2.1. Generation of a Novel Monoclonal Antibody That Specifically Recognizes the Phosphorylated FcεRIγ-Chain ITAM Y47
To investigate the role of FcεRI phosphorylation in Akt phosphorylation we first developed a phospho-specific monoclonal antibody to the γ-chain ITAM Y47. Mice were immunized with an ITAM peptide (as detailed in the Experimental Section) encoding the phosphorylated γ-chain ITAM Y47 (pY47) and six hybridoma clones were successfully established. A phosphorylated peptide-bound ELISA was developed and culture supernatants from the six hybridoma clones were assayed for reactivity to pY47. A peptide encoding the non-phosphorylated Y47 was used as a negative control. Test of culture supernatants (1:10–1:2,430) by ELISA showed that the antibodies produced by all six hybridoma clones were able to recognize the peptide encoding the phosphorylated Y47 antigen but not the peptide encoding the non-phosphorylated Y47 (data not shown).
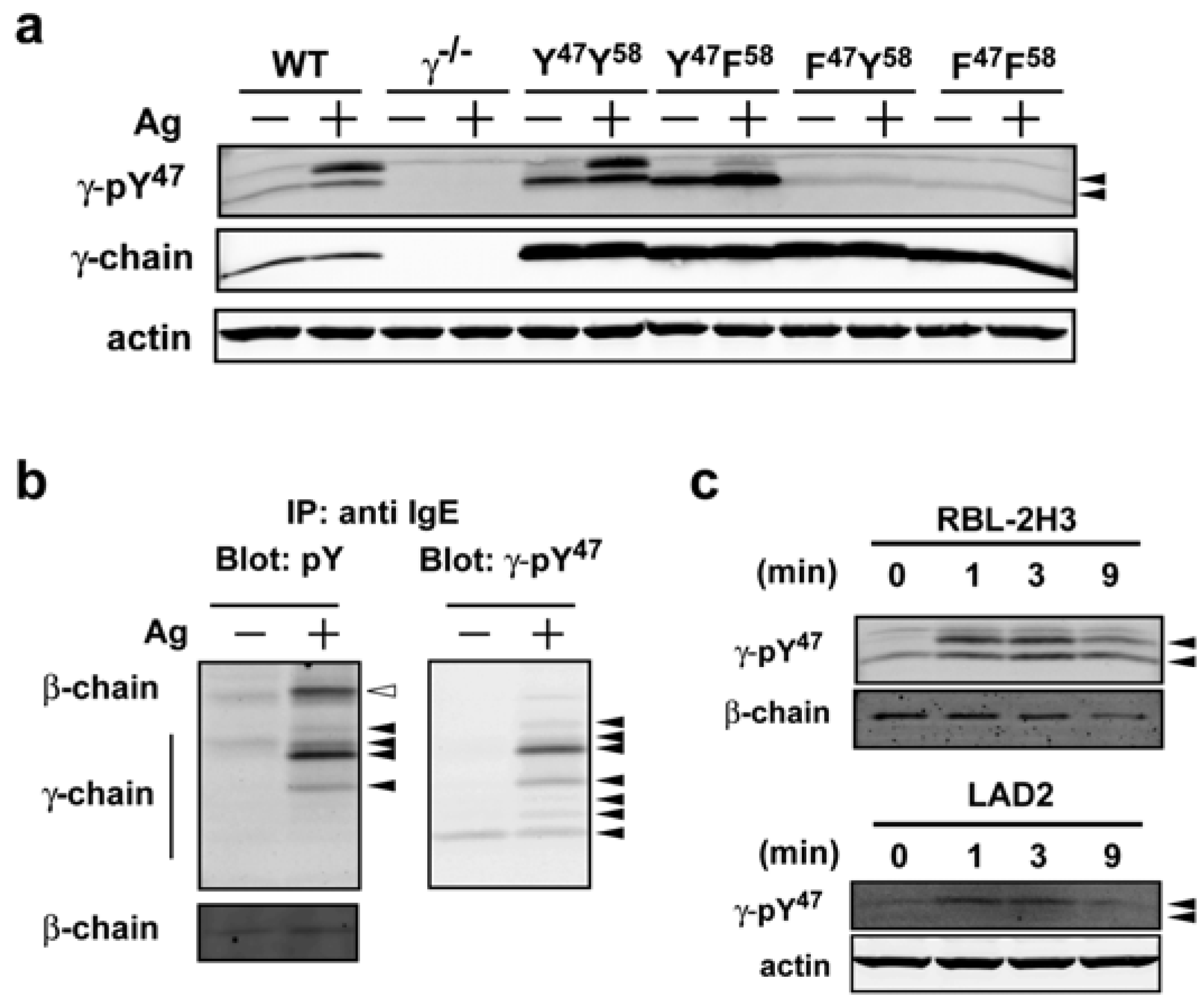
We chose to work with clone 6A5G5 (designated as γ-pY
47 in the following text) based on the sensitivity and selectivity of the observed reactivity by Western blot. Mast cells expressing mutant forms of the FcεRIγγ in which the ITAM tyrosine residues were individually or jointly mutated, were used to verify the specificity of the phospho-specific antibody to phosphorylated Y
47. When we compared lysates from normal (WT) bone marrow-derived mast cells (BMMCs), lysates derived from FcεRIγ-deficient (γ
−/−) BMMCs showed no reactivity with the antibody to pY
47 in Western blots (
Figure 1a). Upon reconstitution of γ
−/− mast cells with wild type (Y
47 Y
58) γ-chain, antibody recognition of pY
47 was restored (
Figure 1a). In contrast, reconstitution with various tyrosine to phenylalanine mutants (YF, FY, FF) resulted in differential recognition of pY
47, with cell lysates from YF mutant-expressing cells showing antibody reactivity by Western blot analysis whereas lysates from FY and FF mutants had no reactivity (
Figure 1a). Since retroviral reconstitution of γ
−/− mast cells resulted in overexpression of YY, YF, FY, and FF ITAMs antibody reactivity was intensified in the resting and activated YY and YF-bearing cells relative to WT cells. Nonetheless, this served to more clearly demonstrate the specificity of the antibody for recognition of pY
47 since lysates from cells overexpressing the FY and FF forms of the FcεRIγ ITAM did not show any detectable antibody reactivity (
Figure 1a). Both WT and YY-expressing mast cells showed two major bands following Ag stimulation. In YF-expressing MCs, one major band was observed in resting mast cells that intensified upon Ag stimulation and was accompanied by the appearance of a weakly reactive upper band comparable to the upper band seen in WT or YY-expressing mast cell lysates (
Figure 1a). This data suggested that majority of signal in upper band corresponds to dual phosphorylation at Y
47 and Y
58 and mutation of the latter resulted in an increase in the lower mono-phosphorylated (pY
47) band of FcεRIγ. We have previously shown that the phosphorylated homodimers of FcεRIγ are detected as multiple bands under non-reducing conditions with differing molecular mass [
12,
20]. Western blotting of immunoprecipitated FcεRI with an antibody to phosphotyrosine identified the phosphorylated β-chain and at least four major species of phosphorylated FcεRIγ (
Figure 1b, left panel) in Ag-stimulated conditions with little or no phosphorylation detected in non-stimulated conditions. Interestingly, the γ-pY
47 antibody blotting of the same stripped and reprobed blot showed a minor reactivity in resting BMMCs, demonstrating that this antibody is more sensitive in detecting phosphorylated FcεRIγthan the widely used anti-phosphotyrosine antibody 4G10 (
