Influence of Powder Surface Contamination in the Ni-Based Superalloy Alloy718 Fabricated by Selective Laser Melting and Hot Isostatic Pressing
Abstract
:1. Introduction
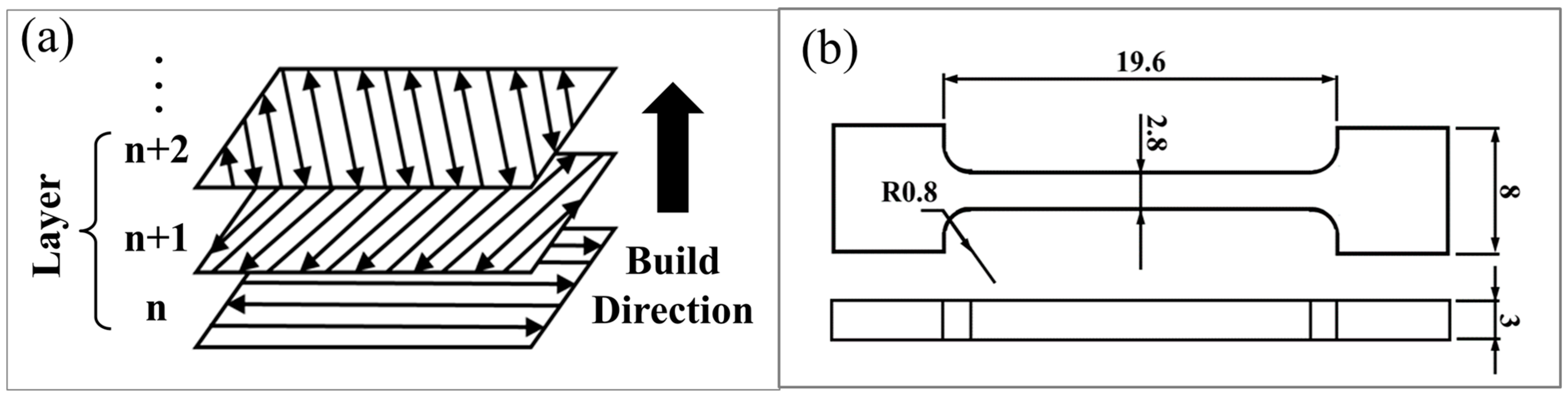
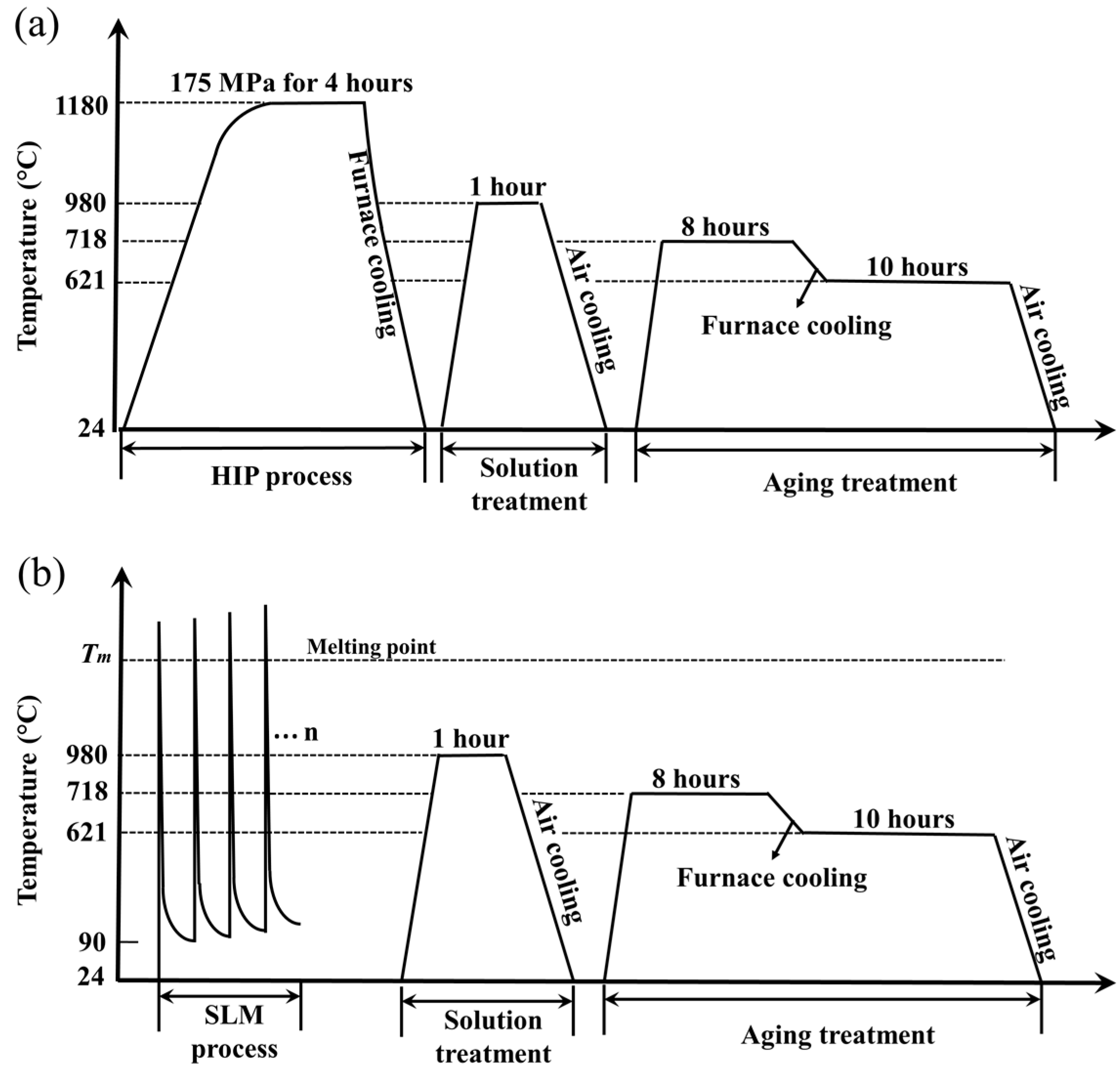
2. Materials and Experimental Procedure
3. Results
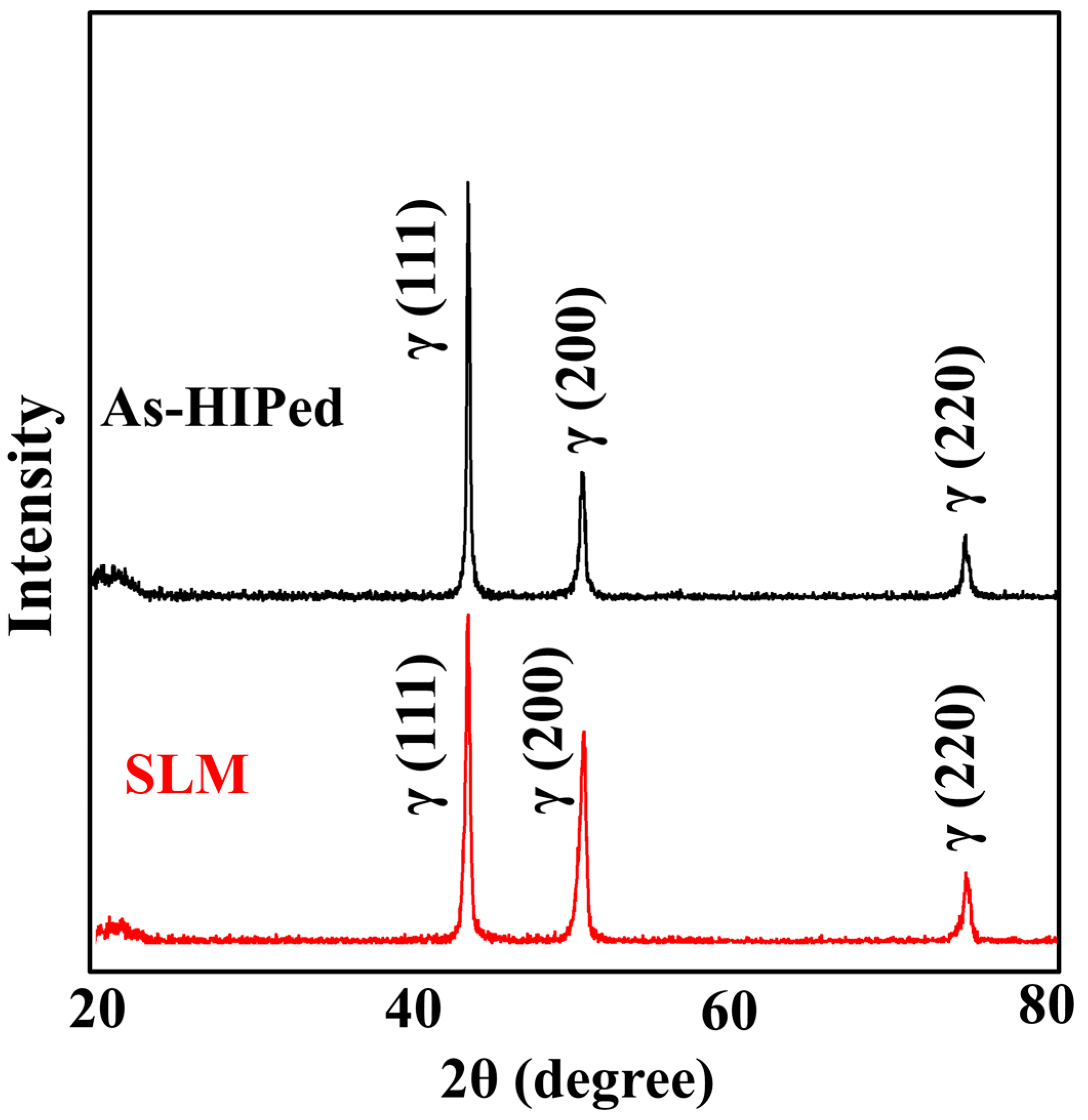
3.1. Microstructures of as-HIPed Specimen and as-Built Specimen
3.2. Influence of the Heat-Treatment Process
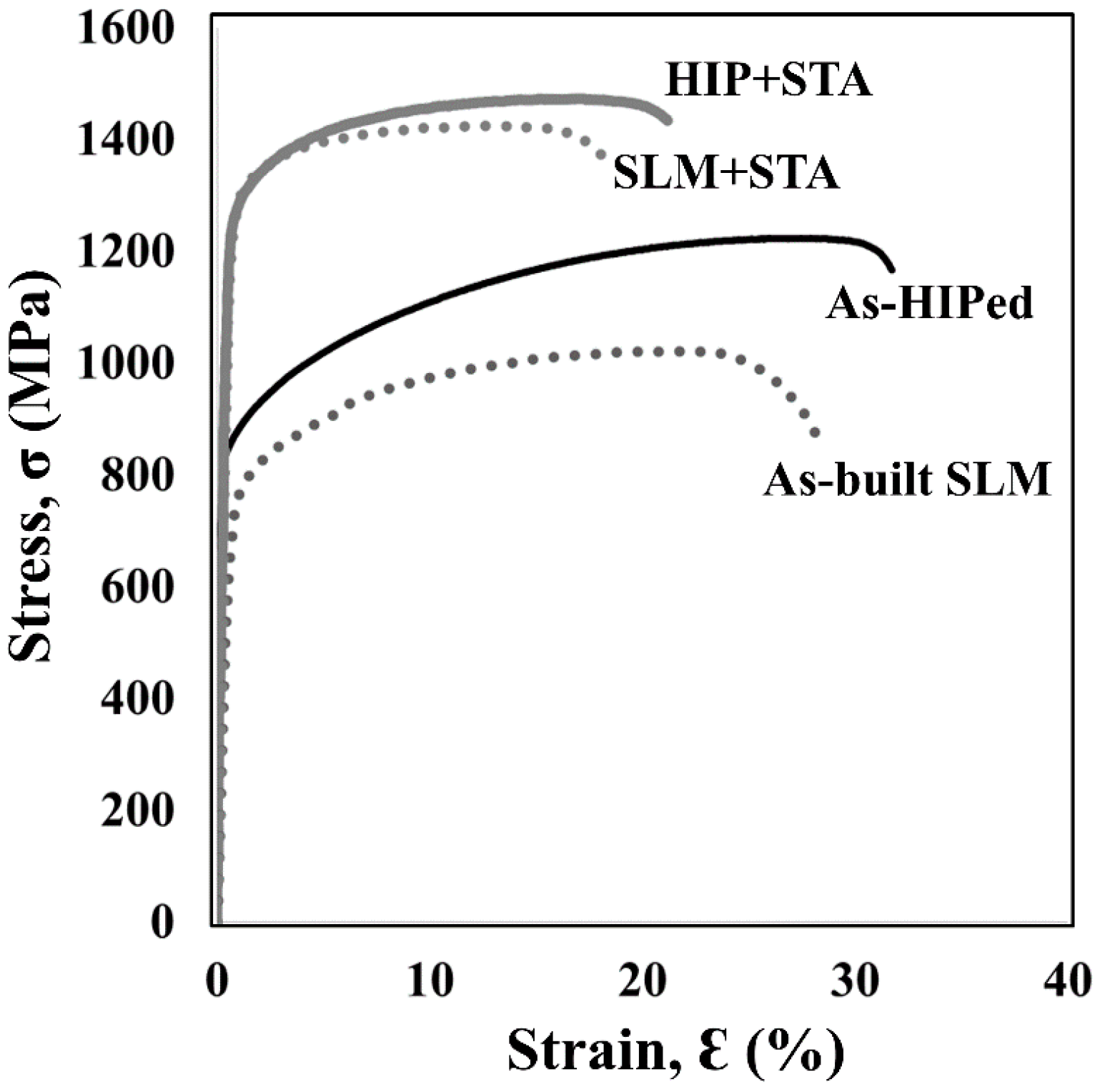
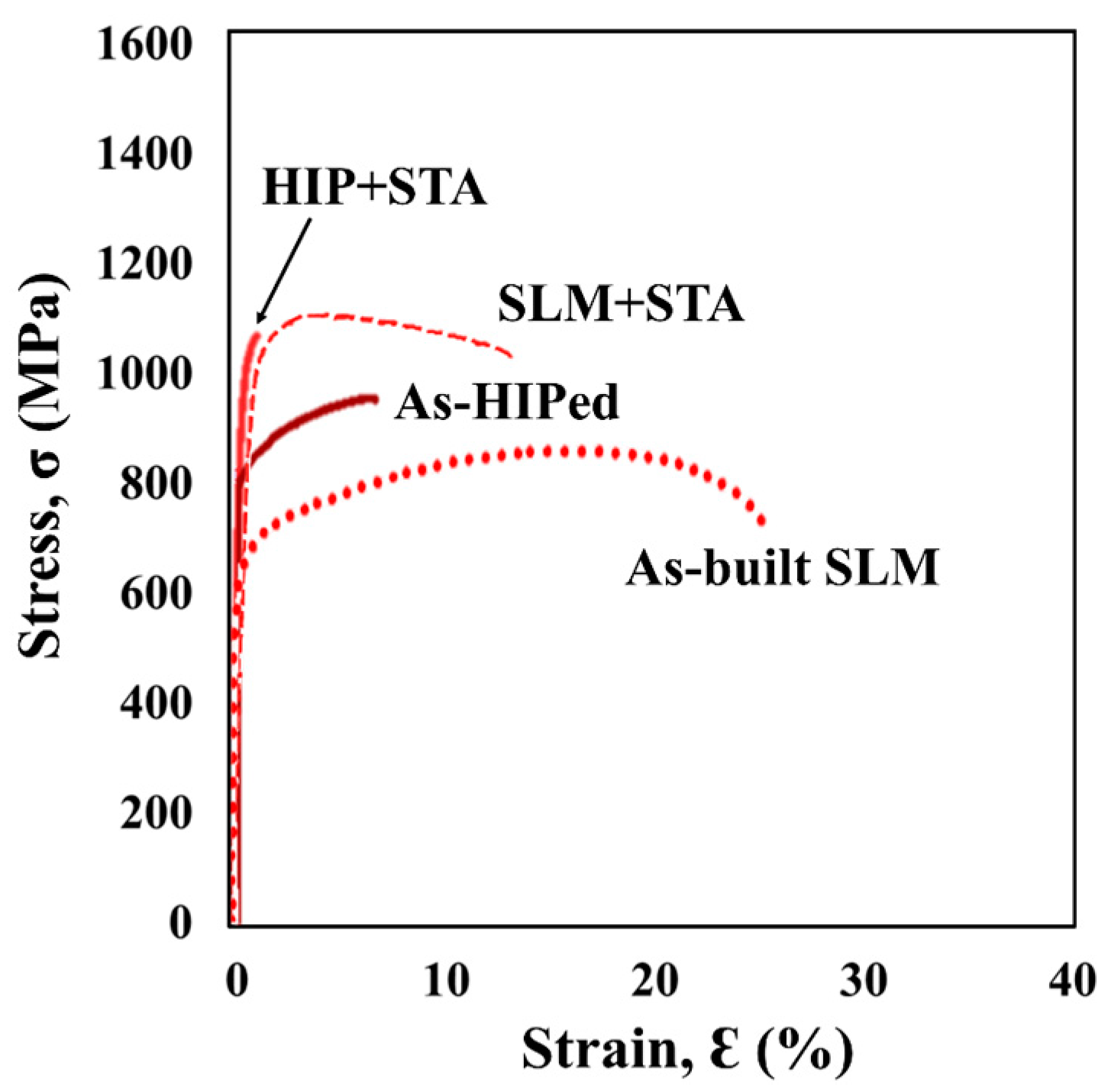
3.3. Tensile Properties
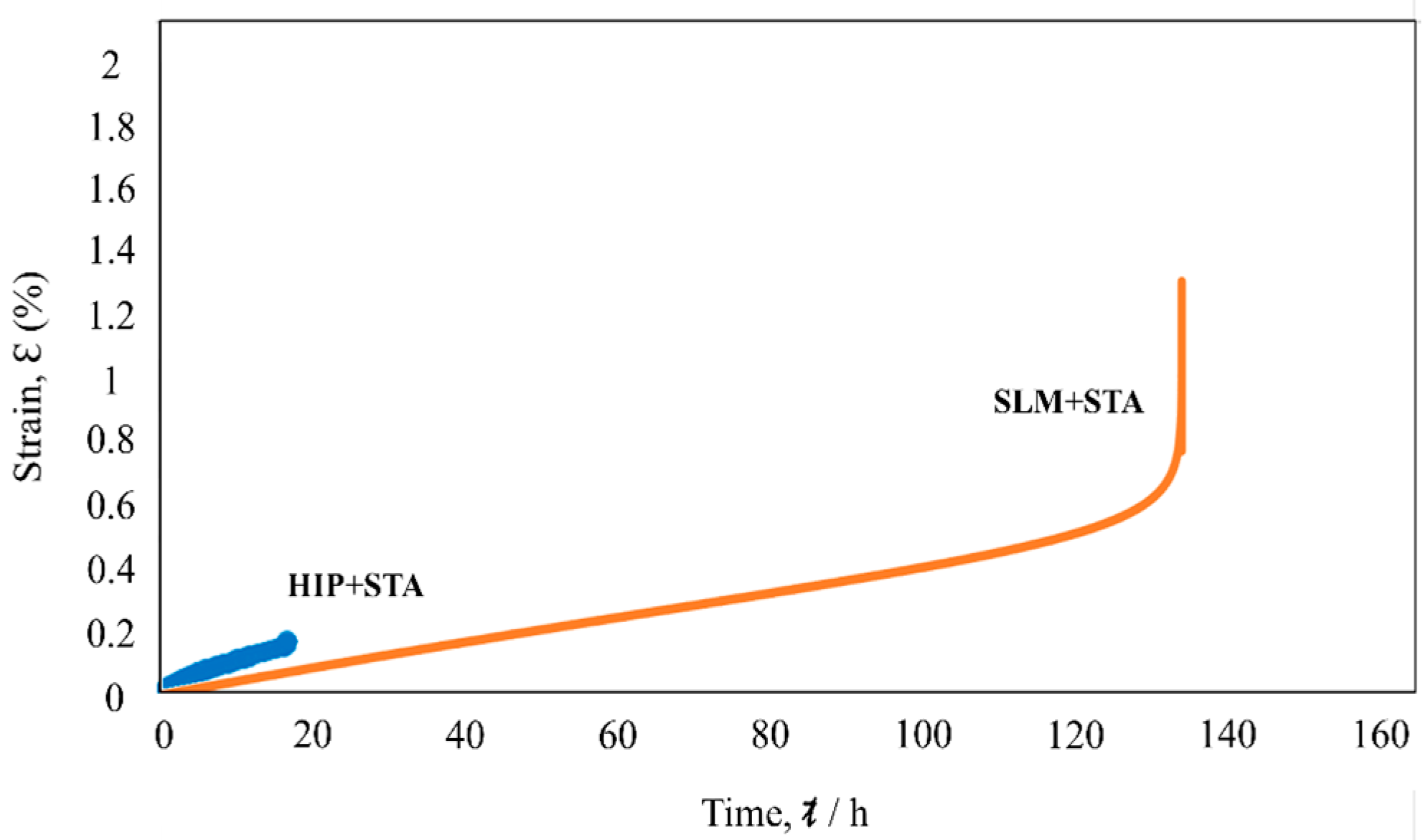
3.4. Creep Properties
4. Discussion
4.1. Grain Morphologies of SLM Materials
4.2. The Formation of PPB in HIP Materials
4.3. The Effects of PPB on the Mechanical Properties of HIP Specimens at 650 °C
4.4. Free-PPB Microstructure in SLM Materials
5. Conclusions
- (1)
- Continuous precipitates were clearly localized along the PPB in HIP materials, while SLM materials showed a microstructure free of PPBs and an extremely low number of contaminants.
- (2)
- The columnar grains in the SLM specimens were attributed to epitaxial growth.
- (3)
- The strengths and ductilities of the SLM + STA specimen were comparable to those of the conventionally wrought specimen both at room temperature and 650 °C. However, a drop was observed in the ductility of the HIP material at 650 °C.
- (4)
- The brittle precipitates formed in the HIP materials, such as oxy-carbides and δ phases, tended to lead to interface decohesion and resulted in premature fracture with poor ductility at 650 °C.
