1. Introduction
Neodymium-iron-boron (NdFeB) magnets were developed in the early 1980s due to the important discovery that modifying Nd
2Fe
17 produces the ternary compound Nd
2Fe
14B [
1,
2]. The NdFeB magnets could substitute samarium-cobalt magnets because neodymium is more abundant than samarium and the use of iron as transition metal is lower-cost than that of cobalt [
1]. The NdFeB magnets have been widely used in various applications such as motors for hard disk and hybrid electric vehicle and magnetic generators for magnetic resonance imaging (MRI) [
3,
4,
5]. Since most Nd is imported into Korea, there is an increasing demand for recycling process of waste NdFeB magnets.
It has been well known that Nd could be obtained from NdFeB magnet scraps by sulfuric acid leaching followed by precipitation of Nd salts [
6,
7,
8]. Because this method dissolves iron as well as neodymium, there are disadvantages such as high consumption of sulfuric acid and large amount of wastewater discharged. Many methods have been reported to extract selectively Nd from waste NdFeB magnets such as roasting followed by sulfuric acid leaching [
9], selective chlorination of Nd using NH
4Cl [
4], selective extraction of Nd using molten Mg [
5,
10] and slag materials [
6], and leaching with hydrochloric acid and oxalic acid [
11]. These methods require strong acid or should be performed at high temperature over 700 °C. In this study, it was important to change the solid phase of NdFeB magnets for selective recovery of Nd.
High-energy ball milling processes result in repeatedly fracturing and cold welding during collision between balls or ball and inner wall of mill container, so mechanical solid phase reaction could occur during milling [
12]. Kim
et al. reported that they improved the yield of La, Nd and Sm in monazite to around 85% by 120 min-milling with NaOH [
12]. Greenberg submitted an application to the US patent regarding Nd recovery where NdFeB magnets are ground mechanically, then leached with acetate, and finally Nd was precipitated selectively as NdF
3 [
13]. However, the experimental conditions and results were not reported in detail.
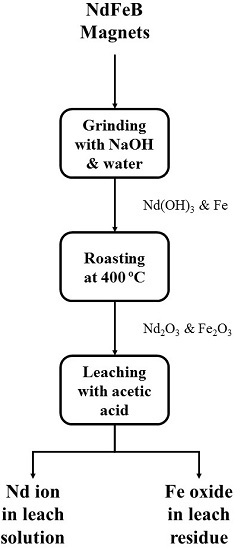
This study is aimed at developing the pretreatment process for selective recovery process of Nd and Dy from waste NdFeB magnets. The magnet scraps were ground with NaOH and water, and then roasted under atmosphere conditions. The effects of various conditions such as milling time, milling interval, amount of water and NaOH added, and roasting temperature were investigated. Finally, acetate leaching tests were performed to investigate the selective leaching of Nd.
3. Results and Discussion
In this study, the pretreatment process consists of grinding and roasting for selective leaching of Nd and Dy from the magnet leaving Fe component as leach residue. The Fe component should be oxidized to ferric ion (Fe
3+), since the solubility products (K
sp) of Fe(OH)
3 and Fe(OH)
2 are 10
−36.51 and 10
−14.18, respectively, using standard Gibbs free energy data in
Table 1 [
14], which means that the solubility of ferric ion (Fe
3+) is much lower than that of ferrous ion (Fe
2+). The roasting tests were performed to oxidize the Fe, but, since Kim
et al. reported that the magnet scraps were not oxidized sufficiently at less than 500 °C [
15], the grinding tests were conducted to increase the specific surface area of magnet powder and change the solid phase before roasting.
Table 1.
Standard Gibbs free energies of species at 25 °C [14].
| Species | Fe2+ | Fe3+ | Fe(OH)2 | Fe(OH)3 | OH− |
|---|
| ΔG0 (kJ/mol) | −91.2 | −16.7 | −486.6 | −696.6 | −157.293 |
The NdFeB magnet scraps were ground with 0 to 5 cm
3 under the following conditions: 15 in stoichiometric molar ratio of NaOH:Nd, 550 rpm in rotational speed, 30 min in grinding time.
