Effect of Sulphur Application on Photosynthesis and Biomass Accumulation of Sesame Varieties under Rainfed Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Research Site and Planting Material
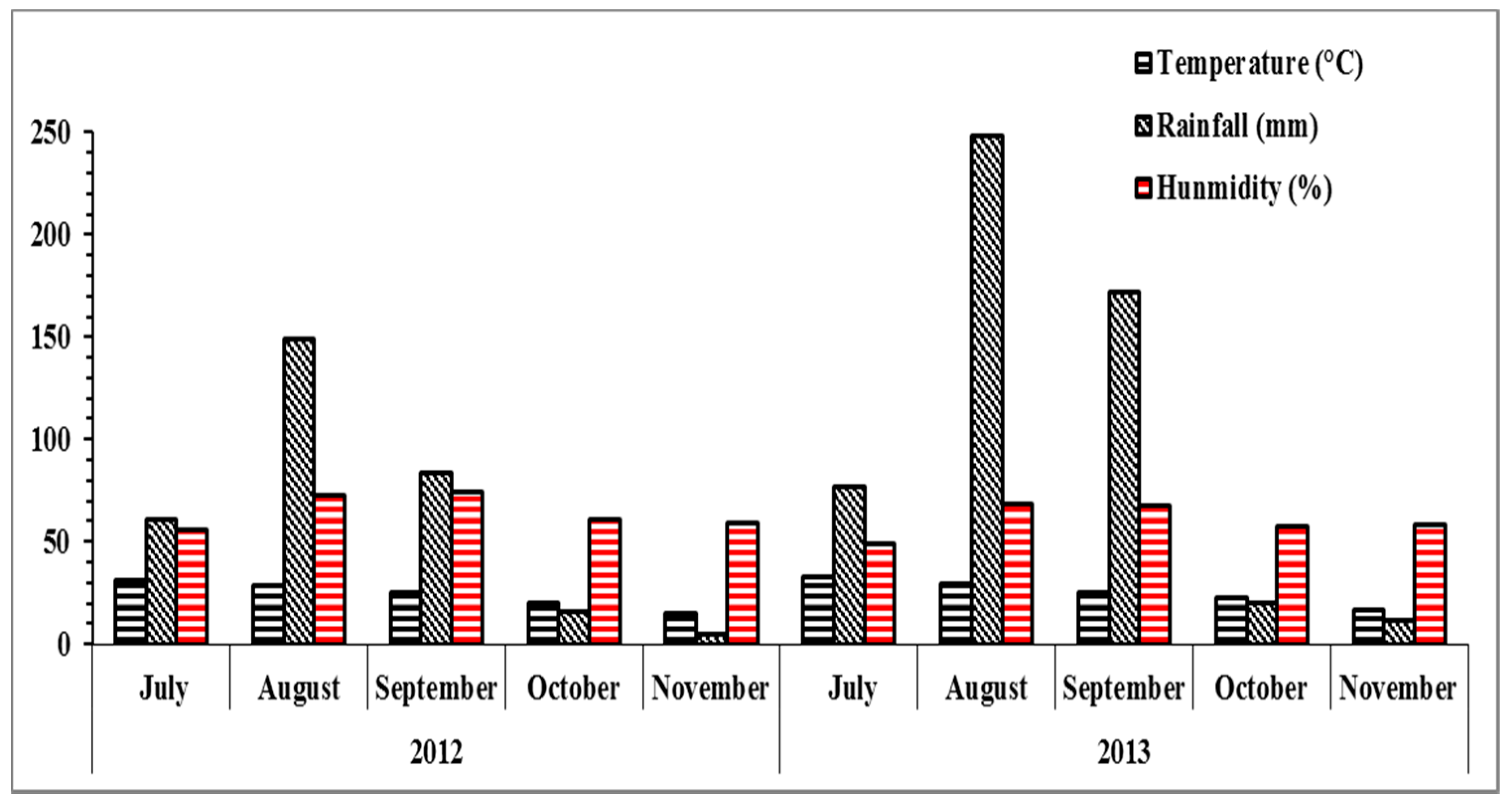
2.3. Weather Description and Soil Characteristics
2.4. Experimental Details and Treatments
2.5. Measurements and Sampling
2.5.1. Changes in Sesame Leaf Area Index
2.5.2. Photosynthetic Rate
2.5.3. Biomass Sampling and Seed Yield
2.5.4. Sulphur Uptake and Distribution
2.5.5. Economic Analysis
2.5.6. Statistical Analysis
3. Results
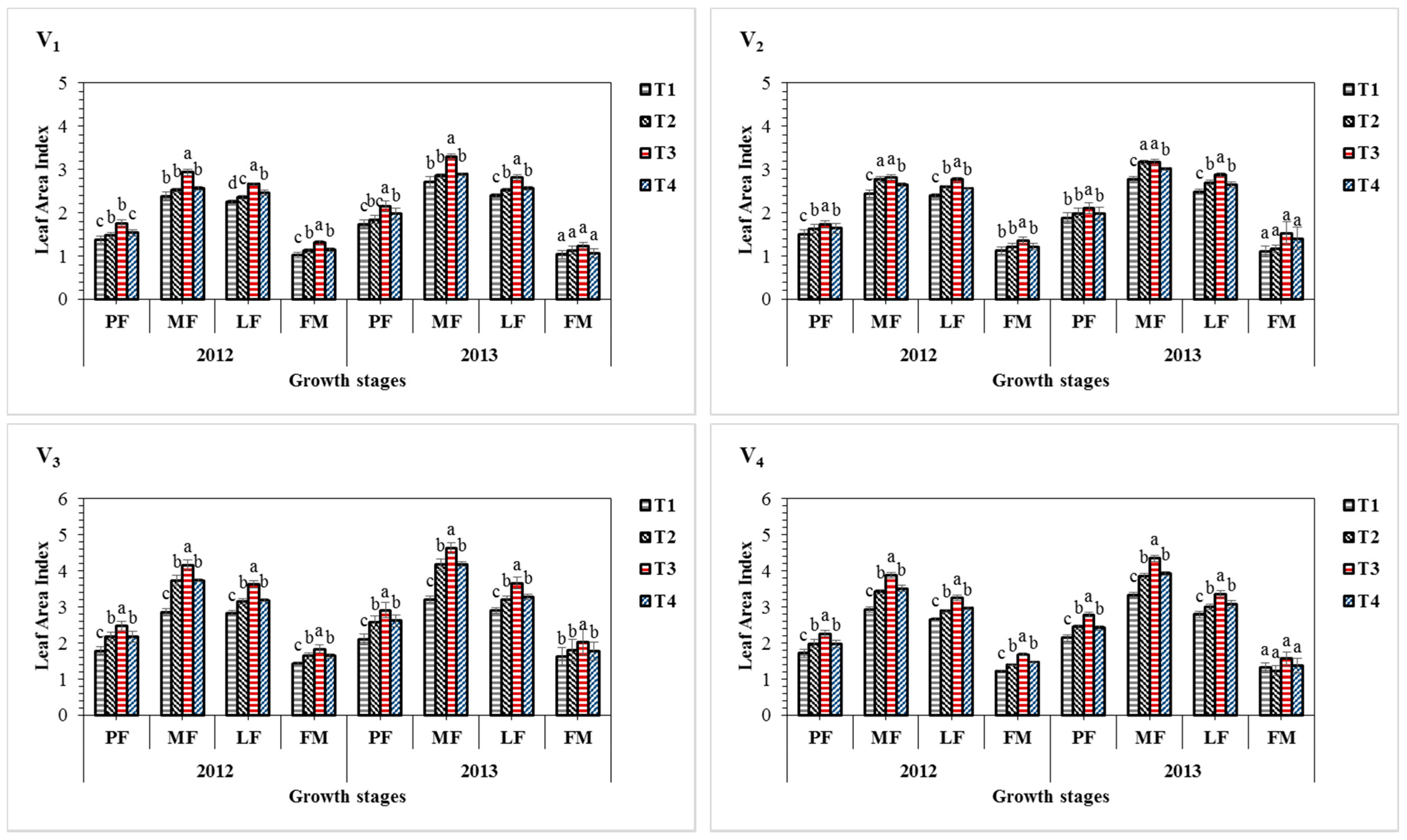
3.1. Effect of Sulphur Treatments on Leaf Area Index of Sesame
3.2. Effect of Sulphur Treatments on Photosynthetic Rate of Sesame
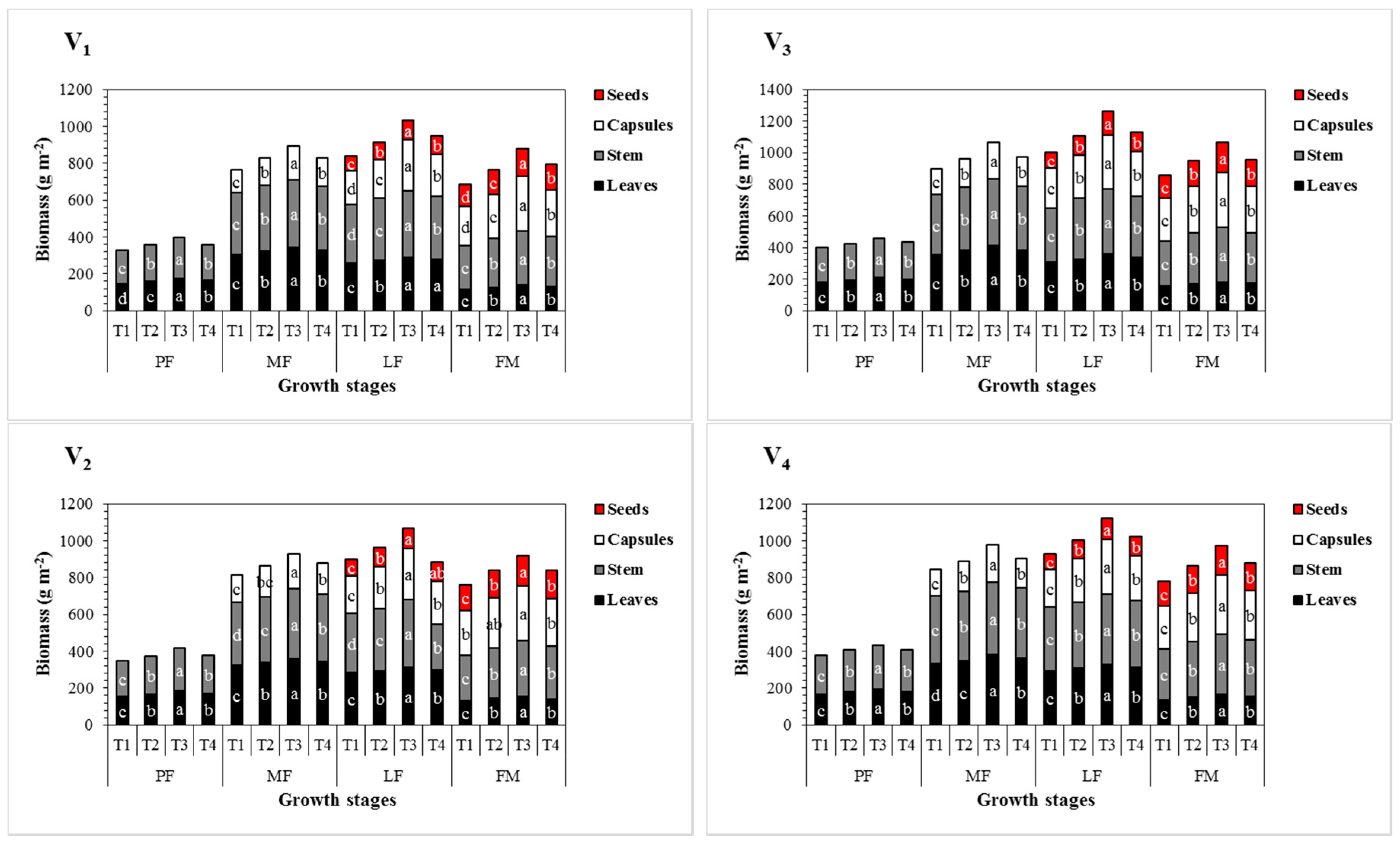
3.3. Effect of Sulphur Treatments on Biomass Accumulation and Seed Yield of Sesame
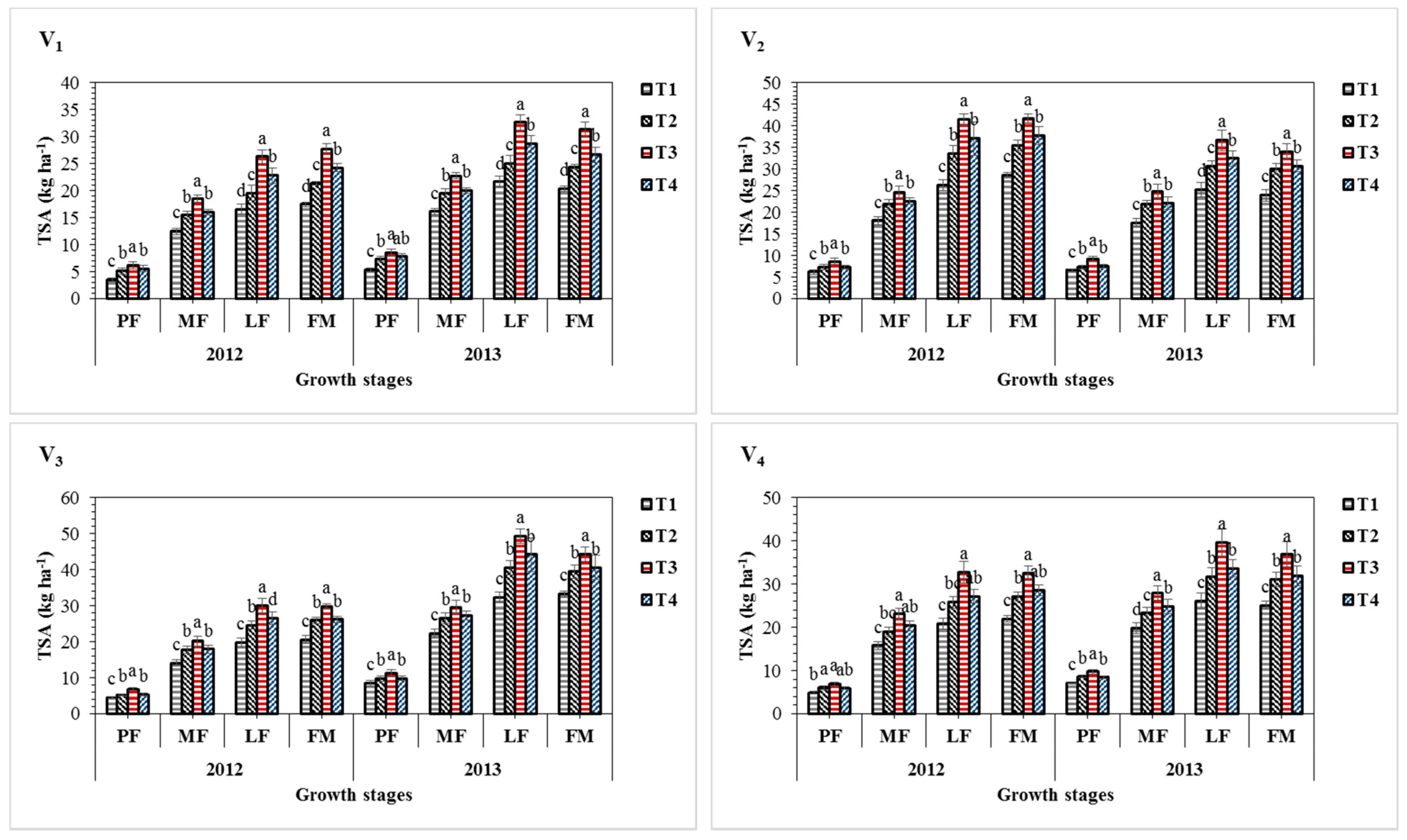
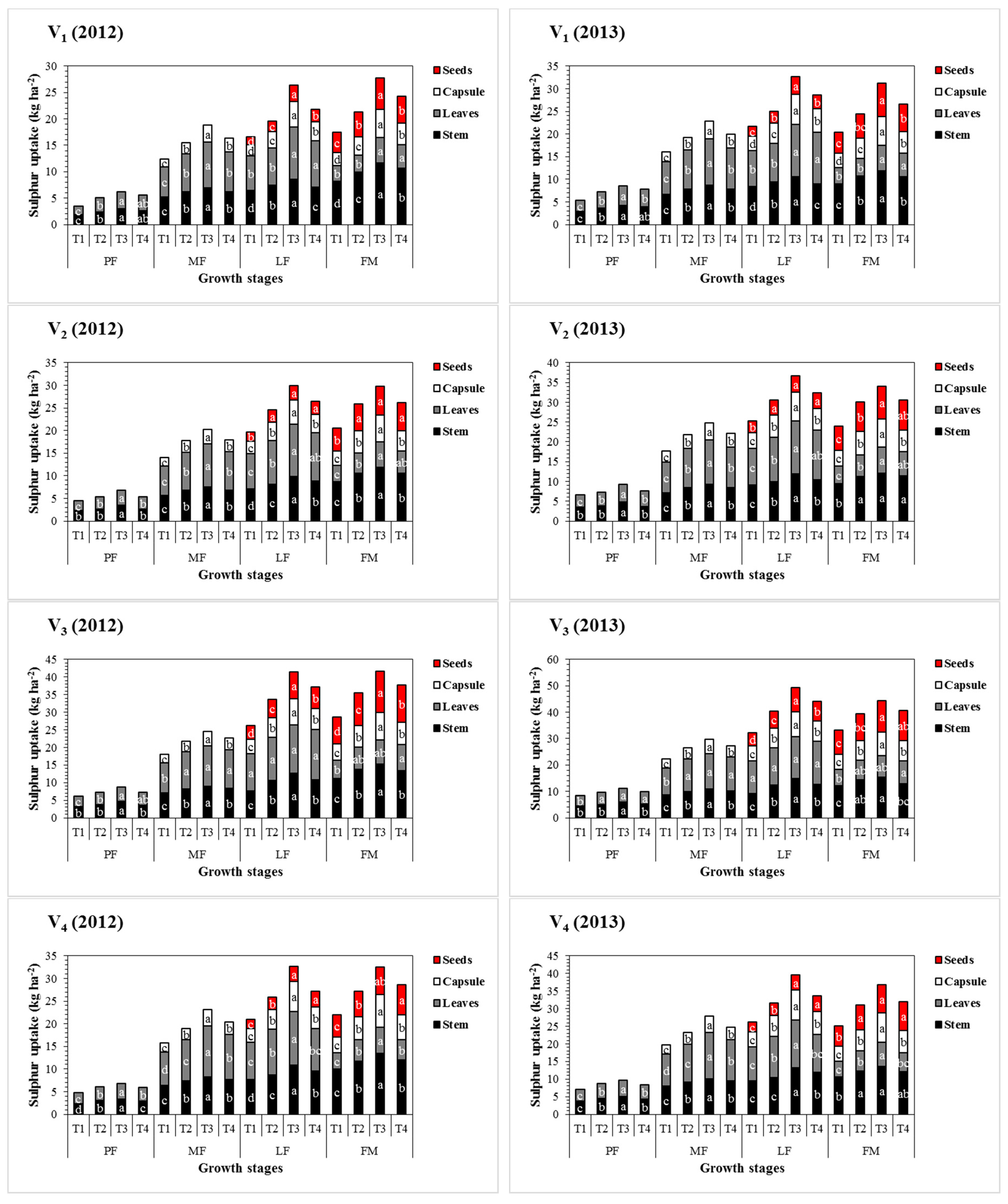
3.4. Effect of Sulphur Treatments on Total Sulphur Accumulation and Distribution
3.5. Effect of Sulphur Treatments on Economic Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Scherer, H.W. Sulphur in crop production. Eur. J. Agron. 2001, 14, 81–111. [Google Scholar] [CrossRef]
- Raja, A.; Hattab, K.O.; Gurusamy, L.; Vembu, G.; Suganya, S. Sulphur application on growth and yield and quality of sesame varieties. Int. J. Agric. Res. 2007, 2, 599–606. [Google Scholar]
- Vaiyapuri, V.; Amudha, A.; Sriramachandrasekharan, M.; Imayavaramban, V. Effect of sulphur levels and organics on growth and yield of sesame. Adv. Plant Sci. 2004, 17, 681–685. [Google Scholar]
- Sarkar, R.; Saha, A. Analysis of growth and productivity of sesame (Sesamum indicum) in relation to nitrogen, sulphur and boron. Indian J. Plant Physiol. 2005, 10, 333. [Google Scholar]
- Raja, A.; Hattab, K.O.; Gurusamy, L.; Suganya, S. Sulphur levels on nutrient uptake and yield of sesame varieties and nutrient availability. Int. J. Soil Sci. 2007, 2, 278–285. [Google Scholar] [CrossRef]
- Shah, M.A.; Manaf, A.; Hussain, M.; Farooq, S.; Zafar-ul-Hye, M. Sulphur fertilization improves the sesame productivity and economic returns under rainfed conditions. Int. J. Agric. Biol. 2013, 15, 1301–1306. [Google Scholar]
