Magnetotactic Bacteria from Extreme Environments
Abstract
:1. Introduction
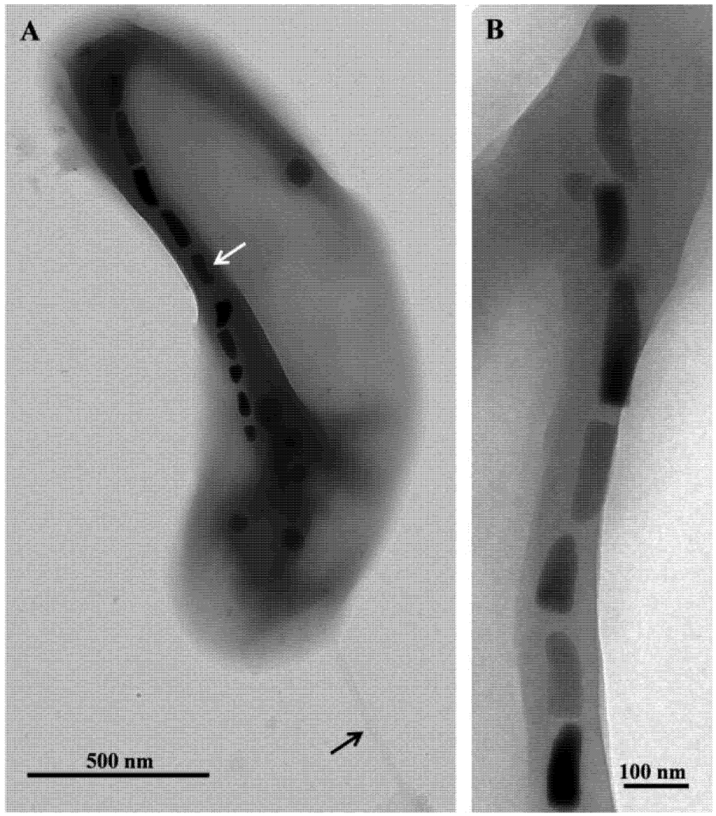
2. Thermophilic Magnetotactic Bacteria

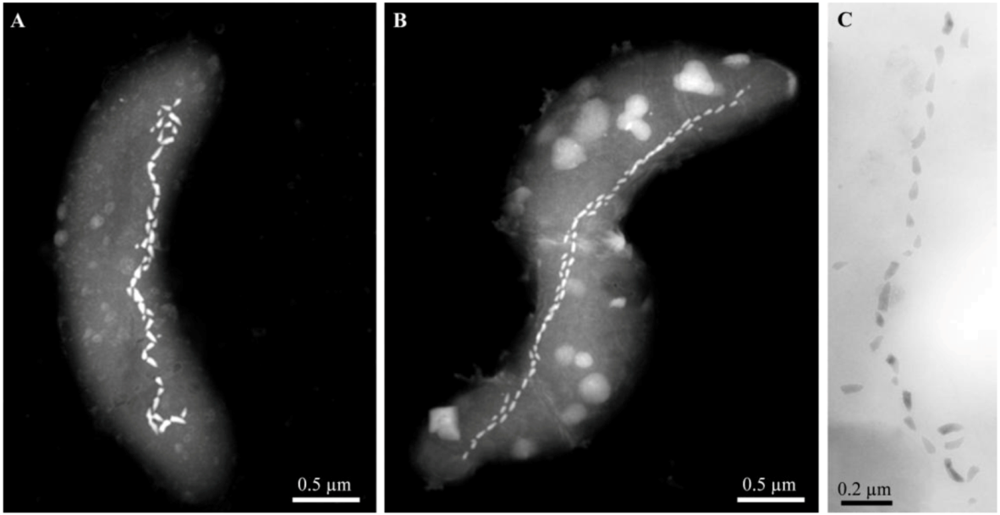
3. Alkaliphilic Magnetotactic Bacteria
4. Potential for the Presence of MTB in Other Extreme Environments
4.1. Piezophilic and Psychrophilic MTB
4.2. Acidophilic MTB
4.3. Halophilic Magnetotactic Bacteria
4.4. Magnetotactic Bacteria as Putative Biosignatures for Extraterrestrial Life
5. Conclusions
Acknowledgements
References
- Kirschvink, J.L.; Walker, M.M.; Diebel, C.E. Magnetite-based magnetoreception. Curr. Opin. Neurobiol. 2001, 11, 462–467. [Google Scholar] [CrossRef]
- Bazylinski, D.A.; Frankel, R.B. Magnetosome formation in prokaryotes. Nat. Rev. Microbiol. 2004, 2, 217–230. [Google Scholar] [CrossRef]
- Frankel, R.B.; Bazylinski, D.A.; Johnson, M.S.; Taylor, B.L. Magneto-aerotaxis in marine coccoid bacteria. Biophys. J. 1997, 73, 994–1000. [Google Scholar] [CrossRef]
- Amann, R.; Peplies, J.; Schüler, D. Diversity and taxonomy of magnetotactic bacteria. In Magnetoreception and Magnetosomes, in Bacteria; Schüler, D., Ed.; Microbiology Monographs; Springer: Berlin/Heidelberg, Germany, 2007; Volume 3, pp. 25–36. [Google Scholar]
- Kolinko, S.; Jogler, C.; Katzmann, E.; Wanner, G.; Peplies, J.; Schüler, D. Single-cell analysis reveals a novel uncultivated magnetotactic bacterium within the candidate division OP3. Environ. Microbiol. 2012, 14, 1709–1721. [Google Scholar]
- Lefèvre, C.T.; Viloria, N.; Schmidt, M.L.; Pósfai, M.; Frankel, R.B.; Bazylinski, D.A. Novel magnetite-producing magnetotactic bacteria belonging to the Gammaproteobacteria. ISME J. 2012, 6, 440–450. [Google Scholar]
- Bazylinski, D.A.; Schübbe, S. Controlled biomineralization by and applications of magnetotactic bacteria. Adv. Appl. Microbiol. 2007, 62, 21–62. [Google Scholar]
- Schleifer, K.H.; Schüler, D.; Spring, S.; Weizenegger, M.; Amann, R.; Ludwig, W.; Kohler, M. The genus Magnetospirillum gen. nov. description of Magnetospirillum gryphiswaldense sp. nov. and transfer of Aquaspirillum magnetotacticum to Magnetospirillum magnetotacticum comb. nov. Syst. Appl. Microbiol. 1991, 14, 379–385. [Google Scholar]
- Sakaguchi, T.; Arakaki, A.; Matsunaga, T. Desulfovibrio magneticus sp nov., a novel sulfate-reducing bacterium that produces intracellular single-domain-sized magnetite particles. Int. J. Syst. Evol. Microbiol. 2002, 52, 215–221. [Google Scholar]
