The Effects of Incorporated Sn in Resynthesized Ni-Rich Cathode Materials on Their Lithium-Ion Battery Performance
Abstract
:1. Introduction
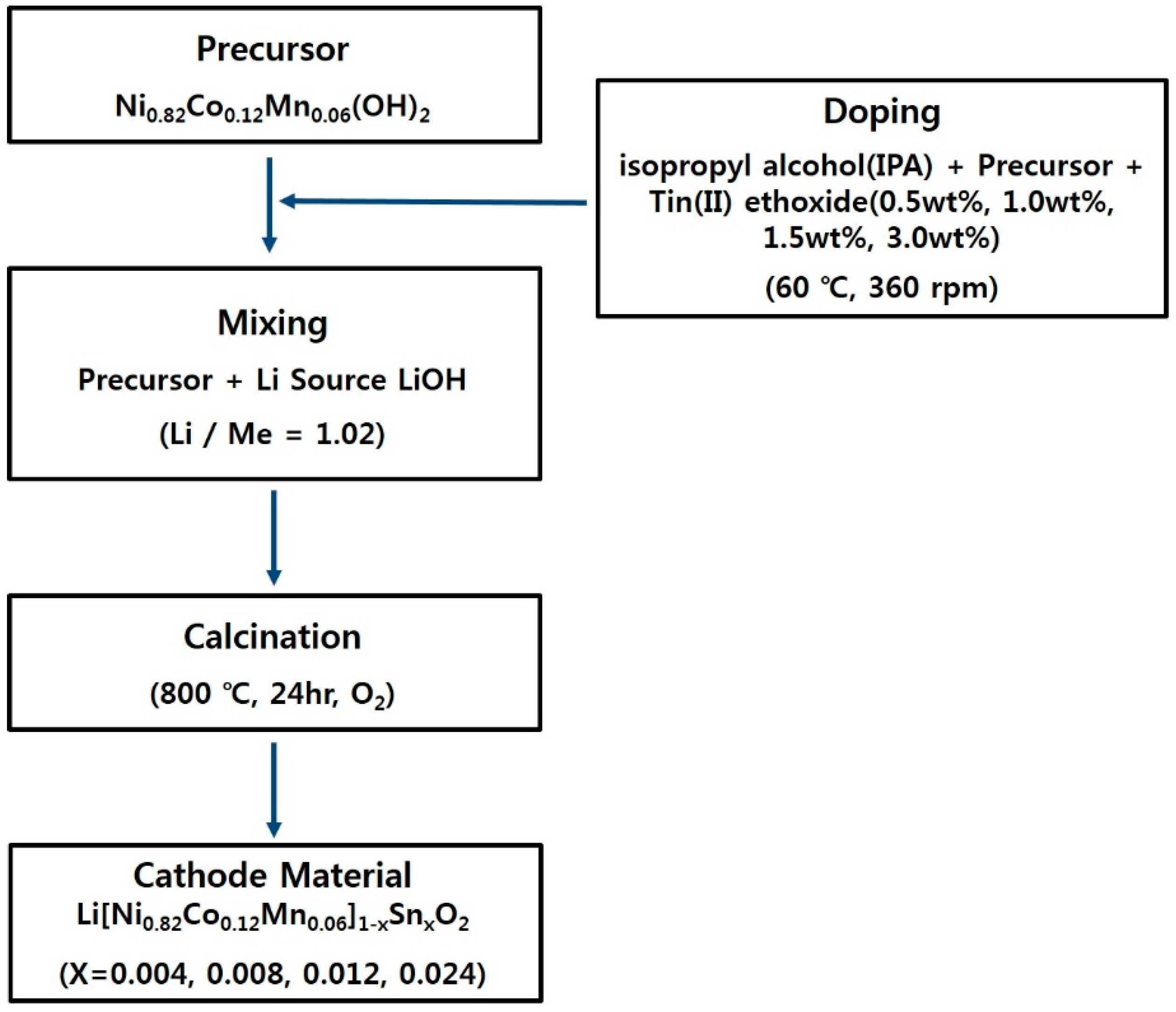
2. Materials and Methods
2.1. Synthesis of Materials
2.2. Physicochemical and Electrochemical Characterizations
3. Results and Discussion
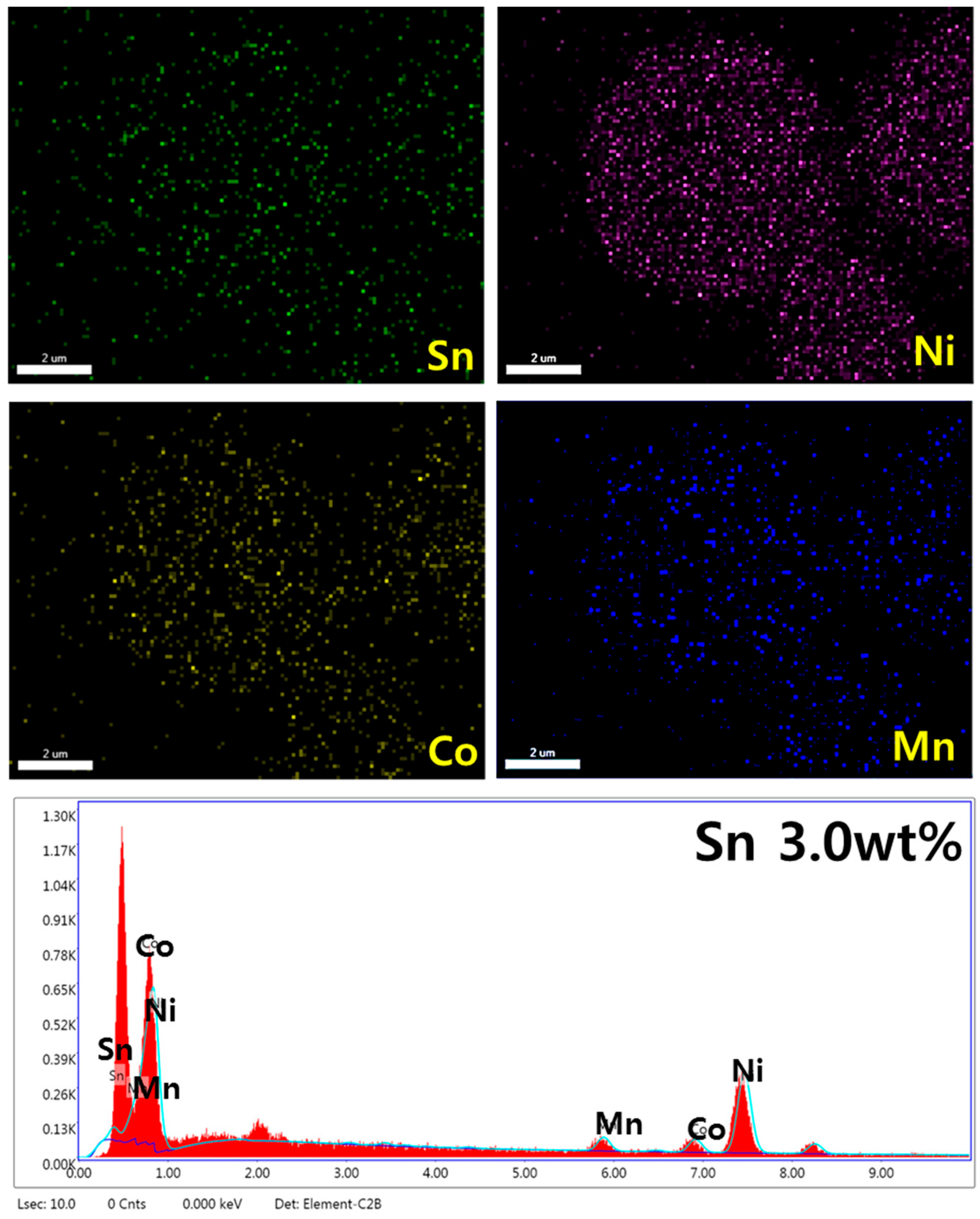
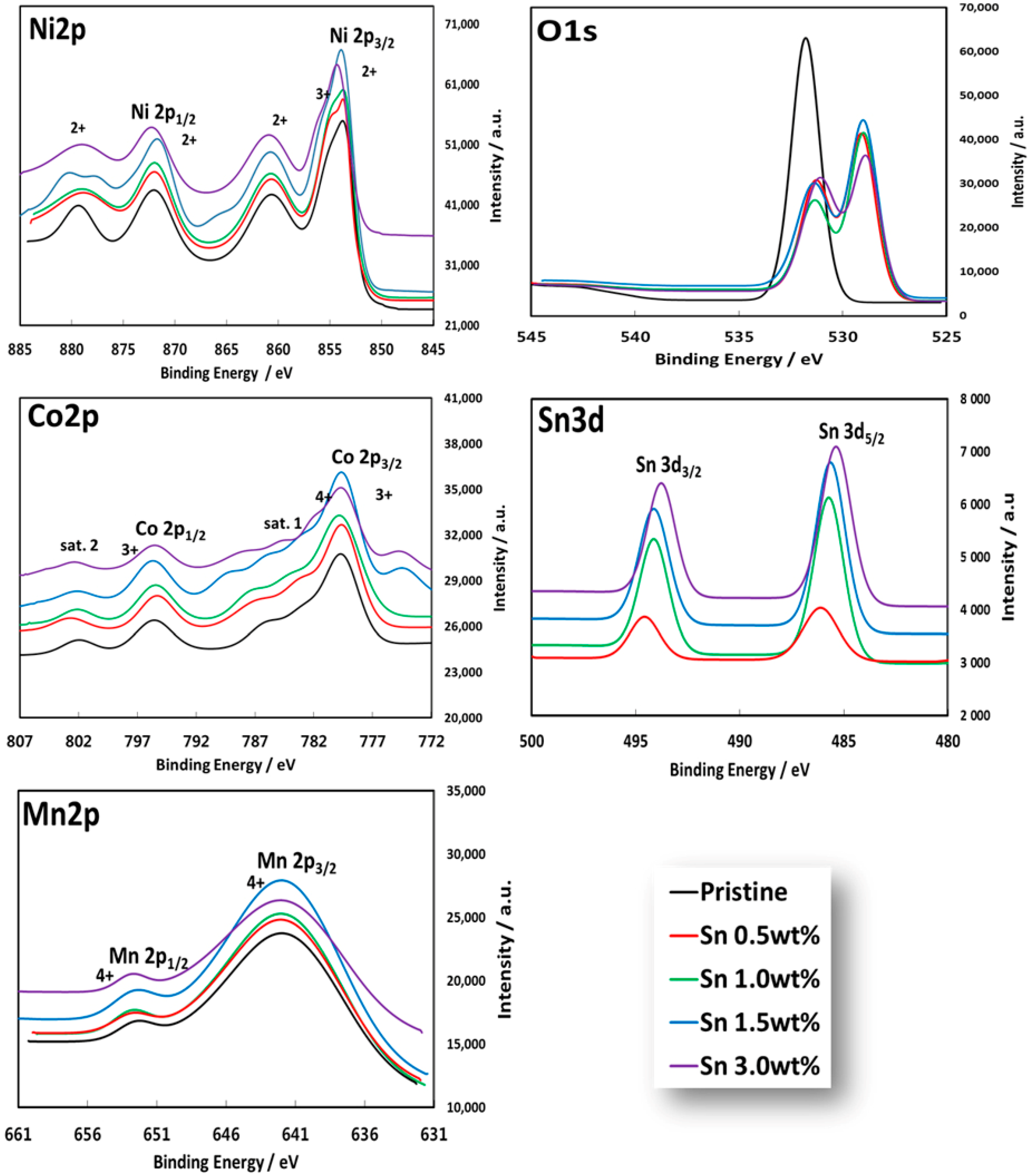
3.1. Physicochemical Characterizations of Sn-Incorporated Ni-Rich Cathode Active Materials
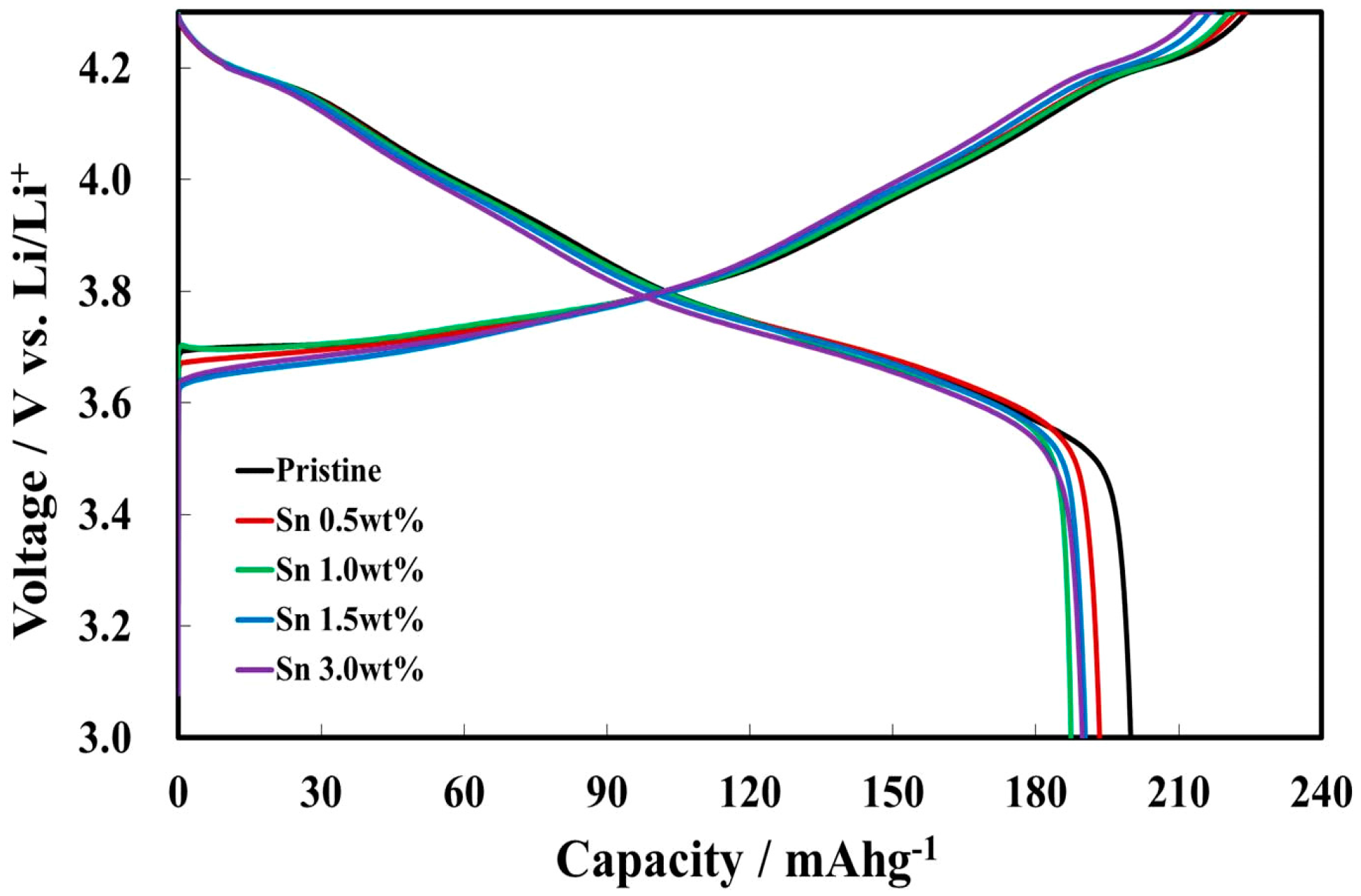
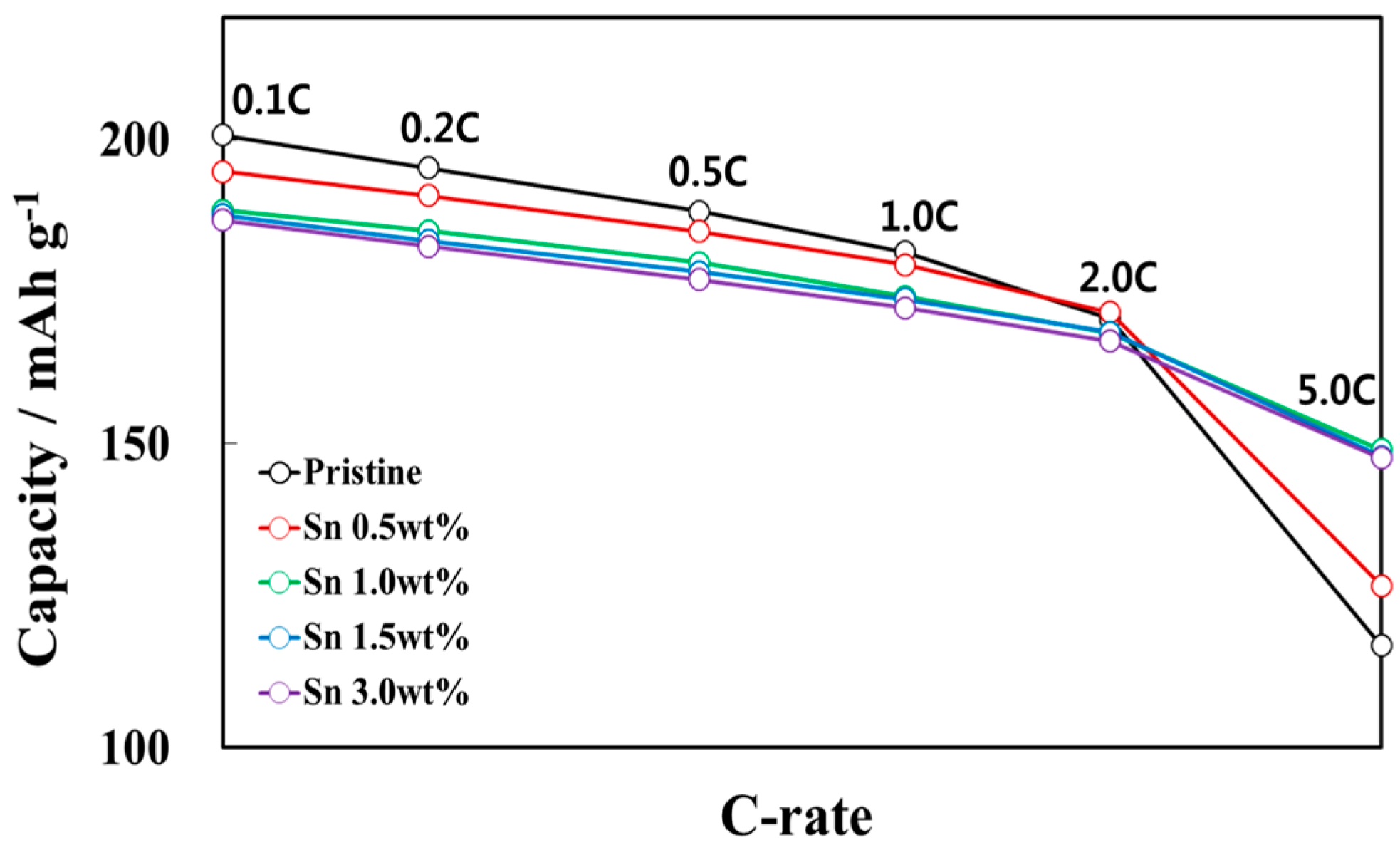
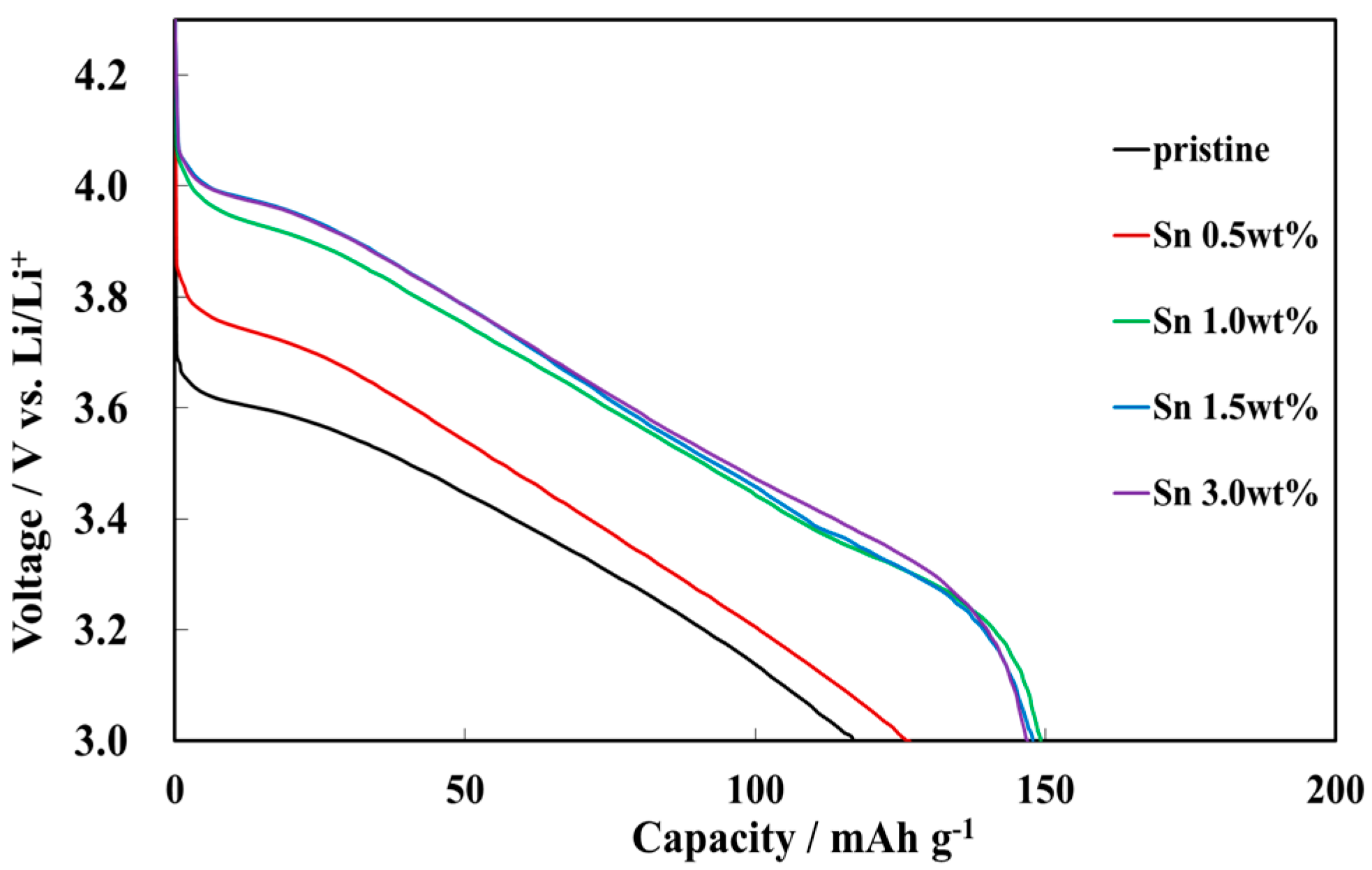
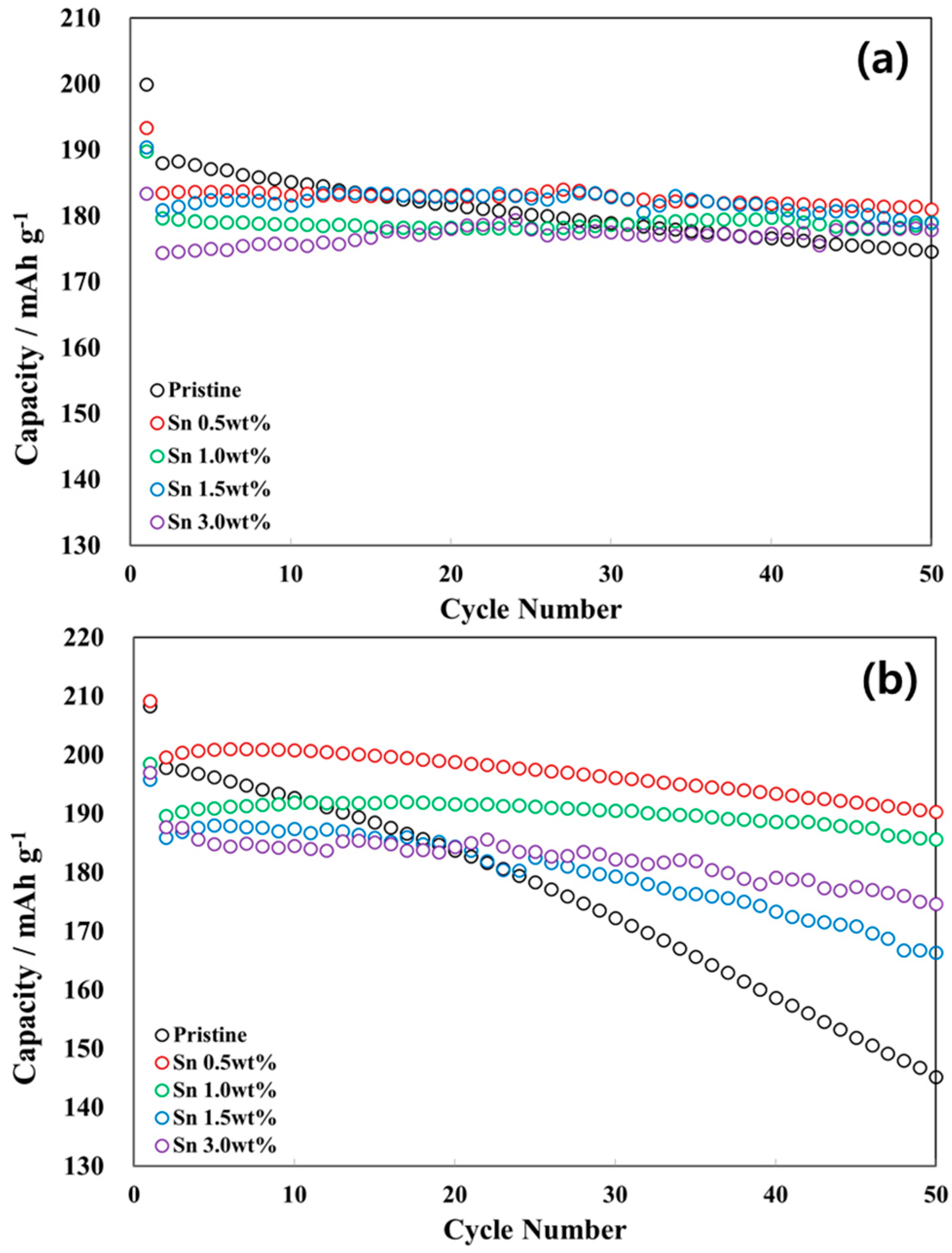
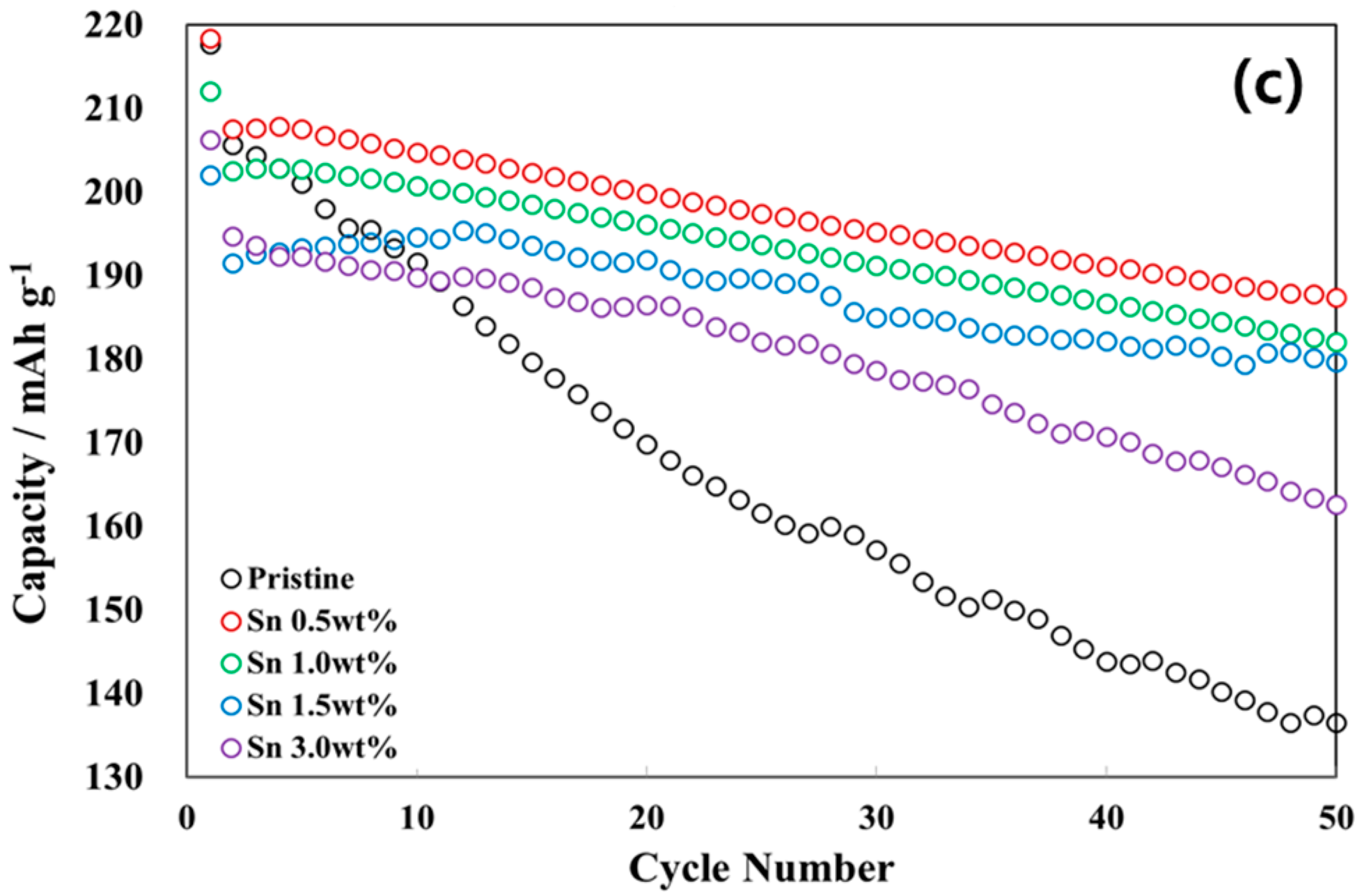
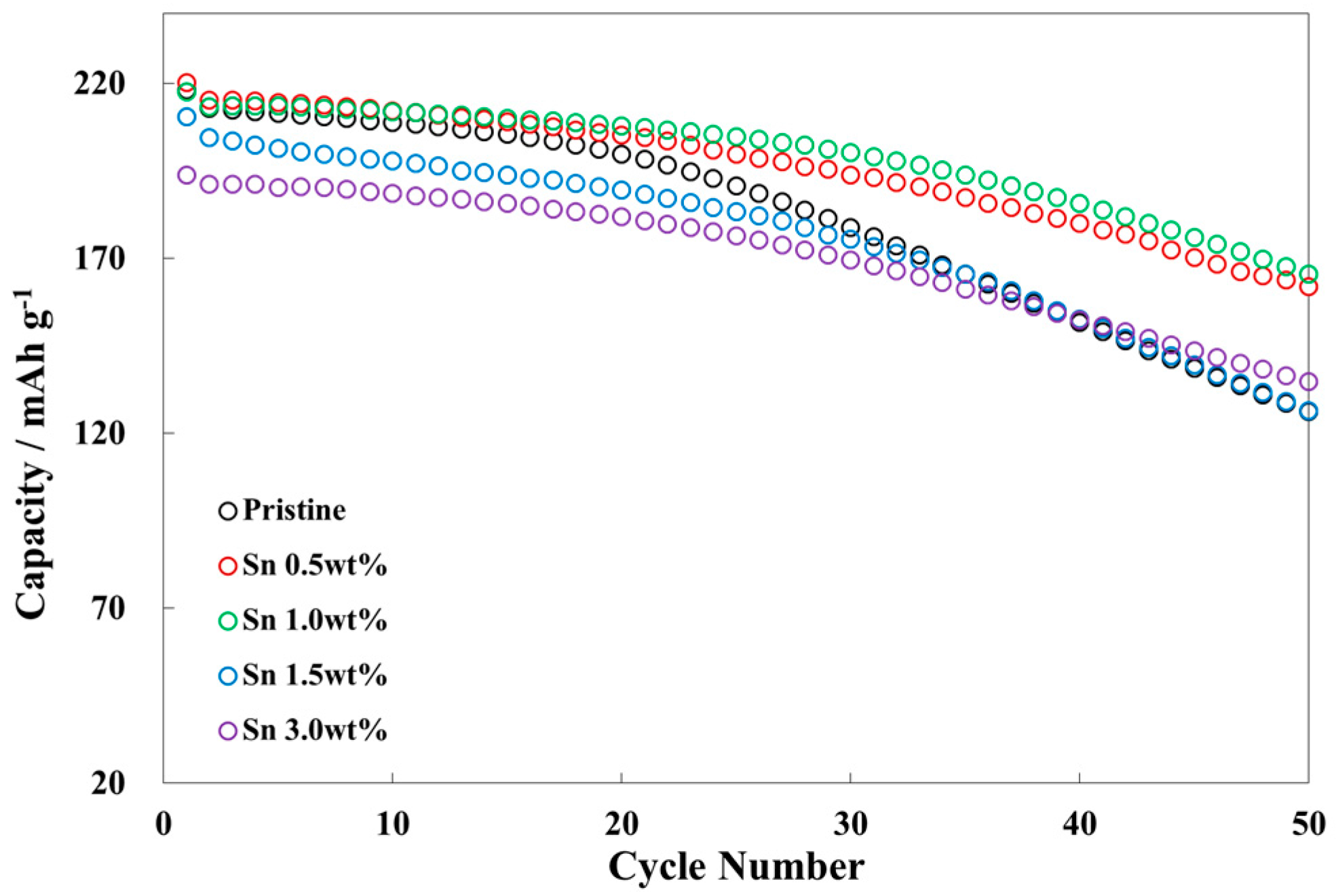
3.2. Electrochemical Characterizations of Sn-Incorporated Ni-Rich Cathode Active Materials
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tarascon, J.M.; Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 2010, 414, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.P.; Zhou, T.; Kong, J.R.; Fang, H.X.; Chen, Y.B. Separation and recovery of metal values from leach liquor of waste lithium nickel cobalt manganese oxide based cathodes. Sep. Purif. Technol. 2015, 141, 76–83. [Google Scholar] [CrossRef]
