Communication between B-Cells and Microbiota for the Maintenance of Intestinal Homeostasis
Abstract
:1. Introduction
2. Differentiation of B-Cells and Generation of IgA Induced by Gut Microbiota
2.1. Evidence of the Participation of Enteric Bacteria in the Differentiation and Activation of B-Cells in Animals and Humans
2.2. Generation of IgA-Producing B-Cells in the Gut-Associated Lymphoid Tissues
2.2.1. Generation of IgA-Producing B-Cells in Peyer’s Patches
2.2.2. Generation of IgA-Producing B-Cells in Isolated Lymphoid Follicles (ILFs) and IgA-Producing Plasma Cells in the Lamina Propria (LP)

3. Control of Gut Microbiota by Intestinal IgA
3.1. Limited Access of Bacteria in the Intestinal Lumen to Intestinal Tissues Mediated by IgA
3.2. Regulation of Bacterial Communities in the Gut Lumen by Secretory IgA
3.3. IgA-Based Immune Complexes As Regulators of Intestinal Homeostasis
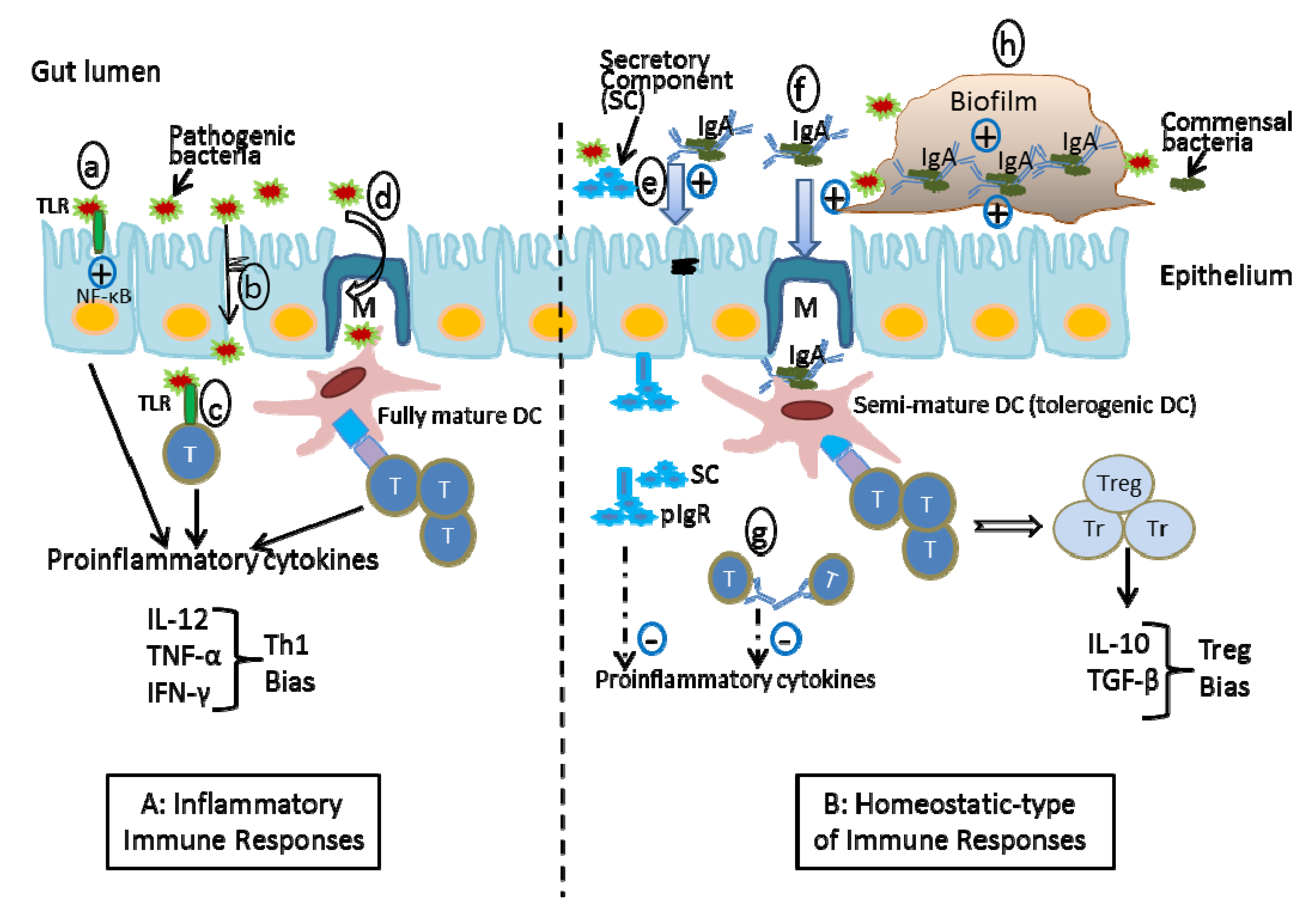
4. The Effect of Probiotics on Intestinal IgA Levels
5. Summary
Acknowledgements
Conflicts of Interest
References
- Sonnenburg, J.L.; Angenent, L.T.; Gordon, J.I. Getting a Grip on Things: How Do Communities of Bacterial Symbionts Become Established in Our Intestine? Nat. Immunol. 2004, 5, 569–573. [Google Scholar] [CrossRef]
- Turnbaugh, P.J.; Ley, R.E.; Hamady, M.; Fraser-Liggett, C.M.; Knight, R.; Gordon, J.I. The Human Microbiome Project. Nature 2007, 449, 804–810. [Google Scholar]
- O'Hara, A.M.; Shanahan, F. The Gut Flora As a Forgotten Organ. EMBO Rep. 2006, 7, 688–693. [Google Scholar] [CrossRef]
- Suzuki, K.; Ha, S.A.; Tsuji, M.; Fagarasan, S. Intestinal IgA Synthesis: A Primitive Form of Adaptive Immunity That Regulates Microbial Communities in the Gut. Semin. Immunol. 2007, 19, 127–135. [Google Scholar] [CrossRef]
- Rakoff-Nahoum, S.; Paglino, J.; Eslami-Varzaneh, F.; Edberg, S.; Medzhitov, R. Recognition of Commensal Microflora by Toll-Like Receptors Is Required for Intestinal Homeostasis. Cell 2004, 118, 229–241. [Google Scholar]
- Ouellette, A.J. Paneth Cells and Innate Immunity in the Crypt Microenvironment. Gastroenterology 1997, 113, 1779–1784. [Google Scholar] [CrossRef]
- Brandl, K.; Plitas, G.; Schnabl, B.; DeMatteo, R.P.; Pamer, E.G. MyD88-Mediated Signals Induce the Bactericidal Lectin RegIII Gamma and Protect Mice Against Intestinal Listeria Monocytogenes Infection. J. Exp. Med. 2007, 204, 1891–1900. [Google Scholar]
- Hooper, L.V.; Stappenbeck, T.S.; Hong, C.V.; Gordon, J.I. Angiogenins: A New Class of Microbicidal Proteins Involved in Innate Immunity. Nat. Immunol. 2003, 4, 269–273. [Google Scholar]
- Salzman, N.H.; Underwood, M.A.; Bevins, C.L. Paneth Cells, Defensins, and the Commensal Microbiota: A Hypothesis on Intimate Interplay at the Intestinal Mucosa. Semin. Immunol. 2007, 19, 70–83. [Google Scholar]
- Golovkina, T.V.; Shlomchik, M.; Hannum, L.; Chervonsky, A. Organogenic Role of B Lymphocytes in Mucosal Immunity. Science 1999, 286, 1965–1968. [Google Scholar]
- Mizoguchi, A.; Mizoguchi, E.; Takedatsu, H.; Blumberg, R.S.; Bhan, A.K. Chronic Intestinal Inflammatory Condition Generates IL-10-Producing Regulatory B Cell Subset Characterized by CD1d Upregulation. Immunity 2002, 16, 219–230. [Google Scholar]
- Cerutti, A.; Chen, K.; Chorny, A. Immunoglobulin Responses at the Mucosal Interface. Annu. Rev. Immunol. 2011, 29, 273–293. [Google Scholar]