- Bazylinski, D.A.; Williams, T.J.; Lefèvre, C.T.; Berg, R.J.; Zhang, C.L.; Bowser, S.S.; Dean, A.J.; Beveridge, T.J. Magnetococcus marinus gen. nov., sp. nov., a marine, magnetotactic bacterium that represents a novel lineage (Magnetococcaceae fam. nov.; Magnetococcales ord. nov.) at the base of the Alphaproteobacteria. Int. J. Syst. Evol. Microbiol. 2013, 63, 801–808. [Google Scholar]
- Williams, T.J.; Lefèvre, C.T.; Zhao, W.; Beveridge, T.J.; Bazylinski, D.A. Magnetospira thiophila gen. nov., sp. nov., a marine magnetotactic bacterium that represents a novel lineage within the Rhodospirillaceae (Alphaproteobacteria). Int. J. Syst. Evol. Microbiol. 2012, 62, 2443–2450. [Google Scholar]
- Bazylinski, D.A.; Williams, T.J.; Lefèvre, C.T.; Trubitsyn, D.; Fang, J.; Beveridge, T.J.; Moskowitz, B.M.; Ward, B.; Schübbe, S.; Dubbels, B.L.; Simpson, B. Magnetovibrio blakemorei, gen. nov. sp. nov., a new magnetotactic bacterium (Alphaproteobacteria: Rhodospirillaceae) isolated from a salt marsh. Int. J. Syst. Evol. Microbiol. 2013. [Google Scholar] [CrossRef]
- McKay, D.S.; Gibson, E.K.; Thomas-Keprta, K.L.; Vali, H.; Romanek, C.S.; Clemett, S.J.; Chillier, X.D.F.; Maechling, C.R.; Zare, R.N. Search for past life on Mars: Possible relic biogenic activity in Martian meteorite ALH84001. Science 1996, 273, 924–930. [Google Scholar] [CrossRef]
- Thomas-Keprta, K.L.; Clemett, S.J.; Bazylinski, D.A.; Kirschvink, J.L.; McKay, D.S.; Wentworth, S.J.; Vali, H.; Gibson, E.K.; McKay, M.F.; Romanek, C.S. Truncated hexa-octahedral magnetite crystals in ALH84001: Presumptive biosignatures. P. Natl. Acad. Sci. USA 2001, 98, 2164–2169. [Google Scholar]
- Lefèvre, C.T.; Abreu, F.; Schmidt, M.L.; Lins, U.; Frankel, R.B.; Hedlund, B.P.; Bazylinski, D.A. Moderately thermophilic magnetotactic bacteria from hot springs in Nevada. Appl. Environ. Microbiol. 2010, 76, 3740–3743. [Google Scholar]
- Anderson, J.P. A geochemical study of the southwest part of the Black Rock Desert and its geothermal areas; Washoe, Pershing, and Humboldt Counties, Nevada. Colo. School Mines Q. 1978, 73, 15–22. [Google Scholar]
- Costa, K.C.; Navarro, J.B.; Shock, E.L.; Zhang, C.L.; Soukup, D.; Hedlund, B.P. Microbiology and geochemistry of great boiling and mud hot springs in the United States Great Basin. Extremophiles 2009, 13, 447–459. [Google Scholar] [CrossRef]