- Raza, M.A.; Feng, L.Y.; Manaf, A.; Wasaya, A.; Ansar, M.; Hussain, A.; Khalid, M.H.B.; Iqbal, N.; Xi, Z.J.; Chen, Y.K. Sulphur application increases seed yield and oil content in sesame seeds under rainfed conditions. Field Crop. Res. 2018, 218, 51–58. [Google Scholar] [CrossRef]
- Hawkesford, M.J. Plant responses to sulphur deficiency and the genetic manipulation of sulphate transporters to improve s-utilization efficiency. J. Exp. Bot. 2000, 51, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Dobermann, A. Rice: Nutrient Disorders & Nutrient Management; International Rice Research Institute: Los Baños, Laguna, Philippines, 2000; Volume 1. [Google Scholar]
- Girondé, A.; Dubousset, L.; Trouverie, J.; Etienne, P.; Avice, J.C. The impact of sulfate restriction on seed yield and quality of winter oilseed rape depends on the ability to remobilize sulfate from vegetative tissues to reproductive organs. Front. Plant Sci. 2014, 5, 695. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Yang, Y.; Deng, X.; Li, M.; Zhang, W.; Zhao, Z. Effects of sulfur and sulfate on selenium uptake and quality of seeds in rapeseed (Brassica napus L.) treated with selenite and selenate. Environ. Exp. Bot. 2017, 135, 13–20. [Google Scholar] [CrossRef]
- Zhao, F.; Evans, E.; Bilsborrow, P.; Syers, J. Sulphur uptake and distribution in double and single low varieties of oilseed rape (Brassica napus L.). Plant Soil 1993, 150, 69–76. [Google Scholar] [CrossRef]
- Syers, J.K.; Curtin, D.; Skinner, R.J. Soil and fertiliser sulphur in UK agriculture. Proceedings—The Fertiliser Society of London; 1987. Available online: http://agris.fao.org/agris-search/search.do?recordID=US201302689611 (accessed on 1 July 2018).
- Pathak, N.; Rai, A.K.; Kumari, R.; Thapa, A.; Bhat, K.V. Sesame crop: An underexploited oilseed holds tremendous potential for enhanced food value. Agric. Sci. 2014, 5, 519. [Google Scholar] [CrossRef]
- Wei, X.; Liu, K.; Zhang, Y.; Feng, Q.; Wang, L.; Zhao, Y.; Li, D.; Zhao, Q.; Zhu, X.; Zhu, X. Genetic discovery for oil production and quality in sesame. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Rangkadilok, N.; Pholphana, N.; Mahidol, C.; Wongyai, W.; Saengsooksree, K.; Nookabkaew, S.; Satayavivad, J. Variation of sesamin, sesamolin and tocopherols in sesame (Sesamum indicum L.) seeds and oil products in Thailand. Food Chem. 2010, 122, 724–730. [Google Scholar] [CrossRef]
