Re-Use of Established Drugs for Anti-Metastatic Indications
Abstract
:1. New Test Methods for Established Drugs Can Reveal Additional Indications
2. Examples of Drug Repurposing in Oncology
| Proven Effect on Cancer Cells | ||
|---|---|---|
| Substance | Current indication | Receptor |
| Propranolol | hypertension | β1/β2-adrenoceptor-antagonist |
| Baclofen | epilepsia | GABA-B agonist |
| Metoclopramide | nausea | dopamine D2 antagonist |
| L-733,060 | anxiety, depression | neurokinin-1 antagonist |
| Further Drugs to be Evaluated | ||
| Substance | Current indication | Receptor |
| Iosartan | hypertension | angiotensin AT1 antagonist |
| Ipratropium | asthma | anticholinergic |
| Montelukast | asthma | leukotriene D4 antagonist |
| Ioratadine | allergies | histamine H1 antagonist |
| Famotidine | ulcers | histamine H2 antagonist |
| Olanzapine | schizophrenia | dopamine D1/D2/5-HT2 |
| Risperidone | psychosis | dopamine D2/5-HT2A |
| Sumatriptan | migrane | serotonin 5-HT1 agonist |
| Fentanyl | pain | opioid agonist |
3. Selection of Candidates by Their Action in Signal Transduction
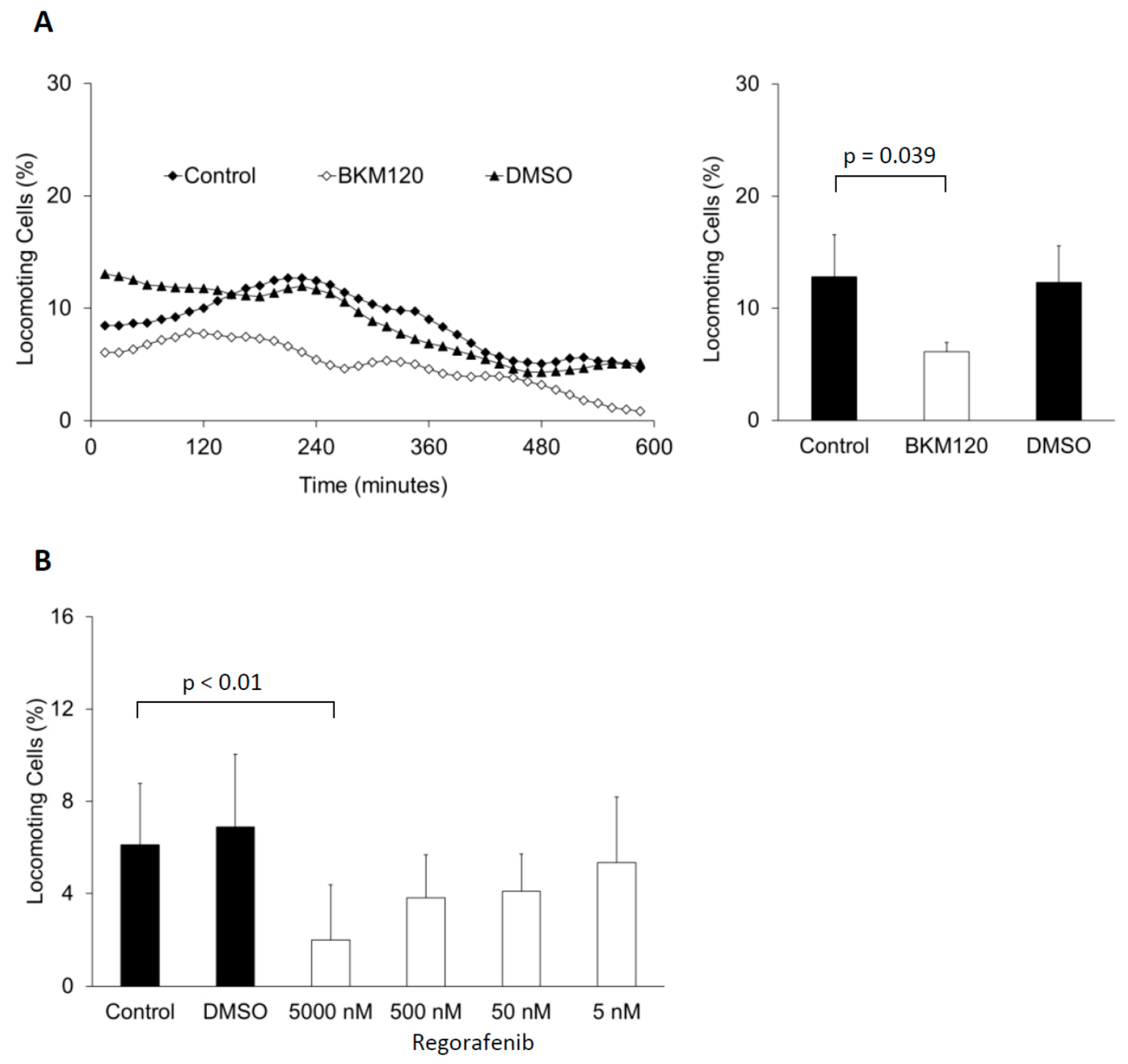
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Walker, I.; Newell, H. Do molecularly targeted agents in oncology have reduced attrition rates? Nat. Rev. Drug Discov. 2009, 8, 15–16. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency; Oncology Working Party. Guideline on the Evaluation of Anticancer Medicinal Products in Man; European Medicines Agency: London, UK, 2011; p. 5. [Google Scholar]
- Drell, T.L.; Zaenker, K.S.; Entschladen, F. Translational research in oncology: The need of additional in vitro preclinical testing methods for new drugs. Curr. Pharm. Des. 2012, 18, 3416–3420. [Google Scholar] [CrossRef] [PubMed]
- Sporn, M.B. The war on cancer. Lancet 1996, 347, 1377–1381. [Google Scholar] [CrossRef]
- Steeg, P.S. Tumor metastasis: Mechanistic insights and clinical challenges. Nat. Med. 2006, 12, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, M.; Rosel, D.; Brabek, J. Translation in solid cancer: Are size-based response criteria an anachronism? Clin. Transl. Oncol. Off. Publ. Fed. Span. Oncol. Soc. Natl. Cancer Inst. Mex. 2015, 17, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, A.S.; Steeg, P.S. Meaningful prevention of breast cancer metastasis: Candidate therapeutics, preclinical validation, and clinical trial concerns. J. Mol. Med. 2015, 93, 13–29. [Google Scholar] [CrossRef] [PubMed]
- Nagaraja, A.S.; Sadaoui, N.C.; Lutgendorf, S.K.; Ramondetta, L.M.; Sood, A.K. Beta-blockers: A new role in cancer chemotherapy? Expert Opin. Investig. Drugs 2013, 22, 1359–1363. [Google Scholar] [CrossRef] [PubMed]
