Diffusion of Molecular Diagnostic Lung Cancer Tests: A Survey of German Oncologists
Abstract
:1. Introduction
2. Methodology
2.1. Research Design
2.2. Population and Sampling Approach
2.3. Research Implementation and Validation
3. Results
3.1. Qualitative Research Findings
| Factor | Description |
|---|---|
| Work setting | The Key Opinion Leader (KOL) interviews suggested that in Germany the work setting has implications on reimbursement: while the costs of diagnostic tests ordered by office-based oncologists are generally fully reimbursed, the costs of tests ordered by hospital-based oncologists are reimbursed by a lump sum as defined by “diagnosis related groups”. Practice variation studies additionally show that the work setting influences physicians’ work patterns [14]. |
| Trial participations and attitude towards R&D | The KOL interviews pointed out that trial participations are positively correlated with the oncologist’s knowledge and general attitude towards innovations. In this context, KOLs see a cultural issue in the specific case of Germany: whereas in some countries such as the U.S. innovations are generally positively perceived, the German culture does not always appreciate innovative outcomes of medical research and development to the same extent. |
| Infrastructure | A lack of infrastructure can slow down the diffusion of innovations [15]. The KOL interviews highlighted that tests may not always be locally available. |
| Test-pathway strategies | Test-pathway strategies can potentially standardize and simplify the collaboration between oncologists and pathologists by defining the type and sequence of tests which are conducted for all patients. The importance of interdisciplinary collaboration is confirmed in the KOL interviews. |
| Cost reimbursement | A variety of studies discussing the diffusion of molecular diagnostic tests point out that a lack of reimbursement can significantly slow down the diffusion process [16,17,18]. This is confirmed in KOL interviews, highlighting reimbursement as the most relevant factor. |
| Information and knowledge | Existing diffusion studies suggest that a lack of knowledge and information can slow down the diffusion process [7,19]. This confirmed in the KOL interviews emphasizing that there is a lack of knowledge concerning targeted lung cancer therapies and respective diagnostic tests among German oncologists. |
| Complexity and compatibility | Studies on the adoption of molecular diagnostics show that novel diagnostic technologies can complicate patient management and may lead to an information overload [20]. Additionally, the diffusion is faster for technologies which are perceived as ordinary rather than revolutionary and fit in with existing procedures and beliefs. |
| Value of diagnostics | The perceived value of an innovation is an integral part of general diffusion research. An innovation must add value in order to be adopted and the added value must be visible to the adopter [21,22]. |
| Attitude of colleagues | This variable is related to the role of the oncologists’ social context emphasized in existing studies suggesting that physicians who work together as colleagues gradually adapt to each other and thus become alike [14,23]. |
| Consensus among colleagues | This variable is related to the role of the social context [9,14,24] as well as to the factors of evidence and uncertainty [21,25]: an individual who associates a personal risk with an innovation is less likely to adopt it. The risk associated with an innovation results from the uncertainty surrounding its value. A perceived consensus among colleagues reduces uncertainty and the risk associated with an innovation. |