- (5)
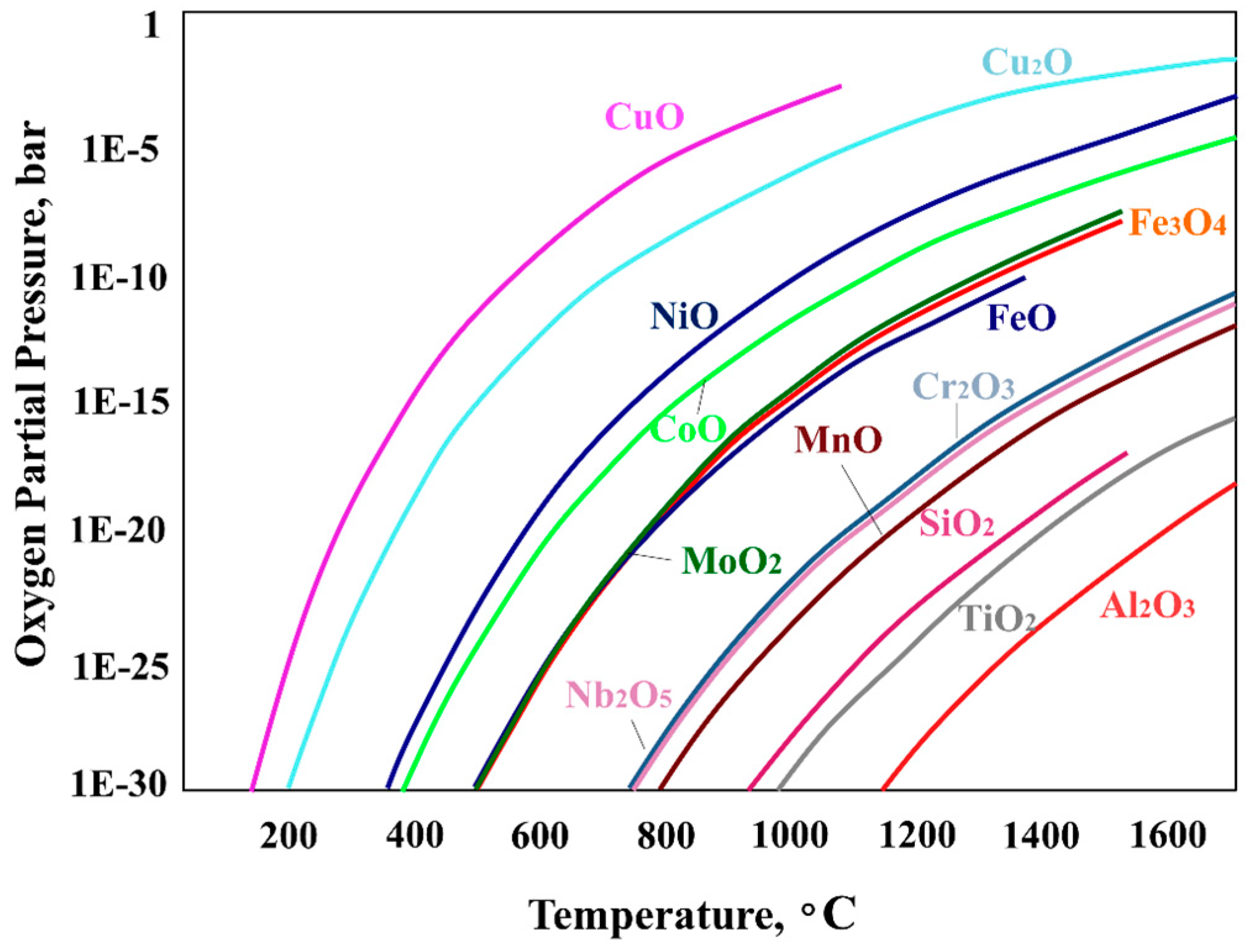
- The main reason that PPB precipitate networks formed during HIPing is that the HIP temperature was held at a temperature suitable for forming Al2O3 for a long time.
- (6)
- In the SLM process, Al2O3, which became a nucleation site of precipitates, is not easily formed, and the PPB precipitates network is churned and spread uniformly during the process.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yeh, A.C.; Lu, K.W.; Kuo, C.M.; Bor, H.Y.; Wei, C.N. Effect of serrated grain boundaries on the creep property of Inconel 718 superalloy. Mater. Sci. Eng. A 2011, 530, 525–529. [Google Scholar] [CrossRef]
- Chamanfar, A.; Sarrat, L.; Jahazi, M.; Asadi, M.; Weck, A.; Koul, A.K. Microstructural characteristics of forged and heat treated Inconel-718 disks. Mater. Des. 2013, 52, 791–800. [Google Scholar] [CrossRef]
- Ghosh, S.; Yadav, S.; Das, G. Study of standard heat treatment on mechanical properties of Inconel 718 using ball indentation technique. Mater. Lett. 2008, 62, 2619–2622. [Google Scholar] [CrossRef]
- Bai, Q.; Lin, J.; Tian, G.; Dean, T.A.J. Review and analysis of powder prior boundary (PPB) formation in powder metallurgy processes for nickel-based super alloys. J. Powder Metall. Min. 2015, 4. [Google Scholar] [CrossRef]
- Lee, S.C.; Chang, S.H.; Tang, T.P.; Ho, H.H.; Chen, J.K. Improvement in the microstructure and tensile properties of Inconel 718 superalloy by HIP treatment. Mater. Trans. 2006, 47, 2877–2881. [Google Scholar] [CrossRef]
