Quantitative DNA Methylation Analysis and Epigenotype-Phenotype Correlations in Taiwanese Patients with Beckwith-Wiedemann Syndrome
Abstract
:1. Introduction
2. Patients and Methods
2.1. Patient Selection
2.2. Clinical Assessments
2.3. Molecular Studies
DNA Extraction and Bisulfite Treatment
2.4. Methylation Analysis Using Methylation-Sensitive High-Resolution Melting
2.5. Methylation Analysis Using the MassARRAY EpiTYPER Platform
2.6. Data and Statistical Analysis
3. Discussion
Limitations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Ethics Approval and Consent to Participate
Consent for Publication
Availability of Data and Material
List of Abbreviations
| BWS | Beckwith–Wiedemann syndrome |
| ART | assisted reproductive technology |
| IC | imprinting center |
| pUPD | paternal uniparental disomy |
| UPD | uniparental disomy |
References
- Weksberg, R.; Shuman, C.; Beckwith, J.B. Beckwith-Wiedemann syndrome. Eur. J. Hum. Genet. 2010, 18, 8–14. [Google Scholar] [CrossRef]
- Choufani, S.; Shuman, C.; Weksberg, R. Beckwith-Wiedemann syndrome. Am. J. Med. Genet. C Semin. Med. Genet. 2010, 154, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Elliott, M.; Bayly, R.; Cole, T.; Temple, I.K.; Maher, E.R. Clinical features and natural history of Beckwith-Wiedemann syndrome: Presentation of 74 new cases. Clin. Genet. 1994, 46, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Choufani, S.; Shuman, C.; Weksberg, R. Molecular findings in Beckwith-Wiedemann syndrome. Am. J. Med. Genet. C Semin. Med. Genet. 2013, 163, 131–140. [Google Scholar] [CrossRef]
- Ibrahim, A.; Kirby, G.; Hardy, C.; Dias, R.P.; Tee, L.; Lim, D.; Berg, J.; MacDonald, F.; Nightingale, P.; Maher, E.R. Methylation analysis and diagnostics of Beckwith-Wiedemann syndrome in 1000 subjects. Clin. Epigenetics 2014, 6, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, C.P. Syndromes and disorders associated with omphalocele (I): Beckwith-Wiedemann syndrome. Taiwan J. Obstet. Gynecol. 2007, 46, 96–102. [Google Scholar] [CrossRef] [Green Version]
- Brioude, F.; Lacoste, A.; Netchine, I.; Vazquez, M.P.; Auber, F.; Audry, G.; Gauthier-Villars, M.; Brugieres, L.; Gicquel, C.; Le Bouc, Y.; et al. Beckwith-Wiedemann syndrome: Growth pattern and tumor risk according to molecular mechanism, and guidelines for tumor surveillance. Horm. Res. Pediatrics 2013, 80, 457–465. [Google Scholar] [CrossRef]