- Cai, G.Q.; Fung, K.Y.; Ng, K.M.; Wibowo, C. Process development for the recycle of spent lithium ion batteries by chemical precipitation. Ind. Eng. Chem. Res. 2014, 53, 18245–18259. [Google Scholar] [CrossRef]
- Zou, H.Y.; Gratz, E.; Apelian, D.; Wang, Y. A novel method to recycle mixed cathode materials for lithium ion batteries. Green Chem. 2013, 15, 1183–1191. [Google Scholar] [CrossRef]
- Park, C.M.; Kim, J.H.; Kim, H.S.; Sohn, H.J. Li-alloy based anode materials for Li secondary batteries. Chem. Soc. Rev. 2010, 39, 3115–3141. [Google Scholar] [CrossRef] [PubMed]
- Chagnes, A.; Pospiech, B. A brief review on hydrometallurgical technologies for recycling spent lithium-ion batteris. J. Chem. Technol. Biotechnol. 2013, 88, 1191–1199. [Google Scholar] [CrossRef]
- Kang, J.; Senanayake, G.; Sohn, J.; Shin, S.M. Recovery of cobalt sulfate from spent lithium ion batteries by reductive leaching and solvent extraction with Cyanex 272. Hydrometallurgy 2010, 100, 168–171. [Google Scholar] [CrossRef]
