Quantitative Assessment of Major Biosecurity Challenges of Poultry Production in Central Ethiopia
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Study Design and Study Population
2.3. Quantification of Biosecurity Score
2.4. Data Collection and Analysis
3. Results and Discussion
3.1. Characteristics of the Study Participants and Poultry Farms
3.2. The Characteristics of the Studied Poultry Farms
3.3. Assessment of External Biosecurity Practices
3.3.1. Routines on Chicken Purchasing, Feeding and Watering Practices
3.3.2. Routines on Management of Wastes and Farm Entry Restriction Practices
3.3.3. Routines on Material Sharing, Farm Infrastructure and Biological Factors
3.3.4. Farms’ Relative Location
3.4. Internal Biosecurity Assessment Practices
3.4.1. Routines on Disease Management Practices
3.4.2. Routines on Cleaning and Disinfection Practices
3.5. Adoption Level of Biosecurity Components
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Milkias, M. Chicken meat production, consumption and constraints in Ethiopia. Food Sci. Qual. Manag. 2016, 54, 1–12. [Google Scholar]
- Ebsa, Y.A.; Harpal, S.; Negia, G.G. Challenges and chicken production status of poultry producers in Bishoftu, Ethiopia. Poult. Sci. 2019, 98, 5452–5455. [Google Scholar] [CrossRef]
- Belay, F.; Oljira, A. Socioeconomic importance and production characteristics of village poultry production in Ethiopia: A review. Niger. J. Anim. Sci. 2019, 21, 112–122. [Google Scholar]
- Tolasa, B. Current status of indigenous and highly productive chicken breeds in Ethiopia. Adv. Agric. 2021, 2021, 8848388. [Google Scholar] [CrossRef]
- The Federal Democratic Republic of Ethiopia, Central Statistical Agency, Agricultural Sample Survey. Volume II Report on Livestock and Livestock Characteristics (Private Peasant Holdings); Statistical Bulletin 589; Central Statistical Authority: Addis Ababa, Ethiopia, 2021.
- Habte, T.; Amare, A.; Bettridge, J.M.; Collins, M.; Christley, R.M.; Wigley, P. Guide to Chicken Health and Management in Ethiopia: For Farmers and Development Agents; ILRI Manual; ILRI: Nairobi, Kenya, 2017. [Google Scholar]
- Asfaw, Y.T.; Ameni, G.; Medhin, G.; Gumi, B.; Hagos, Y.; Wieland, B. Poultry disease occurrences and their impacts in Ethiopia. Trop. Anim. Health Prod. 2021, 53, 1–10. [Google Scholar] [CrossRef]
- Sarrazin, S.; Cay, A.B.; Laureyns, J.; Dewulf, J. A survey on biosecurity and management in selected Belgian cattle farms. Prev. Vet. Med. 2014, 117, 129–139. [Google Scholar] [CrossRef]
- Robertson, I.D. Disease control, prevention and on-farm biosecurity: The role of veterinary epidemiology. Engineering 2020, 6, 20–25. [Google Scholar] [CrossRef]
- Barcelo, J.; Marco, E. On Farm Biosecurity. In Proceedings of the 15th International Pig Veterinary Society Congress, Birmingham, UK, 5–9 July 1998. [Google Scholar]
- Carr, J.; Howells, M. Biosecurity on pig and poultry farms: Principles for the veterinary profession. Practice 2018, 40, 238–248. [Google Scholar] [CrossRef]
- FAO. Biosecurity Guide for Live Poultry Markets; FAO Animal Production and Health Guidelines No. 17; FAO: Rome, Italy, 2015. [Google Scholar]
- Dewulf, J.; Van Immerseel, F. (Eds.) Biosecurity in Animal Production and Veterinary Medicine: From Principles to Practice; CABI: Leuven, Belgium, 2018; pp. 133–154. [Google Scholar]