- Mussa, A.; Russo, S.; Larizza, L.; Riccio, A.; Ferrero, G.B. (Epi)genotype-phenotype correlations in Beckwith-Wiedemann syndrome: A paradigm for genomic medicine. Clin. Genet. 2016, 89, 403–415. [Google Scholar] [CrossRef]
- Brioude, F.; Kalish, J.M.; Mussa, A.; Foster, A.C.; Bliek, J.; Ferrero, G.B.; Boonen, S.E.; Cole, T.; Baker, R.; Bertoletti, M.; et al. Expert consensus document: Clinical and molecular diagnosis, screening and management of Beckwith-Wiedemann syndrome: An international consensus statement. Nat. Rev. Endocrinol. 2018, 14, 229–249. [Google Scholar] [CrossRef]
- Lin, H.Y.; Chuang, C.K.; Tu, R.Y.; Fang, Y.Y.; Su, Y.N.; Chen, C.P.; Chang, C.Y.; Liu, H.C.; Chu, T.H.; Niu, D.M.; et al. Epigenotype, genotype, and phenotype analysis of patients in Taiwan with Beckwith-Wiedemann syndrome. Mol. Genet. Metab. 2016, 119, 8–13. [Google Scholar] [CrossRef]
- Mussa, A.; Russo, S.; De Crescenzo, A.; Chiesa, N.; Molinatto, C.; Selicorni, A.; Richiardi, L.; Larizza, L.; Silengo, M.C.; Riccio, A.; et al. Prevalence of Beckwith-Wiedemann syndrome in North West of Italy. Am. J. Med. Genet. A 2013, 161, 2481–2486. [Google Scholar] [CrossRef] [PubMed]
- Mussa, A.; Molinatto, C.; Cerrato, F.; Palumbo, O.; Carella, M.; Baldassarre, G.; Carli, D.; Peris, C.; Riccio, A.; Ferrero, G.B. Assisted reproductive techniques and Risk of Beckwith-Wiedemann Syndrome. Pediatrics 2017, 140, e20164311. [Google Scholar] [CrossRef] [Green Version]
- Beckwith, J.B. Extreme Cytomegaly of the Adrenal Fetal Cortex, Omphalocele, Hyperplasia of the Kidneys and Pancreas, and Leydig Cell Hyperplasia–Another Syndrome? In Proceedings of the Annual Meeting of Western Society for Pediatric Research, Los Angeles, CA, USA, 11 November 1963. [Google Scholar]
- Wiedemann, H.R. Familial malformation complex with umbilical hernia and macroglossia—A “new syndrome”? J. Genet. Hum. 1964, 13, 223–232. [Google Scholar] [PubMed]
- Eggermann, T.; Algar, E.; Lapunzina, P.; Mackay, D.; Maher, E.R.; Mannens, M.; Netchine, I.; Prawitt, D.; Riccio, A.; Temple, I.K.; et al. Clinical utility gene card for: Beckwith-Wiedemann Syndrome. Eur. J. Hum. Genet. 2014, 22, 435. [Google Scholar] [CrossRef]