Figure 1b, right panel). An additional six species of the phosphorylated FcεRIγ were observed following Ag stimulation. Some of the observed species were identical in both phosphotyrosine antibody and γ-pY
47 antibody blots. However, the three species (with the lowest molecular mass) seemed to be detected only by the γ-pY
47 antibody. Peptide microarray data has demonstrated that all phosphotyrosine antibody clones (
i.e., 4G10, PY20, p-TYR-100) recognize their target in a sequence-specific context and that this recognition differs for each clone [
21]. Our results demonstrate that this novel antibody, γ-pY
47, recognizes its target sequence in the FcεRIγ with high selectivity and sensitivity, and recognizes more of the multiple molecular mass species (or modifications) of the FcεRIγthan the general anti-phosphotyrosine antibody 4G10.
Given the usefulness of such a reagent to assess the status of FcεRI phosphorylation directly in cell lysates, we examined whether γ-pY
47 antibody could recognize phosphorylation of FcεRIγ Y
47 in mast cells from different species. The similarity of the amino acid sequence of the immunizing peptide among mouse, rat, and human was considerable, with mouse to rat being 92.9% identical and mouse to human being 85.7% identical. Thus, it was possible that the γ-pY
47 antibody might react with rat and human FcεRIγ. As shown in
Figure 1c, Western blot analysis showed that the γ-pY
47 antibody reacted with phosphorylated FcεRIγspecies in lysates from non-stimulated and Ag-stimulated RBL-2H3 (rat mast cell line) as well as with LAD2 (human mast cell line) cells. Interestingly, the lower molecular mass species in the LAD2 human mast cell line was only weakly detected and because the homology of mouse with human is considerably lower than that of mouse with rat, this may reflect a contextual difference in recognition of Y
47 or that basal phosphorylation is more restrained in this human mast cell line. Regardless, our findings show γ-pY
47 antibody recognizes phosphorylated FcεRIγ Y
47 of mouse, rat and human origin.
Figure 1.
A novel phospho-specific antibody (γ-pY47) that selectively recognizes the phosphorylated FcεRIγ ITAM Y47 from mouse, rat, and human origin. (a) Murine WT BMMCs or γ−/− BMMCs retrovirally transduced with FcεRIγ mutants (YY, YF, FY, and FF) or control vector (γ−/−) were sensitized with IgE and stimulated with Ag for 1 min at 37 °C. Whole cell lysates were resolved by SDS-PAGE and immunoblotted with γ-pY47 antibody under reducing conditions. (b) FcεRI was immunoprecipitated from whole cell lysates of WT cells (see Experimental Section) and resolved by SDS-PAGE under non-reducing conditions. Immunoblots were analyzed with anti-phosphotyrosine antibody (4G10) or γ-pY47 antibody. (c) Whole cell lysates from stimulated or non-stimulated rat (RBL-2H3) and human (LAD2) mast cell lines were probed for phosphorylation of FcεRI with γ-pY47 antibody.
Figure 1.
A novel phospho-specific antibody (γ-pY47) that selectively recognizes the phosphorylated FcεRIγ ITAM Y47 from mouse, rat, and human origin. (a) Murine WT BMMCs or γ−/− BMMCs retrovirally transduced with FcεRIγ mutants (YY, YF, FY, and FF) or control vector (γ−/−) were sensitized with IgE and stimulated with Ag for 1 min at 37 °C. Whole cell lysates were resolved by SDS-PAGE and immunoblotted with γ-pY47 antibody under reducing conditions. (b) FcεRI was immunoprecipitated from whole cell lysates of WT cells (see Experimental Section) and resolved by SDS-PAGE under non-reducing conditions. Immunoblots were analyzed with anti-phosphotyrosine antibody (4G10) or γ-pY47 antibody. (c) Whole cell lysates from stimulated or non-stimulated rat (RBL-2H3) and human (LAD2) mast cell lines were probed for phosphorylation of FcεRI with γ-pY47 antibody.
