Responses of Different Panicum miliaceum L. Genotypes to Saline and Water Stress in a Marginal Mediterranean Environment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Laboratory Experiment
2.1.1. Seed Water Uptake
2.1.2. Seed Germination
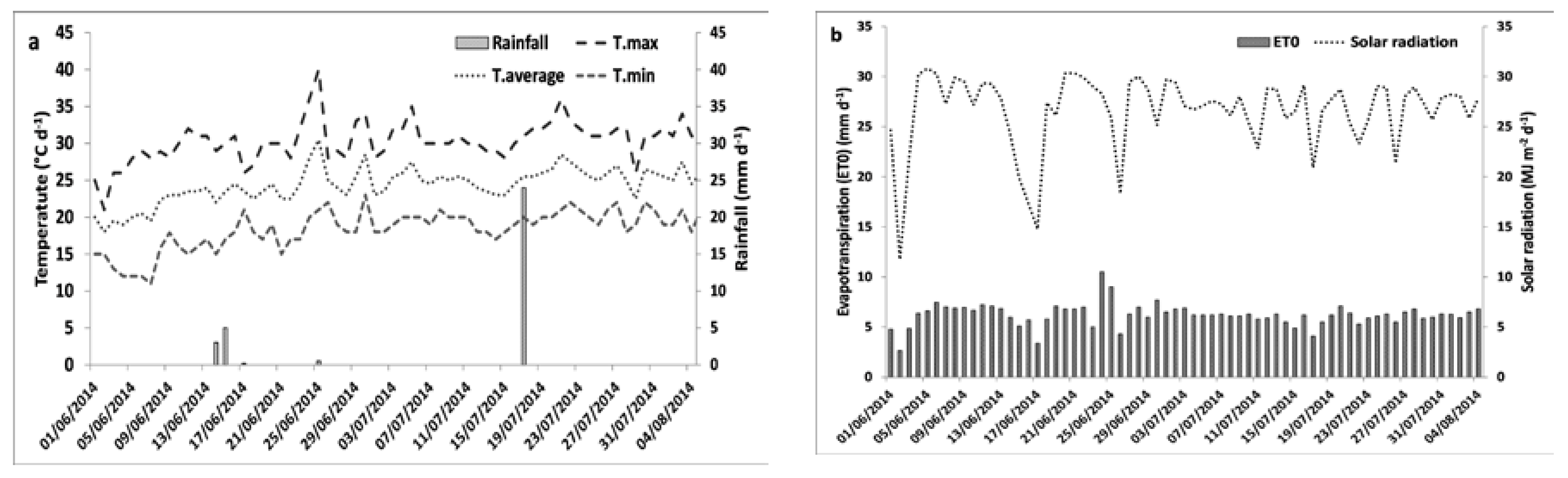
2.2. Field Experiment
2.3. Statistical Analysis
3. Results
3.1. Laboratory Experiment
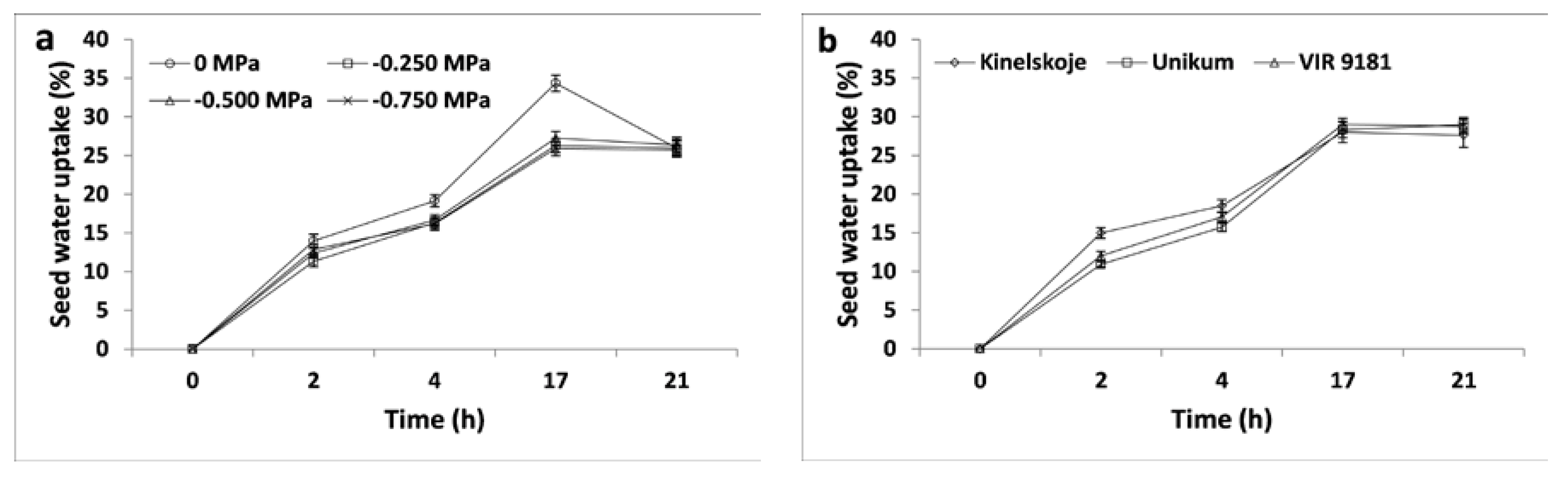
3.1.1. Seed Water Uptake
3.1.2. Seed Germination
3.2. Field Experiment
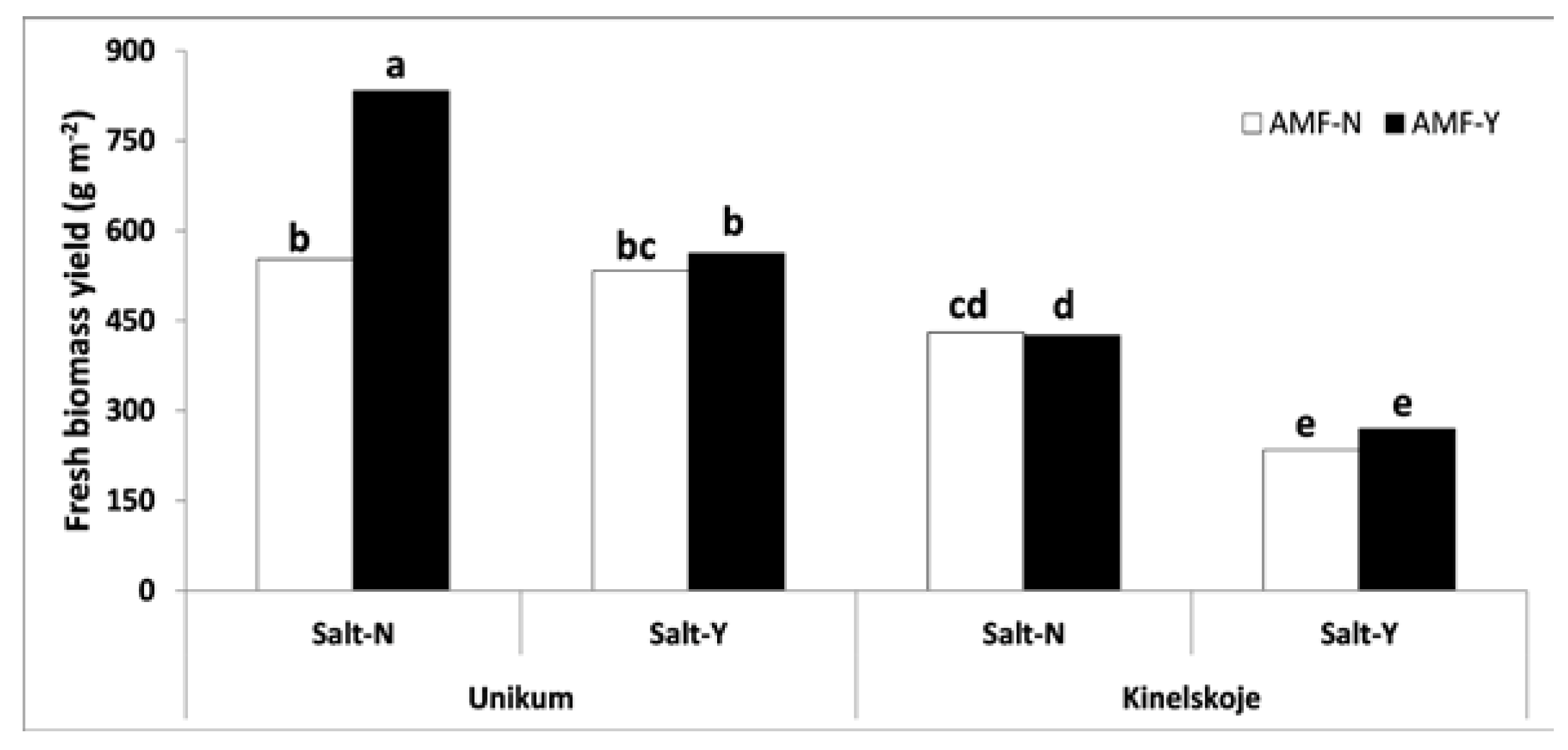
3.2.1. Fresh Biomass Yield (FBY)
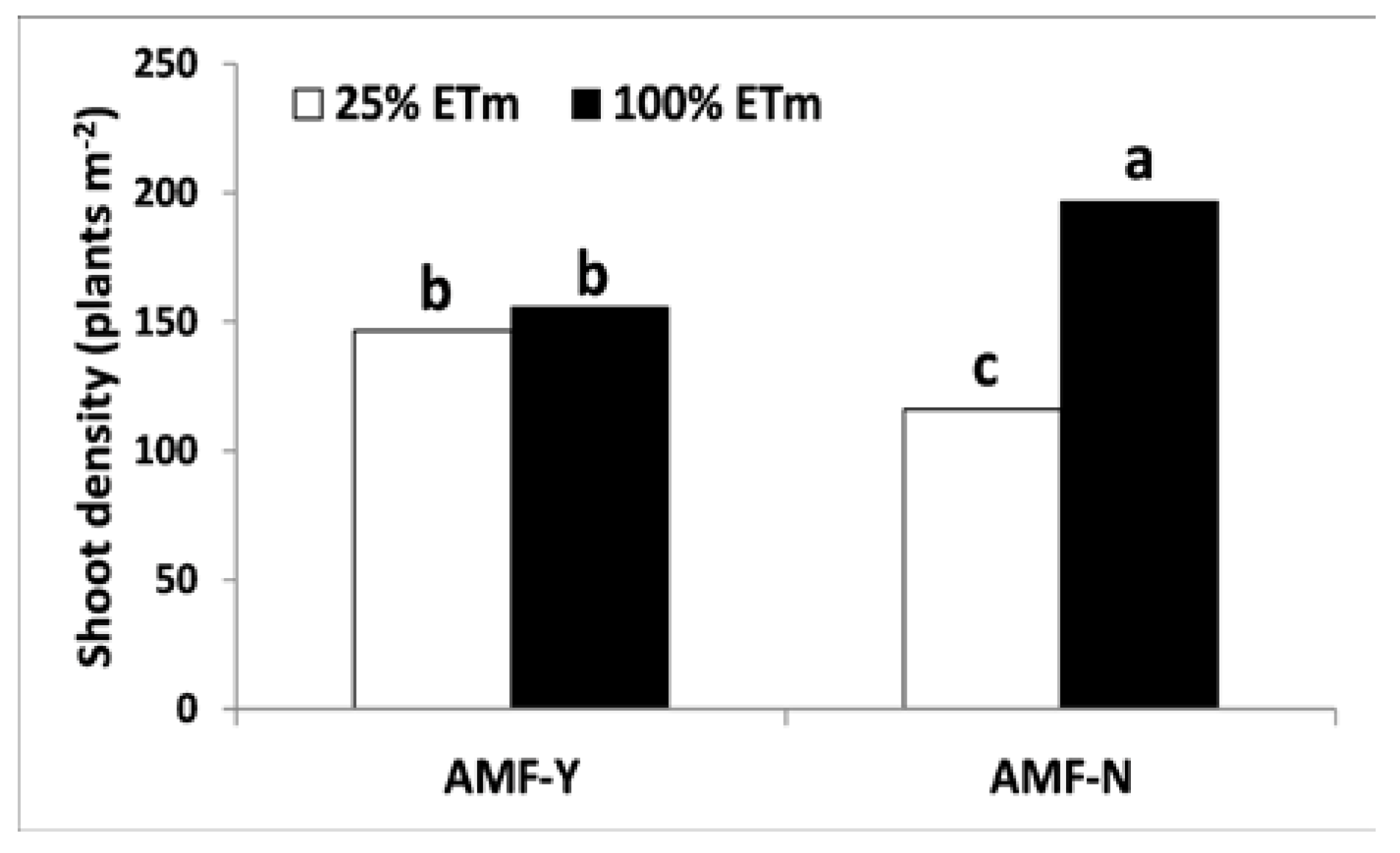
3.2.2. Shoot Density
3.2.3. Culm Height and Fresh Culm Weight
3.2.4. Panicle Length and Fresh Panicle Weight
3.2.5. Leaf Number and Leaf Fresh Weight
3.2.6. Leaf Chlorophyll Content (SPAD index)
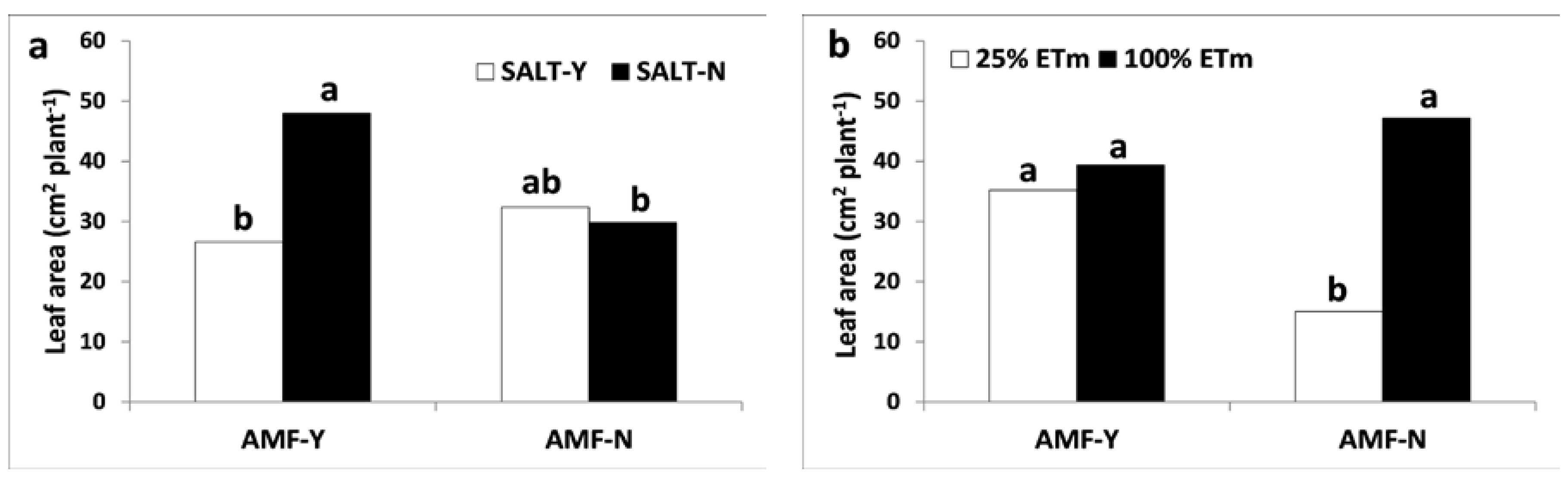
3.2.7. Leaf Area Surface
4. Discussion
4.1. Laboratory Experiment
4.2. Field Experiment
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hasegawa, P.M.; Bressan, R.A.; Zhu, J.K.; Bohnert, H.J. Plant cellular and molecular responses to high salinity. Annu. Rev. Plant Biol. 2000, 51, 463–499. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.K. Regulation of ion homeostasis under salt stress. Curr. Opin. Plant Biol. 2003, 6, 441–445. [Google Scholar] [CrossRef]
- Mahajan, S.; Tuteja, N. Cold, salinity and drought stresses: An overview. Arch. Biochem. Biophys. 2005, 444, 139–158. [Google Scholar] [CrossRef] [PubMed]
- Villa-Castorena, M.; Ulery, A.L.; Catalán-Valencia, E.A.; Remmenga, M.D. Salinity and nitrogen rate effects on the growth and yield of chile pepper plants. Soil Sci. Soc. Am. J. 2003, 67, 1781–1789. [Google Scholar] [CrossRef]
- Al-Karaki, G.N. Nursery inoculation of tomato with arbuscular mycorrhizal fungi and subsequent performance under irrigation with saline water. Sci. Hortic. 2006, 109, 1–7. [Google Scholar] [CrossRef]
- Jones, A.; Panagos, P.; Barcelo, S.; Bouraoui, F.; Bosco, C.; Dewitte, O.; Gardi, C.; Hervás, J.; Hiederer, R.; Jeffery, S.; et al. The State of Soil in Europe—A Contribution of the JRC to the European Environment Agency’s Environment State and Outlook Report—SOER 2010; Publications Office of the European Union: Luxembourg, 2012. [Google Scholar]
- Juniper, S.; Abbott, L. Vesicular-arbuscular mycorrhizas and soil salinity. Mycorrhiza 1993, 4, 45–57. [Google Scholar] [CrossRef]