- Zarate, Y.A.; Mena, R.; Martin, L.J.; Steele, P.; Tinkle, B.T.; Hopkin, R.J. Experience with hemihyperplasia and Beckwith-Wiedemann syndrome surveillance protocol. Am. J. Med. Genet. A 2009, 149, 1691–1697. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.H.; Kim, G.H.; Oh, T.J.; Kim, J.H.; Lee, J.J.; Choi, S.H.; Lee, J.Y.; Kim, J.M.; Choi, I.H.; Kim, Y.M.; et al. Quantitative analysis of methylation status at 11p15 and 7q21 for the genetic diagnosis of Beckwith-Wiedemann syndrome and Silver-Russell syndrome. J. Hum. Genet. 2013, 58, 604–610. [Google Scholar] [CrossRef]
- Calvello, M.; Tabano, S.; Colapietro, P.; Maitz, S.; Pansa, A.; Augello, C.; Lalatta, F.; Gentilin, B.; Spreafico, F.; Calzari, L.; et al. Quantitative DNA methylation analysis improves epigenotype-phenotype correlations in Beckwith-Wiedemann syndrome. Epigenetics 2013, 8, 1053–1060. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sasaki, K.; Soejima, H.; Higashimoto, K.; Yatsuki, H.; Ohashi, H.; Yakabe, S.; Joh, K.; Niikawa, N.; Mukai, T. Japanese and North American/European patients with Beckwith-Wiedemann syndrome have different frequencies of some epigenetic and genetic alterations. Eur. J. Hum. Genet. 2007, 15, 1205–1210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engel, J.R.; Smallwood, A.; Harper, A.; Higgins, M.J.; Oshimura, M.; Reik, W.; Schofield, P.N.; Maher, E.R. Epigenotype-phenotype correlations in Beckwith-Wiedemann syndrome. J. Med. Genet. 2000, 37, 921–926. [Google Scholar] [CrossRef] [PubMed]
- Cooper, W.N.; Luharia, A.; Evans, G.A.; Raza, H.; Haire, A.C.; Grundy, R.; Bowdin, S.C.; Riccio, A.; Sebastio, G.; Bliek, J.; et al. Molecular subtypes and phenotypic expression of Beckwith-Wiedemann syndrome. Eur. J. Hum. Genet. 2005, 13, 1025–1032. [Google Scholar] [CrossRef]
- Mussa, A.; Russo, S.; De Crescenzo, A.; Freschi, A.; Calzari, L.; Maitz, S.; Macchiaiolo, M.; Molinatto, C.; Baldassarre, G.; Mariani, M.; et al. (Epi)genotype-phenotype correlations in Beckwith-Wiedemann syndrome. Eur. J. Hum. Genet. 2016, 24, 183–190. [Google Scholar] [CrossRef] [Green Version]
- Zhang, M.; Sun, C.; Liu, R.; Dong, C.; Cheng, R.; Zheng, Z.; Wu, B.; Luo, F.; Pei, Z.; Lu, W. Phenotypes and epigenetic errors in patients with Beckwith-Wiedemann syndrome in China. Transl. Pediatrics 2020, 9, 653–661. [Google Scholar] [CrossRef]
