Novel Recycling Method for Boron Removal from Silicon by Thermal Plasma Treatment Coupled with Steam and Hydrogen Gases
Abstract
:1. Introduction
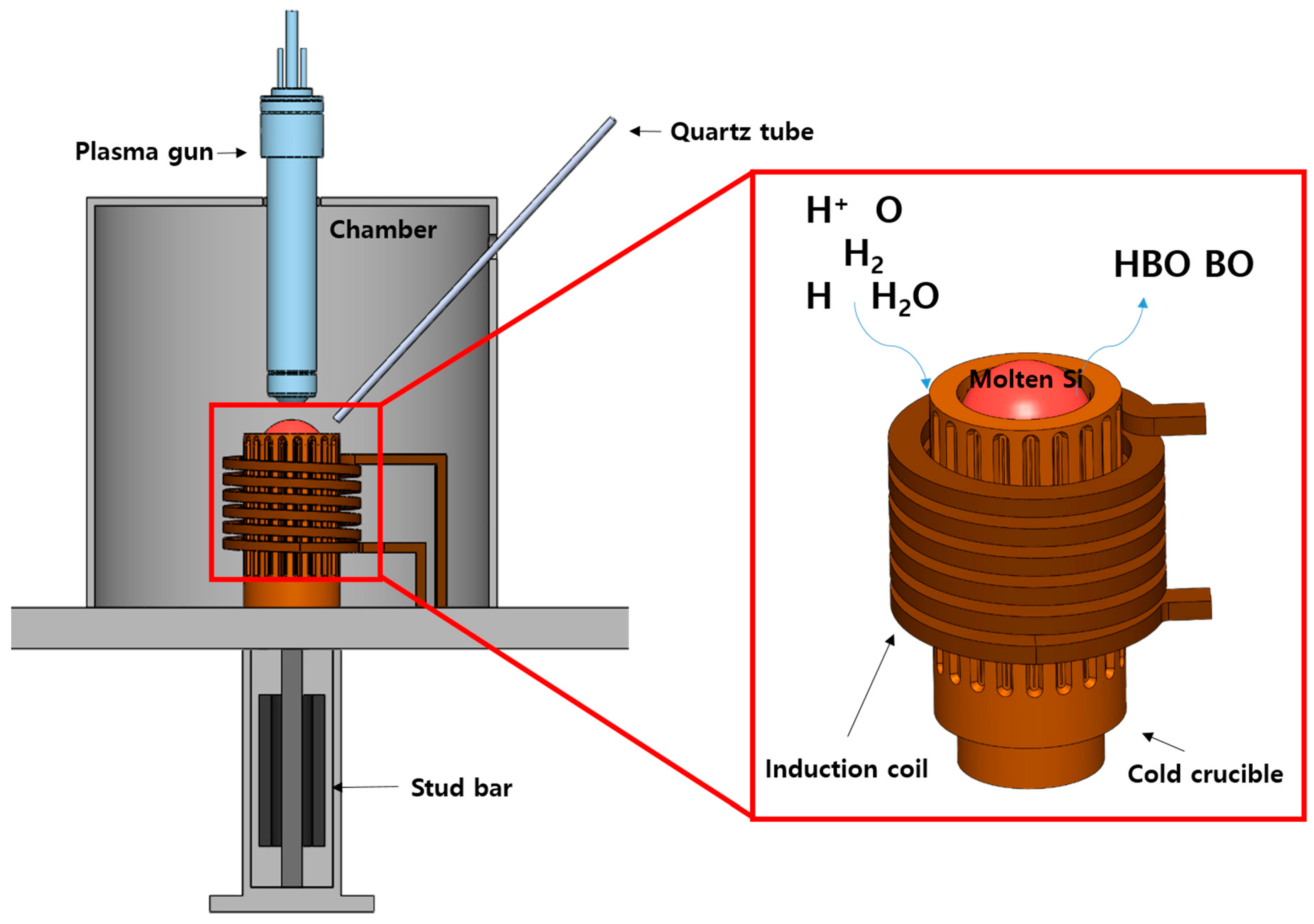
2. Materials and Methods
3. Results and Discussion
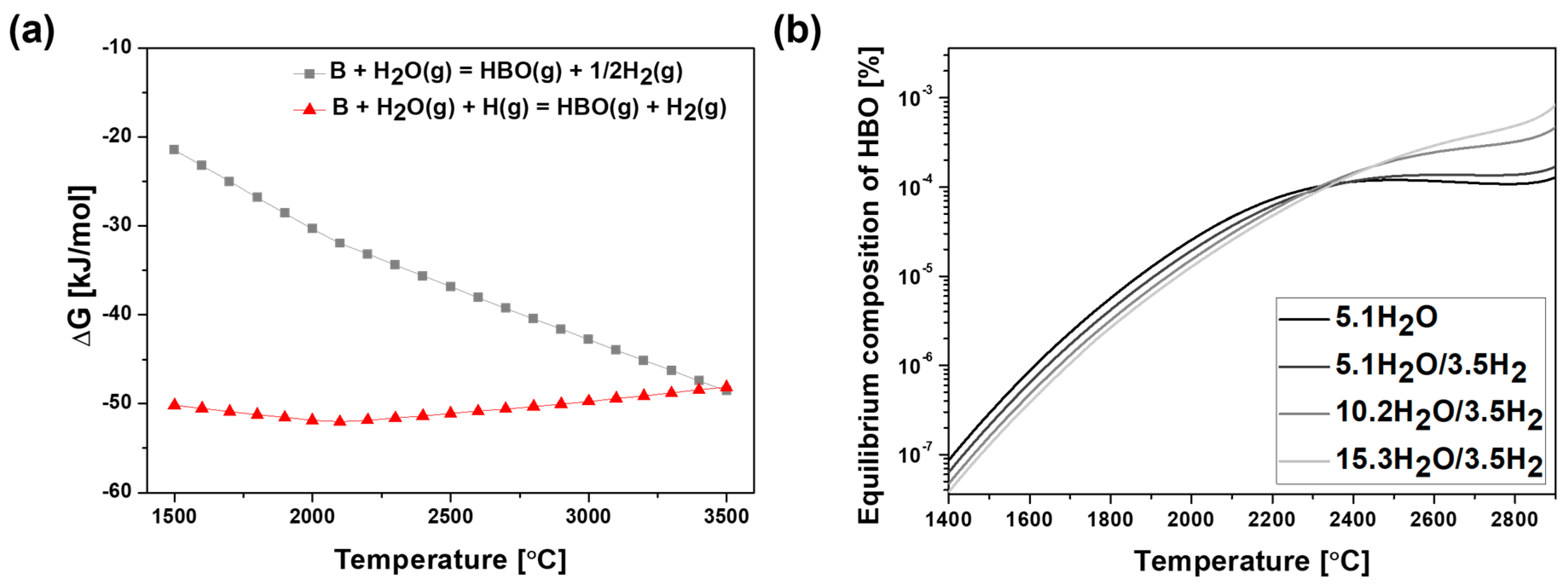
3.1. Thermodynamic Study
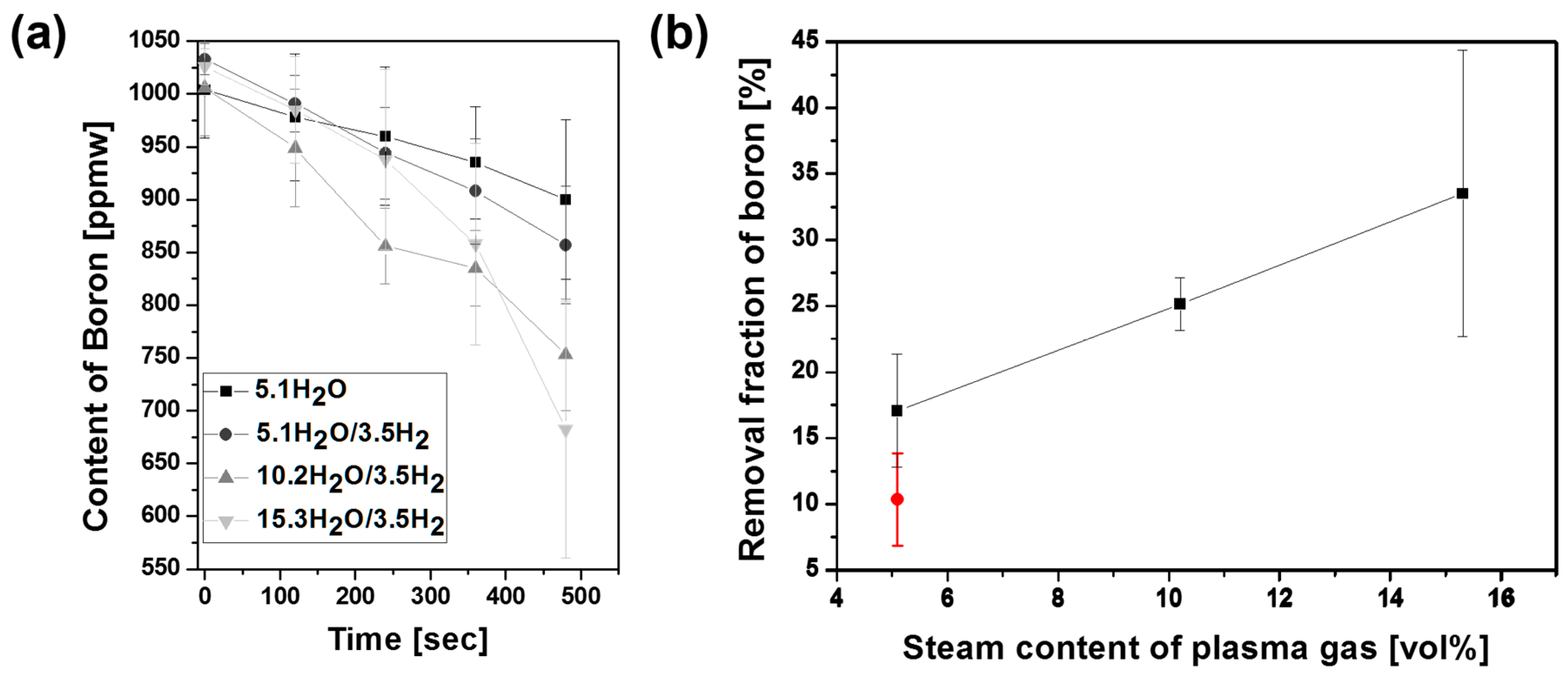
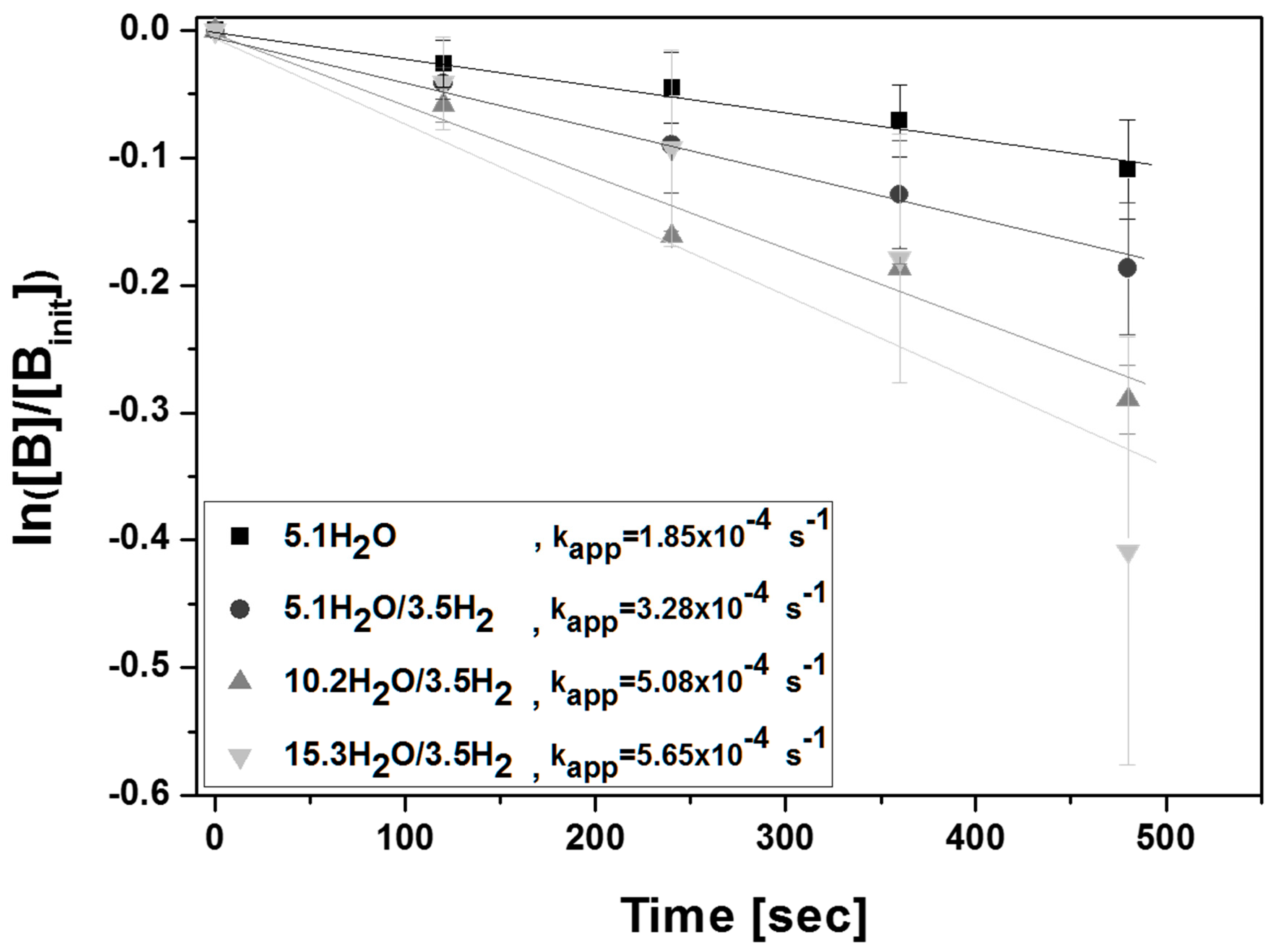
3.2. Experimental Study
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Green, M.A. Silicon photovoltaic modules: A brief history of the first 50 years. Prog. Photovolt. Res. Appl. 2005, 13, 447–455. [Google Scholar] [CrossRef]
- De Wolf, S.; Szlufcik, J.; Delannoy, Y.; Perichaud, I.; Häßler, C.; Einhaus, R. Solar cells from upgraded metallurgical grade (umg) and plasma-purified umg multi-crystalline silicon substrates. Sol. Energy Mater. Sol. Cells 2002, 72, 49–58. [Google Scholar] [CrossRef]
- Saga, T. Advances in crystalline silicon solar cell technology for industrial mass production. NPG Asia Mater. 2010, 2, 96–102. [Google Scholar] [CrossRef]
- Kang, S.; Yoo, S.; Lee, J.; Boo, B.; Ryu, H. Experimental investigations for recycling of silicon and glass from waste photovoltaic modules. Renew. Energy 2012, 47, 152–159. [Google Scholar] [CrossRef]
