The Effect of Indium Content on the Atomic Environment and Cluster Stability of GeSe4Inx=10,15 Glasses
Abstract
:1. Introduction
2. Methodology
| Partial | GeSe4In10 | GeSe4In15 | ||
|---|---|---|---|---|
| rmin | rmax | rmin | rmax | |
| Ge–Se | 2.00 | 2.95 | 1.95 | 2.95 |
| Se–Se | 1.95 | 2.75 | 2.05 | 2.75 |
| Se–In | 2.30 | 2.90 | 2.30 | 2.90 |
3. Results and Discussion
3.1. Pair Distribution Functions
| Total | Ge-Ge | Se-Se | In-In | Ge-In | Se-In | |
|---|---|---|---|---|---|---|
| GeSe4In10 | 0.31 | 0.16 | 0.41 | 0.23 | 0.16 | 0.46 |
| GeSe4In15 | 0.26 | 0.21 | 0.36 | 0.18 | 0.14 | 0.40 |
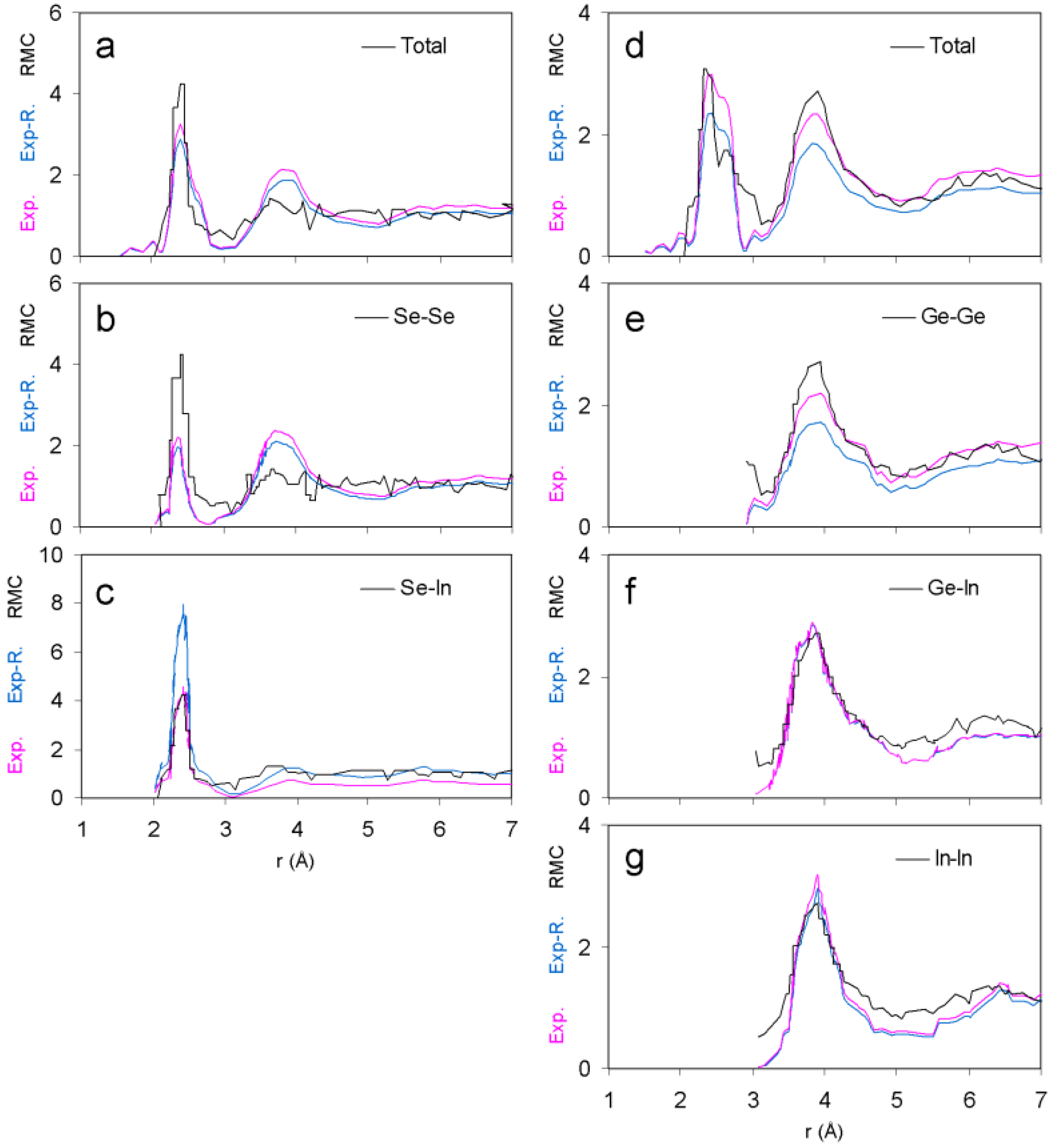