- Macpherson, A.J.; McCoy, K.D.; Johansen, F.E.; Brandtzaeg, P. The Immune Geography of IgA Induction and Function. Mucosal. Immunol. 2008, 1, 11–22. [Google Scholar]
- van der Heijden, P.J.; Stok, W.; Bianchi, A.T. Contribution of Immunoglobulin-Secreting Cells in the Murine Small Intestine to the Total 'Background' Immunoglobulin Production. Immunology 1987, 62, 551–555. [Google Scholar]
- Apter, F.M.; Lencer, W.I.; Finkelstein, R.A.; Mekalanos, J.J.; Neutra, M.R. Monoclonal Immunoglobulin A Antibodies Directed Against Cholera Toxin Prevent the Toxin-Induced Chloride Secretory Response and Block Toxin Binding to Intestinal Epithelial Cells in Vitro. Infect. Immun. 1993, 61, 5271–5278. [Google Scholar]
- Helander, A.; Miller, C.L.; Myers, K.S.; Neutra, M.R.; Nibert, M.L. Protective Immunoglobulin A and G Antibodies Bind to Overlapping Intersubunit Epitopes in the Head Domain of Type 1 Reovirus Adhesin Sigma1. J. Virol. 2004, 78, 10695–10705. [Google Scholar]
- Mantis, N.J.; McGuinness, C.R.; Sonuyi, O.; Edwards, G.; Farrant, S.A. Immunoglobulin A Antibodies Against Ricin A and B Subunits Protect Epithelial Cells From Ricin Intoxication. Infect. Immun. 2006, 74, 3455–3462. [Google Scholar] [CrossRef]
- Hutchings, A.B.; Helander, A.; Silvey, K.J.; Chandran, K.; Lucas, W.T.; Nibert, M.L.; Neutra, M.R. Secretory Immunoglobulin A Antibodies Against the Sigma1 Outer Capsid Protein of Reovirus Type 1 Lang Prevent Infection of Mouse Peyer's Patches. J. Virol. 2004, 78, 947–957. [Google Scholar] [CrossRef]
- Deplancke, B.; Gaskins, H.R. Microbial Modulation of Innate Defense: Goblet Cells and the Intestinal Mucus Layer. Am. J. Clin. Nutr. 2001, 73, 1131S–1141S. [Google Scholar]
- Lievin-Le, M.V.; Servin, A.L. The Front Line of Enteric Host Defense Against Unwelcome Intrusion of Harmful Microorganisms: Mucins, Antimicrobial Peptides, and Microbiota. Clin. Microbiol. Rev. 2006, 19, 315–337. [Google Scholar] [CrossRef]
- Mantis, N.J.; Forbes, S.J. Secretory IgA: Arresting Microbial Pathogens at Epithelial Borders. Immunol. Invest 2010, 39, 383–406. [Google Scholar] [CrossRef]
- Shikina, T.; Hiroi, T.; Iwatani, K.; Jang, M.H.; Fukuyama, S.; Tamura, M.; Kubo, T.; Ishikawa, H.; Kiyono, H. IgA Class Switch Occurs in the Organized Nasopharynx- and Gut-Associated Lymphoid Tissue, but Not in the Diffuse Lamina Propria of Airways and Gut. J. Immunol. 2004, 172, 6259–6264. [Google Scholar]
- Bealmear, P.M.; Mirand, E.A.; Holtermann, O.A. Miscellaneous Immune Defects in Gnotobiotic and SPF Mice. Prog. Clin. Biol. Res. 1983, 132C, 423–432. [Google Scholar]
- Ouwehand, A.; Isolauri, E.; Salminen, S. The Role of the Intestinal Microflora for the Development of the Immune System in Early Childhood. Eur. J. Nutr. 2002, 41, I32–I37. [Google Scholar]
- Macpherson, A.J.; Uhr, T. Induction of Protective IgA by Intestinal Dendritic Cells Carrying Commensal Bacteria. Science 2004, 303, 1662–1665. [Google Scholar] [CrossRef]
- Shulzhenko, N.; Morgun, A.; Hsiao, W.; Battle, M.; Yao, M.; Gavrilova, O.; Orandle, M.; Mayer, L.; Macpherson, A.J.; McCoy, K.D.; et al. Crosstalk Between B Lymphocytes, Microbiota and the Intestinal Epithelium Governs Immunity Versus Metabolism in the Gut. Nat. Med. 2011, 17, 1585–1593. [Google Scholar] [CrossRef]
- Fagarasan, S.; Muramatsu, M.; Suzuki, K.; Nagaoka, H.; Hiai, H.; Honjo, T. Critical Roles of Activation-Induced Cytidine Deaminase in the Homeostasis of Gut Flora. Science 2002, 298, 1424–1427. [Google Scholar] [CrossRef]