- Haouari, O.; Fardeau, M.-L.; Cayol, J.-L.; Fauque, G.; Casiot, C.; Elbaz-Poulichet, F.; Hamdi, M.; Ollivier, B. Thermodesulfovibrio hydrogeniphilus sp. nov., a new thermophilic sulphate-reducing bacterium isolated from a Tunisian hot spring. Syst. Appl. Microbiol. 2008, 31, 38–42. [Google Scholar]
- Nash, C. Mechanisms and evolution of magnetotactic bacteria. Ph.D. thesis, California Institute of Technology, Pasadena, CA, USA, 2008. [Google Scholar]
- Lefèvre, C.T.; Frankel, R.B.; Pósfai, M.; Prozorov, T.; Bazylinski, D.A. Isolation of obligately alkaliphilic magnetotactic bacteria from extremely alkaline environments. Environ. Microbiol. 2011, 13, 2342–2350. [Google Scholar] [CrossRef]
- Oremland, R.S.; Dowdle, P.R.; Hoeft, S.; Sharp, J.O.; Schaefer, J.K.; Miller, L.G.; Blum, J.S.; Smith, R.L.; Bloom, N.S.; Wallschlaeger, D. Bacterial dissimilatory reduction of arsenate and sulfate in meromictic Mono Lake, California. Geochim. Cosmochim. Acta. 2000, 64, 3073–3084. [Google Scholar] [CrossRef]
- Kulp, T.R.; Han, S.; Saltikov, C.W.; Lanoil, B.D.; Zargar, K.; Oremland, R.S. Effects of imposed salinity gradients on dissimilatory arsenate reduction, sulfate reduction, and other microbial processes in sediments from two California soda lakes. Appl. Environ. Microbiol. 2007, 73, 5130–5137. [Google Scholar] [CrossRef]
- Hoeft, S.E.; Kulp, T.R.; Stolz, J.F.; Hollibaugh, J.T.; Oremland, R.S. Dissimilatory arsenate reduction with sulfide as electron donor: experiments with mono lake water and isolation of strain MLMS-1, a chemoautotrophic arsenate respirer. Appl. Environ. Microbiol. 2004, 70, 2741–2747. [Google Scholar] [CrossRef]
- Wiemeyer, S. Metals and trace elements in water, sediment, and vegetation at Ash Meadows National Wildlife Refuge-1993. In Technical Report for the United States Fish and Wildlife Service; Reno, NV, USA, July 2005. [Google Scholar]
- Al-Qudah, O.; Woocay, A.; Walton, J. Identification of probable groundwater paths in the Amargosa Desert vicinity. Appl. Geochem. 2011, 26, 565–574. [Google Scholar] [CrossRef]
- Pikuta, E.V.; Hoover, R.B.; Bej, A.K.; Marsic, D.; Whitman, W.B.; Cleland, D.; Krader, P. Desulfonatronum thiodismutans sp. nov., a novel alkaliphilic, sulfate-reducing bacterium capable of lithoautotrophic growth. Int. J. Syst. Evol. Microbiol. 2003, 53, 1327–1332. [Google Scholar]
- Chavadar, M.S.; Bajekal, S.S. Magnetotactic bacteria from Lonar lake. Curr. Sci. 2009, 96, 957–959. [Google Scholar]
