Plant Growth and Morphogenesis under Different Gravity Conditions: Relevance to Plant Life in Space
Abstract
:1. Introduction
2. Growth and Morphogenesis on a Rotating Clinostat

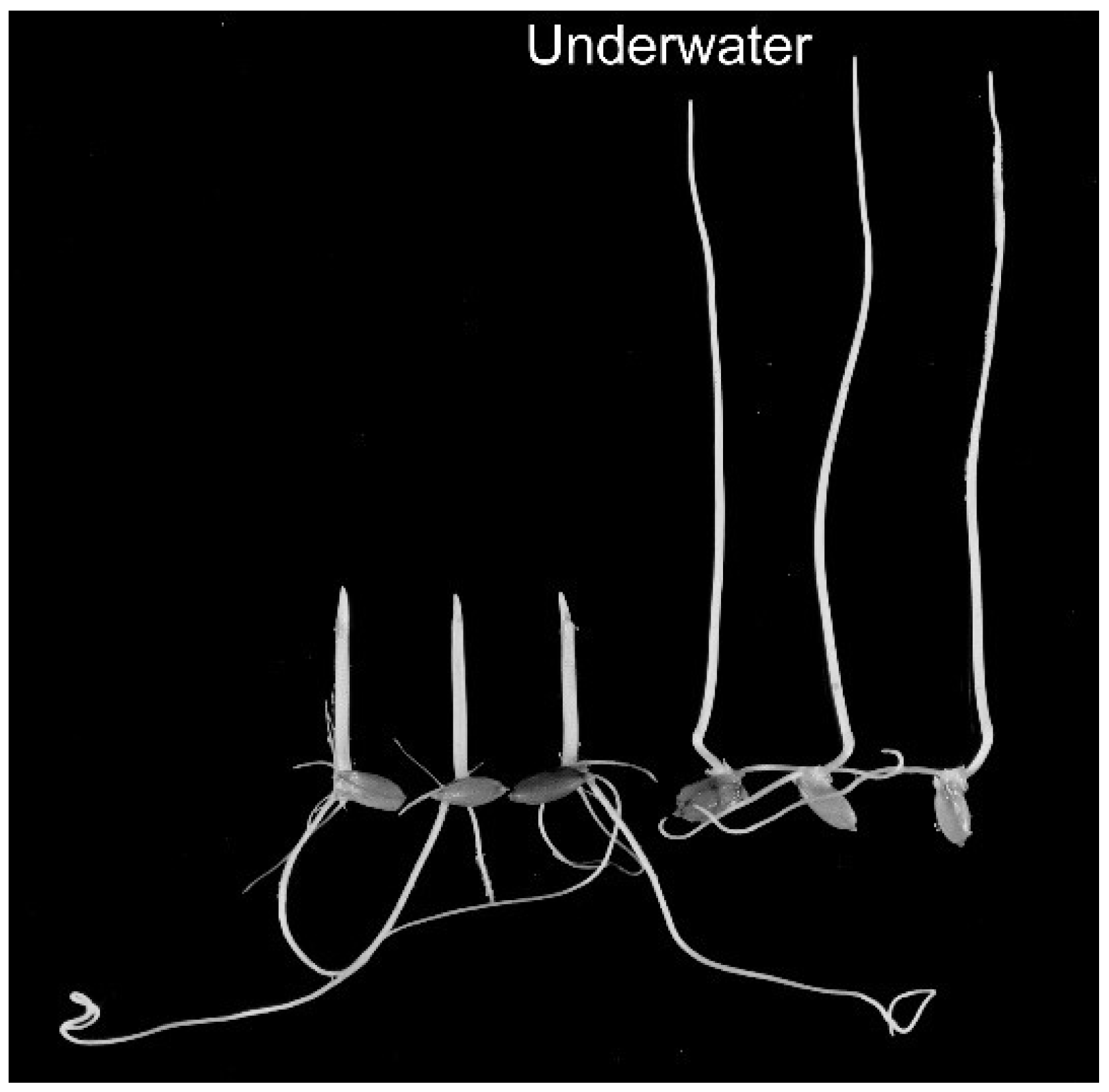
3. Growth and Morphogenesis under Submergence
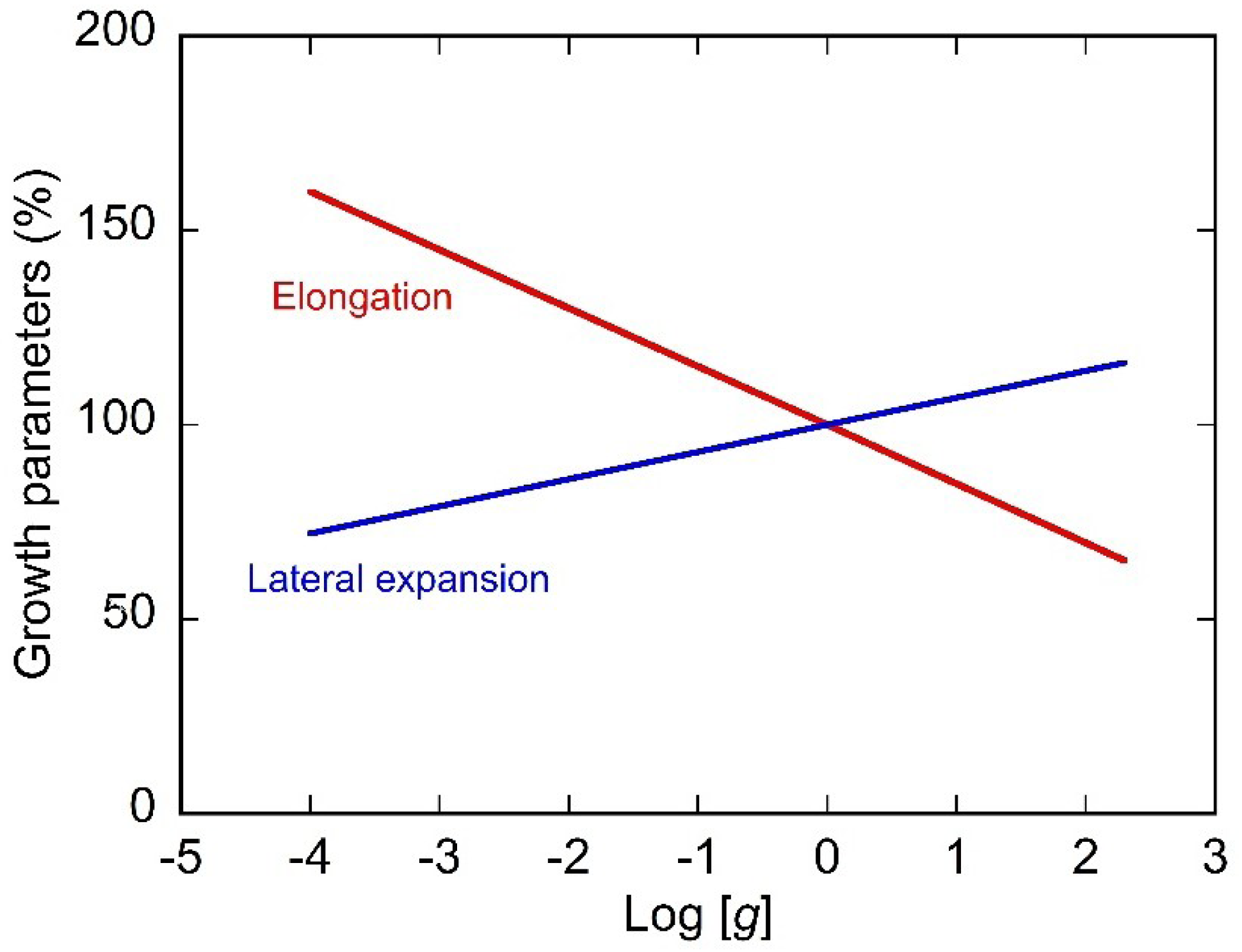
4. Growth and Morphogenesis under Hypergravity Conditions
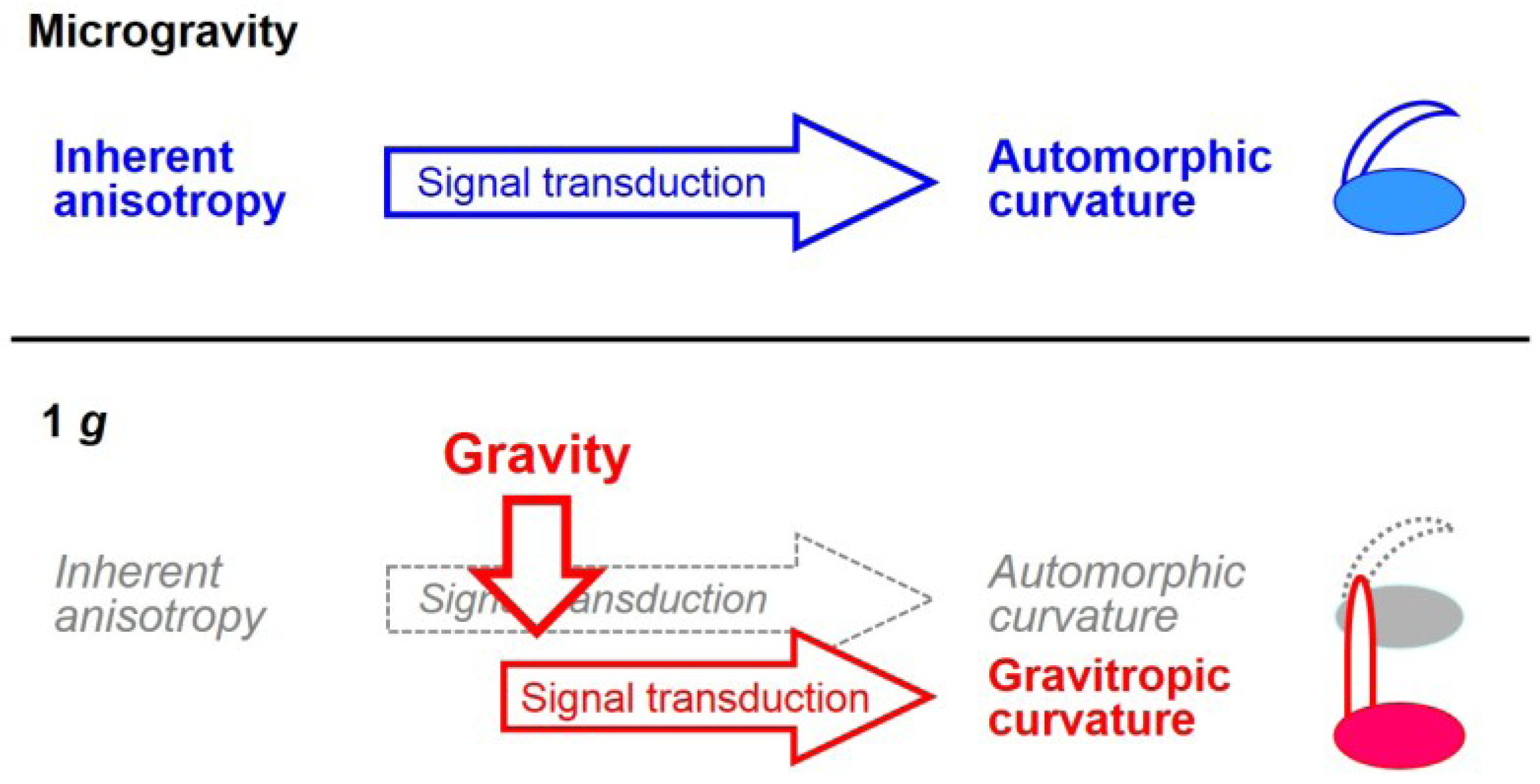
5. Growth and Morphogenesis under Microgravity Conditions in Space
5.1. Growth
5.2. Morphogenesis

6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Hoson, T.; Soga, K. New aspects of gravity responses in plant cells. Int. Rev. Cytol. 2003, 229, 209–244. [Google Scholar] [CrossRef]
- Herranz, R.; Anken, R.; Boonstra, J.; Braun, M.; Christianen, P.C.; de Geest, M.; Hauslage, J.; Hilbig, R.; Hill, R.J.; Lebert, M.; et al. Ground-based facilities for simulation of microgravity: Organism-specific recommendations for their use, and recommended terminology. Astrobiology 2013, 13, 1–17. [Google Scholar]
- Hoson, T.; Kamisaka, S.; Masuda, Y.; Yamashita, M. Changes in plant growth processes under microgravity conditions simulated by a three-dimensional clinostat. Bot. Mag. 1992, 105, 53–70. [Google Scholar] [CrossRef]
- Hensel, W.; Sievers, A. Effects of prolonged omnilateral gravistimulation on the ultrastructure of statocytes and on the graviresponse of roots. Planta 1980, 150, 338–346. [Google Scholar] [CrossRef]
- Hoson, T.; Kamisaka, S.; Masuda, Y.; Yamashita, M.; Buchen, B. Evaluation of the three-dimensional clinostat as a simulator of weightlessness. Planta 1997, 203, S187–S197. [Google Scholar] [CrossRef]
- Stanković, B.; Volkmann, D.; Sack, F.D. Autotropism, automorphogenesis, and gravity. Physiol. Plant. 1998, 102, 328–335. [Google Scholar]
