The Anti-Apoptotic Role of Neuroglobin
Abstract
:1. Introduction: Neuroglobin Discovery and Basic Characteristics

2. Normal Expression and Distribution
3. The Impact of Neuroglobin on Cultured Cells, Animal Models and Human Diseases
3.1. Cells
3.2. Animal Models
3.3. Humans
4. Proposed Modes of Action of Neuroglobin
4.1. The Oxygen Model
4.2. The Nitric Oxide Model
4.3. Protein-Protein Interaction Models
- (1)
- Neuroglobin is found in some brain neurons, retina and some endocrine cells.
- (2)
- The presence of neuroglobin protects cultured cells from various challenges such as hypoxia and NO.
- (3)
- In animal models over-expression of neuroglobin protects against stroke and brain injury.
- (4)
- In humans neuroglobin appears up regulated following stroke, Alzheimer’s and glaucoma and is over expressed in some tumors.
- (5)
- Neuroglobin appears to interact with specific proteins such as cytochrome c, GCPR, 14-3-3.
5. Neuroglobin and Apoptosis
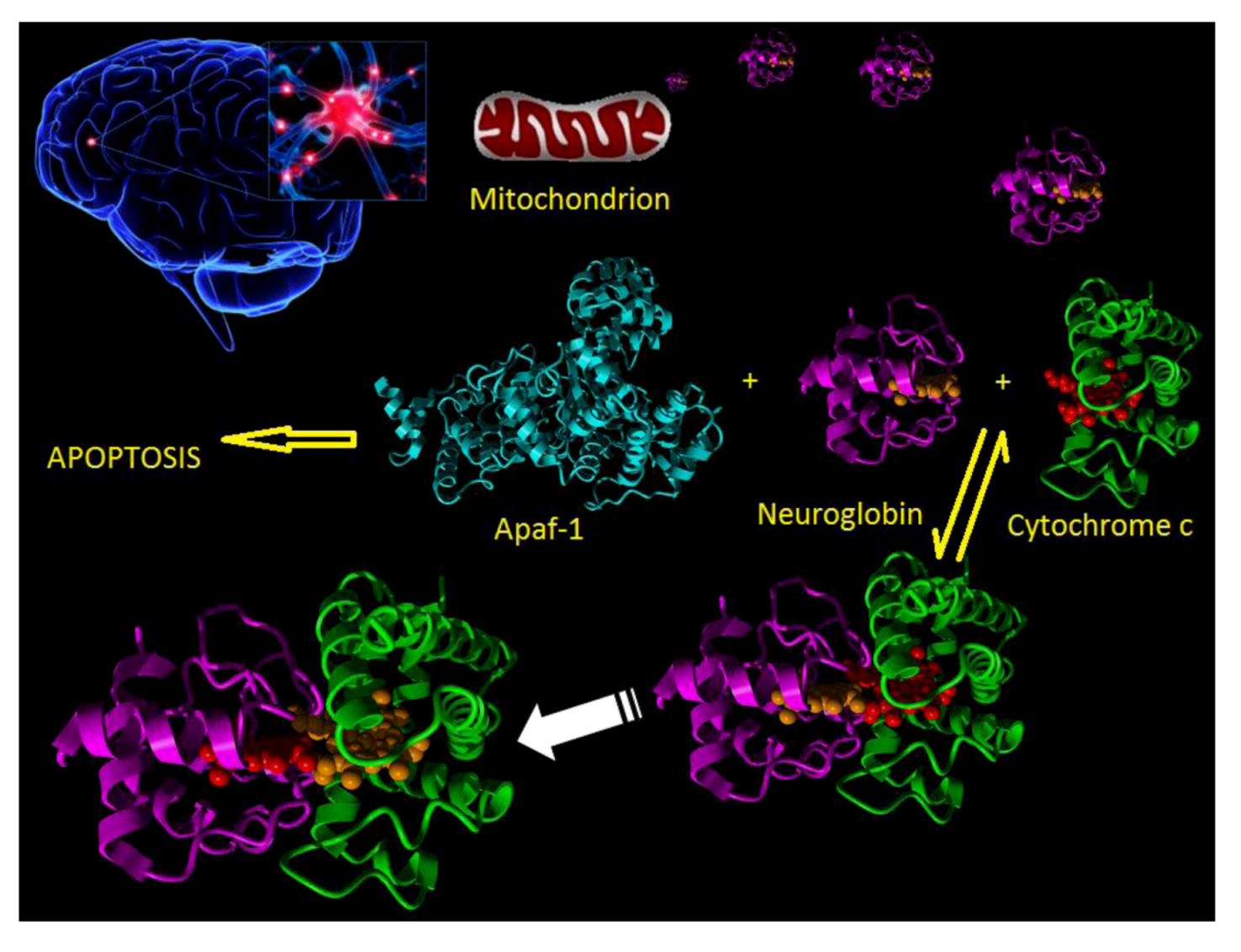
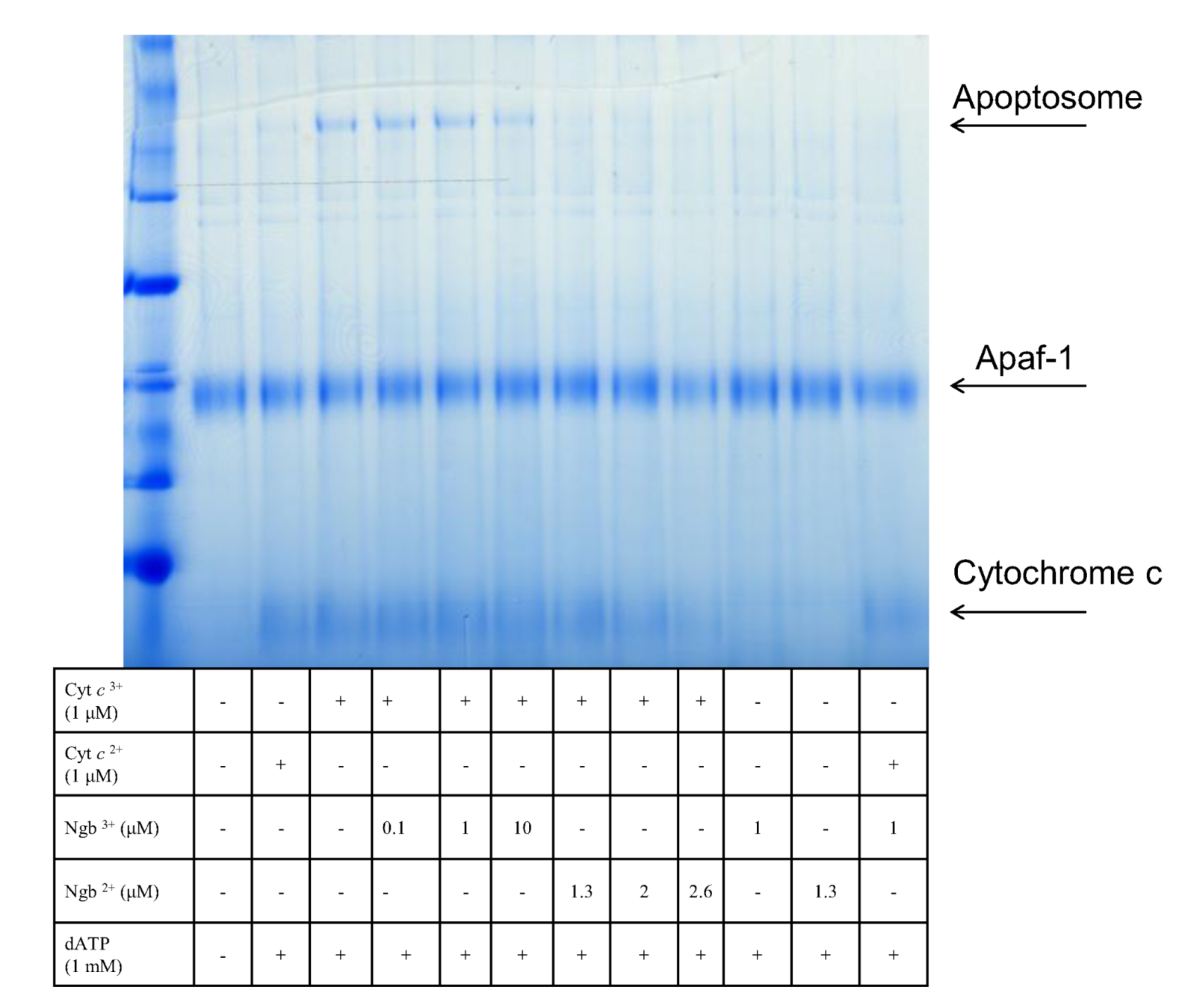

6. Potential Neuroglobin Therapies
7. Conclusions
Conflict of Interest
References
- Burmester, T.; Weich, B.; Reinhardt, S.; Hankeln, T. A vertebrate globin expressed in the brain. Nature 2000, 407, 520–523. [Google Scholar] [CrossRef]
- Vallone, B.; Nienhaus, K.; Matthes, A.; Brunori, M.; Nienhaus, G.U. The structure of carbonmonoxy neuroglobin reveals a heme-sliding mechanism for control of ligand binding. Proc. Natl. Acad. Sci. USA 2004, 101, 17351–17356. [Google Scholar] [CrossRef]
- Pesce, A.; Dewilde, S.; Nardini, M.; Moens, L.; Ascenzi, P.; Hankeln, T.; Burmester, T.; Bolognesi, M. Human brain neuroglobin structure reveals a distinct mode of controlling oxygen affinity. Structure 2003, 11, 1087–1095. [Google Scholar] [CrossRef]
- Vallone, B.; Nienhaus, K.; Brunori, M.; Nienhaus, G.U. The structure of murine neuroglobin: Novel pathways for ligand migration and binding. Proteins 2004, 56, 85–92. [Google Scholar] [CrossRef]
- Wystub, S.; Ebner, B.; Fuchs, C.; Weich, B.; Burmester, T.; Hankeln, T. Interspecies comparison of neuroglobin, cytoglobin and myoglobin: Sequence evolution and candidate regulatory elements. Cytogen. Gen. Res. 2004, 105, 65–78. [Google Scholar] [CrossRef]
- Hankeln, T.; Ebner, B.; Fuchs, C.; Gerlach, F.; Haberkamp, M.; Laufs, T.L.; Roesner, A.; Schmidt, M.; Weich, B.; Wystub, S.; et al. Neuroglobin and cytoglobin in search of their role in the vertebrate globin family. J. Inorg. Biochem. 2005, 99, 110–119. [Google Scholar] [CrossRef]
- Burmester, T.; Haberkamp, M.; Mitz, S.; Roesner, A.; Schmidt, M.; Ebner, B.; Gerlach, F.; Fuchs, C.; Hankeln, T. Neuroglobin and cytoglobin: Genes, proteins and evolution. IUBMB Life 2004, 56, 703–707. [Google Scholar] [CrossRef]
- Fago, A.; Hundahl, C.; Malte, H.; Weber, R.E. Functional properties of neuroglobin and cytoglobin. Insights into the ancestral physiological roles of globins. IUBMB Life 2004, 56, 689–696. [Google Scholar] [CrossRef]
- Dewilde, S.; Mees, K.; Kiger, L.; Lechauve, C.; Marden, M.C.; Pesce, A.; Bolognesi, M.; Moens, L. Expression, purification and crystallization of neuro and cytoglobin. Meth. Enzymol. 2008, 436, 342–357. [Google Scholar]
- Arcovito, A.; Moschetti, T.; D'Angelo, P.; Mancini, G.; Vallone, B.; Brunori, M.; Della Longa, S. An X-ray diffraction and X-ray absorption spectroscopy joint study of neuroglobin. Arch. Biochem. Biophys. 2008, 475, 7–13. [Google Scholar] [CrossRef]
- Dewilde, S.; Kiger, L.; Burmester, T.; Hankeln, T.; Baudin-Creuzal, V.; Aerts, T.; Marden, M.C.; Caubergs, R.; Moens, L. Biochemical characterisation and ligand binding properties of neuroglobin, a novel member of the globin family. J. Biol. Chem. 2001, 276, 38949–38955. [Google Scholar]
- Fago, A.; Mathews, A.J.; Dewilde, S.; Moens, L.; Brittain, T. The reactions of neuroglobin with CO: Evidence for two forms of the ferrous protein. J. Inorg. Biochem. 2006, 100, 1339–1343. [Google Scholar] [CrossRef]
- Brunori, M.; Giuffre, A.; Nienhaus, K.; Nienhaus, G.U.; Scandurra, F.M.; Vallone, B. Neuroglobin, Nitric Oxide and Oxygen: Functional pathways and conformational changes. Proc. Natl. Acad. Sci. USA 2005, 102, 8483–8488. [Google Scholar]
- Kiger, L.; Uzan, J.; Dewilde, S.; Burmester, T.; Hankeln, T.; Moens, L.; Hamdane, D.; Baudin-Creuza, V.; Marden, M. Neuroglobin ligand binding kinetics. IUBMB Life 2004, 56, 709–719. [Google Scholar] [CrossRef]
- Pesce, A.; De Sanctis, D.; Nardini, M.; Dewilde, S.; Moens, L.; Hankeln, T.; Burmester, T.; Ascenzi, P.; Bolognesi, M. Reversible hexa- to penta-coordination of the heme Fe atom modulates ligand binding properties of neuroglobin and cytoglobin. IUBMB Life 2004, 56, 657–664. [Google Scholar] [CrossRef]