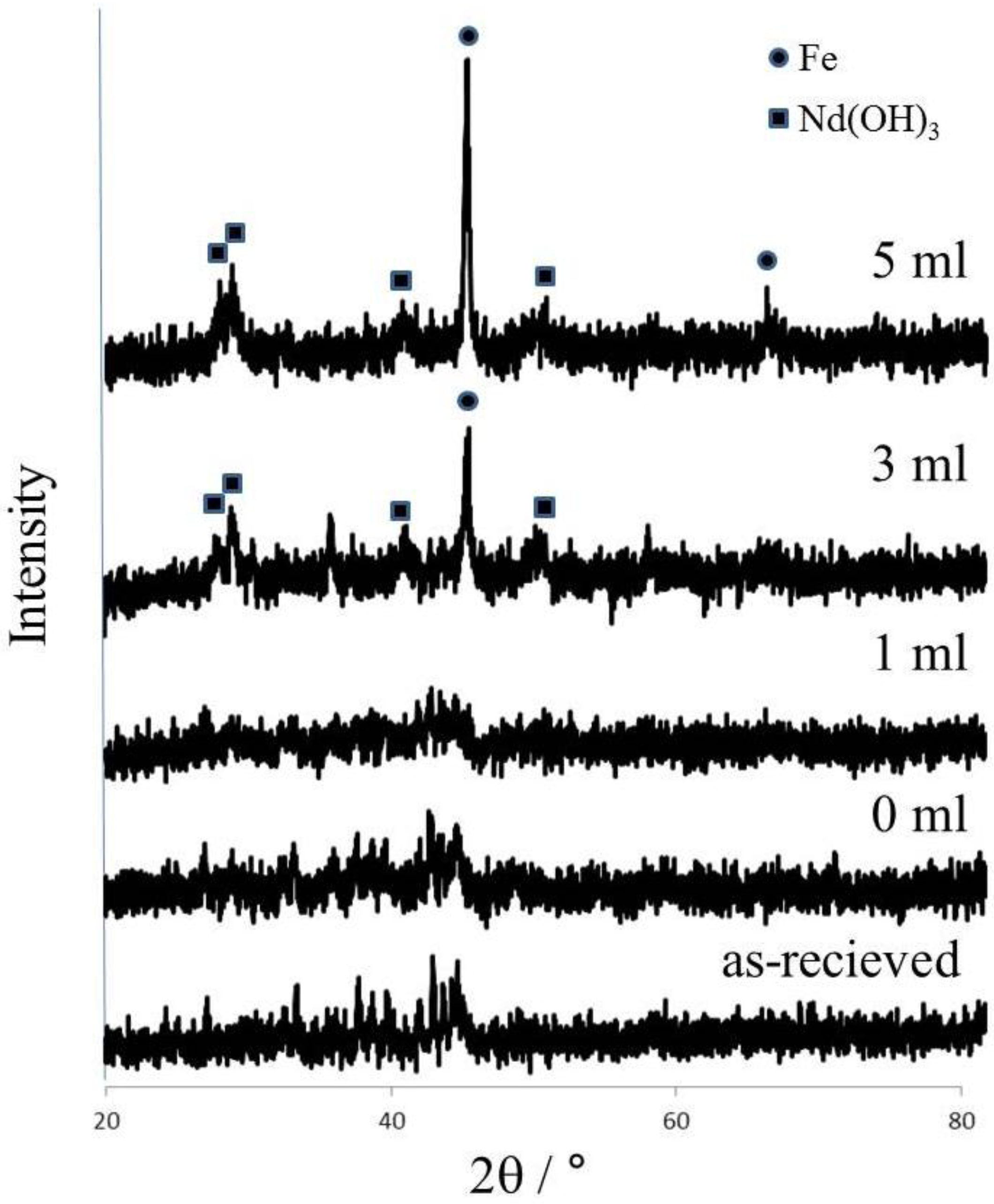
Figure 1 shows the effect of water addition on the formation of Nd(OH)
3 during grinding. When samples were ground without water, XRD peaks show little difference from the as-received sample, while the peaks of Fe and Nd(OH)
3 arose as the amount of water addition increased. It has been found that Nd could be oxidized with H
2O as the following equation.
The standard reduction potentials of Nd (Nd
3+ + 3e = Nd) and water (2H
2O + 2e = H
2 + 2OH
−) are −2.32 V [
16] and −0.83 V [
17], respectively, and because the standard electrode potential of Equation (1) is calculated to be 1.49 V, the reaction must be spontaneous. Therefore, 5 cm
3-water addition could accelerate the formation of Nd(OH)
3 as shown in
Figure 1. However, iron could not be oxidized by water addition, so iron peaks were observed by adding 3 cm
3 or 5 cm
3 water. These results indicate that Nd and Fe of Nd
2Fe
14B was changed successfully into Nd(OH)
3 and Fe, respectively.