- Rao, G.A.; Kumar, M. High performance stainless steel via powder metallurgy hot isostatic pressing. Mater. Sci. Technol. 1997, 13, 1027–1031. [Google Scholar] [CrossRef]
- Gessinger, G.H. Powder Metallurgy of Superalloys; Butteriworth & Co.: London, UK, 1984. [Google Scholar]
- Maurer, G.E.; Castledine, W.; Schweizer, F.A.; Mancuso, S. Development of HIP consolidated P/M superalloys for conventional forging to gas turbine engine components. In Proceedings of the 8th International Symposium on Superalloys, Pittsburgh, PA, USA, 28 September–1 October 2014; pp. 645–652. [Google Scholar]
- Furrer, D.; Fecht, H. Ni-based superalloys for turbine discs. JOM 1999, 51, 14–17. [Google Scholar] [CrossRef]
- Rao, G.A.; Srinivas, M.; Sarma, D.S. Effect of solution treatment temperature on microstructure and mechanical properties of hot isostatically pressed superalloy Inconel* 718. Mater. Sci. Technol. 2004, 20, 1161–1170. [Google Scholar] [CrossRef]
- Bai, Q.; Lin, J.; Jiang, J.; Dean, T.A.; Zou, J.; Tian, G. A study of direct forging process for powder superalloys. Mater. Sci. Eng. A 2015, 621, 68–75. [Google Scholar] [CrossRef]
- Haase, C.; Bültmann, J.; Hof, J.; Ziegler, S.; Bremen, S.; Hinke, C.; Schwedt, A.; Prahl, U.; Bleck, W. Exploiting process-related advantages of selective laser melting for the production of high-manganese steel. Materials 2017, 10, 56. [Google Scholar] [CrossRef] [PubMed]