- Drell, T.L.; Joseph, J.; Lang, K.; Niggemann, B.; Zaenker, K.S.; Entschladen, F. Effects of neurotransmitters on the chemokinesis and chemotaxis of mda-mb-468 human breast carcinoma cells. Breast Cancer Res. Treat. 2003, 80, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Lang, K.; Drell, T.L.; Lindecke, A.; Niggemann, B.; Kaltschmidt, C.; Zaenker, K.S.; Entschladen, F. Induction of a metastatogenic tumor cell type by neurotransmitters and its pharmacological inhibition by established drugs. Int. J. Cancer 2004, 112, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Masur, K.; Niggemann, B.; Zanker, K.S.; Entschladen, F. Norepinephrine-induced migration of sw 480 colon carcinoma cells is inhibited by beta-blockers. Cancer Res. 2001, 61, 2866–2869. [Google Scholar] [PubMed]
- Palm, D.; Lang, K.; Niggemann, B.; Drell, T.L.; Masur, K.; Zaenker, K.S.; Entschladen, F. The norepinephrine-driven metastasis development of pc-3 human prostate cancer cells in balb/c nude mice is inhibited by beta-blockers. Int. J. Cancer 2006, 118, 2744–2749. [Google Scholar] [CrossRef] [PubMed]
- Barron, T.I.; Connolly, R.M.; Sharp, L.; Bennett, K.; Visvanathan, K. Beta blockers and breast cancer mortality: A population-based study. J. Clin. Oncol. 2011, 29, 2635–2644. [Google Scholar] [CrossRef] [PubMed]
- Botteri, E.; Munzone, E.; Rotmensz, N.; Cipolla, C.; de Giorgi, V.; Santillo, B.; Zanelotti, A.; Adamoli, L.; Colleoni, M.; Viale, G.; et al. Therapeutic effect of beta-blockers in triple-negative breast cancer postmenopausal women. Breast Cancer Res. Treat. 2013, 140, 567–575. [Google Scholar] [CrossRef] [PubMed]
- De Giorgi, V.; Grazzini, M.; Gandini, S.; Benemei, S.; Lotti, T.; Marchionni, N.; Geppetti, P. Treatment with beta-blockers and reduced disease progression in patients with thick melanoma. Arch. Intern. Med. 2011, 171, 779–781. [Google Scholar] [CrossRef] [PubMed]
- Diaz, E.S.; Karlan, B.Y.; Li, A.J. Impact of beta blockers on epithelial ovarian cancer survival. Gynecol. Oncol. 2012, 127, 375–378. [Google Scholar] [CrossRef] [PubMed]
- Grytli, H.H.; Fagerland, M.W.; Fossa, S.D.; Tasken, K.A. Association between use of beta-blockers and prostate cancer-specific survival: A cohort study of 3561 prostate cancer patients with high-risk or metastatic disease. Eur. Urol. 2014, 65, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Grytli, H.H.; Fagerland, M.W.; Fossa, S.D.; Tasken, K.A.; Haheim, L.L. Use of beta-blockers is associated with prostate cancer-specific survival in prostate cancer patients on androgen deprivation therapy. Prostate 2013, 73, 250–260. [Google Scholar] [CrossRef] [PubMed]