| Strength of evidence | Diagnostics specific diffusion studies [7,16,17] have shown that scientific evidence serves as a major source of value judgment. Yet, clear evidence often does not emerge until an innovation has been introduced and experimented in practice, which can lead to a slow diffusion process as uncertainty persists. This has been confirmed in the KOL interviews. |
| Reliance on biopharmaceutical industry | The influence of marketing and communication activities of biopharmaceutical companies has been identified in the context of diffusion of medical innovation [26,27] and is assumed to positively correlate with the diffusion process. |
3.2. Quantitative Research Findings: Descriptive Analysis
3.2.1. Survey Sample
| Variable | Valid responses | Minimum | Maximum | Mean |
|---|---|---|---|---|
| Diagnostics usage rate | 109 | 0 | 100 | 64.14 |
| Age | 107 | 30 | 75 | 49.45 |
| Gender | 109 | 0 | 1 | 0.89 |
| Work setting | 109 | 0 | 1 | 0.61 |
| Trial participations | 107 | 0 | 432 | 19.85 |
| Infrastructure | 109 | 0 | 1 | 0.93 |
| Test pathway strategies | 109 | 0 | 1 | 0.53 |
| Cost reimbursement | 109 | 1 | 3 | 2.31 |
| Information and knowledge | 109 | 4 | 10 | 8.61 |
| Perceived value of diagnostics | 109 | 2 | 10 | 8.08 |
| Perceived attitude of colleagues | 109 | 3 | 10 | 7.28 |
| Perceived consensus among colleagues | 109 | 2 | 10 | 7.2 |
| Perceived strength of evidence | 109 | 2 | 10 | 7.73 |
| Attitude towards R&D | 109 | 2 | 10 | 7.35 |
| Reliance on biopharmaceutical industry | 109 | 1 | 10 | 5.72 |
3.2.2. Current Testing Practices
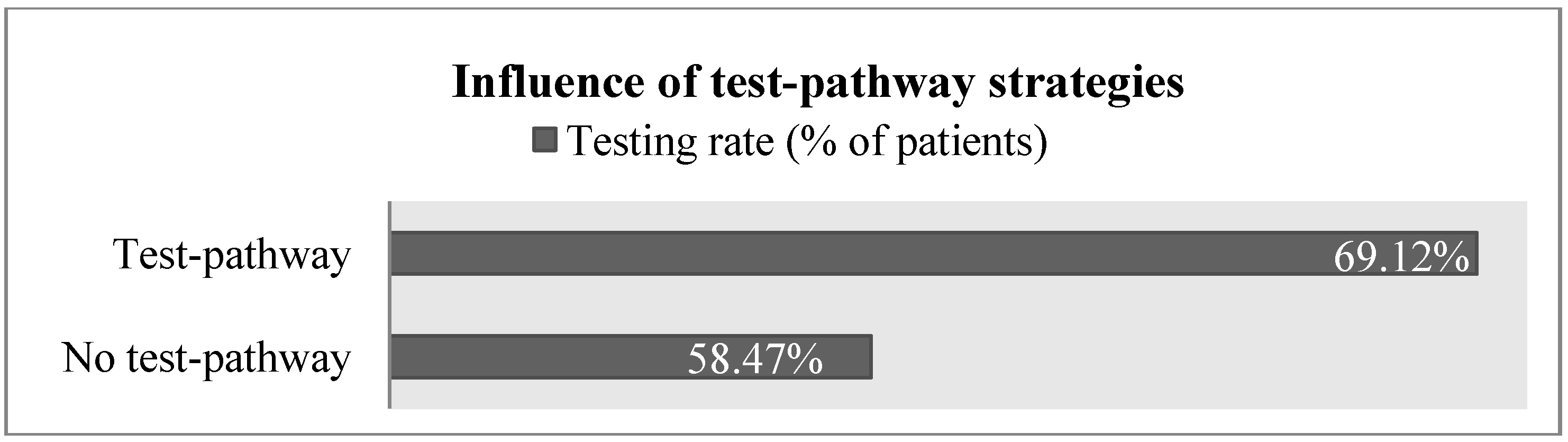
3.2.3. Reimbursement

3.2.4. Perception of Lung Cancer Diagnostics
3.3. Quantitative Research Findings: Multiple Linear Regression
| Variable | Model 1 | Model 2 | Model 3 |
|---|---|---|---|
| R2 = 0.417 | R2 = 0.395 | R2 = 0.393 | |
| Reimbursement | 10.526 *** | 11.467 *** | 12.180 *** |
| 0.305 | 0.329 | 0.35 | |
| Attitude towards R&D | 3.969 *** | 4.534 *** | 4.515 *** |
| 0.265 | 0.302 | 0.301 | |
| Information and knowledge | 4.391 ** | 4.605 ** | 4.605 *** |
| 0.226 | 0.241 | 0.241 | |
| perceived attitude of colleagues | 3.245 * | 3.830 * | 3.945 *** |
| 0.188 | 0.224 | 0.231 | |
| Age | −0.624 ** | −0.679 ** | −0.638 ** |
| −0.171 | −0.176 | −0.178 | |
| Test pathway strategies | 9.641 ** | 10.161 ** | 9.644 ** |
| 0.175 | 0.184 | 0.174 | |
| Work setting | −4.582 | −3.056 | |
| −0.082 | −0.054 | ||
| Gender | −3.889 | −1.083 | |
| −0.045 | −0.012 | ||
| Reliance on biopharmaceutical industry | 1.384 | ||
| 0.108 | |||
| Infrastructure | 7.159 | ||
| 0.069 | |||
| Perceived consensus among colleagues | −0.431 | ||
| −0.033 | |||
| Trial participations | −0.019 | ||
| −0.03 | |||
| Perceived value of diagnostics | 1.159 | ||
| 0.086 | |||
| Perceived strength of evidence | −0.37 | ||
| −0.023 | |||
| Observations | 109 | 109 | 109 |
4. Discussion
4.1. Major Findings and Implications for Clinical Practice
4.2. Limitations, Generalizability and Future Research
5. Conclusions
Acknowledgments
Conflicts of Interest
References and Notes
- Altekruse, S.F.; Kosary, C.L.; Krapcho, M.; Neyman, N.; Aminou, R.; Waldron, W.; Ruhl, J.; Howlader, N.; Tatalovich, Z.; Cho, H.; et al. SEER Cancer Statistics Review. Available online: http://seer.cancer.gov/csr/1975_2007/ (accessed on 12 March 2014).
