In this study, a cold-deformed polycrystalline wire of TiNi shape-memory alloy supplied by Memory-Metalle GmbH (Weil am Rhein, Germany) was used. The average composition of the nickel-titanium ratio was 50.3:49.7 wt. %, as determined by the suppliers by means of a wet chemical method with a tolerance of 0.2 wt. %, and matched the relationship between the Ni content and the martensitic start temperature (M
s) found in these alloys [
7,
31]. A few different samples from the same TiNi wire were selected for the experiments, and the same thermal treatment was applied in order to verify the reproducibility of the various measurements. It consisted of annealing the samples at 950 °C for 1/2 h under high-purity argon and quenching them in ice water. Afterwards, the samples were aged at 400 °C for 1 h and then quenched again in ice water. Previous electron microscopy observations show that the samples exhibit equiaxed grains with diameters between 10 µm and 50 µm and included small 10–15-nm-sized particles of Ni
4Ti
3 precipitates. The 2-mm diameter wires were cut into disks of about 2-mm thickness for DSC measurements. They were carried out in a TA DSC Q2000 calorimeter (TA Instrument, New Castle, DE, USA), which presents high baseline stability and permits the direct measurement of the heat capacity. The specific heat capacity (
Cp) values obtained by this DSC technique have been found to depend slightly on the heating-cooling rate, so we have used a fixed rate of 10 °C/min to avoid ulterior discrepancies due to this rate variation. As it will be later discussed, up to 75 heating-cooling runs from −100 °C to 100 °C at rates of 10 °C/min were required to stabilize the observed drift of the calorimetric curves and obtain reproducible results.
2.1. Transformation Evolution under Cycling
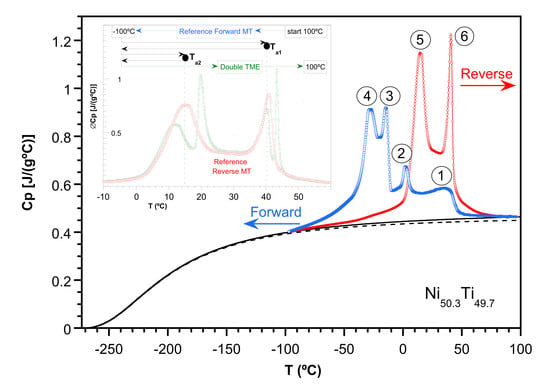
DSC measurements at 10 °C/min, which show the multistage martensitic transformation expected in aged polycrystalline TiNi samples, were performed. The evolution of the forward and reverse MTs multistep processes can be seen in
Figure 1.
In the first cooling run, a
Cp shoulder around 40 °C, followed by a neat high peak around 1 °C appeared. However, after successive cycles, some additional
Cp peaks took place, and the evolution of the traces nearly stabilized after 50 cycles. As regards the reverse MT, only two
Cp peaks were found. This behavior upon heating was maintained after cycling for up to 75 runs, and only small differences in the
Cp traces were observed. The corresponding temperatures, as well as the assigned numbering, are shown in
Figure 2 and
Table 1. We note that the changes in the thermal behavior under cycling did not affect Peak 1 during the forward MT or Peak 6 during the reverse MT, which also presented a good correlation in temperatures.
A direct way to find the evolution of the alloy mass transformed as a function of temperature is to examine the behavior of the enthalpy throughout the MT. However, a detailed study of the transformation and the associated thermodynamic functions requires a previous estimation of the specific heat baseline, which accounts for the alloy lattice contribution. It has been found that the usual linear extrapolation of the
Cp curve from both sides of the anomaly leads to significant errors, both in the actual MT temperature range and in the corresponding enthalpic values. A more accurate alternative, developed in previous works, is based on the previous calculation of the harmonic term (
Ch), anharmonic (
Canh), and electronic (
Ce) contributions to
Cp [
32].