- Ramoliya, P.J.; Patel, H.M.; Pandey, A.N. Effect of salinization of soil on growth and macro-and micro-nutrient accumulation in seedlings of Salvadora persica (Salvadoraceae). For. Ecol. Manag. 2004, 202, 181–193. [Google Scholar] [CrossRef]
- Aggarwal, A.; Kadian, N.; Neetu, K.; Tanwar, A.; Gupta, K.K. Arbuscular mycorrhizal symbiosis and alleviation of salinity stress. J. Appl. Nat. Sci. 2012, 4, 144–155. [Google Scholar]
- Adiku, S.G.K.; Renger, M.; Wessolek, G.; Facklam, M.; Hecht-Bucholtz, C. Simulation of the dry matter production and seed yield of common beans under varying soil water and salinity conditions. Agric. Water Manag. 2001, 47, 55–68. [Google Scholar] [CrossRef]
- Marschner, H. Mineral Nutrition of Higher Plants, 2nd ed.; Academic Press: Cambridge, MA, USA, 1995. [Google Scholar]
- Ipsilantis, I.; Karpouzas, D.G.; Papadopoulou, K.K.; Ehaliotis, C. Effects of soil application of olive mill wastewaters on the structure and function of the community of arbuscular mycorrhizal fungi. Soil Biol. Biochem. 2009, 41, 2466–2476. [Google Scholar] [CrossRef]
- Candido, V.; Campanelli, G.; D’Addabbo, T.; Castronuovo, D.; Renco, M.; Camele, I. Growth and yield promoting effect of artificial mycorrhization combined with different fertiliser rates on field-grown tomato. Ital. J. Agron. 2013, 8, 168–174. [Google Scholar] [CrossRef]
- Sabia, E.; Claps, S.; Morone, G.; Bruno, A.; Sepe, L.; Aleandri, R. Field inoculation of arbuscular mycorrhiza on maize (Zea mays L.) under low inputs: Preliminary study on quantitative and qualitative aspects. Ital. J. Agron. 2015, 10, 30–33. [Google Scholar] [CrossRef]
- Tarraf, W.; Ruta, C.; De Cillis, F.; Tagarelli, A.; Tedone, L.; De Mastro, G. Effects of mycorrhiza on growth and essential oil production in selected aromatic plants. Ital. J. Agron. 2015, 10, 160–162. [Google Scholar] [CrossRef]
- Puia, C.; Vidican, R.; Szabó, G.; Stoian, V. Potential of biofertilisers to improve performance of local genotype tomatoes. Ital. J. Agron. 2017, 12, 192–200. [Google Scholar] [CrossRef]
- Ruiz-Lozano, J.M. Arbuscular mycorrhizal symbiosis and alleviation of osmotic stress. New perspectives for molecular studies. Mycorrhiza 2003, 13, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Evelin, H.; Kapoor, R.; Giri, B. Arbuscular mycorrhizal fungi in alleviation of salt stress: A review. Ann. Bot. 2009, 104, 1263–1280. [Google Scholar] [CrossRef] [PubMed]