- Siekkinen, K.M.; Heikkilä, J.; Tammiranta, N.; Rosengren, H. Measuring the costs of biosecurity on poultry farms: A case study in broiler production in Finland. Acta Vet. Scand. 2012, 54, 12. [Google Scholar] [CrossRef] [PubMed]
- Gelaude, P.; Schlepers, M.; Verlinden, M.; Laanen, M.; Dewulf, J. Biocheck.UGent: A quantitative tool to measure biosecurity at broiler farms and the relationship with technical performances and antimicrobial use. Poult. Sci. 2014, 93, 2740–2751. [Google Scholar] [CrossRef]
- Dawit, A.; Tamirat, D.; Serge, F.S.N.; Devesh, R. Overview and Background Paper on Ethiopia’s Poultry Sector: Relevance for HPAI Research in Ethiopia; Africa/Indonesia Region Report; International Food Policy Research Institute (IFPRI): Washington, DC, USA, 2009; p. 47. [Google Scholar]
- Mtui, G. Biosafety systems in Eastern and Central Africa. Afr. J. Environ. Sci. Technol. 2012, 6, 80–93. [Google Scholar]
- Dessie, T.; Ogle, B. Village poultry production systems in the central highlands of Ethiopia. Trop. Anim. Health Prod. 2001, 33, 521–537. [Google Scholar] [CrossRef] [PubMed]
- Dana, N.; Van der Waaij, L.H.; Dessie, T.; van Arendonk, J.A. Production objectives and trait preferences of village poultry producers of Ethiopia: Implications for designing breeding schemes utilizing indigenous chicken genetic resources. Trop. Anim. Health Prod. 2010, 42, 1519–1529. [Google Scholar] [CrossRef] [PubMed]
- Asfaw, Y.; Ameni, G.; Medhin, G.; Alemayehu, G.; Wieland, B. Infectious and parasitic diseases of poultry in Ethiopia: A systematic review and meta-analysis. Poult. Sci. 2019, 98, 6452–6462. [Google Scholar] [CrossRef] [PubMed]
- FAO. Poultry Sector Ethiopia; FAO Animal Production and Health Livestock Country Reviews No. 11; FAO: Rome, Italy, 2019. [Google Scholar]
- Wondmeneh, E.; Alemayehu, A.; Bewket, S.; Tsigereda, F. Status of Commercial Poultry Production in Ethiopia; Poultry Working Group, Ministry of Livestock and Fisheries: Addis Ababa, Ethiopia, 2017. [Google Scholar]
- Van Limbergen, T.; Dewulf, J.; Klinkenberg, M.; Ducatelle, R.; Gelaude, P.; Méndez, J.; Maes, D. Scoring biosecurity in European conventional broiler production. Poult. Sci. 2018, 97, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Tanquilut, N.C.; Espaldon, M.V.O.; Eslava, D.F.; Ancog, R.C.; Medina, C.D.R.; Paraso, M.G.V.; Dewulf, J. Quantitative assessment of biosecurity in broiler farms using Biocheck.UGent in Central Luzon, Philippines. Poult. Sci. 2020, 99, 3047–3059. [Google Scholar] [CrossRef]
- Caekebeke, N.; Jonquiere, F.J.; Ringenier, M.; Tobias, T.J.; Postma, M.; Van den Hoogen, A.; Dewulf, J. Comparing farm biosecurity and antimicrobial use in high-antimicrobial-consuming broiler and pig farms in the Belgian–Dutch border region. Front. Vet. Sci. 2020, 7, 558455. [Google Scholar] [CrossRef]
- Tadelle, D.; Kijora, C.; Peters, K.J. Indigenous chicken ecotypes in Ethiopia: Growth and feed utilization potentials. Int. J. Poult. Sci. 2003, 2, 144–152. [Google Scholar]
- Moredaa, E.; Mesekel, K.G. Importance of Traditional, Small Scale and Commercial Poultry Production in Ethiopia: A Review. Br. J. Poult. Sci. 2016, 5, 01–08. [Google Scholar]
- Iyiola-Tunji, A.O.; Ojo, I.H.; Hiikyaa, A.N.; Adesina, M.A.; Iyiola-Tunji, M.O.; Ojo, O.A. Neighborhood Acceptability of Poultry Farms Located in Residential Areas in Nigerian Metropolis. Niger. J. Anim. Sci. 2013, 15, 206–215. [Google Scholar]
- Eze, C.O.; Chah, J.M.; Uddin, I.O.; Anugwa, I.J.; Igbokwe, E.M. Bio-security measures employed by poultry farmers in Enugu State Nigeria. J. Agric. Ext. 2017, 21, 89–104. [Google Scholar] [CrossRef][Green Version]