In this case,
Canh has been established from the known values of the elastic constants [
33] and the thermal expansion tensor [
34] in the austenitic phase. This contribution has been extrapolated down to the low-temperature martensitic phase by means of the empirical Nernst-Lindemann law, whereas the electronic contribution was determined by using the free electron model [
35]. This last term is found to represent about 0.5% of the total
Cp at room temperature, around the limits of the experimental resolution. But, the main contribution to the specific heat in the intermediate temperatures comes from the harmonic
Ch. This quantity was obtained from the available phonon density of states g(ν) of a NiTi alloy with a very close atomic proportion [
36]. Small composition differences are found to be not relevant in the phonon spectrum and, as a consequence, in the calculated harmonic specific heat [
35]. Even, the direct Ni [
37] and Ti [
38] density of states data, conveniently weighted for the alloy composition, show very close results. In general, this last method is a reasonable alternative when the complete vibrational spectrum of the alloy is not available [
32]. As can be seen, for example, in
Figure 2, this theoretical calculation provides a confident baseline for the specific heat of the alloy lattice. The usual dynamic DSC measurements finally performed show small discrepancies with this baseline at both sides of the MT temperature range. The fit between the two curves takes place at
T = −63 °C and 97 °C upon heating and at
T = 72 °C and −97 °C upon cooling. That means that the actual MT temperature ranges are about 160 °C and 169 °C for the reverse and forward MTs, respectively. These values are much higher than those normally assigned when a conventional straight line is used below the calorimetric curves of the MT for the enthalpy calculations. However, it should be noted that the contribution to the MT enthalpy is much less significant around the extremes of the transformation, where the experimental curve approaches our theoretical baseline. Nevertheless, we stress that the enthalpy evolves with temperatures of major importance, because it is associated with the transformed mass fraction.
By using this baseline, the enthalpy associated with the complete MT has been determined upon cooling and upon heating for all the
Cp traces of the numerous cycles. In
Table 1, we present the mean values of the enthalpy Δ
H accompanied by the respective standard deviations. Within the limits of the experimental error (0.6 J/g), we can conclude that the forward and reverse MTs attain the same common Δ
H value (21.2 J/g). However, a small decrease (below 5%) is observed through the first 10 cycles, but afterwards this quantity remains practically constant. This small effect may be due to the progressive production of irreversible dislocations under the successive thermal cycles as it gradually reaches saturation. These results do not agree with those reported by other authors, who find striking differences in enthalpy, especially between the direct MT and the reverse MT for samples under thermal cycles [
9,
39,
40]. This notorious discrepancy can be due to the use of a common and precise baseline, which, as noted before, has a noticeable influence on the correct setting of the MT temperature range and also on the Δ
H determination. The calorimetric results after 75 previous thermal cycles are summarized in
Figure 2, which also includes the lattice contribution to the specific heat, as defined above. These experiments clearly show the progressive stabilization of the austenitic microstructure through thermal cycling. Finally, the nucleation of the different variants during the MT extends over a wide temperature range, although the sequential transformation shows up to four preferred temperature ranges associated with the observed specific heat anomalies. As will be discussed later, they must correspond to the nucleation in well-identified areas of the microstructure because of the remarkable heterogeneity of the austenitic framework, while the diversity of the associated thermal hysteresis values will provide direct information on the different energy barriers present during the forward transformation.
2.2. Study of the Transformation Stages
To establish the sequential process of the forward and reverse MTs, an attempt to obtain the correspondence among the various heating and cooling peaks was performed. For this purpose, the enthalpy contents of the various peaks observed upon cooling were compared with those detected upon heating. In order to study the transformation stages, several partial cooling-heating and heating-cooling experiments were performed. For the sake of clarity, as stated in
Figure 2 and
Table 1, the
Cp peaks representing these stages are successively numbered from 1 to 4 with respect to cooling and are numbered 5 and 6 with respect to heating, as shown in
Figure 3 and
Figure 4. The calorimetric experiments consisted of successive partial cooling runs (
Figure 3a) from 100 °C to a variable stop temperature (
Ts), followed by the corresponding heating runs (
Figure 3b) from
Ts back to 100 °C. As can be seen in
Figure 3, the selected
Ts values were 24, 10, −10, −20, −40, and −60 °C, respectively. The vertical lines appearing in the DSC signals are only to indicate the experimental stopping process and, though with no physical meaning, are maintained in the plot as approximate references for
Ts. These temperatures were selected to analyze each individual cooling transformation stage by means of partial forward MT. It has been observed that:
- -
Cooling down to those temperatures that include stage 1 (Ts1 = 24 °C and Ts2 = 10 °C) increases successively the Cp of stage 6 upon heating.
