Cineradiographic Analysis of Mouse Postural Response to Alteration of Gravity and Jerk (Gravity Deceleration Rate)
Abstract
:1. Introduction
2. Experimental Section
2.1. Animals
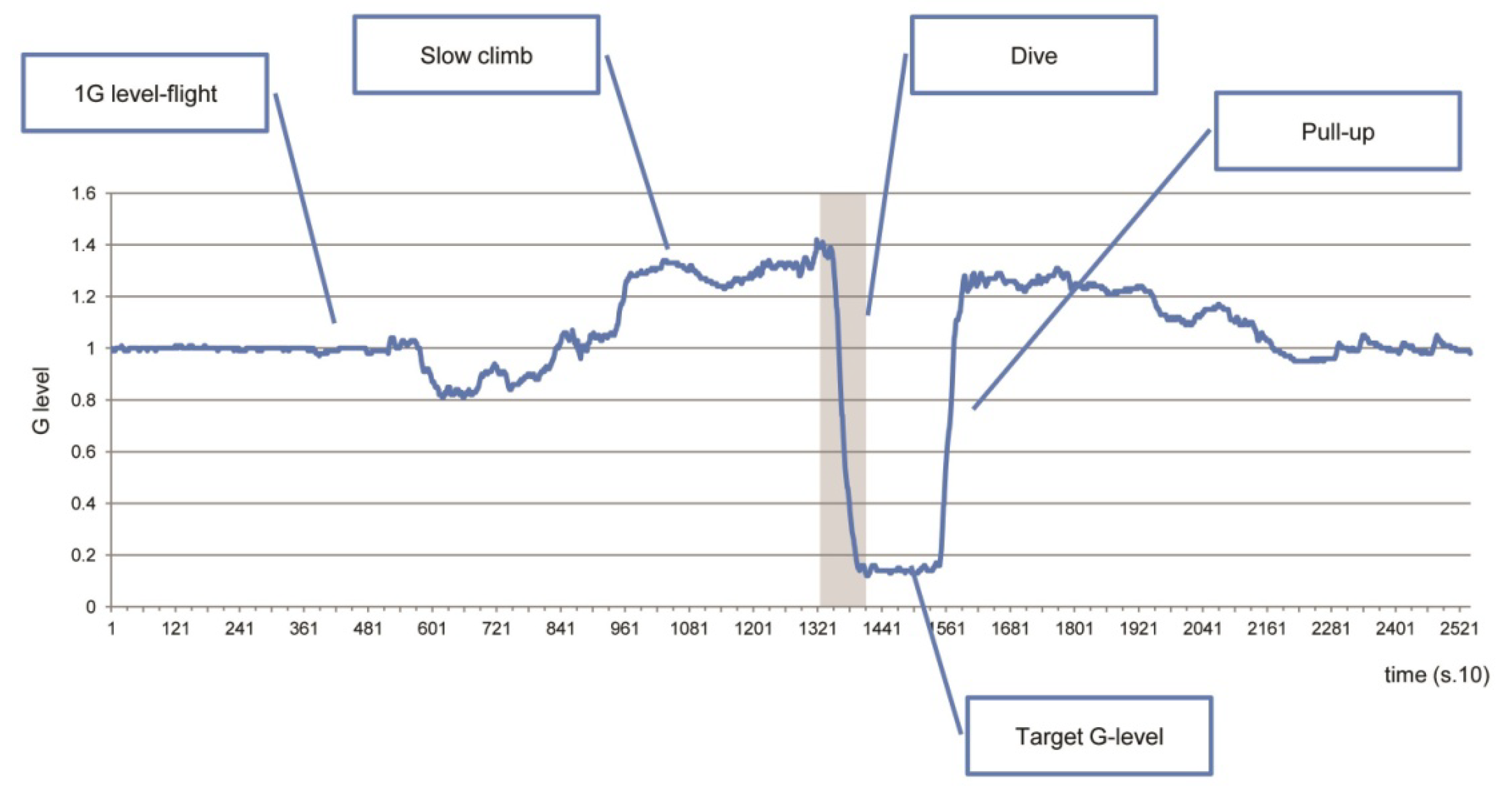
2.2. Parabolic-Flight Protocol
