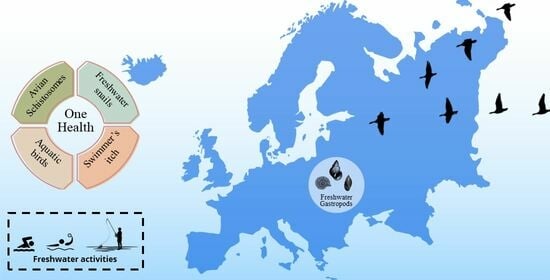
Zoonotic Threats: The (Re)emergence of Cercarial Dermatitis, Its Dynamics, and Impact in Europe
Abstract
:1. Introduction
2. Etiologic Agents
3. Intermediate and Definitive Hosts
4. Parasite Development, Biology, and Pathology
5. Cercarial Dermatitis in Europe: What Do We Know?
6. An Overview of CD Outside Europe
7. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Horák, P.; Mikeš, L.; Lichtenbergová, L.; Skála, V.; Soldánová, M.; Brant, S.V. Avian Schistosomes and Outbreaks of Cercarial Dermatitis. Clin. Microbiol. Rev. 2015, 28, 165–190. [Google Scholar] [CrossRef] [PubMed]
- Juhász, A.; Majoros, G.; Cech, G. Threat of cercarial dermatitis in Hungary: A first report of Trichobilharzia franki from the mallard (Anas platyrhynchos) and European ear snail (Radix auricularia) using molecular methods. Parasitol. Parasites Wildl. 2022, 18, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Ashrafi, K.; Nouroosta, A.; Sharifdini, M.; Mahmoudi, M.R.; Rahmati, B.; Brant, S.V. Genetic diversity of an avian nasal schistosome causing cercarial dermatitis in the Black Sea-Mediterranean migratory route. Parasitol. Res. 2018, 117, 3821–3833. [Google Scholar] [CrossRef] [PubMed]
- Cort, W.W. Studies on Schistosome Dermatitis. Am. J. Epidemiol. 1950, 52, 251–307. [Google Scholar] [CrossRef]
- Soldánová, M.; Selbach, C.; Kalbe, M.; Kostadinova, A.; Sures, B. Swimmer’s itch: Etiology, impact, and risk factors in Europe. Trends Parasitol. 2013, 29, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Caron, Y.; Cabaraux, A.; Marechal, F.; Losson, B. Swimmer’s Itch in Belgium: First Recorded Outbreaks, Molecular Identification of the Parasite Species and Intermediate Hosts. Vector-Borne Zoonotic Dis. 2017, 17, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Brant, S.V.; Loker, E.S. Discovery-based studies of schistosome diversity stimulate new hypotheses about parasite biology. Trends Parasitol. 2013, 29, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Horák, P.; Kolářová, L. Snails, waterfowl and cercarial dermatitis. Freshw. Biol. 2011, 56, 779–790. [Google Scholar] [CrossRef]
- Hoeffler, D.F. Cercarial Dermatitis. Arch. Environ. Health Int. J. 1974, 29, 225–229. [Google Scholar] [CrossRef]
- Brant, S.V.; Cohen, A.N.; James, D.; Hui, L.; Hom, A.; Loker, E.S. Cercarial Dermatitis Transmitted by Exotic Marine Snail. Emerg. Infect. Dis. 2010, 16, 1357–1365. [Google Scholar] [CrossRef]
- Rao, V.G.; Dash, A.P.; Agrawal, M.C.; Yadav, R.S.; Anvikar, A.R.; Vohra, S.; Bhondeley, M.K.; Ukey, M.J.; Das, S.K.; Minocha, R.K.; et al. Cercarial dermatitis in central India: An emerging health problem among tribal communities. Ann. Trop. Med. Parasitol. 2007, 101, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Ashrafi, K.; Sharifdini, M.; Darjani, A.; Brant, S.V. Migratory routes, domesticated birds and cercarial dermatitis: The distribution of Trichobilharzia franki in Northern Iran. Parasite 2021, 28, 4. [Google Scholar] [CrossRef] [PubMed]
- Kolářová, L.; Horák, P.; Skírnisson, K.; Marečková, H.; Doenhoff, M. Cercarial Dermatitis, a Neglected Allergic Disease. Clin. Rev. Allergy Immunol. 2013, 45, 63–74. [Google Scholar] [CrossRef] [PubMed]
- De Liberato, C.; Berrilli, F.; Bossù, T.; Magliano, A.; Montalbano Di Filippo, M.; Di Cave, D.; Sigismondi, M.; Cannavacciuolo, A.; Scaramozzino, P. Outbreak of swimmer’s itch in Central Italy: Description, causative agent and preventive measures. Zoonoses Public Health 2019, 66, 377–381. [Google Scholar] [CrossRef]
- Kolářová, L. Schistosomes causing cercarial dermatitis: A mini-review of current trends in systematics and of host specificity and pathogenicity. Folia Parasitol. 2007, 54, 81–87. [Google Scholar] [CrossRef]
- Lashaki, E.K.; Teshnizi, S.H.; Gholami, S.; Fakhar, M.; Brant, S.V.; Dodangeh, S. Global prevalence status of avian schistosomes: A systematic review with meta-analysis. Parasite Epidemiol. Control 2020, 9, e00142. [Google Scholar] [CrossRef] [PubMed]
- Macháček, T.; Turjanicová, L.; Bulantová, J.; Hrdý, J.; Horák, P.; Mikeš, L. Cercarial dermatitis: A systematic follow-up study of human cases with implications for diagnostics. Parasitol. Res. 2018, 117, 3881–3895. [Google Scholar] [CrossRef]
- Horák, P.; Kolářová, L.; Adema, C.M. Biology of the schistosome genus Trichobilharzia. Adv. Parasitol. 2002, 52, 155–233. [Google Scholar] [CrossRef] [PubMed]
- Neuhaus, W. Biologie und Entwicklung von Trichobilharzia szidati n. sp. (Trematoda, Schistosomatidae), einem Erreger von Dermatitis beim Menschen. Z. Parasitenkd. 1952, 15, 203–266. [Google Scholar] [CrossRef]
- Horák, P.; Dvořák, J.; Kolářová, L.; Trefil, L. Trichobilharzia regenti, a pathogen of the avian and mammalian central nervous systems. Parasitology 1999, 119, 577–581. [Google Scholar] [CrossRef]
- Müller, V.; Kimmig, P. Trichobilharzia franki n. sp.—The cause of swimmer’s dermatitis in southwest German dredged lakes. Appl. Parasitol. 1994, 35, 12–31. Available online: https://pubmed.ncbi.nlm.nih.gov/8173581/ (accessed on 14 December 2023). [PubMed]