- -
Cooling down to Ts below stage 2 (Ts3 = −10 °C) shows an equal increase for Cp at both stages 5 and 6 upon heating.
- -
Cooling down to Ts below stage 3 (Ts4 = −20 °C) mainly increases the Cp values around stage 6 up to its total final value upon heating.
- -
Finally, cooling down to Ts below stage 4 (Ts5 = −40, Ts6 = −60 °C) only increases the Cp of stage 5, approaching its total final value upon heating.
In summary, we can conclude the following: stages 1 and 3 are mainly related to stage 6 and stage 4 is (only) related to stage 5, but stage 2 is related to both stages 5 and 6. Thus, stage 2 can be divided into 2a (related to 5) and 2b (related to 6).
These results can be strengthened and complemented by a partial reverse MT, as shown in
Figure 4.
In this experiment, the sample has been heated from −100 °C to a selected intermediate temperature between peaks 5 and 6:
Ts7 = 30 °C. The subsequent cooling trace down to −100 °C presents peak 2 and peak 4, but stage 3 is absent. This confirms that stage 5 is only related to stages 2 and 4, but not to stages 1 and 3. These results are consistent with the reference forward MT obtained by cooling from 100 °C to −100 °C. Within this scheme, calculations of the peak areas lead to the corresponding enthalpy assignment to the various stages, the values of which are shown in
Table 1. In addition, the relationships among the various MT stages are summarized in
Figure 5.
The microscopic description of the multistage MT together with the identification of the different phases have already been studied in previous works [
12,
15,
23]. Very similar complex phase sequences, close to the one presented here, have been found in the works by G. Fan et al. [
19] and B. Karbakhsh Ravari et al. [
16,
41]. In the latter study, up to a four-step transformation process was found in the forward MT of the samples after very similar thermal treatments, involving the same annealing temperature and time (950 °C, 1 h) and aging pattern (500 °C, 1 h), but no systematic thermal cycling seems to have been performed. However, their samples present a 50.6% Ni content, higher than that in our samples (50.3%), which would favor the Ni
4Ti
3 precipitation and, therefore, the presence of a heterogeneous nucleation. As we have described above, those authors also propose diverse relationships among the observed forward and reverse DSC peaks, but their results are somewhat different. In some cases [
16,
41], they have identified the various low-temperature phases (R and B19′ martensites) by means of in situ TEM measurements. As the phase sequences are extremely dependent on multiple physical parameters (mainly the composition and the thermomechanical history) these results cannot be confidently applied to our samples. In order to avoid these ambiguities, we have performed preliminary in situ TEM experiments in our quenched samples. Both the R and B19′ phases were identified, but it should be noted that the former is also found at very low temperatures well below the expected temperatures, as can be seen in
Figure 6. The typical R phase spots at 1/3 <111> B2 positions were observed in the microdiffraction pattern acquired in the zone marked with circle 1, and spots occurring at 1/2 of the <110> B2 reflections characteristic of the B19′ martensite were clearly observed for the pattern acquired in the zone marked with circle 2.
This observation is not in agreement with the low thermal hysteresis usually claimed for this transformation [
5,
20,
42]. However, in relation to this result, we must underline the following considerations. As it is well-known, the observed thermal hysteresis (Δ
TH) in this kind of first-order phase transition is a consequence of the presence of dissipative effects usually grouped in the general term “frictional work” (
Wf), which in general represents any kind of physical impediment requiring an additional energy to be overcome in order to observe the transition [
43].