- Wang, R.; Xiao, Y.; Li, D.; Hu, H.; Li, X.; Ge, T.; Yu, R.; Wang, Y.; Zhang, T. Clinical and molecular features of children with Beckwith-Wiedemann syndrome in China: A single-center retrospective cohort study. Ital. J. Pediatrics 2020, 46, 55. [Google Scholar] [CrossRef]
- Ehrich, M.; Nelson, M.R.; Stanssens, P.; Zabeau, M.; Liloglou, T.; Xinarianos, G.; Cantor, C.R.; Field, J.K.; van den Boom, D. Quantitative high-throughput analysis of DNA methylation patterns by base-specific cleavage and mass spectrometry. Proc. Natl. Acad. Sci. USA 2005, 102, 15785–15790. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.; Chang, M.H. New growth charts for Taiwanese children and adolescents based on World Health Organization standards and health-related physical fitness. Pediatrics Neonatol. 2010, 51, 69–79. [Google Scholar] [CrossRef] [Green Version]
- Suchiman, H.E.; Slieker, R.C.; Kremer, D.; Slagboom, P.E.; Heijmans, B.T.; Tobi, E.W. Design, measurement and processing of region-specific DNA methylation assays: The mass spectrometry-based method EpiTYPER. Front. Genet. 2015, 6, 287. [Google Scholar] [CrossRef] [Green Version]
- Jacob, K.J.; Robinson, W.P.; Lefebvre, L. Beckwith-Wiedemann and Silver-Russell syndromes: Opposite developmental imbalances in imprinted regulators of placental function and embryonic growth. Clin. Genet. 2013, 84, 326–334. [Google Scholar] [CrossRef]
- Alders, M.; Bliek, J.; vd Lip, K.; vd Bogaard, R.; Mannens, M. Determination of KCNQ1OT1 and H19 methylation levels in BWS and SRS patients using methylation-sensitive high-resolution melting analysis. Eur. J. Hum. Genet. 2009, 17, 467–473. [Google Scholar] [CrossRef] [Green Version]
- Wojdacz, T.K.; Dobrovic, A.; Algar, E.M. Rapid detection of methylation change at H19 in human imprinting disorders using methylation-sensitive high-resolution melting. Hum. Mutat. 2008, 29, 1255–1260. [Google Scholar] [CrossRef]
- Kalish, J.M.; Conlin, L.K.; Mostoufi-Moab, S.; Wilkens, A.B.; Mulchandani, S.; Zelley, K.; Kowalski, M.; Bhatti, T.R.; Russo, P.; Mattei, P.; et al. Bilateral pheochromocytomas, hemihyperplasia, and subtle somatic mosaicism: The importance of detecting low-level uniparental disomy. Am. J. Med. Genet. A 2013, 161, 993–1001. [Google Scholar] [CrossRef] [Green Version]