- Wei, B.; Su, T.T.; Dalwadi, H.; Stephan, R.P.; Fujiwara, D.; Huang, T.T.; Brewer, S.; Chen, L.; Arditi, M.; Borneman, J.; et al. Resident Enteric Microbiota and CD8+ T Cells Shape the Abundance of Marginal Zone B Cells. Eur. J. Immunol. 2008, 38, 3411–3425. [Google Scholar] [CrossRef]
- Lundell, A.C.; Bjornsson, V.; Ljung, A.; Ceder, M.; Johansen, S.; Lindhagen, G.; Tornhage, C.J.; Adlerberth, I.; Wold, A.E.; Rudin, A. Infant B Cell Memory Differentiation and Early Gut Bacterial Colonization. J. Immunol. 2012, 188, 4315–4322. [Google Scholar] [CrossRef]
- Rudin, A.; Lundell, A.C. Infant B Cell Memory and Gut Bacterial Colonization. Gut Microbes. 2012, 3, 474–475. [Google Scholar]
- Mellander, L.; Carlsson, B.; Jalil, F.; Soderstrom, T.; Hanson, L.A. Secretory IgA Antibody Response Against Escherichia Coli Antigens in Infants in Relation to Exposure. J. Pediatr. 1985, 107, 430–433. [Google Scholar] [CrossRef]
- Adlerberth, I.; Carlsson, B.; de, M.P.; Jalil, F.; Khan, S.R.; Larsson, P.; Mellander, L.; Svanborg, C.; Wold, A.E.; Hanson, L.A. Intestinal Colonization With Enterobacteriaceae in Pakistani and Swedish Hospital-Delivered Infants. Acta Paediatr. Scand. 1991, 80, 602–610. [Google Scholar] [CrossRef]
- Adlerberth, I.; Wold, A.E. Establishment of the Gut Microbiota in Western Infants. Acta Paediatr. 2009, 98, 229–238. [Google Scholar] [CrossRef]
- Nowrouzian, F.; Hesselmar, B.; Saalman, R.; Strannegard, I.L.; Aberg, N.; Wold, A.E.; Adlerberth, I. Escherichia Coli in Infants' Intestinal Microflora: Colonization Rate, Strain Turnover, and Virulence Gene Carriage. Pediatr. Res. 2003, 54, 8–14. [Google Scholar] [CrossRef]
- Janzi, M.; Kull, I.; Sjoberg, R.; Wan, J.; Melen, E.; Bayat, N.; Ostblom, E.; Pan-Hammarstrom, Q.; Nilsson, P.; Hammarstrom, L. Selective IgA Deficiency in Early Life: Association to Infections and Allergic Diseases During Childhood. Clin. Immunol. 2009, 133, 78–85. [Google Scholar] [CrossRef]
- Malamut, G.; Verkarre, V.; Suarez, F.; Viallard, J.F.; Lascaux, A.S.; Cosnes, J.; Bouhnik, Y.; Lambotte, O.; Bechade, D.; Ziol, M.; et al. The Enteropathy Associated With Common Variable Immunodeficiency: the Delineated Frontiers With Celiac Disease. Am. J. Gastroenterol. 2010, 105, 2262–2275. [Google Scholar] [CrossRef]
- Meini, A.; Pillan, N.M.; Villanacci, V.; Monafo, V.; Ugazio, A.G.; Plebani, A. Prevalence and Diagnosis of Celiac Disease in IgA-Deficient Children. Ann. Allergy Asthma Immunol. 1996, 77, 333–336. [Google Scholar] [CrossRef]
- Hamada, H.; Hiroi, T.; Nishiyama, Y.; Takahashi, H.; Masunaga, Y.; Hachimura, S.; Kaminogawa, S.; Takahashi-Iwanaga, H.; Iwanaga, T.; Kiyono, H.; et al. Identification of Multiple Isolated Lymphoid Follicles on the Antimesenteric Wall of the Mouse Small Intestine. J. Immunol. 2002, 168, 57–64. [Google Scholar]
- Randall, T.D.; Carragher, D.M.; Rangel-Moreno, J. Development of Secondary Lymphoid Organs. Annu. Rev. Immunol. 2008, 26, 627–650. [Google Scholar] [CrossRef]
- Nishikawa, S.; Honda, K.; Vieira, P.; Yoshida, H. Organogenesis of Peripheral Lymphoid Organs. Immunol. Rev. 2003, 195, 72–80. [Google Scholar] [CrossRef]
- King, C.; Tangye, S.G.; Mackay, C.R. T Follicular Helper (TFH) Cells in Normal and Dysregulated Immune Responses. Annu. Rev. Immunol. 2008, 26, 741–766. [Google Scholar] [CrossRef]