2.2. Use of the γ-pY47Antibody in Various Applications
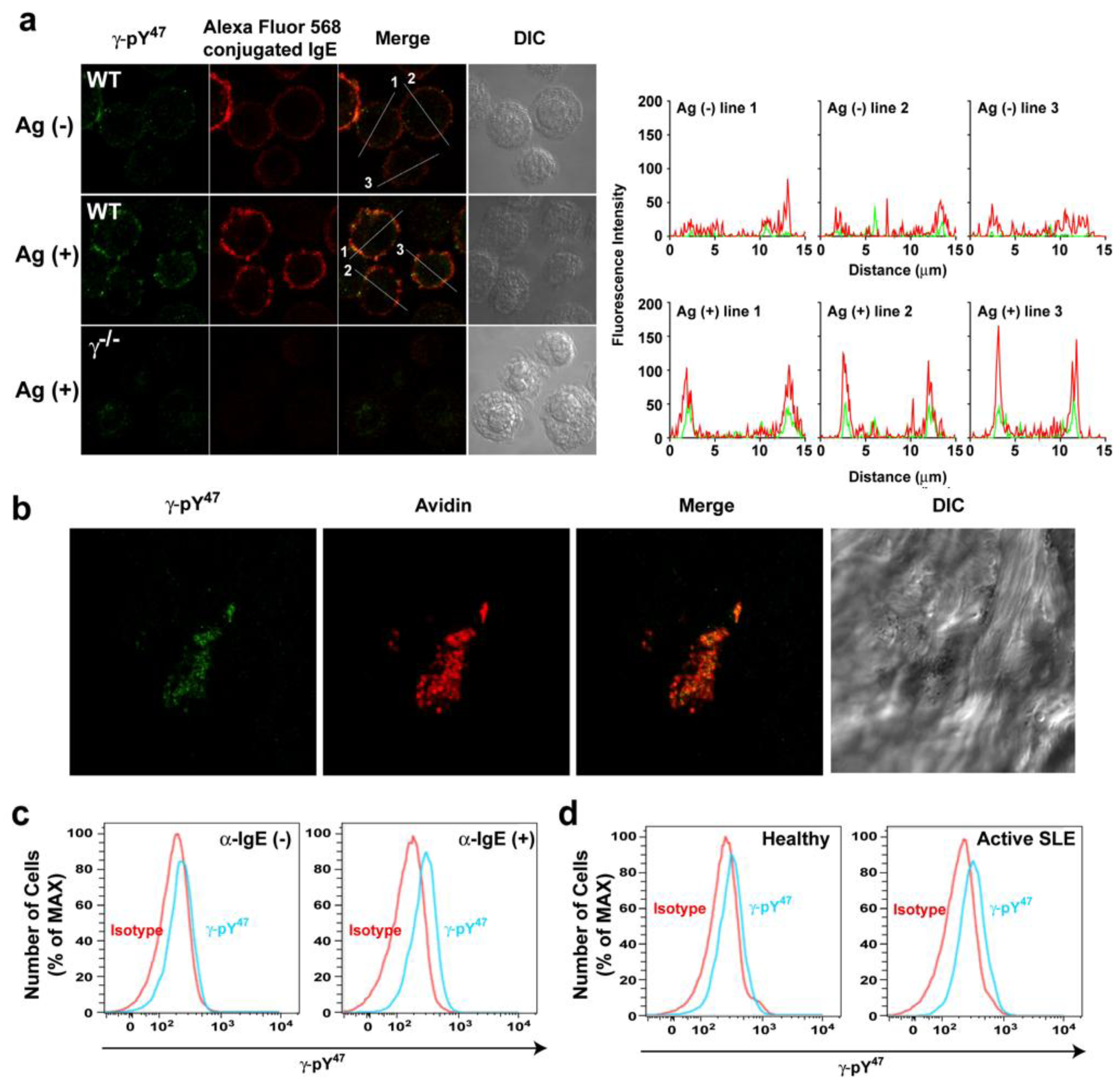
The findings show that the γ-pY
47 antibody can selectively, and with high sensitivity, recognize phosphorylated FcεRIγ in whole cell lysates from various species by Western blot. However, the usefulness of such a reagent for other applications was unclear. Thus, we tested its efficacy in detecting phosphorylated FcεRIγ in immunocytochemistry, immunohistochemistry, and FACS. Double-immunostaining using the Alexa Fluor 568-labeled IgEand the γ-pY
47 antibody detected with Alexa Fluor 488 conjugated secondly antibody was performed in resting and Ag-stimulated BMMCs (
Figure 2a). The BMMCs were then visualized by confocal laser scanning microscopy. In resting BMMCs, Alexa Fluor 568-labeled IgE (red) was uniformly distributed on the plasma membrane (intensity plot: red). Fluorescent signals from the γ-pY
47 antibody (in green) were very weakly localized in the plasma membrane (intensity plot: green). Following Ag-stimulation, Alexa Fluor 568-labeled IgE appeared more clustered and γ-pY
47 antibody staining was more co-localized with these FcεRI clusters (
Figure 2a, middle panel). Quantitative fluorescence intensity plots across a line section of these images showed that γ-pY
47 antibody staining increased its intensity following stimulation with Ag (as might be expected upon increased FcεRI phosphorylation) and co-localized with IgE aggregates (intensity plot: Ag (+)). No obvious immunostaining was observed in BMMC derived from γ chain-deficient mice (
Figure 2a: bottom panel). We further tested the utility of γ-pY
47 antibody for immunohistochemistry using skin tissue from the mast cell-dependent mouse model of passive cutaneous anaphylaxis (PCA). Skin samples from the ear of PCA challenged (5 min) WT mice were stained with Alexa Fluor 488 conjugated γ-pY
47 antibody (
Figure 2b). Alexa Fluor 647 conjugated-avidin was used to visualize mast cells in skin tissues [
22,
23]. As shown in
Figure 2b, following PCA challenge, mast cells (a single representative cell is shown in red) was observed in the skin and the staining with γ-pY
47 antibody (green) showed punctate pattern reminiscent of FcεRI clusters.