- Mu, J.; Li, L.; Guo, Y.; Qiu, Z.; Tan, X. Spectroscopic study on acid-induced unfolding and refolding of apo-neuroglobin. Spectrochim. Acta Part A Mol. Biomol. Spec. 2010, 75, 1600–1604. [Google Scholar] [CrossRef]
- Picotti, P.; Dewilde, S.; Fago, A.; Hundahl, C.; De Filippis, V.; Moens, L.; Fontana, A. Unusual stability of human neuroglobin at low pH—Molecular mechanisms and biological significance. FEBS J. 2009, 276, 7027–7039. [Google Scholar]
- Hamdane, D.; Kiger, L.; Dewilde, S.; Uzan, J.; Burmester, T.; Hankeln, T.; Moens, L.; Marden, M.C. Hyperthermal stability of neuroglobin and cytoglobin. FEBS J. 2005, 272, 2076–2084. [Google Scholar] [CrossRef]
- Burmester, T.; Hankeln, T. Neuroglobin: A respiratory protein of the nervous system. News Physiol. Sci. 2004, 19, 110–113. [Google Scholar]
- Bentmann, A.; Schmidt, M.; Reuss, S.; Wolfrum, U.; Hankeln, T.; Burmester, T. Divergent distribution in vascular and avascular mammalian retinae links neuroglobin to cellular respiration. J. Biol. Chem. 2005, 280, 20660–20665. [Google Scholar]
- Geuens, E.; Brouns, I.; Flamez, D.; Dewilde, S.; Timmermans, J.P.; Moens, L. A globin in the nucleus. J. Biol. Chem. 2003, 278, 30417–30420. [Google Scholar]
- Mammen, P.P.; Shelton, J.M.; Goetsch, S.C.; Williams, S.C.; Richardson, J.A.; Garry, M.G.; Garry, D.J. Neuroglobin, a novel member of the globin family, is expressed in focal regions of the brain. J. Histochem. Cytochem. 2002, 50, 1591–1598. [Google Scholar] [CrossRef]
- Reuss, S.; Saaler-Reinhardt, S.; Weich, B.; Wystub, S.; Reuss, M.H.; Burmester, T.; Hankeln, T. Expression analysis of neuroglobin mRNA in rodent tissues. Neuroscience 2002, 11, 645–656. [Google Scholar]
- Wystub, S.; Laufs, T.; Schmidt, M.; Burmester, T.; Maas, U.; Saaler-Reinhardt, S.; Hankeln, T.; Reuss, S. Localization of neuroglobin protein in the mouse brain. Neurosci. Lett. 2003, 346, 114–116. [Google Scholar]
- Laufs, T.L.; Wystub, S.; Reuss, S.; Burmester, T.; Saaler-Reinhardt, S.; Hankeln, T. Neuron-specific expression of neuroglobin in mammals. Neurosci. Lett. 2004, 362, 83–86. [Google Scholar] [CrossRef]
- Hundahl, C.A.; Allen, G.C.; Hannibal, J.; Kjaer, K.; Rehfeld, J.F.; Dewilde, S.; Nyengaard, J.R.; Kelsen, J.; Hay-Schmidt, A. Anatomical characterization of Cytoglobin and Neuroglobin mRNA and protein expression in the mouse brain. Brain Res. 2010, 1355, 58–63. [Google Scholar]
- Hundahl, C.A.; Allen, G.C.; Nyengaard, J.R.; Dewilde, S.; Carter, B.D.; Kelsen, J.; Ay-Schmidt, A. Neuroglobin in the rat brain: Localization. Neuroendocrin 2008, 88, 173–182. [Google Scholar]
- Hundahl, C.A.; Kelsen, J.; Dewilde, S.; Hay-Schmidt, A. Neuroglobin in the rat brain (II): Co-localisation with neurotransmitters. Neuroendocrinology 2008, 88, 183–198. [Google Scholar] [CrossRef]
- Hundahl, C.A.; Hannibal, J.; Fahrenkrug, J.; Dewilde, S.; Hay-Schmidt, A. Neuroglobin expression in the rat suprachiasmatic nucleus: Colocalization, innervation, and response to light. J. Comp. Neurol. 2010, 518, 1556–1569. [Google Scholar] [CrossRef]
- Schmidt, M.; Giessl, A.; Laufs, T.; Hankeln, T.; Wolfrum, U.; Burmester, T. How does the eye breathe. J. Biol. Chem. 2003, 278, 1932–1935. [Google Scholar]
- Schmidt, M.; Laufs, T.; Reuss, S.; Hankeln, T.; Burmester, T. Divergent distribution of cytoglobin and neuroglobin in the murine eye. Neurosci. Lett. 2005, 374, 207–211. [Google Scholar]
- Ostojić, J.; Sakaguchi, D.S.; de Lathouder, Y.; Hargrove, M.S.; Trent, J.T.; Kwon, Y.H.; Kardon, R.H.; Kuehn, M.H.; Betts, D.M.; Grozdanic, S. Nuroglobin and cytoglobin: Oxygen-binding proteins in retinal neurons. IOVS 2006, 47, 1016–1023. [Google Scholar]
- Hankeln, T. Institute for Molecular Genetics, Johannes Gutenberg University, Germany. personal communication, 2012.
- Hundahl, C.A.; Fahrenkrug, J.; Luuk, H.; Hay-Schmidt, A.; Hannibal, J. Restricted expression of neuroglobin in the mouse retina and co-localization with Melanopsin and Tyrosine Hydroxylase. Biochem. Biophys. Res. Commun. 2012, 425, 100–106. [Google Scholar] [CrossRef]
- Fordel, E.; Geuens, E.; Dewilde, S.; De Coen, W.; Moens, L. Hypoxia/ischemia and the regulation of neuroglobin and cytoglobin expression. IUBMB Life 2004, 56, 681–687. [Google Scholar] [CrossRef]
- Zhang, W.; Tian, Z.; Sha, S.; Cheng, L.Y.; Philipsen, S.; Tan-Un, K.C. Functional and sequence analysis of human neuroglobin gene promoter region. Biochim. Biophys. Acta 2011, 1809, 236–244. [Google Scholar]
- Liu, N.; Yu, Z.; Xiang, S.; Zhao, S.; Tjarnlund-Wolf, A.; Xing, C.; Zhang, J.; Wang, X. Transcriptional regulation mechanisms of hypoxia-induced neuroglobin gene expression. Biochem. J. 2012, 443, 153–164. [Google Scholar] [CrossRef]