- Ellyson, B.; Brochu, M.; Brochu, M. Characterization of bending vibration fatigue of SLM fabricated Ti-6Al-4V. Int. J. Fatigue 2011, 19, 389–395. [Google Scholar] [CrossRef]
- AlMangour, B.; Grzesiakc, D.; Yang, J.M. Selective laser melting of TiB2/H13 steel nanocomposites: Influence of hot isostatic pressing post-treatment. J. Mater. Process. Technol. 2017, 244, 344–353. [Google Scholar] [CrossRef]
- Kuo, Y.L.; Horikawa, S.; Kakehi, K. The effect of interdendritic δ phase on the mechanical properties of Alloy 718 built up by additive manufacturing. Mater. Des. 2017, 116, 411–418. [Google Scholar] [CrossRef]
- Kuo, Y.L.; Horikawa, S.; Kakehi, K. Effects of build direction and heat treatment on creep properties of Ni-base superalloy built up by additive manufacturing. Scr. Mater. 2017, 129, 74–78. [Google Scholar] [CrossRef]
- Donachie, M.J.; Donachie, S.J. Superalloys: A Technical Guide, 2nd ed.; ASM International: Geauga County, OH, USA, 2002. [Google Scholar]
- Zhang, J.; Singer, R.F. Hot tearing of nickel-based superalloys during directional solidification. Acta Mater. 2002, 50, 1869–1879. [Google Scholar] [CrossRef]
- Chang, S.; Lee, S.; Tang, T.; Ho, H. Influences of soaking time in hot isostatic pressing on strength of inconel 718 superalloy. Mater. Trans. 2006, 47, 426–432. [Google Scholar] [CrossRef]
- Jia, Q.; Gu, D. Selective laser melting additive manufacturing of Inconel 718 superalloy parts: Densification, microstructure and properties. J. Alloys Compd. 2014, 585, 713–721. [Google Scholar] [CrossRef]