- Li, J.; Cao, P.; Ni, P.; Li, Y.; Tan, Y. Enhanced boron removal from metallurgical grade silicon by the slag refining method with the addition of tin. Sep. Sci. Technol. 2016, 51, 1598–1603. [Google Scholar] [CrossRef]
- Bathey, B.; Cretella, M. Solar-grade silicon. J. Mater. Sci. 1982, 17, 3077–3096. [Google Scholar] [CrossRef]
- Polverini, D.; Field, M.; Dunlop, E.; Zaaiman, W. Polycrystalline silicon PV modules performance and degradation over 20 years. Prog. Photovolt. Res. Appl. 2013, 21, 1004–1015. [Google Scholar] [CrossRef]
- McDonald, N.C.; Pearce, J.M. Producer responsibility and recycling solar photovoltaic modules. Energy Policy 2010, 38, 7041–7047. [Google Scholar] [CrossRef]
- Müller, A.; Wambach, K.; Alsema, E. Life cycle analysis of solar module recycling process. MRS Online Proc. Libr. Arch. 2005. [Google Scholar] [CrossRef]
- Khajavi, L.T.; Barati, M. Thermodynamics of phosphorus in solvent refining of silicon using ferrosilicon alloys. Metall. Mater. Trans. B 2017, 48, 268–275. [Google Scholar] [CrossRef]
- Yang, H.; Wang, H. The materials characteristic and the efficiency degradation of solar cells from solar grade silicon from a metallurgical process route. J. Mater. Sci. 2011, 46, 1044–1048. [Google Scholar] [CrossRef]
- Fthenakis, V.M. End-of-life management and recycling of PV modules. Energy Policy 2000, 28, 1051–1058. [Google Scholar] [CrossRef]