- Helmer, N.; Blatterer, H.; Hörweg, C.; Reier, S.; Sattmann, H.; Schindelar, J.; Szucsich, N.U.; Haring, E. First Record of Trichobilharzia physellae (Talbot, 1936) in Europe, a Possible Causative Agent of Cercarial Dermatitis. Pathogens 2021, 10, 1473. [Google Scholar] [CrossRef] [PubMed]
- Jouet, D.; Kolářová, L.; Patrelle, C.; Ferté, H.; Skírnisson, K. Trichobilharzia anseri n. sp. (Schistosomatidae: Digenea), a new visceral species of avian schistosomes isolated from greylag goose (Anser anser L.) in Iceland and France. Infect. Genet. Evol. 2015, 34, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Kolářová, L.; Skírnisson, K.; Ferté, H.; Jouet, D. Trichobilharzia mergi sp. nov. (Trematoda: Digenea: Schistosomatidae), a visceral schistosome of Mergus serrator (L.) (Aves: Anatidae). Parasitol. Int. 2013, 62, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Simon Martin, F.; Simon Vicente, F. The life cycle of Trichobilharzia salmanticensis n. sp. (Digenea: Schistosomatidae), related to cases of human dermatitis. Res. Rev. Parasitol. 1999, 59, 13–18. [Google Scholar] [CrossRef]
- Rizevsky, S.V.; Cherviakovsky, E.M.; Kurchenko, V.P. Molecular taxonomic identification of Schistosomatidae from Naroch Lake and Polonevichi Lake in Belarus. Biochem. Syst. Ecol. 2011, 39, 14–21. [Google Scholar] [CrossRef]
- Reier, S.; Haring, E.; Billinger, F.; Blatterer, H.; Duda, M.; Gorofsky, C.; Grasser, H.P.; Heinisch, W.; Hörweg, C.; Kruckenhauser, L.; et al. First confirmed record of Trichobilharzia franki Müller & Kimmig, 1994, from Radix auricularia (Linnaeus, 1758) for Austria. Parasitol. Res. 2020, 119, 4135–4141. [Google Scholar] [CrossRef] [PubMed]
- Semyenova, S.K.; Chrisanfova, G.G.; Guliaev, A.S.; Yesakova, A.P.; Ryskov, A.P. Structural and Population Polymorphism of RT-Like Sequences in Avian Schistosomes Trichobilharzia szidati (Platyhelminthes: Digenea: Schistosomatidae). Biomed. Res. Int. 2015, 2015, 315312. [Google Scholar] [CrossRef] [PubMed]
- Chrisanfova, G.; Mozharovskaya, L.; Zhukova, T.; Nefedova, D.; Semyenova, S. Non-coding Regions of Mitochondrial DNA and the cox1 Gene Reveal Genetic Variability Among Local Belarusian Populations of the Causative Agent of Cercarial Dermatitis, Bird Schistosome Trichobilharzia szidati (Digenea: Schistosomatidae). Acta Parasitol. 2021, 66, 1193–1203. [Google Scholar] [CrossRef]
- Schols, R.; Smitz, N.; Vanderheyden, A.; Huyse, T. Expanding the swimmer’s itch pool of the Benelux: A first record of the neurotropic Trichobilharzia regenti and potential link to human infection. Parasites Vectors. 2024, 17, 1. [Google Scholar] [CrossRef]
- Dvořák, J.; Vaňáčová, Š.; Hampl, V.; Flegr, J.; Horák, P. Comparison of European Trichobilharzia species based on ITS1 and ITS2 sequences. Parasitology 2002, 124, 307–313. [Google Scholar] [CrossRef]
- Lockyer, A.E.; Olson, P.D.; Østergaard, P.; Rollinson, D.; Johnston, D.A.; Attwood, S.W.; Southgate, V.R.; Horak, P.; Snyder, S.D.; Le, T.H.; et al. The phylogeny of the Schistosomatidae based on three genes with emphasis on the interrelationships of Schistosoma Weinland, 1858. Parasitology 2003, 126, 203–224. [Google Scholar] [CrossRef]
- Rudolfová, J.; Hampl, V.; Bayssade-Dufour, C.; Lockyer, A.E.; Littlewood, D.T.J.; Horák, P. Validity reassessment of Trichobilharzia species using Lymnaea stagnalis as the intermediate host. Parasitol. Res. 2005, 95, 79–89. [Google Scholar] [CrossRef]
- Rudolfová, J.; Littlewood, D.T.; Sitko, J.; Horák, P. Bird schistosomes of wildfowl in the Czech Republic and Poland. Folia Parasitol. 2007, 54, 88–93. [Google Scholar] [CrossRef]
- Brant, S.V.; Loker, E.S. Molecular Systematics of the Avian Schistosome Genus Trichobilharzia (Trematoda: Schistosomatidae) in North America. J. Parasitol. 2009, 95, 941–963. [Google Scholar] [CrossRef]
- Aldhoun, J.A.; Podhorský, M.; Holická, M.; Horák, P. Bird schistosomes in planorbid snails in the Czech Republic. Parasitol. Int. 2012, 61, 250–259. [Google Scholar] [CrossRef]
- Christiansen, A.Ø.; Olsen, A.; Buchmann, K.; Kania, P.W.; Nejsum, P.; Vennervald, B.J. Molecular diversity of avian schistosomes in Danish freshwater snails. Parasitol. Res. 2016, 115, 1027–1037. [Google Scholar] [CrossRef]
- Al-Jubury, A.; Duan, Y.; Kania, P.; Tracz, E.; Bygum, A.; Jørgensen, L.; Horák, P.; Buchmann, K. Avian schistosome species in Danish freshwater lakes: Relation to biotic and abiotic factors. J. Helminthol. 2021, 95, e22. [Google Scholar] [CrossRef]
- Lawton, S.P.; Lim, R.M.; Dukes, J.P.; Cook, R.T.; Walker, A.J.; Kirk, R.S. Identification of a major causative agent of human cercarial dermatitis, Trichobilharzia franki (Müller and Kimmig 1994), in southern England and its evolutionary relationships with other European populations. Parasites Vectors 2014, 7, 277. [Google Scholar] [CrossRef]
