Graphite and Hybrid Nanomaterials as Lubricant Additives
Abstract
:1. Introduction
2. Results and Discussion
2.1. Conventional Carbon-Based Lubricants
2.1.1. Graphite and Diamond

2.1.2. Nanotubes
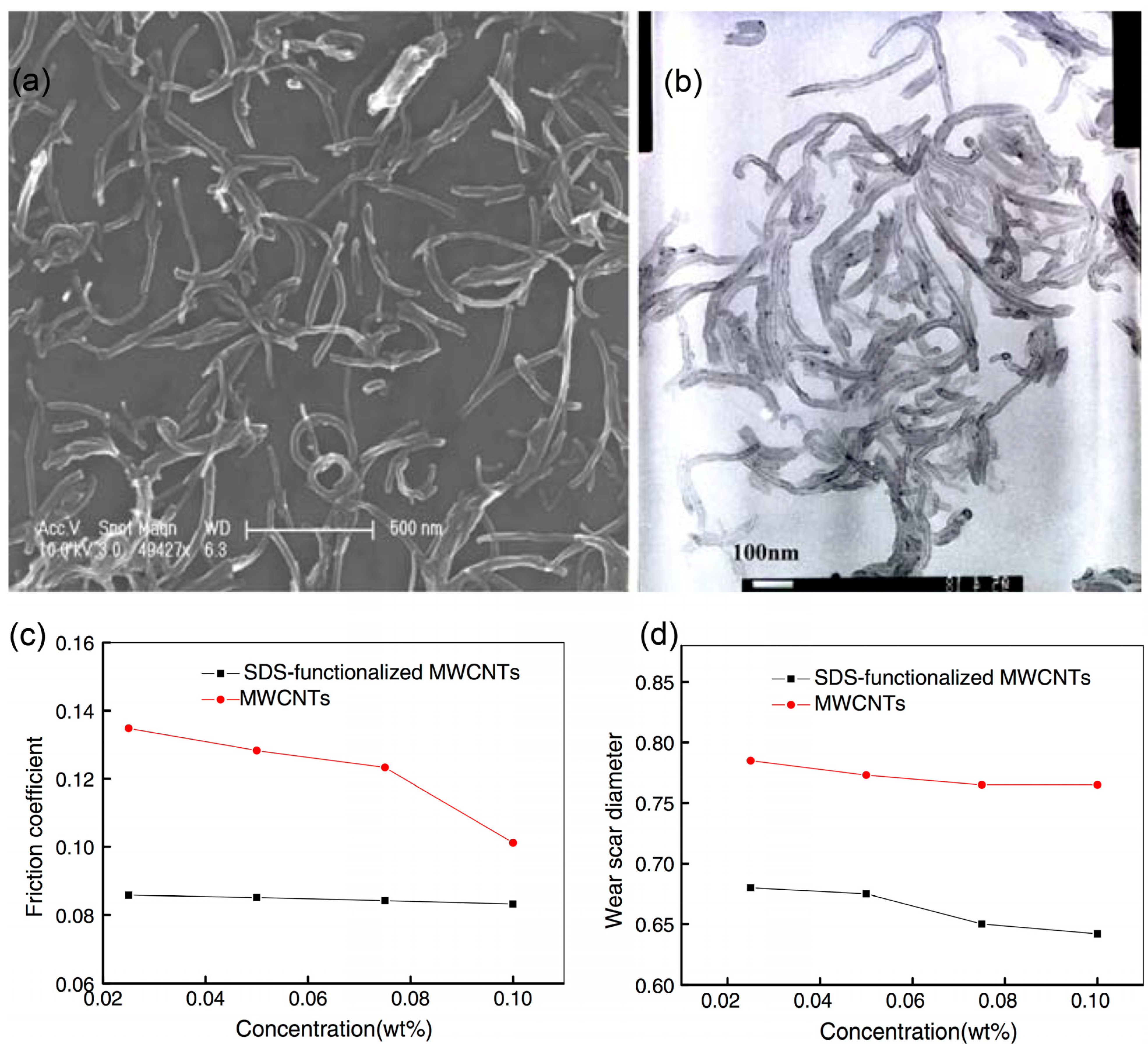
2.2. Metal-Based Nanoparticles with Capping Agents

2.3. Polymer Coated Nanoparticles

| Nanoparticle | Capping agent | Shape | Base liquid | Size/nm | Reference |
|---|---|---|---|---|---|
| Nickel-based Ni(HCOO)2·2H2O | Oleylamine Oleic acid | Face-centered Cubic | Poly-alpha-olefin (PAO6) | 7.5, 13.5, 28.5 | [9] |
| ZnS | Octadecylamine | Rod-like | Dodecane | Length ~ 5 Width ~ 1 | [18] |
| Cu | Dialkyldithiophosphate (DDP) | Spherical | Liquid paraffin | 5 | [22] |
| Cu | DDP | Face-centered Cubic | Liquid paraffin | 8 | [75,78] |
| TiO2 | Stearic acid | Spherical | Liquid paraffin | 10 | [30] |
| Gold, silver | Oleate, amine | Spherical | Hexadecane | 5–20 | [49] |
| Nano-diamond, SiO2 | Oleic acid | Spherical | Liquid paraffin | 110, 92 | [19] |
| SiO2 | Base-stabilized organic silane | Spherical | PAO | Core size: 12 | [45] |
| Multi-walled carbon nanotubes | Imidazolium cation-based ionic liquid | Tube | Ionic liquid ((Bmim) (PF6)) | Diameter: 20–40 | [71] |
| Multi-walled carbon nanotubes | Poly(ionic liquids) | Tube | Base lubricant LP104 | Diameter: 20–40 | [73] |
| Multi-walled carbon nanotubes | Room temperature ionic liquid (HEHImPF6) | Tube | Toluene, dichloromethane ionic liquid P106 | N/A | [74] |
| Cu, Ag, LaF3 | DDP | Spherical | Non-polar solvents, mineral oil | 5, 4, 2 | [85] |
| Cu | SDS, PVP, CTAB, AAS | Spherical | PEGASUS 1005 | 60, 130 | [79] |
| Cu | Tween, SDS | Spherical | Aqueous solutions | 4.8–12.1 | [81] |
| Cu | CTAB | Spherical | Toluene | 1–45 | [83] |
| Cu | EDTA | Spherical | 50CC oil | 40 | [90] |
| Cu | Carbon-coating | Spherical | PAO6 | 25 | [91] |
| ZnO | Oleic acid | Spherical | Paraffin oil | 40–100 | [88] |
| CeO2, TiO2 | Tween, Span, sodium sodecylbenzenesulfonate | Spherical | 500SN | 10.4, 15.2 | [93] |
| SiO2 | Silane coupling agents | Spherical | Mobile oil | 15–20 | [94] |
| Megnetite | Poly(methyl methacrylate) | Spherical | THF, DCM, toluene | Core size: 13 | [110] |
| Iron oxide | Poly(ethylene glycol) | Spherical | Aqueous solutions | 23–82 | [112] |
| Silica | Poly(ethylene oxide) | Spherical | Aqueous solutions | Core size: 12, 16 | [116,118] |
| Silica | Poly-l-lysine | Spherical | Aqueous solutions | Core size: 22 | [117] |
3. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Stachowiak, G.W.; Batchelor, A.W. Engineering Tribology, 4th ed.; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Abdullah, S.F. Study on nanoparticles as an additive in lubricant towards sustainability of energy in industrial engineering. J. Energy Environ. 2009, 1, 34–37. [Google Scholar]
- Hsu, S.M. Nano-lubrication: Concept and design. Tribol. Int. 2004, 37, 537–545. [Google Scholar] [CrossRef]
- Dorinson, A.; Ludema, K.C. Mechanics and Chemistry in Lubrication; Elsevier: Amsterdam, The Netherlands, 1985. [Google Scholar]
