Influence of Micron-Ti and Nano-Cu Additions on the Microstructure and Mechanical Properties of Pure Magnesium
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis
2.2. Density and Porosity
| S. No. | Material | Density Measurements | ||
|---|---|---|---|---|
| Theoretical Density | Experimental Density | Porosity | ||
| (g/cc) | (g/cc) | (%) | ||
| 1 | Pure Mg | 1.7400 | 1.7397 ± 0.0015 | 0.02 |
| 2 | Mg-Ti | 1.8019 | 1.8002 ± 0.0017 | 0.09 |
| 3 | Mg-Cu | 1.9685 | 1.9681 ± 0.0010 | 0.04 |
| 4 | Mg-Ti-Cu | 2.1106 | 2.1089 ± 0.0019 | 0.16 |
| 5 | Mg-(Ti + Cu)BM | 2.1106 | 2.1096 ± 0.0016 | 0.09 |
2.3. X-ray Diffraction

2.4. Microstructure

| S. No. | Material | Grain size (µm) | Aspect ratio (µm) |
|---|---|---|---|
| 1 | Pure Mg | 21 ± 3 | 1.63 ± 0.44 |
| 2 | Mg-Ti | 15 ± 4 | 1.58 ± 0.40 |
| 3 | Mg-Cu | 11 ± 2 | 1.66 ± 0.24 |
| 4 | Mg-Ti-Cu | 9 ± 3 | 1.60 ± 0.52 |
| 5 | Mg-(Ti + Cu)BM | 7 ± 3 | 1.56 ± 0.50 |


2.5. Mechanical Properties
| S. No. | Material | Microhardness [Hv] |
|---|---|---|
| 1 | Pure Mg | 48 ± 1 |
| 2 | Mg-Ti | 71 ± 2 |
| 3 | Mg-Cu | 82 ± 4 |
| 4 | Mg-Ti-Cu | 86 ± 2 |
| 5 | Mg-(Ti + Cu)BM | 91 ± 3 |
| S. No. | Material | Tensile properties | Compressive properties | ||||
|---|---|---|---|---|---|---|---|
| 0.2 YS [MPa] | UTS [MPa] | Failure Strain [%] | 0.2 CYS [MPa] | UCS [MPa] | Failure Strain [%] | ||