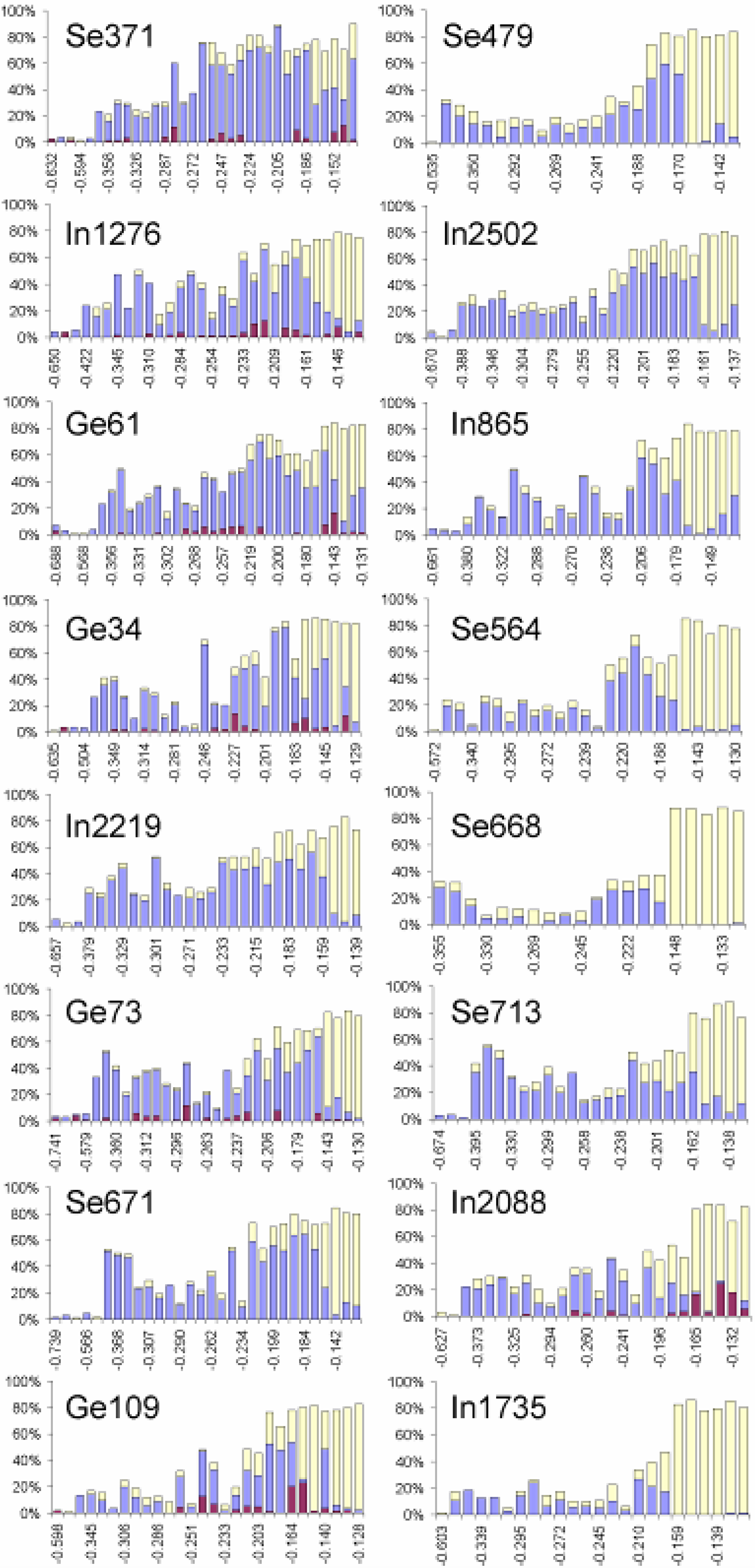
3.2. Atomic Environment Statistics and Cluster Selection
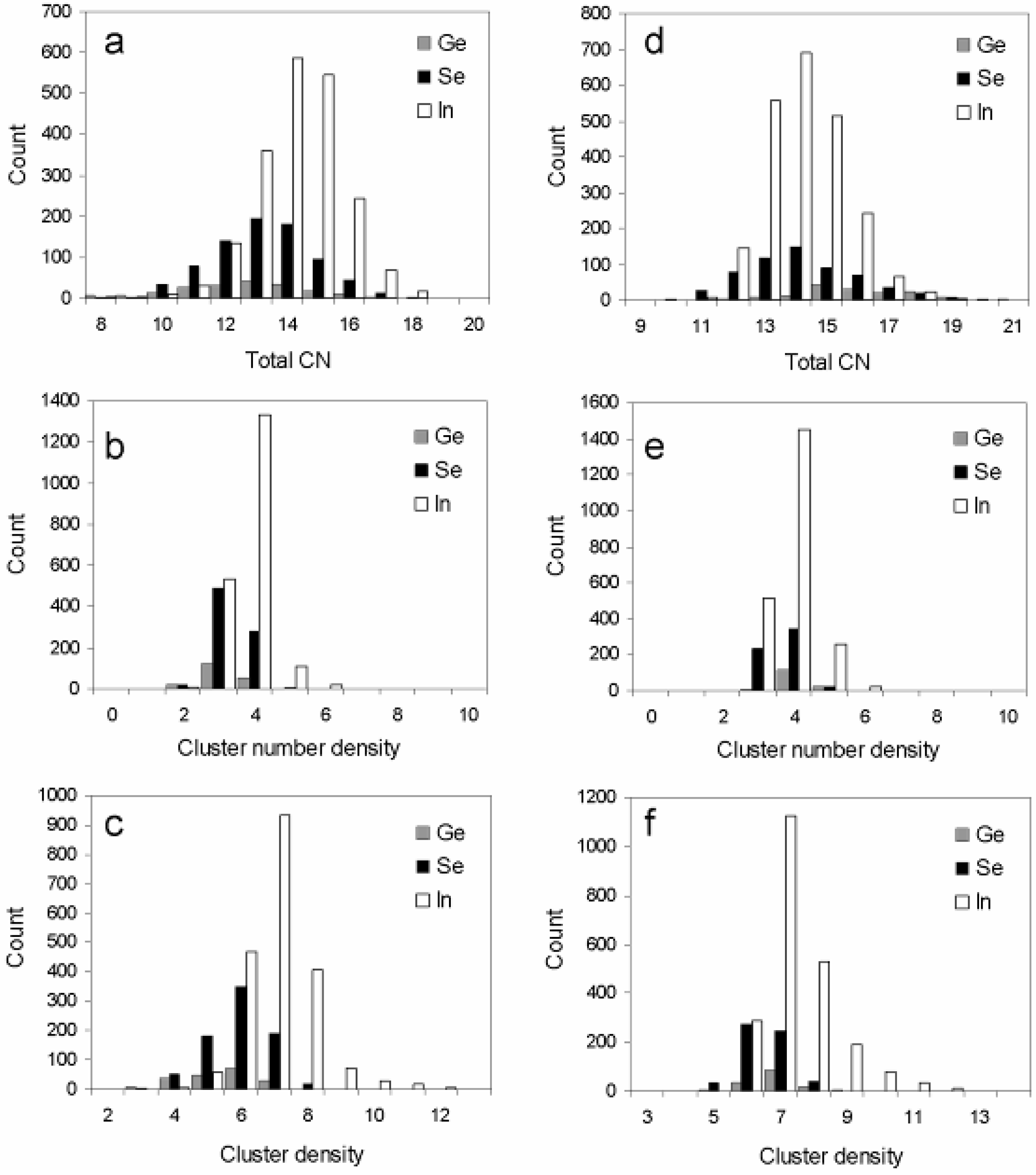

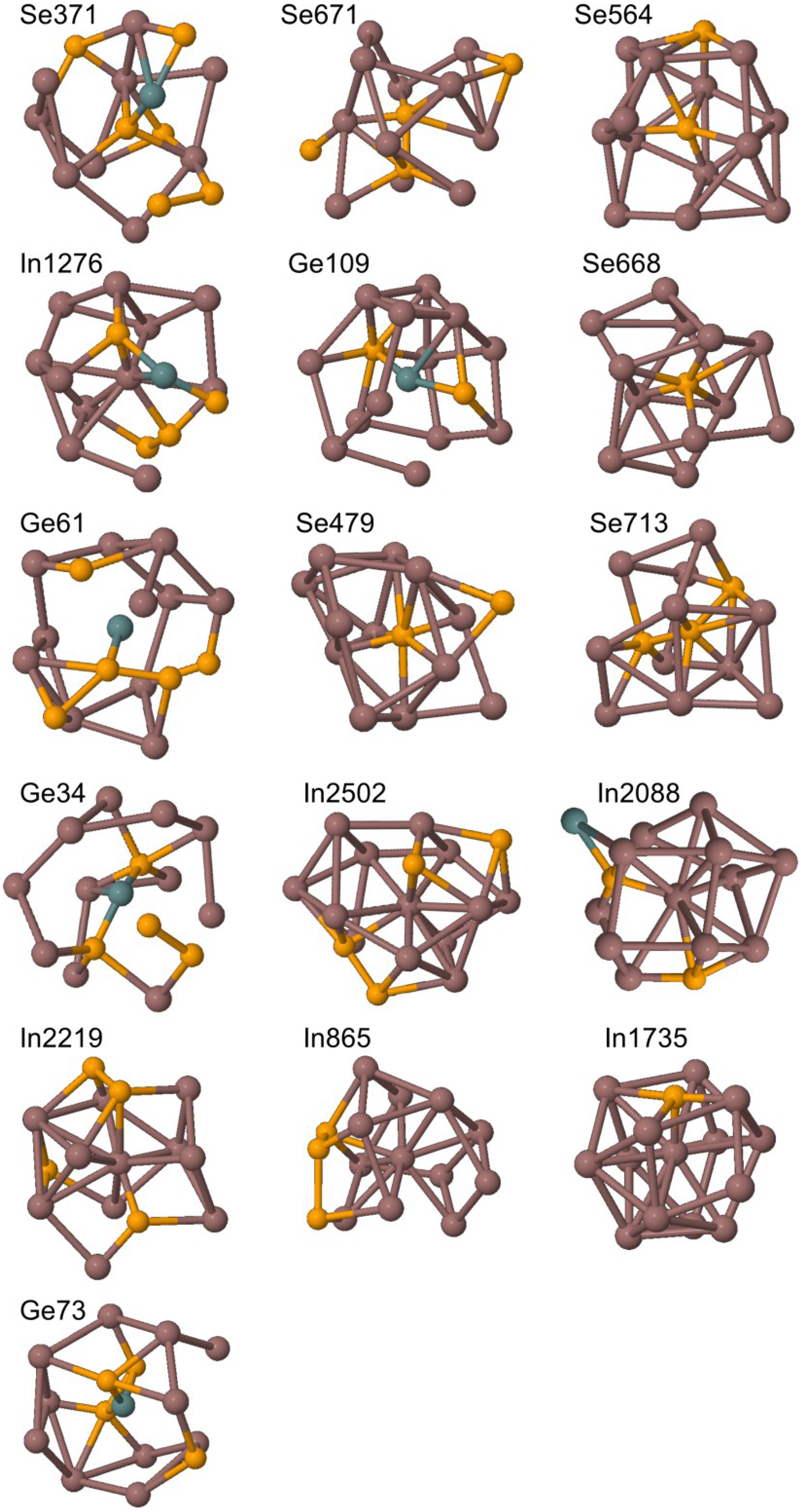
| Reference system | Center atom | Cluster designation | Average coordination of cluster center | Cluster density (g/mL) | Cluster number density (Atoms/Å3) | Central atom distance from RMC box origin (Å) | Number of Ge surface atoms | Number of Se surface atoms | Number of In surface atoms |
|---|---|---|---|---|---|---|---|---|---|
| GeSe4In10 | Ge | 34 | 14 | 6.268 | 0.03(2859) | 2.865 | 0 | 2 | 12 |
| GeSe4In10 | Se | 248 | 14 | 6.410 | 0.03(3600) | 3.037 | 1 | 1 | 12 |