- Bakunin, V.N.; Suslov, A.Y.; Kuzmina, G.N.; Parenago, O.P. Synthesis and application of inorganic nanoparticles as lubricant components—A review. J. Nanopart. Res. 2004, 6, 273–284. [Google Scholar] [CrossRef]
- Bowden, F.P.; Tabor, D. The Friction and Lubrication of Solids; Clarendon Press: Oxford, UK, 1950. [Google Scholar]
- Choi, S.U.S. Enhancing Thermal Conductivity of Fluids with Nanoparticles. In Developments and Applications of Non-Newtonian Flows; Siginer, D.A., Wang, H.P., Eds.; ASME: New York, NY, USA, 1995; pp. 99–105. [Google Scholar]
- Buongiorno, J.; Venerus, D.C.; Prabhat, N.; McKrell, T.; Townsend, J.; Christianson, R.; Tolmachev, Y.V.; Keblinski, P.; Hu, L.; Alvarado, J.L.; et al. A benchmark study on the thermal conductivity of nanofluids. J. Appl. Phys. 2009, 106. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, Y.; Zhang, S.; Yu, L.; Zhang, P.; Zhang, Z. Preparation of nickel-based nanolubricants via a facile in situ one-step route and investigation of their tribological properties. Tribol. Lett. 2013, 51, 73–83. [Google Scholar] [CrossRef]
- Ji, X.; Chen, Y.; Zhao, G.; Wang, X.; Liu, W. Tribological properties of CaCO3 nanoparticles as an additive in lithium grease. Tribol. Lett. 2011, 41, 113–119. [Google Scholar] [CrossRef]
- Friedman, H.; Eidelman, O.; Feldman, Y.; Moshkovich, A.; Perfiliev, V.; Rapoport, L.; Cohen, H.; Yoffe, A.; Tenne, R. Fabrication of self-lubricating cobalt coatings on metal surfaces. Nanotechnology 2007, 18. [Google Scholar] [CrossRef]
- Chaudhury, M.K. Complex fluids: Spread the word about nanofluids. Nature 2003, 423, 131–132. [Google Scholar] [CrossRef]
- Elechiguerra, J.L.; Reyes-Gasga, J.; Yacaman, M.J. The role of twinning in shape evolution of anisotropic noble metal nanostructures. J. Mater. Chem. 2006, 16, 3906–3919. [Google Scholar] [CrossRef]
- Kleinstreuer, C.; Li, J.; Koo, J. Microfluidics of nano-drug delivery. Int. J. Heat Mass Transfer 2008, 51, 5590–5597. [Google Scholar] [CrossRef]
- Rapoport, L.; Fleischer, N.; Tenne, R. Fullerene-like WS2 nanoparticles: Superior lubricants for harsh conditions. Adv. Mater. 2003, 15, 651–655. [Google Scholar] [CrossRef]
- Cumings, J.; Zettl, A. Low-friction nanoscale linear bearing realized from multiwall carbon nanotubes. Science 2000, 289, 602–604. [Google Scholar] [CrossRef]
- Falvo, M.R.; Taylor, R.M., II; Helser, A.; Chi, V.; Brooks, F.P., Jr.; Washburn, S.; Superfine, R. Nanometre-scale rolling and sliding of carbon nanotubes. Nature 1999, 397, 236–238. [Google Scholar] [CrossRef]
- Akbulut, M.; Belman, N.; Golan, Y.; Israelachvili, J. Frictional properties of confined nanorods. Adv. Mater. 2006, 18, 2589–2592. [Google Scholar] [CrossRef]
- Peng, D.X.; Kang, Y.; Hwang, R.M.; Shyr, S.S.; Chang, Y.P. Tribological properties of diamond and SiO2 nanoparticles added in paraffin. Tribol. Int. 2009, 42, 911–917. [Google Scholar] [CrossRef]
- Rapoport, L.; Leshchinsky, V.; Lvovsky, M.; Nepomnyashchy, O.; Volovik, Y.; Tenne, R. Mechanism of friction of fullerenes. Ind. Lubr. Tribol. 2002, 54, 171–176. [Google Scholar] [CrossRef]
- Tao, X.; Jiazheng, Z.; Kang, X. The ball-bearing effect of diamond nanoparticles as an oil additive. J. Phys. D: Appl. Phys. 1996, 29, 2932–2937. [Google Scholar] [CrossRef]