- Li, R.C.; Pouranfar, F.; Lee, S.K.; Morris, M.W.; Wang, Y.; Gozal, D. Neuroglobin protects PC12 cells against beta-amyloid-induced cell injury. Neurobiol. Aging 2008, 29, 1815–1822. [Google Scholar] [CrossRef]
- Li, R.C.; Morris, M.W.; Lee, S.K.; Pouranfar, F.; Wang, Y.; Gozal, D. Neuroglobin protects PC12 cells against oxidative stress. Brain Res. 2008, 1190, 159–166. [Google Scholar] [CrossRef]
- Fordel, E.; Thijs, L.; Martinet, W.; Lenjou, M.; Laufs, T.; Van Bockstaele, D.; Moens, L.; Dewilde, S. Neuroglobin and cytoglobin overexpression protects human SH-SY5Y neuroblastoma cells against oxidative stress-induced cell death. Neurosci. Lett. 2006, 410, 146–151. [Google Scholar] [CrossRef]
- Fordel, E.; Thijs, L.; Martinet, W.; Schrijvers, D.; Moens, L.; Dewilde, S. Anoxia or oxygen and glucose deprivation in SH-SY5Y cells: A step closer to the unraveling of neuroglobin and cytoglobin functions. Gene 2007, 398, 114–122. [Google Scholar] [CrossRef]
- Skommer, J.; Brittain, T. Extended survival of SH-SY5Y cells following overexpression of Lys67Glu neuroglobin is associated with stabilization of ΔΨΜ. Cytometry Part A 2012, 81, 602–610. [Google Scholar] [CrossRef]
- Jin, K.; Mao, X.O.; Xie, L.; Khan, A.A.; Greenberg, D.A. Neuroglobin protects against nitric oxide toxicity. Neurosci. Lett. 2008, 430, 135–137. [Google Scholar]
- Yu, Z.; Liu, J.; Guo, S.; Xing, C.; Fan, X.; Ning, M.; Yuan, J.C.; Lo, E.H.; Wang, X. Neuroglobin-overexpression alters hypoxic response gene expression in primary neuron culture following oxygen glucose deprivation. Neuroscience 2009, 162, 396–403. [Google Scholar] [CrossRef]
- Yu, Z.; Xu, J.; Liu, N.; Wang, Y.; Li, X.; Pallast, S.; van Leyen, K.; Wang, X. Mitochondrial distribution of neuroglobin and its response to oxygen-glucose deprivation in primary-cultured mouse cortical neurons. Neuroscience 2012, 218, 235–242. [Google Scholar] [CrossRef]
- Liu, J.; Yu, Z.; Guo, S.; Lee, S.R.; Xing, C.; Zhang, C.; Gao, Y.; Nicholls, D.G.; Lo, E.H.; Wang, X. Effects of neuroglobin overexpression on mitochondrial function and oxidative stress following hypoxia/reoxygenation in cultured neurons. J. Neurosci. Res. 2009, 87, 164–170. [Google Scholar] [CrossRef]
- Antao, S.T.; Duong, T.T.; Aran, R.; Witting, P.K. Neuroglobin overexpression in cultured human neuronal cells protects against hydrogen peroxide insult via activating phosphoinositide-3 kinase and opening the mitochondrial K(ATP) channel. Antioxid. Redox Signal. 2010, 13, 769–781. [Google Scholar] [CrossRef]
- Sun, Y.; Jin, K.; Peel, A.; Mao, X.O.; Xie, L.; Greenberg, D.A. Neuroglobin protects the brain from experimental stroke in vivo. Proc. Natl. Acad. Sci. USA 2003, 100, 3497–3500. [Google Scholar]
- Wang, X.; Liu, J.; Zhu, H.; Tejima, E.; Tsuji, K.; Murata, Y.; Atochin, D.N.; Huang, P.L.; Zhang, C.; Lo, E.H. Effects of neuroglobin overexpression on acute brain injury and long-term outcomes after focal cerebral ischemia. Stroke 2008, 39, 1869–1874. [Google Scholar]
- Zhu, J.; Tang, W. Cerebroprotection with recombinant neuroglobin plasmid in a rat model of focal cerebral ischemia. Neural. Regen. Res. 2010, 5, 52–57. [Google Scholar]
- Li, R.C.; Guo, S.Z.; Lee, S.K.; Gozal, D. Neuroglobin protects neurons against oxidative stress in global ischemia. J. Cereb. Blood Flow Metab. 2010, 30, 1874–1882. [Google Scholar] [CrossRef]
- Khan, A.A.; Sun, Y.; Jin, K.; Mao, X.O.; Chen, S.; Ellerby, L.M.; Greenberg, D.A. A neuroglobin-overexpressing transgenic mouse. Gene 2007, 398, 172–176. [Google Scholar] [CrossRef]
- Khan, A.A.; Wang, Y.; Sun, Y.; Mao, X.O.; Xie, L.; Miles, E.; Graboski, J.; Chen, S.; Ellerby, L.M.; Jin, K.; et al. Neuroglobin-overexpressing transgenic mice are resistant to cerebral and myocardial ischemia. Proc. Natl. Acad. Sci. USA 2006, 103, 17944–17948. [Google Scholar]
- Hundahl, C.A.; Luuk, H.; Ilmjarv, S.; Falktoft, B.; Raida, Z.; Vikesaa, J.; Friis-Hansen, L.; Hay-Schmidt, A. Neuroglobin-deficiency exacerbates Hif1A and c-FOS response, but does not affect neuronal survival during severe hypoxia in vivo. PLoS one 2011, 6, e28160. [Google Scholar]
- Raida, Z.; Hundahl, C.A.; Kelsen, J.; Nyengaard, J.R.; Hay-Schmidt, A. A reduced infarct size in neuroglobin-null mice after experimental stroke in vivo. Expt. Trans. Stroke Med. 2012, 4, 1–12. [Google Scholar] [CrossRef]
- Li, R.C.; Lee, S.K.; Pouranfar, F.; Brittian, K.R.; Clair, H.B.; Row, B.W.; Wang, Y.; Gozal, D. Hypoxia differentially regulates the expression of neuroglobin and cytoglobin in rat brain. Brain Res. 2006, 1096, 173–179. [Google Scholar] [CrossRef]
- Schmidt-Kastner, R.; Haberkamp, M.; Schmitz, C.; Hankeln, T.; Burmester, T. Neuroglobin mRNA expression after transient global brain ischemia and prolonged hypoxia in cell culture. Brain Res. 2006, 1103, 173–180. [Google Scholar] [CrossRef]