- Amass, S.F. Biosecurity: Stopping the bugs from getting in. Pig J. 2005, 55, 104. [Google Scholar]
- Ornelas-Eusebio, E.; García-Espinosa, G.; Laroucau, K.; Zanella, G. Characterization of commercial poultry farms in Mexico: Towards a better understanding of biosecurity practices and antibiotic usage patterns. PLoS ONE 2020, 15, e0242354. [Google Scholar] [CrossRef]
- Laanen, M.; Maes, D.; Hendriksen, C.; Gelaude, P.; De Vliegher, S.; Rosseel, Y.; Dewulf, J. Pig, cattle and poultry farmers with a known interest in research have comparable perspectives on disease prevention and on-farm biosecurity. Prev. Vet. Med. 2014, 115, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Eltholth, M.M.; Mohamed, R.A.; Elgohary, F.A.; Elfadl, E.A.A. Assessment of Biosecurity Practices in Broiler Chicken Farms in Gharbia Governorate, Egypt. Alexandria. J. Vet. Sci. 2016, 49, 68–77. [Google Scholar]
- Amass, S.F.; Baysinger, A. Disease surveillance in swine population. In Diseases of Swine; Blackwell Publisher: Hoboken, NJ, USA, 2006; pp. 1075–1098. [Google Scholar]
- Lister, S.A. Biosecurity in poultry management. In Poultry Diseases; WB Saunders: Philadelphia, PA, USA, 2008; pp. 48–65. [Google Scholar]
- Thomas, M.E.; Bouma, A.; Ekker, H.M.; Fonken, A.J.M.; Stegeman, J.A.; Nielen, M. Risk factors for the introduction of high pathogenicity Avian Influenza virus into poultry farms during the epidemic in the Netherlands in 2003. Prev. Vet. Med. 2005, 69, 1–11. [Google Scholar] [CrossRef]
- Vieira, A.R.; Hofacre, C.L.; Smith, J.A.; Cole, D. Human contacts and potential pathways of disease introduction on Georgia poultry farms. Avian Dis. 2009, 53, 55–62. [Google Scholar] [CrossRef]
- Dorea, F.C.; Berghaus, R.; Hofacre, C.; Cole, D.J. Survey of biosecurity protocols and practices adopted by growers on commercial poultry farms in Georgia—USA. Avian Dis. 2010, 54, 1007–1015. [Google Scholar] [CrossRef]
- Racicot, M.; Venne, D.; Durivage, A.; Vaillancourt, J.P. Evaluation of strategies to enhance biosecurity compliance on poultry farms in Québec: Effect of audits and cameras. Prev. Vet. Med. 2012, 103, 208–218. [Google Scholar] [CrossRef]
- CDC (US Center of Disease Control). Preliminary Incidence and Trends of Infections with Pathogens Transmitted Commonly Through Food—Foodborne Diseases Active Surveillance Network, 10 U.S. Sites, 2016–2019. MMWR. Morb. Mortal. Wkly Rep. 2020, 69, 509–514. [Google Scholar] [CrossRef]
- Abah, H.O.; Abdu, P.A.; Assam, A. Assessment of biosecurity measures against Newcastle disease in commercial poultry farms in Benue state, Nigeria. Sokoto J. Vet. Sci. 2017, 15, 32–37. [Google Scholar] [CrossRef]
- Liljebjelke, K.A.; Hofacre, C.L.; Liu, T.; White, D.G.; Ayers, S.; Young, S.; Maurer, J.J. Vertical and horizontal transmission of Salmonella within integrated broiler production system. Foodborne Pathog. Dis. 2005, 2, 90–102. [Google Scholar] [CrossRef] [PubMed]
- Garber, L.; Bjork, K.; Patyk, K.; Rawdon, T.; Antognoli, M.; Delgado, A.; McCluskey, B. Factors associated with highly pathogenic avian influenza H5N2 infection on table-egg layer farms in the Midwestern United States, 2015. Avian Dis. 2016, 60, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, J.L.; Elbers, A.R.W.; Beerens, N. Risk factors of primary introduction of highly pathogenic and low pathogenic avian influenza virus into European poultry holdings, considering at least material contaminated by wild birds and contact with wild birds. EFSA Support. Publ. 2017, 14, 1282E. [Google Scholar]