- Hoson, T.; Kamisaka, S.; Buchen, B.; Sievers, A.; Yamashita, M.; Masuda, Y. Possible use of a 3-D clinostat to analyze plant growth processes under simulated microgravity conditions. Adv. Space Res. 1996, 17, 47–53. [Google Scholar]
- Hoson, T. Automorphogenesis of maize roots under simulated microgravity conditions. Plant Soil 1994, 165, 309–314. [Google Scholar] [CrossRef]
- Saiki, M.; Fujita, H.; Soga, K.; Wakabayashi, K.; Kamisaka, S.; Yamashita, M.; Hoson, T. Cellular basis for the automorphic curvature of rice coleoptiles on a three-dimensional clinostat: Possible involvement of reorientation of cortical microtubules. J. Plant Res. 2005, 118, 199–205. [Google Scholar] [CrossRef]
- Ueda, J.; Miyamoto, K.; Uheda, E.; Oka, M.; Yano, S.; Higashibata, A.; Ishioka, N. Close relationships between polar auxin transport and graviresponse in plants. Plant. Biol. 2014, 16, 43–49. [Google Scholar]
- Kutschera, U.; Siebert, C.; Masuda, Y.; Sievers, A. Effects of submergence on development and gravitropism in the coleoptile of Oryza sativa L. Planta 1991, 183, 112–119. [Google Scholar]
- Hoson, T. Apoplast as the site of response to environmental signals. J. Plant. Res. 1998, 111, 167–177. [Google Scholar] [CrossRef]
- Waldron, K.W.; Brett, C.T. Effects of extreme acceleration on the germination growth and cell wall composition of pea epicotyls. J. Exp. Bot. 1990, 41, 71–77. [Google Scholar] [CrossRef]
- Hoson, T.; Nishitani, K.; Miyamoto, K.; Ueda, J.; Kamisaka, S.; Yamamoto, R.; Masuda, Y. Effects of hypergravity on growth and cell wall properties of cress hypocotyls. J. Exp. Bot. 1996, 47, 513–517. [Google Scholar] [CrossRef]
- Soga, K.; Wakabayashi, K.; Kamisaka, S.; Hoson, T. Hypergravity induces reorientation of cortical microtubules and modifies growth anisotropy in azuki bean epicotyls. Planta 2006, 224, 1485–1494. [Google Scholar] [CrossRef]