Figure 2.
The γ-pY47 antibody detects phosphorylation of FcεRIγ ITAM Y47 in vitro and in vivo. (a) Single cell analysis of FcεRIγ ITAM Y47 phosphorylation of mast cells stimulated with Ag for 1 min. Distribution of γ-pY47 antibody was detected with Alexa Fluor 488 conjugated secondly antibody (green) and IgE was conjugated with Alexa Fluor 568 (red). An increased fluorescence intensity of Alexa Fluor 568 conjugated IgE was observed upon FcεRI clustering following Ag stimulation. The merged images (yellow) indicate colocalization of γ-pY47 antibody and Alexa Fluor 568 conjugated IgE. The fluorescence intensity in cross-section of individual cells is shown as an intensity plot. (b) Detection of FcεRIγ ITAM Y47 phosphorylation in ear skin tissue of WT mice locally sensitized with saline or (anti-DNP) IgE in the contralateral ear. The next day, mice were challenged retroorbitally with Ag. Following a 5-min incubation, mice were euthanized, tissue collected and double whole-mount immunostaining was done with γ-pY47 antibody (green) and Alexa Fluor 647 avidin (for mast cell staining).Mice sensitized with saline and subsequently challenged with Ag showed no detectable fluorescencewith γ-pY47antibody in tissues (data not shown). (c) Human blood from healthy donors was stimulated with anti-human IgE antibody for 2 min and then cells were fixed and stained for the surface markers, APC-conjugated anti-human CD123, PE-conjugated anti-human CD303, Pacific Blue-conjugated anti-human FcεRIα, and PercPCy5.5-conjugated anti-human CD203c, followed by intracellular staining with Alexa Fluor 488 conjugated γ-pY47 antibody.Mean fluorescence intensity (MFI) for non-stimulated or anti-IgE stimulated (2 min) conditions was 228 and 269, respectively. (d) γ-pY47 antibody can be used to determine the activation status of basophils in human blood from subjects with active SLE. Representative data from 4 healthy and 4 SLE subjects is shown.
Figure 2.
The γ-pY47 antibody detects phosphorylation of FcεRIγ ITAM Y47 in vitro and in vivo. (a) Single cell analysis of FcεRIγ ITAM Y47 phosphorylation of mast cells stimulated with Ag for 1 min. Distribution of γ-pY47 antibody was detected with Alexa Fluor 488 conjugated secondly antibody (green) and IgE was conjugated with Alexa Fluor 568 (red). An increased fluorescence intensity of Alexa Fluor 568 conjugated IgE was observed upon FcεRI clustering following Ag stimulation. The merged images (yellow) indicate colocalization of γ-pY47 antibody and Alexa Fluor 568 conjugated IgE. The fluorescence intensity in cross-section of individual cells is shown as an intensity plot. (b) Detection of FcεRIγ ITAM Y47 phosphorylation in ear skin tissue of WT mice locally sensitized with saline or (anti-DNP) IgE in the contralateral ear. The next day, mice were challenged retroorbitally with Ag. Following a 5-min incubation, mice were euthanized, tissue collected and double whole-mount immunostaining was done with γ-pY47 antibody (green) and Alexa Fluor 647 avidin (for mast cell staining).Mice sensitized with saline and subsequently challenged with Ag showed no detectable fluorescencewith γ-pY47antibody in tissues (data not shown). (c) Human blood from healthy donors was stimulated with anti-human IgE antibody for 2 min and then cells were fixed and stained for the surface markers, APC-conjugated anti-human CD123, PE-conjugated anti-human CD303, Pacific Blue-conjugated anti-human FcεRIα, and PercPCy5.5-conjugated anti-human CD203c, followed by intracellular staining with Alexa Fluor 488 conjugated γ-pY47 antibody.Mean fluorescence intensity (MFI) for non-stimulated or anti-IgE stimulated (2 min) conditions was 228 and 269, respectively. (d) γ-pY47 antibody can be used to determine the activation status of basophils in human blood from subjects with active SLE. Representative data from 4 healthy and 4 SLE subjects is shown.
![Antibodies 02 00321 g002]()
Given that our previous findings showed that γ-pY
47 antibody recognized phosphorylated FcεRIγ from human origin (
Figure 1c), we hypothesized that γ-pY
47 antibody might detect the activation status of FcεRI in human mast cells or basophils and thus could be useful to analyze such cells in human disease. We previously published that, in systemic lupus erythematosus (SLE), activation of basophils by autoreactive IgE-containing immune complexes serves to amplify the production of autoantibodies and contributes to disease progression and severity [
24]. Thus, we first examined whether γ-pY
47 antibody could detect phosphorylated FcεRI in human basophil by FACS. Human blood basophils were activated with anti-human IgE antibody for 2 min and fixed and stained with antibodies against different surface marker proteins used to identify human basophils (see Experimental Section). As shown in
Figure 2c (left panel), γ-pY
47 antibody staining of non-stimulated human basophils did not differ with isotype control. Following a 2 min stimulation with anti-IgE, γ-pY
47 antibody staining showed a shift in fluorescence intensity relative to isotype control (
Figure 2c, right panel). This demonstrated that γ-pY
47 antibody could detect the phosphorylation of FcεRI in human basophils following their activation. To test if one could distinguish the phosphorylation status of FcεRI in human basophils during disease we tested the reactivity of the γ-pY
47 antibody from healthy donors or donors with active SLE (
Figure 2d). Staining of basophils from healthy donors was very similar to isotype control staining (
Figure 2d, left panel). However, the staining with γ-pY
47 antibody in basophils from donors with active SLE was considerably higher than that of isotype control or that of the healthy donors (
Figure 2d, right panel). These findings demonstrate the utility of the γ-pY
47 antibody in determining the phosphorylation status of FcεRI during cell activation or in disease and could be a useful diagnostic tool in allergic and other diseases where IgE antibodies may play a role.