- Chavadar, M.S.; Bajekal, S.S. Microaerophilic magnetotactic bacteria from Lonar Lake, India. J. Pure Appl. Microbiol. 2010, 4, 681–685. [Google Scholar]
- Rajasekhar, R.P.; Mishra, D.C. Analysis of gravity and magnetic anomalies over Lonar Lake, India: An impact crater in a basalt province. Curr. Sci. 2005, 88, 1836–1840. [Google Scholar]
- Krulwich, T.A. Alkaliphilic prokaryotes. In The Prokaryotes; Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K.-H., Stackebrandt, E., Eds.; Springer: New York, NY, USA, 2006; pp. 283–308. [Google Scholar]
- Jimenez-Lopez, C.; Romanek, C.S.; Bazylinski, D.A. Magnetite as a prokaryotic biomarker: A review. J. Geophys. Res. Biogeosci. 2010, 115, G00G03. [Google Scholar] [CrossRef]
- Schübbe, S.; Kube, M.; Scheffel, A.; Wawer, C.; Heyen, U.; Meyerdierks, A.; Madkour, M.H.; Mayer, F.; Reinhardt, R.; Schüler, D. Characterization of a spontaneous nonmagnetic mutant of Magnetospirillum gryphiswaldense reveals a large deletion comprising a putative magnetosome island. J. Bacteriol. 2003, 185, 5779–5790. [Google Scholar] [CrossRef]
- Dubbels, B.L.; DiSpirito, A.A.; Morton, J.D.; Semrau, J.D.; Neto, J.N.E.; Bazylinski, D.A. Evidence for a copper-dependent iron transport system in the marine, magnetotactic bacterium strain MV-1. Microbiology 2004, 150, 2931–2945. [Google Scholar] [CrossRef]
- Schüler, D. Genetics and cell biology of magnetosome formation in magnetotactic bacteria. FEMS Microbiol. Rev. 2008, 32, 654–672. [Google Scholar] [CrossRef]
- Pósfai, M.; Moskowitz, B.M.; Arató, B.; Schüler, D.; Flies, C.; Bazylinski, D.A.; Frankel, R.B. Properties of intracellular magnetite crystals produced by Desulfovibrio magneticus strain RS-1. Earth Planet Sci. Lett. 2006, 249, 444–455. [Google Scholar] [CrossRef]
- Fang, J.; Zhang, L.; Bazylinski, D.A. Deep-sea piezosphere and piezophiles: geomicrobiology and biogeochemistry. Trends Microbiol. 2010, 18, 413–422. [Google Scholar] [CrossRef]
- Stolz, J.; Chang, S.B.R.; Kirschvink, J.L. Magnetotactic bacteria and single-domain magnetite in hemipelagic sediments. Nature 1986, 321, 849–851. [Google Scholar]
- Petermann, H.; Bleil, U. Detection of live magnetotactic bacteria in South Atlantic deep-sea sediments. Earth Planet. Sci. Lett. 1993, 117, 223–228. [Google Scholar] [CrossRef]
- Abreu, F.; Lins, U. Personal communication, Universidade Federal do Rio de Janeiro: Rio de Janeiro, Brazil, 2013.