- Allen, J.; Bisbee, P.A.; Darnell, R.L.; Kuang, A.; Levine, L.H.; Musgrave, M.E.; van Loon, J.J. Gravity control of growth form in Brassica. rapa and Arabidopsis thaliana (Brassicaceae): Consequences for secondary metabolism. Am. J. Bot. 2009, 96, 652–660. [Google Scholar] [CrossRef]
- Hoson, T.; Saito, Y.; Soga, K.; Wakabayashi, K. Signal perception, transduction, and response in gravity resistance. Another graviresponse in plants. Adv. Space Res. 2005, 36, 1196–1202. [Google Scholar] [CrossRef]
- Yoshioka, R.; Soga, K.; Wakabayashi, K.; Takeba, G.; Hoson, T. Hypergravity-induced changes in gene expression in Arabidopsis hypocotyls. Adv. Space Res. 2003, 31, 2187–2193. [Google Scholar] [CrossRef]
- Matsumoto, S.; Saito, Y.; Kumasaki, S.; Soga, K.; Wakabayashi, K.; Hoson, T. Up-regulation of tubulin genes and roles of microtubules in hypergravity-induced growth modifications in Arabidopsis hypocotyls. Adv. Space Res. 2007, 39, 1176–1181. [Google Scholar] [CrossRef]
- Matsumoto, S.; Kumasaki, S.; Soga, K.; Wakabayashi, K.; Hashimoto, T.; Hoson, T. Gravity-induced modifications to development in hypocotyls of Arabidopsis tubulin mutants. Plant Physiol. 2010, 152, 918–926. [Google Scholar] [CrossRef]
- Hoson, T. Interaction of gravity with other environmental factors in growth and development: An introduction. Adv. Space Res. 1999, 23, 1971–1974. [Google Scholar] [CrossRef]
- Kiss, J.Z.; Katembe, W.J.; Edelmann, R.E. Gravitropism and development of wild-type and starch-deficient mutants of Arabidopsis during spaceflight. Physiol. Plant. 1998, 102, 493–502. [Google Scholar]