Figure 1.
XRD patterns of ground products with water addition (Grinding conditions: 15 in stoichiometric molar ratio, 550 rpm in rotational speed, 30 min in grinding time).
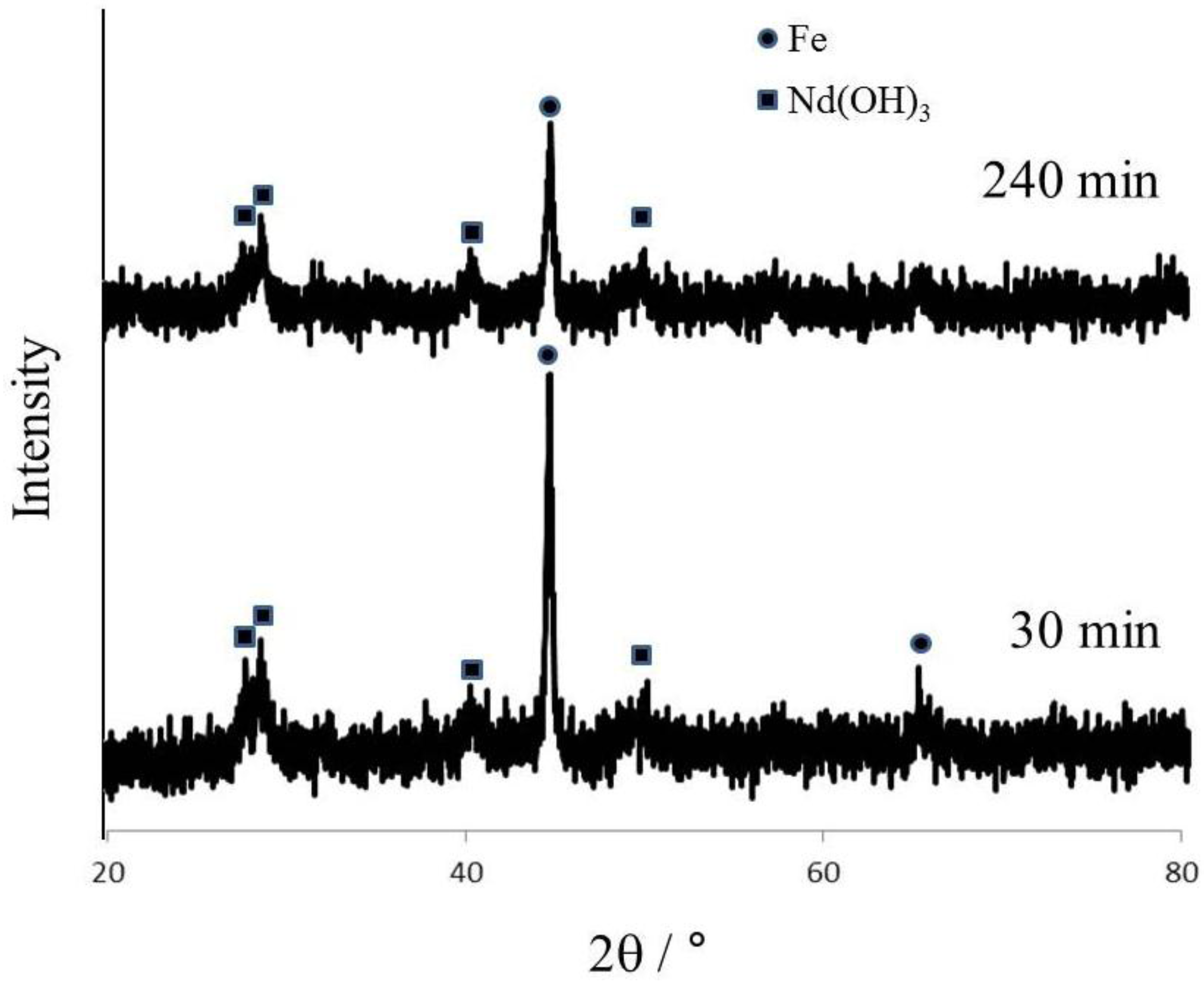
Greenburg proposed the recycling process of NdFeB magnet scraps [
13], where Nd was selectively leached after grinding with NaOH.