2.3. Phosphorylation of FcεRIγ Y47 is Dependent on FcεRIβ Y219
Our previous work [
12] demonstrated that FcεRIγ Y
58 (C-terminal ITAM tyrosine) is highly susceptible to dephosphorylation. Thus, the aforementioned findings of a predominance in detection of phosphorylation of Y
47 relative to Y
58, using cells carry mutated FcεRIγ at these sites (
Figure 1a), led us to further characterize the requirements for phosphorylation of Y
47and how Y
47 phosphorylation influenced downstream signaling events.
Lyn kinase has been shown to phosphorylate FcεRI [
25,
26] and associates with the FcεRIβ subunit and the phosphorylation of ITAM Y
219 in FcεRIβ is required for this association [
6]. Thus, we investigated the role of the canonical (Y
219 and Y
229) or noncanonical (Y
225) tyrosine’s of FcεRIβ on the phosphorylation of FcεRIγ Y
47. Mutation of each tyrosine residue (Y
219 Y
225 Y
229) individually or in combination to phenylalanine was done. These mutant forms of FcεRIβ were transduced (lentivirus) into BMMCs derived from β-chain deficient (β
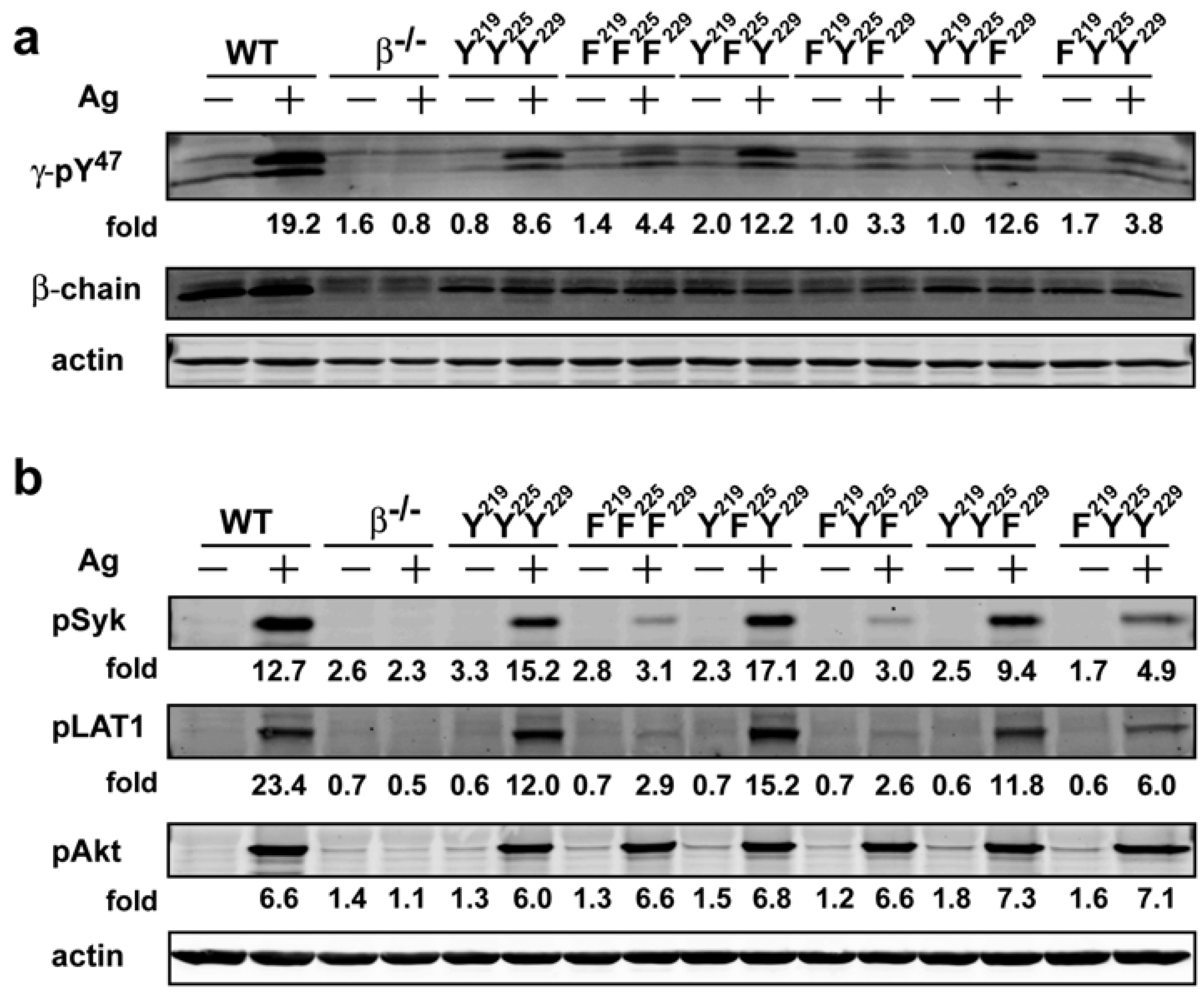
−/−) mice. All transduced BMMCs were more than 95% positive for FcεRI expression on their cell surface with similar flourescence intensity (data not shown) demonstrating that the expression of the mutant forms of FcεRIβ did not alter receptor expression. FcεRIβ-deficient cells transduced with wild type (YYY) FcεRIβ showed a phosphorylation pattern of FcεRIγ (as detected by the γ-pY
47 antibody) that was similar to the FcεRIγ phosphorylation pattern from WT cells, whereas no detectable phosphorylation was observed in FcεRIβ-deficient cells transduced with a control vector (
Figure 3a). Phosphorylation of FcεRIγ Y
47 in YFY or YYF transduced BMMCs was remarkably similar to that of YYY-transduced BMMCs. In contrast, FFF, FYF or FYY transduced cells showed considerable reduction (~40%) of FcεRIγ pY
47 phosphorylation upon Ag stimulation (
Figure 3a). Taken together, the findings suggested that the phosphorylation of the FcεRIβ Y
219 (canonical N-terminal tyrosine) played an important role in phosphorylation of the FcεRIγ Y
47. This finding is consistent with previous studies demonstrating the requirement of Lyn kinase for FcεRI phosphorylation [
25] and the importance of FcεRIβ Y
219 for association of Lyn with FcεRI [
6]. Because both FcεRIY
47and Y
58 are required for the full extent of Syk kinase activation in mast cells, we analyzed the extent of Syk phosphorylation at the activation loop Y
519/520 (which when phosphorylated reflects a state of Syk activation) in the cells expressing the different FcεRIβ tyrosine mutants. As shown in
Figure 3b, while phosphorylation of Syk Y
519/520 in YFY and YYF mutant was comparable to the YYY expressing cells, FcεRIβ tyrosine mutants expressing FFF or FYF ITAMs showed a marked reduction (~80%) in Syk phosphorylation whereas FYY expressing cells showed a considerable reduction (>60%) in phosphorylation of Syk activation loop Y
519/520. Thus, these findings showed that the loss of phosphorylation of the FcεRIβ ITAM tyrosine’s has a significant impact on Syk activation. Importantly, this also translates to defective molecular signaling further downstream as the extent of LAT1 (linker for activation of T cells) phosphorylation at Y
191 (a downstream target of Syk) was similarly affected (
Figure 3b). Moreover, they clearly demonstrate the importance of FcεRIβ ITAM in regulating the phosphorylation of FcεRIγ and further show that the FcεRIβ Y