- Hoson, T.; Soga, K.; Mori, R.; Saiki, M.; Nakamura, Y.; Wakabayashi, K.; Kamisaka, S. Stimulation of elongation growth and cell wall loosening in rice coleoptiles under microgravity conditions in space. Plant Cell Physiol. 2002, 43, 1067–1071. [Google Scholar] [CrossRef]
- Soga, K.; Wakabayashi, K.; Kamisaka, S.; Hoson, T. Stimulation of elongation growth and xyloglucan breakdown in Arabidopsis hypocotyls under microgravity conditions in space. Planta 2002, 215, 1040–1046. [Google Scholar] [CrossRef]
- Matía, I.; González-Camacho, F.; Herranz, R.; Kiss, J.Z.; Gasset, G.; van Loon, J.J.; Marco, R.; Medina, F.J. Plant cell proliferation and growth are altered by microgravity conditions in spaceflight. J. Plant Physiol. 2010, 167, 184–193. [Google Scholar] [CrossRef]
- Hoson, T.; Soga, K.; Wakabayashi, K.; Kamisaka, S.; Tanimoto, E. Growth and cell wall changes in rice roots during spaceflight. Plant Soil 2003, 255, 19–26. [Google Scholar] [CrossRef]
- Hoson, T.; Soga, K.; Wakabayashi, K.; Hashimoto, T.; Karahara, I.; Yano, S.; Tanigaki, F.; Shimazu, T.; Kasahara, H.; Masuda, D.; et al. Growth stimulation in inflorescences of an Arabidopsis tubulin mutant under microgravity conditions in space. Plant Biol. 2014, 16, 91–96. [Google Scholar] [CrossRef]
- Heathcote, D.G.; Chapman, D.K.; Brown, A.H. Nastic curvatures of wheat coleoptiles that develop in true microgravity. Plant Cell Environ. 1995, 18, 818–822. [Google Scholar] [CrossRef]
- Ueda, J.; Miyamoto, K.; Yuda, T.; Hoshino, T.; Fujii, S.; Mukai, C.; Kamigaichi, S.; Aizawa, S.; Yoshizaki, I.; Shimazu, T.; Fukui, K. Growth and development, and auxin polar transport in higher plants under microgravity conditions in space: BRIC-AUX on STS-95 space experiment. J. Plant Res. 1999, 112, 487–492. [Google Scholar] [CrossRef]