- Zhou, J.; Yang, J.; Zhang, Z.; Liu, W.; Xue, Q. Study on the structure and tribological properties of surface-modified Cu nanoparticles. Mater. Res. Bull. 1999, 34, 1361–1367. [Google Scholar] [CrossRef]
- Liu, G.; Li, X.; Qin, B.; Xing, D.; Guo, Y.; Fan, R. Investigatoin of the mending effect and mechanism of copper nano-particles on a tribologically stressed surface. Tribol. Lett. 2004, 17, 961–966. [Google Scholar] [CrossRef]
- Lee, K.; Hwang, Y.; Cheong, S.; Choi, Y.; Kwon, L.; Lee, J.; Kim, S.H. Understanding the role of nanoparticles in nano-oil lubrication. Tribol. Lett. 2009, 35, 127–131. [Google Scholar] [CrossRef]
- Pilkington, G.A.; Briscoe, W.H. Nanofluids mediating surface forces. Adv. Colloid Interface Sci. 2012, 179–182, 68–84. [Google Scholar] [CrossRef]
- Daniel, M.-C.; Astruc, D. Gold nanoparticles: Assembly, supramolecular chemistry, quantum-size-related properties, and applications toward biology, catalysis, and nanotechnology. Chem. Rev. 2004, 104, 293–346. [Google Scholar] [CrossRef]
- Zhang, B.-S.; Xu, B.-S.; Xu, Y.; Gao, F.; Shi, P.-J.; Wu, Y.-X. Cu nanoparticles effect on the tribological properties of hydrosilicate powders as lubricant additive for steel-steel contacts. Tribol. Int. 2011, 44, 878–886. [Google Scholar] [CrossRef]
- Hernández Battez, A.; González, R.; Viesca, J.L.; Fernández, J.E.; Díaz Fernández, J.M.; Machado, A.; Chou, R.; Riba, J. CuO, ZrO2 and ZnO nanoparticles as antiwear additive in oil lubricants. Wear 2008, 265, 422–428. [Google Scholar] [CrossRef]
- Guerrero-Martínez, A.; Pérez-Juste, J.; Liz-Marzán, L.M. Recent progress on silica coating of nanoparticles and related nanomaterials. Adv. Mater. 2010, 22, 1182–1195. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, L.; Wan, H.; Chen, J.; Zhou, H. Synthesis and tribological properties of stearic acid-modified anatase (TiO2) nanoparticles. Tribol. Lett. 2011, 41, 409–416. [Google Scholar] [CrossRef]
- Fan, Z.; Lu, J.G. Zinc oxide nanostructures: Synthesis and properties. J. Nanosci. Nanotechnol. 2005, 5, 1561–1573. [Google Scholar] [CrossRef]
- Qiu, S.; Dong, J.; Chen, G. Tribological properties of CeF3 nanoparticles as additives in lubricating oils. Wear 1999, 230, 35–38. [Google Scholar] [CrossRef]
- Rapoport, L.; Feldman, Y.; Homyonfer, M.; Cohen, H.; Sloan, J.; Hutchison, J.L.; Tenne, R. Inorganic fullerene-like material as additives to lubricants: Structure-function relationship. Wear 1999, 225, 975–982. [Google Scholar]
- Chiñas-Castillo, F.; Spikes, H.A. The behavior of colloidal solid particles in elastohydrodynamic contacts. Tribol. Trans. 2000, 43, 387–394. [Google Scholar] [CrossRef]
- Stachowiak, G.W.; Batchelor, A.W.; Stachowiak, G.B. Experimental Methods in Tribology; Elsevier: Amsterdam, The Netherlands, 2004; Volume 44. [Google Scholar]
- Martin, J.M.; Ohmae, N. Nanolubricants; John Wiley & Sons, Ltd.: Chichester, UK, 2008. [Google Scholar]
- Akbulut, M. Nanoparticle-based lubrication systems. J. Powder Metall. Min. 2012, 1. [Google Scholar] [CrossRef]
- Spalla, O.; Kékicheff, P. Adhesion between oxide nanoparticles: Influence of surface complexation. J. Colloid Interface Sci. 1997, 192, 43–65. [Google Scholar] [CrossRef]