- Meshram, P.; Pandey, B.D.; Mabkhand, T.R. Extraction of lithium from primary and secondary sources by pre-treatment, leaching and separation: A comprehensive review. Hydrometallurgy 2014, 150, 192–208. [Google Scholar] [CrossRef]
- Ordones, J.; Gago, E.J.; Girard, A. Processes and technologies for the recycling and recovery of spent lithium-ion batteries. Energy Rev. 2016, 60, 195–205. [Google Scholar]
- Zeng, X.; Li, J.; Singh, N. Recycling of spent lithium-ion battery: A critical review. Environ. Sci. Technol. 2014, 44, 1129–1165. [Google Scholar] [CrossRef]
- Huang, K.; Li, J.; Xu, Z. A novel process for recovering valuable metals from waste nickel-cadmium batteries. Environ. Sci. Technol. 2009, 43, 8974–8978. [Google Scholar] [CrossRef] [PubMed]
- Pietrelli, L.; Bellomo, B.; Fontana, D.; Montereali, M. Characterization and leaching of NiCd and NiMH spent batteries for the recovery of metals. Waste Manag. 2005, 25, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Ku, H.; Jung, Y.; Jo, M.; Park, S.; Kim, S.; Yang, D.; Rhee, K.; An, E.-M.; Sohn, J.; Kwon, K. Recycling of spent lithium-ion battery cathode materials by ammoniacal leaching. J. Hazard. Mater. 2016, 313, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Jha, M.K.; Kumari, A.; Jha, A.K.; Kumar, V.; Hait, J.; Pandey, B.D. Recovery of lithium and cobalt from waste lithium ion batteries of mobile phone. Waste Manag. 2013, 33, 1890–1897. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.K.; Rhee, K.I. Reductive leaching of cathodic active materials from lithium ion battery wastes. Hydrometallurgy 2003, 68, 5–10. [Google Scholar] [CrossRef]
- Nan, J.M.; Han, D.M.; Yang, M.J.; Cui, M.; Hou, X.L. Recovery of metal values from a mixture of spent lithium-ion batteries and nickel-metal hydride batteries. Hydrometallurgy 2006, 84, 75–80. [Google Scholar] [CrossRef]
- Shin, S.M.; Kim, N.H.; Sohn, J.S.; Yang, D.H.; Kim, Y.H. Development of a metal recovery process from Li-ion battery wastes. Hydrometallurgy 2005, 79, 172–181. [Google Scholar] [CrossRef]
- Krüger, S.; Hanisch, C.; Kwade, A.; Winter, M.; Nowak, S. Effect of impurities caused by a recycling process on the electrochemical performance of Li[Ni0.33Co0.33Mn0.33]O2. J. Electroanal. Chem. 2014, 726, 91–96. [Google Scholar]