- Casola, S.; Otipoby, K.L.; Alimzhanov, M.; Humme, S.; Uyttersprot, N.; Kutok, J.L.; Carroll, M.C.; Rajewsky, K. B Cell Receptor Signal Strength Determines B Cell Fate. Nat. Immunol. 2004, 5, 317–327. [Google Scholar]
- Cebra, J.J. Influences of Microbiota on Intestinal Immune System Development. Am. J. Clin. Nutr. 1999, 69, 1046S–1051S. [Google Scholar]
- Muramatsu, M.; Kinoshita, K.; Fagarasan, S.; Yamada, S.; Shinkai, Y.; Honjo, T. Class Switch Recombination and Hypermutation Require Activation-Induced Cytidine Deaminase (AID), a Potential RNA Editing Enzyme. Cell 2000, 102, 553–563. [Google Scholar] [CrossRef]
- Chieppa, M.; Rescigno, M.; Huang, A.Y.; Germain, R.N. Dynamic Imaging of Dendritic Cell Extension into the Small Bowel Lumen in Response to Epithelial Cell TLR Engagement. J. Exp. Med. 2006, 203, 2841–2852. [Google Scholar] [CrossRef]
- Hooper, L.V.; Macpherson, A.J. Immune Adaptations That Maintain Homeostasis With the Intestinal Microbiota. Nat. Rev. Immunol. 2010, 10, 159–169. [Google Scholar] [CrossRef]
- Sato, A.; Hashiguchi, M.; Toda, E.; Iwasaki, A.; Hachimura, S.; Kaminogawa, S. CD11b+ Peyer's Patch Dendritic Cells Secrete IL-6 and Induce IgA Secretion From Naive B Cells. J. Immunol. 2003, 171, 3684–3690. [Google Scholar]
- Fan, H.; Rothstein, T.L. Lymphokine Dependence of STAT3 Activation Produced by Surface Immunoglobulin Cross-Linking and by Phorbol Ester Plus Calcium Ionophore Treatment in B Cells. Eur. J. Immunol. 2001, 31, 665–671. [Google Scholar] [CrossRef]
- Weber-Nordt, R.M.; Riley, J.K.; Greenlund, A.C.; Moore, K.W.; Darnell, J.E.; Schreiber, R.D. Stat3 Recruitment by Two Distinct Ligand-Induced, Tyrosine-Phosphorylated Docking Sites in the Interleukin-10 Receptor Intracellular Domain. J. Biol. Chem. 1996, 271, 27954–27961. [Google Scholar]
- Lafarge, S.; Hamzeh-Cognasse, H.; Richard, Y.; Pozzetto, B.; Cogne, M.; Cognasse, F.; Garraud, O. Complexes Between Nuclear Factor-KappaB P65 and Signal Transducer and Activator of Transcription 3 Are Key Actors in Inducing Activation-Induced Cytidine Deaminase Expression and Immunoglobulin A Production in CD40L Plus Interleukin-10-Treated Human Blood B Cells. Clin. Exp. Immunol. 2011, 166, 171–183. [Google Scholar] [CrossRef]
- Macpherson, A.J.; Lamarre, A.; McCoy, K.; Harriman, G.R.; Odermatt, B.; Dougan, G.; Hengartner, H.; Zinkernagel, R.M. IgA Production Without Mu or Delta Chain Expression in Developing B Cells. Nat. Immunol. 2001, 2, 625–631. [Google Scholar] [CrossRef]
- Taylor, R.T.; Patel, S.R.; Lin, E.; Butler, B.R.; Lake, J.G.; Newberry, R.D.; Williams, I.R. Lymphotoxin-Independent Expression of TNF-Related Activation-Induced Cytokine by Stromal Cells in Cryptopatches, Isolated Lymphoid Follicles, and Peyer's Patches. J. Immunol. 2007, 178, 5659–5667. [Google Scholar]
- Tsuji, M.; Suzuki, K.; Kitamura, H.; Maruya, M.; Kinoshita, K.; Ivanov, I.I.; Itoh, K.; Littman, D.R.; Fagarasan, S. Requirement for Lymphoid Tissue-Inducer Cells in Isolated Follicle Formation and T Cell-Independent Immunoglobulin A Generation in the Gut. Immunity 2008, 29, 261–271. [Google Scholar] [CrossRef]
- Lorenz, R.G.; Chaplin, D.D.; McDonald, K.G.; McDonough, J.S.; Newberry, R.D. Isolated Lymphoid Follicle Formation Is Inducible and Dependent Upon Lymphotoxin-Sufficient B Lymphocytes, Lymphotoxin Beta Receptor, and TNF Receptor I Function. J. Immunol. 2003, 170, 5475–5482. [Google Scholar]