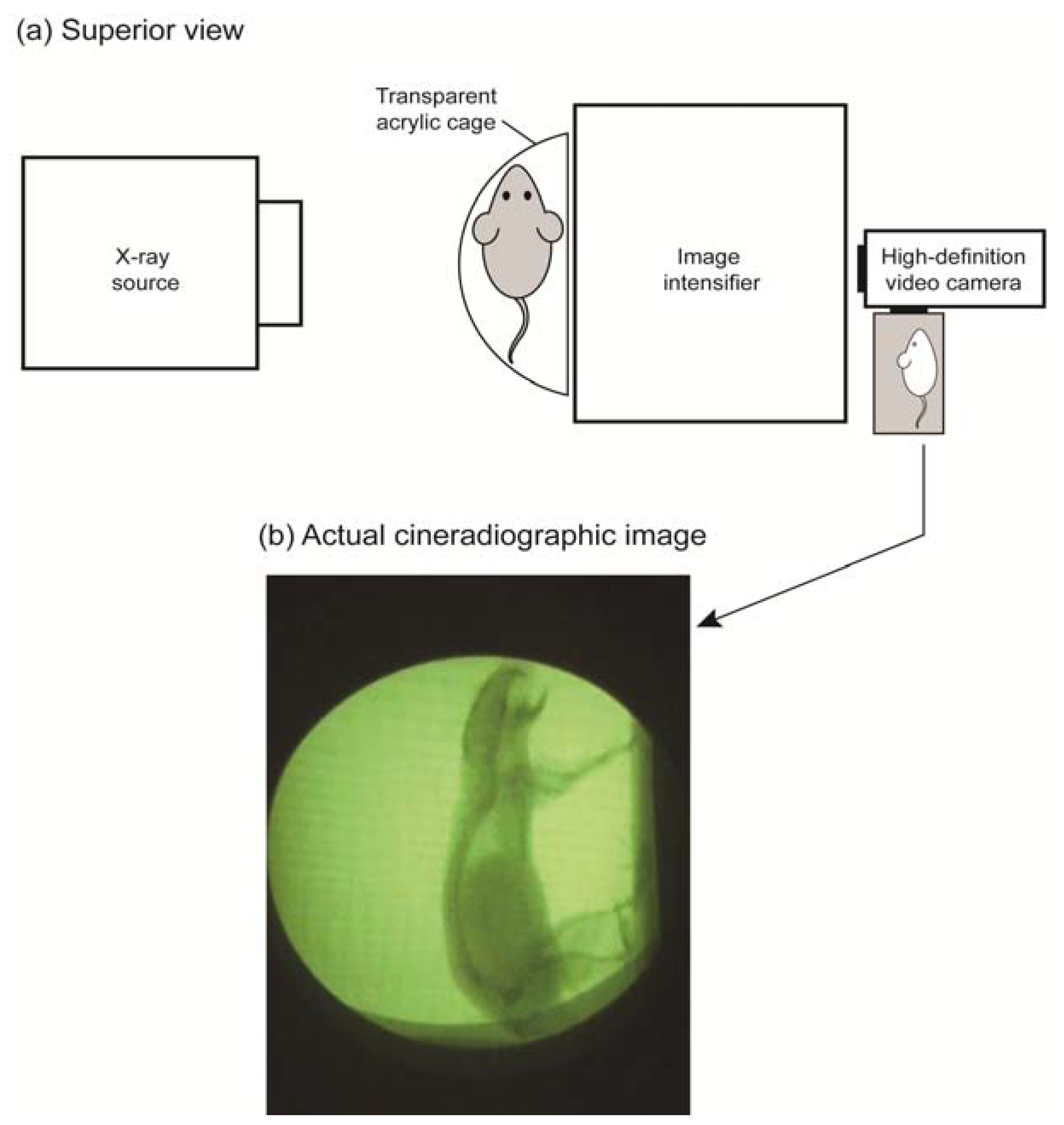
2.3. Cineradiographic Imaging
2.4. Data Collection
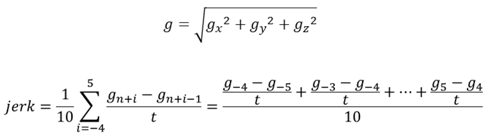
n: time (sec × 10) in gravity-data acquisition at 10Hz. t = 0.1.
gn: gravity-data at time “n” when the photo frame was obtained.