- Miransari, M. Contribution of arbuscular mycorrhizal symbiosis to plant growth under different types of soil stress. Plant Biol. 2010, 12, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Porcel, R.; Aroca, R.; Ruiz-Lozano, J.M. Salinity stress alleviation using arbuscular mycorrhizal fungi. A review. Agron. Sustain. Dev. 2012, 32, 181–200. [Google Scholar] [CrossRef]
- Langeroodi, A.R.S.; Ghooshchi, F.; Dadgar, T. Alleviatory activities in mycorrhizal tobacco plants subjected to increasing chloride in irrigation water. Ital. J. Agron. 2017. [Google Scholar] [CrossRef]
- Wu, Q.S.; Zou, Y.N.; He, X.H. Contributions of arbuscular mycorrhizal fungi to growth, photosynthesis, root morphology and ionic balance of citrus seedlings under salt stress. Acta Physiol. Plant. 2010, 32, 297–304. [Google Scholar] [CrossRef]
- Al-Karaki, G.N. Growth of mycorrhizal tomato and mineral acquisition under salt stress. Mycorrhiza 2000, 10, 51–54. [Google Scholar] [CrossRef]
- Al-Karaki, G.N.; Hammad, R.; Rusan, M. Response of two tomato cultivars differing in salt tolerance to inoculation with mycorrhizal fungi under salt stress. Mycorrhiza 2001, 11, 43–47. [Google Scholar] [CrossRef]
- Asghari, H.R.; Marschner, P.; Smith, S.E.; Smith, F.A. Growth response of Atriplex nummularia to inoculation with arbuscular mycorrhizal fungi at different salinity levels. Plant Soil 2005, 273, 245–256. [Google Scholar] [CrossRef]
- Feng, G.; Zhang, F.; Li, X.; Tian, C.; Tang, C.; Rengel, Z. Improved tolerance of maize plants to salt stress by arbuscular mycorrhiza is related to higher accumulation of soluble sugars in roots. Mycorrhiza 2002, 12, 185–190. [Google Scholar] [PubMed]
- Giri, B.; Kapoor, R.; Mukerji, K.G. Improved tolerance of Acacia nilotica to salt stress by arbuscular mycorrhiza, Glomus fasciculatum may be partly related to elevated K/Na ratios in root and shoot tissues. Microb. Ecol. 2007, 54, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Asghari, H.R. Vesicular-arbuscular (VA) mycorrhizae improve salinity tolerance in pre-inoculation subterranean clover (Trifolium subterraneum) seedlings. Int. J. Plant Prod. 2012, 2, 243–256. [Google Scholar]
- He, Z.; He, C.; Zhang, Z.; Zou, Z.; Wang, H. Changes of antioxidative enzymes and cell membrane osmosis in tomato colonized by arbuscular mycorrhizae under NaCl stress. Colloids Surf. B. 2007, 59, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Tian, C.Y.; Feng, G.; Li, X.L.; Zhang, F.S. Different effects of arbuscular mycorrhizal fungal isolates from saline or non-saline soil on salinity tolerance of plants. Appl. Soil Ecol. 2004, 26, 143–148. [Google Scholar] [CrossRef]