- Weng, Y.; Xu, S.; Huang, G.; Jiang, C. Synthesis and performance of Li[(Ni1/3Co1/3Mn1/3)1−xMgx]O2 prepared from spent lithium ion batteries. J. Hazard. Mater. 2013, 246, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.M.; Hwang, T.; Kim, D.; Park, M.S.; Cho, K.; Cho, M. Intrinsic origins of crack generation in Ni-rich LiNi0.8Co0.1Mn0.1O2 layered oxide cathode material. Sci. Rep. 2017, 8, 39669. [Google Scholar] [CrossRef] [PubMed]
- Shu, J.; Ma, R.; Shao, L.Y.; Shui, M.; Wu, K.G.; Lao, M.M.; Wang, D.J.; Long, N.B.; Ren, Y.L. In-situ X-ray diffraction study on the structural evolutions of LiNi0.5Co0.3Mn0.2O2 in different working potential windows. J. Power Sources 2014, 245, 7–18. [Google Scholar] [CrossRef]
- Chen, Z.; Wang, J.; Chao, D.L.; Baikie, T.; Bai, L.Y.; Chen, S.; Zhao, Y.L.; Sum, T.C.; Lin, J.Y.; Shen, Z.X. Hierarchical porous LiNi1/3Co1/3Mn1/3O2 nano-/micro spherical cathode material: Minimized cation mixing and improved Li+ mobility for enhanced electrochemical performance. Sci. Rep. 2016, 6, 25771. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.L.; Wang, C.W.; Cheng, J.G.; Sun, J.T. Effects of Sn doping on the structural and electrochemical properties of LiNi0.8Co0.2O2 cathode materials. Solid State Ion. 2007, 178, 125–129. [Google Scholar] [CrossRef]
- Ren, H.B.; Mu, X.M.; Huang, Y.H.; Li, Z.J.; Wang, Y.H.; Cai, P.; Peng, Z.H.; Zhou, Y.H. Effects of Sn doping on electrochemical characterizations of Li[Ni1/3Co1/3Mn1/3]O2 cathode material. Ionics 2010, 16, 497–502. [Google Scholar] [CrossRef]
- Zhao, Y.J.; Xia, M.H.; Hu, X.S.; Zhao, Z.K.; Wang, Y.; Lv, Z. Effects of Sn doping on the structural and electrochemical properties of Li1.2Ni0.2Mn0.8O2 Li-rich cathode materials. Electrochim. Acta 2015, 174, 1167–1174. [Google Scholar] [CrossRef]
- Jonsen, P.; Haggblad, H.A.; Sommer, K. Tensile strength and fracture energy of pressed metal powder by diametral compression test. Powder Technol. 2007, 176, 148–155. [Google Scholar] [CrossRef]
- Araki, K.; Taguchi, N.; Sakaebe, H.; Tatsumi, K.; Ogumi, Z. Electrochemical properties of Li Ni1/3Co1/3Mn1/3O2 cathode material modified by coating with Al2O3 nanoparticles. J. Power Sources 2014, 269, 236–243. [Google Scholar] [CrossRef]