- Huemmler, N.; Schneider, C.; Giessl, A.; Bauer, R.; Walkinshaw, G.; Gassmann, M.; Rascher, W.; Trollmann, R. Acute hypoxia modifies regulation of neuroglobin in the neonatal mouse brain. Expt. Neurol. 2012, 236, 112–121. [Google Scholar] [Green Version]
- Hundahl, C.; Kelsen, J.; Kjaer, K.; Ronn, L.C.; Weber, R.E.; Geuens, E.; Hay-Schmidt, A.; Nyengaard, J.R. Does neuroglobin protect neurons from ischemic insult? A quantitative investigation of neuroglobin expression following transient MCAo in spontaneously hypertensive rats. Brain Res. 2006, 1085, 19–27. [Google Scholar] [CrossRef]
- Shao, G.; Gong, K.R.; Li, J.; Xu, X.J.; Gao, C.Y.; Zeng, X.Z.; Lu, G.W.; Huo, X. Antihypoxic effects of neuroglobin in hypoxia-preconditioned mice and SH-SY5Y cells. Neurosignals 2009, 17, 196–202. [Google Scholar] [CrossRef]
- Shang, A.; Zhou, D.; Wang, L.; Gao, Y.; Fan, M.; Wang, X.; Zhou, R.; Zhang, C. Increased neuroglobin levels in the cerebral cortex and serum after ischemia-reperfusion insults. Brain Res. 2006, 1078, 219–226. [Google Scholar] [CrossRef]
- Hundahl, C.; Stoltenberg, M.; Fago, A.; Weber, R.E.; Dewilde, S.; Fordel, E.; Danscher, G. Effects of short-term hypoxia on neuroglobin levels and localization in mouse brain tissues. Neuropathol. App. Neurobiol. 2005, 31, 610–617. [Google Scholar] [CrossRef]
- Liu, C.; Sun, S.; Yu, J. Up-regulation of expression of neuroglobin in rats with brain injury induced by endotoxin. Chinese J. Biol. 2009, 22, 845–849. [Google Scholar]
- Hundahl, C.A.; Hannibal, J.; Fahrenkrug, J.; Dewilde, S.; Hay-Schmidt, A. Neuroglobin expression in the rat suprachiasmatic nucleus: Colocalization, innervation, and response to light. J. Comp. Neurol. 2010, 518, 1556–1569. [Google Scholar] [CrossRef]
- Beltran-Parrazal, L.; Acuna, D.; Ngan, A.M.; Kim, E.; Ngan, A.; Kawakami, K.; Edmond, J.; Lopez, I.A. Neuroglobin, cytoglobin, and transcriptional profiling of hypoxia-related genes in the rat cerebellum after prenatal chronic very mild carbon monoxide exposure (25 ppm). Brain Res. 2010, 1330, 61–71. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, W.; Sun, D.; Song, L.; Li, Y.; Xu, C. Analysis of neuroglobin mRNA expression in rat brain due to arsenite-induced oxidative stress. Environ. Toxicol. 2012, 27, 503–509. [Google Scholar] [CrossRef]
- Shang, A.; Liu, K.; Wang, H.; Wang, J.; Hang, X.; Yang, Y.; Wang, Z.; Zhang, C.; Zhou, D. Neuroprotective effects of neuroglobin after mechanical injury. Neurol. Sci. 2012, 33, 551–558. [Google Scholar] [CrossRef]
- Shang, A.; Feng, X.; Wang, H.; Wang, J.; Hang, X.; Yang, Y.; Wang, Z.; Zhou, D. Neuroglobin upregulation offers neuroprotection in traumatic brain injury. Neurol. Res. 2012, 34, 588–594. [Google Scholar]
- Shi, S.Y.; Feng, X.M.; Li, Y.; Li, X.; Chen, X.L. Expression of neuroglobin in ocular hypertension induced acute hypoxic-ischemic retinal injury in rats. Int. J. Ophthal. Mol. 2011, 4, 393–395. [Google Scholar]
- Lopez, I.A.; Acuna, D.; Shahram, Y.; Mowlds, D.; Ngan, A.M.; Rungvivatjarus, T.; Sharma, Y.; Edmond, J. Neuroglobin expression in the cochlea of rat pups exposed to chronic very mild carbon monoxide (25ppm) in air during and after the prenatal period. Brain Res. 2010, 1327, 56–68. [Google Scholar] [CrossRef]
- Sun, Y.; Jin, K.; Mao, X.O.; Xie, L.; Peel, A.; Childs, J.T.; Logvinova, A.; Wang, X.; Greenberg, D.A. Effect of aging on neuroglobin expression in rodent brain. Neurobiol. Aging 2005, 26, 275–278. [Google Scholar] [CrossRef]
- Chen, X.Q.; Qin, L.Y.; Zhang, C.G.; Yang, L.T.; Gao, Z.; Liu, S.; Lau, L.T.; Fung, Y.W.; Greenberg, D.A.; Yu, A.C. Presence of neuroglobin in cultured astrocytes. GLIA 2005, 50, 182–186. [Google Scholar] [CrossRef]
- DellaValle, B.; Hempel, C.; Kurtzhals, J.A.; Penkowa, M. In vivo expression of neuroglobin in reactive astrocytes during neuropathology in murine models of traumatic brain injury, cerebral malaria, and autoimmune encephalitis. GLIA 2010, 58, 1220–1227. [Google Scholar]
- Jin, K.; Mao, Y.; Mao, X.; Xie, L.; Greenberg, D.A. Neuroglobin expression in ischemic stroke. Stroke 2010, 41, 557–559. [Google Scholar] [CrossRef]
- Yu, Z.; Liu, N.; Liu, J.; Yang, K.; Wang, X. Neuroglobin, a novel target for endogenous neuroprotection against stroke and neurodegenerative disorders. Int. J. Mol. Sci. 2012, 13, 6995–7014. [Google Scholar] [CrossRef]
- Wang, X.; Liu, J.; Zhu, H.; Tejima, E.; Tsuji, K.; Murata, Y.; Atochin, D.N.; Huang, P.L.; Zhang, C.; Lo, E.H. Effects of neuroglobin overexpression on acute brain injury and long-term outcomes after focal cerebral ischemia. Stroke 2008, 39, 1869–1874. [Google Scholar] [CrossRef]