- Wang, F.; Wu, J.; Ma, W.; Xu, M.; Lei, Y.; Yang, B. Removal of impurities from metallurgical grade silicon by addition of ZnO to calcium silicate slag. Sep. Purif. Technol. 2016, 170, 248–255. [Google Scholar] [CrossRef]
- Khattak, C.P.; Joyce, D.B.; Schmid, F. In Production of Low-cost Solar Grade (SoG) Silicon Feedstock. In Proceedings of the Conference Record of the Twenty-Ninth IEEE, Photovoltaic Specialists Conference, New Orleans, LA, USA, 19–24 May 2002; pp. 364–367. [Google Scholar]
- Mukashev, B.; Tamendarov, M.; Kikkarin, S.; Ustimenko, A. Development of a technology of silicon production by recycling phosphorous industry wastes. Sol. Energy Mater. Sol. Cells 2002, 72, 605–611. [Google Scholar] [CrossRef]
- Khattak, C.P.; Joyce, D.B.; Schmid, F. A simple process to remove boron from metallurgical grade silicon. Sol. Energy Mater. Sol. Cells 2002, 74, 77–89. [Google Scholar] [CrossRef]
- Alemany, C.; Trassy, C.; Pateyron, B.; Li, K.-I.; Delannoy, Y. Refining of metallurgical-grade silicon by inductive plasma. Sol. Energy Mater. Sol. Cells 2002, 72, 41–48. [Google Scholar] [CrossRef]
- Martorano, M.; Neto, J.F.; Oliveira, T.; Tsubaki, T. Refining of metallurgical silicon by directional solidification. Mater. Sci. Eng. B 2011, 176, 217–226. [Google Scholar] [CrossRef]