- Wiseman, A.; Berman, E.M.; Klement, E. Risk factors for Newcastle disease in broiler farms in Israel. Prev. Vet. Med. 2018, 149, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Sims, L.D. Risks Associated with Poultry Production Systems. In Proceedings of the International Conference: Poultry in the 21st Century, Bangkok, Thailand, 5–7 November 2008; pp. 1–24. [Google Scholar]
- Van Steenwinkel, S.; Ribbens, S.; Ducheyne, E.; Goossens, E.; Dewulf, J. Assessing biosecurity practices, movements and densities of poultry sites across Belgium, resulting in different farm risk-groups for infectious disease introduction and spread. Prev. Vet. Med. 2011, 98, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Correia-Gomes, C.; Henry, M.K.; Reeves, A.; Sparks, N. Management and biosecurity practices by small to medium egg producers in Scotland. Poult. Sci. 2021, 62, 499–508. [Google Scholar] [CrossRef]
- Aondo, E.O.; Jackson, N.O.; Joshua, O.; Onduso, R.; Simion, K.O. Poultry Farming and Disease Management Practices in Small- Scale Farmers in Kisii County, Kenya. Glob. J. Sci. Front. Res. D Agric. Vet. 2020, 20, 1–9. [Google Scholar]
- Morton, B. Vaccines and animal welfare. Rev. Sci. Tech. 2007, 26, 157–163. [Google Scholar] [CrossRef]
- Cserep, T. Vaccines and Vaccination. In Poultry Diseases, 6th ed.; Patisson, M., McMullin, P.F., Bradburry, J.M., Alexander, D.J., Eds.; Saunders Elsevier: Shanghai, China, 2008; p. 66. [Google Scholar]
- Gibbens, J.C.; Pascoe, S.J.S.; Evans, S.J.; Davies, R.H.; Sayers, A.R. A trial of biosecurity as a means to control Campylobacter infection of broiler chickens. Prev. Vet. Med. 2001, 48, 85–99. [Google Scholar] [CrossRef]
- Tilli, G.; Laconi, A.; Galuppo, F.; Mughini-Gras, L.; Piccirillo, A. Assessing Biosecurity Compliance in Poultry Farms: A Survey in a Densely Populated Poultry Area in North East Italy. Animals 2022, 12, 1409. [Google Scholar] [CrossRef]
- Butcher, G.D.; Miles, R.D. The Avian Immune System, UF/IFAS Extension; Institute of Food and Agricultural Sciences, University of Florida: Gainesville, FL, USA, 2012. [Google Scholar]
- Meroz, M.; Samberg, Y. Disinfecting poultry production premises. Rev. Sci. Tech. 1995, 14, 273–291. [Google Scholar] [CrossRef] [PubMed]
- Huneau-Salaün, A.; Michel, V.; Balaine, L.; Petetin, I.; Eono, F.; Ecobichon, F.; Bouquin, S.L. Evaluation of common cleaning and disinfection programmes in battery cage and on-floor layer houses in France. Br. Poult. Sci. 2010, 51, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Studer, E.; Lüthy, J.; Hübner, P. Study of the presence of Campylobacter jejuni and C. coli in sand samples from four Swiss chicken farms. Res. Microbiol. 1999, 150, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Skarp, C.P.A.; Hänninen, M.L.; Rautelin, H.I.K. Campylobacteriosis: The role of poultry meat. Clin. Microbiol. Infect. 2016, 22, 103–109. [Google Scholar] [CrossRef]
| External Biosecurity Components | Definition |
|---|---|
| Chicken Purchasing Practice (CPP) | Refers to type of chicken purchased, source of purchased chicken, delivery of chicken from same source, inspection routine while purchasing and separate delivery of purchased chicken |