219 contributes to the extent of Syk and LAT1 phosphorylation following FcεRI engagement.
The findings also raised the question of whether all signaling downstream of FcεRI engagement were similarly regulated by the ITAM tyrosine’s of FcεRIβ or FcεRIγ. We previously demonstrated that Fyn kinase initiates complementary signals in mast cells [
14], which are less dependent on Lyn, Syk, or LAT1. Our findings showed that Fyn activation was important for the phosphorylation of the adapter molecule Gab2 and the activation of PI3K and the phosphorylation of Akt [
14]. In cells expressing the various FcεRIβ ITAM mutants, analysis of Akt phosphorylation on T
308 (the site phosphorylated by the PI3K-dependent kinase 1) demonstrated no marked effect on its phosphorylation (
Figure 3b). However, phosphorylation of Akt was clearly dependent on expression of FcεRI because FcεRIβ
−/− BMMCs (which do express FcεRI on their cell surface [
9]) failed to induce Akt T
308 phosphorylation. Thus, the data suggested that phosphorylation of Akt was likely to be dependent on the FcεRIγ, a hypothesis we subsequently explored.
Figure 3.
The FcεRIβ Y219 regulates phosphorylation of FcεRIγ ITAM Y47 and proximal signaling molecules. Whole cell lysates from WT cells were resolved under reducing conditions. (a) Immunoblots were probed withγ-pY47 antibody. (b) Anti-phosphoSyk antibody (Y519/520), anti-phospho LAT1 antibody (Y191), and anti-phosphoAkt antibody (T308) were used to detect site specific phosphorylation. Fold induction was calculated by densitometry and normalized to the respective protein or to actin.
Figure 3.
The FcεRIβ Y219 regulates phosphorylation of FcεRIγ ITAM Y47 and proximal signaling molecules. Whole cell lysates from WT cells were resolved under reducing conditions. (a) Immunoblots were probed withγ-pY47 antibody. (b) Anti-phosphoSyk antibody (Y519/520), anti-phospho LAT1 antibody (Y191), and anti-phosphoAkt antibody (T308) were used to detect site specific phosphorylation. Fold induction was calculated by densitometry and normalized to the respective protein or to actin.
2.4. Phosphorylation of Akt Is Largely Dependent on the Phosphorylation of FcεRIγ Y58
Our subsequent experiments were initially aimed at addressing whether the phosphorylation of Akt was dependent on the continuous aggregation of the FcεRI. As shown in
Figure 4a the use of monovalent hapten (DNP-L-Lys), which disrupts the receptor aggregation induced by a multivalent Ag (DNP
30–40-HSA) [
27,
28], demonstrated that phosphorylation of FcεRI (as measured with γ-pY
47 antibody) is markedly abrogated by such treatment, however, phosphorylation can be restored when a general inhibitor of tyrosine phosphatases (pervanadate) is added to the cells. This suggested that disaggregation of FcεRI by hapten (30 s after Ag addition) makes the receptor susceptible to dephosphorylation by tyrosine phosphatases. Given this finding, we asked if the phosphorylation of downstream molecules (whether tyrosine or serine/threonine phosphorylated) was similarly affected by disaggregation of FcεRI. As shown in
Figure 4b, Syk (Y
519/520) and LAT1 (Y
191) phosphorylation was markedly affected by hapten addition whereas that of Akt (T
308) was largely unaffected. This suggested that phosphorylation of Akt on T
308 was less susceptible to dephosphorylation upon disaggregation of FcεRI.