| 1 | Mg | 125 ± 9 | 169 ± 11 | 6.2 ± 0.7 | 74 ± 3 | 273 ± 11 | 22.7 ± 4.9 |
| 2 | Mg-Ti | 158 ± 6 | 226 ± 6 | 8.0 ± 1.5 | 85 ± 3 | 360 ± 5 | 13.6 ± 1.2 |
| 3 | Mg-Cu | 182 ± 4 | 220 ± 4 | 8.9 ± 0.9 | 102 ± 8 | 386 ± 4 | 21.1± 1.7 |
| 4 | Mg-Ti-Cu | 196 ± 9 | 227 ± 4 | 5.7 ± 1.6 | 116 ± 5 | 342 ± 3 | 16.1 ± 1.4 |
| 5 | Mg-(Ti + Cu)BM | 201 ± 7 | 265 ± 11 | 7.5 ± 0.8 | 126 ± 8 | 380 ± 6 | 19.1 ± 2.9 |
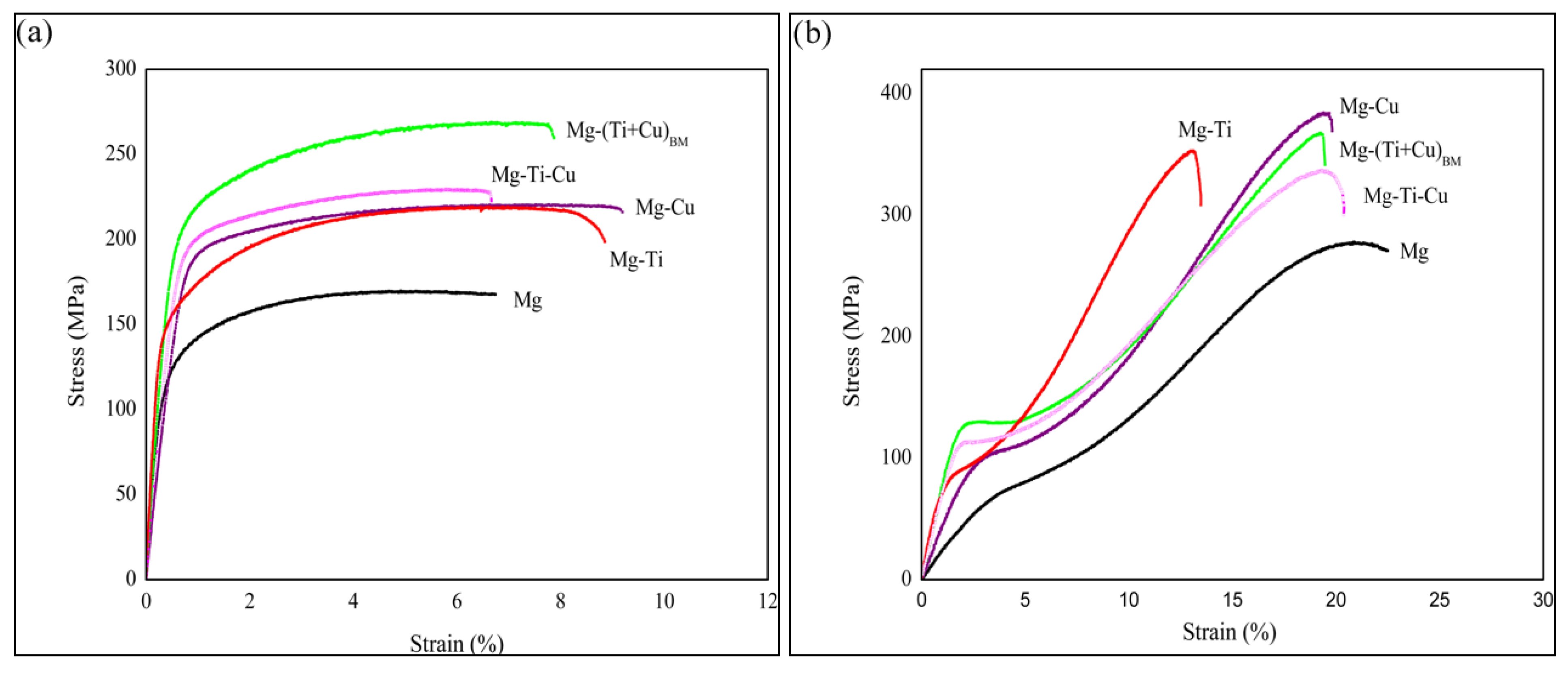
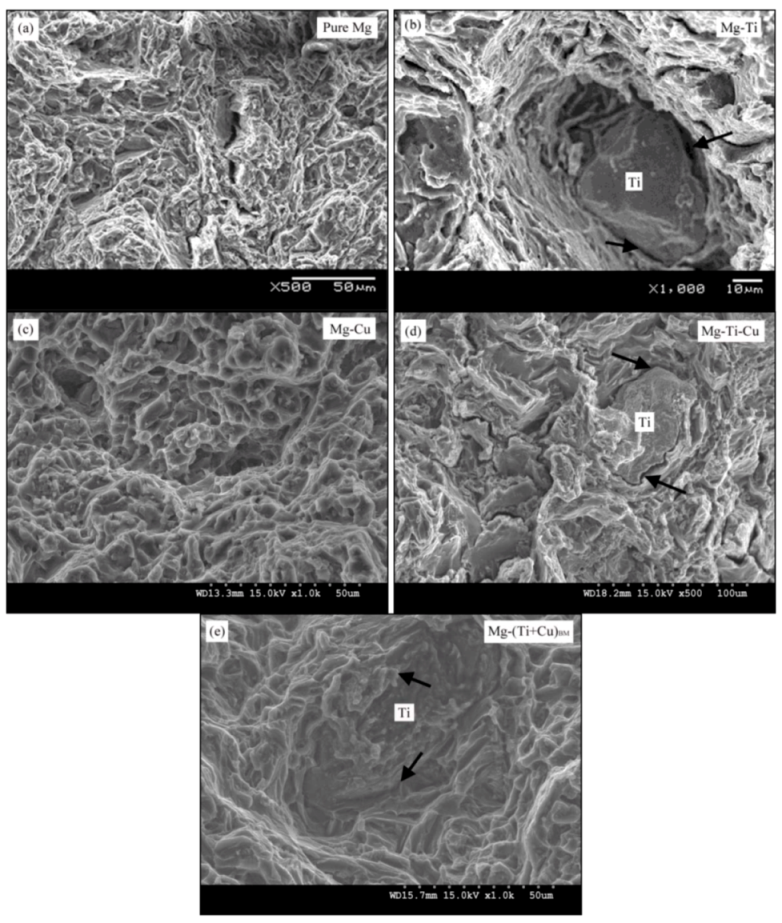
2.5.1. Influence of Individual Ti Addition

2.5.2. Influence of Individual Nano-Cu Addition
2.5.3. Influence of Direct addition of Hybrid (Ti and nano-Cu) Metallic Particulates
2.5.4. Influence of Prior Ball-milling of Hybrid (Ti and nano-Cu) Metallic Particulates
3. Experimental Procedure
3.1. Materials
3.2. Processing
3.2.1. Pre-processing of Metallic Additions
3.2.2. Melting and Casting
3.2.3. Hot Extrusion
3.3. Materials Characterization
3.3.1. Density Measurements
3.3.2. X-ray Diffraction Studies
3.3.3. Microstructural Characterization
3.3.4. Mechanical Properties Measurements
4. Conclusions
- (1) The addition of micron sized (Ti) particulates with negligible solubility and nano-sized Cu particulates with limited solubility in Mg, resulted in the formation of Mg-Ti composite and Mg-Cu alloy, respectively.