- Özerinç, S.; Kakaç, S.; Yazicioglu, A.G. Enhanced thermal conductivity of nanofluids: A state-of-the-art review. Microfluid. Nanofluid. 2010, 8, 145–170. [Google Scholar] [CrossRef]
- Kalin, M.; Roman, E.; Vižintin, J. The effect of temperature on the tribological mechanisms and reactivity of hydrogenated, amorphous diamond-like carbon coatings under oil-lubricated conditions. Thin Solid Films 2007, 515, 3644–3652. [Google Scholar] [CrossRef]
- Alves, S.M.; Barros, B.S.; Trajano, M.F.; Ribeiro, K.S.B.; Moura, E. Tribological behavior of vegetable oil-based lubricants with nanoparticles of oxides in boundary lubrication conditions. Tribol. Int. 2013, 65, 28–36. [Google Scholar] [CrossRef]
- Wu, Y.Y.; Tsui, W.C.; Liu, T.C. Experimental analysis of tribological properties of lubricating oils with nanoparticle additives. Wear 2007, 262, 819–825. [Google Scholar] [CrossRef]
- Rapoport, L.; Lvovsky, M.; Lapsker, I.; Leshchinsky, W.; Volovik, Y.; Feldman, Y.; Tenne, R. Friction and wear of bronze powder composites including fullerene-like WS2 nanoparticles. Wear 2001, 249, 150–157. [Google Scholar]
- Hu, Z.S.; Dong, J.X. Study on Antiwear and Reducing Friction Additives of Nanometer Titanium Oxide. Wear 1998, 216, 92–96. [Google Scholar] [CrossRef]
- Kim, D.; Archer, L.A. Nanoscale organic-inorganic hybrid lubricants. Langmuir 2011, 27, 3083–3094. [Google Scholar] [CrossRef]
- Bhushan, B.; Gupta, B.K.; van Cleef, G.W.; Capp, C.; Coe, J.V. Fullerene (C60) films for solid lubrication. Tribol. Trans. 1993, 36, 573–580. [Google Scholar] [CrossRef]
- Israelachvili, J.; Maeda, N.; Rosenberg, K.J.; Akbulut, M. Effects of sub-angstrom (pico-scale) structure of surfaces on adhesion, friction, and bulk mechanical properties. J. Mater. Res. 2005, 20, 1952–1972. [Google Scholar] [CrossRef]
- Xue, Q.; Liu, W.; Zhang, Z. Friction and wear properties of a suface-modified TiO2 nanoparticle as an additive in liquid paraffin. Wear 1997, 213, 29–32. [Google Scholar] [CrossRef]
- Chiñas-Castillo, F.; Spikes, H.A. Mechanism of action of colloidal solid dispersions. J. Tribol. Trans. ASME 2003, 125, 552–557. [Google Scholar] [CrossRef]
- Rapoport, L.; Leshchinsky, V.; Lapsker, I.; Volovik, Y.; Nepomnyashchy, O.; Lvovsky, M.; Popovitz-Biro, R.; Feldman, Y.; Tenne, R. Tribological properties of WS2 nanoparticles under mixed lubrication. Wear 2003, 255, 785–793. [Google Scholar] [CrossRef]
- Rapoport, L.; Nepomnyashchy, O.; Lapsker, I.; Verdyan, A.; Moshkovich, A.; Feldman, Y.; Tenne, R. Behaviour of fullerene-like WS2 nanoparticles under severe contact conditions. Wear 2005, 259, 703–707. [Google Scholar] [CrossRef]
- Savage, R.H. Graphite lubrication. J. Appl. Phys. 1948, 19, 1–10. [Google Scholar] [CrossRef]
- Huang, H.D.; Tu, J.P.; Gan, L.P.; Li, C.Z. An investigation on tribological properties of graphite nanosheets as oil additive. Wear 2006, 261, 140–144. [Google Scholar] [CrossRef]
- Bay, N.; Nakamura, T.; Schmid, S. Green Lubricants for Metal Forming. In Proceedings of the International Conference on Tribology in Manufacturing Processes, Nice, France, 13–15 June 2010; pp. 5–28.
