Synchrotron Nano-Diffraction Study of Thermally Treated Asbestos Tremolite from Val d’Ala, Turin (Italy)
Abstract
:1. Introduction
2. Materials and Methods
3. Results
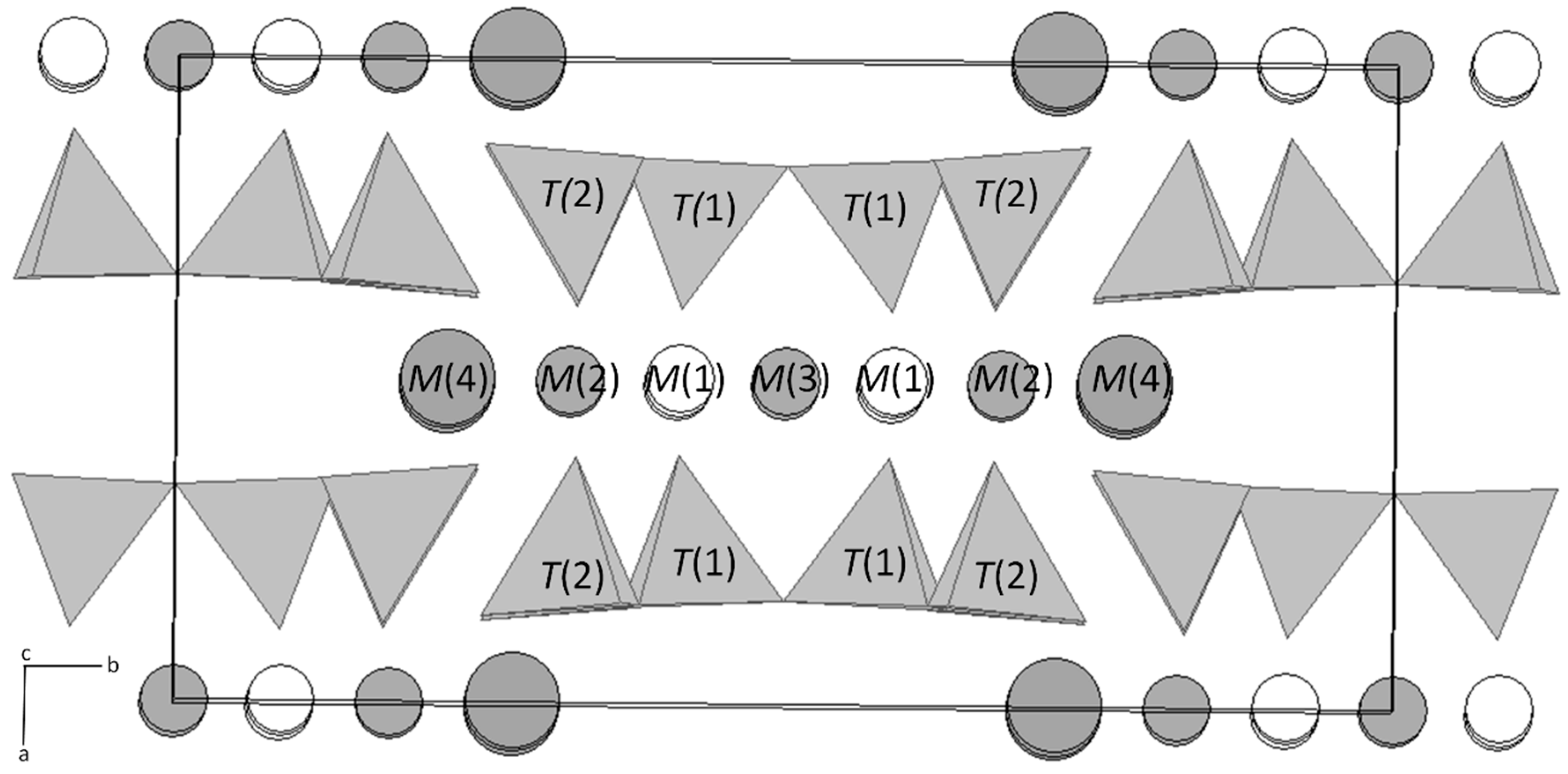
3.1. X-ray Single Crystal Nano-Diffraction
3.2. X-ray Powder Nano-Diffraction
3.3. FEG-SEM Imaging
4. Discussion
4.1. Single Crystal Nano-Diffraction
4.2. Tremolite Asbestos Fibres Heated at 1000 °C
5. Conclusions
- The mean T-(O) distances have no sensitivity to heat, as they undergo a very slight expansion;
- The M(1), M(2), M(3), and M(4) polyhedra exhibit larger thermal expansions than do the silicate tetrahedra, as a result of the weaker M-O bonds.