- Jiang, D.; Yue, H.; Wollenweber, B.; Tan, W.; Mu, H.; Bo, Y.; Dai, T.; Jing, Q.; Cao, W. Effects of post-anthesis drought and waterlogging on accumulation of high-molecular-weight glutenin subunits and glutenin macropolymers content in wheat grain. J. Crop Sci. 2009, 195, 89–97. [Google Scholar] [CrossRef]
- Motzo, R.; Fois, S.; Giunta, F. Protein content and gluten quality of durum wheat (Triticum turgidum subsp. durum) as affected by sowing date. J. Sci. Food Agric. 2007, 87, 1480–1488. [Google Scholar] [CrossRef]
- Ciaffi, M.; Tozzi, L.; Borghi, B.; Corbellini, M.; Lafiandra, D. Effect of heat shock during grain filling on the gluten protein composition of bread wheat. J. Cereal Sci. 1996, 24, 91–100. [Google Scholar] [CrossRef]
- Thornton, P.K.; Ericksen, P.J.; Herrero, M.; Challinor, A.J. Climate variability and vulnerability to climate change: A review. Glob. Chang. Biol. 2014, 20, 3313–3328. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M. Response of spring wheat (Triticum aestivum L.) quality traits and yield to sowing date. PLoS ONE 2015, 10, e0126097. [Google Scholar] [CrossRef] [PubMed]
- Abuelsoud, W.; Hirschmann, F.; Papenbrock, J. Sulfur metabolism and drought stress tolerance in plants. In Drought Stress Tolerance in Plants; Springer: Cham, Switzerland, 2016; Volume 1, pp. 227–249. [Google Scholar]
- Ahmad, N.; Malagoli, M.; Wirtz, M.; Hell, R. Drought stress in maize causes differential acclimation responses of glutathione and sulfur metabolism in leaves and roots. BMC Plant Biol. 2016, 16, 247. [Google Scholar] [CrossRef] [PubMed]
- Girma, K.; Holtz, S.; Tubaña, B.; Solie, J.; Raun, W. Nitrogen accumulation in shoots as a function of growth stage of corn and winter wheat. J. Plant Nutr. 2010, 34, 165–182. [Google Scholar] [CrossRef]
- Bender, R.R.; Haegele, J.W.; Ruffo, M.L.; Below, F.E. Nutrient uptake, partitioning and remobilization in modern, transgenic insect-protected maize hybrids. Agron. J. 2013, 105, 161–170. [Google Scholar] [CrossRef]
- Ciampitti, I.A.; Vyn, T.J. Grain nitrogen source changes over time in maize: A review. Crop Sci. 2013, 53, 366–377. [Google Scholar] [CrossRef]
- McGrath, S.P.; Zhao, F.; Blake-Kalff, M. History and outlook for sulphur fertilizers in Europe. Fertili. Fertilization 2003, 2, 5–27. [Google Scholar]