- Moustafa, S.F.; El-Badry, S.A.; Sanad, A.M.; Kieback, B. Friction and wear of copper-graphite composites made with Cu-coated and uncoated graphite powders. Wear 2002, 253, 699–710. [Google Scholar] [CrossRef]
- Liu, Y.B.; Lim, S.C.; Ray, S.; Rohatgi, P.K. Friction and wear of aluminium-graphite composites: The smearing process of graphite during sliding. Wear 1992, 159, 201–205. [Google Scholar] [CrossRef]
- Rohatgi, P.K.; Ray, S.; Liu, Y. Tribological properties of metal matrix-graphite particle composites. Int. Mater. Rev. 1992, 37, 129–149. [Google Scholar] [CrossRef]
- Buldum, A.; Lu, J.P. Atomic scale sliding and rolling of carbon nanotubes. Phys. Rev. Lett. 1999, 83. [Google Scholar] [CrossRef]
- Sinnott, S.B.; Andrews, R. Carbon nanotubes: Synthesis, properties, and applications. Crit. Rev. Solid State Mater. Sci. 2001, 26, 145–249. [Google Scholar] [CrossRef]
- Chen, W.X.; Tu, J.P.; Wang, L.Y.; Gan, H.Y.; Xu, Z.D.; Zhang, X.B. Tribological application of carbon nanotubes in a metal-based composite coating and composites. Carbon 2003, 41, 215–222. [Google Scholar] [CrossRef]
- Shi, L.; Sun, C.; Gao, P.; Zhou, F.; Liu, W. Mechanical properties and wear and corrosion resistance of electrodeposited Ni-Co/SiC nanocomposite coating. Appl. Surf. Sci. 2006, 252, 3591–3599. [Google Scholar] [CrossRef]
- Yao, Y.; Yao, S.; Zhang, L.; Wang, H. Electrodeposition and mechanical and corrosion resistance properties of Ni-W/SiC nanocomposite coatings. Mater. Lett. 2007, 61, 67–70. [Google Scholar] [CrossRef]
- Chen, W.X.; Li, F.; Han, G.; Xia, J.B.; Wang, L.Y.; Tu, J.P.; Xu, Z.D. Tribological behavior of carbon-nanotube-filled PTFE composites. Tribol. Lett. 2003, 15, 275–278. [Google Scholar] [CrossRef]
- Joly-Pottuz, L.; Dassenoy, F.; Vacher, B.; Martin, J.M.; Mieno, T. Ultralow friction and wear behaviour of Ni/Y-based single wall carbon nanotubes (SWNTs). Tribol. Int. 2004, 37, 1013–1018. [Google Scholar] [CrossRef]
- Vander Wal, R.L.; Miyoshi, K.; Street, K.W.; Tomasek, A.J.; Peng, H.; Liu, Y.; Margrave, J.L.; Khabashesku, V.N. Friction properties of surface-fluorinated carbon nanotubes. Wear 2005, 259, 738–743. [Google Scholar] [CrossRef]
- Joly-Pottuz, L.; Ohmae, N. Carbon-Based Nanolubricants. In Nanolubricants; Martin, J.M., Ohmae, N., Eds.; John Wiley & Sons: Chichester, UK, 2008; pp. 93–147. [Google Scholar]
- Tasis, D.; Tagmatarchis, N.; Bianco, A.; Prato, M. Chemistry of carbon nanotubes. Chem. Rev. 2006, 106, 1105–1136. [Google Scholar] [CrossRef]
- Bonhôte, P.; Dias, A.-P.; Papageorgiou, N.; Kalyanasundaram, K.; Grätzel, M. Hydrophobic, highly conductive ambient-temperature molten salts. Inorg. Chem. 1996, 35, 1168–1178. [Google Scholar] [CrossRef]
- Swatloski, R.P.; Visser, A.E.; Reichert, W.M.; Broker, G.A.; Farina, L.M.; Holbrey, J.D.; Rogers, R.D. On the solubilization of water with ethanol in hydrophobic hexafluorophosphate ionic liquids. Green Chem. 2002, 4, 81–87. [Google Scholar] [CrossRef]
- Liu, Y.; Yu, L.; Zhang, S.; Yuan, J.; Shi, L.; Zheng, L. Dispersion of multiwalled carbon nanotubes by ionic liquid-type gemini imidazolium surfactants in aqueous solution. Colloids Surf. A 2010, 359, 66–70. [Google Scholar] [CrossRef]
- Wang, B.; Wang, X.; Lou, W.; Hao, J. Rheological and tribological properties of ionic liquid-based nanofluids containing functionalized multi-walled carbon nanotubes. J. Phys. Chem. C 2010, 114, 8749–8754. [Google Scholar] [CrossRef]
- Peng, Y.; Hu, Y.; Wang, H. Tribological behaviors of surfactant-functionalized carbon nanotubes as lubricant additive in water. Tribol. Lett. 2007, 25, 247–253. [Google Scholar] [CrossRef]