- Juhász, A.; Barlow, S.E.J.; Williams, H.; Johnson, B.; Walsh, N.D.; Cunningham, L.C.; Jones, S.; LaCourse, E.J.; Stothard, J.R. A report of Bilharziella polonica cercariae in Knowsley Safari, Prescot, United Kingdom, with notes on other trematodes implicated in human cercarial dermatitis. J. Helminthol. 2022, 96, e79. [Google Scholar] [CrossRef]
- Aldhoun, J.A.; Faltýnková, A.; Karvonen, A.; Horák, P. Schistosomes in the North: A unique finding from a prosobranch snail using molecular tools. Parasitol. Int. 2009, 58, 314–317. [Google Scholar] [CrossRef]
- Ferté, H.; Depaquit, J.; Carré, S.; Villena, I.; Léger, N. Presence of Trichobilharzia szidati in Lymnaea stagnalis and T. franki in Radix auricularia in northeastern France: Molecular evidence. Parasitol. Res. 2005, 95, 150–154. [Google Scholar] [CrossRef]
- Bayssade-Dufour, C.; Jouet, D.; Rudolfova, J.; Horák, P.; Ferté, H. Seasonal morphological variations in bird schistosomes. Parasite 2006, 13, 205–214. [Google Scholar] [CrossRef]
- Jouet, D.; Ferté, H.; Depaquit, J.; Rudolfová, J.; Latour, P.; Zanella, D.; Kaltenbach, M.L.; Léger, N. Trichobilharzia spp. in natural conditions in Annecy Lake, France. Parasitol. Res. 2008, 103, 51–58. [Google Scholar] [CrossRef]
- Jouet, D.; Ferté, H.; Hologne, C.; Kaltenbach, M.L.; Depaquit, J. Avian schistosomes in French aquatic birds: A molecular approach. J. Helminthol. 2009, 83, 181–189. [Google Scholar] [CrossRef]
- Jouet, D.; Skírnisson, K.; Kolářová, L.; Ferté, H. Final hosts and variability of Trichobilharzia regenti under natural conditions. Parasitol. Res. 2010, 107, 923–930. [Google Scholar] [CrossRef]
- Jouet, D.; Skírnisson, K.; Kolářová, L.; Ferté, H. Molecular diversity of Trichobilharzia franki in two intermediate hosts (Radix auricularia and Radix peregra): A complex of species. Infect. Genet. Evol. 2010, 10, 1218–1227. [Google Scholar] [CrossRef]
- Hertel, J.; Hamburger, J.; Haberl, B.; Haas, W. Detection of bird schistosomes in lakes by PCR and filter-hybridization. Exp. Parasitol. 2002, 101, 57–63. [Google Scholar] [CrossRef]
- Majoros, G.; Dán, Á.; Erdélyi, K. A natural focus of the blood fluke Orientobilharzia turkestanica (Skrjabin, 1913) (Trematoda: Schistosomatidae) in red deer (Cervus elaphus) in Hungary. Vet. Parasitol. 2010, 170, 218–223. [Google Scholar] [CrossRef]
- Kolářová, L.; Rudolfová, J.; Hampl, V.; Skírnisson, K. Allobilharzia visceralis gen. nov., sp. nov. (Schistosomatidae-Trematoda) from Cygnus cygnus (L.) (Anatidae). Parasitol. Int. 2006, 55, 179–186. [Google Scholar] [CrossRef]
- Aldhoun, J.A.; Kolářová, L.; Horák, P.; Skírnisson, K. Bird schistosome diversity in Iceland: Molecular evidence. J. Helminthol. 2009, 83, 173–180. [Google Scholar] [CrossRef]
- Cipriani, P.; Mattiucci, S.; Paoletti, M.; Scialanca, F.; Nascetti, G. Molecular evidence of Trichobilharzia franki Müller and Kimmig, 1994 (Digenea: Schistosomatidae) in Radix auricularia from Central Italy. Parasitol. Res. 2011, 109, 935–940. [Google Scholar] [CrossRef]
- Soldánová, M.; Georgieva, S.; Roháčová, J.; Knudsen, R.; Kuhn, J.A.; Henriksen, E.H.; Siwertsson, A.; Shaw, J.C.; Kuris, A.M.; Amundsen, P.A.; et al. Molecular analyses reveal high species diversity of trematodes in a sub-Arctic lake. Int. J. Parasitol. 2017, 47, 327–345. [Google Scholar] [CrossRef]
- Marszewska, A.; Strzała, T.; Cichy, A.; Dąbrowska, G.B.; Żbikowska, E. Agents of swimmer’s itch—Dangerous minority in the Digenea invasion of Lymnaeidae in water bodies and the first report of Trichobilharzia regenti in Poland. Parasitol. Res. 2018, 117, 3695–3704. [Google Scholar] [CrossRef]
- Stanicka, A.; Migdalski, Ł.; Zając, K.S.; Cichy, A.; Lachowska-Cierlik, D.; Żbikowska, E. The genus Bilharziella vs. other bird schistosomes in snail hosts from one of the major recreational lakes in Poland. Knowl. Manag. Aquat. Ecosyst. 2021, 2020, 12. [Google Scholar] [CrossRef]
- Korsunenko, A.V.; Chrisanfova, G.G.; Ryskov, A.P.; Movsessian, S.O.; Vasilyev, V.A.; Semyenova, S.K. Detection of European Trichobilharzia Schistosomes (T. franki, T. szidati, and T. regenti) Based on Novel Genome Sequences. J. Parasitol. 2010, 96, 802–806. [Google Scholar] [CrossRef]
- Korsunenko, A.; Chrisanfova, G.; Lopatkin, A.; Beer, S.A.; Voronin, M.; Ryskov, A.P.; Semyenova, S.K. Genetic differentiation of cercariae infrapopulations of the avian schistosome Trichobilharzia szidati based on RAPD markers and mitochondrial cox1 gene. Parasitol. Res. 2012, 110, 833–841. [Google Scholar] [CrossRef]
- Picard, D.; Jousson, O. Genetic variability among cecariae of the shistomatidae (Trematoda: Digenea) causing swimmer’s itch in Europe. Parasite 2001, 8, 237–242. [Google Scholar] [CrossRef]
- Webster, B.L.; Rudolfová, J.; Horák, P.; Littlewood, D.T.J. The complete mitochondrial genome of the bird Schistosome Trichobilharzia regenti (PLATYHELMINTHES: DIGENEA), causative agent of cercarial dermatitis. J. Parasitol. 2007, 93, 553–561. [Google Scholar] [CrossRef]
- Soldánová, M.; Selbach, C.; Sures, B. The Early Worm Catches the Bird? Productivity and Patterns of Trichobilharzia szidati Cercarial Emission from Lymnaea stagnalis. Knight M, editor. PLoS ONE 2016, 11, e0149678. [Google Scholar] [CrossRef]