- Jin, K.; Mao, X.; Xie, L.; Greenberg, D.A. Neuroglobin expression in human arteriovenous malformation and intracerebral hemorrhage. Acta Neurochir. 2011, 111, 315–319. [Google Scholar] [CrossRef]
- Lin, Y.; Fang, L.; Xue, X.H.; Murong, S.X.; Wang, N.; Wu, Z.Y. Association between Ngb polymorphisms and ischemic stroke in the Southern Chinese Han population. BMC Med. Gen. 2008, 9, 110–120. [Google Scholar]
- Emara, M.; Salloum, N.; Allalunis-Turner, J. Expression and hypoxic up-regulation of neuroglobin in human glioblastoma cells. Mol. Oncol. 2009, 3, 45–53. [Google Scholar] [CrossRef]
- Qin, H.; Guo, Y.; Zhang, C.; Zhang, L.; Li, M.; Guan, P. The expression of neuroglobin in astrocytoma. Brain Tumor Pathol. 2012, 29, 10–16. [Google Scholar] [CrossRef]
- Oleksiewicz, U.; Daskoulidou, N.; Liloglou, T.; Tasopoulou, K.; Bryan, J.; Gosney, J.R.; Field, J.K.; Xinarianos, G. Neuroglobin and myoglobin in non-small cell lung cancer: Expression, regulation and prognosis. Lung Cancer 2011, 74, 411–418. [Google Scholar]
- Emara, M.; Turner, A.R.; Allalunis-Turner, J. Hypoxic regulation of cytoglobin and neuroglobin expression in human normal and tumor tissues. Cancer Cell Int. 2010, 10, 33. [Google Scholar] [CrossRef]
- Khan, A.A.; Mao, X.O.; Banwait, S.; Jin, K.; Greenberg, D.A. Neuroglobin attenuates beta-amyloid neurotoxicity in vitro and transgenic Alzheimer phenotype in vivo. Proc. Natl. Acad. Sci. USA 2007, 104, 19114–19119. [Google Scholar] [CrossRef]
- Szymanski, M.; Wang, R.; Fallin, M.D.; Bassett, S.S.; Avramopoulos, D. Neuroglobin and Alzheimer's dementia: genetic association and gene expression changes. Neurobiol. Aging 2010, 3, 1835–1842. [Google Scholar]
- Chuang, P.Y.; Conley, Y.P.; Poloyac, S.M.; Okonkwo, D.O.; Ren, D.; Sherwood, P.R.; Hravnak, M.; Alexander, S.A. Neuroglobin genetic polymorphisms and their relationship to functional outcomes after traumatic brain injury. J. Neurotrauma 2010, 27, 999–1006. [Google Scholar] [CrossRef]
- Shang, A.; Feng, X.; Wang, H.; Wang, J.; Hang, X.; Yang, Y.; Wang, Z.; Zhou, D. Neuroglobin upregulation offers neuroprotection in traumatic brain injury. Neurol. Res. 2012, 34, 588–594. [Google Scholar] [CrossRef]
- Ostojic, J.; Grozdanic, S.D.; Syed, N.A.; Hargrove, M.S.; Trent, J.T.; Kuehn, M.H.; Kwon, Y.H.; Kardon, R.H.; Sakaguchi, D.S. Patterns of distribution of oxygen-binding globins, neuroglobin and cytoglobin in human retina. Arch. Ophthal. Mol. 2008, 126, 1530–1536. [Google Scholar] [CrossRef]
- Ostojic, J.; Grozdanic, S.; Syed, N.A.; Hargrove, M.S.; Trent, J.T.; Kuehn, M.H.; Kardon, R.H.; Kwon, Y.H.; Sakaguchi, D.S. Neuroglobin and cytoglobin distribution in the anterior eye segment: A comparative immunohistochemical study. J. Histochem. Cytochem. 2008, 56, 863–872. [Google Scholar] [CrossRef]
- Ostojic, J.; Sakaguchi, D.S.; de Lathouder, Y.; Hargrove, M.S.; Trent, J.T.; Kwon, Y.H.; Kardon, R.H.; Kuehn, M.H.; Betts, D.M.; Grozdanic, S. Neuroglobin and cytoglobin: oxygen-binding proteins in retinal neurons. Invest. Ophthal. Mol. Vis. Sci. 2006, 47, 1016–1023. [Google Scholar] [CrossRef]
- Chan, A.S.; Saraswathy, S.; Rehak, M.; Ueki, M.; Rao, N.A. Neuroglobin protection in retinal ischemia. Invest. Ophthal. Mol. Vis. Sci. 2012, 53, 704–711. [Google Scholar]
- Rajendram, R.; Rao, N.A. Neuroglobin in normal retina and retina from eyes with advanced glaucoma. Brit. J. Ophthal. Mol. 2007, 91, 663–666. [Google Scholar] [CrossRef]
- Wei, X.; Yu, Z.; Cho, K.S.; Chen, H.; Malik, M.T.; Chen, X.; Lo, E.H.; Wang, X.; Chen, D.F. Neuroglobin is an endogenous neuroprotectant for retinal ganglion cells against glaucomatous damage. Amer. J. Pathol. 2011, 179, 2788–2797. [Google Scholar] [CrossRef]
- Burmester, T.; Hankeln, T. What is the function of neuroglobin? J. Expt. Biol. 2009, 212, 1423–1428. [Google Scholar] [CrossRef]
- Giuffre, A.; Moschetti, T.; Vallone, B.; Brunori, M. Is there an answer? Is neuroglobin a signal transducer? IUBMB Life 2008, 60, 410–413. [Google Scholar] [CrossRef]
- Yu, Z.; Fan, X.; Lo, E.H.; Wang, X. Neuroprotective roles and mechanisms of neuroglobin. Neurol. Res. 2009, 31, 122–127. [Google Scholar] [CrossRef]
- Brunori, M.; Vallone, B. A globin for the brain. FASEB J. 2006, 20, 2192–2197. [Google Scholar] [CrossRef]
- Nienhaus, K.; Nienhaus, G.U. Searching for neuroglobin's role in the brain. IUBMB Life 2007, 59, 490–497. [Google Scholar] [CrossRef]
- Brunori, M.; Vallone, B. Neuroglobin, seven years after. Cell Mol. Life Sci. 2007, 64, 1259–1268. [Google Scholar] [CrossRef]
- Garry, D.J.; Mammen, P.P. Neuroprotection and the role of neuroglobin. Lancet 2003, 362, 342–343. [Google Scholar] [CrossRef]
- Venis, S. Neuroglobin might protect brain cells during stroke. Lancet 2001, 358, 2055. [Google Scholar]