Author Contributions
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). Asbestos and Other Natural Mineral Fibres: Environmental Health Criteria 53; World Health Organization: Geneva, Switzerland, 1986; pp. 1–194. ISBN 92-4-154193-8. [Google Scholar]
- European Community (EU) Parliament. Directive 2003/18/EC of the Council of 27th March 2003 amending Council Directive 83/477/EEC on the protection of workers from the risks related to exposure to asbestos at work. Off. J. Eur. Union 2003, 97, 48–52. [Google Scholar]
- Gualtieri, A.F. Mineral fibre-based building materials and their health hazards. In Toxicity of Building Materials; Pacheco-Torgal, F., Jalali, S., Fucic, A., Eds.; Woodhead Publishing: Cambridge, UK, 2012; pp. 166–195. ISBN 978-0-85709-122-2. [Google Scholar]
- Gualtieri, A.F. Introduction. In Mineral Fibres: Crystal Chemistry, Chemical-Physical Properties, Biological Interaction and Toxicity; Gualtieri, A.F., Ed.; European Mineralogical Union: London, UK, 2017; Volume 18, pp. 1–15. ISBN 0-903-05665-6. [Google Scholar]
- International Agency for Research on Cancer (IARC). Overall Evaluations of Carcinogenicity: An Updating of IARC Monographs Volumes 1 to 42; International Agency for Research on Cancer: Lyon, France, 1987; pp. 1–440. ISBN 92-8-32-1411-0. [Google Scholar]
- Gualtieri, A.F.; Giacobbe, C.; Sardisco, L.; Saraceno, M.; Gualtieri, M.L.; Lusvardi, G.; Cavenati, C.; Zanatto, I. Recycling of the product of thermal inertization of cement–asbestos for various industrial applications. Waste Manag. 2011, 31, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Gualtieri, A.F.; Boccaletti, M. Recycling of the product of thermal inertization of cement-asbestos for the production of concrete. Constr. Build. Mater. 2011, 25, 3561–3569. [Google Scholar] [CrossRef]
- Kusiorowski, R.; Zaremba, T.; Piotrowski, J.; Podwórny, J. Utilisation of cement-asbestos wastes by thermal treatment and the potential possibility use of obtained product for the clinker bricks manufacture. J. Mater. Sci. 2015, 50, 6757–6767. [Google Scholar] [CrossRef] [Green Version]
- European Commission (EC). Directive 2008/98/EC of the European Parliament and of the Council of 19 November 2008 on waste. Off. J. Eur. Union 2008, 312, 22. [Google Scholar]
- Leonelli, C.; Veronesi, P.; Boccaccini, D.N.; Rivasi, M.R.; Barbieri, L.; Andreola, F.; Lancellotti, I.; Rabitti, D.; Pellacani, G.C. Microwave thermal inertisation of asbestos containing waste and its recycling in traditional ceramics. J. Hazard. Mater. 2006, 135, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Gualtieri, A.F.; Cavenati, C.; Zanatto, I.; Meloni, M.; Elmi, G.; Gualtieri, M.L. The transformation sequence of cement-asbestos slates up to 1200 °C and safe recycling of the reaction product in stoneware tile mixtures. J. Hazard. Mater. 2008, 152, 563–570. [Google Scholar] [CrossRef] [PubMed]
- Dellisanti, F.; Rossi, P.L.; Valdrè, G. Remediation of asbestos containing materials by Joule heating vitrification performed in a pre-pilot apparatus. Int. J. Miner. Process. 2009, 91, 61–67. [Google Scholar] [CrossRef]