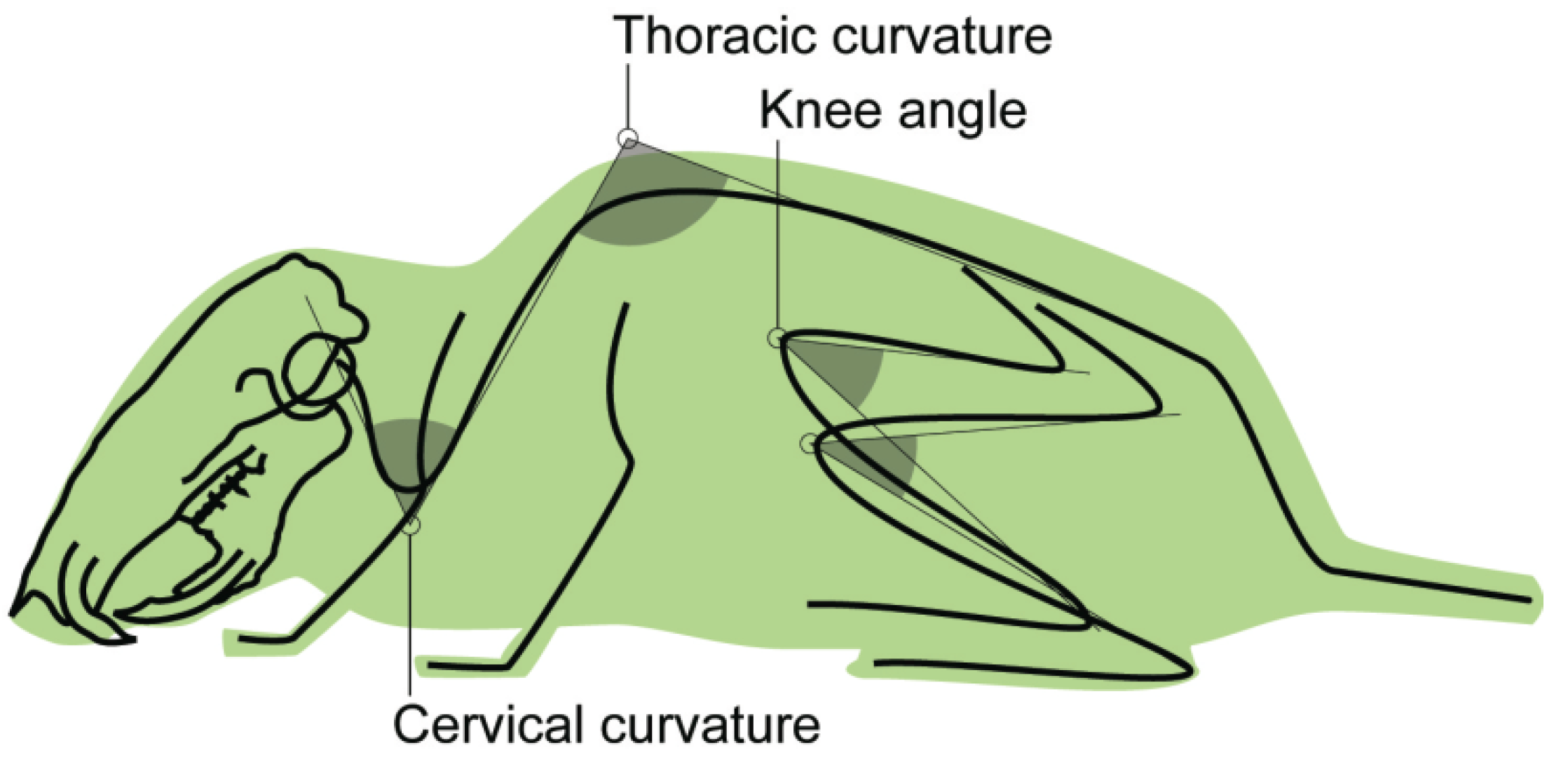
| Type | Label | Measurement |
|---|---|---|
| angle | cervical curvature | angle between cervical and thoracic segments of the spinal column |
| angle | thoracic curvature | angle between thoracic and lumbar segments of the spinal column |
| angle | knee angle | angle between greater trochanter, lateral epicondyle, and lateral malleolus [24] |
2.5. Statistical Analysis
3. Results and Discussion
3.1. Mouse Skeletal Angles in Parabolic Flight

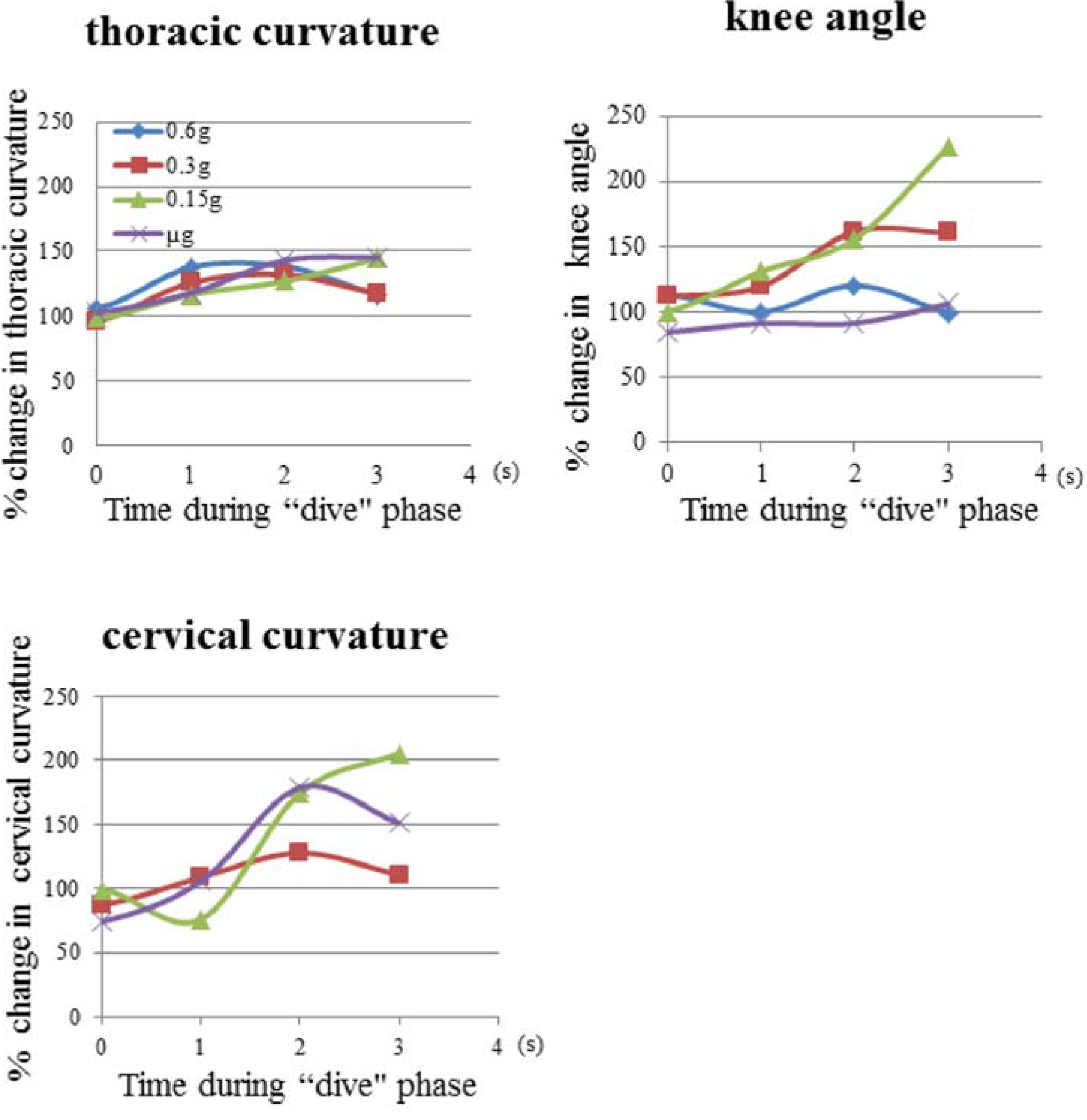
3.2. Thoracic Stretching
| Time (s) in the “dive” phase | 0.6 g-flight | 0.3 g-flight | 0.15 g-flight | μ g-flight |
|---|---|---|---|---|
| −1.0 | 94.4º ± 5.2 | 82.9º ± 6.2 | 93.3º ± 6.3 | 89.9º ± 6.5 |
| 0.0 | 94.7º ± 4.6 | 86.1º ± 4.4 | 88.1º ± 4.7 | 91.9º ± 4.3 |
| 1.0 | 122.9º ± 8.8 | 112.6º ± 8.4 | 104.4º ± 11.6 | 105.4º ± 14.3 |
| 2.0 | 123.7º ± 8.7 | 118.0º ± 7.3 | 113.5º ± 6.4 | 127.8º ± 8.7 |
| 3.0 | 104.0º ± 10.4 | 105.1º ± 14.7 | 129.0º ± 10.2 | 130.1º ± 7.7 |
| 4.0 | 96.6º ± 3.5 | 99.81º ± 3.8 | 107.9º ± 10.7 | 96.1º ± 7.3 |
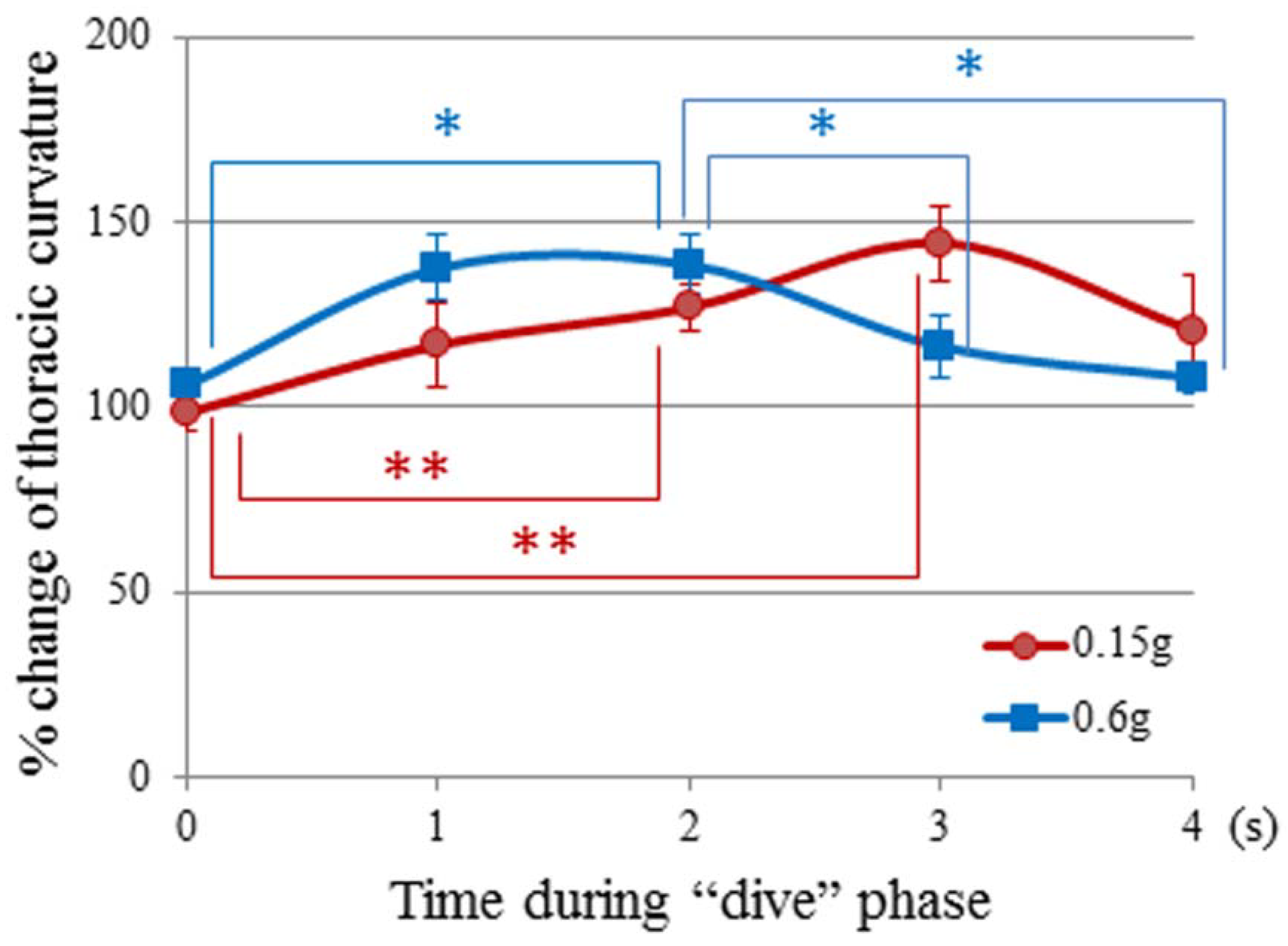
3.3. Time History of Gravity and Jerk

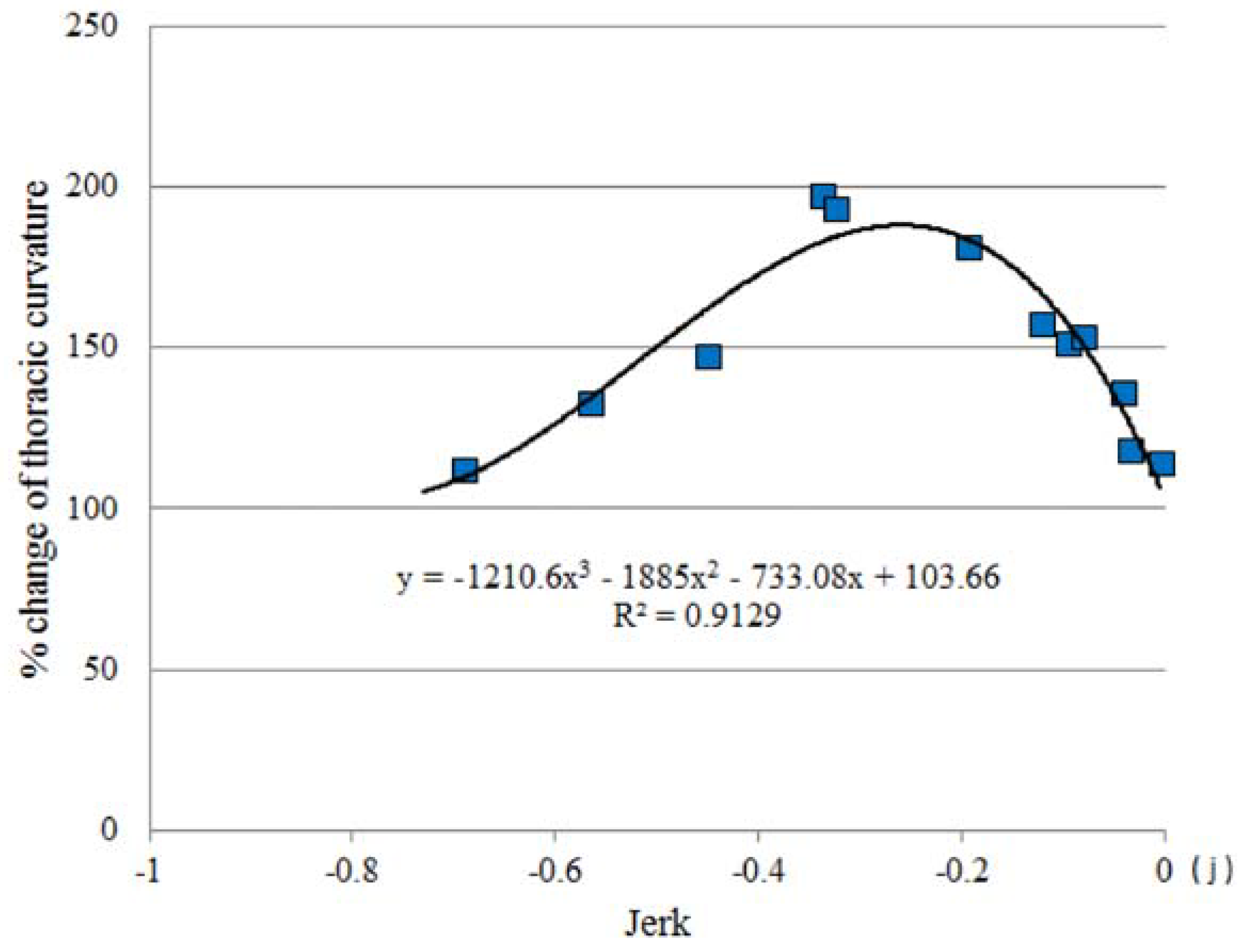
3.4. Jerk and Thoracic Stretching

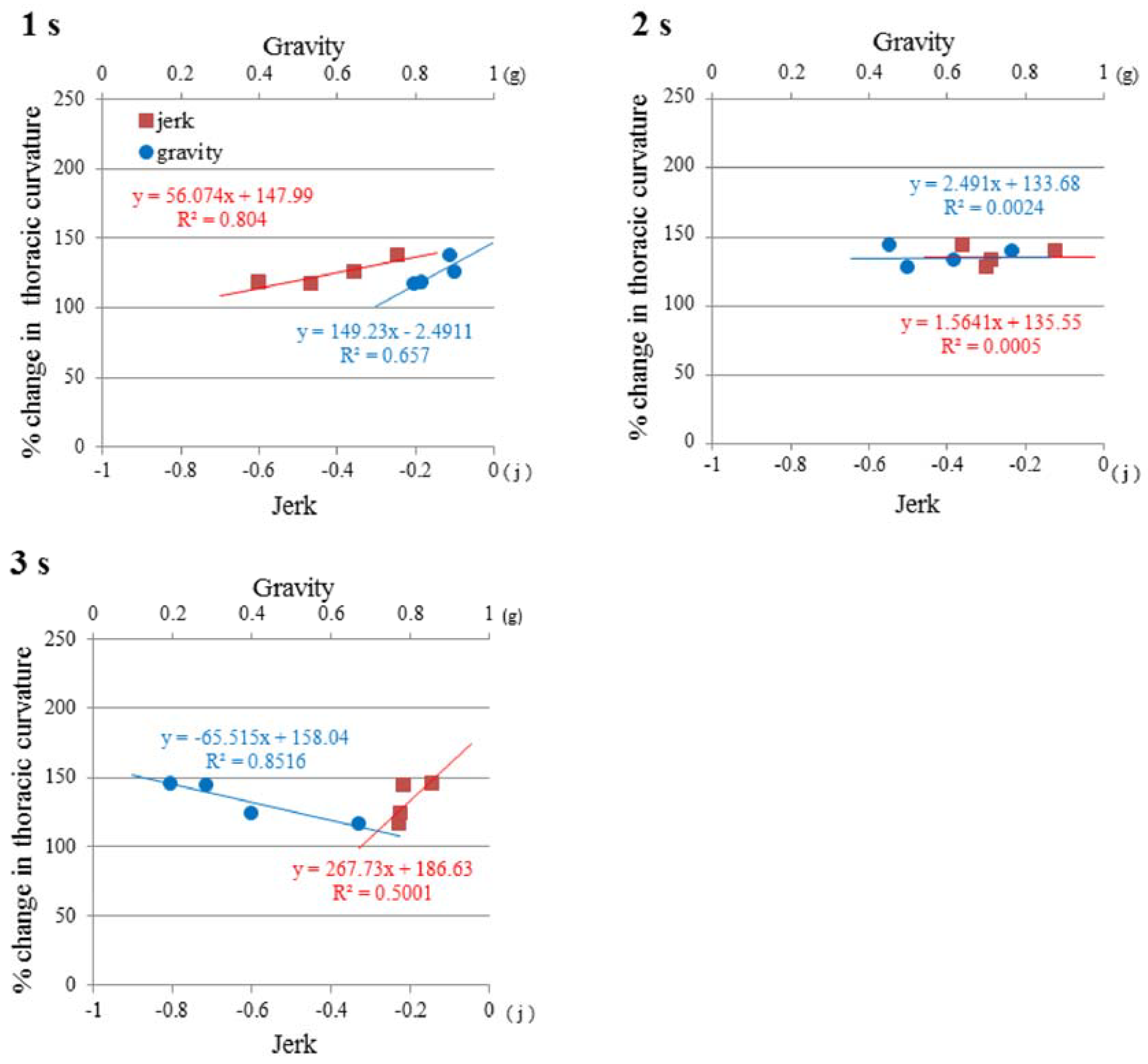