- Baker-Austin, C.; Dopson, M. Life in acid: pH homeostasis in acidophiles. Trends Microbiol. 2007, 15, 165–171. [Google Scholar] [CrossRef]
- Frankel, R.B.; Bazylinski, D.A. Biologically induced mineralization by bacteria. Rev. Mineral. Geochem. 2003, 54, 95–114. [Google Scholar] [CrossRef]
- Bell, P.E.; Mills, A.L.; Herman, J.S. Biogeochemical conditions favoring magnetite formation during anaerobic iron reduction. Appl. Environ. Microbiol. 1987, 53, 2610–2616. [Google Scholar]
- Martins, J.L.; Silveira, T.S.; Silva, K.T.; Lins, U. Salinity dependence of the distribution of multicellular magnetotactic prokaryotes in a hypersaline lagoon. Int. Microbiol. 2009, 12, 193–201. [Google Scholar]
- Lefèvre, C.T.; Abreu, F.; Lins, U.; Bazylinski, D.A. Nonmagnetotactic multicellular prokaryotes from low-saline, nonmarine aquatic environments and their unusual negative phototactic behavior. Appl. Environ. Microbiol. 2010, 76, 3220–3227. [Google Scholar] [CrossRef]
- Ollivier, B.; Caumette, P.; Garcia, J.L.; Mah, R.A. Anaerobic bacteria from hypersaline environments. Microbiol. Rev. 1994, 58, 27–38. [Google Scholar]
- Thomas-Keprta, K.L.; Bazylinski, D.A.; Kirschvink, J.L.; Clemett, S.J.; McKay, D.S.; Wentworth, S.J.; Vali, H.; Gibson, E.K.; Romanek, C.S. Elongated prismatic magnetite crystals in ALH84001 carbonate globules: Potential Martian magnetofossils. Geochim. Cosmochim. Acta 2000, 64, 4049–4081. [Google Scholar] [CrossRef]
- Thomas-Keprta, K.L.; Clemett, S.J.; Bazylinski, D.A.; Kirschvink, J.L.; McKay, D.S.; Wentworth, S.J.; Vali, H.; Gibson, E.K.; Romanek, C.S. Magnetofossils from ancient Mars: a robust biosignature in the Martian meteorite ALH84001. Appl. Environ. Microbiol. 2002, 68, 3663–3672. [Google Scholar] [CrossRef]
- Buseck, P.R.; Dunin-Borkowski, R.E.; Devouard, B.; Frankel, R.B.; McCartney, M.R.; Midgley, P.A.; Posfai, M.; Weyland, M. Magnetite morphology and life on Mars. P. Natl. Acad. Sci. USA 2001, 98, 13490–13495. [Google Scholar] [CrossRef]
- Clemett, S.J.; Thomas-Keprta, K.L.; Shimmin, J.; Morphew, M.; McIntosh, J.R.; Bazylinski, D.A.; Kirschvink, J.L.; Wentworth, S.J.; McKay, D.S.; Vali, H.; et al. Crystal morphology of MV-1 magnetite. Am. Mineral. 2002, 87, 1727–1730. [Google Scholar]
- Chang, S.B.R.; Kirschvink, J.L. Magnetofossils, the magnetization of sediments, and the evolution of magnetite biomineralization. Annu. Rev. Earth Planet Sci. 1989, 17, 169–195. [Google Scholar] [CrossRef]
- Golden, D.C.; Ming, D.W.; Morris, R.V.; Brearley, A.J.; Lauer, H.V., Jr.; Treiman, A.H.; Zolensky, M.E.; Schwandt, C.S.; Lofgren, G.E.; McKay, G.A. Evidence for exclusively inorganic formation of magnetite in Martian meteorite ALH84001. Am. Mineral. 2004, 89, 681–695. [Google Scholar]
- Martel, J.; Young, D.; Peng, H.-H.; Wu, C.-Y.; Young, J.D. Biomimetic properties of minerals and the search for life in the Martian meteorite ALH84001. Annu. Rev. Earth Planet. Sci. 2012, 40, 167–193. [Google Scholar]
- Weiss, B.P.; Kim, S.S.; Kirschvink, J.L.; Kopp, R.E.; Sankaran, M.; Kobayashi, A.; Komeili, A. Magnetic tests for magnetosome chains in Martian meteorite ALH84001. P. Natl. Acad. Sci. USA 2004, 101, 8281–8284. [Google Scholar]