- (2) While the inherent properties of Ti such as hardness, ductility and the good wettability of Ti and Mg enhanced the strength and ductility of the Mg-Ti composite, the Mg2Cu eutectic phase formation refined the grain size and significantly improved the mechanical properties in Mg-Cu alloy.
- (3) The effects of the combined addition of (micro-Ti and nano-Cu) metallic elements on the properties of Mg primarily depend on their method of addition.
- (4) In Mg-Ti-Cu, involving direct addition of micro-Ti and nano-Cu, strength properties improvement with an adverse effect on ductility occurred and the reduction in ductility is attributed to the overall higher volume fraction of the metallic particulates and intermetallics, morphology of micron-sized Ti-particle, formation and agglomeration of Mg2Cu/Cu.
- (5) The best strength properties were obtained in Mg-(Ti + Cu)BM, through the combined addition of micron-sized Ti particles and nano-sized Cu particles after prior ball-milling. The elimination of sharp corners of Ti particulates and Ti3Cu intermetallic formation during ball milling attributes to the simultaneous improvement in strength and ductility when compared to Mg-5.6Ti-3Cu.
Conflict of Interest
Acknowledgements
References
- Kainer, K.U. Metal Matrix Composites: Custom-Made Materials for Automotive and Aerospace Engineering; Wiley-VCH: Weinheim, Germany, 2006. [Google Scholar]
- Emley, E.F. Principles of Magnesium Technology; Pergamon Press: Oxford, UK, 1966. [Google Scholar]
- Smithells, C.J. Metals Reference Book, 5th ed; Butterworth’s & Co. Ltd.: London, UK, 1976. [Google Scholar]
- Mordike, B.L.; Ebert, T. Magnesium: Properties-applications-potential. Mater. Sci. Eng. A 2001, 302(1), 37–45. [Google Scholar] [CrossRef]
- Tharumarajah, A.; Koltun, P. Is there an environmental advantage of using magnesium components for light-weighting cars? J. Cleaner Prod. 2007, 15, 1007–1013. [Google Scholar] [CrossRef]
- Polmear, I.J. Magnesium alloys and Applications. Mater. Sci. Technol. 1994, 10, 1–16. [Google Scholar] [CrossRef]
- Pettersen, G.; Westengen, H.; Høier, R.; Lohne, O. Microstructure of a pressure die cast magnesium—4 wt% aluminium alloy modified with rare earth additions. Mater. Sci. Eng. A 1996, 207, 115–120. [Google Scholar] [CrossRef]
- Du, W.; Sun, Y.; Min, X.; Xue, F.; Zhu, M.; Wu, D. Microstructure and mechanical properties of Mg-Al based alloy with calcium and rare earth additions. Mater. Sci. Eng. A 2003, 356, 1–7. [Google Scholar] [CrossRef]
- Luo, A.; Perquleryuz, M.O. Cast magnesium alloys for elevated temperature applications. J. Mater. Sci. 1994, 29, 5259–5271. [Google Scholar] [CrossRef]
- Tang, W.N.; Park, S.S.; You, B.S. Effect of the Zn content on the microstructure and mechanical properties of indirect-extruded Mg-5Sn-xZn alloys. Mater. Des. 2011, 32, 3537–3543. [Google Scholar] [CrossRef]