- Lemeshow, S.; Sorensen, H.T.; Phillips, G.; Yang, E.V.; Antonsen, S.; Riis, A.H.; Lesinski, G.B.; Jackson, R.; Glaser, R. Beta-blockers and survival among danish patients with malignant melanoma: A population-based cohort study. Cancer Epidemiol. Biomark. Prev. 2011, 20, 2273–2279. [Google Scholar] [CrossRef] [PubMed]
- Melhem-Bertrandt, A.; Chavez-Macgregor, M.; Lei, X.; Brown, E.N.; Lee, R.T.; Meric-Bernstam, F.; Sood, A.K.; Conzen, S.D.; Hortobagyi, G.N.; Gonzalez-Angulo, A.M. Beta-blocker use is associated with improved relapse-free survival in patients with triple-negative breast cancer. J. Clin. Oncol. 2011, 29, 2645–2652. [Google Scholar] [CrossRef] [PubMed]
- Powe, D.G.; Voss, M.J.; Zaenker, K.S.; Habashi, H.O.; Green, A.R.; Ellis, I.O.; Entschladen, F. Beta-blocker drug therapy reduces secondary cancer formation in breast cancer and improves cancer specific survival. Oncotarget 2010, 1, 628–638. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.M.; Liao, Z.X.; Komaki, R.; Welsh, J.W.; O’Reilly, M.S.; Chang, J.Y.; Zhuang, Y.; Levy, L.B.; Lu, C.; Gomez, D.R. Improved survival outcomes with the incidental use of beta-blockers among patients with non-small-cell lung cancer treated with definitive radiation therapy. Ann. Oncol. 2013, 24, 1312–1319. [Google Scholar] [CrossRef] [PubMed]
- De Giorgi, V.; Gandini, S.; Grazzini, M.; Benemei, S.; Marchionni, N.; Geppetti, P. Effect of beta-blockers and other antihypertensive drugs on the risk of melanoma recurrence and death. Mayo Clin. Proc. 2013, 88, 1196–1203. [Google Scholar] [CrossRef] [PubMed]
- Jansen, L.; Hoffmeister, M.; Arndt, V.; Chang-Claude, J.; Brenner, H. Stage-specific associations between beta blocker use and prognosis after colorectal cancer. Cancer 2014, 120, 1178–1186. [Google Scholar] [CrossRef] [PubMed]
- Nkontchou, G.; Aout, M.; Mahmoudi, A.; Roulot, D.; Bourcier, V.; Grando-Lemaire, V.; Ganne-Carrie, N.; Trinchet, J.C.; Vicaut, E.; Beaugrand, M. Effect of long-term propranolol treatment on hepatocellular carcinoma incidence in patients with hcv-associated cirrhosis. Cancer Prev. Res. 2012, 5, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.H.; Song, T.; Kim, T.H.; Choi, J.K.; Park, J.Y.; Yoon, A.; Lee, Y.Y.; Kim, T.J.; Bae, D.S.; Lee, J.W.; et al. Meta-analysis of the effects of beta blocker on survival time in cancer patients. J. Cancer Res. Clin. Oncol. 2014, 140, 1179–1188. [Google Scholar] [CrossRef] [PubMed]
- Childers, W.K.; Hollenbeak, C.S.; Cheriyath, P. Beta-blockers reduce breast cancer recurrence and breast cancer death: A meta-analysis. Clin. Breast Cancer 2015, 15, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Giampieri, R.; Scartozzi, M.; del Prete, M.; Faloppi, L.; Bianconi, M.; Ridolfi, F.; Cascinu, S. Prognostic value for incidental antihypertensive therapy with beta-blockers in metastatic colorectal cancer. Medicine (Baltimore) 2015, 94, e719. [Google Scholar] [CrossRef] [PubMed]