- Zhang, B.; Lee, X.; Bai, J.; Guo, J.; Wang, P.; Sun, C.; Nai, M.; Qi, G.; Wei, J. Study of selective laser melting (SLM) Inconel 718 part surface improvement by electrochemical polishing. Mater. Des. 2017, 116, 531–537. [Google Scholar] [CrossRef]
- Kanagarajah, P.; Brenne, F.; Niendorf, T.; Maier, H.J. Inconel939 processed by selective laser melting: Effect of microstructure and temperature on the mechanical properties under static and cyclic loading. Mater. Sci. Eng. A 2013, 588, 188–195. [Google Scholar] [CrossRef]
- Rao, G.A.; Kumar, M.; Srinivas, M.; Sarma, D.S. Effect of standard heat treatment on the microstructure and mechanical properties of hot isostatically pressed superalloy inconel 718. Mater. Sci. Eng. A 2003, A355, 114–125. [Google Scholar] [CrossRef]
- Crompton, J.S.; Hertzberg, R.W. Analysis of second phase particles in a powder metallurgy HIP nickel-base superalloy. J. Mater. Sci. 1986, 21, 3445–3454. [Google Scholar] [CrossRef]
- Olakanmi, E.O.; Cochrane, R.F.; Dalgarno, K.W. A review on selective laser sintering/melting (SLS/SLM) of aluminium alloy powders: Processing, microstructure, and properties. Prog. Mater. Sci. 2015, 74, 401–477. [Google Scholar] [CrossRef]
- DuPont, J.N.; Lippold, J.C.; Kiser, S.D. Welding Metallurgy and Weldability of Nickel Base Alloys; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009. [Google Scholar]