- Bloise, A.; Catalano, M.; Gualtieri, A.F. Effect of Grinding on Chrysotile, Amosite and Crocidolite and Implications for Thermal Treatment. Minerals 2018, 8, 135. [Google Scholar] [CrossRef]
- Hawthorne, F.C.; Oberti, R. Amphiboles: Crystal Chemistry. Rev. Mineral. Geochem. 2007, 67, 1–54. [Google Scholar] [CrossRef]
- Dichicco, M.C.; Laurita, S.; Sinisi, R.; Battiloro, R.; Rizzo, G. Environmental and Health: The Importance of Tremolite Occurence in the Pollino Geopark (Southern Italy). Geosciences 2018, 8, 98. [Google Scholar] [CrossRef]
- Bloise, A.; Kusiorowski, R.; Gualtieri, A.F. The Effect of Grinding on Tremolite Asbestos and Anthophyllite Asbestos. Minerals 2018, 8, 274. [Google Scholar] [CrossRef]
- Bloise, A.; Kusiorowski, R.; Gualtieri, M.L.; Gualtieri, A.F. Thermal behaviour of mineral fibres. In Mineral Fibres: Crystal Chemistry, Chemical-Physical Properties, Biological Interaction and Toxicity; Gualtieri, A.F., Ed.; European Mineralogical Union: London, UK, 2017; Volume 18, pp. 215–252. ISBN 0-903-05665-6. [Google Scholar]
- Wittels, M. Structural disintegration of some amphiboles. Am. Mineral. 1952, 52, 28–36. [Google Scholar]
- Kusiorowski, R.; Zaremba, T.; Piotrowski, J.; Adamek, J. Thermal decomposition of different types of asbestos. J. Therm. Anal. Calorim. 2012, 109, 693–704. [Google Scholar] [CrossRef] [Green Version]
- Bloise, A.; Catalano, M.; Barrese, E.; Gualtieri, A.F.; Gandolfi, N.B.; Capella, S.; Belluso, E. TG/DSC study of the thermal behaviour of hazardous mineral fibres. J. Therm. Anal. Calorim. 2016, 123, 2225–2239. [Google Scholar] [CrossRef]
- Johnson, N.M.; Fegley, B. Tremolite decomposition on Venus II. Products, kinetics, and mechanism. Icarus 2003, 164, 317–333. [Google Scholar] [CrossRef]
- Sueno, S.; Cameron, M.; Papike, J.J.; Prewitt, C.T. The High Temperature Crystal Ghemistry of Tremolite. Am. Mineral. 1973, 58, 649–664. [Google Scholar]
- Pollastri, S.; Gualtieri, A.F.; Gualtieri, M.L.; Hanuskova, M.; Cavallo, A.; Gaudino, G. The zeta potential of mineral fibers. J. Hazard. Mater. 2014, 276, 469–479. [Google Scholar] [CrossRef] [PubMed]
- Snigirev, A.; Kohn, V.; Snigireva, I.; Lengeler, B. A compound refractive lens for focusing high-energy X-rays. Nature 1996, 384, 49–51. [Google Scholar] [CrossRef]
- Labiche, J.C.; Mathon, O.; Pascarelli, S.; Newton, M.A.; Guilera Ferre, G.; Curfs, C.; Vaughan, G.; Homs, A. The fast readout low noise camera as a versatile x-ray detector for time resolved dispersive extended x-ray absorption fine structure and diffraction studies of dynamic problems in materials science, chemistry, and catalysis. Rev. Sci. Instrum. 2007, 78, 09130. [Google Scholar] [CrossRef] [PubMed]
- Rigaku Oxford Diffraction Ltd. CrysAlis PRO; Rigaku Oxford Diffraction Ltd.: Yarnton, UK, 2015. [Google Scholar]
- Sheldrick, G.M. SHELX97—Program Suite for the Solution and Refinement of Crystal Structures (Release 97-2); University of Göttingen: Göttingen, Germany, 1998. [Google Scholar]
- Larson, A.C.; Von Dreele, R.B. Generalized Structure Analysis System (GSAS); Los Alamos National Lab: Los Alamos, NM, USA, 1994.