- Hoson, T.; Soga, K.; Mori, R.; Saiki, M.; Wakabayashi, K.; Kamisaka, S.; Kamigaichi, S.; Aizawa, S.; Yoshizaki, I.; Shimazu, T.; et al. Morphogenesis of rice and Arabidopsis seedlings in space. J. Plant Res. 1999, 112, 477–486. [Google Scholar] [CrossRef]
- Schulze, A.; Jensen, P.J.; Desrosiers, M.; Buta, J.G.; Bandurski, R.S. Studies on the growth and indole-3-acetic acid and abscisic acid content of Zea. mays seedlings grown in microgravity. Plant Physiol. 1992, 100, 692–698. [Google Scholar] [CrossRef]
- Kiss, J.Z.; Edelmann, R.E.; Wood, P.C. Gravitropism of hypocotyls of wild-type and starch-deficient Arabidopsis seedlings in spaceflight studies. Planta 1999, 209, 96–103. [Google Scholar] [CrossRef]
- Volkmann, D.; Behrens, H.M.; Sievers, A. Development and gravity sensing of cress roots under microgravity. Naturwissenschaften 1986, 73, 438–441. [Google Scholar] [CrossRef]
- Johnsson, A.; Karlsson, C.; Iversen, T.H.; Chapman, D.K. Random root movements in weightlessness. Physiol. Plant. 1996, 96, 169–178. [Google Scholar] [CrossRef]
- Lorenzi, G.; Perbal, G. Root growth and statocyte polarity in lentil seedling roots grown in microgravity or on a slowly rotating clinostat. Physiol. Plant. 1990, 78, 532–537. [Google Scholar] [CrossRef]
- Driss-Ecole, D.; Legue, V.; Carnero-Diaza, E.; Perbal, G. Gravisensitivity and automorphogenesis of lentil seedling roots grown on board the International Space Station. Physiol. Plant. 2008, 134, 191–201. [Google Scholar] [CrossRef]
- Millar, K.D.; Johnson, C.M.; Edelmann, R.E.; Kiss, J.Z. An endogenous growth pattern of roots is revealed in seedlings grown in microgravity. Astrobiology 2011, 11, 787–797. [Google Scholar] [CrossRef]
- Scherer, G.F.E.; Pietrzyk, P. Gravity-dependent differentiation and root coils in Arabidopsis thaliana wild type and phospholipase-A-I knockdown mutant grown on the International Space Station. Plant Biol. 2014, 16, 97–106. [Google Scholar] [CrossRef]