| Feed and Water Management (FWM) | Indicates practices that assess the source of feed, sealed feed storage against water and vermin and source of drinking water |
| Waste and Manure Management (WMM) | Denotes waste and manure management practices comprising separated waste disposal area, way of handling wastes, use of gloves during waste handling, habit of handwashing after waste handling and destination of farm wastes |
| Farm Entry Restriction (FER) | Shows practices consist of obligation of visitors to register, presence of farm specific clothes, handwashing and disinfection during farm entry, employees working in different farms |
| Inter-farm Material Sharing (IMS) | Encompasses inter-farm material sharing practices and disinfect shared materials when receiving to use |
| Farm Infrastructure Status (FIS) | States farm infrastructure status mainly the material used to construct the wall of the chicken farm |
| Control of Biological Vector (CBV) | Include the situation of control of biological vector practices that encompasses access of birds to outside (open air), vegetation potentially harbors other animals, manifestation of vermin (e.g., rats, mice, etc.) and access of pet animals (cats and dogs) |
| Farm Relative Location (FRL) | Denotes the position of farm relative location in terms of farm relative location from main road, residence area close to farm location and the approximate distance from nearest poultry farm |
| Internal Biosecurity Components | Definition |
| Disease Management (DM) | Deals with disease management practices including the extent and degree of fixed vaccination program, monitory of health status, professional help for health status monitory, isolation of sick birds and removal of dead birds |
| Cleaning and Disinfection (CD) | Refers to cleaning and disinfection practices that include biosecurity practices pertaining to cleaning of poultry farm after each production cycle, presence of footbath facility, probability of accessing farm without footbath, frequency of changing footbath and cleaning and disinfection of farm materials. |
| Variables | Categories | Addis Ababa N = 56 | Bishoftu N = 70 | West of Shaggar N = 100 |
|---|---|---|---|---|
| Role of respondent | Farm owner/co-owner | 82.1% | 52.9% | 88% |
| Employee | 16.1% | 41.4% | 8% | |
| Owner’s relative | 1.8% | 5.7% | 4% | |
| Sex | Male | 60.7% | 62.9% | 58% |
| Female | 39.3% | 37.1% | 42% | |
| Age | 18–25 years | 1.8% | 20% | 15% |
| 26–35 years | 51.8% | 52.9% | 49% | |
| 36–45 years | 35.7% | 22.9% | 27% | |
| >45 years | 10.7% | 4.3% | 9% | |
| Educational Status | No formal education | 7.1% | 7.1% | 9% |
| Primary school | 26.8% | 35.7% | 28% | |
| Secondary school | 42.9% | 28.6% | 26% | |
| Diploma and above | 23.2% | 28.6% | 37% |
| Biosecurity Measures | Responses | Addis Ababa N = 56 | Bishoftu N = 70 | West of Shaggar N = 100 | Chi-square | p-Value |
|---|---|---|---|---|---|---|
| Purpose of chicken production | Egg | 85.7% | 64.3% | 64% | 40.2 | <0.001 |
| Meat | 14.3% | 30% | 8% | |||
| Dual | 0% | 5.7% | 28% | |||
| Keeping different types of breeds | Yes * | 14.3% | 2.9% | 7% | 5.9 | 0.052 |
| No | 85.7% | 97.1% | 93% | |||
| Age of chickens | <2 months | 14.3% | 30% | 14% | 17.1 | 0.009 |
| 2–6 months | 14.3% | 25.7% | 29% | |||
| 7–12 months | 42.8% | 37.1% | 35% | |||
| >12 months | 28.6% | 7.1% | 22% | |||
| Housing system | Deep litter system | 83.9% | 78.6% | 99% | 19.9 | 0.001 |
| Cage system | 12.5% | 18.6% | 1% | |||