- Lei, Y.; Ma, W.; Sun, L.; Wu, J.; Dai, Y.; Morita, K. Removal of B from Si by Hf addition during Al–Si solvent refining process. Sci. Technol. Adv. Mater. 2016, 17, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Lee, H.; Park, D.; Shin, J.; Yu, T.; Moon, B. Refining of Mg-Si by hybrid melting using steam plasma and EMC. Sol. Energy Mater. Sol. Cells 2011, 95, 56–58. [Google Scholar] [CrossRef]
- Yoshikawa, T.; Morita, K. Removal of B from Si by solidification refining with Si-Al melts. Metall. Mater. Trans. B 2005, 36, 731–736. [Google Scholar] [CrossRef]
- Khajavi, L.T.; Morita, K.; Yoshikawa, T.; Barati, M. Thermodynamics of boron distribution in solvent refining of silicon using ferrosilicon alloys. J. Alloys Compd. 2015, 619, 634–638. [Google Scholar] [CrossRef]
- Safarian, J.; Tang, K.; Hildal, K.; Tranell, G. Boron removal from silicon by humidified gases. Metall. Mater. Trans. E 2014, 1, 41–47. [Google Scholar] [CrossRef]
- Nordstrand, E.F.; Tangstad, M. Removal of boron from silicon by moist hydrogen gas. Metall. Mater. Trans. B 2012, 43, 814–822. [Google Scholar] [CrossRef]
- Suzuki, K.; Kumagai, T.; Sano, N. Removal of boron from metallurgical-grade silicon by applying the plasma treatment. ISIJ Int. 1992, 32, 630–634. [Google Scholar] [CrossRef]