- Ferlay, J.; Parkin, D.M.; Steliarova-Foucher, E. Estimates of cancer incidence and mortality. Eur. J. Can. 2010, 46, 765–781. [Google Scholar] [CrossRef]
- Paz-Ares, L.; Soulières, D.; Melezinek, I.; Moecks, J.; Keil, L.; Mok, T.; Rosell, R.; Klughammer, B. Clinical outcomes in non-small-cell lung cancer patients with EGFR mutations: Pooled analysis. J. Cell. Mol. Med. 2010, 14, 51–69. [Google Scholar] [CrossRef]
- Zhou, Q.; Zhang, X.C.; Chen, Z.H.; Yin, X.L.; Yang, J.J.; Xu, C.R.; Yan, H.H.; Chen, H.J.; Su, J.; Zhong, W.Z.; et al. Relative abundance of EGFR mutations predicts benefit from gefitinib treatment for advanced non-small-cell lung cancer. J. Clin. Oncol. 2011, 29, 3316–3321. [Google Scholar] [CrossRef]
- Rosell, R.; Bivona, T.G.; Karachaliou, N. Genetics and biomarkers in personalisation of lung cancer treatment. Lancet 2013, 382, 720–731. [Google Scholar]
- Johnson, B.E.; Kris, M.G.; Berry, L.D.; Kwiatkowski, D.J.; Lafrate, A.J.; Varella-Garcia, M.; Wistuba, I.I.; Franklin, W.A.; Ladanyi, M.; Su, P.F.; et al. A multicenter effort to identify driver mutations and employ targeted therapy in patients with lung adenocarcinomas: The Lung Cancer Mutation Consortium (LCMC). J. Clin. Oncol. 2013, 31, 8019. [Google Scholar]
- Lindeman, N.I.; Cagle, P.T.; Beasley, M.B.; Chitale, D.A.; Dacic, S.; Giaccone, G.; Jenkins, R.B.; Kwiatkowski, D.J.; Saldivar, J.S.; Squire, J.; et al. Molecular Testing Guideline for Selection of Lung Cancer Patients for EGFR and ALK Tyrosine Kinase Inhibitors: Guideline from the College of American Pathologists, International Association for the Study of Lung Cancer, and Association for Molecular Pathology. J. Mol. Diagn. 2013, 15, 415–453. [Google Scholar] [CrossRef]
- This is Pfizer internal information which cannot be published openly.
- Creswell, J.W. Research Design; SAGE: Thousand Oaks, CA, USA, 2009. [Google Scholar]
- Organization for Economic Cooperation and Development. OECD Health Data 2013—Frequently Requested Data. 2013. Available online: http://www.oecd.org/els/health-systems/oecdhealthdata2013-frequentlyrequesteddata.htm (accessed on 1 August 2013).
- Federal Health Monitoring. Practitioners with additional postgraduate education. Available online: http://www.gbebund.de/oowa921install/servlet/oowa/aw92/dboowasys921.xwdevkit/ (accessed on 2 June 2013).
- Berufsverband der Niedergelassenen Hämatologen und Onkologen in Deutschland e.V. Arztsuche. Available online: http://www.bnho.de/arztsuche.html (accessed on 10 June 2013).
- German Hospital Directory. Regional Search. Available online: http://www.german-hospital-directory.com/en/159/Search.html (accessed on 8 June 2013).
- Westert, G.P.; Groenevegen, P.P. Medical practice variation: Changing the theoretical approach. Scand. J. Public Health 1999, 27, 173–180. [Google Scholar] [CrossRef]
- Grüber, H.; Verboven, F. The diffusion of mobile telecommunications innovations in the European Union. Eur. Econ. Rev. 2001, 45, 577–588. [Google Scholar] [CrossRef]
- Pletcher, M.J.; Pignone, M. Evaluating the clinical utility of a biomarker: A review for methods estimating health impact. Available online: http://circ.ahajournals.org/content/123/10/1116/ (accessed on 1 August 2013).