- Gardby, E.; Kagrdic, D.; Kjerrulf, M.; Bromander, A.; Vajdy, M.; Hornquist, E.; Lycke, N. The Influence of Costimulation and Regulatory CD4+ T Cells on Intestinal IgA Immune Responses. Dev. Immunol. 1998, 6, 53–60. [Google Scholar] [CrossRef]
- Tezuka, H.; Abe, Y.; Iwata, M.; Takeuchi, H.; Ishikawa, H.; Matsushita, M.; Shiohara, T.; Akira, S.; Ohteki, T. Regulation of IgA Production by Naturally Occurring TNF/INOS-Producing Dendritic Cells. Nature 2007, 448, 929–933. [Google Scholar] [CrossRef]
- He, B.; Xu, W.; Santini, P.A.; Polydorides, A.D.; Chiu, A.; Estrella, J.; Shan, M.; Chadburn, A.; Villanacci, V.; Plebani, A.; et al. Intestinal Bacteria Trigger T Cell-Independent Immunoglobulin A(2) Class Switching by Inducing Epithelial-Cell Secretion of the Cytokine APRIL. Immunity 2007, 26, 812–826. [Google Scholar] [CrossRef]
- Mackay, F.; Schneider, P. TACI, an Enigmatic BAFF/APRIL Receptor, With New Unappreciated Biochemical and Biological Properties. Cytokine Growth Factor Rev. 2008, 19, 263–276. [Google Scholar] [CrossRef]
- Tsuji, M.; Suzuki, K.; Kinoshita, K.; Fagarasan, S. Dynamic Interactions Between Bacteria and Immune Cells Leading to Intestinal IgA Synthesis. Semin. Immunol. 2008, 20, 59–66. [Google Scholar] [CrossRef]
- Mackay, F.; Leung, H. The Role of the BAFF/APRIL System on T Cell Function. Semin. Immunol. 2006, 18, 284–289. [Google Scholar] [CrossRef]
- Pabst, O. New Concepts in the Generation and Functions of IgA. Nat. Rev. Immunol. 2012, 12, 821–832. [Google Scholar] [CrossRef]
- Fritz, J.H.; Rojas, O.L.; Simard, N.; McCarthy, D.D.; Hapfelmeier, S.; Rubino, S.; Robertson, S.J.; Larijani, M.; Gosselin, J.; Ivanov, I.I.; et al. Acquisition of a Multifunctional IgA+ Plasma Cell Phenotype in the Gut. Nature 2012, 481, 199–203. [Google Scholar]
- Berlin, C.; Berg, E.L.; Briskin, M.J.; Andrew, D.P.; Kilshaw, P.J.; Holzmann, B.; Weissman, I.L.; Hamann, A.; Butcher, E.C. Alpha 4 Beta 7 Integrin Mediates Lymphocyte Binding to the Mucosal Vascular Addressin MAdCAM-1. Cell 1993, 74, 185–195. [Google Scholar] [CrossRef]
- Bowman, E.P.; Kuklin, N.A.; Youngman, K.R.; Lazarus, N.H.; Kunkel, E.J.; Pan, J.; Greenberg, H.B.; Butcher, E.C. The Intestinal Chemokine Thymus-Expressed Chemokine (CCL25) Attracts IgA Antibody-Secreting Cells. J. Exp. Med. 2002, 195, 269–275. [Google Scholar] [CrossRef]
- Sekirov, I.; Russell, S.L.; Antunes, L.C.; Finlay, B.B. Gut Microbiota in Health and Disease. Physiol. Rev. 2010, 90, 859–904. [Google Scholar] [CrossRef]
- Walter, J.; Ley, R. The Human Gut Microbiome: Ecology and Recent Evolutionary Changes. Annu. Rev. Microbiol. 2011, 65, 411–429. [Google Scholar] [CrossRef]
- Slack, E.; Balmer, M.L.; Fritz, J.H.; Hapfelmeier, S. Functional Flexibility of Intestinal IgA—Broadening the Fine Line. Front. Immunol. 2012, 3, 100. [Google Scholar]
- Harris, N.L.; Spoerri, I.; Schopfer, J.F.; Nembrini, C.; Merky, P.; Massacand, J.; Urban, J.F., Jr.; Lamarre, A.; Burki, K.; Odermatt, B.; et al. Mechanisms of Neonatal Mucosal Antibody Protection. J. Immunol. 2006, 177, 6256–6262. [Google Scholar]
- Peterson, D.A.; McNulty, N.P.; Guruge, J.L.; Gordon, J.I. IgA Response to Symbiotic Bacteria As a Mediator of Gut Homeostasis. Cell Host Microbe 2007, 2, 328–339. [Google Scholar] [CrossRef]