- Pei, X.; Xia, Y.; Liu, W.; Yu, B.; Hao, J. polyelectrolyte-grafted carbon nanotubes: Synthesis, reversible phase-transition behavior, and tribological properties as lubricant additives. J. Polym. Sci. Part A: Polym. Chem. 2008, 46, 7225–7237. [Google Scholar] [CrossRef]
- Yu, B.; Liu, Z.; Zhou, F.; Liu, W.; Liang, Y. A novel lubricant additive based on carbon nanotubes for ionic liquids. Mater. Lett. 2008, 62, 2967–2969. [Google Scholar] [CrossRef]
- Zhou, J.; Wu, Z.; Zhang, Z.; Liu, W.; Xue, Q. Tribological behavior and lubricating mechanism of Cu nanoparticles in oil. Tribol. Lett. 2000, 8, 213–218. [Google Scholar] [CrossRef]
- Padgurskas, J.; Rukuiza, R.; Prosyčevas, I.; Kreivaitis, R. Tribological properties of lubricant additives of Fe, Cu and Co nanoparticles. Tribol. Int. 2013, 60, 224–232. [Google Scholar] [CrossRef]
- Choi, Y.; Lee, C.; Hwang, Y.; Park, M.; Lee, J.; Choi, C.; Jung, M. Tribological behavior of copper nanoparticles as additives in oil. Curr. Appl. Phys. 2009, 9, 124–127. [Google Scholar]
- Liu, W.; Wang, X. Nanolubricants Made of Metals. In Nanolubricants; Martin, J.M., Ohmae, N., Eds.; John Wiley & Sons: Chichester, UK, 2008; pp. 93–147. [Google Scholar]
- Zin, V.; Agresti, F.; Barison, S.; Colla, L.; Gondolini, A.; Fabrizio, M. The synthesis and effect of copper nanoparticles on the tribological properties of lubricant oils. IEEE Trans. Nanotechnol. 2013, 12, 751–759. [Google Scholar] [CrossRef]
- Lisiecki, I.; Billoudet, F.; Pileni, M.P. Control of the shape and the size of copper metallic particles. J. Phys. Chem. 1996, 100, 4160–4166. [Google Scholar] [CrossRef]
- Zhang, X.; Yin, H.; Cheng, X.; Hu, H.; Yu, Q.; Wang, A. Effects of various polyoxyethylen sorbitan monooils (tweens) and sodium dodecyl sulfate on reflux synthesis of copper nanoparticles. Mater. Res. Bull. 2006, 41, 2041–2048. [Google Scholar] [CrossRef]
- Parka, B.K.; Jeong, S.; Kim, D.; Moon, J.; Lim, S.; Kim, J.S. Synthesis and size control of monodisperse copper nanoparticles by polypol method. J. Colloid Interface Sci. 2007, 311, 417–424. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, D.; Chen, J.; Zhou, H.; Wan, H. The use of CTAB to control the size of copper nanoparticles and the concentration of alkylthiols on their surfaces. Mater. Sci. Eng. A 2006, 415, 156–161. [Google Scholar] [CrossRef]
- Cheng, X.; Zhang, X.; Yin, H.; Wang, A.; Xu, Y. Modifier effects on chemical reduction synthesis of nanostructured copper. Appl. Surf. Sci. 2006, 253, 2727–2732. [Google Scholar] [CrossRef]
- Li, B.; Wang, X.; Liu, W.; Xue, Q. Tribochemistry and antiwear mechanism of organic-inorganic nanoparticles as lubricant additives. Tribol. Lett. 2006, 22, 79–84. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, X.; Liu, W.; Fu, X. Performance and anti-wear mechanism of Cu nanoparticles as lubricating oil additives. Ind. Lubr. Tribol. 2009, 61, 311–318. [Google Scholar] [CrossRef]
- Li, Z.; Zhu, Y. Surface-modification of SiO2 nanoparticles with oleic acid. Appl. Surf. Sci. 2003, 211, 315–320. [Google Scholar] [CrossRef]
- Gara, L.; Zou, Q. Friction and wear characteristics of oil-based ZnO nanofluids. Tribol. Trans. 2013, 56, 236–244. [Google Scholar] [CrossRef]
- Wan, Y.; Sun, B.; Liu, W.; Qi, C. Tribological performance of fatty acid modification of sol-gel TiO2 coating. J. Sol-Gel Sci. Technol. 2012, 61, 558–564. [Google Scholar] [CrossRef]
