Efficiency of Tris-Based Extender Steridyl for Semen Cryopreservation in Stallions
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Animals
2.3. Chemicals and Extenders Preparation
2.4. Semen Collection and Preparation
2.5. Cryopreservation
2.6. Semen Evaluation after Thawing
2.7. Artificial Insemination (AI) and Pregnancy Control
2.8. Statistical Analysis
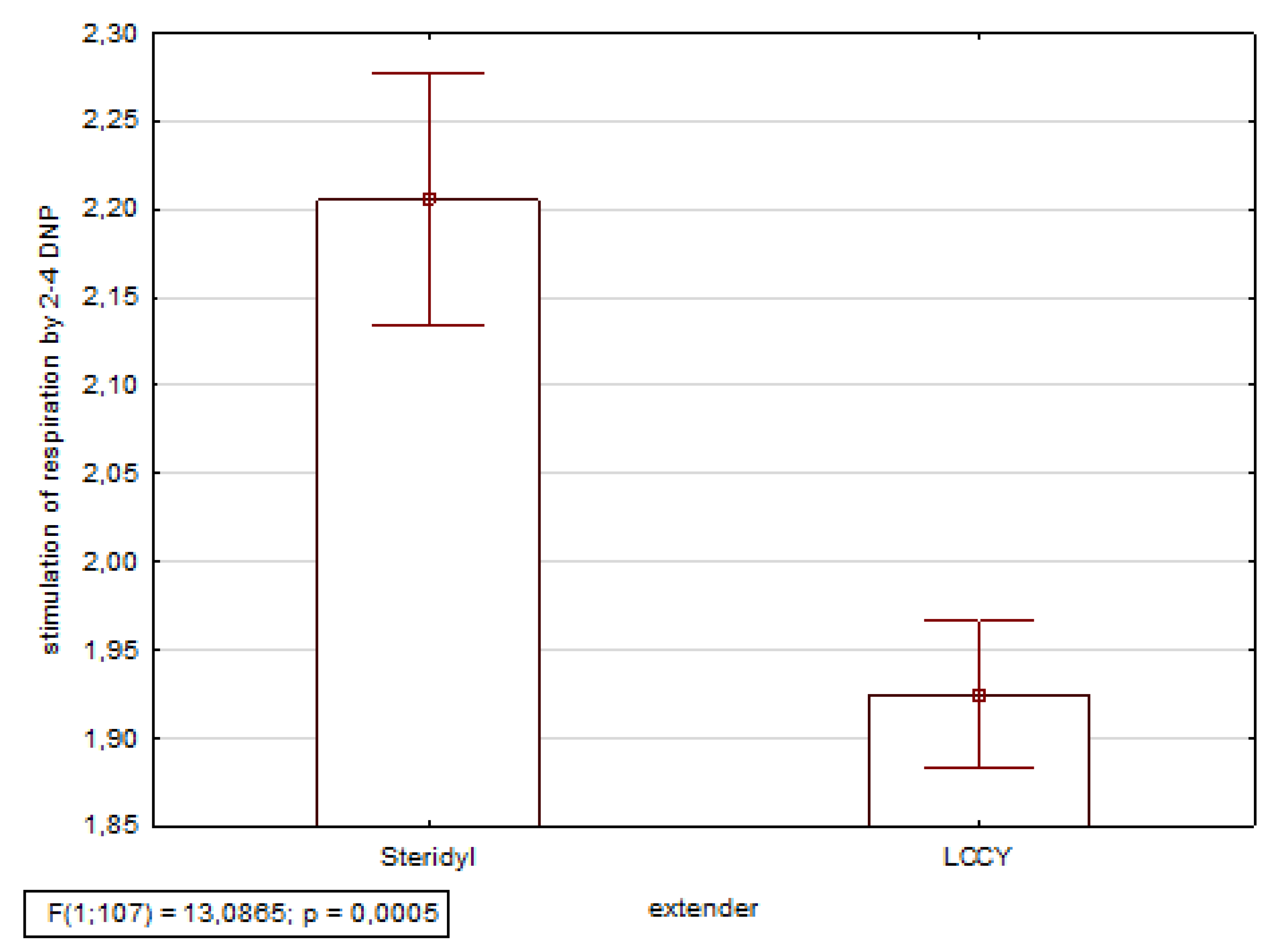
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aurich, J.E. Artificial Insemination in Horses-More than a Century of Practice and Research. J. Equine Vet. Sci. 2012, 32, 458–463. [Google Scholar] [CrossRef]
- Loomis, P.R.; Graham, J.K. Commercial semen freezing: Individual male variation in cryosurvival and the response of stallion sperm to customized freezing protocols. Anim. Reprod. Sci. 2008, 105, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Blottner, S.; Warnke, C.; Tuchscherer, A.; Heinen, V.; Torner, H. Morphological and functional changes of stallion spermatozoa after cryopreservation during breeding and non-breeding season. Anim. Reprod. Sci. 2001, 65, 75–88. [Google Scholar] [CrossRef]
- Contreras, M.J.; Treulen, F.; Arias, M.E.; Silva, M.; Fuentes, F.; Cabrera, P.; Felmer, R. Cryopreservation of stallion semen: Effect of adding antioxidants to the freezing medium on sperm physiology. Reprod. Domest. Anim. 2020, 55, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Clulow, J.R.; Maxwell, W.M.C.; Evans, G.; Morris, L.H.A. A comparison of duck and chicken egg yolk for cryopreservation of stallion sperm. Aust. Vet. J. 2007, 85, 232–235. [Google Scholar] [CrossRef]
- Scherzer, J.; Fayrer-Hosken, R.A.; Aceves, M.; Hurley, D.J.; Ray, L.E.; Jones, L.; Heusner, G.L. Freezing equine semen: The effect of combinations of semen extenders and glycerol on post-thaw motility. Aust. Vet. J. 2009, 87, 275–279. [Google Scholar] [CrossRef]
- Atroshchenko, M.M.; Bragina, E.E.; Zaitsev, A.M.; Kalashnikov, V.V.; Naumenkova, V.A.; Kudlaeva, A.M.; Nikitkina, E.V. Conservation of genetic resources in horse breeding and major structural damages of sperm during semen cryopreservation in stallions. Nat. Conserv. Res. 2019, 4, 78–82. [Google Scholar] [CrossRef]
- Atroshchenko, M.M.; Arkhangelskaya, E.; Isaev, D.A.; Stavitsky, S.B.; Zaitsev, A.M.; Kalaschnikov, V.V.; Leonov, S.; Osipov, A.N. Reproductive characteristics of thawed stallion sperm. Animals 2019, 9, 1099. [Google Scholar] [CrossRef] [Green Version]
- Alvarenga, M.A.; Papa, F.O.; Landim-Alvarenga, F.C.; Medeiros, A.S.L. Amides as cryoprotectants for freezing stallion semen: A review. Anim. Reprod. Sci. 2005, 89, 105–113. [Google Scholar] [CrossRef]