- Fernandez, M.I.; Pedron, T.; Tournebize, R.; Olivo-Marin, J.C.; Sansonetti, P.J.; Phalipon, A. Anti-Inflammatory Role for Intracellular Dimeric Immunoglobulin a by Neutralization of Lipopolysaccharide in Epithelial Cells. Immunity 2003, 18, 739–749. [Google Scholar] [CrossRef]
- Suzuki, K.; Meek, B.; Doi, Y.; Muramatsu, M.; Chiba, T.; Honjo, T.; Fagarasan, S. Aberrant Expansion of Segmented Filamentous Bacteria in IgA-Deficient Gut. Proc. Natl. Acad. Sci. USA 2004, 101, 1981–1986. [Google Scholar]
- Hapfelmeier, S.; Lawson, M.A.; Slack, E.; Kirundi, J.K.; Stoel, M.; Heikenwalder, M.; Cahenzli, J.; Velykoredko, Y.; Balmer, M.L.; Endt, K.; et al. Reversible Microbial Colonization of Germ-Free Mice Reveals the Dynamics of IgA Immune Responses. Science 2010, 328, 1705–1709. [Google Scholar] [CrossRef]
- Tsuruta, T.; Inoue, R.; Nojima, I.; Tsukahara, T.; Hara, H.; Yajima, T. The Amount of Secreted IgA May Not Determine the Secretory IgA Coating Ratio of Gastrointestinal Bacteria. FEMS Immunol. Med. Microbiol. 2009, 56, 185–189. [Google Scholar] [CrossRef]
- van der Waaij, L.A.; Limburg, P.C.; Mesander, G.; van der Waaij, D. In Vivo IgA Coating of Anaerobic Bacteria in Human Faeces. Gut 1996, 38, 348–354. [Google Scholar] [CrossRef]
- Mestecky, J.; Russell, M.W. Specific Antibody Activity, Glycan Heterogeneity and Polyreactivity Contribute to the Protective Activity of S-IgA at Mucosal Surfaces. Immunol. Lett. 2009, 124, 57–62. [Google Scholar] [CrossRef]
- Brandtzaeg, P. Homeostatic Impact of Indigenous Microbiota and Secretory Immunity. Benef. Microbes. 2010, 1, 211–227. [Google Scholar] [CrossRef]
- Johansen, F.E.; Kaetzel, C.S. Regulation of the Polymeric Immunoglobulin Receptor and IgA Transport: New Advances in Environmental Factors That Stimulate PIgR Expression and Its Role in Mucosal Immunity. Mucosal. Immunol. 2011, 4, 598–602. [Google Scholar] [CrossRef]
- Bruno, M.E.; Frantz, A.L.; Rogier, E.W.; Johansen, F.E.; Kaetzel, C.S. Regulation of the Polymeric Immunoglobulin Receptor by the Classical and Alternative NF-KappaB Pathways in Intestinal Epithelial Cells. Mucosal. Immunol. 2011, 4, 468–478. [Google Scholar] [CrossRef]
- Roopenian, D.C.; Akilesh, S. FcRn: the Neonatal Fc Receptor Comes of Age. Nat. Rev. Immunol. 2007, 7, 715–725. [Google Scholar] [CrossRef]
- Hase, K.; Kawano, K.; Nochi, T.; Pontes, G.S.; Fukuda, S.; Ebisawa, M.; Kadokura, K.; Tobe, T.; Fujimura, Y.; Kawano, S.; et al. Uptake Through Glycoprotein 2 of FimH(+) Bacteria by M Cells Initiates Mucosal Immune Response. Nature 2009, 462, 226–230. [Google Scholar] [CrossRef]
- Neutra, M.R.; Kraehenbuhl, J.P. Transepithelial Transport and Mucosal Defence I: The Role of M Cells. Trends Cell Biol. 1992, 2, 134–138. [Google Scholar] [CrossRef]
- Mantis, N.J.; Rol, N.; Corthesy, B. Secretory IgA's Complex Roles in Immunity and Mucosal Homeostasis in the Gut. Mucosal. Immunol. 2011, 4, 603–611. [Google Scholar] [CrossRef]
- Rey, J.; Garin, N.; Spertini, F.; Corthesy, B. Targeting of Secretory IgA to Peyer's Patch Dendritic and T Cells After Transport by Intestinal M Cells. J. Immunol. 2004, 172, 3026–3033. [Google Scholar]
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial Biofilms: A Common Cause of Persistent Infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef]