- Schnug, E.; Haneklaus, S. Theoretical principles for the indirect determination of the total glucosinolate content in rapeseed and meal quantifying the sulphur concentration via x-ray fluorescence (x-rf method). J. Sci. Food Agric. 1988, 45, 243–254. [Google Scholar] [CrossRef]
- Schnug, E. Double low oilseed rape in west Germany; sulphur nutrition and glucosinolate levels. In Aspects of Applied Biology (United Kingdom); Food and Agriculture Organization of the United Nations: Rome, Italy, 1989. [Google Scholar]
- Langham, D.R. Phenology of sesame. In Issues in New Crops and New Uses; Janick, J., Whipkey, A., Eds.; ASHS (American Society for Horticultural Science) Press: Alexandria, VA, Egypt, 2007; pp. 144–182. [Google Scholar]
- Verma, B.C.; Swaminathan, K.; Sud, K. An improved turbidimetric procedure for the determination of sulphate in plants and soils. Talanta 1977, 24, 49–50. [Google Scholar] [CrossRef]
- CIMMYT Economics Program. From Agronomic Data to Farmer Recommendations: An Economics Training Manual; International Maize and Wheat Improvement Center: Edo Mex, Mexico, 1988. [Google Scholar]
- Wasaya, A.; Tahir, M.; Ali, H.; Hussain, M.; Yasir, T.A.; Sher, A.; Ijaz, M. Influence of varying tillage systems and nitrogen application on crop allometry, chlorophyll contents, biomass production and net returns of maize (Zea mays L.). Soil Till. Res. 2017, 170, 18–26. [Google Scholar] [CrossRef]
- Steel, R.G.; Torrie, J.H. Analysis of variance. 2. Multiway classifications. In Principles and Procedures of Statistics; McGraw-Hill Education: New York, NY, USA, 1960; p. 132. [Google Scholar]
- Jun, X.; Ling, G.; SHI, Z.G.; ZHAO, Y.S.; ZHANG, W.F. Effect of leaf removal on photosynthetically active radiation distribution in maize canopy and stalk strength. J. Integr. Agric. 2017, 16, 85–96. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, X.; Sun, H.; Ren, T.; Wang, Y. Effects of winter wheat row spacing on evapotranpsiration, grain yield and water use efficiency. Agric. Water Manag. 2010, 97, 1126–1132. [Google Scholar] [CrossRef]
- Rahman, T.; Liu, X.; Hussain, S.; Ahmed, S.; Chen, G.; Yang, F.; Chen, L.; Du, J.; Liu, W.; Yang, W. Water use efficiency and evapotranspiration in maize-soybean relay strip intercrop systems as affected by planting geometries. PLoS ONE 2017, 12, e0178332. [Google Scholar] [CrossRef] [PubMed]