- Zhang, X.H.; Yu, C.; Huang, X.D.; Zheng, J.; Guan, X.F.; Luo, D.; Li, L.P. Novel composites Li[LixNi0.34−xMn0.47Co0.19]O2 (0.18 ≤ x ≤ 0.21): Synthesis and application as high-voltage cathode with improved electrochemical performance for lithium ion batteries. J. Mater. Chem. 2012, 81, 233–238. [Google Scholar] [CrossRef]
- Darma, M.S.D.; Lang, M.; Kleiner, K.; Mereacre, L.; Liebau, V.; Fauth, F.; Bergfeldt, T.; Ehrenberg, H. The influence of cycling temperature and cycling rate on the phase specific degradation of a positive electrode in lithium ion batteries: A post mortem analysis. J. Power Sources 2016, 327, 714–725. [Google Scholar] [CrossRef]

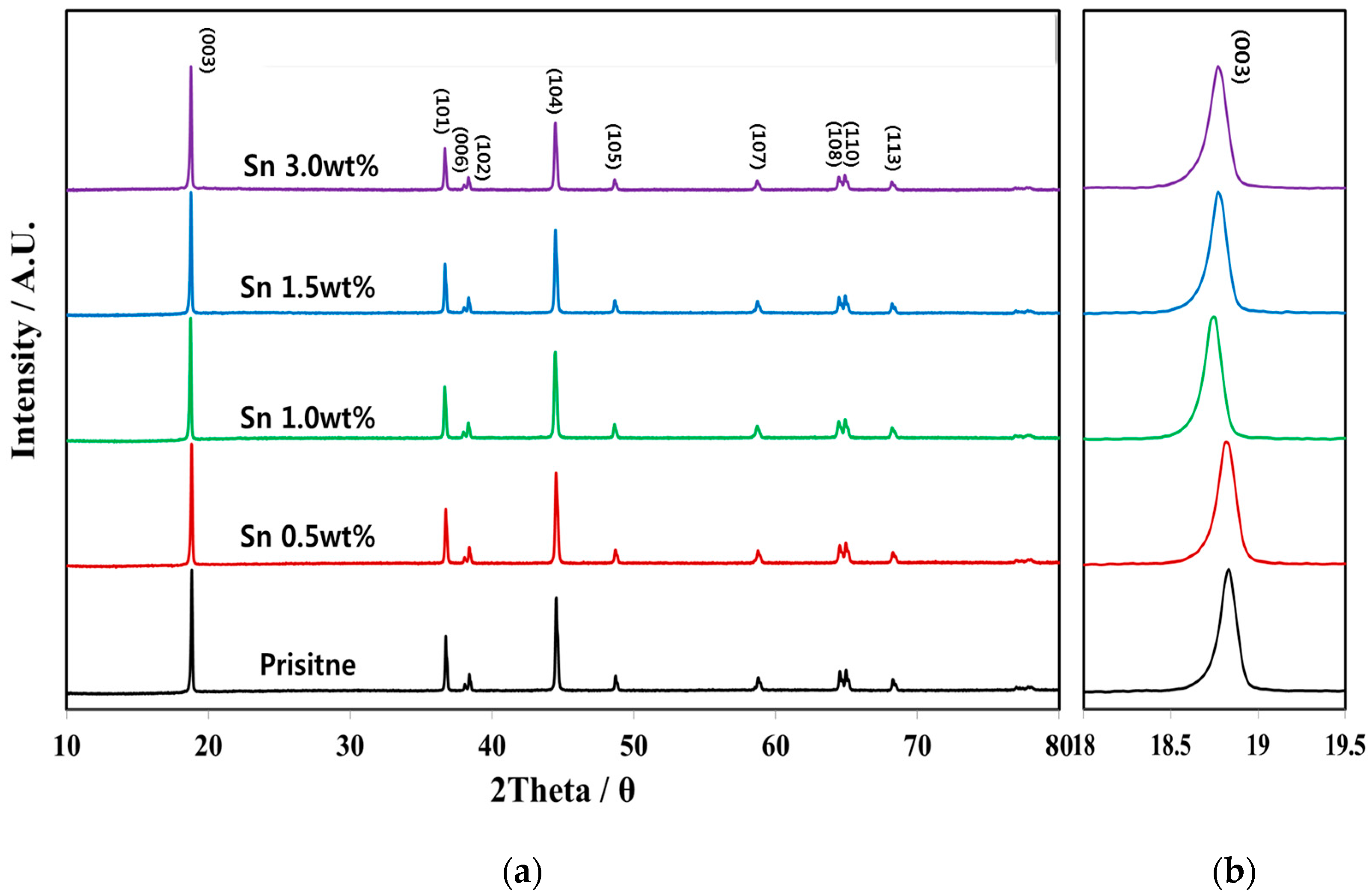
| Lattice Parameter | Pristine | Sn 0.5 wt % | Sn 1.0 wt % | Sn 1.5 wt % | Sn 3.0 wt % |
|---|---|---|---|---|---|
| a [Å] | 2.86769 | 2.86794 | 2.87031 | 2.87007 | 2.86819 |
| c [Å] | 14.13716 | 14.14405 | 14.19612 | 14.17667 | 14.14698 |
| Sample | Force P (mN) | Diameter D (µm) | σt (MPa) |
|---|---|---|---|
| Pristine | 8.29 | 9.39 | 83.86 |