- Watkins, J.L.; Thaker, P.H.; Nick, A.M.; Ramondetta, L.M.; Kumar, S.; Urbauer, D.L.; Matsuo, K.; Squires, K.C.; Coleman, R.L.; Lutgendorf, S.K.; et al. Clinical impact of selective and nonselective beta-blockers on survival in patients with ovarian cancer. Cancer 2015, 121, 3444–3451. [Google Scholar] [CrossRef] [PubMed]
- Palm, D.; Lang, K.; Brandt, B.; Zaenker, K.S.; Entschladen, F. In vitro and in vivo imaging of cell migration: Two interdepending methods to unravel metastasis formation. Semin. Cancer Biol. 2005, 15, 396–404. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.J.; Hannanta-anan, P.; Chau, M.; Kim, Y.S.; Swartz, M.A.; Wu, M. Cooperative roles of SDF-1α and EGF gradients on tumor cell migration revealed by a robust 3D microfluidic model. PLoS ONE 2013, 8, e68422. [Google Scholar] [CrossRef] [PubMed]
- Jeon, J.S.; Bersini, S.; Gilardi, M.; Dubini, G.; Charest, J.L.; Moretti, M.; Kamm, R.D. Human 3D vascularized organotypic microfluidic assays to study breast cancer cell extravasation. Proc. Natl. Acad. Sci. USA 2015, 112, 214–219. [Google Scholar] [CrossRef] [PubMed]
- Mosadegh, B.; Lockett, M.R.; Minn, K.T.; Simon, K.A.; Gilbert, K.; Hillier, S.; Newsome, D.; Li, H.; Hall, A.B.; Boucher, D.M.; et al. A paper-based invasion assay: Assessing chemotaxis of cancer cells in gradients of oxygen. Biomaterials 2015, 52, 262–271. [Google Scholar] [CrossRef] [PubMed]
- Pantziarka, P.; Bouche, G.; Meheus, L.; Sukhatme, V.; Sukhatme, V.P.; Vikas, P. The repurposing drugs in oncology (redo) project. Ecancermedicalscience 2014, 8, 442. [Google Scholar] [CrossRef] [PubMed]
- Azuma, H.; Inamoto, T.; Sakamoto, T.; Kiyama, S.; Ubai, T.; Shinohara, Y.; Maemura, K.; Tsuji, M.; Segawa, N.; Masuda, H.; et al. Gamma-aminobutyric acid as a promoting factor of cancer metastasis; induction of matrix metalloproteinase production is potentially its underlying mechanism. Cancer Res. 2003, 63, 8090–8096. [Google Scholar] [PubMed]
- Zhang, D.; Li, X.; Yao, Z.; Wei, C.; Ning, N.; Li, J. Gabaergic signaling facilitates breast cancer metastasis by promoting erk1/2-dependent phosphorylation. Cancer Lett. 2014, 348, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Ortega, A. A new role for gaba: Inhibition of tumor cell migration. Trends Pharmacol. Sci. 2003, 24, 151–154. [Google Scholar] [CrossRef]
- Neman, J.; Termini, J.; Wilczynski, S.; Vaidehi, N.; Choy, C.; Kowolik, C.M.; Li, H.; Hambrecht, A.C.; Roberts, E.; Jandial, R. Human breast cancer metastases to the brain display gabaergic properties in the neural niche. Proc. Natl. Acad. Sci. USA 2014, 111, 984–989. [Google Scholar] [CrossRef] [PubMed]