- Begum, F.; Hossain, F.; Mondal, M.R.I. Influence of sulphur on morpho-physiological and yield parameters of rapeseed (Brassica campestris L.). Bangl. J. Agric. Res. 2013, 37, 645–652. [Google Scholar] [CrossRef]
- Ahmad, A.; Khan, I.; Anjum, N.; Diva, I.; Abdin, M.; Iqbal, M. Effect of timing of sulfur fertilizer application on growth and yield of rapeseed. J. Plant Nutr. 2005, 28, 1049–1059. [Google Scholar] [CrossRef]
- CHOPRA, R.K. Effects of temperature on the in vivo assay of nitrate reductase in some C3 and C4 species. Ann. Bot. 1983, 51, 617–620. [Google Scholar] [CrossRef]
- Liu, X.; Rahman, T.; Yang, F.; Song, C.; Yong, T.; Liu, J.; Zhang, C.; Yang, W. Par interception and utilization in different maize and soybean intercropping patterns. PLoS ONE 2017, 12, e0169218. [Google Scholar] [CrossRef] [PubMed]
- D’Hooghe, P.; Escamez, S.; Trouverie, J.; Avice, J.C. Sulphur limitation provokes physiological and leaf proteome changes in oilseed rape that lead to perturbation of sulphur, carbon and oxidative metabolisms. BMC Plant Biol. 2013, 13, 23. [Google Scholar] [CrossRef] [PubMed]
- ur Rehman, H.; Iqbal, Q.; Farooq, M.; Wahid, A.; Afzal, I.; Basra, S.M. Sulphur application improves the growth, seed yield and oil quality of canola. Acta Physiol. Plant. 2013, 35, 2999–3006. [Google Scholar] [CrossRef]
- Ahmad, A.; Abdin, M. Photosynthesis and its related physiological variables in the leaves of brassica genotypes as influenced by sulphur fertilization. Physiol. Plant. 2000, 110, 144–149. [Google Scholar] [CrossRef]
- Zhang, Z.; Sun, K.; Lu, A.; Zhang, X. Study on the effect of s fertilizer application on crops and the balance of s in soil. J. Agric. Sci. 1999, 5, 25–27. [Google Scholar]
- Liu, T.; Gu, L.; Dong, S.; Zhang, J.; Liu, P.; Zhao, B. Optimum leaf removal increases canopy apparent photosynthesis, 13 c-photosynthate distribution and grain yield of maize crops grown at high density. Field Crop. Res. 2015, 170, 32–39. [Google Scholar] [CrossRef]