| Sn 0.5 wt % | 12.12 | 9.87 | 110.98 |
| Sn 1.0 wt % | 12.34 | 8.91 | 138.49 |
| Sn 1.5 wt % | 13.75 | 11.47 | 93.16 |
| Sn 3.0 wt % | 10.52 | 10.24 | 89.43 |
| Sample | Charge (mAh g−1) | Discharge (mAh g−1) |
|---|---|---|
| Pristine | 225.98 | 199.92 |
| Sn 0.5 wt % | 224.03 | 193.38 |
| Sn 1.0 wt % | 221.39 | 187.39 |
| Sn 1.5 wt % | 217.43 | 190.38 |
| Sn 3.0 wt % | 216.80 | 189.74 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, G.-h.; Lee, K.-w.; Kwon, K.; Song, J. The Effects of Incorporated Sn in Resynthesized Ni-Rich Cathode Materials on Their Lithium-Ion Battery Performance. Metals 2017, 7, 395. https://doi.org/10.3390/met7100395
Kang G-h, Lee K-w, Kwon K, Song J. The Effects of Incorporated Sn in Resynthesized Ni-Rich Cathode Materials on Their Lithium-Ion Battery Performance. Metals. 2017; 7(10):395. https://doi.org/10.3390/met7100395
Chicago/Turabian StyleKang, Ga-hee, Ko-woon Lee, Kyungjung Kwon, and Junho Song. 2017. "The Effects of Incorporated Sn in Resynthesized Ni-Rich Cathode Materials on Their Lithium-Ion Battery Performance" Metals 7, no. 10: 395. https://doi.org/10.3390/met7100395