- Fritze, C.; Berek, H.; Kainer, K.U.; Mielke, S.; Wielage, B. Magnesium Alloys and Their Applications; Mordike, B.L., Kainer, K.U., Eds.; Werkstoff-Information-sgesellschaft: Frankfurt, Germany, 1998; pp. 635–640. [Google Scholar]
- Gupta, M.; Lai, M.O.; Sarvanaranganathan, D. Synthesis, microstructure and properties characterization of disintegrated melt deposited Mg/SiC composites. J. Mater. Sci. 2000, 35, 2155–2165. [Google Scholar] [CrossRef]
- Ugandhar, S.; Gupta, M.; Sinha, S.K. Enhancing strength and ductility of Mg/SiC composites using recrystallization heat treatment. Compos. Struct. 2006, 72, 266–272. [Google Scholar] [CrossRef]
- Hassan, S.F.; Gupta, M. Development of a novel magnesium/nickel composite with improved mechanical properties. J. Alloys Compd. 2002, 335, L10–L15. [Google Scholar] [CrossRef]
- Hassan, S.F.; Gupta, M. Development of a novel magnesium-copper based composite with improved mechanical properties. Mater. Res. Bull. 2002, 37, 377–389. [Google Scholar] [CrossRef]
- Pérez, P.; Garcés, G.; Adeva, P. Mechanical properties of a Mg-10 (vol%)Ti composite. Compos. Sci. Technol. 2004, 64, 145–151. [Google Scholar] [CrossRef]
- Ferkel, H.; Mordike, B.L. Magnesium strengthened by SiC nanoparticles. Mater. Sci. Eng. A. 2001, 298, 193–199. [Google Scholar] [CrossRef]
- Dieringa, H. Properties of magnesium alloys reinforced with nanoparticles and carbon nanotubes: A review. J. Mater. Sci. 2011, 46, 289–306. [Google Scholar] [CrossRef]
- Goh, C.S.; Wei, J.; Lee, L.C.; Gupta, M. Simultaneous enhancement in strength and ductility by reinforcing magnesium with carbon nanotubes. Mater. Sci. Eng. A. 2006, 423, 153–156. [Google Scholar] [CrossRef]
- Hassan, S.F.; Gupta, M. Effect of particulate size of Al2O3 reinforcement on microstructure and mechanical behavior of solidification processed elemental Mg. J. Alloys Compd. 2006, 419, 84–90. [Google Scholar] [CrossRef]
- Sankaranarayanan, S.; Jayalakshmi, S.; Gupta, M. Effect of individual and combined addition of micro/nano-sized metallic elements on the microstructure and mechanical properties of pure Mg. Mater. Des. 2012, 37, 274–284. [Google Scholar] [CrossRef]
- Sankaranarayanan, S.; Jayalakshmi, S.; Gupta, M. Effect of addition of mutually soluble and insoluble metallic elements on the microstructure, tensile and compressive properties of pure magnesium. Mater. Sci. Eng A. 2011, 530, 149–160. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, H.; Liao, L.; Teng, X.; Ma, N. The mechanical properties of magnesium matrix composites reinforced with (TiB2 + TiC) ceramic particulates. Mater. Lett. 2005, 59, 2105–2109. [Google Scholar] [CrossRef]
- Binary Alloy Phase Diagram [Electronic Resource], 2nd ed; ASM International: Materials Park, OH, USA, 1996.