- Parmar, S.; Buchner, P.; Hawkesford, M. Leaf developmental stage affects sulfate depletion and specific sulfate transporter expression during sulfur deprivation in Brassica napus L. Plant Biol. 2007, 9, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Blake-Kalff, M.M.; Harrison, K.R.; Hawkesford, M.J.; Zhao, F.J.; McGrath, S.P. Distribution of sulfur within oilseed rape leaves in response to sulfur deficiency during vegetative growth. Plant Physiol. 1998, 118, 1337–1344. [Google Scholar] [CrossRef] [PubMed]
- Couch, A.; Jani, A.; Mulvaney, M.; Hochmuth, G.; Bennett, J.; Gloaguen, R.; Langham, R.; Rowland, D. Nitrogen accumulation, partitioning and remobilization by diverse sesame cultivars in the humid southeastern USA. Field Crop. Res. 2017, 203, 55–64. [Google Scholar] [CrossRef]
- Zhao, F.; Salmon, S.; Withers, P.; Monaghan, J.; Evans, E.; Shewry, P.; McGrath, S. Variation in the breadmaking quality and rheological properties of wheat in relation to sulphur nutrition under field conditions. J. Cereal Sci. 1999, 30, 19–31. [Google Scholar] [CrossRef]
- Riekman, M.R. The Effect of Canola Cultivar on Water Extraction and Nitrogen and Sulphur Uptake. Master’s Thesis, University of Manitoba, Winnipeg, MB, Canada, October 2005. [Google Scholar]
- Flinn, A.; Pate, J. A quantitative study of carbon transfer from pod and subtending leaf to the ripening seeds of the field pea (Pisum arvense L.). J. Exp. Bot. 1970, 21, 71–82. [Google Scholar] [CrossRef]
- Andrews, A.; Svec, L. Photosynthetic activity of soybean pods at different growth stages compared to leaves. Can. J. Plant Sci. 1975, 55, 501–505. [Google Scholar] [CrossRef]
- Garg, B.; Burman, U.; Kathju, S. Influence of thiourea on photosynthesis, nitrogen metabolism and yield of clusterbean (Cyamopsis tetragonoloba (L.) taub.) under rainfed conditions of Indian arid zone. Plant Growth Regul. 2006, 48, 237–245. [Google Scholar] [CrossRef]
- Rotundo, J.L.; Borrás, L.; De Bruin, J.; Pedersen, P. Physiological strategies for seed number determination in soybean: Biomass accumulation, partitioning and seed set efficiency. Field Crop. Res. 2012, 135, 58–66. [Google Scholar] [CrossRef]
- Khan, M.B.; Khan, M.; Hussain, M.; Farooq, M.; Jabran, K.; Lee, D.J. Bio-economic assessment of different wheat-canola intercropping systems. Int. J. Agric. Biol. 2012, 14, 769–774. [Google Scholar]
| Serial No. | Physiological Stage | Growth Period | DAS 2012 * | DAS 2013 * |
|---|---|---|---|---|
| 1 | Seed germination | Vegetative | 04 | 03 |
| 2 | Juvenile | Vegetative | 23 | 21 |