- Costerton, J.W.; Lewandowski, Z.; Caldwell, D.E.; Korber, D.R.; Lappin-Scott, H.M. Microbial Biofilms. Annu. Rev. Microbiol. 1995, 49, 711–745. [Google Scholar] [CrossRef]
- Bollinger, R.R.; Everett, M.L.; Palestrant, D.; Love, S.D.; Lin, S.S.; Parker, W. Human Secretory Immunoglobulin A May Contribute to Biofilm Formation in the Gut. Immunology 2003, 109, 580–587. [Google Scholar] [CrossRef]
- Bollinger, R.R.; Everett, M.L.; Wahl, S.D.; Lee, Y.H.; Orndorff, P.E.; Parker, W. Secretory IgA and Mucin-Mediated Biofilm Formation by Environmental Strains of Escherichia Coli: Role of Type 1 Pili. Mol. Immunol. 2006, 43, 378–387. [Google Scholar] [CrossRef]
- Palestrant, D.; Holzknecht, Z.E.; Collins, B.H.; Parker, W.; Miller, S.E.; Bollinger, R.R. Microbial Biofilms in the Gut: Visualization by Electron Microscopy and by Acridine Orange Staining. Ultrastruct. Pathol. 2004, 28, 23–27. [Google Scholar]
- Friman, V.; Adlerberth, I.; Connell, H.; Svanborg, C.; Hanson, L.A.; Wold, A.E. Decreased Expression of Mannose-Specific Adhesins by Escherichia Coli in the Colonic Microflora of Immunoglobulin A-Deficient Individuals. Infect. Immun. 1996, 64, 2794–2798. [Google Scholar]
- Liu, Y.; Fatheree, N.Y.; Mangalat, N.; Rhoads, J.M. Lactobacillus Reuteri Strains Reduce Incidence and Severity of Experimental Necrotizing Enterocolitis Via Modulation of TLR4 and NFkappaB Signaling in the Intestine. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 302, G608–G617. [Google Scholar] [CrossRef]
- Neu, J.; Walker, W.A. Necrotizing Enterocolitis. N. Engl. J. Med. 2011, 364, 255–264. [Google Scholar] [CrossRef]
- Huang, J.S.; Bousvaros, A.; Lee, J.W.; Diaz, A.; Davidson, E.J. Efficacy of Probiotic Use in Acute Diarrhea in Children: a Meta-Analysis. Dig. Dis. Sci. 2002, 47, 2625–2634. [Google Scholar] [CrossRef]
- Rea, M.C.; Alemayehu, D.; Ross, R.P.; Hill, C. Gut Solutions to a Gut Problem: Bacteriocins, Probiotics and Bacteriophage for Control of Clostridium Difficile Infection. J. Med. Microbiol. 2013, 62, 1369–1378. [Google Scholar] [CrossRef]
- Ohland, C.L.; MacNaughton, W.K. Probiotic Bacteria and Intestinal Epithelial Barrier Function. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 298, G807–G819. [Google Scholar] [CrossRef]
- Hart, A.L.; Lammers, K.; Brigidi, P.; Vitali, B.; Rizzello, F.; Gionchetti, P.; Campieri, M.; Kamm, M.A.; Knight, S.C.; Stagg, A.J. Modulation of Human Dendritic Cell Phenotype and Function by Probiotic Bacteria. Gut 2004, 53, 1602–1609. [Google Scholar] [CrossRef]
- Liu, Y.; Fatheree, N.Y.; Dingle, B.M.; Tran, D.Q.; Rhoads, M. Lactobacillus Reuteri DSM 17938 Changes the Frequency of Foxp3+ Regulatory T Cells in the Intestine and Mesenteric Lymph Node in Experimental Necrotizing Enterocolitis. PLoS One 2013, 8, e56547. [Google Scholar]
- Liu, Y.; Fatheree, N.Y.; Mangalat, N.; Rhoads, J.M. Human Derived Probiotic Lactobacillus Reuteri Strains Differentially Reduce Intestinal Inflammation. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 299, G1087–G1096. [Google Scholar] [CrossRef]
- Ng, S.C.; Hart, A.L.; Kamm, M.A.; Stagg, A.J.; Knight, S.C. Mechanisms of Action of Probiotics: Recent Advances. Inflamm. Bowel. Dis. 2009, 15, 300–310. [Google Scholar] [CrossRef]
- Fukushima, Y.; Kawata, Y.; Hara, H.; Terada, A.; Mitsuoka, T. Effect of a Probiotic Formula on Intestinal Immunoglobulin A Production in Healthy Children. Int. J. Food Microbiol. 1998, 42, 39–44. [Google Scholar] [CrossRef]