Figure 2 shows XRD patterns of products ground for 30 min and 240 min. However, the intensity of the Fe peak decreased with increasing the grinding time, since Fe remained after 240 min grinding and Fe could be dissolved by acetic acid leaching; therefore, it is difficult to expect selective leaching of Nd. Furthermore, in the preliminary experiments with 30 in stoichiometric molar ratio, the peaks of Fe were observed (data not shown).
Figure 2.
XRD patterns of ground products with grinding time (Grinding conditions: 15 in stoichiometric molar ratio, 550 rpm in rotational speed, 5 cm3 in water addition).
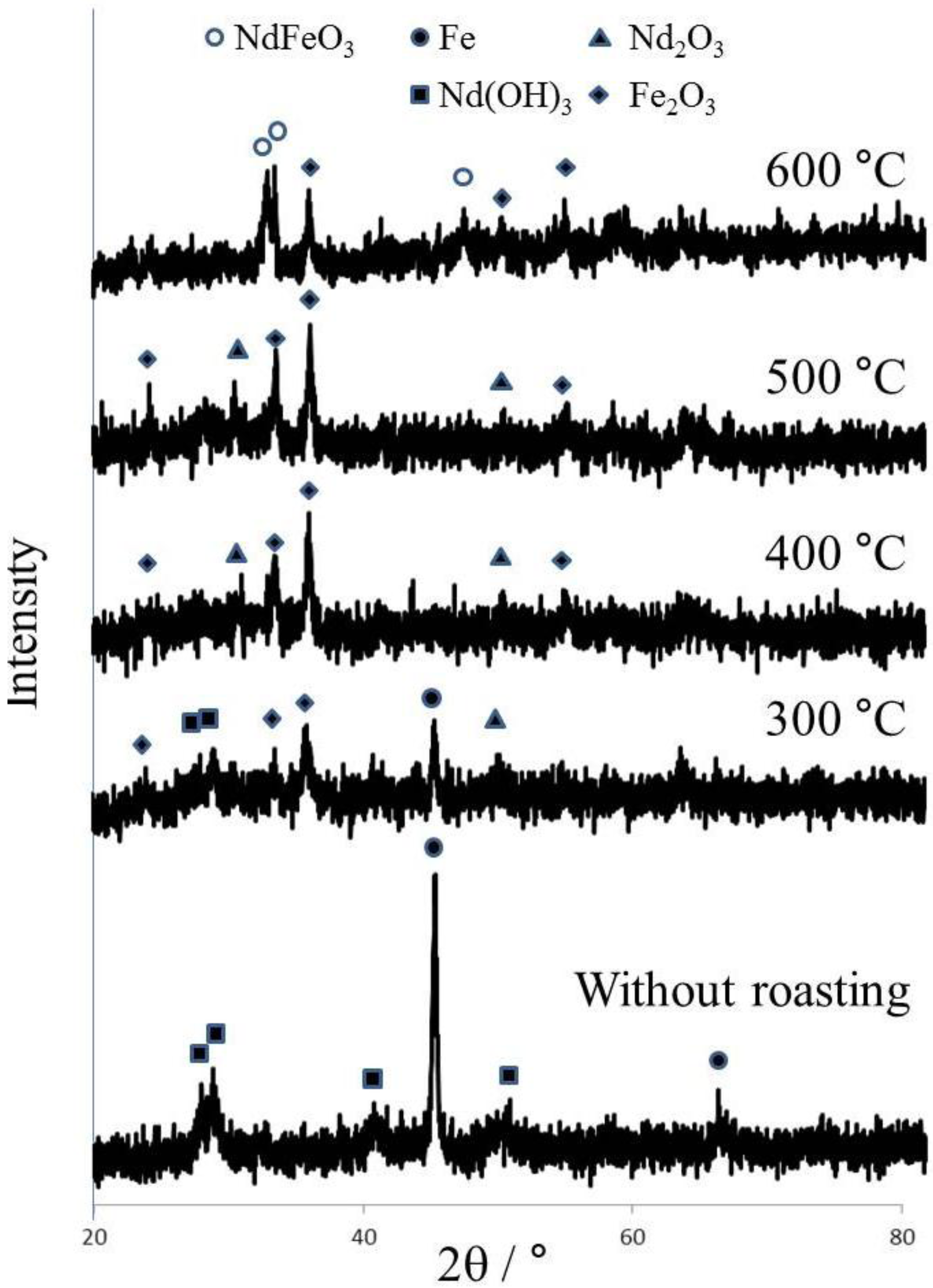
Therefore, in this study, the magnet scraps were roasted after grinding to oxidize Fe for improving selective leaching efficiency. The ground products were prepared under the conditions such as 15 in stoichiometric molar ratio, 550 rpm in rotational speed, 30 min in total grinding time, and 5 cm
3 in water addition, and then was roasted at 300 °C to 600 °C for 2 h. As shown in
Figure 3, the peaks of Fe
2O
3 and Nd
2O
3 as well as Nd(OH)
3 and Fe were shown after roasting at 300 °C. After roasting at 400 °C and 500 °C, the peaks of Nd(OH)
3 and Fe disappeared, and only peaks of Fe
2O
3 and Nd
2O
3 were observed. The peaks of NdFeO
3 emerged after roasting at 600 °C, and the reaction was given as the following equation [
9].