| 3 | Pre-flowering | Vegetative | 35 | 35 |
| 4 | Initiation of flowering | Pre-reproductive | 37 | 36 |
| 5 | Mid-flowering | Reproductive | 65 | 65 |
| 6 | Late-flowering | Reproductive | 95 | 95 |
| 7 | Physiological maturity | Reproductive | 109 | 103 |
| 8 | Full-maturity | Reproductive | 125 | 125 |
| Years | Pre-Flowering | Mid-Flowering | Late-Flowering | ||
|---|---|---|---|---|---|
| 2012 | Varieties (V) | V1 | 20.4 c | 25.2 c | 19.7 c |
| V2 | 20.6 bc | 25.4 c | 20.5 b | ||
| V3 | 22.3 a | 26.9 b | 22.1 a | ||
| V4 | 21.6 ab | 26.0 b | 21.6 a | ||
| LSD (0.05) | 1.12 | 0.47 | 0.73 | ||
| Sulphur Treatments (T) | T1 | 20.2 d | 24.9 c | 20.1 d | |
| T2 | 21.1 c | 25.8 b | 20.8 c | ||
| T3 | 22.2 a | 27.0 a | 22.0 a | ||
| T4 | 21.6 b | 25.9 b | 21.1 b | ||
| LSD (0.05) | 0.39 | 0.22 | 0.28 | ||
| Interaction (V × T) | NS | * | NS | ||
| 2013 | Varieties (V) | V1 | 21.1 c | 26.2 c | 20.5 c |
| V2 | 21.2 bc | 26.5 bc | 21.3 b | ||
| V3 | 23.0 a | 27.9 a | 22.9 a | ||
| V4 | 22.4 ab | 26.9 b | 22.4 | ||
| LSD (0.05) | 1.11 | 0.54 | 0.73 | ||
| Sulphur Treatments (T) | T1 | 20.9 d | 25.9 c | 20.8 d | |
| T2 | 21.7 c | 26.7 b | 21.6 c | ||
| T3 | 22.8 a | 28.0 a | 22.8 a | ||
| T4 | 22.3 | 26.9 b | 21.9 b | ||
| LSD (0.05) | 0.38 | 0.23 | 0.29 | ||
| Interaction (V × T) | NS | NS | NS | ||
| Years | Total Biomass Accumulation (g m–2) | Grain Yield | |||||
|---|---|---|---|---|---|---|---|
| PF | MF | LF | FM | (g m–2) | |||
| 2012 | Varieties (V) | V1 | 335.5 d | 785.3 d | 883.5 d | 648.4 b | 128.5 d |
| V2 | 355.2 c | 827.6 c | 927.1 c | 656.0 b | 138.0 b | ||
| V3 | 404.0 a | 931.3 a | 1071.5 a | 760.1 a | 162.1 a | ||
| V4 | 379.5 b | 858.6 b | 969.1 b | 697.6 b | 133.8 c | ||
| LSD (0.05) | 9.97 | 16.25 | 12.91 | 54.42 | 2.88 | ||
| Sulphur Treatments (T) | T1 | 339.4 c | 786.4 d | 866.9 d | 612.4 c | 126.0 d | |
| T2 | 366.3 b | 840.6 c | 945.1 c | 684.5 b | 140.2 c | ||
| T3 | 400.9 a | 922.1 a | 1070.3 a | 771.2 a | 152.9 a | ||
| T4 | 368.4 b | 853.6 b | 969.1 b | 694.1 b | 143.1 b | ||
| LSD (0.05) | 4.30 | 8.14 | 8.70 | 12.3 | 2.57 | ||
| Interaction (V × T) | * | * | * | NS | * | ||
| 2013 | Varieties (V) | V1 | 384.2 d | 872.6 d | 982.2 d | 815.7 c | 144.3 c |
| V2 | 404.0 c | 913.5 c | 1028.2 c | 881.8 b | 164.3 ab | ||