- Sheng, M.; Tang, M.; Chen, H.; Yang, B.; Zhang, F.; Huang, Y. Influence of arbuscular mycorrhizae on photosynthesis and water status of maize plants under salt stress. Mycorrhiza 2008, 18, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Juniper, S.; Abbott, L.K. Soil salinity delays germination and limits growth of hyphae from propagules of arbuscular mycorrhizal fungi. Mycorrhiza 2006, 16, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Upadhyaya, H.D.; Sharma, S.; Gowda, C.L.L.; Reddy, V.G.; Singh, S. Developing proso millet (Panicum miliaceum L.) core collection using geographic and morpho-agronomic data. Crop Pasture Sci. 2011, 62, 383–389. [Google Scholar] [CrossRef]
- Channabasava, A.; Lakshman, H.C.; Jorquera, M.A. Effect of fungicides on association of arbuscular mycorrhiza fungus Rhizophagus fasciculatus and growth of proso millet (Panicum miliaceum L.). J. Soil Sci. Plant Nutr. 2015, 15, 35–45. [Google Scholar] [CrossRef]
- Arab, A.; Bradaran, R.; Vahidipour, T.H. Effect of irrigation and mycorrhizal bio-fertilizers on yield and agronomic traits of millet (Panicum miliaceum L.). Int. J. Agri. Crop Sci. 2013, 6, 103–109. [Google Scholar]
- Ledér, I. Sorghum and millet in cultivated plants, primarily as food sources. In Encyclopedia of Life Support Systems (EOLSS), Developed under the Auspices of the UNESCO; Eolss Publishers: Oxford, UK, 2004. [Google Scholar]
- Food and Agriculture Organization (FAO). FAOSTAT Statistical Database 2014; FAO: Rome, Italy, 2014. [Google Scholar]
- Yue, H.; Wang, M.; Liu, S.; Du, X.; Song, W.; Nie, X. Transcriptome-wide identification and expression profiles of the WRKY transcription factor family in Broomcorn millet (Panicum miliaceum L.). BMC Genom. 2016, 17, 343. [Google Scholar] [CrossRef] [PubMed]
- Hunt, H.V.; Campana, M.G.; Lawes, M.C.; Park, Y.J.; Bower, M.A.; Howe, C.J.; Jones, M.K. Genetic diversity and phylogeography of broomcorn millet (Panicum miliaceum L.) across Eurasia. Mol. Ecol. 2011, 20, 4756–4771. [Google Scholar] [CrossRef] [PubMed]
- Saleh, A.S.; Zhang, Q.; Chen, J.; Shen, Q. Millet grains: Nutritional quality, processing, and potential health benefits. Compr. Rev. Food Sci. Food Saf. 2013, 12, 281–295. [Google Scholar] [CrossRef]
- Morales, J.U.; Alatorre, J.A.H.; Nieto, C.A.R.; Becerra, J.F.C. Forage production and nutritional content of silage from three varieties of pearl millet (Pennisetum glaucum) harvested at two maturity stages. J. Anim. Plant Sci. 2015, 27, 4161–4169. [Google Scholar]
