegradability of some antibiotics, elimination of the genotoxicity and affection of wastewater bacteria in a simple test. Chemosphere 2000, 40, 701–710. [Google Scholar] [CrossRef]
- Wang, Q.-Q.; Bradford, S.A.; Zheng, W.; Yates, S.R. Sulfadimethoxine degradation kinetics in manure as affected by initial concentration, moisture, and temperature. J. Environ. Qual. 2006, 35, 2162–2169. [Google Scholar] [CrossRef]
- Shi, J.C.; Liao, X.D.; Wu, Y.B.; Liang, J.B. Effect of antibiotics on methane arising from anaerobic digestion of pig manure. Anim. Feed Sci. Technol. 2011, 166–167, 457–463. [Google Scholar] [CrossRef]
- Loke, M.-L.; Ingerslev, F.; Halling-Sørensen, B.; Tjørnelund, J. Stability of tylosin a in manure containing test systems determined by high performance liquid chromatography. Chemosphere 2000, 40, 759–765. [Google Scholar] [CrossRef]
- Varel, V.H.; Wells, J.E.; Shelver, W.L.; Rice, C.P.; Armstrong, D.L.; Parker, D.B. Effect of anaerobic digestion temperature on odour, coliforms and chlortetracycline in swine manure or monensin in cattle manure. J. Appl. Microbiol. 2012, 112, 705–715. [Google Scholar] [CrossRef]
- Turker, G.; Ince, O.; Ertekin, E.; Akyol, C.; Ince, B. Changes in performance and active microbial communities due to single and multiple effects of mixing and solid content in anaerobic digestion process of otc medicated cattle manure. Int. J. Renew. Energ. Res. 2013, 3, 144–148. [Google Scholar]
- Chelliapan, S.; Wilby, T.; Sallis, P.J. Performance of an up-flow anaerobic stage reactor (UASR) in the treatment of pharmaceutical wastewater containing macrolide antibiotics. Water Res. 2006, 40, 507–516. [Google Scholar] [CrossRef]
- Dolliver, H.; Gupta, S.; Noll, S. Antibiotic degradation during manure composting. J. Environ. Qual. 2008, 37, 1245–1253. [Google Scholar] [CrossRef]
- Hamscher, G.; Sczesny, S.; Hoper, H.; Nau, H. Determination of persistent tetracycline residues in soil fertilized with liquid manure by high-performance liquid chromatography with electrospray ionization tandem mass spectrometry. Anal. Chem. 2002, 74, 1509–1518. [Google Scholar] [CrossRef]
- Frankenberger, W.T.; Tabatabai, M.A. Transformations of amide nitrogen in soils1. Soil Sci. Soc. Am. J. 1982, 46, 280–284. [Google Scholar] [CrossRef]
- Gavalchin, J.; Katz, S.E. The persistence of fecal-borne antibiotics in soil. J. AOAC Int. 1994, 77, 481–485. [Google Scholar]
- Amin, M.M.; Zilles, J.L.; Greiner, J.; Charbonneau, S.; Raskin, L.; Morgenroth, E. Influence of the antibiotic erythromycin on anaerobic treatment of a pharmaceutical wastewater. Environ. Sci. Technol. 2006, 40, 3971–3977. [Google Scholar] [CrossRef]
- lvarez, J.A.; Otero, L.; Lema, J.M.; Omil, F. The effect and fate of antibiotics during the anaerobic digestion of pig manure. Bioresource Technol. 2010, 101, 8581–8586. [Google Scholar] [CrossRef]
- Loke, M.L.; Jespersen, S.; Vreeken, R.; Halling-Sørensen, B.; Tjørnelund, J. Determination of oxytetracycline and its degradation products by high-performance liquid chromatography-tandem mass spectrometry in manure-containing anaerobic test systems. J. Chromatogr. B 2003, 783, 11–23. [Google Scholar] [CrossRef]
- Søeborg, T.; Ingerslev, F.; Halling-Sørensen, B. Chemical stability of chlortetracycline and chlortetracycline degradation products and epimers in soil interstitial water. Chemosphere 2004, 57, 1515–1524. [Google Scholar] [CrossRef]
- Massé, D.I.; Lu, D.; Masse, L.; Droste, R.L. Effect of antibiotics on psychrophilic anaerobic digestion of swine manure slurry in sequencing batch reactors. Bioresource Technol. 2000, 75, 205–211. [Google Scholar] [CrossRef]
- Poels, J.; Van Assche, P.; Verstraete, W. Effects of disinfectants and antibiotics on the anaerobic digestion of piggery waste. Agr. Wastes 1984, 9, 239–247. [Google Scholar] [CrossRef]
- Rabølle, M.; Spliid, N.H. Sorption and mobility of metronidazole, olaquindox, oxytetracycline and tylosin in soil. Chemosphere 2000, 40, 715–722. [Google Scholar] [CrossRef]
- Chelliapan, S.; Wilby, T.; Sallis, P.J.; Yuzir, A. Tolerance of the antibiotic tylosin on treatment performance of an up-flow anaerobic stage reactor (UASR). Water Sci. Technol. 2011, 63, 1599–1606. [Google Scholar] [CrossRef]
- Drillia, P.; Dokianakis, S.N.; Fountoulakis, M.S.; Kornaros, M.; Stamatelatou, K.; Lyberatos, G. On the occasional biodegradation of pharmaceuticals in the activated sludge process: The example of the antibiotic sulfamethoxazole. J. Hazard. Mater. 2005, 122, 259–265. [Google Scholar] [CrossRef]
- Carballa, M.; Omil, F.; Ternes, T.; Lema, J.M. Fate of pharmaceutical and personal care products (ppcps) during anaerobic digestion of sewage sludge. Water Res. 2007, 41, 2139–2150. [Google Scholar] [CrossRef]
- Kim, K.R.; Owens, G.; Ok, Y.S.; Park, W.K.; Lee, D.B.; Kwon, S.I. Decline in extractable antibiotics in manure-based composts during composting. Waste Manag. 2012, 32, 110–116. [Google Scholar] [CrossRef]
- Fedler, C.B.; Day, D.L. Anaerobic Digestion of Swine Manure Containing an Antibiotic Inhibitor. Trans. ASAE 1985, 28, 523–530. [Google Scholar]
- Halling-Sørensen, B.; Sengeløv, G.; Tjørnelund, J. Toxicity of tetracyclines and tetracycline degradation products to environmentally relevant bacteria, including selected tetracycline-resistant bacteria. Arch. Environ. Contam. Toxicol. 2002, 42, 263–271. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Massé, D.I.; Saady, N.M.C.; Gilbert, Y. Potential of Biological Processes to Eliminate Antibiotics in Livestock Manure: An Overview. Animals 2014, 4, 146-163. https://doi.org/10.3390/ani4020146
Massé DI, Saady NMC, Gilbert Y. Potential of Biological Processes to Eliminate Antibiotics in Livestock Manure: An Overview. Animals. 2014; 4(2):146-163. https://doi.org/10.3390/ani4020146
Chicago/Turabian StyleMassé, Daniel I., Noori M. Cata Saady, and Yan Gilbert. 2014. "Potential of Biological Processes to Eliminate Antibiotics in Livestock Manure: An Overview" Animals 4, no. 2: 146-163. https://doi.org/10.3390/ani4020146