| Both | 3.6% | 2.9% | 0% | |||
| Breeds of chickens | Bovans Brown | 85.7% | 47.1% | 57% | 56.7 | <0.001 |
| Sasso | 0% | 5.7% | 28% | |||
| Cobb 500 | 14.3% | 30% | 8% | |||
| Lohmann Brown | 0% | 17.1% | 7% | |||
| Size of farm | Small scale | 73.2% | 47.1% | 79% | 51.6 | <0.001 |
| Medium scale | 26.8% | 24.3% | 21% | |||
| Large scale | 0% | 28.6% | 0% | |||
| All-in/all-out practice | Yes * | 78.6% | 84.3% | 65% | 8.7 | 0.013 |
| No | 21.4% | 15.7% | 35% | |||
| Presence of pet animals | Yes | 100% | 41.4% | 97% | 99.1 | <0.001 |
| No | 0% | 58.6% | 3% |
| Biosecurity Measures | Responses | Addis Ababa N = 56 | Bishoftu N = 70 | West of Shaggar N = 100 | Chi-square | p-Value |
|---|---|---|---|---|---|---|
| Type of chicken purchased | Day-old chicken | 53.6 | 65.7% | 41% | 10.2 | 0.006 |
| Pullets | 46.4% | 34.3% | 59% | |||
| Source of purchased chicken | Well-known supplier | 78.6% | 85.7% | 82% | 8.6 | 0.196 |
| Local chicken supplier | 14.3% | 14.3% | 10% | |||
| Middleman | 7.1% | 0% | 8% | |||
| Delivery of chicken from same source | Yes, always the same source | 10.7% | 72.9% | 55% | 89.3 | <0.001 |
| No, sometimes vary | 46.4% | 25.7% | 43% | |||
| No, mostly vary | 42.9% | 1.4% | 2% | |||
| Batch mixing practices | Yes | 21.4 | 15.7% | 35% | 8.7 | 0.013 |
| No | 78.6% | 84.3% | 65% | |||
| Inspection routine while purchasing | Overall examination | 17.9% | 15.7% | 18% | 23.3 | <0.001 |
| Random size and/or weight | 12.5% | 34.3% | 7% | |||
| No known inspection | 69.6% | 50% | 75% | |||
| Separate delivery of purchased chicken | Yes * | 26.8% | 17.1% | 2% | 21.4 | <0.001 |
| No | 73.2% | 82.9% | 98% | |||
| Source of feed | Purchased from companies | 69.6% | 94.3% | 95% | 42.7 | <0.001 |
| In-house manual feed mix | 7.1% | 5.7% | 55% | |||
| Mixed | 23.2% | 0% | 0% | |||
| Sealed feed storage against water | Yes * | 71.4% | 98.6% | 77% | 19.2 | <0.001 |
| No | 28.6% | 1.4% | 23% | |||
| Sealed feed storage against vermin | Yes * | 8.9% | 82.9% | 58% | 69.8 | <0.001 |
| No | 91.1% | 17.1% | 42% | |||
| Source of drinking water | Tap water | 87.5% | 94.3% | 1% | 221.2 | <0.001 |
| Well water | 0% | 0% | 98% | |||
| Mixed | 7 (12.5%) | 5.7% | 1% |
| Biosecurity Measures | Responses | Addis Ababa N = 56 | Bishoftu N = 70 | West of Shaggar N = 100 | Chi-square | p-Value |
|---|---|---|---|---|---|---|
| Separated waste disposal area | Yes * | 17.9% | 22.9% | 74% | 64.7 | <0.001 |
| No | 82.1% | 77.1% | 26% | |||
| Handling waste | Composting in pit | 16.1% | 54.3% | 67% | 69.3 | <0.001 |
| Stored in sealed bag | 25% | 15.7% | 15% | |||
| Immediate removal | 17.9% | 24.3% | 16% | |||
| No recognized system | 41.1% | 5.7% | 2% | |||
| Use of gloves during waste handling | Always | 1.8% | 2.9% | 6% | 6.8 | 0.555 |
| Sometimes | 28.6% | 31.4% | 25% | |||
| Never | 69.6% | 65.7% | 69% | |||
| Washing after waste handling | Always | 48.2% | 80% | 70% | 17.2 | 0.002 |
| Sometimes | 50% | 18.6% | 26% | |||
| Never | 1.8% | 1.4% | 4% | |||
| Destination of farm waste | Disposed around farm | 28.6% | 62.9% | 12% | 155.8 | <0.001 |
| Sell for other uses | 5.4% | 25.7% | 51% | |||
| Taken by dirt collectors | 12.5% | 1.4% | 37% | |||
| Mixed | 53.6% | 10% | 0% | |||
| Visitors register | Yes * | 10.7% | 15.7% | 4% | 6.9 | 0.032 |
| No | 89.3% | 84.3% | 96% | |||
| Presence of farm specific clothes | Yes, always | 1.8% | 20% | 4% | 55 | <0.001 |
| Yes, sometimes | 33.9% | 41.4% | 7% | |||