- Jahansouz, M.R.; Afshar, R.K.; Heidari, H.; Hashemi, M. Evaluation of yield and quality of sorghum and millet as alternative forage crops to corn under normal and deficit irrigation regimes. Jordan J. Agric. Sci. 2014, 10, 699–715. [Google Scholar]
- Mohajer, S.; Ghods, H.; Taha, R.M.; Talati, A. Effect of different harvest time on yield and forage quality of three varieties of common millet (Panicum miliaceum). Sci. Res. Essays 2012, 7, 3020–3025. [Google Scholar] [CrossRef]
- Ali, S.A.; Idris, A.Y. Germination and seedling growth of pearl millet (Pennisetum glaucum L.) cultivars under salinity conditions. Int. J. Plant Res. 2015, 1, 1–5. [Google Scholar]
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. Single crop coefficient (KC). In Crop Evapotranspiration—Guidelines for Computing Crop Water Requirements; FAO Irrigation and Drainage Paper 56; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 1998; ISBN 92-5-104219-5. [Google Scholar]
- Hess, M.; Barralis, G.; Bleiholder, H.; Buhr, L.; Eggers, T.H.; Hack, H.; Stauss, R. Use of the extended BBCH scale—General for the descriptions of the growth stages of mono; and dicotyledonous weed species. Weed Res. 1997, 37, 433–441. [Google Scholar] [CrossRef]
- Dodd, G.L.; Donovan, L.A. Water potential and ionic effects on germination and seedling growth of two cold desert shrubs. Am. J. Bot. 1999, 86, 1146–1153. [Google Scholar] [CrossRef] [PubMed]
- Almansouri, M.; Kinet, J.M.; Lutts, S. Effect of salt and osmotic stresses on germination in durum wheat (Triticum durum Desf.). Plant Soil 2001, 231, 243–254. [Google Scholar] [CrossRef]
- Ryan, J.; Miyamoto, S.; Stroehlein, J.L. Salt and specific ion effects on germination of four grass. J. Range Manag. 1975, 28, 61–64. [Google Scholar] [CrossRef]
- Cavallaro, V.; Barbera, A.C.; Maucieri, C.; Gimma, G.; Scalisi, C.; Patanè, C. Evaluation of variability to drought and saline stress through the germination of different ecotypes of carob (Ceratonia siliqua L.) using a hydrotime model. Ecol. Eng. 2016, 95, 557–566. [Google Scholar] [CrossRef]
- Sabir, P.; Ashraf, M. Inter-cultivar variation for salt tolerance in proso millet (Panicum miliaceum L.) at the germination stage. Pak. J. Bot. 2008, 40, 677–682. [Google Scholar]