- Arato, B.; Szanyi, Z.; Flies, C.; Schüler, D.; Frankel, R.B.; Buseck, P.R.; Pósfai, M. Crystal-size and shape distributions of magnetite from uncultured magnetotactic bacteria as a potential biomarker. Am. Mineral. 2005, 90, 1233–1240. [Google Scholar] [CrossRef]
- Kopp, R.E.; Kirschvink, J.L. The identification and biogeochemical interpretation of fossil magnetotactic bacteria. Earth Sci. Rev. 2008, 86, 42–61. [Google Scholar] [CrossRef]
- Gehring, A.U.; Kind, J.; Charilaou, M.; Garcia-Rubio, I. The detection of magnetotactic bacteria and magnetofossils by means of magnetic anisotropy. Earth Planet. Sci. Lett. 2011, 309, 113–117. [Google Scholar] [CrossRef]
- Kind, J.; Gehring, A.U.; Winklhofer, M.; Hirt, A.M. Combined use of magnetometry and spectroscopy for identifying magnetofossils in sediments. Geochem. Geophys. Geosyst. 2011, 12, Q08008. [Google Scholar]
- Kempe, S.; Degens, E. An early soda ocean? Chem. Geol. 1985, 53, 95–108. [Google Scholar] [CrossRef]
- Hecht, M.H.; Kounaves, S.P.; Quinn, R.C.; West, S.J.; Young, S.M.M.; Ming, D.W.; Catling, D.C.; Clark, B.C.; Boynton, W.V.; Hoffman, J.; et al. Detection of perchlorate and the soluble chemistry of martian soil at the Phoenix lander site. Science 2009, 325, 64–67. [Google Scholar]
- Kounaves, S.P.; Hecht, M.H.; Kapit, J.; Gospodinova, K.; DeFlores, L.; Quinn, R.C.; Boynton, W.V.; Clark, B.C.; Catling, D.C.; Hredzak, P.; et al. Wet chemistry experiments on the 2007 Phoenix Mars Scout Lander mission: Data analysis and results. J. Geophys. Res. Planets 2010, 115. [Google Scholar]
- Kempe, S.; Kazmierczak, J. A terrestrial model for an alkaline martian hydrosphere. Planet Space Sci. 1997, 45, 1493–1499. [Google Scholar] [CrossRef]
- McNeill, D.F.; Ginsburg, R.N.; Chang, S.B.R.; Kirschvink, J.L. Magnetostratigraphic dating of shallow-water carbonates from San-Salvador, Bahamas. Geology 1988, 16, 8–12. [Google Scholar] [CrossRef]
- Sakai, S.; Jige, M. Characterization of magnetic particles and magnetostratigraphic dating of shallow-water carbonates in the Ryukyu Islands, northwestern Pacific. Isl. Arc. 2006, 15, 468–475. [Google Scholar] [CrossRef]
- Bellini, S. On a unique behavior of freshwater bacteria. Chin. J. Oceanol. Limn. 2009, 27, 3–5. [Google Scholar] [CrossRef]
- Bellini, S. Further studies on “magnetosensitive bacteria”. Chin. J. Oceanol. Limn. 2009, 27, 6–12. [Google Scholar] [CrossRef]
- Blakemore, R.P. Magnetotactic bacteria. Science 1975, 190, 377–379. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bazylinski, D.A.; Lefèvre, C.T. Magnetotactic Bacteria from Extreme Environments. Life 2013, 3, 295-307. https://doi.org/10.3390/life3020295
Bazylinski DA, Lefèvre CT. Magnetotactic Bacteria from Extreme Environments. Life. 2013; 3(2):295-307. https://doi.org/10.3390/life3020295
Chicago/Turabian StyleBazylinski, Dennis A., and Christopher T. Lefèvre. 2013. "Magnetotactic Bacteria from Extreme Environments" Life 3, no. 2: 295-307. https://doi.org/10.3390/life3020295