- Goldman, M.; Smith, A.; Shuman, C.; Caluseriu, O.; Wei, C.; Steele, L.; Ray, P.; Sadowski, P.; Squire, J.; Weksberg, R.; et al. Renal abnormalities in beckwith-wiedemann syndrome are associated with 11p15.5 uniparental disomy. J. Am. Soc. Nephrol. 2002, 13, 2077–2084. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mussa, A.; Peruzzi, L.; Chiesa, N.; De Crescenzo, A.; Russo, S.; Melis, D.; Tarani, L.; Baldassarre, G.; Larizza, L.; Riccio, A.; et al. Nephrological findings and genotype-phenotype correlation in Beckwith-Wiedemann syndrome. Pediatrics Nephrol. 2012, 27, 397–406. [Google Scholar] [CrossRef]
- Chen, C.P.; Su, Y.N.; Chen, S.U.; Chang, T.Y.; Wu, P.C.; Chern, S.R.; Wu, P.S.; Kuo, Y.L.; Wang, W. Prenatal diagnosis of hypomethylation at KvDMR1 and Beckwith-Wiedemann syndrome in a pregnancy conceived by intracytoplasmic sperm injection and in vitro fertilization and embryo transfer. Taiwan J. Obstet. Gynecol. 2014, 53, 90–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cox, G.F.; Bürger, J.; Lip, V.; Mau, U.A.; Sperling, K.; Wu, B.L.; Horsthemke, B. Intracytoplasmic sperm injection may increase the risk of imprinting defects. Am. J. Hum. Genet. 2002, 71, 162–164. [Google Scholar] [CrossRef] [Green Version]
- Lim, D.; Bowdin, S.C.; Tee, L.; Kirby, G.A.; Blair, E.; Fryer, A.; Lam, W.; Oley, C.; Cole, T.; Brueton, L.A.; et al. Clinical and molecular genetic features of Beckwith-Wiedemann syndrome associated with assisted reproductive technologies. Hum. Reprod. 2009, 24, 741–747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Halliday, J.; Oke, K.; Breheny, S.; Algar, E.; Amor, D.J. Beckwith-Wiedemann syndrome and IVF: A case-control study. Am. J. Hum. Genet. 2004, 75, 526–528. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gicquel, C.; Gaston, V.; Mandelbaum, J.; Siffroi, J.P.; Flahault, A.; Le Bouc, Y. In vitro fertilization may increase the risk of Beckwith-Wiedemann syndrome related to the abnormal imprinting of the KCN1OT gene. Am. J. Hum. Genet. 2003, 72, 1338–1341. [Google Scholar] [CrossRef] [Green Version]