- Skála, V.; Walker, A.J.; Horák, P. Snail defence responses to parasite infection: The Lymnaea stagnalis-Trichobilharzia szidati model. Dev. Comp. Immunol. 2020, 102, 103464. [Google Scholar] [CrossRef]
- Haas, W. Parasitic worms: Strategies of host finding, recognition and invasion. Zoology 2003, 106, 349–364. [Google Scholar] [CrossRef]
- Horák, P.; Kolářová, L. Molluscan and vertebrate immune responses to bird schistosomes. Parasite Immunol. 2005, 27, 247–255. [Google Scholar] [CrossRef]
- Horák, P.; Knaap, W.P.W. Lectins in snail-trematode immune interactions: A review. Folia Parasitol. 1997, 44, 161–172. Available online: https://folia.paru.cas.cz/ (accessed on 11 March 2024).
- Skírnisson, K.; Kolářová, L. Diversity of bird schistosomes in anseriform birds in Iceland based on egg measurements and egg morphology. Parasitol. Res. 2008, 103, 43–50. [Google Scholar] [CrossRef]
- Hrádková, K.; Horák, P. Neurotropic behaviour of Trichobilharzia regenti in ducks and mice. J. Helminthol. 2002, 76, 137–141. [Google Scholar] [CrossRef]
- Gyimesi, A.; Lensink, R. Egyptian Goose Alopochen aegyptiaca: An introduced species spreading in and from the Netherlands. Wildfowl 2012, 62, 128–145. Available online: https://wildfowl.wwt.org.uk/index.php/wildfowl/article/view/1331 (accessed on 16 January 2024).
- Fischer, E.F.; Recht, S.; Vélez, J.; Rogge, L.; Taubert, A.; Hermosilla, C.R. Occurrence of Gastrointestinal Parasites in Synanthropic Neozoan Egyptian Geese (Alopochen aegyptiaca, Linnaeus 1766) in Germany. Diversity 2023, 15, 388. [Google Scholar] [CrossRef]
- Fain, A. Nasal Trichobilharziasis: A New Avian Schistosomiasis. Nature 1956, 177, 389. [Google Scholar] [CrossRef]
- Appleton, C.C. The eggs of some blood-flukes (Trematoda: Schistosomatidae) from South African birds. S. Afr. J. Zool. 1982, 17, 147–150. [Google Scholar] [CrossRef]
- Muñoz-Fuentes, V.; Green, A.J.; Sorenson, M.D.; Negro, J.J.; Vilà, C. The ruddy duck Oxyura jamaicensis in Europe: Natural colonization or human introduction? Mol. Ecol. 2006, 15, 1441–1453. [Google Scholar] [CrossRef]
- Anastácio, P.M.; Ribeiro, F.; Capinha, C.; Banha, F.; Gama, M.; Filipe, A.F.; Rebelo, R.; Sousa, R. Non-native freshwater fauna in Portugal: A review. Sci. Total Environ. 2019, 650, 1923–1934. [Google Scholar] [CrossRef]
- Padilla-Aguilar, P.; Romero-Callejas, E.; Osorio-Sarabia, D.; Pérez–Ponce de León, G.; Alcalá-Canto, Y. New records of helminth parasites of nine species of waterfowl in Mexico, and a checklist of the helminth fauna of Anatidae occurring in Mexican wetlands. J. Helminthol. 2020, 94, e176. [Google Scholar] [CrossRef]
- Jamieson, B.; Haas, W. Miracidium of Schistosoma. In Schistosoma: Biology, Pathology and Control, 1st ed.; CRC Press: Boca Raton, FL, USA, 2017; pp. 77–117. [Google Scholar] [CrossRef]
- Kalbe, M.; Haberl, B.; Haas, W. Snail Host Finding by Fasciola hepatica and Trichobilharzia ocellata: Compound Analysis of “Miracidia-Attracting Glycoproteins. Exp. Parasitol. 2000, 96, 231–242. [Google Scholar] [CrossRef]
- Nelwan, M.L. Schistosomiasis: Life Cycle, Diagnosis, and Control. Curr. Ther. Res. 2019, 91, 5–9. [Google Scholar] [CrossRef]
- Al-Jubury, A.; Kania, P.; Bygum, A.; Buchmann, K. Temperature and light effects on Trichobilharzia szidati cercariae with implications for a risk analysis. Acta Vet. Scand. 2020, 62, 54. [Google Scholar] [CrossRef]
- Auer, H.; Aspöck, H. Helminths and helminthoses in Central Europe: General overview and diseases caused by trematodes (flukes). Wien. Med. Wochenschr. 2014, 164, 405–413. [Google Scholar] [CrossRef]
- Selbach, C.; Soldánová, M.; Sures, B. Estimating the risk of swimmer’s itch in surface waters—A case study from Lake Baldeney, River Ruhr. Int. J. Hyg. Environ. Health 2016, 219, 693–699. [Google Scholar] [CrossRef]
- Kourilová, P.; Hogg, K.G.; Kolárová, L.; Mountford, A.P. Cercarial Dermatitis Caused by Bird Schistosomes Comprises Both Immediate and Late Phase Cutaneous Hypersensitivity Reactions. J. Immunol. 2004, 172, 3766–3774. [Google Scholar] [CrossRef]
- Horák, P.; Kolářová, L. Bird schistosomes: Do they die in mammalian skin? Trends Parasitol. 2001, 17, 66–69. [Google Scholar] [CrossRef]
- Semenza, J.C.; Paz, S. Climate change and infectious disease in Europe: Impact, projection and adaptation. Lancet Reg. Health—Eur. 2021, 9, 100230. [Google Scholar] [CrossRef]
- Jones, K.E.; Patel, N.G.; Levy, M.A.; Storeygard, A.; Balk, D.; Gittleman, J.L.; Daszak, P. Global trends in emerging infectious diseases. Nature 2008, 451, 990–993. [Google Scholar] [CrossRef]
- Tracz, E.; Al-Jubury, A.; Buchmann, K.; Bygum, A. Outbreak of Swimmer’s Itch in Denmark. Acta Derm. Venereol. 2019, 99, 1116–1120. [Google Scholar] [CrossRef]
- Fernandes, G.; de Castro, E.; Gomes, H. Water Resources and Tourism Development in Estrela Geopark Territory: Meaning and Contributions of Fluvial Beaches to Valorise the Destination. Eur. Countrys. 2020, 12, 551–567. [Google Scholar] [CrossRef]