- Toby, B.H. EXPGUI, a graphical user interface for GSAS. J. App. Cryst. 2001, 34, 210–213. [Google Scholar] [CrossRef]
- Pollastri, S.; D’Acapito, F.; Trapanati, A.; Colantoni, I.; Andreozzi, G.B.; Gualtieri, A.F. The chemical environment of iron in mineral fibres. A combined X-ray absorption and Mössbauer spectroscopic study. J. Hazard. Mater. 2015, 298, 282–293. [Google Scholar]
- Pacella, A.; Andreozzi, G.B.; Ballirano, P.; Gianfagna, A. Crystal chemical and structural characterization of fibrous tremolite from Ala di Stura (Lanzo Valley, Italy). Period. Mineral. 2008, 77, 51–62. [Google Scholar]
- Fahrnbauer, F.; Rosenthal, T.; Schmutzler, T.; Wagner, G.; Vaughan, G.B.M.; Wright, J.P.; Oeckler, O. Discovery and Structure Determination of an Unusual Sulfide Telluride through an Effective Combination of TEM and Synchrotron Microdiffraction. Angew. Chem. 2015, 54, 10020–10023. [Google Scholar] [CrossRef] [PubMed]
- Hawthorne, F.C. Crystal chemistry of the amphiboles. Rev. Mineral. Geochem. 1981, 9A, 1–102. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised effective ionic radii andsystematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr. 1976, A32, 751–767. [Google Scholar] [CrossRef]
- Giacobbe, C.; Gualtieri, A.F.; Quartieri, S.; Rinaudo, C.; Allegrina, M.; Andreozzi, G.B. Spectroscopic study of the product of thermal transformation of chrysotile-asbestos containing materials (ACM). Eur. J. Mineral. 2010, 22, 535–546. [Google Scholar] [CrossRef]




| RT | 800 °C | |
|---|---|---|
| a (Å) | 9.8536(2) | 9.9044(2) |
| b (Å) | 18.0774(3) | 18.1311(2) |
| c (Å) | 5.2843(1) | 5.3009(3) |
| β (°) | 104.709(2) | 104.687(2) |
| V (Å3) | 910.44 | 917.1 |
| Space group | C2/m | C2/m |
| Size (µm) | 4.3 | 5.8 |
| GooF | 1.021 | 1.022 |
| R1 | 4.07 | 4.99 |
| wR2 | 0.1175 | 0.118 |
| Tremolite RT | Refinement | Possible Site Partition | Chemical Data |
|---|---|---|---|
| M(4) | 40.00 | ||
| B site sum | 40.00 | Ca1.91Na0.06K0.01 | 39.05 |
| M(1) | 25.23 | Mg1.884Fe2+0.088Mn0.02 | 25.40 |
| M(2) | 26.30 | Mg1.884Fe2+0.088Fe3+0.08 | 26.98 |
| M(3) | 12.62 | Mg0.942Fe2+0.044 | 12.44 |
| C sites sum | 64.15 | 64.82 |
| Site | ss | x/a | y/b | z/c | β11 | β22 | β33 | β12 | β13 | β23 | Beq |
|---|---|---|---|---|---|---|---|---|---|---|---|
| tremolite RT | |||||||||||
| M(4) | Ca | 0 | 0.27763(4) | 0.5 | 0.01141 | 0.01154 | 0.00881 | 0 | 0.00689 | 0 | 0.00981 |
| M(1) | Mg0.96Fe0.04 | 0 | 0.08763(6) | 0.5 | 0.00659 | 0.00786 | 0.00159 | 0 | 0.00197 | 0 | 0.00518 |