- Kelsen, J.; Hundahl, C.A.; Hay-Schmidt, A. Neuroglobin: Endogenous neuroprotectant or maintenance of homeostasis? Stroke 2008, 39, e177–e178. [Google Scholar] [CrossRef]
- Greenberg, D.A.; Jin, K.; Khan, A.A. Neuroglobin: An endogenous neuroprotectant. Curr. Opin. Pharmacol. 2008, 8, 20–24. [Google Scholar] [CrossRef]
- Hankeln, T.; Wystub, S.; Laufs, T.; Schmidt, M.; Gerlach, F.; Saaler-Reinhardt, S.; Reuss, S.; Burmester, T. The cellular and subcellular localization of neuroglobin and cytoglobin—a clue to their function? IUBMB Life 2004, 56, 671–679. [Google Scholar] [CrossRef]
- Schmidt, M.; Giessl, A.; Laufs, T.; Hankeln, T.; Wolfrum, U.; Burmester, T. How does the eye breathe? Evidence for neuroglobin-mediated oxygen supply in the mammalian retina. J. Biol. Chem. 2003, 278, 1932–1935. [Google Scholar]
- Fago, A.; Hundahl, C.; Dewilde, S.; Gilany, K.; Moens, L.; Weber, R.E. Allosteric regulation and temperature dependence of oxygen binding in human neuroglobin and cytoglobin. J. Biol. Chem. 2004, 279, 44417–44426. [Google Scholar]
- Trandafir, F.; Hoogewijs, D.; Altieri, F.; Rivetti de val Cervo, P.; Ramser, K.; van Doorslaer, S.; Vanfleteren, J.R.; Moens, L.; Dewilde, S. Neuroglobin and cytoglobin as potential enzymes or substrates. Gene 2007, 398, 103–113. [Google Scholar] [CrossRef]
- Guiffre, A.; Moschetti, T.; Vallone, B.; Brunori, M. Neuroglobin: Enzymatic reduction and oxygen affinity. Biochem. Biophys. Res. Co. 2008, 367, 893–898. [Google Scholar] [CrossRef]
- Moschetti, T.; Giuffrè, A.; Ardiccioni, C.; Vallone, B.; Modjtahedi, N.; Kroemer, G.; Brunori, M. Failure of apoptosis-inducing factor to act as neuroglobin reductase. Biochem. Biophy. Res. Co. 2009, 390, 121–124. [Google Scholar] [CrossRef]
- Fordel, E.; Thijs, L.; Moens, L.; Dewilde, S. Neuroglobin and cytoglobin expression in mice. Evidence for a correlation with reactive oxygen species scavenging. FEBS J. 2007, 274, 1312–1317. [Google Scholar] [CrossRef]
- Li, W.; Wu, Y.; Ren, C.; Lu, Y.; Gao, Y.; Zheng, X.; Zhang, C. The activity of recombinant human neuroglobin as an antioxidant and free radical scavenger. Proteins 2011, 79, 115–125. [Google Scholar] [CrossRef]
- Van Doorslaer, S.; Dewilde, S.; Kiger, L.; Nistor, S.V.; Goovaertts, E.; Marden, M.C.; Moens, L. Nitric oxide binding properties of neuroglobin. J. Biol. Chem. 2003, 278, 4919–4925. [Google Scholar]
- Petersen, M.G.; Dewilde, S.; Fago, A. Reactions of ferrous neuroglobin and cytoglobin with nitrite under anaerobic conditions. J. Inorg. Biochem. 2008, 102, 1777–1782. [Google Scholar] [CrossRef]
- Tiso, M.; Tejero, J.; Basu, S.; Azarov, I.; Wang, X.; Simplaceanu, V.; Frizell, S.; Jayaraman, T.; Geary, L.; Shapiro, C.; et al. Human neuroglobin functions as a redox-regulated nitrite reductase. J. Biol. Chem. 2011, 286, 18277–18289. [Google Scholar]
- Smagghe, B.J.; Trent, J.T.; Hargrove, M.S. NO dioxygenase activity in hemoglobins is ubiquitous in vitro but limited by reduction in vivo. PloS one 2008, 3, e2039. [Google Scholar]
- Herold, S.; Fago, A.; Weber, R.E.; Dewilde, S.; Moens, L. Reactivity studies of the Fe(III) and Fe(II)NO forms of human neuroglobin reveal a potential role against oxidative stress. J. Biol. Chem. 2004, 279, 22841–22847. [Google Scholar]
- Herold, S.; Fago, A. Reactions of peroxynitrite with globin proteins and their possible physiological role. Comp. Biochem. Physiol. 2005, 142, 124–129. [Google Scholar] [CrossRef]
- Haines, B.A.; Davis, D.A.; Zykovich, A.; Peng, B.; Rao, R.; Mooney, S.D.; Jin, K.; Greenberg, D.A. Comparative protein interactomics of neuroglobin and myoglobin. J. Neurochem. 2012, 123, 192–198. [Google Scholar] [CrossRef]
- Yu, Z.; Liu, N.; Wang, Y.; Li, X.; Wang, X. Identification of Neuroglobin-interacting proteins using yeast two-hybrid screening. Neuroscience 2012, 200, 99–105. [Google Scholar] [CrossRef]
- Wakasugi, K.; Nakano, T.; Morishima, I. Oxidized human neuroglobin acts as a heterotrimeric Galpha protein guanine nucleotide dissociation inhibitor. J. Biol. Chem. 2003, 278, 36505–36512. [Google Scholar] [CrossRef]
- Wakasugi, K.; Morishima, I. Identification of residues in human neuroglobin crucial for Guanine nucleotide dissociation inhibitor activity. Biochemistry 2005, 44, 2943–2948. [Google Scholar] [CrossRef]
- Wakasugi, K.; Kitatsuji, C.; Morishima, I. Possible neuroprotective mechanism of human neuroglobin. Ann. NY Acad. Sci. 2005, 1053, 220–230. [Google Scholar] [CrossRef]
- Kitatsuji, C.; Kurogochi, M.; Nishimura, S.; Ishimori, K.; Wakasugi, K. Molecular basis of guanine nucleotide dissociation inhibitor activity of human neuroglobin by chemical cross-linking and mass spectrometry. J. Mol. Biol. 2007, 368, 150–160. [Google Scholar] [CrossRef]
- Schwindinger, W.F.; Robishaw, J.D. Heterotrimeric G-protein βγ -dimers in growth and differentiation. Oncogene 2001, 20, 1653–1660. [Google Scholar] [CrossRef]