| GeSe4In10 | Se | 371 | 11 | 5.598 | 0.03(0197) | 2.734 | 1 | 2 | 8 |
| GeSe4In10 | In | 1276 | 14 | 7.709 | 0.04(1329) | 1.256 | 1 | 3 | 10 |
| GeSe4In10 | In | 2088 | 15 | 6.716 | 0.03(5206) | 4.513 | 1 | 2 | 12 |
| GeSe4In15 | Ge | 61 | 16 | 6.995 | 0.03(8950) | 9.441 | 0 | 5 | 11 |
| GeSe4In15 | Ge | 73 | 15 | 7.224 | 0.03(9512) | 23.148 | 0 | 4 | 11 |
| GeSe4In15 | Ge | 109 | 15 | 7.053 | 0.03(6971) | 23.075 | 0 | 2 | 13 |
| GeSe4In15 | Se | 479 | 12 | 6.779 | 0.03(4477) | 6.715 | 0 | 1 | 11 |
| GeSe4In15 | Se | 564 | 14 | 6.765 | 0.03(4554) | 8.228 | 0 | 1 | 13 |
| GeSe4In15 | Se | 668 | 11 | 7.518 | 0.03(7109) | 2.086 | 0 | 0 | 11 |
| GeSe4In15 | Se | 671 | 14 | 7.713 | 0.04(1187) | 8.277 | 0 | 3 | 11 |
| GeSe4In15 | Se | 713 | 13 | 5.878 | 0.03(0678) | 5.771 | 0 | 2 | 11 |
| GeSe4In15 | In | 865 | 14 | 7.563 | 0.03(9488) | 6.124 | 0 | 3 | 11 |
| GeSe4In15 | In | 1735 | 14 | 6.772 | 0.03(3854) | 5.810 | 0 | 1 | 13 |
| GeSe4In15 | In | 2219 | 14 | 7.458 | 0.03(9824) | 5.467 | 0 | 4 | 10 |
| GeSe4In15 | In | 2502 | 17 | 7.582 | 0.04(0358) | 6.267 | 0 | 4 | 13 |
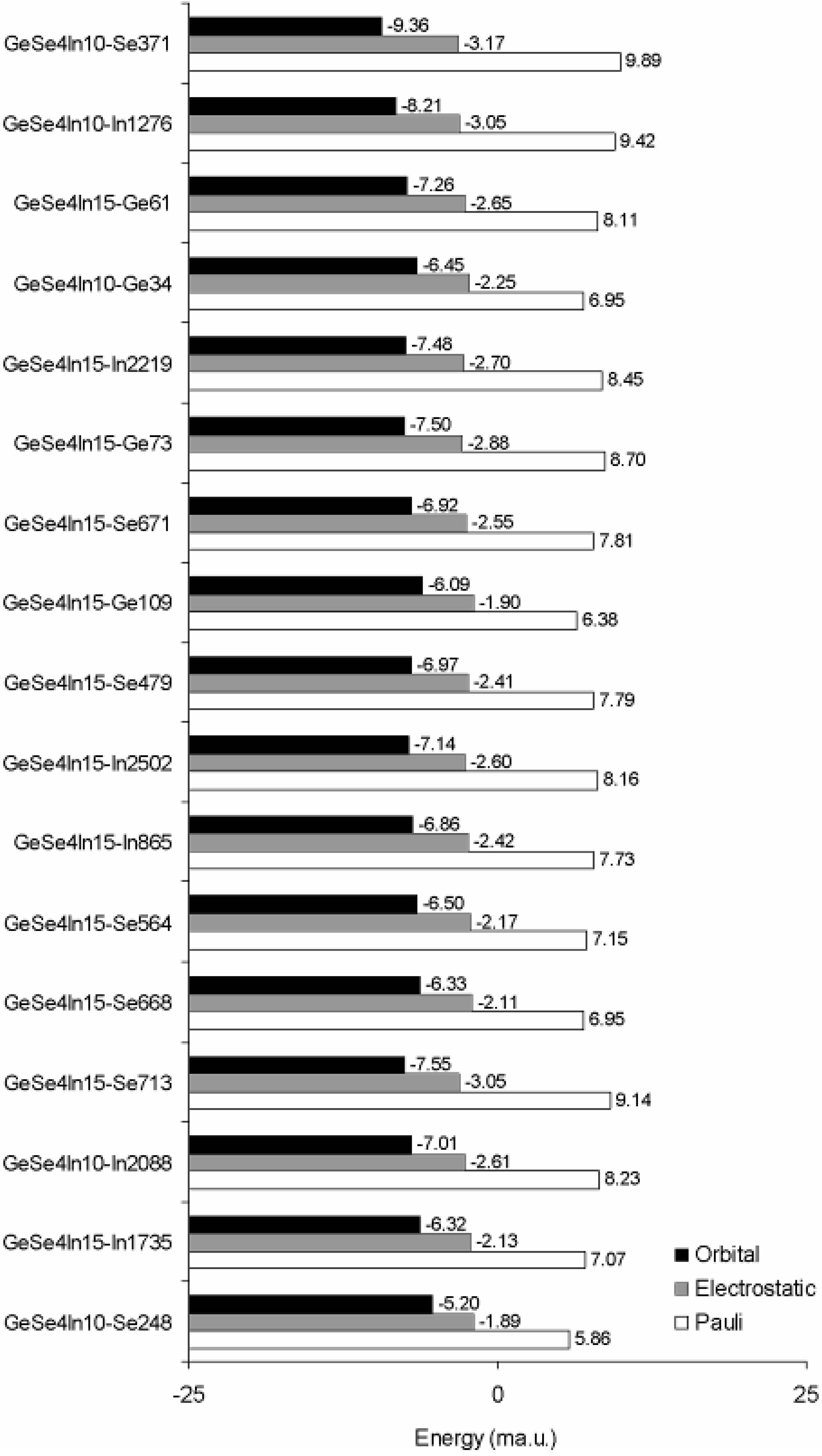
3.3. Binding Energy Decomposition and Molecular Orbital Interactions
| Cluster name | Spin polarization | Ne | Pauli (a.u.) | Electrostatic (a.u.) | Orbital Interactions (a.u.) |