| M(2) | Mg0.92Fe0.08 | 0 | 0.17701(6) | 0 | 0.0056 | 0.0097 | 0.00412 | 0 | 0.00183 | 0 | 0.00636 |
| M(3) | Mg0.96Fe0.04 | 0 | 0 | 0 | 0.00646 | 0.01077 | 0.00341 | 0 | 0.00116 | 0 | 0.0069 |
| T(1) | Si | 0.28028(7) | 0.0841(4) | 0.29657(1) | 0.00479 | 0.00775 | 0.00248 | 0.00012 | 0.0012 | 0.00016 | 0.00496 |
| T(2) | Si | 0.2883(7) | 0.17109(4) | 0.80399(1) | 0.00523 | 0.00834 | 0.00201 | 0.00009 | 0.00134 | 0.00099 | 0.00512 |
| O(1) | O | 0.111(1) | 0.0862(1) | 0.2175(2) | 0.00667 | 0.00987 | 0.00183 | 0.00005 | 0.00168 | 0.00034 | 0.00602 |
| O(2) | O | 0.1188(2) | 0.1712(1) | 0.7243(3) | 0.00755 | 0.01017 | 0.00307 | 0.00044 | 0.00285 | 0.00083 | 0.00666 |
| O(3) | O | 0.1113(2) | 0 | 0.7181(3) | 0.00832 | 0.00911 | 0.00363 | 0 | 0.00251 | 0 | 0.00684 |
| O(4) | O | 0.3644(2) | 0.2476(1) | 0.7912(3) | 0.00947 | 0.00985 | 0.00691 | 0.00017 | 0.00319 | 0.00245 | 0.00854 |
| O(5) | O | 0.3462(2) | 0.1337(1) | 0.0988(3) | 0.00713 | 0.01187 | 0.00474 | 0.00367 | 0.00128 | 0.00068 | 0.00795 |
| O(6) | O | 0.3435(2) | 0.1185(1) | 0.5889(3) | 0.00601 | 0.0128 | 0.00484 | 0.00305 | 0.00175 | 0 | 0.00782 |
| O(7) | O | 0.3364(3) | 0 | 0.2922(1) | 0.0098 | 0.00926 | 0.00986 | 0 | 0.00285 | 0 | 0.00958 |
| H * | H | 0.206 | 0 | 0.771 | 0.03 | ||||||
| tremolite 800 °C | |||||||||||
| M(4) | Ca | 0 | 0.27742(5) | 0.5 | 0.01916 | 0.01465 | 0.01425 | 0 | 0.00871 | 0 | 0.01521 |
| M(1) | Mg0.96Fe0.04 | 0 | 0.08723(6) | 0.5 | 0.00758 | 0.00968 | 0.00366 | 0 | 0.00283 | 0 | 0.00672 |
| M(2) | Mg0.92Fe0.08 | 0 | 0.17679(7) | 0 | 0.01146 | 0.01112 | 0.00792 | 0 | 0.00303 | 0 | 0.01006 |
| M(3) | Mg0.96Fe0.04 | 0 | 0 | 0 | 0.01448 | 0.00967 | 0.00675 | 0 | 0.00159 | 0 | 0.0105 |
| T(1) | Si | 0.28112(9) | 0.08405(4) | 0.29629(1) | 0.01099 | 0.00973 | 0.00652 | 0.00027 | 0.00237 | 0.00022 | 0.00905 |
| T(2) | Si | 0.28851(9) | 0.17096(4) | 0.80355(2) | 0.01159 | 0.0102 | 0.00668 | 0.00014 | 0.00257 | 0.00086 | 0.00944 |
| O(1) | O | 0.1121(2) | 0.0860(1) | 0.218(3) | 0.01285 | 0.0115 | 0.00884 | 0.00028 | 0.00376 | 0.00003 | 0.01088 |
| O(2) | O | 0.1187(2) | 0.1707(1) | 0.7236(4) | 0.01348 | 0.01295 | 0.00888 | 0.00067 | 0.00385 | 0.00085 | 0.01158 |
| O(3) | O | 0.1132(3) | 0 | 0.7213(4) | 0.01888 | 0.01288 | 0.01211 | 0 | 0.00542 | 0 | 0.01435 |
| O(4) | O | 0.3635(2) | 0.2479(1) | 0.7904(4) | 0.01596 | 0.01214 | 0.01132 | 0.00102 | 0.00414 | 0.00227 | 0.01302 |