3.5. Regulation of Mouse Posture by Gravity and Jerk
3.6. Limitations of This Study
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Counil, L.; Kerlirzin, Y.; Dietrich, G. Cognitive style in attainment of an upside-down posture in water with and without vision. Percept. Mot. Skills 2012, 114, 51–58. [Google Scholar] [CrossRef]
- Jarchow, T.; Mast, F.W. The effect of water immersion on postural and visual orientation. Aviat. Space Environ. Med. 1999, 70, 879–886. [Google Scholar]
- Newman, D.J.; Alexander, H.L. Human locomotion and workload for simulated lunar and martian environments. Acta Astronaut. 1993, 29, 613–620. [Google Scholar] [CrossRef]
- Wickman, L.A.; Luna, B. Locomotion while load-carrying in reduced gravities. Aviat. Space Environ. Med. 1996, 67, 940–946. [Google Scholar]
- Iwasaki, K.; Levine, B.D.; Zhang, R.; Zuckerman, J.H.; Pawelczyk, J.A.; Diedrich, A.; Ertl, A.C.; Cox, J.F.; Cooke, W.H.; Giller, C.A.; et al. Human cerebral autoregulation before, during and after spaceflight. J. Physiol. 2007, 579, 799–810. [Google Scholar] [CrossRef]
- Williams, D.; Kuipers, A.; Mukai, C.; Thirsk, R. Acclimation during space flight: Effects on human physiology. CMAJ 2009, 180, 1317–1323. [Google Scholar] [CrossRef]
