Noise Reduction Potential of Cellular Metals
Abstract
:1. Introduction
2. Material Characterization



| Material | Alloy | Porosity | Cell/pore size * | Average pore size |
|---|---|---|---|---|
| MF-50 | Haynes Alloy 230 | 82% | n/a | 121 ± 9 µm |
| MF-450 | NiFe22Cr22Al6 | 90% | 450 ± 45 µm | 380 ± 19 ** µm |
| MF-580 | NiFe22Cr22Al6 | 92% | 580 ± 60 µm | 445 ± 18 ** µm |
| MF-800 | NiFe22Cr22Al6 | 94% | 800 ± 80 µm | 538 ± 58 ** µm |
| MF-1200 | NiFe22Cr22Al6 | 95% | 1200 ± 120 µm | 656 ± 59 ** µm |
| SF-7.5 | Hastelloy X | 20% | 1.4~8 µm | 8.6 ± 1.2 µm |
| SF-20 | Hastelloy X | 35% | 12~25 µm | 22 ± 2 µm |
| SF-115 | Hastelloy X | 50% | 24~117 µm | 113 ± 13 µm |
| SS-25 | CuSn12 | 30% | 8~21 µm | 25 ± 2 µm |
| SS-80 | CuSn12 | 25% | 37~175 µm | 80 ± 12 µm |
| SFF-1 | Fe-22Cr-5Al (316 L) | 60 *% | 3~10 µm | 6.7 * µm |
| SFF-15 | Fe-22Cr-5Al (316 L) | 77 *% | 13~26 µm | 18 * µm |
| SFF-25 | Fe-22Cr-5Al (316 L) | 80 *% | 22~52 µm | 29 * µm |
| SFF-50 | Fe-22Cr-5Al (316 L) | 68 *% | 33~98 µm | 51 * µm |
| SFF-100 | Fe-22Cr-5Al (316 L) | 84 *% | 38~112 µm | 99 ± 8 µm |
| SFF-120 | Fe-22Cr-5Al (316 L) | 92% | n/a | 337 ± 83 µm |
| SFF-150 | Fe-22Cr-5Al (316 L) | 96% | n/a | 386 ± 14 µm |
3. Acoustic Absorption Measurement Using an Impedance Tube
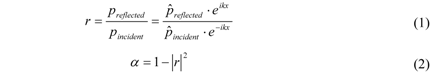
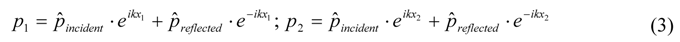
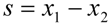 ) gives the transfer functions of incident wave H1, reflected wave H2, and entire interference field H3. p1incident is the incident part of p1 and p1reflected is the reflected part of p1. The same applies for p2.
) gives the transfer functions of incident wave H1, reflected wave H2, and entire interference field H3. p1incident is the incident part of p1 and p1reflected is the reflected part of p1. The same applies for p2.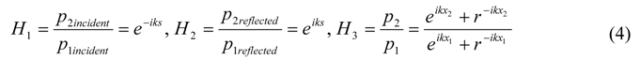
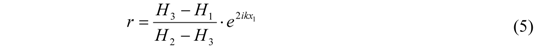

4. Discussion
5. Conclusions
Conflict of Interest
Acknowledgments
References
- Haines, M.M.; Stansfeld, S.A.; Job, R.F.S.; Berglund, B. Chronic aircraft noise exposure, stress responses, mental health and cognitive performance in school children. Psychl. Med. 2001, 31, 265–277. [Google Scholar] [CrossRef]
- Franssen, E.; van Wiechen, C.; Nagelkerke, N.; Lebret, E. Aircraft noise around a large international airport and its impact on general health and medication use. Occup. Environ. Med. 2004, 61, 405–413. [Google Scholar] [CrossRef]
- Golosnoy, I.O.; Tan, J.C.; Clyne, T.W. Ferrous fibre network materials for jet noise reduction in aeroengines. Part I: Acoustic effects. Adv. Eng. Mater. 2008, 10, 192–200. [Google Scholar]
- Yu, C.-J.; Eifert, H.H.; Banhart, J.; Baumeister, J. Metal foaming by a powder metallurgy method: Production, properties and applications. Mater. Res. Innovat. 1998, 2, 181–188. [Google Scholar] [CrossRef]
- Pickering, S. Inco invests in its nickel future at Clydach. Met. Powder Rep. 1998, 53, 24–27. [Google Scholar]
- Brothers, A.; Scheunemann, R.; de Fouw, J.; Dunand, D. Processing and structure of open-celled amorphous metal foams. Scripta. Mater. 2005, 52, 335–339. [Google Scholar] [CrossRef]
- Hinze, B.; Rösler, J. Characterization of open-pored metals using image processing. In Characterization of Minerals, Metals and Materials; Hwang, J.-Y., Monteiro, S.N., Bai, C.-G., Carpenter, J., Cai, M., Firrao, D., Kim, B.-G., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2012; pp. 185–192. [Google Scholar]
- Hinze, B.; Rösler, J.; Schmitz, F. Production of nanoporous superalloy membranes by load-free coarsening of γ′-precipitates. Acta Mater. 2011, 59, 3049–3060. [Google Scholar] [CrossRef]
- Akustik—Bestimmung des Schallabsorptionsgrades und der Impedanz in Impedanzrohren—Teil 2: Verfahren mit Übertragungsfunktion. EN ISO 10534-2, Brussels, Belgium, 2001.
- Schirmer, W.; Lotze, E. Technischer Lärmschutz, 2nd ed; Springer: Berlin, Germany, 2006; pp. 208–216. [Google Scholar]
- Delany, M.E.; Bazley, E.N. Acoustical properties of fibrous absorbent materials. Appl. Acoust. 1970, 3, 105–116. [Google Scholar] [CrossRef]
- Allard, J.-F. New empirical equations for sound propagation in rigid frame fibrous materials. J. Acoust. Soc. Am. 1992, 91, 3346–3353. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hinze, B.; Rösler, J.; Lippitz, N. Noise Reduction Potential of Cellular Metals. Metals 2012, 2, 195-201. https://doi.org/10.3390/met2020195
Hinze B, Rösler J, Lippitz N. Noise Reduction Potential of Cellular Metals. Metals. 2012; 2(2):195-201. https://doi.org/10.3390/met2020195
Chicago/Turabian StyleHinze, Björn, Joachim Rösler, and Nicolas Lippitz. 2012. "Noise Reduction Potential of Cellular Metals" Metals 2, no. 2: 195-201. https://doi.org/10.3390/met2020195