- Maher, E.R.; Brueton, L.A.; Bowdin, S.C.; Luharia, A.; Cooper, W.; Cole, T.R.; Macdonald, F.; Sampson, J.R.; Barratt, C.L.; Reik, W.; et al. Beckwith-Wiedemann syndrome and assisted reproduction technology (ART). J. Med. Genet. 2003, 40, 62–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eggermann, T.; Brioude, F.; Russo, S.; Lombardi, M.P.; Bliek, J.; Maher, E.R.; Larizza, L.; Prawitt, D.; Netchine, I.; Gonzales, M.; et al. Prenatal molecular testing for Beckwith-Wiedemann and Silver-Russell syndromes: A challenge for molecular analysis and genetic counseling. Eur. J. Hum. Genet. 2016, 24, 784–793. [Google Scholar] [CrossRef] [Green Version]

| Clinical Classification | BWS Score (Maximum = 8) | Epigenetic Defects | ||||
|---|---|---|---|---|---|---|
| IC2 Hypomethylation (%) | IC1 Hypermethylation (%) | pUPD (%) | Unknown (%) | Molecular Diagnosis Rate | ||
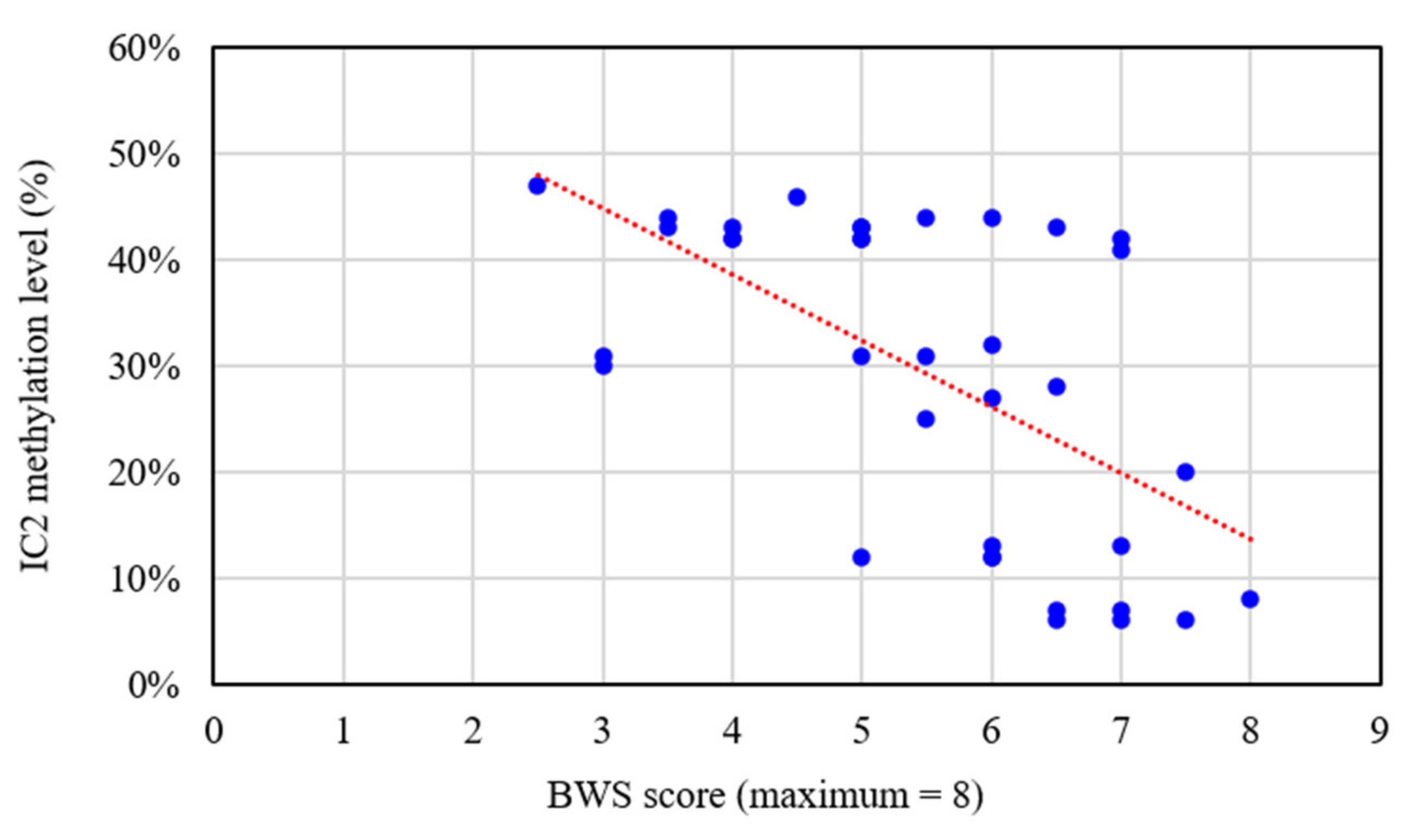
| Clinical diagnosis (n = 36) | 5.5 ±1.4 | 12 (33%) | 2 (5%) | 8 (22%) | 14 (39%) | 61% |
| Suspected BWS (n = 38) | 2.5 ± 1.0 | 6 (16%) | 0 | 1 (3%) | 31 (82%) | 18% |
| All (n = 74) | 4.0 ± 1.9 | 18 (24%) | 2 (3%) | 9 (12%) | 45 (61%) | 39% |
| Only minor criteria (n = 30) | 0.9 ± 0.5 | 1 (3%) | 0 | 1 (3%) | 28 (93%) | 7% |
| Clinical Features | IC2 Hypomethylation (n = 19) | pUPD (n = 10) | p Value | IC1 Hypermethylation (n = 2) |
|---|---|---|---|---|
| BWS score (maximum = 8) | 5.3 ± 1.9 | 4.7 ± 2.0 | 0.646 | 6.5 ± 0.7 |
| Assisted reproductive technology | 3 (16%) | 1 (10%) | 0.681 | 0 |
| Major features | ||||
| Macroglossia | 18 (95%) | 6 (60%) | 0.018 | 2 (100%) |
| Pre- or postnatal gigantism (growth >90th centile) | 11 (58%) | 9 (90%) | 0.081 | 2 (100%) |
| Abdominal wall defects | 11 (58%) | 6 (60%) | 0.917 | 2 (100%) |
| Minor features | ||||
| Ear creases/pits | 11 (58%) | 3 (30%) | 0.164 | 2 (100%) |
| Renal abnormalities | 5 (26%) | 5 (50%) | 0.216 | 2 (100%) |
| Facial naevus flammeus | 10 (53%) | 3 (30%) | 0.260 | 1 (50%) |
| Neonatal hypoglycemia | 5 (26%) | 0 | 0.079 | 0 |
| Hemihypertrophy | 4 (21%) | 9 (90%) | 0.0001 | 0 |
| Congenital cardiac malformations | 5 (26%) | 1 (10%) | 0.32 | 1 (50%) |
| Neoplasia | 1 (5%) | 0 | 0.478 | 0 |
| Moderate or severe mental retardation | 2 (11%) | 0 | 0.305 | 0 |
| Polydactyly | 0 | 0 | — | 0 |
| Cleft palate | 0 | 0 | — | 0 |
| Intra-abdominal visceral organomegaly | 13 (68%) | 6 (60%) | 0.664 | 2 (100%) |
| Major and Minor Features | With or Without Certain Features | N | *Mean IC2 Methylation Level (%) | p Value |
|---|---|---|---|---|
| Major features | ||||
| Macroglossia | With | 18 | 11.1 | 0.005 |