As reported in previous works [
12,
16,
19,
23], the presence of a highly heterogeneous microstructure makes the martensitic plates’ nucleation highly dependent on the different environments in the austenitic frame. Up to now, some of these locations have been identified [
13,
14,
15,
16], and among them are the grain boundaries, grain interiors, defects, and some other parts of the austenitic crystal, which hinder the transformation. As this affects both the martensitic B19′ and R phases, it can explain a wide variety of thermal hysteresis values and, consequently, the presence of the multiple and highly overlapped specific heat peaks. In this context, the observed thermal hysteresis should not be considered as an intrinsic property, fixed for each structural transformation. To analyze this problem, we can resort to the study of the Gibbs energy determined from the specific heat data together with the temperatures of transformation, which allow us to estimate the values associated with the elastic contribution and the dissipative works. As shown in previous studies [
1], the intrinsic Gibbs energy (sometimes called the “chemical Gibbs energy”), as a function of temperature, corresponding both to the austenitic phase (
GA) and to the low-temperature martensitic state (
GM) well below the transformation temperature range, is directly obtained from the enthalpy and entropy of the crystal lattice. For this purpose, the general relation
G =
H −
TS can be used, together with the total enthalpy (Δ
HT) and entropy (Δ
ST) changes associated with the complete transformation. Within this frame, the true thermodynamic equilibrium temperature (
T0) between phases must be established by the
GA =
GM condition. However, the peculiarities of the plates’ nucleation in these alloys do not permit such a simple description. The difficulties for nucleation, which arise mainly from the crystallographic distortion of the crystal lattice during the transformation from the high-temperature cubic phase to the less symmetric low-temperature phases, lead to an elastic deformation of the variants [
5,
43]. This fact alters the effective value of the Gibbs energy in the martensitic phases, which can be now written as
=
GM +
Gel. So, the actual phase equilibrium temperature (
) for every individual plate must satisfy the condition:
=
GA. As the values for
Gel are different for the various single plates, so are their local equilibrium temperatures, providing a satisfactory description of the usual spread of the first-order martensitic transformations into a wide temperature range. That means that the distribution of the
Gel among the plates explains the sequential character of these transformations, whereas, as cited above,
Wf remains responsible for the observed Δ
TH. For the sake of clarity,
Figure 7 offers a schematic drawing to show the role of the quantities involved in the transformation of each plate.
In some cases, when a common thermal hysteresis value can be assigned for all the plates, the
Gel and
Wf values can be easily separated, as it is the case, for example, for some single crystals of Cu-Al-Ni shape-memory alloys [
28].The direct relationship between
Wf and Δ
TH makes this last quantity a good measure of the extra energy required for the nucleation of the different martensite variants in the austenitic matrix. It is found that Δ
TH is higher in polycrystalline samples than in single crystals [
28,
44,
45] and shows a marked dispersion of values when we have a complex austenitic microstructure. This is the case of the thermally aged TiNi sample, showing a convoluted phase transformation sequence, where no common value for the plates phase transition Δ
TH is found. It is manifest from the lack of symmetry between the observed
Cp curves upon cooling and upon heating, which precludes a precise evaluation both of
Gel and of
Wf for each plate. However, the wide range of Δ
TH observed values indicates the highly heterogeneous austenitic framework. The Δ
TH values found for the various
Cp peaks (using the tip of the peaks temperatures
Ti as reference) can be obtained from the data in
Table 1. The minimum value corresponds to peaks 1 and 6 (3.7 °C), probably related to the Austenite-R phase transformation near the grain boundaries. On the other hand, the maximum one is about 58 °C between peaks 3 and 6 and corresponds to the massive martensitic nucleation in the grains bulk, in agreement with the highest enthalpy value shown by this last forward transformation step (11.6 J/g, about 50% of the total transformation enthalpy). It leads to an estimation of about 4 J/g for
Wf and 2.5 J/g to
Gel, if we assume
= (
T3 +
T6)/2 = 286 °C. In total, they represent 6% of the average value of G at this temperature. As expected, all these figures are significantly higher than those found in more homogeneous microstructures [
28].
Leaving apart the evident limitations, we finally stress the fact that the procedure described above constitutes an efficient tool for a first estimation of the energies associated with the multistep martensitic transformations, common in these materials, obtained by means of simple and easy experiments. There is another point that must be noted. Traditionally it has been argued that the R phase was buffered during the nucleation of martensite B19′, which justifies the presence of only one
Cp peak in the reverse transformation. Nevertheless, our experiments summarized in
Figure 3,
Figure 4 and
Figure 5 and in
Table 1, show that the R phase nucleated in particular environments is not transformed to B19′. This fact becomes evident in the in situ TEM observations, where the R phase was undoubtedly identified at temperatures well below the end of transformation stage 4, which should correspond to the austenite–B19′ transformation. Finally, the elastic energy relaxation processes in the low-temperature phases may be the origin of the reduction of the observed steps in the reverse transformation, but more experimental work is necessary to clarify these points. Further TEM (using a Philips CM200 microscope, Amsterdam, The Netherlands) experiments are in progress in order to accomplish this task.