- Stensgaard, A.S.; Vounatsou, P.; Sengupta, M.E.; Utzinger, J. Schistosomes, snails and climate change: Current trends and future expectations. Acta Trop. 2019, 190, 257–268. [Google Scholar] [CrossRef]
- Kolářová, L.; Horák, P.; Skírnisson, K. Methodical approaches in the identification of areas with a potential risk of infection by bird schistosomes causing cercarial dermatitis. J. Helminthol. 2010, 84, 327–335. [Google Scholar] [CrossRef]
- Ebbs, E.T.; Loker, E.S.; Davis, N.E.; Flores, V.; Veleizan, A.; Brant, S.V. Schistosomes with wings: How host phylogeny and ecology shape the global distribution of Trichobilharzia querquedulae (Schistosomatidae). Int. J. Parasitol. 2016, 46, 669–677. [Google Scholar] [CrossRef]
- Marszewska, A.; Cichy, A.; Heese, T.; Żbikowska, E. The real threat of swimmers’ itch in anthropogenic recreational water body of the Polish Lowland. Parasitol. Res. 2016, 115, 3049–3056. [Google Scholar] [CrossRef]
- Malone, J.B.; Nieto, P.; Tadesse, A. Biology-based mapping of vector-borne parasites by geographic information systems and remote sensing. Parassitologia 2006, 48, 77–79. Available online: https://pubmed.ncbi.nlm.nih.gov/16881402/ (accessed on 19 January 2024). [PubMed]
- Cotton, P.A. Avian migration phenology and global climate change. Proc. Natl. Acad. Sci. USA 2003, 100, 12219–12222. [Google Scholar] [CrossRef]
- Mas-Coma, S.; Valero, M.A.; Bargues, M.D. Efectos del cambio climático en las helmintiasis animales y zoonóticas. Rev. Sci. Tech. L’oie 2008, 27, 443–457. [Google Scholar] [CrossRef]
- Żbikowska, E. Is there a potential danger of “swimmer’s itch in Poland? Parasitol. Res. 2002, 89, 59–62. [Google Scholar] [CrossRef]
- Korycińska, J.; Rybak-d’Obyrn, J.; Kubiak, D.; Kubiak, K.; Dzika, E. Dermatological and Molecular Evidence of Human Cercarial Dermatitis in North-Eastern Poland. Vector-Borne Zoonotic Dis. 2021, 21, 269–274. [Google Scholar] [CrossRef]
- Soldánová, M.; Selbach, C.; Sures, B.; Kostadinova, A.; Pérez-del-Olmo, A. Larval trematode communities in Radix auricularia and Lymnaea stagnalis in a reservoir system of the Ruhr River. Parasites Vectors 2010, 3, 56. [Google Scholar] [CrossRef]
- Strathmann, M.; Horstkott, M.; Koch, C.; Gayer, U.; Wingender, J. The River Ruhr—An urban river under particular interest for recreational use and as a raw water source for drinking water: The collaborative research project “Safe Ruhr”—Microbiological aspects. Int. J. Hyg. Environ. Health 2016, 219, 643–661. [Google Scholar] [CrossRef]
- Schets, F.M.; Lodder, W.J.; van Duynhoven, Y.T.H.P.; de Roda Husman, A.M. Cercarial dermatitis in the Netherlands caused by Trichobilharzia spp. J. Water Health 2008, 6, 187–195. [Google Scholar] [CrossRef]
- Schets, F.M.; Lodder, W.J.; De Roda Husman, A.M. Confirmation of the presence of Trichobilharzia by examination of water samples and snails following reports of cases of cercarial dermatitis. Parasitology 2010, 137, 77–83. [Google Scholar] [CrossRef]
- De Gentile, L.; Picot, H.; Bourdeau, P.; Bardet, R.; Kerjan, A.; Piriou, M.; Le Guennic, A.; Bayssade-Dufour, C.; Chabasse, D.; Mott, K. Cercarial dermatitis in Europe: A new public health problem? Bull. World Health Organ. 1996, 74, 159–163. Available online: http://www.ncbi.nlm.nih.gov/pubmed/8706231 (accessed on 25 January 2024). [PubMed]
- Caumes, E.; Felder-Moinet, S.; Couzigou, C.; Darras-Joly, C.; Latour, P.; Léger, N. Failure of an ointment based on IR3535 (ethyl butylacetylaminopropionate) to prevent an outbreak of cercarial dermatitis during swimming races across Lake Annecy, France. Ann. Trop. Med. Parasitol. 2003, 97, 157–163. [Google Scholar] [CrossRef]
- Eklu-Natey, D.T.; Al-Khudri, M.; Gauthey, D.; Dubois, J.P.; Wüest, J.; Vaucher, C. Epidémiologie de la dermatite des baigneurs et morphologie de Trichobilharzia cf. ocellata dans le lac Léman. Rev. Suisse Zool. 1985, 92, 939–953. [Google Scholar] [CrossRef]
- Chamot, E.; Toscani, L.; Rougemont, A. Public health importance and risk factors for cercarial dermatitis associated with swimming in Lake Leman at Geneva, Switzerland. Epidemiol. Infect. 1998, 120, 305–314. [Google Scholar] [CrossRef]
- Gulyás, K.; Soldánová, M.; Orosová, M.; Oros, M. Confirmation of the presence of zoonotic Trichobilharzia franki following a human cercarial dermatitis outbreak in recreational water in Slovakia. Parasitol. Res. 2020, 119, 2531–2537. [Google Scholar] [CrossRef]
- Faltýnková, A.; Našincová, V.; Kablásková, L. Larval trematodes (Digenea) of planorbid snails (Gastropoda: Pulmonata) in Central Europe: A survey of species and key to their identification. Syst. Parasitol. 2008, 69, 155–178. [Google Scholar] [CrossRef]
- Allerberger, F.; Wotzer, G.; Dierich, M.P.; Moritz, C.; Fritsch, P.; Haas, W. Occurrence of swimmer’s itch in the Tyrol|Auftreten Von Badedermatitis in Tirol. Immun. Infekt. 1994, 22, 30–32. Available online: https://europepmc.org/article/med/7927463 (accessed on 16 January 2024).