- Nakashima, J.; Liao, F.; Sparks, J.A.; Tang, Y.; Blancaflor, E.B. The actin cytoskeleton is a suppressor of the endogenous skewing behaviour of Arabidopsis primary roots in microgravity. Plant Biol. 2014, 16, 142–150. [Google Scholar] [CrossRef]
- Hoson, T.; Soga, K.; Mori, R.; Saiki, M.; Nakamura, Y.; Wakabayashi, K.; Kamisaka, S. Cell wall changes involved in the automorphic curvature of rice coleoptiles under microgravity conditions in space. J. Plant. Res. 2004, 117, 449–455. [Google Scholar] [CrossRef]
- Kiss, J.Z. Plant biology in reduced gravity on the Moon and Mars. Plant Biol. 2014, 16, 12–17. [Google Scholar] [CrossRef]
- Paul, A.L.; Zupanska, A.K.; Schultz, E.R.; Ferl, R.J. Organ-specific remodeling of the Arabidopsis transcriptome in response to spaceflight. BMC Plant Biol. 2013, 13, 1–11. [Google Scholar] [CrossRef]
- Hoson, T. The mechanism and significance of gravity resistance in plants. J. Gravit. Physiol. 2006, 13, 97–100. [Google Scholar]
- Volkmann, D.; Baluška, F. Gravity: One of the driving forces for evolution. Protoplasma 2006, 229, 143–148. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hoson, T. Plant Growth and Morphogenesis under Different Gravity Conditions: Relevance to Plant Life in Space. Life 2014, 4, 205-216. https://doi.org/10.3390/life4020205
Hoson T. Plant Growth and Morphogenesis under Different Gravity Conditions: Relevance to Plant Life in Space. Life. 2014; 4(2):205-216. https://doi.org/10.3390/life4020205
Chicago/Turabian StyleHoson, Takayuki. 2014. "Plant Growth and Morphogenesis under Different Gravity Conditions: Relevance to Plant Life in Space" Life 4, no. 2: 205-216. https://doi.org/10.3390/life4020205