- He, L.M.; Su, Y.-F.; Allard, L.F.; Lance, M.J.; Lee, W.Y. Effects of preoxidation on the nucleation and growth behavior of chemically vapor-deposited α-Al2O3 on a single-crystal Ni-based superalloy. Metall. Trans. A 2004, 35, 1113–1124. [Google Scholar] [CrossRef]
- Hryha, E.; Dudrova, E.; Nyborg, L. On-line control of processing atmospheres for proper sintering of oxidation-sensitive PM steels. J. Mater. Process. Technol. 2012, 212, 977–987. [Google Scholar] [CrossRef]
- Wallace, W.; Wiebe, W.; Whelan, W.P.; Dainty, R.V.; Terada, T. The effect of grain-boundary structure on the tensile fracture behavior of hot-isostatically pressed 713 LC Alloy Compacts. Powder Metall. 1972, 16, 416–435. [Google Scholar] [CrossRef]
- Tetelman, A.S.; Mcevily, A.J., Jr. Fracture of Structural Materials; John Wiley & Sons Inc.: New York, NY, USA, 1967; pp. 193–233. [Google Scholar]
- Broek, D. Elementary Engineering Fracture Mechanics; Martinus Nijhoff: Leiden, The Netherlands, 1984. [Google Scholar]
- Idell, Y.; Levine, L.E.; Allen, A.J.; Zhang, F.; Campbell, C.E.; Olson, G.B.; Gong, J.; Snyder, D.R.; Deutchman, H.Z. Unexpected δ-phase formation in additive-manufactured Ni-based superalloy. JOM 2016, 68, 950–959. [Google Scholar] [CrossRef]
- Campo, E.; Turco, C.; Catena, V. The correlation between heat treatment, structure and mechanical characteristics in Inconel 718. Metall. Sci. Technol. 1985, 3, 16–21. [Google Scholar]