- Hörweg, C.; Sattmann, H.; Auer, H. Cercarial dermatitis in Austria: Questionnaires as useful tools to estimate risk factors? Wien. Klin. Wochenschr. 2006, 118, 77–80. [Google Scholar] [CrossRef]
- Juhász, A.; Dán, Á.; Dénes, B.; Kucsera, I.; Danka, J.; Majoros, G. Egy ritka zoonosis: A Schistosoma turkestanicum vérmétely által okozott cercaria dermatitis Magyarországon. Orvosi Hetil. 2016, 157, 1579–1586. [Google Scholar] [CrossRef]
- Rizevsky, S.V.; Bodilovskaya, O.A.; Golubev, A.P.; Kurchenko, V.P. Capacity for long-term self-fertilization of the pulmonate mollusk Lymnaea stagnalis as a factor of preservation of human cercarial dermatitis foci. Dokl. Biol. Sci. 2012, 443, 109–112. [Google Scholar] [CrossRef]
- Arias, M.; Lomba, C.; Dacal, V.; Vázquez, L.; Pedreira, J.; Francisco, I.; Piñeiro, P.; Cazapal-Monteiro, C.; Suárez, J.L.; Díez-Baños, P.; et al. Prevalence of mixed trematode infections in an abattoir receiving cattle from northern Portugal and north-west Spain. Vet. Rec. 2011, 168, 408. [Google Scholar] [CrossRef]
- Mas-Coma, S.; Valero, M.A.; Bargues, M.D. Human and Animal Fascioliasis: Origins and Worldwide Evolving Scenario. Clin. Microbiol. Rev. 2022, 35, e00088-19. [Google Scholar] [CrossRef]
- Fraga de Azevedo, J.; Da Silva, J.B.; Coito, A.D.; Coelho, M.F.; Colaco, A.A. Portuguese focus of schistosomiasis. An. Inst. Med. Trop. 1948, 5, 175–222. [Google Scholar] [CrossRef]
- Salas-Coronas, J.; Bargues, M.D.; Lozano-Serrano, A.B.; Artigas, P.; Martínez-Ortí, A.; Mas-Coma, S.; Merino-Salas, S.; Vivas-Pérez, J.I.A. Evidence of autochthonous transmission of urinary schistosomiasis in Almeria (southeast Spain): An outbreak analysis. Travel Med. Infect. Dis. 2021, 44, 102165. [Google Scholar] [CrossRef]
- Rudko, S.P.; McPhail, B.A.; Reimink, R.L.; Froelich, K.; Turnbull, A.; Hanington, P.C. Non-resident definitive host presence is sufficient to sustain avian schistosome populations. Int. J. Parasitol. 2022, 52, 305–315. [Google Scholar] [CrossRef]
- McPhail, B.A.; Froelich, K.; Reimink, R.L.; Hanington, P.C. Simplifying Schistosome Surveillance: Using Molecular Cercariometry to Detect and Quantify Cercariae in Water. Pathogens 2022, 11, 565. [Google Scholar] [CrossRef]
- Loker, E.S.; DeJong, R.J.; Brant, S.V. Scratching the Itch: Updated Perspectives on the Schistosomes Responsible for Swimmer’s Itch around the World. Pathogens 2022, 11, 587. [Google Scholar] [CrossRef]
- Gordy, M.A.; Cobb, T.P.; Hanington, P.C. Swimmer’s itch in Canada: A look at the past and a survey of the present to plan for the future. Environ. Health 2018, 17, 73. [Google Scholar] [CrossRef]
- Pinto, H.A.; Pulido-Murillo, E.A.; de Melo, A.L.; Brant, S.V. Putative new genera and species of avian schistosomes potentially involved in human cercarial dermatitis in the Americas, Europe and Africa. Acta Trop. 2017, 176, 415–420. [Google Scholar] [CrossRef]
- Gohardehi, S.; Fakhar, M.; Madjidaei, M. Avian Schistosomes and Human Cercarial Dermatitis in a Wildlife Refuge in Mazandaran Province, Northern Iran. Zoonoses Public Health 2013, 60, 442–447. [Google Scholar] [CrossRef]
- Wulff, C.; Haeberlein, S.; Haas, W. Cream formulations protecting against cercarial dermatitis by Trichobilharzia. Parasitol. Res. 2007, 101, 91–97. [Google Scholar] [CrossRef]


| Country | Locality | Stage | Species | Accession Number | Reference |
|---|---|---|---|---|---|
| Austria | Reichersberger Au | cercaria | Trichobilharzia franki | MT763194-98 | Reier et al., 2020 [27] |
| Lake Pleschinger See | cercaria | Trichobilharzia physellae | OL434662-65 | Helmer et al., 2021 [22] | |
| Belarus | Lakes Naroch and Polonevichi | cercaria | Trichobilharzia szidati | GU350726; HM001253; HM001260-61 | Rizevsky et al., 2011 [26] |
| cercaria | Trichobilharzia franki | HM001254; HM001256 | |||
| cercaria | Trichobilharzia sp. | HM001257-59 | |||
| cercaria | Bilharziella polonica | HM001255; HM001262 | |||
| Lake Naroch | sporocyst/ cercaria | Trichobilharzia szidati | KP889985-KP890002 | Semyenova et al., 2015 [28] | |
| Lakes Great Shwakshty and Naroch | cercaria | Trichobilharzia szidati | MT112075-106; MT708486-99 | Chrisanfova et al., 2021 [29] | |
| Belgium | Lake Eau d’Heure | cercaria | Trichobilharzia franki | KX034088 | Caron et al., 2017 [6] |
| Kampenhout | cercaria | Trichobilharzia regenti | PP232105 | Schols et al., 2024 [30] | |