- Rai, B.; Kaur, J.; Foing, B.H. Evaluation by an aeronautic dentist on the adverse effects of a six-week period of microgravity on the oral cavity. Int. J. Dent. 2011, 2011, 548068. [Google Scholar]
- Clement, G.; Arnesen, T.N.; Olsen, M.H.; Sylvestre, B. Perception of longitudinal body axis in microgravity during parabolic flight. Neurosci. Lett. 2007, 413, 150–153. [Google Scholar] [CrossRef]
- Clement, G.; Demel, M. Perceptual reversal of bi-stable figures in microgravity and hypergravity during parabolic flight. Neurosci. Lett. 2012, 507, 143–146. [Google Scholar] [CrossRef]
- Clement, G.; Andre-Deshays, C. Motor activity and visually induced postural reactions during two-g and zero-g phases of parabolic flight. Neurosci. Lett. 1987, 79, 113–116. [Google Scholar] [CrossRef]
- Clement, G.; Reschke, M.F.; Verrett, C.M.; Wood, S.J. Effects of gravitoinertial force variations on optokinetic nystagmus and on perception of visual stimulus orientation. Aviat. Space Environ. Med. 1992, 63, 771–777. [Google Scholar]
- Dalecki, M.; Drager, T.; Mierau, A.; Bock, O. Production of finely graded forces in humans: Effects of simulated weightlessness by water immersion. Exp. Brain Res. 2012, 218, 41–47. [Google Scholar] [CrossRef]