2.3. Evidence of the Temperature Memory Effect
The TME appears when partial heating cycles are performed from the low-temperature martensitic phase to an intermediate temperature (the so-called “arrested temperature”,
Ta) between austenitic start temperature (
As) and austenitic finish temperature (
Af). It is characterized by a noticeable delay of the MT up to higher temperatures in the subsequent total heating cycle (the “reverse” MT), and it is considerably increased by the repetition of the partial cycles to
Ta, previous to the final total MT (the so-called “hammer effect”) [
25,
46]. As two independent stages (5, 6) are present in the reverse transformation, we have examined the presence of the expected TME under partial heating/cooling cycles in both cases by means of the thermal sequence shown in
Figure 8.
The experiments were started at 100 °C to guarantee that any previous TME is previously erased. The arrested temperatures were
Ta1 = 40 °C and
Ta2 = 15 °C close to the 6 and 5 peak temperatures, respectively. We note that the second partial heating up to
Ta2 does not eliminate the first effect associated with
Ta1, as
Ta1 >
Ta2 [
25,
47]. The final heating run undoubtedly shows the characteristic
Cp peak split and confirms the presence of a clear TME in both stages. In the same figure, the
Cp reference curve and the one showing the TME are plotted. Both the additional
Cp peaks and the associated delay of the MT to higher temperatures are significantly larger in these TiNi alloys when compared, for example, with those observed in CuAlNi [
32].
The repetition of the partial reverse MT from the martensitic state to the same arrested temperature
Ta, is found to increase the TME [
25,
46]. To characterize this effect (the so-called “hammer” effect), we have focused only on stage 5 and selected
Ta = 17 °C. After cooling from the austenitic phase, the partial sequence (
ps): −100 °C → 17 °C → −100 °C is repeated
n times (
ps = 0, 1, 2…,
n−1). Then, a complete reverse MT: −100 °C → 100 °C is made, and this last result is called the “Hammer
n” curve. Independent experiments for
n = 1 to
n = 6 and
n = 20 were performed. An example of these experiments for
n = 6 is presented in
Figure 9, where the various heating and cooling
Cp curves are depicted after subtracting the calculated baseline. The reference cooling and heating curves (those which do not exhibit TME) are also plotted for comparison. The progressive delay of the MT is clearly shown by the
ps heating curves 0–5 below
Ta when compared with the reference curve. This means that at
Ta the remaining martensite phase fraction is increased with cycles. Accordingly, the corresponding cooling curves also confirm this fact, as the area below the peaks clearly diminish after each cycle. It should be noted that only peaks 2 and 4 show this effect, but, as expected, peak 3 does not, being not present in all these cooling curves. This result again confirms that stage 5, upon heating, is only related to stages 2 and 4 on cooling as discussed before (see
Figure 5 and
Table 1). After the multiple partial cycling, the final complete MT shows the “hammer 6” TME curve with a high increase of the secondary peak (5 s) when compared with the single TME (
n = 1), as seen in
Figure 9. We finally note that, once the TME has been erased, the last cooling curve corresponding to the complete forward MT fits quite well the initial “reference” curve.
In
Figure 10, the final complete MT heating traces for “Hammer
n” (
n = 1–6 and
n = 20) around stage 5 are presented. As defined above, each “Hammer
n” curve represents the complete reverse MT after
n previous partial cycles. The progressive increase of the secondary peak on stage 5 (5 s) is evident from these results, whereas stage 6 remains practically unchanged. The enthalpy decrease below
Ta, is found to match the corresponding increase of the secondary peak (5 s). An example of the evolution of the Δ
Cp below
Ta in the 20 previous partial cycles, before the “Hammer 20” curve, is shown in
Figure 11.
These measurements make it possible to calculate, by means of numerical integration, the enthalpy associated with each one of the partial transformations (
), which are directly related to the mass transformed over the successive heating cycles up to
Ta:
As can be seen in
Figure 12, an almost exponential decrease in this quantity is observed as a function of
ps. The delay of the transformation approaches 50% of the initial value for
n > 20. In previous works, similar results were explained by the progressive redistribution of the elastic energy among the martensitic plates by means of a simple relaxation model [
28,
45]. However, the complexity of the multistage transformation in these TiNi polycrystalline samples does not permit such an approach.