|---|---|---|---|---|---|
| GeSe4In10-Se371 | 1 | 526 | 5.201 | −1.666 | −4.925 |
| GeSe4In10-In1276 | 1 | 673 | 6.341 | −2.053 | −5.522 |
| GeSe4In15-Ge61 | 1 | 741 | 6.011 | −1.966 | −5.379 |
| GeSe4In10-Ge34 | 1 | 688 | 4.781 | −1.545 | −4.439 |
| GeSe4In15-In2219 | 1 | 675 | 5.701 | −1.823 | −5.046 |
| GeSe4In15-Ge73 | 1 | 707 | 6.151 | −2.035 | −5.304 |
| GeSe4In15-Se671 | 1 | 675 | 5.269 | −1.721 | −4.671 |
| GeSe4In15-Ge109 | 1 | 737 | 4.701 | −1.401 | −4.491 |
| GeSe4In15-Se479 | 0 | 607 | 4.729 | −1.465 | −4.231 |
| GeSe4In15-In2502 | 1 | 822 | 6.708 | −2.134 | −5.866 |
| GeSe4In15-In865 | 0 | 690 | 5.335 | −1.673 | −4.737 |
| GeSe4In15-Se564 | 1 | 705 | 5.040 | −1.528 | −4.586 |
| GeSe4In15-Se668 | 0 | 573 | 3.983 | −1.207 | −3.626 |
| GeSe4In15-Se713 | 1 | 641 | 5.860 | −1.955 | −4.839 |
| GeSe4In10-In2088 | 0 | 737 | 6.065 | −1.924 | −5.166 |
| GeSe4In15-In1735 | 0 | 720 | 5.094 | −1.535 | −4.551 |
| GeSe4In10-Se248 | 1 | 688 | 4.032 | −1.301 | −3.576 |

4. Conclusions
- (1)
- The extra In content of the GeSe4In15 system appeared to have substantially demoted contributions from Ge–Se in favor of Se–In, which shaped the shell almost exclusively. The role of Se–Se was faint, in similarity to GeSe4In10.