- Andrieux, J.; Dezellus, O.; Bosselet, F.; Sacerdote-Peronnet, M.; Sigala, C.; Chiriac, R. Details on the formation of Ti2Cu3 in the Ag-Cu-Ti system in the temperature range 790 to 860 C. J. Phase Equilib. Diffus. 2008, 29(2), 156–162. [Google Scholar] [CrossRef]
- Konieczny, M. Processing and microstructural characterisation of laminated Ti-intermetallic composites synthesised using Ti and Cu foils. Mater. Lett. 2008, 62, 2600–2602. [Google Scholar] [CrossRef]
- Lütjering, G.; Weissmann, S. Mechanical properties and structure of age-hardened Ti-Cu alloys. Metall. Mater. Trans. B 1970, 1(6), 1641–1649. [Google Scholar] [CrossRef]
- Anisimova, M.V.; Nuzhdin, G.A. New functional compositions of mixture for superhard composites based on bn blende. Refract. Ind. Ceram. 1999, 40(3–4), 98–100. [Google Scholar] [CrossRef]
- Kondoh, K.; Kawakami, M.; Imai, H.; Umeda, J.; Fujii, H. Wettability of pure Ti by molten pure Mg droplets. Acta Mater. 2010, 58, 606–614. [Google Scholar] [CrossRef]
- Gupta, M.; Srivatsan, T.S. Microstructure and grain growth behavior of an aluminum alloy metal matrix composite processed by disintegrated melt deposition. J. Mater. Eng. Perform. 1999, 8(4), 473–478. [Google Scholar] [CrossRef]
- Lloyd, D.J. Particle reinforced aluminium and magnesium matrix composites. Int Mater. Rev. 1994, 39(1), 1–23. [Google Scholar] [CrossRef]
- Ahmed, S.; Jones, F.R. Effect of particulate agglomeration and the residual stress state on the modulus of filled resin. Part II: Moduli of untreated sand and glass bead filled composites. Composites 1990, 21, 81–84. [Google Scholar] [CrossRef]
- Suryanarayana, C. Mechanical Alloying and Milling; CRC Press: NewYork, NY, USA, 2004. [Google Scholar]
- Habibi, M.K.; Pouriayevali, H.; Gupta, M. Effect of strain rate and ball milling of reinforcement on the compressive response of magnesium composites. Composites Part A 2011, 42(12), 1920–1929. [Google Scholar]
- Qin, S.; Chen, C.; Zhang, G.; Wang, W.; Wang, Z. The effect of particle shape on ductility of SiCp reinforced 6061 Al matrix composites. Mater. Sci. Eng. A 1999, 272, 363–370. [Google Scholar] [CrossRef]
- Dieter, G. Mechanical Metallurgy; McGraw-Hill: New York, NY, USA, 1986. [Google Scholar]
- Ibrahim, I.A.; Mohamed, F.A.; Lavernia, E.J. Particulate reinforced metal matrix composites—A review. J. Mater. Sci. 1991, 26, 1137–1156. [Google Scholar] [CrossRef]
- Arsenault, R.J.; Shi, N. Dislocation generation due to differences between coefficients of thermal expansion. Mater. Sci. Eng. 1986, 81, 175–187. [Google Scholar] [CrossRef]
- Mordike, B.L.; Lukac, P. Interfaces in magnesium-based composites. Surf. Interface Anal. 2001, 31, 682–685. [Google Scholar] [CrossRef]
- Barrett, P.F. Compressive Properties of Titanium Sheet at Elevated Temperatures; Technical Note 2038; National Advisory Committee for Aeronautics: Washington, DC, USA, 1950. [Google Scholar]
- Meijer, G.; Ellyin, F.; Xia, Z. Aspects of residual thermal stress/strain in particle reinforced metal matrix composites. Composites B 2000, 31, 29–37. [Google Scholar]
- Agnew, S.R.; Duygulu, O. Plastic anisotropy and the role of non-basal slip in magnesium alloy AZ31B. Int. J. Plast. 2005, 21, 1161–1193. [Google Scholar]
- Gupta, M.; Nai, S. Magnesium, Magnesium Alloys and Magnesium Composites; John Wiley: Hoboken, NJ, USA, 2011. [Google Scholar]
- Ksiazek, M.; Sobczak, N.; Mikulowski, B.; Radziwill, W.; Surowiak, I. Wetting and bonding strength in Al/Al2O3 system. Mater. Sci. Eng. A 2002, 324(1–2), 162–167. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Seetharaman, S.; Subramanian, J.; Gupta, M.; Hamouda, A.S. Influence of Micron-Ti and Nano-Cu Additions on the Microstructure and Mechanical Properties of Pure Magnesium. Metals 2012, 2, 274-291. https://doi.org/10.3390/met2030274
Seetharaman S, Subramanian J, Gupta M, Hamouda AS. Influence of Micron-Ti and Nano-Cu Additions on the Microstructure and Mechanical Properties of Pure Magnesium. Metals. 2012; 2(3):274-291. https://doi.org/10.3390/met2030274
Chicago/Turabian StyleSeetharaman, Sankaranarayanan, Jayalakshmi Subramanian, Manoj Gupta, and Abdelmagid S. Hamouda. 2012. "Influence of Micron-Ti and Nano-Cu Additions on the Microstructure and Mechanical Properties of Pure Magnesium" Metals 2, no. 3: 274-291. https://doi.org/10.3390/met2030274