- Wagner, E.B.; Fulford-Jones, T.R.F. Sensorimotor investigations for the mars gravity biosatellite: A rotating spacecraft for partial gravity research. Brain Res. 2006, 1091, 75–78. [Google Scholar]
- Aou, S.; Narikiyo, K.; Masuda, A.; Zeredo, J.L.; Hasegawa, K.; Inoue, K.A.; Kumei, Y. Obese mice lacking leptin receptors showed lower struggling with longer social contact in partial gravity conditions than control lean mice. Neurosci. Res. 2011, 71, e52. [Google Scholar]
- Inoue, K.A.; Narikiyo, K.; Zeredo, J.L.; Masuda, A.; Aou, S.; Kumei, Y. Analysis of neurotransmitters/neuromodulators released under low gravity using microdialysis technique. Neurosci. Res. 2011, 71, e371. [Google Scholar]
- Kumei, Y.; Inoue, K.A.; Zeredo, J.L.; Hasegawa, K.; Maezawa, Y.; Narikiyo, K.; Masuda, A.; Aou, S. Rodent neuroscience in low-gravity conditions. Neurosci. Res. 2011, 71, e168. [Google Scholar]
- Zeredo, J.L.; Toda, K.; Matsuura, M.; Kumei, Y. Behavioral responses to partial-gravity conditions in rats. Neurosci. Lett. 2012, 529, 108–111. [Google Scholar] [CrossRef]
- De Winkel, K.N.; Clement, G.; Groen, E.L.; Werkhoven, P.J. The perception of verticality in lunar and martian gravity conditions. Neurosci. Lett. 2012, 529, 7–11. [Google Scholar] [CrossRef]