- (2)
- In both glasses studied, interactions beyond the first coordination shell revealed great similarity among the shapes of the partials involved. The Se–In partial was instrumental towards second coordination shell formation; Ge–In contributed only fractionally towards second coordination and this behavior was common to both alloys. Third shell interatomic interactions comprised two overlapping peaks at 3.3 and 3.5 Å due to Se–In and In–In interactions.
- (3)
- The excess In content in GeSe4In15 mediated the formation of rich Ge-centered clusters at radial distances further than 4 Å from the RMC center, an effect which also flagged the reduction of bonding between Ge and Se near the RMC center.
- (4)
- Ge–Se and Se–In bonding promoted overall cluster stability and the intervention of excess In caused breaking of these bonds contributed towards a lower binding energy.
- (5)
- The introduction of excess In resulted in higher Se–In cooperation towards frontier orbitals. Direct interactions between Ge and In were scarce and limited to the GeSe4In10 glass. On the whole, Ge and Se competed for connectivity with Se over the whole range of valence electron energies; however, Indium, particularly in the GeSe4In15 system, was far more effective in bonding with Se.
Author Contributions
Conflicts of Interest
References
- Saffarini, G.; Saiter, J.M.; Schmitt, H. The composition dependence of the optical band gap in Ge–Se–In thin films. Opt. Mater. 2007, 29, 1143–1147. [Google Scholar] [CrossRef]
- Kumar, A.; Husain, M.; Swarup, S.; Nigam, A.N.; Kumar, A. X-ray spectroscopic studies in glassy semiconducting Ge20Se80 and Ge20Se80-xInx alloys. Physica B 1990, 162, 177–180. [Google Scholar] [CrossRef]
- Dejus, R.J.; Susman, S.; Volin, K.J.; Montague, D.G.; Price, D.L. Structure of vitreous AgGeSe. J. Non-Cryst. Solids 1992, 143, 162–180. [Google Scholar] [CrossRef]
- Usuki, T.; Uemura, O.; Fujimura, K.; Kameda, Y. Local arrangement in Ge-Se-In glasses. J. Non-Cryst. Solids 1995, 192–193, 69–73. [Google Scholar] [CrossRef]
- Conde Garrido, J.M.; Ureña, M.A.; Arcondo, B. Ion selective electrodes based on chalcogenide glasses. J. Alloys Compd. 2010, 495, 356–359. [Google Scholar] [CrossRef]
- Sharma, I.; Tripathi, S.K.; Monga, A.; Barman, P.B. Electrical properties of a-Ge-Se-In thin films. J. Non-Cryst. Solids 2008, 354, 3215–3219. [Google Scholar] [CrossRef]
- Goyal, D.R.; Maan, A.S. Far-infrared absorption in amorphous Sb15GexSe85−x glasses. J. Non-Cryst. Solids 1995, 183, 182–185. [Google Scholar] [CrossRef]
- Tohge, N.; Minami, T.; Yamamoto, Y.; Tanaka, M. Electrical and optical properties of n-type semiconducting chalcogenide glasses in the system Ge-Bi-Se. J. Appl. Phys. 1980, 51, 1048–1053. [Google Scholar] [CrossRef]
- Giridhar, A.; Mahadevan, S. The Tg versus Z dependence of glasses of the Ge-In-Se system. J. Non-Cryst. Solids 1992, 151, 245–252. [Google Scholar] [CrossRef]
- Rabinal, M.K.; Sangunni, K.S.; Gopal, E.S.R. Chemical ordering in Ge20Se80−xInx glasses. J. Non-Cryst. Solids 1995, 188, 98–106. [Google Scholar] [CrossRef]
- Tanaka, K. Structural phase transitions in chalcogenide glasses. Phys. Review B 1989, 39, 1270–1279. [Google Scholar] [CrossRef]
- Liang, Z. Chemical bond approach to the chalcogenide glass forming tendency. J. Non-Cryst. Solids 1991, 127, 298–305. [Google Scholar] [CrossRef]