- Xu, Y.; Yu, H.; Xu, B.; Wang, X.; Liu, Q. Preparation and tribological properties of surface-coated nano-copper additives. Key Eng. Mater. 2008, 373–374, 580–584. [Google Scholar]
- Viesca, J.L.; Hernández Battez, A.; González, R.; Chou, R.; Cabello, J.J. Antiwear properties of carbon-coated copper nanoparticles used as an additive to a polyalphaolefin. Tribol. Int. 2011, 44, 829–833. [Google Scholar] [CrossRef]
- Yu, W.; Xie, H. A review of nanofluids: Preparation, stability mechanisms, and applications. J. Nanomater. 2012, 2012. [Google Scholar] [CrossRef]
- Gu, C.; Zhu, G.; Li, L.; Tian, X.; Zhu, G. Tribological effects of oxide based nanoparticles in lubricating oils. J. Mar. Sci. Appl. 2009, 8, 71–76. [Google Scholar] [CrossRef]
- Li, X.; Cao, Z.; Zhang, Z.; Dang, H. Surface-modification in situ of Nano-SiO2 and its structure and tribological properties. Appl. Surf. Sci. 2006, 252, 7856–7861. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, Z.; Dang, H. A novel solution route for preparing indium nanoparticles. J. Phys. Chem. B 2003, 107, 7574–7576. [Google Scholar] [CrossRef]
- Klein, J. Hydration lubrication. Friction 2013, 1, 1–23. [Google Scholar] [CrossRef]
- Klein, J.; Kumacheva, E.; Mahalu, D.; Perahia, D.; Fetters, L. Reduction of frictional forces between solid surfaces bearing polymer brushes. Nature 1994, 370, 634–636. [Google Scholar] [CrossRef]
- Raviv, U.; Giasson, S.; Kampf, N.; Gohy, J.F.; Jérôme, R.; Klein, J. Lubrication by charged polymers. Nature 2003, 425, 163–165. [Google Scholar] [CrossRef]
- Raviv, U.; Giasson, S.; Gohy, J.F.; Jérôme, R.; Klein, J. Normal and frictional forces between surfaces bearing polyelectrolyte brushes. Langmuir 2008, 24, 8678–8687. [Google Scholar] [CrossRef]
- Zhang, Z.; Morse, A.J.; Armes, S.P.; Lewis, A.L.; Geoghegan, M.; Leggett, G.J. Effect of brush thickness and solvent composition on the friction force response of poly(2-(methacryloyloxy)ethylphosphorylcholine) brushes. Langmuir 2011, 27, 2514–2521. [Google Scholar]
- Perry, S.S.; Yan, X.; Limpoco, F.T.; Lee, S.; Müller, M.; Spencer, N.D. Tribological properties of poly(l-lysine)-graft-poly(ethylene glycol) films: Influence of polymer architecture and adsorbed conformation. ACS Appl. Mater. Interfaces 2009, 1, 1224–1230. [Google Scholar] [CrossRef]
- Li, A.; Ramakrishna, S.N.; Kooij, E.S.; Espinosa-Marzal, R.M.; Spencer, N.D. Poly(acrylamide) films at the solvent-induced glass transition: Adhesion, tribology, and the influence of crosslinking. Soft Matter 2012, 8, 9092–9100. [Google Scholar] [CrossRef]
- Limpoco, F.T.; Advincula, R.C.; Perry, S.S. Solvent dependent friction force response of polystyrene brushes prepared by surface initiated polymerization. Langmuir 2007, 23, 12196–12201. [Google Scholar] [CrossRef]
- Kobayashi, M.; Terayama, Y.; Hosaka, N.; Kaido, M.; Suzuki, A.; Yamada, N.; Torikai, N.; Ishihara, K.; Takahara, A. Friction behavior of high-density poly(2-methacryloyloxyethyl phosphorylcholine) brush in aqueous media. Soft Matter 2007, 3, 740–746. [Google Scholar] [CrossRef]
- Klein, J.; Kamiyama, Y.; Yoshizawa, H.; Israelachvili, J.N.; Fredrickson, G.H.; Pincus, P.; Fetters, L.J. Lubrication forces between surfaces bearing polymer brushes. Macromolecules 1993, 26, 5552–5560. [Google Scholar] [CrossRef]
- Ishikawa, T.; Kobayashi, M.; Takahara, A. Macroscopic frictional properties of poly(1-(2-methacryloyloxy)ethyl-3-butyl imidazolium bis(trifluoromethanesulfonyl)-imide) brush surfaces in an ionic liquid. ACS Appl. Mater. Interfaces 2010, 2, 1120–1128. [Google Scholar] [CrossRef]