- Watanabe, S.; Wakasugi, K. Neuroprotective function of human neuroglobin is correlated with its guanine nucleotide dissociation inhibitor activity. Biochem. Biophy. Res. Co. 2008, 369, 695–700. [Google Scholar] [CrossRef]
- Watanabe, S.; Takahashi, N.; Uchida, H.; Wakasugi, K. Human neuroglobin functions as an oxidative stress-responsive sensor for neuroprotection. J. Biol. Chem. 2012, 287, 30128–30138. [Google Scholar]
- Palladino, P.; Scaglione, G.L.; Arcovito, A.; Maria Vitale, R.; Amodeo, P.; Vallone, B.; Brunori, M.; Benedetti, E.; Rossi, F. Neuroglobin-prion protein interaction: What's the function? J. Pept. Sci. 2011, 17, 387–391. [Google Scholar] [CrossRef]
- Lechauve, C.; Rezaei, H.; Celier, C.; Kiger, L.; Corral-Debrinski, M.; Noinville, S.; Chauvierre, C.; Hamdane, D.; Pato, C.; Marden, M.C. Neuroglobin and Prion Cellular Localization: Investigation of a Potential Interaction. J. Mol. Biol. 2009, 388, 968–977. [Google Scholar] [CrossRef]
- Chen, L.; Xiong, Y.; Kong, F.; Qu, M.; Wang, Q.; Chen, X.; Wang, J.; Zhu, L. Neuroglobin attenuates Alzheimer-like tau hyperphosphorylation by activating Akt signaling. J. Neurochem. 2012, 120, 157–164. [Google Scholar]
- Ye, S.; Zhou, X.; Lai, X.; Zheng, L.; Chen, X. Silencing neuroglobin enhances neuronal vulnerability to oxidative injury by down-regulating 14–3-3 gamma Acta. Pharmacol. Sin. 2009, 30, 913–918. [Google Scholar] [CrossRef]
- Dong, Y.; Zhao, R.; Chen, X.Q.; Yu, A.C. 14–3-3gamma and neuroglobin are new intrinsic protective factors for cerebral ischemia. Mol. Neurobiol. 2010, 41, 218–231. [Google Scholar] [CrossRef]
- Jayaraman, T.; Tejero, J.; Chen, B.B.; Blood, A.B.; Frizzell, S.; Shapiro, C.; Tiso, M.; Hood, B.L.; Wang, X.; Zhao, X.; et al. 14–3-3 Binding and Phosphorylation of Neuroglobin during Hypoxia Modulate Six-to-Five Heme Pocket Coordination and Rate of Nitrite Reduction to Nitric Oxide. J. Biol. Chem. 2011, 286, 42679–42689. [Google Scholar]
- Fago, A.; Mathews, A.J.; Moens, L.; Dewilde, S.; Brittain, T. The reaction of neuroglobin with potential redox protein partners cytochrome b5 and cytochrome c. FEBS Letts. 2006, 580, 4884–4888. [Google Scholar] [CrossRef]
- Bonding, S.H.; Henty, K.; Dingley, A.J.; Brittain, T. The binding of cytochrome c to neuroglobin: a docking and surface plasmon resonance study. Int. J. Biol. Macromol. 2008, 43, 295–299. [Google Scholar]
- Fago, A.; Mathews, A.J.; Brittain, T. A role for neuroglobin: resetting the trigger level for apoptosis in neuronal and retinal cells. IUBMB Life 2008, 60, 398–401. [Google Scholar]
- Brittain, T.; Skommer, J.; Henty, K.; Birch, N.; Raychaudhuri, S. A role for human neuroglobin in apoptosis. IUBMB Life 2010, 62, 878–885. [Google Scholar] [CrossRef]
- Brittain, T.; Skommer, J.; Raychaudhuri, S.; Birch, N. An antiapoptotic neuroprotective role for neuroglobin. Int. J. Mol. Sci. 2010, 11, 2306–2321. [Google Scholar] [CrossRef]
- Kang, Y.; Kim, P.K.; Choi, B.; Chung, H.; Ha, K.; Kwon, Y.; Kim, Y. Regulation of programmed cell death in neuronal cells by nitric oxide. In Vivo 2004, 18, 367–376. [Google Scholar]
- Crompton, M. The mitochondrial permeability transition pore and its role in cell death. Biochem. J. 1999, 341, 233–249. [Google Scholar] [CrossRef]
- Borutaite, V.; Brown, G.C. Mitochondrial regulation of caspase activation by cytochrome c oxidase and tetramethylenephenylenediamine via cytosolic cytochrome c redox state. J. Biol. Chem. 2007, 282, 31124–31130. [Google Scholar] [CrossRef]
- Raychaudhuri, S.; Skommer, J.; Henty, K.; Birch, N.; Brittain, T. Neuroglobin protects nerve cells from apoptosis by inhibiting the intrinsic pathway of cell death. Apoptosis 2010, 15, 401–411. [Google Scholar] [CrossRef]
- Skommer, J.; Brittain, T.; Raychaudhuri, S. Bcl-2 inhibits apoptosis by modulating intrinsic cell-to-cell variations in the mitochondrial pathway of cell death. Apoptosis 2012, 15, 1223–1233. [Google Scholar]
- Skommer, J.; Brittain, T. Extended survival of SH-SY5Y cells following over-expression of Lys67Glu neuroglobin is associated with stabilization of ΔψM. Cytometry 2012, 81, 602–610. [Google Scholar]
- Boehning, D.; Patterson, R.C.; Sedaghat, L.; Glebova, N.O.; Kurosaki, T.; Snyder, S.H. Cytochrome c binds to inositol (1,4,5) trisphosphate receptors, amplifying calcium dependent apoptosis. Nat. Cell Biol. 2003, 5, 1051–1061. [Google Scholar] [CrossRef]
- Pinton, P.; Giorgi, G.; Siviero, R.; Zuchini, E.; Rizutto, R. Calcium and apoptosis: ER-mitochondrial Ca2+ transfer in the control of apoptosis. Oncogene 2008, 27, 6407–6418. [Google Scholar] [CrossRef]