- Kaban, I.; Jovari, P.; Petkova, T.; Petkov, P.; Stoilova, A.; Hoyer, W.; Beuneu, B. Structure of GeSe4-In and GeSe5-In glasses. J. Phys. Condens. Matter 2010, 22, 404205. [Google Scholar] [CrossRef] [PubMed]
- Faber, T.E.; Ziman, J.M. A theory of the electrical properties of liquid metals. Philos. Mag. 1965, 11, 153–173. [Google Scholar] [CrossRef]
- Gereben, O.; Pusztai, L. RMC_POT: A computer code for reverse Monte Carlo modeling the structure of disordered systems containing molecules of arbitrary complexity. J. Comput. Chem. 2012, 33, 2285–2291. [Google Scholar] [CrossRef] [PubMed]
- Saffarini, G. Fragmentation of Ge1−xSnxSe2 glasses. Solid State Commun. 1994, 10, 677–681. [Google Scholar] [CrossRef]
- Saffarini, G. Atomic density versus average coordination number in Ge–In–Se glasses. Phys. Status Solidi 1999, 2, 261–265. [Google Scholar] [CrossRef]
- Antipas, G.S.E. Molecular orbital interactions in glass-forming Zr70Cu30 liquid quasicrystals. J. Alloys Compd. 2013, 578, 565–570. [Google Scholar] [CrossRef]
- Becke, A.D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 1988, 38, 3098–3100. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef]
- Van Lenthe, E.; Baerends, E.J.; Snijders, J.G. Relativistic total energy using regular approximations. J. Chem. Phys. 1994, 101, 9783–9792. [Google Scholar] [CrossRef]
- Belanzoni, P.; van Lenthe, E.; Baerends, E.J. An evaluation of the density functional approach in the zero order regular approximation for relativistic effects: Magnetic interactions in small metal compounds. J. Chem. Phys. 2001, 114, 4421–4433. [Google Scholar] [CrossRef]
- Van Lenthe, E.; van der Avoird, A.; Wormer, P.E.S. Density functional calculations of molecular hyperfine interactions in the zero order regular approximation for relativistic effects. J. Chem. Phys. 1998, 108, 4783–4796. [Google Scholar]
- Amsterdam Density Functional Program; Vrije Universiteit: Amsterdam, The Netherlands, 2010.
- Salmon, P.S. Structure of liquids and glasses in the Ge–Se binary system. J. Non-Cryst. Solids 2007, 353, 2959–2974. [Google Scholar] [CrossRef]
- Ledru, J.; Saiter, J.M.; Saffarini, G.; Benazeth, S. An EXAFS investigation of the local order around indium in Ge–Se–In glasses. J. Non-Cryst. Solids 1998, 232–234, 634–637. [Google Scholar] [CrossRef]
- Antipas, G.S.E.; Mangiorou, E.; Hristoforou, E. Solute-solvent interactions and atomic cohesion in GeSe4 and GeSe4In5 metallic glasses. Mater. Res. Express 2014, 1, 015202. [Google Scholar] [CrossRef]
- Abdel-Rahim, M.A.; Hafiz, M.M.; El-Nahass, M.M.; Shamekh, A.M. Influence of composition on optical and electrical properties of Ge–Se–In thin films. Physica B 2007, 387, 383–391. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antipas, G.S.E.; Mangiorou, E.; Hristoforou, E. The Effect of Indium Content on the Atomic Environment and Cluster Stability of GeSe4Inx=10,15 Glasses. Metals 2015, 5, 102-118. https://doi.org/10.3390/met5010102
Antipas GSE, Mangiorou E, Hristoforou E. The Effect of Indium Content on the Atomic Environment and Cluster Stability of GeSe4Inx=10,15 Glasses. Metals. 2015; 5(1):102-118. https://doi.org/10.3390/met5010102
Chicago/Turabian StyleAntipas, Georgios S. E., Eleni Mangiorou, and Evangelos Hristoforou. 2015. "The Effect of Indium Content on the Atomic Environment and Cluster Stability of GeSe4Inx=10,15 Glasses" Metals 5, no. 1: 102-118. https://doi.org/10.3390/met5010102