- Daei, G.; Ardekani, M.R.; Rejali, F.; Teimuri, S.; Miransari, M. Alleviation of salinity stress on wheat yield, yield components, and nutrient uptake using arbuscular mycorrhizal fungi under field conditions. J. Plant Physiol. 2009, 166, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Porcel, R.; Ruiz-Lozano, J.M. Arbuscular mycorrhizal influence on leaf water potential, solute accumulation, and oxidative stress in soybean plants subjected to drought stress. J. Exp. Bot. 2004, 55, 1743–1750. [Google Scholar] [CrossRef] [PubMed]
- Saia, S.; Amato, G.; Frenda, A.S.; Giambalvo, D.; Ruisi, P. Influence of arbuscular mycorrhizae on biomass production and nitrogen fixation of berseem clover plants subjected to water stress. PLoS ONE 2014, 9, e90738. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oladele, S.; Awodun, M. Response of lowland rice to biofertilizers inoculation and their effects on growth and yield in southwestern Nigeria. J. Agric. Environ. Sci. 2014, 3, 371–390. [Google Scholar]
- Augé, R.M.; Toler, H.D.; Saxton, A.M. Arbuscular mycorrhizal symbiosis alters stomatal conductance of host plants more under drought than under amply watered conditions: A meta-analysis. Mycorrhiza 2015, 25, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Fagbola, O.; Osonubi, O.; Mulongoy, K.; Odunfa, S. Effects of drought stress and arbuscular mycorrhiza on the growth of Gliricidia sepium (Jacq). Walp, and Leucaena leucocephala (Lam.) de Wit. in simulated eroded soil conditions. Mycorrhiza 2001, 11, 215–223. [Google Scholar] [CrossRef]
- Zhou, Q.; Ravnskov, S.; Jiang, D.; Wollenweber, B. Changes in carbon and nitrogen allocation, growth and grain yield induced by arbuscular mycorrhizal fungi in wheat (Triticum aestivum L.) subjected to a period of water deficit. Plant Growth Regul. 2015, 75, 751–760. [Google Scholar] [CrossRef]
- Fidalgo, F.; Santos, A.; Santos, I.; Salema, R. Effects of long-term salt stress on antioxidant defense systems, leaf water relations and chloroplast ultrastructure of potato plants. Ann. Appl. Biol. 2004, 145, 185–192. [Google Scholar] [CrossRef]
- Shi, L.X.; Guo, J.X. Changes in photosynthetic and growth characteristics of Leymus chinensis community along the retrogression on the Songnen grassland in northeastern China. Photosynthetica 2006, 44, 542–547. [Google Scholar] [CrossRef]
- Beltrano, J.; Ronco, M.G. Improved tolerance of wheat plants (Triticum aestivum L.) to drought stress and rewatering by the arbuscular mycorrhizal fungus Glomus claroideum: Effect on growth and cell membrane stability. Braz. J. Plant Physiol. 2008, 20, 29–37. [Google Scholar] [CrossRef]