- Bielecki, R.M.; Crobu, M.; Spencer, N.D. Polymer-brush lubrication in oil: Sliding beyond the stribeck curve. Tribol. Lett. 2013, 49, 263–272. [Google Scholar] [CrossRef]
- Espinosa-Marzal, R.M.; Bielecki, R.M.; Spencer, N.D. Understanding the role of viscous solvent confinement in the tribological behavior of polymer brushes: A bioinspired approach. Soft Matter 2013, 9, 10572–10585. [Google Scholar] [CrossRef]
- Goyal, S.; Escobedo, F.A. Structure and transport properties of polymer grafted nanoparticles. J. Chem. Phys. 2011, 135. [Google Scholar] [CrossRef]
- Babu, K.; Dhamodharan, R. Grafting of poly(methyl methacrylate) brushes from magnetite nanoparticles using a phosphonic acid based initiator by ambient temperature atom transfer radical polymerization (ATATRP). Nanoscale Res. Lett. 2008, 3, 109–117. [Google Scholar] [CrossRef]
- Gong, R.; Maclaughlin, S.; Zhu, S. Surface modification of active metals through atom transfer radical polymerization grafting of acrylics. Appl. Surf. Sci. 2008, 254, 6802–6809. [Google Scholar] [CrossRef]
- Amstad, E.; Gillich, T.; Bilecka, I.; Textor, M.; Reimhult, E. Ultrastable iron oxide nanoparticle colloidal suspensions using dispersants with catechol-derived anchor groups. Nano Lett. 2009, 9, 4042–4048. [Google Scholar]
- Chaudhuri, R.G.; Paria, S. Core/shell nanoparticles: Classes, properties, synthesis mechanisms, characterization, and applications. Chem. Rev. 2011, 112, 2373–2433. [Google Scholar] [CrossRef]
- Kalele, S.; Gosavi, S.W.; Urban, J.; Kulkarni, S.K. Nanoshell particles: Synthesis, properties and applications. Curr. Sci. 2006, 91, 1038–1052. [Google Scholar]
- Bharali, D.J.; Klejbor, I.; Stachowiak, E.K.; Dutta, P.; Roy, I.; Kaur, N.; Bergey, E.J.; Prasad, P.N.; Stachowiak, M.K. Organically modified silica nanoparticles: A nonviral vector for in vivo gene delivery and expression in the Brain. Proc. Natl. Acad. Sci. USA 2005, 102, 11539–11544. [Google Scholar] [CrossRef]
- Fernandes, N.J.; Akbarzadeh, J.; Peterlik, H.; Giannelis, E.P. Synthesis and properties of highly dispersed ionic silica-poly(ethylene oxide) nanohybrids. ACS Nano 2013, 7, 1265–1271. [Google Scholar] [CrossRef]
- Kar, M.; Vijayakumar, P.S.; Prasad, B.L.V.; Gupta, S.S. Synthesis and characterization of poly-l-lysine-grafted silica nanoparticles synthesized via NCA polymerization and click chemistry. Langmuir 2010, 26, 5772–5781. [Google Scholar] [CrossRef]
- Zhang, Q.; Archer, L.A. Poly(ethylene oxide)/silica nanocomposites: Structure and rheology. Langmuir 2002, 18, 10435–10442. [Google Scholar] [CrossRef]
- Voevodin, A.A.; Vaia, R.A.; Patton, S.T.; Diamanti, S.; Pender, M.; Yoonessi, M.; Brubaker, J.; Hu, J.-J.; Sanders, J.H.; Phillips, B.S.; et al. Nanoparticle-wetted surfaces for relays and energy transmission contacts. Small 2007, 3, 1957–1963. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, Z.J.; Simionesie, D.; Schaschke, C. Graphite and Hybrid Nanomaterials as Lubricant Additives. Lubricants 2014, 2, 44-65. https://doi.org/10.3390/lubricants2020044
Zhang ZJ, Simionesie D, Schaschke C. Graphite and Hybrid Nanomaterials as Lubricant Additives. Lubricants. 2014; 2(2):44-65. https://doi.org/10.3390/lubricants2020044
Chicago/Turabian StyleZhang, Zhenyu J., Dorin Simionesie, and Carl Schaschke. 2014. "Graphite and Hybrid Nanomaterials as Lubricant Additives" Lubricants 2, no. 2: 44-65. https://doi.org/10.3390/lubricants2020044
APA StyleZhang, Z. J., Simionesie, D., & Schaschke, C. (2014). Graphite and Hybrid Nanomaterials as Lubricant Additives. Lubricants, 2(2), 44-65. https://doi.org/10.3390/lubricants2020044




