Overview of Five-Years of Experience Performing Non-Invasive Fetal Sex Assessment in Maternal Blood
Abstract
:1. Introduction
2. Experimental Section
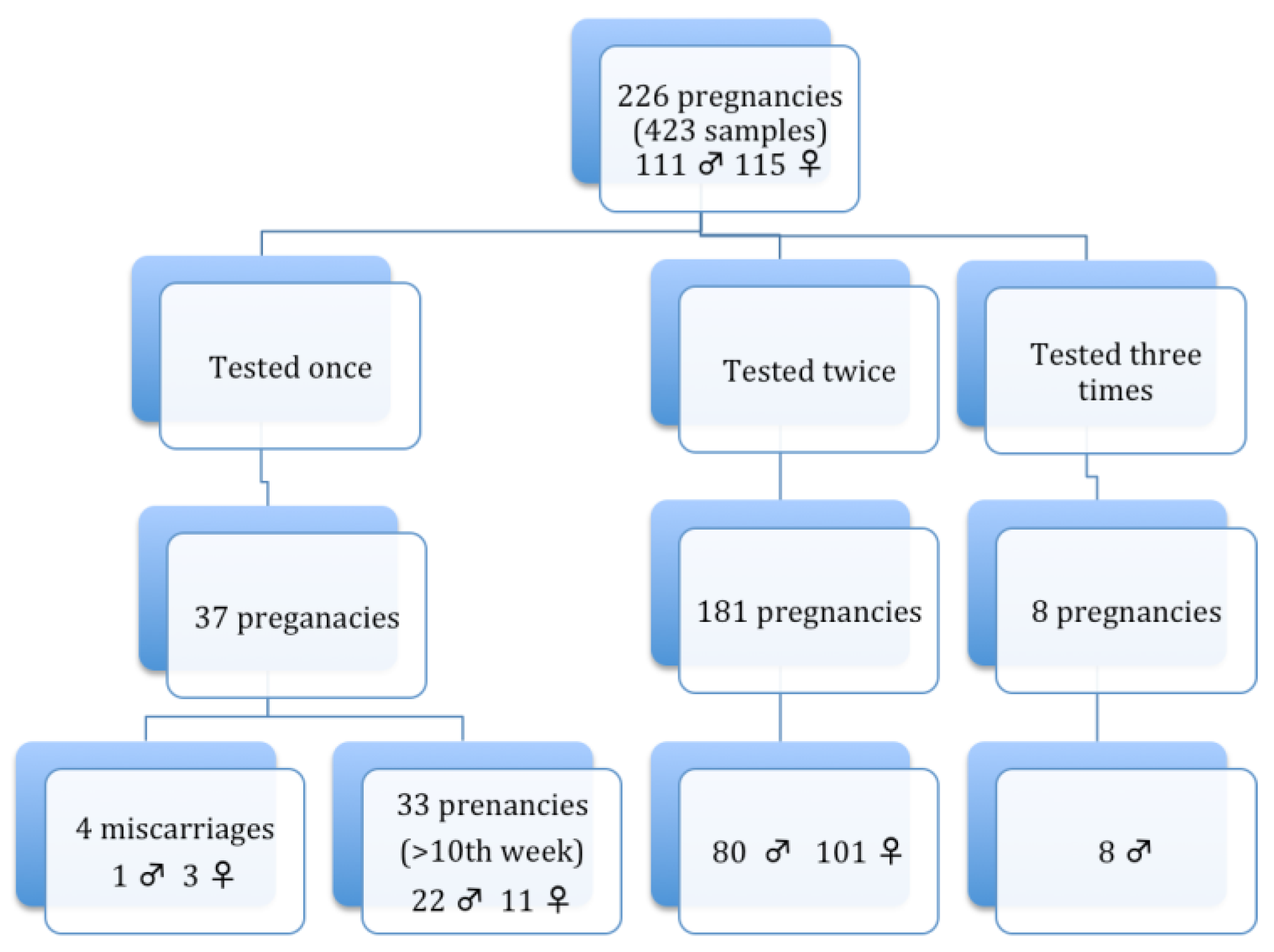
2.1. Sample Collection
| Haemophilia (93) | Incontinentia Pigmenti (2) |
| Duchenne/Becker Muscular Distrophy (42) | X-linked Cardiomyopathy (1) |
| Congenital Adrenal Hyperplasia (9) | X-linked Mental Retardation (1) |
| Hypophosphatemic dysplasia (6) | Morris Syndrome (1) |
| Norrie Syndrome (6) | Ambiguous Genitalia (3) |
| Agammaglobulinemia (6) | Menkes Disease (2) |
| X-linked Retinitis Pigmentosa (6) | X-linked Deafness (1) |
| X-linked inmunodeficiency (5) | Alport Syndrome (1) |
| Fragile X (6) | X-linked Tubular Myopathy (1) |
| PGD Confirmation (5) | Lowe Syndrome (1) |
| Retinoschisis (6) | Bruton Syndrome (1) |
| Kennedy Disease (4) | Androgen insensitivity syndrom (1) |
| X-linked Ocular Albinism (4) | Barth Syndrome (1) |
| Adenoleucodistrophy (4) | X-linked Fabry Disease (1) |
| Hunter Syndrome (5) | Ornitiltranscarbamilasa Deficiency (1) |
2.2. Sample Processing and DNA Extraction
2.3. Real-Time PCR
2.4. Scoring Model
- -
- for positive results (male fetus), only one replicate of the SRY gene is allowed to fail among replicates of both samples. Ct < 38 is considered as positive.