Figure 4.
Akt phosphorylation is largely initiated by FcεRIγ ITAM Y58 butonce phosphorylated it does not require persistent FcεRI aggregation. (a) WT cells were sensitized with IgE and stimulated with Ag. At 30 s of Ag addition hapten (DNP-L-Lys, 100 µM) was added in the presence or absence of pervanadate (PV: 1 mM) and cells were incubated for the indicated time. Whole cell lysates were resolved and immunoblotted with γ-pY47 antibody. (b) Effects of FcεRI disaggregation on proximal signaling proteins as analyzed using indicated antibodies. (c) IgE-sensitized BMMCs expressing WT (YY) or mutated (YF, FY) FcεRIγ were stimulated for 1 min with Ag. Whole cell lysate proteins were resolved by SDS-PAGE and immunoblotted with indicated antibodies. (d) BMMCs expressing WT or mutant FcεRIγ were sensitized with IgE and were either not stimulated or stimulated with Ag in the presence or absence of pervanadate (PV: 1mM). Proteins in whole cell lysates were resolved and immunobloted with phospho-specific (T308) Akt antibody.
Figure 4.
Akt phosphorylation is largely initiated by FcεRIγ ITAM Y58 butonce phosphorylated it does not require persistent FcεRI aggregation. (a) WT cells were sensitized with IgE and stimulated with Ag. At 30 s of Ag addition hapten (DNP-L-Lys, 100 µM) was added in the presence or absence of pervanadate (PV: 1 mM) and cells were incubated for the indicated time. Whole cell lysates were resolved and immunoblotted with γ-pY47 antibody. (b) Effects of FcεRI disaggregation on proximal signaling proteins as analyzed using indicated antibodies. (c) IgE-sensitized BMMCs expressing WT (YY) or mutated (YF, FY) FcεRIγ were stimulated for 1 min with Ag. Whole cell lysate proteins were resolved by SDS-PAGE and immunoblotted with indicated antibodies. (d) BMMCs expressing WT or mutant FcεRIγ were sensitized with IgE and were either not stimulated or stimulated with Ag in the presence or absence of pervanadate (PV: 1mM). Proteins in whole cell lysates were resolved and immunobloted with phospho-specific (T308) Akt antibody.
![Antibodies 02 00321 g004]()
To assess the importance of the individual FcεRIγ ITAM tyrosine’s to Akt T
308phosphorylation we used BMMCs expressing FcεRIγ ITAM tyrosine mutants (Y
47F or Y
58F). We first verified that mutation at either of these sites had an impact on Syk and LAT1 phosphorylation as previously described [
12]. As shown in
Figure 4c, phosphorylation of Syk Y
519/520 and LAT1 Y
191 was dramatically reduced in BMMCs expressing either Y
47or Y
58 mutant FcεRIγ. In contrast, analysis of Akt T
308 phosphorylation revealed a modest decrease (<35%) in BMMCs expressing the FY mutant form of FcεRIγ, whereas cells expressing the YF form of FcεRIγ showed a marked reduction (~80%) in Akt phosphorylation (
Figure 4d, left panel). These findings suggested that initiation of Akt phosphorylation is primarily dependent on the phosphorylation of FcεRIγ Y
58. We next investigated whether phosphorylation of FcεRIγ Y
58 in initiating Akt phosphorylation could be by-passed by inhibiting tyrosine phosphatases. As shown in
Figure 4d, right panel, treatment of cells carrying the YF mutant form of FcεRIγ with pervanadate, results in the restoration of Akt T
308 phosphorylation demonstrating that the key steps for PI3K-dependent Akt phosphorylation are downstream of FcεRIγ Y
58 phosphorylation. These findings are consistent with the view that FcεRIγ Y
58 is key in initiating FcεRI-dependent Akt phosphorylation (see Model,
Figure 5) and this occurs upstream of the key signals (like Fyn kinase) that are essential for the activation of Akt [
14]. This is also consistent with the finding that the absence of Lyn, which causes an increase in Fyn kinase activity [
29], results in defective FcεRI phosphorylation but enhanced Akt phosphorylation. In contrast, Fyn-deficiency does not affect FcεRI phosphorylation but causes a marked dampening of Akt phosphorylation [
14].