These results indicate that iron could be oxidized at 400 °C and 500 °C, and the components of Nd and Fe were changed into NdFeO3 at 600 °C.
The selective leaching with acetic acid was tested using the roasted product.
Figure 4 shows the leaching efficiencies of Nd, Fe, and Dy using product ground under the following conditions: 15 in stoichiometric molar ratio, 550 rpm in rotational speed, 5 cm
3 in water addition, 30 min in grinding time and roasted at 400 °C. The leaching efficiencies of Nd and Dy increased rapidly to more than 90% and remained almost constant after 60 min, and that of Fe remained at less than 1.5%, which indicates that Nd and Dy were selectively leached from scraps. Therefore, the leaching efficiencies measured at 60 min were used for further discussion.
Figure 3.
XRD patterns of roasted sample with temperature (Grinding conditions: 15 in stoichiometric molar ratio, 550 rpm in rotational speed, 5 cm3 in water addition, 30 min in grinding time).
Figure 4.
Leaching efficiencies of Nd, Fe, Dy with leaching time (Grinding conditions: 15 in stoichiometric molar ratio, 550 rpm in rotational speed, 5 cm3 in water addition, 30 min in grinding time, roasting condition; 2 h at 400 °C, leaching conditions: 400 rpm, 1 kmol·m−3 acetic acid, 90 °C).
Figure 5 shows the effect of roasting temperature on the leaching efficiencies of Nd, Fe and Dy. The samples were prepared under the grinding conditions: 550 rpm in rotational speed, 5 cm
3 in water addition, 30 min in grinding time and 15 in stoichiometric ratio, and then were leached under the conditions: 1% in pulp density, 1 kmol·m
−3 in acetic acid, 400 rpm in agitation speed, and 90 °C in temperature. The leaching efficiencies of Nd and Dy decreased rapidly with increasing temperature from 500 °C to 600 °C, and that of Fe decreased and then increased gradually with increasing roasting temperature. The decrease in leaching efficiencies of Nd and Dy would be formation of NdFeO
3 shown in Equation (2).
Figure 5.
The effect of roasting temperature on the leaching efficiency (Grinding conditions: 550 rpm in rotational speed, 5 cm3 in water addition, 30 min in grinding time, 15 in stoichiometric molar ratio).
Figure 6.
The comparison of leaching efficiencies using samples as as-received, only roasted, and roasted after being ground.
Figure 6 shows the comparison of leaching efficiencies using samples such as “as-received”, “roasting only”, which was roasted at 400 °C for 2 h using “as-received” sample, and “roasting after grinding”, which was prepared under the grinding conditions: 15 in stoichiometric molar ratio, 550 rpm in rotational speed, 5 cm
3 in water addition, 30 min in grinding time and roasting conditions: 400 °C in temperature and 2 h in roasting time. The leaching efficiency of Fe is calculated to be more than 40% in the cases of “as-received” and “only roasting” samples, and the results indicate that Nd and Dy were not leached selectively from the scraps. The dissolution of Fe would result from insufficient oxidation of Fe as reported by Kim
et al. [
15] However, 30-min grinding with NaOH changes the components of Nd and Fe into Nd(OH)
3 and Fe, respectively, which could oxidize Fe easily during roasting. Finally, more than 90% of Nd and Dy could be leached but the leaching efficiency of Fe was 1% at 60 min leaching time, which indicates that selective leaching of Nd and Dy could be achieved by roasting after grinding but not by only roasting or by only grinding. The leaching efficiencies of Nd and Dy increased to 94.2% and 93.1%, respectively, with increasing time to 180 min. These results indicate that the selective leaching of Nd and Dy was achieved successfully.