| Without | 1 | 30.0 | ||
| Pre- or postnatal overgrowth (growth >90th centile) | With | 11 | 8.5 | 0.007 |
| Without | 8 | 16.9 | ||
| Abdominal wall defect | With | 11 | 10.5 | 0.258 |
| Without | 8 | 14.3 | ||
| Minor features | ||||
| Ear creases/pits | With | 11 | 9.9 | 0.123 |
| Without | 8 | 15.0 | ||
| Renal abnormalities | With | 5 | 11.2 | 0.763 |
| Without | 14 | 12.4 | ||
| Facial naevus flammeus | With | 10 | 12.0 | 0.974 |
| Without | 9 | 12.1 | ||
| Neonatal hypoglycemia | With | 5 | 10.4 | 0.557 |
| Without | 14 | 12.6 | ||
| Hemihypertrophy | With | 4 | 12.0 | 0.987 |
| Without | 15 | 12.1 | ||
| Congenital cardiac malformations | With | 5 | 12.4 | 0.709 |
| Without | 14 | 11.0 | ||
| Neoplasia | With | 1 | 30.0 | 0.005 |
| Without | 18 | 11.1 | ||
| Moderate/severe mental retardation | With | 2 | 7.0 | 0.297 |
| Without | 17 | 12.6 | ||
| Polydactyly | With | 0 | — | 1.000 |
| Without | 19 | 12.1 | ||
| Cleft palate | With | 0 | — | 1.000 |
| Without | 19 | 12.1 | ||
| Intra-abdominal visceral organomegaly | With | 13 | 11.2 | 0.470 |
| Without | 6 | 13.8 |
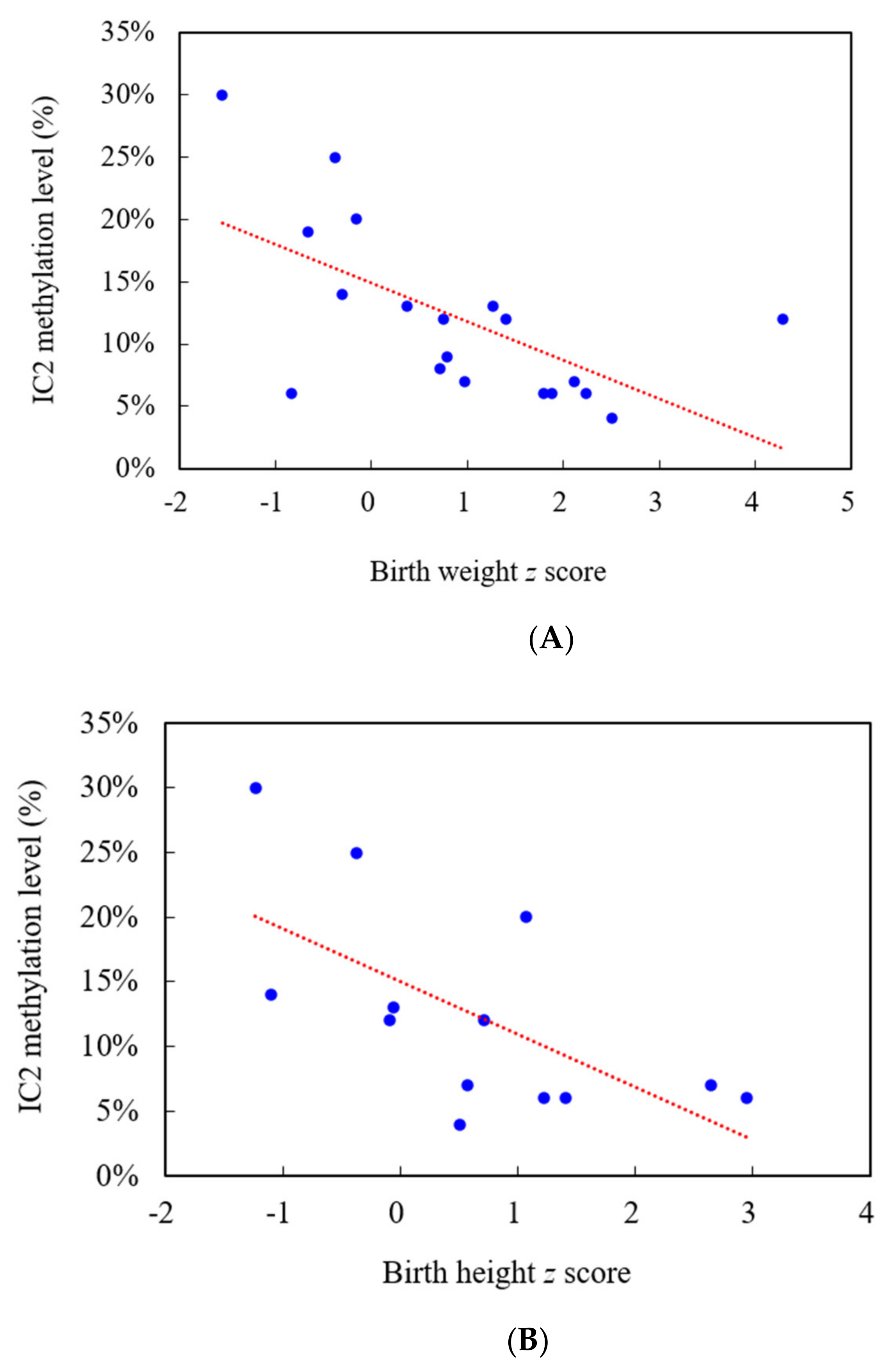
| No. | Gender | Gestational Age (Weeks) | Birth Weight z Score | Birth Height z Score | BWS Score (Maximum = 8) | IC1 Methylation Level * | IC2 Methylation Level ** |
|---|---|---|---|---|---|---|---|
| 1 | F | 32 | 0.71 | NA | 8 | 43% | 8% |
| 2 | M | 33 | 1.79 | 1.22 | 7.5 | 43% | 6% |
| 3 | M | 38 | 2.23 | 2.94 | 7 | 40% | 6% |
| 4 | M | 32 | 0.97 | 0.57 | 6.5 | 39% | 7% |
| 5 | F | 40 | 1.40 | NA | 6 | 36% | 12% |
| 6 | M | 31 | −0.38 | −0.37 | 5.5 | 41% | 25% |
| 7 | F | 39 | 0.75 | 0.71 | 5 | 41% | 12% |
| 8 | F | 36 | −0.31 | −1.11 | 4.5 | 40% | 14% |
| 9 | F | 37 | −0.67 | NA | 3 | 42% | 19% |
| 10 | M | 35 | 0.79 | NA | 4 | 41% | 9% |
| 11 | F | 38 | −1.56 | −1.24 | 1 | 44% | 30% |
| 12 | M | 38 | −0.83 | NA | 2.5 | 43% | 6% |
| 13 | M | 39 | 2.11 | 2.65 | 7 | 40% | 7% |
| 14 | F | 41 | −0.16 | 1.07 | 3.5 | 42% | 20% |
| 15 | F | 38 | 1.27 | −0.06 | 7 | 42% | 13% |
| 16 | M | 35 | 4.28 | −0.10 | 6 | 37% | 12% |
| 17 | M | 36 | 1.88 | 1.40 | 6.5 | 45% | 6% |
| 18 | F | 40 | 2.51 | 0.50 | 3.5 | 43% | 4% |
| 19 | F | 40 | 0.37 | NA | 6 | 42% | 13% |
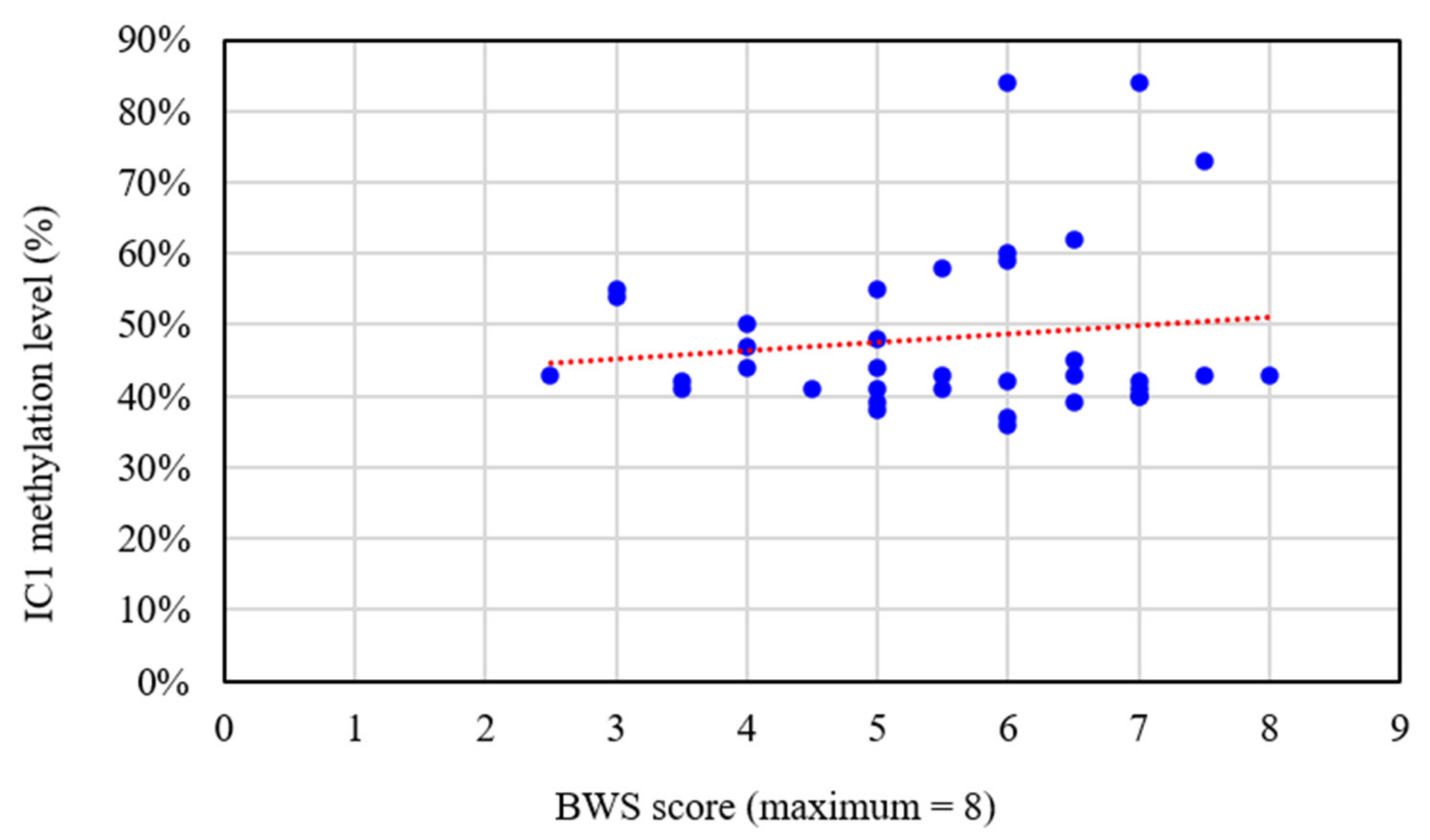
| Major and Minor Features | With or Without Certain Features | N | * Mean IC1 Methylation Level (%) | p Value |