| Czech | cercaria | Trichobilharzia franki | AF356845 | Dvořák et al., 2002 [31] | |
| cercaria | Trichobilharzia regenti | AF263829 | |||
| cercaria | Trichobilharzia szidati | AF263828 | |||
| Trichobilharzia regenti | AY157190; AY157218; AY157244 | Lockyer et al., 2003 [32] | |||
| Trichobilharzia szidati | AY157191; AY157219; AY157245 | ||||
| cercaria | Trichobilharzia franki | AY713969; AY713973 | Rudolfova et al., 2005 [33] | ||
| cercaria | Trichobilarzia szidati | AY713961; AY713968; AY713972 | |||
| cercaria | Avian schistosomatid sp. | AY713963; AY713969 | |||
| Tovačov | egg/ miracidium | Trichobilharzia regenti | EF094538; EF094540 | Rudolfova et al., 2007 [34] | |
| fluke | Trichobilharzia szidati | EF094541 | |||
| fluke | Bilharziella polonica | EF094539 | |||
| cercaria | Trichobilharzia franki | FJ174530 | Brant & Loker, 2009 [35] | ||
| cercaria | Trichobilharzia szidati | GU233735-36 * | Rizevsky et al. 2011 [26] | ||
| cercaria | Trichobilharzia szidati | GU233739 * | Aldhoun et al., 2012 [36] | ||
| egg | Trichobilharzia regenti | GU233740 * | |||
| Novozámecký, Litovický, and Dolní Svitavský Ponds, Modřany and Loužek | sporocyst/cercaria/fluke | Avian schistosomatid sp. | FJ786027-30; JF734335; JF694008 | Aldhoun et al., 2012 [36] | |
| Denmark | Copenhagen (North) | cercaria | Trichobilharzia franki | KP271013 | Christiansen et al., 2016 [37] |
| cercaria | Trichobilharzia szidati | KP271014 | |||
| cercaria | Trichobilharzia regenti | KP271015 | |||
| Zealand, Jutland, and Funen | cercaria | Trichobilharzia sp. (Trichobilharzia anseri) | FJ469784-85; FJ469791 | Al-Jubury et al., 2021 [38] | |
| cercaria | Trichobilharzia anseri | MW538530; MW482445 | |||
| cercaria | Trichobilharzia franki | MW538531; MW482439-41; MW482443-44; MW482446 | |||
| fluke/ cercaria | Trichobilharzia szidati | MW482436-37; MW482447-49 | |||
| fluke | Trichobilharzia regenti | MW482450 | |||
| England | Tundry Pond (Hampshire) | cercaria | Trichobilharzia franki | KJ775865-69 | Lawton et al., 2014 [39] |
| Knowsley Safari (Prescot) | cercaria | Trichobilharzia sp. | ON987329-30 | Juhász et al., 2022 [40] | |
| cercaria | Trichobilharzia anseri | ON987331 | |||
| cercaria | Bilharziella polonica | ON987332-34 | |||
| Finland | Lakes Vuojärvi, Peurunkajärvi, and Konnevesi | cercaria | Trichobilharzia szidati | FJ609409-10 | Aldhoun et al., 2009 [41] |
| cercaria | Trichobilharzia franki | FJ609411 | |||
| cercaria | Avian schistosomatid sp. | FJ609412-14 | |||
| France | Marne and Lake Der-Chantecoq | cercaria | Trichobilharzia szidati | AY795570-71 | Ferté et al., 2005 [42] |
| cercaria | Trichobilharzia franki | AY795572-73 | |||
| Champagne region | fluke | Bilharziella polonica | DQ813437-42 | Bayssade-Dufour et al., 2006 [43] | |
| fluke | Dendritobilharzia pulverulenta | DQ813443 | |||
| Lake Annecy | cercaria/egg/ miracidium | Trichobilharzia regenti | EU413960; EU413967-70; EU413977-79 | Jouet et al., 2008 [44] | |
| cercaria | Trichobilharzia sp. | EU413961; EU413964; EU413970 | |||
| cercaria | Trichobilharzia franki | EU413962-63; EU413965-66; EU413971-76 | |||
| Lakes Der-Chantecoq and Annecy, Vanault les Dames, Beauvais, and Forêt d’Orient | fluke/egg | Trichobilharzia franki | FJ793813-22; FJ793874-83 | Jouet et al., 2009 [45] | |
| fluke/egg | Trichobilharzia regenti | FJ793823-49; FJ793884-95 | |||
| fluke/egg | Trichobilharzia szidati | FJ793896-97 | |||
| fluke/egg | Bilharziella polonica | FJ793850-57; FJ793898-99; FJ793900-07 | |||
| fluke/egg | Schistosomatidae sp. | FJ793858-73; FJ793908-22 | |||
| Lakes Der-Chantecoq and Annecy, and Beauvais | cercaria/fluke/egg | Trichobilharzia regenti | HM439484; HM439487; HM439494-99; HM439500-02 | Jouet et al., 2010 [46] | |
| egg | Trichobilharzia sp. | HM439493; HM439505 | |||
| Lakes Der-Chantecoq and Annecy, Beauvais, and Strasbourg | cercaria | Trichobilharzia franki | HM131131-41; HM131158-67; HM131176-84; HM13197-99; HM131200-02 | Jouet et al., 2010 [47] | |
| cercaria | Trichobilharzia sp. | HM131156-57; HM131192-96; HM131203-05 | |||
| Lake Annecy | cercaria | Trichobilharzia mergi | JX456170 | Kolarová et al., 2013 [24] | |
| Lakes Der-Chantecoq and Annecy, and Beauvais | fluke/egg | Trichobilharzia anseri | KP901355-56; KP901369; KP901376-79; KP901382-85 | Jouet et al., 2015 [23] | |