- Radavich, J.F.; Korth, G.E.; Antolovich, S.D.; Stusrud, R.W.; MacKay, R.A.; Khan, T.; Kissinger, R.D.; Klarstrom, D.L. High temperature degradation of Alloy 718 after longtime exposures. In Proceedings of the 7th International Symposium on Superalloys, Seven Springs, PA, USA, 20–24 September 1992; pp. 497–506. [Google Scholar]
- Davis, J.R. ASM Specialty Handbook: Heat-Resistant Materials; ASM International: Geauga County, OH, USA, 1997. [Google Scholar]
- Chiou, M.S.; Jian, S.R.; Yeh, A.C.; Kuo, C.M. Effects of aluminum addition on the high temperature oxidation behavior of CM-247LC Ni-based superalloy. Int. J. Electrochem. Sci. 2015, 10, 5981–5993. [Google Scholar]
- German, R. Powder Metallurgy and Particulate Materials Processing: The Processes, Materials, Products, Properties and Applications; Metal Powder Industries Federation: Princeton, NJ, USA, 2005. [Google Scholar]
- Abe, F.; Osakada, K.; Shiomi, M.; Matsumoto, M.; Shiomi, M. The manufacturing of hard tools from metallic powders by selective laser melting. J. Mater. Process. Technol. 2001, 111, 210–213. [Google Scholar] [CrossRef]
- Das, S.; Beaman, J.J. Direct Selective Laser Sintering of Metals. U.S. Patent 6,676,892 B2, 13 January 2004. [Google Scholar]
- Wang, X.; Read, N.; Carter, L.N.; Ward, R.M.; Attallah, M.M. Defect formation and mitigation in selective laser melting of high γ′ Ni-base superalloys. In Proceedings of the 13th International Symposium on Superalloys, Seven Springs, PA, USA, 11–15 September 2016; pp. 351–358. [Google Scholar]