- -
- for negative results (female fetus), only one positive result for 139 the SRY gene is allowed among replicates of both samples.
3. Results and Discussion
3.1. Results
3.2. Discussion
4. Conclusion
Conflict of Interest
Acknowledgements
References
- Lo, Y.M.; Corbetta, N.; Chamberlain, P.F.; Rai, V.; Sargent, I.L.; Redman, C.W.; Wainscoat, J.S. Presence of fetal DNA in maternal plasma and serum. Lancet 1997, 350, 485–487. [Google Scholar] [CrossRef]
- Johnson, K.L.; Dukes, K.A.; Vidaver, J.; LeShane, E.S.; Ramirez, I.; Weber, W.D.; Bischoff, F.Z.; Hahn, S.; Sharma, A.; Dang, D.X.; et al. Interlaboratory comparison of fetal male DNA detection from common maternal plasma samples by real-time PCR. Clin. Chem. 2004, 50, 516–521. [Google Scholar] [CrossRef]
- Ho, S.S.; Damayanti, Z.; Chua, W.Y.; Ng, B.L.; Peh, C.M.; Biswas, A.; Choolani, M. Non-invasive prenatal diagnosis of fetal gender using real-time polymerase chain reaction amplification of SRY in maternal plasma. Ann. Acad. Med. Singap. 2004, 5, S61–S62. [Google Scholar]
- Tachdjian, G.; Frydman, N.; Audibert, F.; Ray, P.; Kerbrat, V.; Ernault, P.; Frydman, R.; Costa, J.M. Clinical applications of fetal sex determination in maternal blood in a preimplantation genetic diagnosis centre. Hum. Reprod. 2002, 17, 2183–2186. [Google Scholar] [CrossRef]
- Bustamante-Aragones, A.; Rodriguez de Alba, M.; Gonzalez-Gonzalez, C.; Trujillo-Tiebas, J.; Diego-Alvarez, D.; Vallespin, E.; Plaza, J.; Ayuso, C.; Ramos, C. Foetal sex determination in maternal blood from the seventh week of gestation and its role in diagnosing haemophilia in the foetuses of female carriers. Haemophilia 2008, 14, 593–598. [Google Scholar] [CrossRef]
- Avent, N.D.; Chitty, L.S. Non-invasive diagnosis of fetal sex; utilisation of free fetal DNA in maternal plasma and ultrasound. Prenat. Diagn. 2006, 26, 598–603. [Google Scholar] [CrossRef]
- Finning, K.M.; Chitty, L.S. Non-invasive fetal sex determination: Impact on clinical practice. Semin. Fetal Neonatal Med. 2008, 13, 69–75. [Google Scholar] [CrossRef]
- Scheffer, P.G.; van der Schoot, C.E.; Page-Christiaens, G.C.; Bossers, B.; van Erp, F.; de Haas, M. Reliability of fetal sex determination using maternal plasma. Obstet. Gynecol. 2010, 115, 117–126. [Google Scholar] [CrossRef]
- Akolekar, R.; Farkas, D.H.; van Agtmael, A.L.; Bombard, A.T.; Nicolaides, K.H. Fetal sex determination using circulating cell-free fetal DNA (ccffDNA) at 11 to 13 weeks of gestation. Prenat. Diagn. 2010, 30, 918–923. [Google Scholar] [CrossRef]
- Miura, K.; Higashijima, A.; Shimada, T.; Miura, S.; Yamasaki, K.; Abe, S.; Jo, O.; Kinoshita, A.; Yoshida, A.; Yoshimura, S.; et al. Clinical application of fetal sex determination using cell-free fetal DNA in pregnant carriers of X-linked genetic disorders. J. Hum. Genet. 2011, 56, 296–299. [Google Scholar] [CrossRef]
- Hill, M.; Finning, K.; Martin, P.; Hogg, J.; Meaney, C.; Norbury, G.; Daniels, G.; Chitty, L.S. Non-invasive prenatal determination of fetal sex: Translating research into clinical practice. Clin. Genet. 2011, 80, 68–75. [Google Scholar] [CrossRef]
- Lim, J.H.; Park, S.Y.; Kim, S.Y.; Kim, D.J.; Choi, J.E.; Kim, M.H.; Choi, J.S.; Kim, M.Y.; Yang, J.H.; Ryu, H.M. Effective detection of fetal sex using circulating fetal DNA in first-trimester maternal plasma. FASEB J. 2012, 26, 250–258. [Google Scholar] [CrossRef]