- Alghamdi, A.S.; Troedsson, M.H.T.; Xue, J.L.; Crabo, B.G. Effect of seminal plasma concentration and various extenders on postthaw motility and glass wool-Sephadex filtration of cryopreserved stallion semen. Am. J. Vet. Res. 2002, 63, 880–885. [Google Scholar] [CrossRef]
- Pace, M.M.; Graham, E.F. Components in egg yolk which protect bovine spermatozoa during freezing. J. Anim. Sci. 1974, 39, 1144–1149. [Google Scholar] [CrossRef] [PubMed]
- Batellier, F.; Vidament, M.; Fauquant, J.; Duchamp, G.; Arnaud, G.; Yvon, J.M.; Magistrini, M. Advances in cooled semen technology. Anim. Reprod. Sci. 2001, 68, 181–190. [Google Scholar] [CrossRef]
- Panahi, F.; Niasari-Naslaji, A.; Seyedasgari, F.; Ararooti, T.; Razavi, K.; Moosavi-Movaheddi, A.A. Supplementation of tris-based extender with plasma egg yolk of six avian species and camel skim milk for chilled preservation of dromedary camel semen. Anim. Reprod. Sci. 2017, 184, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Naz, S.; Umair, M.; Iqbal, S. Ostrich egg yolk improves post thaw quality and in vivo fertility of Nili Ravi buffalo (Bubalus bubalis) bull spermatozoa. Theriogenology 2019, 126, 140–144. [Google Scholar] [CrossRef] [PubMed]
- Swelum, A.A.A.; Saadeldin, I.M.; Ba-Awadh, H.; Al-Mutary, M.G.; Moumen, A.F.; Alowaimer, A.N.; Abdalla, H. Efficiency of commercial egg yolk-free and egg yolk-supplemented tris-based extenders for dromedary camel semen cryopreservation. Animals 2019, 9, 999. [Google Scholar] [CrossRef] [Green Version]
- Fernández, J.L.; Muriel, L.; Rivero, M.T.; Goyanes, V.; Vazquez, R.; Alvarez, J.G. The sperm chromatin dispersion test: A simple method for the determination of sperm DNA fragmentation. J. Androl. 2003, 24, 59–66. [Google Scholar] [CrossRef]
- Moroz, L.G.; Shapiev, I. Polarographic method for assessing energy metabolism in sperm by stimulating respiration with 2.4-dinitrophenol. Bull. State Sci. Inst. All-Russian Res. Inst. Farm Anim. Genet. Breed. 1978, 33, 28–30. [Google Scholar]
- Pojprasath, T.; Lohachit, C.; Techakumphu, M.; Stout, T.; Tharasanit, T. Improved cryopreservability of stallion sperm using a sorbitol-based freezing extender. Theriogenology 2011, 75, 1742–1749. [Google Scholar] [CrossRef]
- Vidament, M.; Dupere, A.M.; Julienne, P.; Evain, A.; Noue, P.; Palmer, E. Equine frozen semen: Freezability and fertility field results. Theriogenology 1997, 48, 907–917. [Google Scholar] [CrossRef]
- Vidament, M. French field results (1985–2005) on factors affecting fertility of frozen stallion semen. Anim. Reprod. Sci. 2005, 89, 115–136. [Google Scholar] [CrossRef]
- Šichtař, J.; Bubeníčková, F.; Sirohi, J.; Šimoník, O. How to increase post-thaw semen quality in poor freezing stallions: Preliminary results of the promising role of seminal plasma added after thawing. Animals 2019, 9, 414. [Google Scholar] [CrossRef] [Green Version]