- Van Bebber, S.L.; Trosman, J.R.; Liang, S.Y.; Wang, G.; Marshall, D.A.; Knight, S.; Philips, K.A. Capacity building for assessing new technologies: Approaches to examining personalized medicine in practice. Pers. Med. 2010, 7, 427–439. [Google Scholar]
- Engstrom, P.F.; Bloom, M.G.; Demetri, G.D.; Febbo, P.G.; Goeckeler, W.; Ladanyi, M.; Loy, B.; Murphy, K.; Nerenberg, M.; Pagagni, P.; et al. NCCN molecular testing white paper: Effectiveness, efficiency, and reimbursement. J. Nat. Comp. Cancer Net. 2011, 9, S1–S16. [Google Scholar]
- Stanek, E.J.; Sanders, C.L.; Johansen Taber, K.A.; Khalid, A.; Patel, A.; Verbrugge, R.R.; Agatep, B.B.; Aubert, R.E.; Epstein, R.S.; Frueh, F.W. Adoption of pharmacogenomic testing by U.S. physicians results of a nationwide survey. Clin. Pharmacol. Ther. 2012, 91, 450–458. [Google Scholar] [CrossRef]
- Hedgecoe, A. At the point at which you can do something about it, then it becomes more relevant: Informed Consent in the Pharmacogenetic Clinic. Soc. Sci. Med. 2005, 61, 1201–1210. [Google Scholar] [CrossRef]
- Greenhalgh, T.; Robert, G.; Macfarlane, F.; Bate, P.; Kyriakidou, O. Diffusion of innovations in service organisations: Systematic review and recommendations. Milbank Q 2004, 82, 581–629. [Google Scholar] [CrossRef]
- Rogers, E.M. Diffusion of Innovations, 5th ed.; Free Press: New York, NY, USA, 2003. [Google Scholar]
- Coleman, J.S.; Katz, E.; Menzel, H. Medical Innovation; Bobbs-Merrill: New York, NY, USA, 1966. [Google Scholar]
- Burt, R.S. Social contagion and innovation: Cohesion versus structural equivalence. Am. J. Sociol. 1987, 92, 1287–1335. [Google Scholar]
- Denis, J.L.; Hébert, Y.; Langley, A.; Lozeau, D.; Trottier, L.H. Explaining diffusion patterns for complex health care innovations. Health Care Manag. Rev. 2002, 27, 60–73. [Google Scholar] [CrossRef]
- Ratna, N.; Dray, A.; Perez, P. Diffusion and Social Networks: Revisiting Medical Innovation with Agents. In Complex Decision Making: Theory and Practice; Qudrat-Ullah, H., Spector, J.M., Davidsen, P.I., Eds.; Springer: New York, NY, USA, 2008; pp. 247–268. [Google Scholar]
- Van den Bulte, C.; Lilien, G. Medical innovation revisited: Social contagion versus marketing effort. Am. J. Sociol. 2001, 106, 1409–1435. [Google Scholar] [CrossRef]
- De Jong, J. Explaining Medical Practice Variation; NIVEL: Utrecht, The Netherlands, 2008. [Google Scholar]
- Canestaro, W.J.; Martell, L.A.; Wassman, E.R.; Schatzberg, R. Healthcare Payers: A Gate or Translational Bridge to Personalised Medicine? Pers. Med. 2012, 9, 73–84. [Google Scholar] [CrossRef]
- Davis, J.C.; Furstenthal, L.; Desai, A.A.; Norris, T.; Sutaria, S.; Fleming, E.; Ma, P. The microeconomics of personalised medicine: Today’s challenge and tomorrow’s promise. Nat. Rev. Drug Discov. 2009, 8, 279–286. [Google Scholar] [CrossRef]
- Bonter, K.; Desjardins, C.; Currier, N.; Pun, J.; Ashbury, F.D. Personalised medicine in Canada: A survey of adoption and practice in oncology, cardiology and family medicine. Br. Med. J. Open 2011. [Google Scholar] [CrossRef]
- Kadafour, M.; Haugh, R.; Posin, M.; Kayser, S.R.; Shin, J. Survey on warfarin pharmacogenetic testing among anticoagulation providers. Pharmacogenomics 2009, 10, 1853–1860. [Google Scholar] [CrossRef]
- Dacic, S. Molecular genetic testing for lung adenocarcinomas: A practical approach to clinically relevant mutations and translocations. J. Clin. Pathol. 2013, 66, 870–874. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Steffen, J.A. Diffusion of Molecular Diagnostic Lung Cancer Tests: A Survey of German Oncologists. J. Pers. Med. 2014, 4, 102-114. https://doi.org/10.3390/jpm4010102
Steffen JA. Diffusion of Molecular Diagnostic Lung Cancer Tests: A Survey of German Oncologists. Journal of Personalized Medicine. 2014; 4(1):102-114. https://doi.org/10.3390/jpm4010102
Chicago/Turabian StyleSteffen, Julius Alexander. 2014. "Diffusion of Molecular Diagnostic Lung Cancer Tests: A Survey of German Oncologists" Journal of Personalized Medicine 4, no. 1: 102-114. https://doi.org/10.3390/jpm4010102
APA StyleSteffen, J. A. (2014). Diffusion of Molecular Diagnostic Lung Cancer Tests: A Survey of German Oncologists. Journal of Personalized Medicine, 4(1), 102-114. https://doi.org/10.3390/jpm4010102