- Ikeda, T.; Maeda, M. Elimination of boron in molten silicon by reactive rotating plasma arc melting. Mater. Trans. JIM 1996, 37, 983–987. [Google Scholar] [CrossRef]
- Wu, J.; Li, Y.; Ma, W.; Liu, K.; Wei, K.; Xie, K.; Yang, B.; Dai, Y. Impurities removal from metallurgical grade silicon using gas blowing refining techniques. Silicon 2014, 6, 79–85. [Google Scholar] [CrossRef]
- Tan, Y.; Guo, X.; Shi, S.; Dong, W.; Jiang, D. Study on the removal process of phosphorus from silicon by electron beam melting. Vacuum 2013, 93, 65–70. [Google Scholar] [CrossRef]
- Yuge, N.; Abe, M.; Hanazawa, K.; Baba, H.; Nakamura, N.; Kato, Y.; Sakaguchi, Y.; Hiwasa, S.; Aratani, F. Purification of metallurgical-grade silicon up to solar grade. Prog. Photovolt. Res. Appl. 2001, 9, 203–209. [Google Scholar] [CrossRef]
- Morita, K.; Miki, T. Thermodynamics of solar-grade-silicon refining. Intermetallics 2003, 11, 1111–1117. [Google Scholar] [CrossRef]
- Theuerer, H. Removal of boron from silicon by hydrogen water vapor treatment. JOM 1956, 8, 1316–1319. [Google Scholar] [CrossRef]
- Johnston, M.D.; Khajavi, L.T.; Li, M.; Sokhanvaran, S.; Barati, M. High-temperature refining of metallurgical-grade silicon: A review. JOM 2012, 64, 935–945. [Google Scholar] [CrossRef]