- El-Shamaa, I.; El-Seify, E.S.; Hussein, A.; El-Sherbieny, M.; El-Sharawy, M. A comparison of duck and chicken egg yolk for cryopreservation of Egyptian buffalo bull spermatozoa Scientific Papers. Sci. Pap. Ser. D Anim. Sci. 2012, 55, 109–113. [Google Scholar]
- Minitube Insemination Results When Applying Triladyl® or Steridyl as Preservation Media for Bull Semen. Technical Report; Minitube: Tiefenbach, Germany, 2012; Volume 2. [Google Scholar]
- Barrier Battut, I.; Kempfer, A.; Becker, J.; Lebailly, L.; Camugli, S.; Chevrier, L. Development of a new fertility prediction model for stallion semen, including flow cytometry. Theriogenology 2016, 86, 1111–1131. [Google Scholar] [CrossRef]
- Prien, S. Cryoprotectants & Cryopreservation of Equine Semen: A Review of Industry Cryoprotectants and the Effects of Cryopreservation on Equine Semen Membranes. J. Dairy Vet. Anim. Res. 2016, 3. [Google Scholar] [CrossRef]
- Alamaary, M.S.; Haron, A.W.; Ali, M.; Hiew, M.W.H.; Adamu, L.; Peter, I.D. Effects of four extenders on the quality of frozen semen in Arabian stallions. Vet. World 2019, 12, 34–40. [Google Scholar] [CrossRef]
- Varner, D.D. Approaches to Breeding Soundness Examination and Interpretation of Results. J. Equine Vet. Sci. 2016, 43, S37–S44. [Google Scholar] [CrossRef]
- Teodora, V.; Groza, M.I. the Effect of Different Freezing Procedures on Sperm Head Morphometry in Stallions. Bull. Univ. Agric. Sci. Vet. Med. Cluj-Napoca Vet. Med. 2008, 65, 146–151. [Google Scholar] [CrossRef]
- Nikitkina, E.S.I. Assessment of the respiratory activity in equine sperm. Reproduction in Domestic Animals. Reprod. Domest. Anim. 2014, 49, 49–50. [Google Scholar]
- Moraes, C.R.; Meyers, S. The sperm mitochondrion: Organelle of many functions. Anim. Reprod. Sci. 2018, 194, 71–80. [Google Scholar] [CrossRef]
- Davila, M.P.; Muñoz, P.M.; Bolaños, J.M.G.; Stout, T.A.E.; Gadella, B.M.; Tapia, J.A.; Balao Da Silva, C.; Ortega Ferrusola, C.; Peña, F.J. Mitochondrial ATP is required for the maintenance of membrane integrity in stallion spermatozoa, whereas motility requires both glycolysis and oxidative phosphorylation. Reproduction 2016, 152, 683–694. [Google Scholar] [CrossRef] [Green Version]
- Gibb, Z.; Lambourne, S.R.; Aitken, R.J. The paradoxical relationship between stallion fertility and oxidative stress. Biol. Reprod. 2014, 91. [Google Scholar] [CrossRef]
- García, B.M.; Moran, A.M.; Fernández, L.G.; Ferrusola, C.O.; Rodriguez, A.M.; Bolaños, J.M.G.; Da Silva, C.M.B.; Martínez, H.R.; Tapia, J.A.; Peña, F.J. The mitochondria of stallion spermatozoa are more sensitive than the plasmalemma to osmotic-induced stress: Role of c-Jun N-terminal kinase (JNK) pathway. J. Androl. 2012, 33, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Nikitkina, E.; Shapiev, I.; Moroz, L.K.V.P. Correlation between the level of respiratory stimulation by 2,4-dnp and fertilizing ability of frozen sperm. Reprod. Domest. Anim. 2016, 51, 121–122. [Google Scholar]