- Beltrano, J.; Ruscitti, M.; Arango, M.C.; Ronco, M. Effects of arbuscular mycorrhiza inoculation on plant growth, biological and physiological parameters and mineral nutrition in pepper grown under different salinity and p levels. J. Soil Sci. Plant Nutr. 2013, 13, 123–141. [Google Scholar] [CrossRef]
- Grümberg, B.C.; Urcelay, C.; Shroeder, M.A.; Vargas-Gil, S.; Luna, C.M. The role of inoculum identity in drought stress mitigation by arbuscular mycorrhizal fungi in soybean. Biol. Fertil. Soils 2015, 51, 1–10. [Google Scholar] [CrossRef]



| Treatment Number | Osmotic Agent | Osmotic Agent Weight (g L−1) | Osmotic Pressure (MPa) |
|---|---|---|---|
| 1 | Distilled water | 0 | 0 |
| 2 | NaCl | 4.1 | −0.250 |
| 3 | NaCl | 7.6 | −0.500 |
| 4 | NaCl | 11.1 | −0.750 |
| 5 | Mannitol | 21.6 | −0.250 |
| 6 | Mannitol | 42.5 | −0.500 |
| 7 | Mannitol | 63.4 | −0.750 |
| Osmotic Pressure (MPa) | Genotypes | Mean | ||||||
|---|---|---|---|---|---|---|---|---|
| Kinelskoje | Unikum | VIR 9181 | ||||||
| 0.0 | 3.93 ± 0.17 | 5.48 ± 0.07 | 5.35 ± 0.16 | 4.92 ± 0.19 | ||||
| −0.250 | 2.48 ± 0.29 | 4.08 ± 0.46 | 2.66 ± 0.23 | 3.07 ± 0.25 | ||||
| −0.500 | 2.45 ± 0.32 | 3.01 ± 0.36 | 2.93 ± 0.36 | 2.79 ± 0.20 | ||||
| −0.750 | 1.08 ± 0.13 | 2.04 ± 2.04 | 1.18 ± 0.10 | 1.43 ± 0.13 | ||||
| Mean | 2.48 ± 0.24 | 3.65 ± 0.30 | 3.03 ± 0.33 | 3.05 ± 0.18 | ||||
| ANOVA | SS | DF | MS | F | Prob. F | Sign. F | LSD | |
| p < 0.05 | p < 0.01 | |||||||
| G | 15.7309 | 2 | 7.8654 | 17.2022 | 2.33 × 10−6 | ** | 0.39247 | 0.52356 |
| OP | 108.764 | 3 | 36.256 | 79.2911 | 1.30 × 10−18 | ** | 0.45319 | 0.60456 |
| S | 0.0147 | 1 | 0.0147 | 0.0321 | 8.59 × 10−1 | 0.32045 | 0.42749 | |
| G × OP | 6.1833 | 6 | 1.0306 | 2.2539 | 5.37 × 10−2 | 0.78495 | 1.04713 | |
| G × S | 0.3383 | 2 | 0.1692 | 0.3700 | 6.93 × 10−1 | 0.55504 | 0.74043 | |
| OP × S | 0.5102 | 3 | 0.1701 | 0.3719 | 7.74 × 10−1 | 0.64091 | 0.85498 | |
| G × OP × S | 4.1026 | 6 | 0.6838 | 1.4954 | 2.00× 10−1 | 1.11009 | 1.48086 | |
| Residual | 21.9472 | 48 | 0.4572 | |||||
| Total | 157.5909 | 71 | ||||||
| Treatments | Culm Height (cm) | Panicle Length (cm) | Leaf Number (plant−1) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Kinelskoje | AMF-Y | Salt-Y | 25% ETm | 13.3 ± 2.37 | 2.06 ± 1.35 | 3.75 ± 0.45 | |||
| 100% ETm | 25.1 ± 2.09 | 10.6 ± 1.11 | 5.50 ± 0.33 | ||||||
| Salt-N | 25% ETm | 18.1 ± 3.59 | 11.2 ± 0.64 | 4.88 ± 0.23 | |||||