| O(5) | O | 0.3468(2) | 0.1339(1) | 0.0985(4) | 0.013 | 0.01615 | 0.00913 | 0.00307 | 0.00313 | 0.00031 | 0.0127 |
| O(6) | O | 0.3444(2) | 0.1188(1) | 0.5877(3) | 0.01307 | 0.01475 | 0.00883 | 0.00251 | 0.00285 | 0.00051 | 0.0122 |
| O(7) | O | 0.3373(3) | 0 | 0.2925(5) | 0.01578 | 0.01021 | 0.01607 | 0 | 0.00525 | 0 | 0.0138 |
| H * | H | 0.206 | 0 | 0.771 | 0.03 |
| RT | 800 °C | RT | 800 °C | ||||
|---|---|---|---|---|---|---|---|
| T(1) | -O1 | 1.604592) | 1.6124(2) | T(2) | -O2 | 1.6147(2) | 1.6201(2) |
| T(1) | -O5 | 1.6325(1) | 1.6377(2) | T(2) | -O4 | 1.5839(2) | 1.5895(1) |
| T(1) | -O6 | 1.6357(2) | 1.6391(2) | T(2) | -O5 | 1.6605(2) | 1.6644(2) |
| T(1) | -O7 | 1.6202(2) | 1.6236(1) | T(2) | -O6 | 1.674(2) | 1.6807(2) |
| average | 1.623225 | 1.6282 | average | 1.633275 | 1.63867 | ||
| Δ | 0.03483 | 0.079895 | Δ | 6.217863 | 6.577533 | ||
| M(1) | -O1 ×2 | 2.0693(2) | 2.0707(2) | M(2) | -O1 ×2 | 2.1418(2) | 2.1499(2) |
| M(1) | -O2 ×2 | 2.0874(3) | 2.1063(2) | M(2) | -O2 ×2 | 2.0908(2) | 2.0974(2) |
| M(1) | -O3 ×3 | 2.0977(2) | 2.0946(1) | M(2) | -O4 ×2 | 2.0265(2) | 2.0374(2) |
| average | 2.0848 | 2.090533 | average | 2.086367 | 2.0949 | ||
| Δ | 0.31706 | 0.50224 | Δ | 5.112674 | 4.813601 | ||
| M(3) | -O1 ×4 | 2.0798(3) | 2.0862(3) | M(4) | -O2 ×2 | 2.4024(2) | 2.4116(3) |
| M(3) | -O3 ×2 | 2.0623(2) | 2.0658(2) | M(4) | -O4 ×2 | 2.3248(3) | 2.3334(2) |
| M(4) | -O5 ×2 | 2.779(2) | 2.7863(3) | ||||
| M(4) | -O6 ×2 | 2.5466(3) | 2.5438(2) | ||||
| average | 2.07105 | 2.076 | average | 2.5132 | 2.518775 | ||
| Δ | 0.178499 | 0.241405 | Δ | 44.88401 | 44.25401 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giacobbe, C.; Wright, J.P.; Di Giuseppe, D.; Zoboli, A.; Zapparoli, M.; Gualtieri, A.F. Synchrotron Nano-Diffraction Study of Thermally Treated Asbestos Tremolite from Val d’Ala, Turin (Italy). Minerals 2018, 8, 311. https://doi.org/10.3390/min8080311
Giacobbe C, Wright JP, Di Giuseppe D, Zoboli A, Zapparoli M, Gualtieri AF. Synchrotron Nano-Diffraction Study of Thermally Treated Asbestos Tremolite from Val d’Ala, Turin (Italy). Minerals. 2018; 8(8):311. https://doi.org/10.3390/min8080311
Chicago/Turabian StyleGiacobbe, Carlotta, Jonathan P. Wright, Dario Di Giuseppe, Alessandro Zoboli, Mauro Zapparoli, and Alessandro F. Gualtieri. 2018. "Synchrotron Nano-Diffraction Study of Thermally Treated Asbestos Tremolite from Val d’Ala, Turin (Italy)" Minerals 8, no. 8: 311. https://doi.org/10.3390/min8080311