- Evenson, D.; Jost, L. Sperm chromatin structure assay is useful for fertility assessment. Methods Cell Sci. 2000, 22, 169–189. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Avilés, C.; Zambrano-Varón, J.; Jiménez-Escobar, C. Current trends on stallion semen evaluation: What other methods can be used to improve our capacity for semen assessement? J. Vet. Androl. 2019, 4, 1–19. [Google Scholar]
- Benchaib, M.; Braun, V.; Lornage, J.; Hadj, S.; Salle, B.; Lejeune, H.; Guérin, J.F. Sperm DNA fragentation decreases the pregnancy rate in an assisted reproductive technique. Hum. Reprod. 2003, 18, 1023–1028. [Google Scholar] [CrossRef]
- Morrell, J.M.; Johannisson, A.; Dalin, A.M.; Hammar, L.; Sandebert, T.; Rodriguez-Martinez, H. Sperm morphology and chromatin integrity in Swedish warmblood stallions and their relationship to pregnancy rates. Acta Vet. Scand. 2008, 50. [Google Scholar] [CrossRef] [Green Version]
- Burruel, V.; Klooster, K.L.; Chitwood, J.; Ross, P.J.; Meyers, S.A. Oxidative damage to rhesus macaque spermatozoa results in mitotic arrest and transcript abundance changes in early embryos. Biol. Reprod. 2013, 89. [Google Scholar] [CrossRef]
| Steridyl | LCCY | |
|---|---|---|
| Total Motility, % | 83.2 ± 1.56 | 83.6 ± 1.97 |
| Progressive Motility, % | 75.3 ± 1.93 | 75.3 ± 1.38 |
| Spermatozoa of Normal Morphology, % | 75.8 ± 2.03 | 76.2 ± 1.82 |
| Steridyl | LCCY | |
|---|---|---|
| Total Motility, % | 43.1 ± 1.86 a | 39.6 ± 0.93 b |
| Progressive Motility, % | 36.3 ± 2.14 a | 31.7 ± 1.13 b |
| Spermatozoa of Normal Morphology, % | 72.4 ± 2.10 c | 60.4 ± 1.72 d |
| Spermatozoa with Damaged DNA, % | 27.2 ± 6.16 | 29.3 ± 4.32 |
| Pregnancy Rates (Pregnancy Confirmation) at Day 14, % | 75 ± 12.5 | 60 ± 15.5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nikitkina, E.; Musidray, A.; Krutikova, A.; Anipchenko, P.; Plemyashov, K.; Shiryaev, G. Efficiency of Tris-Based Extender Steridyl for Semen Cryopreservation in Stallions. Animals 2020, 10, 1801. https://doi.org/10.3390/ani10101801
Nikitkina E, Musidray A, Krutikova A, Anipchenko P, Plemyashov K, Shiryaev G. Efficiency of Tris-Based Extender Steridyl for Semen Cryopreservation in Stallions. Animals. 2020; 10(10):1801. https://doi.org/10.3390/ani10101801
Chicago/Turabian StyleNikitkina, Elena, Artem Musidray, Anna Krutikova, Polina Anipchenko, Kirill Plemyashov, and Gennadiy Shiryaev. 2020. "Efficiency of Tris-Based Extender Steridyl for Semen Cryopreservation in Stallions" Animals 10, no. 10: 1801. https://doi.org/10.3390/ani10101801