- Medinger, M.; Halter, J.; Heim, D.; Buser, A.; Gerull, S.; Stern, M.; Passweg, J. Angiogenic markers in plasma cell myeloma patients treated with novel agents. Anticancer Res. 2015, 35, 1085–1090. [Google Scholar] [PubMed]
- Vargesson, N. Thalidomide-induced teratogenesis: History and mechanisms. Birth Defects Res. C Embryo Today 2015, 105, 140–156. [Google Scholar] [CrossRef] [PubMed]
- Beedie, S.L.; Peer, C.J.; Pisle, S.; Gardner, E.R.; Mahony, C.; Barnett, S.; Ambrozak, A.; Gutschow, M.; Chau, C.H.; Vargesson, N.; et al. Anticancer properties of a novel class of tetrafluorinated thalidomide analogues. Mol. Cancer Ther. 2015, 14, 2228–2237. [Google Scholar] [CrossRef] [PubMed]
- Joseph, J.; Niggemann, B.; Zaenker, K.S.; Entschladen, F. Anandamide is an endogenous inhibitor for the migration of tumor cells and t lymphocytes. Cancer Immunol. Immunother. 2004, 53, 723–728. [Google Scholar] [CrossRef] [PubMed]
- Schaap-Nutt, A.; Thyssen, D.A.; Drell, D.W.; Entschladen, F. Anti-metastatics: An overview of drug candidates in current pipelines. Curr. Pharm. Des. 2014, 20, 6522–6528. [Google Scholar] [CrossRef] [PubMed]
- Sorlie, T.; Perou, C.M.; Tibshirani, R.; Aas, T.; Geisler, S.; Johnsen, H.; Hastie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. USA 2001, 98, 10869–10874. [Google Scholar] [CrossRef] [PubMed]
- Masur, K.; Lang, K.; Niggemann, B.; Zaenker, K.S.; Entschladen, F. High PKC alpha and low E-cadherin exporession contribute to high migratory activity of colon carcinoma cells. Mol. Biol. Cell 2001, 12, 1973–1982. [Google Scholar] [CrossRef] [PubMed]
- Niggemann, B.; Drell, T.L.; Joseph, J.; Weidt, C.; Lang, K.; Zaenker, K.S.; Entschladen, F. Tumor cell locomotion: Differential dynamics of spontaneous and induced migration in a 3D collagen matrix. Exp. Cell Res. 2004, 298, 178–187. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Entschladen, F.; Thyssen, D.A.; Drell, D.W. Re-Use of Established Drugs for Anti-Metastatic Indications. Cells 2016, 5, 2. https://doi.org/10.3390/cells5010002
Entschladen F, Thyssen DA, Drell DW. Re-Use of Established Drugs for Anti-Metastatic Indications. Cells. 2016; 5(1):2. https://doi.org/10.3390/cells5010002
Chicago/Turabian StyleEntschladen, Frank, Dane A. Thyssen, and David W. Drell. 2016. "Re-Use of Established Drugs for Anti-Metastatic Indications" Cells 5, no. 1: 2. https://doi.org/10.3390/cells5010002
APA StyleEntschladen, F., Thyssen, D. A., & Drell, D. W. (2016). Re-Use of Established Drugs for Anti-Metastatic Indications. Cells, 5(1), 2. https://doi.org/10.3390/cells5010002