- Kaila, M.; Isolauri, E.; Soppi, E.; Virtanen, E.; Laine, S.; Arvilommi, H. Enhancement of the Circulating Antibody Secreting Cell Response in Human Diarrhea by a Human Lactobacillus Strain. Pediatr. Res. 1992, 32, 141–144. [Google Scholar] [CrossRef]
- Holscher, H.D.; Czerkies, L.A.; Cekola, P.; Litov, R.; Benbow, M.; Santema, S.; Alexander, D.D.; Perez, V.; Sun, S.; Saavedra, J.M.; et al. Bifidobacterium Lactis Bb12 Enhances Intestinal Antibody Response in Formula-Fed Infants: a Randomized, Double-Blind, Controlled Trial. JPEN J. Parenter. Enteral Nutr. 2012, 36, 106S–117S. [Google Scholar] [CrossRef]
- Mohan, R.; Koebnick, C.; Schildt, J.; Mueller, M.; Radke, M.; Blaut, M. Effects of Bifidobacterium Lactis Bb12 Supplementation on Body Weight, Fecal PH, Acetate, Lactate, Calprotectin, and IgA in Preterm Infants. Pediatr. Res. 2008, 64, 418–422. [Google Scholar] [CrossRef]
- Galdeano, C.M.; Perdigon, G. The Probiotic Bacterium Lactobacillus Casei Induces Activation of the Gut Mucosal Immune System Through Innate Immunity. Clin. Vaccine Immunol. 2006, 13, 219–226. [Google Scholar] [CrossRef]
- Qiao, H.; Duffy, L.C.; Griffiths, E.; Dryja, D.; Leavens, A.; Rossman, J.; Rich, G.; Riepenhoff-Talty, M.; Locniskar, M. Immune Responses in Rhesus Rotavirus-Challenged BALB/c Mice Treated With Bifidobacteria and Prebiotic Supplements. Pediatr. Res. 2002, 51, 750–755. [Google Scholar] [CrossRef]
- Shu, Q.; Gill, H.S. A Dietary Probiotic (Bifidobacterium Lactis HN019) Reduces the Severity of Escherichia Coli O157:H7 Infection in Mice. Med. Microbiol. Immunol. 2001, 189, 147–152. [Google Scholar] [CrossRef]
- Ogawa, M.; Shimizu, K.; Nomoto, K.; Takahashi, M.; Watanuki, M.; Tanaka, R.; Tanaka, T.; Hamabata, T.; Yamasaki, S.; Takeda, Y. Protective Effect of Lactobacillus Casei Strain Shirota on Shiga Toxin-Producing Escherichia Coli O157:H7 Infection in Infant Rabbits. Infect. Immun. 2001, 69, 1101–1108. [Google Scholar] [CrossRef]
- Zhang, W.; Azevedo, M.S.; Gonzalez, A.M.; Saif, L.J.; Van, N.T.; Wen, K.; Yousef, A.E.; Yuan, L. Influence of Probiotic Lactobacilli Colonization on Neonatal B Cell Responses in a Gnotobiotic Pig Model of Human Rotavirus Infection and Disease. Vet. Immunol. Immunopathol. 2008, 122, 175–181. [Google Scholar] [CrossRef]
- Roller, M.; Rechkemmer, G.; Watzl, B. Prebiotic Inulin Enriched With Oligofructose in Combination With the Probiotics Lactobacillus Rhamnosus and Bifidobacterium Lactis Modulates Intestinal Immune Functions in Rats. J. Nutr. 2004, 134, 153–156. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liu, Y.; Rhoads, J.M. Communication between B-Cells and Microbiota for the Maintenance of Intestinal Homeostasis. Antibodies 2013, 2, 535-553. https://doi.org/10.3390/antib2040535
Liu Y, Rhoads JM. Communication between B-Cells and Microbiota for the Maintenance of Intestinal Homeostasis. Antibodies. 2013; 2(4):535-553. https://doi.org/10.3390/antib2040535
Chicago/Turabian StyleLiu, Yuying, and Jon Marc Rhoads. 2013. "Communication between B-Cells and Microbiota for the Maintenance of Intestinal Homeostasis" Antibodies 2, no. 4: 535-553. https://doi.org/10.3390/antib2040535
APA StyleLiu, Y., & Rhoads, J. M. (2013). Communication between B-Cells and Microbiota for the Maintenance of Intestinal Homeostasis. Antibodies, 2(4), 535-553. https://doi.org/10.3390/antib2040535