| IN718 | Cr | Nb | Mo | Ti | Al | Co | Cu | C | Si, Mn | P, S | B | O | Fe | Ni |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HIP | 18.4 | 4.98 | 3.3 | 0.84 | 0.57 | 0.1 | 0.01 | 0.026 | 0.08 | 0.02 | 0.001 | 0.014 | 17.2 | Bal. |
| SLM | 19.6 | 5.05 | 2.85 | 1.10 | 0.46 | 0.03 | 0.05 | 0.04 | 0.04 | 0.0 | 0.002 | 0.019 | Bal. | 52.59 |
| σ0.2 (MPa) | σT (MPa) | εf (%) | |
|---|---|---|---|
| As-HIPed | 833 | 1225 | 32.1 |
| As-built SLM | 677 | 1023 | 28.1 |
| HIP + STA | 1125 | 1473 | 21.2 |
| SLM + STA | 1271 | 1425 | 18.6 |
| Wrought + STA | 1216 | 1435 | 24.0 |
| σ0.2 (MPa) | σT (MPa) | εf (%) | |
|---|---|---|---|
| As-HIPed | 844 | 978.0 | 6.8 |
| As-built SLM | 594 | 862.0 | 25.1 |
| HIP + STA | 1031 | 1093 | 0.9 |
| SLM + STA | 1042 | 1142 | 10.1 |
| Wrought + STA | 1028 | 1139 | 10.5 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuo, Y.-L.; Kakehi, K. Influence of Powder Surface Contamination in the Ni-Based Superalloy Alloy718 Fabricated by Selective Laser Melting and Hot Isostatic Pressing. Metals 2017, 7, 367. https://doi.org/10.3390/met7090367
Kuo Y-L, Kakehi K. Influence of Powder Surface Contamination in the Ni-Based Superalloy Alloy718 Fabricated by Selective Laser Melting and Hot Isostatic Pressing. Metals. 2017; 7(9):367. https://doi.org/10.3390/met7090367
Chicago/Turabian StyleKuo, Yen-Ling, and Koji Kakehi. 2017. "Influence of Powder Surface Contamination in the Ni-Based Superalloy Alloy718 Fabricated by Selective Laser Melting and Hot Isostatic Pressing" Metals 7, no. 9: 367. https://doi.org/10.3390/met7090367