| No | 64.3% | 38.6% | 89% | |||
| Handwashing and disinfection during farm entry | Always | 10.7% | 35.7% | 4% | 95.6 | <0.001 |
| Sometimes | 53.6% | 44.3% | 9% | |||
| Never | 35.7% | 20% | 87% | |||
| Employees working in different farms | Yes * | 26.8% | 5.7% | 54% | 44.9 | <0.001 |
| No | 73.2% | 94.3% | 46% |
| Biosecurity Measures | Responses | Addis Ababa N = 56 | Bishoftu N = 70 | West of Shaggar N = 100 | Chi-square | p-Value |
|---|---|---|---|---|---|---|
| Material being shared with other farms | Always | 6 (10.71%) | 0% | 1% | 96.6 | <0.001 |
| Sometimes | 19 (33.93%) | 97.1% | 93% | |||
| Never | 55.4% | 2.9% | 6% | |||
| Disinfect materials after receiving prior usage | Always | 16.1% | 28.6% | 4% | 68.1 | <0.001 |
| Sometimes | 46.4% | 44.3% | 11% | |||
| Never | 37.5% | 27.1% | 85% | |||
| Material of chicken farm wall made of | Brick wall | 42.9% | 40% | 6% | 57 | <0.001 |
| Wire mesh | 51.8% | 34.3% | 53% | |||
| Soil plastered wall | 3.6% | 10% | 31% | |||
| Bamboo and others | 1.79% | 15.7% | 10% | |||
| Access of chickens to the outside (open air) | Yes | 26.9% | 5.7% | 23% | 11.4 | 0.003 |
| No * | 73.2% | 94.3% | 77% | |||
| Access of wild birds to the farm | Yes | 60.7% | 64.3% | 18% | 45.6 | <0.001 |
| No | 39.3% | 35.7% | 82% | |||
| Vegetation potentially harbors other animals | Yes | 91.1% | 32.9% | 0% | 135.2 | <0.001 |
| No* | 9% | 67.1% | 100% | |||
| Manifestation of vermin (e.g., rats, mice, etc.) | Yes | 98.2% | 38.6% | 65% | 48.7 | <0.001 |
| No * | 1.8% | 61.4% | 35% | |||
| Access of pet animals (cats and dogs) | Yes | 26.8% | 42.9% | 4% | 37.8 | <0.001 |
| No * | 73.2% | 57.4 | 96% |
| Biosecurity Measures | Responses | Addis Ababa N = 56 | Bishoftu N = 50 | West of Shaggar N = 100 | Chi-square | p-Value |
|---|---|---|---|---|---|---|
| Farm relative location from main road | Main road within 100 m | 55.4% | 34.3% | 42% | 27.5 | <0.001 |
| Main road 100–200 m | 41.1% | 47.1% | 23% | |||
| Main road > 200 m | 3.6% | 18.6% | 35% | |||
| Residence area close to farm location | House within 100 m | 82.1% | 62.9% | 95% | 29.6 | <0.001 |
| House within 100 –200 m | 8.9% | 24.3% | 3% | |||
| House > 200 m | 8.9% | 12.9% | 2% | |||
| Approximate distance from nearest poultry farm | <500 m | 66.1% | 34.3% | 69% | 30.1 | <0.001 |
| Between 500 m and 1 km | 21.4% | 41.4% | 25% | |||
| >1 km | 12.5% | 24.3% | 6% |
| Biosecurity Measures | Responses | Addis Ababa N = 56 | Bishoftu N = 50 | West of Shaggar N = 100 | Chi-square | p-Value |
|---|---|---|---|---|---|---|
| Fixed vaccination program | Always followed | 71.4% | 95.7% | 53% | 36.4 | <0.001 |
| Sometimes followed | 28.6% | 4.3% | 47% | |||
| Monitory of health status | Every week or less | 71.4% | 12.9% | 2% | 135.5 | <0.001 |
| Every two weeks | 19.6% | 40% | 12% | |||
| More than two weeks | 8.9% | 47.1% | 86% | |||
| Professional help for health status monitory | Private animal health workers | 16.1% | 84% | 58% | 186.3 | <0.001 |
| Government animal health worker | 8.9% | 4.3% | 40% | |||
| By farm workers | 75% | 7.1% | 2% | |||
| Keeping different age groups | Yes | 78.6% | 10% | 8% | 6.5 | 0.038 |
| No | 21.4% | 90% | 92% | |||
| Removal of dead birds | Separated in isolation room | 8.9% | 27.1% | 59% | 42 | <0.001 |
| Separated at corner of room with apparently healthy flocks | 37.5% | 64.3% | 17% | |||
| No separation room | 53.6% | 27.1% | 59% | |||