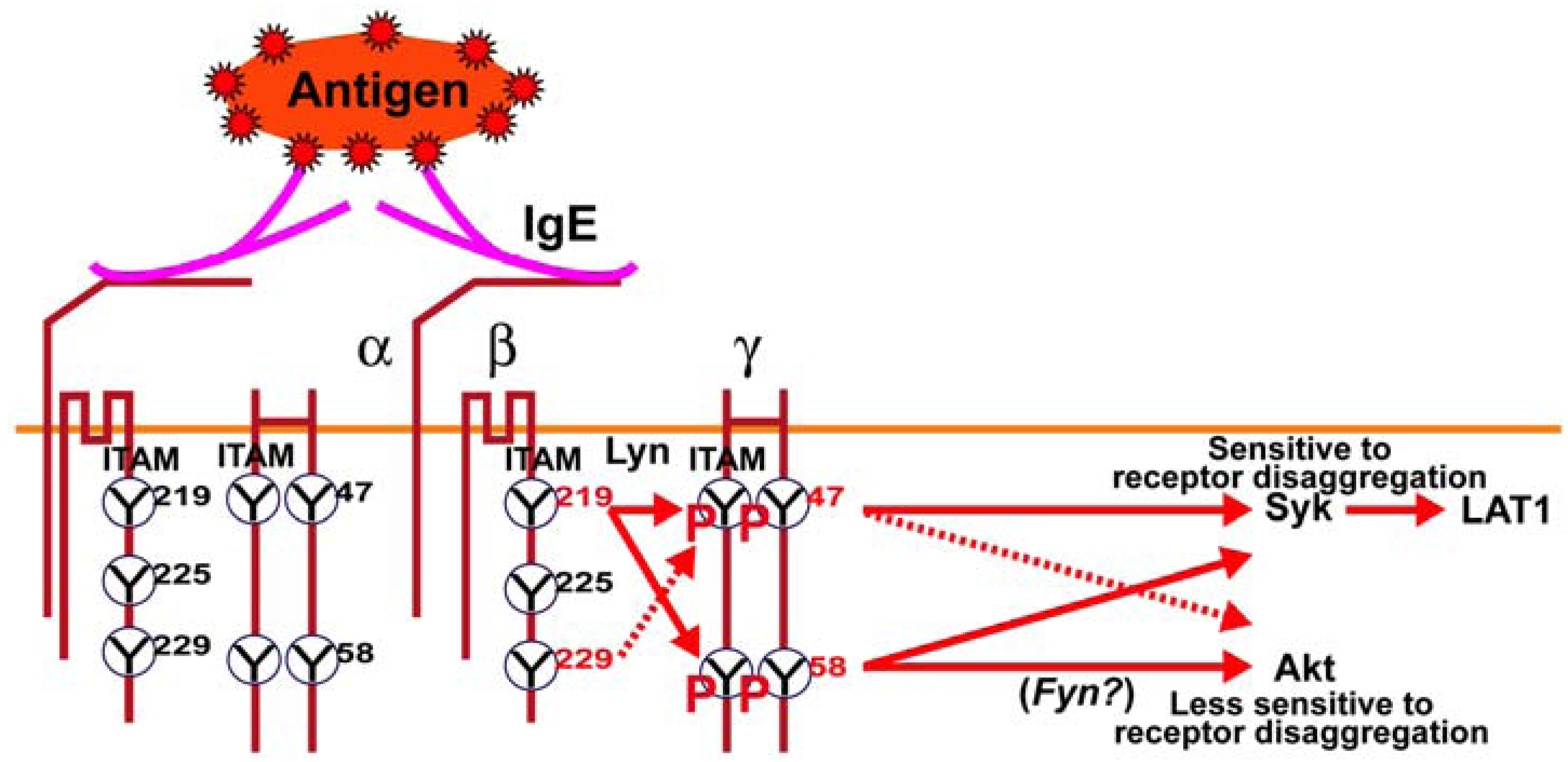
Figure 5.
Schematic model of the regulatory role of FcεRIβ and γ ITAM tyrosine’s on downstream signaling in mast cells. Ag-dependent aggregation of IgE antibody-occupied FcεRI on mast cells results in the Lyn kinase-dependent phosphorylation of the FcεRIβ and γ ITAM tyrosine’s. The FcεRIβY
219 is important for the full phosphorylation of FcεRIγ and was previously shown to be important for Lyn association with FcεRI [
6]. Loss of phosphorylation at FcεRIγ Y
47 or Y
58 had a marked effect on Syk and LAT1 phosphorylation; molecules known to be important for mast cell degranulation. However, Akt phosphorylation (T
308), whose activity is required for cytoskeletal reorganization and gene expression, was largely unaffected by loss of phosphorylation at FcεRIγ Y
47 whereas loss of phosphorylation at FcεRIγY
58 greatly reducedAkt phosphorylation. This can be overcome by inhibition of tyrosine phosphatases, which fully restored Akt phosphorylation in FcεRIγ Y
58F expressing mutant mast cells. Previous findings demonstrate that Fyn kinase is critical for Akt phosphorylation, thus we propose that FcεRIγY
58 is an upstream regulator of the Fyn-Gab2-Akt pathway.
Figure 5.
Schematic model of the regulatory role of FcεRIβ and γ ITAM tyrosine’s on downstream signaling in mast cells. Ag-dependent aggregation of IgE antibody-occupied FcεRI on mast cells results in the Lyn kinase-dependent phosphorylation of the FcεRIβ and γ ITAM tyrosine’s. The FcεRIβY
219 is important for the full phosphorylation of FcεRIγ and was previously shown to be important for Lyn association with FcεRI [
6]. Loss of phosphorylation at FcεRIγ Y
47 or Y
58 had a marked effect on Syk and LAT1 phosphorylation; molecules known to be important for mast cell degranulation. However, Akt phosphorylation (T
308), whose activity is required for cytoskeletal reorganization and gene expression, was largely unaffected by loss of phosphorylation at FcεRIγ Y
47 whereas loss of phosphorylation at FcεRIγY
58 greatly reducedAkt phosphorylation. This can be overcome by inhibition of tyrosine phosphatases, which fully restored Akt phosphorylation in FcεRIγ Y
58F expressing mutant mast cells. Previous findings demonstrate that Fyn kinase is critical for Akt phosphorylation, thus we propose that FcεRIγY
58 is an upstream regulator of the Fyn-Gab2-Akt pathway.
![Antibodies 02 00321 g005]()