|---|---|---|---|---|
| Major features | ||||
| Macroglossia | With | 31 | 48.2 | 0.945 |
| Without | 5 | 48.6 | ||
| Pre- or postnatal overgrowth (growth >90th centile) | With | 33 | 48.9 | 0.283 |
| Without | 3 | 41.0 | ||
| Abdominal wall defects | With | 29 | 48.7 | 0.638 |
| Without | 7 | 46.3 | ||
| Minor features | ||||
| Ear creases/pits | With | 20 | 47.9 | 0.849 |
| Without | 16 | 48.7 | ||
| Renal abnormalities | With | 17 | 49.6 | 0.519 |
| Without | 19 | 47.0 | ||
| Facial naevus flammeus | With | 14 | 49.4 | 0.795 |
| Without | 19 | 48.3 | ||
| Neonatal hypoglycemia | With | |||
| Without | ||||
| Hemihypertrophy | With | 13 | 52.2 | 0.147 |
| Without | 23 | 46.0 | ||
| Congenital cardiac malformations | With | 12 | 46.2 | 0.472 |
| Without | 24 | 49.3 | ||
| Neoplasia | With | 0 | — | 1.000 |
| Without | 36 | 48.3 | ||
| Moderate/severe mental retardation | With | 4 | 49.0 | 0.319 |
| Without | 32 | 42.5 | ||
| Polydactyly | With | 1 | 41.0 | 0.550 |
| Without | 35 | 48.5 | ||
| Cleft palate | With | 1 | 41.0 | 0.550 |
| Without | 35 | 48.5 | ||
| Intra-abdominal visceral organomegaly | With | 20 | 49.6 | 0.478 |
| Without | 16 | 46.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, H.-Y.; Lee, C.-L.; Fran, S.; Tu, R.-Y.; Chang, Y.-H.; Niu, D.-M.; Chang, C.-Y.; Chiu, P.C.; Chou, Y.-Y.; Hsiao, H.-P.; et al. Quantitative DNA Methylation Analysis and Epigenotype-Phenotype Correlations in Taiwanese Patients with Beckwith-Wiedemann Syndrome. J. Pers. Med. 2021, 11, 1066. https://doi.org/10.3390/jpm11111066
Lin H-Y, Lee C-L, Fran S, Tu R-Y, Chang Y-H, Niu D-M, Chang C-Y, Chiu PC, Chou Y-Y, Hsiao H-P, et al. Quantitative DNA Methylation Analysis and Epigenotype-Phenotype Correlations in Taiwanese Patients with Beckwith-Wiedemann Syndrome. Journal of Personalized Medicine. 2021; 11(11):1066. https://doi.org/10.3390/jpm11111066
Chicago/Turabian StyleLin, Hsiang-Yu, Chung-Lin Lee, Sisca Fran, Ru-Yi Tu, Ya-Hui Chang, Dau-Ming Niu, Chia-Ying Chang, Pao Chin Chiu, Yen-Yin Chou, Hui-Pin Hsiao, and et al. 2021. "Quantitative DNA Methylation Analysis and Epigenotype-Phenotype Correlations in Taiwanese Patients with Beckwith-Wiedemann Syndrome" Journal of Personalized Medicine 11, no. 11: 1066. https://doi.org/10.3390/jpm11111066