- Duong, T.T.H.; Witting, P.K.; Antao, S.T.; Parry, S.N.; Kennerson, M.; Lai, B.; Vogt, S.; Lay, P.A.; Harris, H.M. Multiple protective activities of neuroglobin in cultured neuronal cells exposed to hypoxia re-oxygenation injury. J. Neurochem. 2009, 108, 1143–1154. [Google Scholar] [CrossRef]
- Liu, J.; Yu, Z.; Guo, S.; Lee, S.; Xing, C.; Zhang, C.; Gao, Y.; Nicholls, D.G.; Lo, E.H.; Wang, X. Effects of neuroglobin overexpression on mitochondrial function and oxidative stress following hypoxia/reoxygenation in cultured neurons. J. Neurosci. Res. 2009, 87, 164–170. [Google Scholar] [CrossRef]
- Antao, S.T.; Duong, T.T.; Aran, R.; Witting, P.K. Neuroglobin over-expression in cultured human neuronal cells protects against hydrogen peroxide insult via activating phosphoinositide-3 kinase and opening the mitochondrial K-ATP channel. Antioxid. Redox. Signal. 2010, 13, 769–781. [Google Scholar] [CrossRef]
- Brittain, T.; Skommer, J. Does a redox cycle provide a mechanism for setting the capacity of neuroglobin to protect cells from apoptosis? IUBMB Life 2012, 64, 419–422. [Google Scholar] [CrossRef]
- Skommer, J.; Helbo, S.; Henty, K.; Brittain, T. Ligand binding, reactivity and biological activity of a distal pocket mutant of neuroglobin. Int. J. Biol. Macromol. 2012, 51, 284–290. [Google Scholar] [CrossRef]
- Mendoza, V.; Klein, D.; Ichii, H.; Ribeiro, M.M.; Ricordi, C.; Hankeln, T.; Burmester, T.; Pastori, R.L. Protection of islets in culture by delivery of oxygen binding neuroglobin via protein transduction. Transplant Proc. 2005, 37, 237–240. [Google Scholar] [CrossRef]
- Zhou, G.Y.; Zhou, S.N.; Lou, Z.Y.; Zhu, C.S.; Zheng, X.P.; Hu, X.Q. Translocation and neuroprotective properties of transactivator-of-transcription protein-transduction domain-neuroglobin fusion protein in primary cultured cortical neurons. Biotechnol. Appl. Biochem. 2008, 49, 25–33. [Google Scholar] [CrossRef]
- Peroni, D.; Negro, A.; Bahr, M.; Dietz, G.P. Intracellular delivery of Neuroglobin using HIV-1 TAT protein transduction domain fails to protect against oxygen and glucose deprivation. Neurosci. Lett. 2007, 421, 110–114. [Google Scholar] [CrossRef]
- Cai, B.; Lin, Y.; Xue, X.H.; Fang, L.; Wang, N.; Wu, Z.Y. TAT-mediated delivery of neuroglobin protects against focal cerebral ischemia in mice. Expt. Neurol. 2011, 227, 224–231. [Google Scholar]
- Dietz, G.P. Protection by neuroglobin and cell-penetrating peptide-mediated deliveryin vivo: A decade of research. Exp. Neurol. 2011, 227, 224–231. [Google Scholar] [CrossRef]
- Watanabe, S.; Wakasugi, K. Zebrafish neuroglobin is a cell membrane-penetrating globin. Biochemistry 2008, 47, 5266–5270. [Google Scholar]
- Watanabe, S.; Wakasugi, K. Identification of residues critical for the cell-membrane-penetrating activity of zebrafish neuroglobin. FEBS Lett. 2010, 584, 2467–2472. [Google Scholar] [CrossRef]
- Chen, X.W.; Lin, W.P.; Lin, J.H.; Wu, C.Y.; Zhang, L.Q.; Huang, Z.D.; Lai, J.M. The protective effects of the lentivirus-mediated neuroglobin gene transfer on spinal cord injury in rabbits. Spinal. Cord. 2012, 50, 467–471. [Google Scholar] [CrossRef]
- Jin, K.; Mao, X.O.; Xie, L.; John, V.; Greenberg, D.A. Pharmacological induction of neuroglobin expression. Pharmacology 2011, 87, 81–84. [Google Scholar]
- De Marinis, E.; Ascenzi, P.; Pellegrini, M.; Galluzzo, P.; Bulzomi, P.; Angeles, A.M.; Miguel Garcia-Segura, L.; Marino, M. 17 beta-Estradiol—A new modulator of neuroglobin levels in neurons: Role in neuroprotection against H2O2-Induced toxicity. Neurosignals 2010, 18, 20223–20235. [Google Scholar]
- Gillies, G.E.; McArthur, S. Estrogen actions in the brain and the basis for differential action in men and women: A case for sex-specific medicines. Pharmacol. Rev. 2010, 62, 155–198. [Google Scholar] [CrossRef]
- Henderson, B.E.; Feigelson, H.S. Hormonal carcinogenesis. Carcinogenesis 2000, 21, 427–433. [Google Scholar] [CrossRef]
- Key, T.J. Serum oestradiol and breast cancer risk. Endocr. Relat. Cancer 1999, 6, 175–180. [Google Scholar] [CrossRef]
- Yu, Z.; Liu, N.; Liu, J.; Yang, K.; Wang, X. Neuroglobin, a Novel Target for Endogenous Neuroprotection against Stroke and Neurodegenerative Disorders. Int. J. Mol. Sci. 2012, 13, 6995–7014. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Brittain, T. The Anti-Apoptotic Role of Neuroglobin. Cells 2012, 1, 1133-1155. https://doi.org/10.3390/cells1041133
Brittain T. The Anti-Apoptotic Role of Neuroglobin. Cells. 2012; 1(4):1133-1155. https://doi.org/10.3390/cells1041133
Chicago/Turabian StyleBrittain, Thomas. 2012. "The Anti-Apoptotic Role of Neuroglobin" Cells 1, no. 4: 1133-1155. https://doi.org/10.3390/cells1041133
APA StyleBrittain, T. (2012). The Anti-Apoptotic Role of Neuroglobin. Cells, 1(4), 1133-1155. https://doi.org/10.3390/cells1041133