- Schultheis, L.; Ruff, C.B.; Rastogi, S.; Bloomfield, S.; Hogan, H.A.; Fedarko, N.; Thierry-Palmer, M.; Ruiz, J.; Bauss, F.; Shapiro, J.R. Disuse bone loss in hindquarter suspended rats: Partial weightbearing, exercise and ibandronate treatment as countermeasures. J. Gravit. Physiol. 2000, 7, P13–P14. [Google Scholar]
- Swift, J.M.; Lima, F.; Macias, B.R.; Allen, M.R.; Greene, E.S.; Shirazi-Fard, Y.; Kupke, J.S.; Hogan, H.A.; Bloomfield, S.A. Partial weight bearing does not prevent musculoskeletal losses associated with disuse. Med. Sci. Sports Exerc. 2013, 45, 2052–2060. [Google Scholar] [CrossRef]
- Harris, L.R.; Jenkin, M.; Dyde, R.T.; Jenkin, H. Enhancing visual cues to orientation: Suggestions for space travelers and the elderly. Prog. Brain Res. 2011, 191, 133–142. [Google Scholar] [CrossRef]
- Mori, S. Disorientation of animals in microgravity. Nagoya J. Med. Sci. 1995, 58, 71–81. [Google Scholar]
- Thornton, W.E.; Bonato, F. Space motion sickness and motion sickness: Symptoms and etiology. Aviat. Space Environ. Med. 2013, 84, 716–721. [Google Scholar] [CrossRef]
- Bauman, J.M.; Chang, Y.H. High-speed x-ray video demonstrates significant skin movement errors with standard optical kinematics during rat locomotion. J. Neurosci. Methods 2010, 186, 18–24. [Google Scholar] [CrossRef]