- Fernández-Martínez, F.J.; Galindo, A.; Garcia-Burguillo, A.; Vargas-Gallego, C.; Nogués, N.; Moreno-García, M.; Moreno-Izquierdo, A. Non inavsive fetal sex determination in maternal plasma: A prospective feasibility study. Genet. Med. 2012, 14, 101–106. [Google Scholar] [CrossRef]
- Kim, S.Y.; Lim, J.H.; Park, S.Y.; Kim, M.Y.; Choi, J.S.; Ryu, H.M. Non-invasive prenatal determination of fetal gender using QF-PCR analysis of cell-free fetal DNA in maternal plasma. Clin. Chim. Acta 2012, 413, 600–604. [Google Scholar] [CrossRef]
- Rong, Y.; Gao, J.; Jiang, X.; Zheng, F. Multiplex PCR for 17 Y-chromosome specific short tandem repeats (STR) to enhance the reliability of fetal sex determination in maternal plasma. Int. J. Mol. Sci. 2012, 13, 5972–5981. [Google Scholar] [CrossRef]
- White, H.E.; Dent, C.L.; Hall, V.J.; Crolla, J.A.; Chitty, L.S. Evaluation of a novel assay for detection of the fetal marker RASSF1A: Facilitating improved diagnostic reliability of non-invasive prenatal diagnosis. PLoS One 2012, 7, e45073. [Google Scholar] [CrossRef]
- Aghanoori, M.R.; Vafaei, H.; Kavoshi, H.; Mohamadi, S.; Goodarzi, H.R. Sex determination using 291 free fetal DNA at early gestational ages: A comparison between modified mini-STR genotyping method and real-time PCR. AJOG 2012, 202, e1–e8. [Google Scholar]
- Kolialexi, A.; Tounta, G.; Apostolou, P.; Vrettou, C.; Papantoniou, N.; Kanavakis, E.; Antsaklis, A.; Mavrou, A. Early non-invasive detection of fetal Y chromosome sequences in maternal plasma using multiplex PCR. Eur. J. Obstet. Gynecol. Reprod. Biol. 2012, 161, 34–37. [Google Scholar] [CrossRef]
- Devaney, S.A.; Palomaki, G.E.; Scott, J.A.; Bianchi, D.W. Noninvasive fetal sex determination using cell-free fetal DNA: A systematic review and meta-analysis. JAMA 2011, 306, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Galbiati, S.; Smid, M.; Gambini, D.; Ferrari, A.; Restagno, G.; Viora, E.; Campogrande, M.; Bastonero, S.; Pagliano, M.; Calza, S.; et al. Fetal DNA detection in maternal plasma throughout gestation. Hum. Genet. 2005, 117, 243–248. [Google Scholar] [CrossRef] [PubMed]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Perlado-Marina, S.; Bustamante-Aragones, A.; Horcajada, L.; Trujillo-Tiebas, M.J.; Lorda-Sanchez, I.; Ruiz Ramos, M.; Plaza, J.; Rodriguez de Alba, M. Overview of Five-Years of Experience Performing Non-Invasive Fetal Sex Assessment in Maternal Blood. Diagnostics 2013, 3, 283-290. https://doi.org/10.3390/diagnostics3020283
Perlado-Marina S, Bustamante-Aragones A, Horcajada L, Trujillo-Tiebas MJ, Lorda-Sanchez I, Ruiz Ramos M, Plaza J, Rodriguez de Alba M. Overview of Five-Years of Experience Performing Non-Invasive Fetal Sex Assessment in Maternal Blood. Diagnostics. 2013; 3(2):283-290. https://doi.org/10.3390/diagnostics3020283
Chicago/Turabian StylePerlado-Marina, Sara, Ana Bustamante-Aragones, Laura Horcajada, Maria Jose Trujillo-Tiebas, Isabel Lorda-Sanchez, Marta Ruiz Ramos, Javier Plaza, and Marta Rodriguez de Alba. 2013. "Overview of Five-Years of Experience Performing Non-Invasive Fetal Sex Assessment in Maternal Blood" Diagnostics 3, no. 2: 283-290. https://doi.org/10.3390/diagnostics3020283