| Experimental Conditions | 5.1H2O | 5.1H2O/3.5H2 | 10.2H2O/3.5H2 | 15.3H2O/3.5H2 |
|---|---|---|---|---|
| Ar (vol %) | 94.9 | 91.4 | 86.3 | 81.2 |
| H2O (vol %) | 5.1 | 5.1 | 10.2 | 15.3 |
| H2 (vol %) | 0 | 3.5 | 3.5 | 3.5 |
| Plasma Power (kW) | 10 | 10 | 11 | 11 |
| Molten Si Temperature (°C) | 2090 | 2121 | 2104 | 2113 |
| Reaction Area of Molten Si, A (m2) | 3.85 × 10−5 | |||
| Volume of Molten Si, V (m3) | 3.92 × 10−5 | |||
| V/A (m) | 1.02 | |||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baek, S.-H.; Lee, H.; Min, D.-J.; Choi, S.-J.; Moon, B.-M.; Jung, H.-D. Novel Recycling Method for Boron Removal from Silicon by Thermal Plasma Treatment Coupled with Steam and Hydrogen Gases. Metals 2017, 7, 401. https://doi.org/10.3390/met7100401
Baek S-H, Lee H, Min D-J, Choi S-J, Moon B-M, Jung H-D. Novel Recycling Method for Boron Removal from Silicon by Thermal Plasma Treatment Coupled with Steam and Hydrogen Gases. Metals. 2017; 7(10):401. https://doi.org/10.3390/met7100401
Chicago/Turabian StyleBaek, Su-Hyun, Hyunjae Lee, Dong-Joon Min, Su-Ji Choi, Byung-Moon Moon, and Hyun-Do Jung. 2017. "Novel Recycling Method for Boron Removal from Silicon by Thermal Plasma Treatment Coupled with Steam and Hydrogen Gases" Metals 7, no. 10: 401. https://doi.org/10.3390/met7100401