| Isolation of sick birds | Immediately after observation | 5.4% | 67.1% | 98% | 81.1 | <0.001 |
| Can be kept up to 24 h | 28.6% | 32.9% | 2% | |||
| Can be kept for > 24 h | 5.4% | 0% | 0% |
| Biosecurity Measures | Responses | Addis Ababa N = 56 | Bishoftu N = 50 | West of Shaggar N = 100 | Chi-square | p-Value |
|---|---|---|---|---|---|---|
| Cleaning of poultry farm after each production cycle | Always | 91.1% | 77.1% | 56% | 71.3 | <0.001 |
| Sometimes | 8.9% | 22.7% | 44% | |||
| Presence of footbath facility | Yes * | 41.1% | 100% | 29% | 88.6 | <0.001 |
| No | 58.9% | 0% | 71% | |||
| Probability of accessing farm without footbath | Never | 41.1% | 81.4% | 29% | 91.8 | <0.001 |
| Sometimes | 0% | 15.7% | 0% | |||
| Frequently | 58.9% | 2.9% | 71% | |||
| Frequency of changing footbath | Everyday | 1.8% | 2.9% | 1% | 100.7 | <0.001 |
| Every two days | 12.5% | 55.7% | 5% | |||
| Every three or more days | 26.8% | 40% | 23% | |||
| Not applicable | 58.9% | 1.4% | 71% | |||
| Cleaning and disinfection of farm materials | Always | 91.1% | 77.1% | 56% | 23.3 | <0.001 |
| Sometimes | 8.9% | 22.9% | 44% |
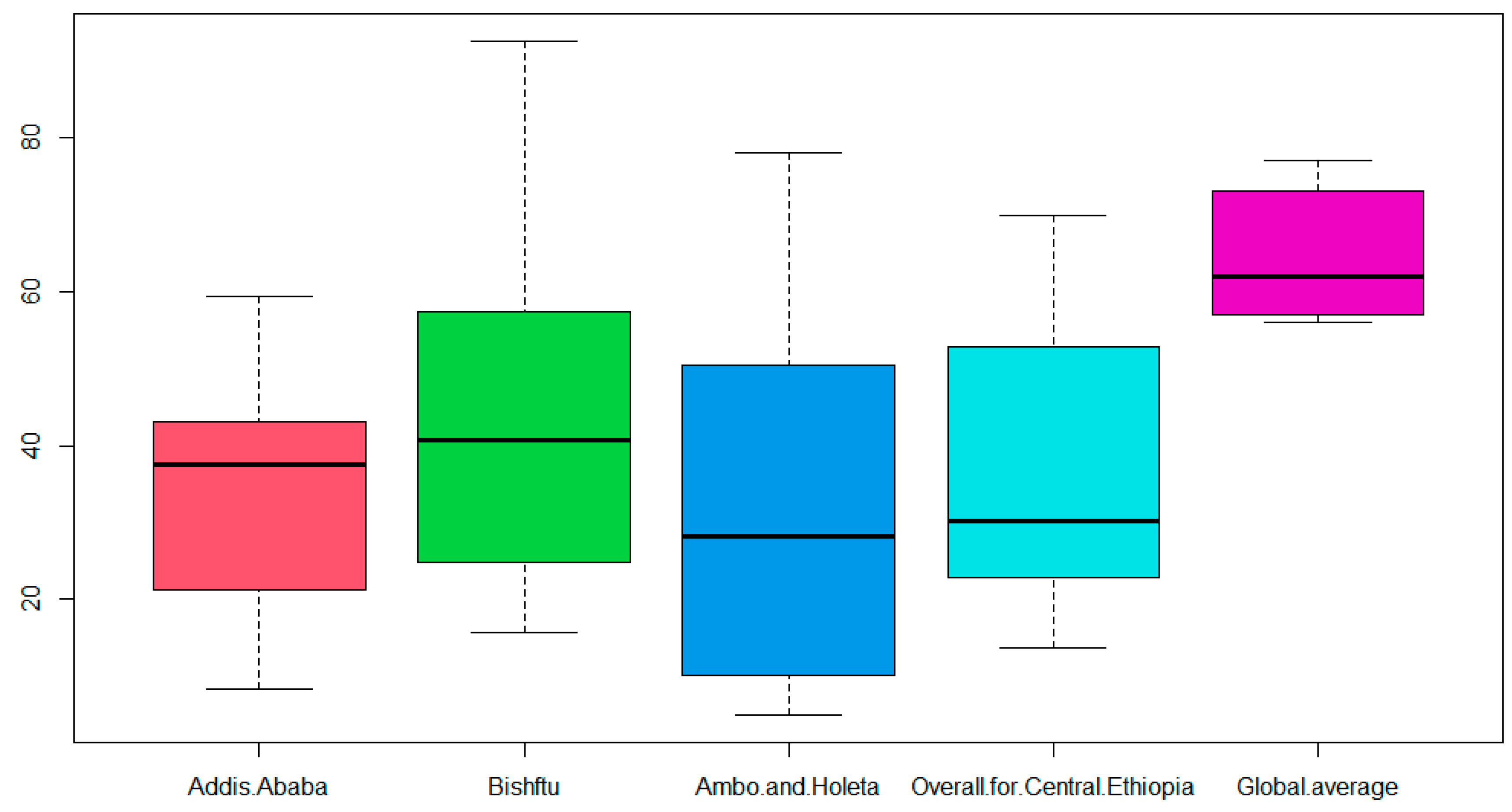
| External Biosecurity Components | Addis Ababa (%) | Bishoftu (%) | West of Shaggar (%) | Overall for Central Ethiopia (%) | Global Average (%) | t-Test, p Value |
|---|---|---|---|---|---|---|
| CPP | 43.2 | 51.4 | 41.8 | 45.5 | 56 | 0.1440 |
| FWM | 59.4 | 92.5 | 57.6 | 69.9 | 58 | 0.2347 |
| WMM | 18.2 | 31.1 | 43 | 30.8 | 59 | 0.0012 |
| FER | 24.1 | 41.4 | 14.5 | 26.7 | 69 | 0.0005 |
| IMS | 35.7 | 15.7 | 5 | 18.8 | 56 | 0.0066 |
| FIS | 42.9 | 40 | 6 | 29.6 | 77 | 0.0571 |
| CBV | 39.3 | 63.1 | 78 | 60.1 | 77 | 0.0494 |
| FRL | 8.3 | 18.6 | 14.3 | 13.8 | 65 | 0.0000 |
| Overall external biosecurity | 40.7 | 64.6 | 0.0000 | |||
| Overall external biosecurity computed out of 80% | 32.6 | |||||
| DM | 34.6 | 58.3 | 54 | 49 | 73 | 0.0163 |
| CD | 55.4 | 78.3 | 35 | 56.2 | 61 | 0.5423 |
| Overall internal biosecurity | 52.6 | 64 | 0.0578 | |||
| Overall internal biosecurity computed out of 20% | 10.5 | |||||
| Overall biosecurity practices * | 43.1 | |||||
| Overall biosecurity global average | 64.3 | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Waktole, H.; Muluneh, T.; Miressa, Y.; Ayane, S.; Berhane, G.; Kabeta, T.; Borena, B.M.; Amenu, K.; Ashenafi, H.; Antonissen, G. Quantitative Assessment of Major Biosecurity Challenges of Poultry Production in Central Ethiopia. Animals 2023, 13, 3719. https://doi.org/10.3390/ani13233719
Waktole H, Muluneh T, Miressa Y, Ayane S, Berhane G, Kabeta T, Borena BM, Amenu K, Ashenafi H, Antonissen G. Quantitative Assessment of Major Biosecurity Challenges of Poultry Production in Central Ethiopia. Animals. 2023; 13(23):3719. https://doi.org/10.3390/ani13233719
Chicago/Turabian StyleWaktole, Hika, Tsedal Muluneh, Yemsrach Miressa, Sisay Ayane, Gebreyohannes Berhane, Tadele Kabeta, Bizunesh Mideksa Borena, Kebede Amenu, Hagos Ashenafi, and Gunther Antonissen. 2023. "Quantitative Assessment of Major Biosecurity Challenges of Poultry Production in Central Ethiopia" Animals 13, no. 23: 3719. https://doi.org/10.3390/ani13233719
APA StyleWaktole, H., Muluneh, T., Miressa, Y., Ayane, S., Berhane, G., Kabeta, T., Borena, B. M., Amenu, K., Ashenafi, H., & Antonissen, G. (2023). Quantitative Assessment of Major Biosecurity Challenges of Poultry Production in Central Ethiopia. Animals, 13(23), 3719. https://doi.org/10.3390/ani13233719