- Asslander, L.; Hettich, G.; Gollhofer, A.; Mergner, T. Contribution of visual velocity and displacement cues to human balancing of support surface tilt. Exp. Brain Res. 2013, 228, 297–304. [Google Scholar] [CrossRef]
- Vaitl, D.; Mittelstaedt, H.; Saborowski, R.; Stark, R.; Baisch, F. Shifts in blood volume alter the perception of posture: Further evidence for somatic graviception. Int. J. Psychophysiol. 2002, 44, 1–11. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hasegawa, K.; De Campos, P.S.; Zeredo, J.L.; Kumei, Y. Cineradiographic Analysis of Mouse Postural Response to Alteration of Gravity and Jerk (Gravity Deceleration Rate). Life 2014, 4, 174-188. https://doi.org/10.3390/life4020174
Hasegawa K, De Campos PS, Zeredo JL, Kumei Y. Cineradiographic Analysis of Mouse Postural Response to Alteration of Gravity and Jerk (Gravity Deceleration Rate). Life. 2014; 4(2):174-188. https://doi.org/10.3390/life4020174
Chicago/Turabian StyleHasegawa, Katsuya, Priscila S. De Campos, Jorge L. Zeredo, and Yasuhiro Kumei. 2014. "Cineradiographic Analysis of Mouse Postural Response to Alteration of Gravity and Jerk (Gravity Deceleration Rate)" Life 4, no. 2: 174-188. https://doi.org/10.3390/life4020174