| V3 | 454.6 a | 1017.1 a | 1174.2 a | 992.7 a | 170.6 a | ||
| V4 | 432.6 b | 945.7 b | 1068.4 b | 912.0 b | 159.8 b | ||
| LSD (0.05) | 12.58 | 16.3 | 15.94 | 37.8 | 10.08 | ||
| Sulphur Treatments (T) | T1 | 389.9 c | 872.5 d | 967.3 d | 807.4 c | 142.1 c | |
| T2 | 415.0 b | 928.0 c | 1045.4 c | 893.6 b | 159.0 b | ||
| T3 | 450.0 a | 1009.6 a | 1170.9 a | 998.5 a | 175.7 a | ||
| T4 | 418.6 b | 938.9 b | 1069.3 b | 903.7 b | 162.2 b | ||
| LSD (0.05) | 4.70 | 7.97 | 8.44 | 13.63 | 5.38 | ||
| Interaction (V × T) | * | * | NS | NS | NS | ||
| Treatments | Total Expenses | Gross Income | Net Income | Benefit-Cost Ratio | ||||
|---|---|---|---|---|---|---|---|---|
| 2012 | 2013 | 2012 | 2013 | 2012 | 2013 | 2012 | 2013 | |
| V1 | 405.1 | 402.9 | 1007.8 | 1179.4 | 602.7 | 776.5 | 2.5 | 2.9 |
| V2 | 407.5 | 405.3 | 1082.4 | 1342.8 | 674.8 | 937.5 | 2.7 | 3.3 |
| V3 | 410.0 | 407.7 | 1271.4 | 1394.3 | 861.4 | 986.6 | 3.1 | 3.4 |
| V4 | 412.5 | 410.1 | 1049.4 | 1306.1 | 637.0 | 896.0 | 2.5 | 3.2 |
| T1 | 405.1 | 402.9 | 988.4 | 1161.4 | 583.3 | 758.5 | 2.4 | 2.9 |
| T2 | 407.5 | 405.3 | 1100.5 | 1299.5 | 692.9 | 894.2 | 2.7 | 3.2 |
| T3 | 410.0 | 407.7 | 1199.9 | 1436.0 | 789.9 | 1028.3 | 2.9 | 3.5 |
| T4 | 412.5 | 410.1 | 1122.6 | 1325.7 | 710.1 | 915.6 | 2.7 | 3.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raza, M.A.; Feng, L.Y.; Iqbal, N.; Manaf, A.; Khalid, M.H.B.; Ur Rehman, S.; Wasaya, A.; Ansar, M.; Billah, M.; Yang, F.; et al. Effect of Sulphur Application on Photosynthesis and Biomass Accumulation of Sesame Varieties under Rainfed Conditions. Agronomy 2018, 8, 149. https://doi.org/10.3390/agronomy8080149
Raza MA, Feng LY, Iqbal N, Manaf A, Khalid MHB, Ur Rehman S, Wasaya A, Ansar M, Billah M, Yang F, et al. Effect of Sulphur Application on Photosynthesis and Biomass Accumulation of Sesame Varieties under Rainfed Conditions. Agronomy. 2018; 8(8):149. https://doi.org/10.3390/agronomy8080149
Chicago/Turabian StyleRaza, Muhammad Ali, Ling Yang Feng, Nasir Iqbal, Abdul Manaf, Muhammad Hayder Bin Khalid, Sana Ur Rehman, Allah Wasaya, Muhammad Ansar, Masum Billah, Feng Yang, and et al. 2018. "Effect of Sulphur Application on Photosynthesis and Biomass Accumulation of Sesame Varieties under Rainfed Conditions" Agronomy 8, no. 8: 149. https://doi.org/10.3390/agronomy8080149