| Germany | Erlangen | cercaria | Trichobilharzia occelata | AF442689 | Helter et al., 2002 [48] |
| cercaria | Trichobilharzia regenti | AF442688 | |||
| Trichobilharzia occelata | AY157189; AY157217; AY157243 | Lockyer et al., 2003 [32] | |||
| cercaria | Trichobilharzia szidati | AY713971 | Rudolfova et al., 2005 [33] | ||
| cercaria | Trichobilharzia franki | FJ711767-68 | Brant & Loker, 2009 [35] | ||
| Hungary | fluke | Orientobilharzia turkestanicum | EU702749 | Majoros et al., 2010 [49] | |
| Eger, Hortobágy, and Gyulaj | cercaria | Trichobilharzia franki | MZ560932-4; MZ562961-63; MZ562965-66 | Juhász et al., 2022 [2] | |
| cercaria | Bilharziella polonica | MZ562959-60; MZ562964 | |||
| Iceland | fluke | Allobilharzia visceralis | DQ067561 | Kolarova et al., 2006 [50] | |
| Reykjavík, Hrísatjorn, Osland, Botnsvatn, Mývatn, Víkingavatn, and Landmannalaugar | cercaria | Trichobilharzia sp. | FJ469784-99; FJ469803-04; FJ469807 | Aldhoun et al., 2009 [51] | |
| cercaria | Trichobilharzia franki | FJ469805; FJ469806; FJ469808-12; FJ469816-17; FJ469819-21 | |||
| cercaria | Avian schistosomatid sp. | FJ469813; FJ469815; FJ469818; FJ469822 | |||
| Landmannalaugar and Reykjavik | fluke/egg | Trichobilharzia regenti | HM439484-86; HM439488-92; HM439503-04 | Jouet et al., 2010 [46] | |
| Botnsvatn, Helgavogur-Myvatn, and Raudavatn | cercaria | Trichobilharzia sp. | HM131142-55; HM131168-75; HM131185-91 | Jouet et al., 2010 [47] | |
| Botnsvatn | fluke/egg | Trichobilharzia mergi | JX456151-69; JX456170-72 | Kolarova et al., 2013 [24] | |
| cercaria/fluke/egg | Trichobilharzia anseri | KP901348-54; KP901357-68; KP901370-75; KP901380-81 | Jouet et al., 2015 [23] | ||
| Italy | Lake Vico | cercaria | Trichobilharzia franki | HM596077 | Cipriani et al., 2011 [52] |
| Lake Albano | cercaria | Trichobilharzia franki | MK053632; MK046867 | De Liberato et al., 2019 [14] | |
| Netherlands | cercaria | Trichobilharzia szidati | AY713970 | Rudolfova et al., 2005 [33] | |
| Norway | Lake Takvatn | cercaria | Trichobilharzia franki | KY513270-75 | Soldanová et al., 2017 [53] |
| Poland | cercaria | Trichobilharzia szidati | AY713965 | Rudolfova et al., 2005 [33] | |
| cercaria | Trichobilharzia franki | AY713964; AY713966 | |||
| Gdansk | egg | Trichobilharzia szidati | EF094530; EF094536 | Rudolfova et al., 2007 [34] | |
| egg | Trichobilharzia regenti | EF094533-35; EF094537 | |||
| egg | Trichobilharzia sp. | EF094531-32 | |||
| cercaria | Trichobilharzia szidati | MH190225-28 | Marszewska et al., 2018 [54] | ||
| cercaria | Trichobilharzia regenti | MH190224 | |||
| Lake Drawsko (West Pomerania Voivodeship) | cercaria | Trichobilharzia szidati | MT785880-82 | Stanicka et al., 2021 [55] | |
| Russia | cercaria | Trichobilharzia franki | GU980749-50 | Korsunenko et al., 2010 [56] | |
| cercaria | Trichobilharzia szidati | GU980751-53 | |||
| cercaria | Trichobilharzia regenti | GU980754-55 | |||
| Kargat River, Altufyevo and Olympiyskaya derevnya Ponds | cercariae | Trichobilharzia szidati | HM016851-57; JF838197-99; JF8381200-03 | Korsunenko et al., 2012 [57] | |
| Moscow ponds and Lake Onega | sporocyst/ cercaria | Trichobilharzia szidati | KP890003-21 | Semyenova et al., 2015 [28] | |
| Switzerland | Lake Geneva | cercaria | Trichobilharzia franki | AJ312041-46 | Picard & Jousson, 2001 [58] |
| cercaria/fluke | Trichobilharzia regenti | AJ312047-49 | |||
| Ukraine | Bilharziella polonica | AY157186; AY157214; AY157240 | Lockyer et al., 2003 [32] | ||
| Others (collaborations) | cercaria | Trichobilharzia regenti | DQ859919 | Webster et al., 2007 [59] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bispo, M.T.; Calado, M.; Maurício, I.L.; Ferreira, P.M.; Belo, S. Zoonotic Threats: The (Re)emergence of Cercarial Dermatitis, Its Dynamics, and Impact in Europe. Pathogens 2024, 13, 282. https://doi.org/10.3390/pathogens13040282
Bispo MT, Calado M, Maurício IL, Ferreira PM, Belo S. Zoonotic Threats: The (Re)emergence of Cercarial Dermatitis, Its Dynamics, and Impact in Europe. Pathogens. 2024; 13(4):282. https://doi.org/10.3390/pathogens13040282
Chicago/Turabian StyleBispo, Maria Teresa, Manuela Calado, Isabel Larguinho Maurício, Pedro Manuel Ferreira, and Silvana Belo. 2024. "Zoonotic Threats: The (Re)emergence of Cercarial Dermatitis, Its Dynamics, and Impact in Europe" Pathogens 13, no. 4: 282. https://doi.org/10.3390/pathogens13040282