| 100% ETm | 42.8 ± 2.27 | 13.7 ± 0.39 | 4.50 ± 0.46 | ||||||
| AMF-N | Salt-Y | 25% ETm | 12.1 ± 1.43 | 0.00 ± 0.00 | 3.38 ± 0.26 | ||||
| 100% ETm | 16.2 ± 1.02 | 10.9 ± 0.33 | 5.00 ± 0.19 | ||||||
| Salt-N | 25% ETm | 11.0 ± 0.58 | 0.00 ± 0.00 | 1.63 ± 0.63 | |||||
| 100% ETm | 27.1 ± 2.54 | 12.5 ± 2.39 | 5.00 ± 0.27 | ||||||
| Unikum | AMF-Y | Salt-Y | 25% ETm | 15.5 ± 1.39 | 0.00 ± 0.00 | 4.38 ± 0.18 | |||
| 100% ETm | 30.5 ± 1.79 | 16.0 ± 1.27 | 4.75 ± 0.25 | ||||||
| Salt-N | 25% ETm | 17.1 ± 2.79 | 9.81 ± 2.28 | 4.25 ± 0.25 | |||||
| 100% ETm | 47.4 ± 1.68 | 21.2 ± 0.87 | 4.75 ± 0.25 | ||||||
| AMF-N | Salt-Y | 25% ETm | 17.5 ± 2.44 | 0.00 ± 0.00 | 4.00 ± 0.38 | ||||
| 100% ETm | 33.6 ± 0.86 | 17.3 ± 0.60 | 5.13 ± 0.13 | ||||||
| Salt-N | 25% ETm | 12.0 ± 0.92 | 0.00 ± 0.00 | 4.38 ± 0.18 | |||||
| 100% ETm | 31.1 ± 0.95 | 13.7 ± 1.15 | 5.25 ± 0.16 | ||||||
| ANOVA | Sign. | LSD | Sign. | LSD | Sign. | LSD | |||
| Genotype (G) | ** | 2.476 | ** | 1.410 | * | 0.314 | |||
| Mycorrhizal inoculation (M) | ** | 2.476 | ** | 1.410 | ns | - | |||
| Water salinity (S) | ** | 2.476 | ** | 1.410 | * | 0.314 | |||
| Water restitution (W) | ** | 2.476 | ** | 1.410 | ** | 0.416 | |||
| G × M | * | 2.647 | ns | - | ** | 0.588 | |||
| G × S | ** | 3.502 | ns | - | ns | - | |||
| G × W | ** | 3.502 | ** | 1.994 | ** | 0.588 | |||
| M × S | ** | 3.502 | ** | 1.994 | ns | - | |||
| M × W | ** | 3.502 | ** | 1.994 | ** | 0.588 | |||
| S × W | ** | 3.502 | ** | 1.994 | ns | - | |||
| G × M × S | ns | - | ns | - | ns | - | |||
| G × M × W | ns | - | * | 2.820 | ns | - | |||
| G × S × W | ns | - | ns | - | ns | - | |||
| M × S × W | ns | - | * | 2.820 | ns | - | |||
| G × M × S × W | ns | - | ns | - | ns | - | |||
| Treatments | Culm (g) | Panicle (g) | Leaves (g) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Kinelskoje | AMF-Y | Salt-Y | 25% ETm | 0.37 ± 0.11 | 0.16 ± 0.11 | 0.63 ± 0.23 | |||
| 100% ETm | 1.36 ± 0.29 | 0.42 ± 0.11 | 1.10 ± 0.16 | ||||||
| Salt-N | 25% ETm | 0.96 ± 0.17 | 0.47 ± 0.07 | 0.93 ± 0.08 | |||||
| 100% ETm | 2.82 ± 0.24 | 0.57 ± 0.06 | 1.13 ± 0.15 | ||||||
| AMF-N | Salt-Y | 25% ETm | 0.22 ± 0.04 | 0.00 ± 0.00 | 0.22 ± 0.04 | ||||
| 100% ETm | 0.81 ± 0.09 | 0.54 ± 0.08 | 0.67 ± 0.12 | ||||||
| Salt-N | 25% ETm | 0.34 ± 0.04 | 0.00 ± 0.00 | 0.15 ± 0.08 | |||||
| 100% ETm | 1.74 ± 0.27 | 0.59 ± 0.11 | 1.10 ± 0.18 | ||||||
| Unikum | AMF-Y | Salt-Y | 25% ETm | 0.41 ± 0.18 | 0.00 ± 0.00 | 0.58 ± 0.17 | |||
| 100% ETm | 1.69 ± 0.29 | 0.84 ± 0.15 | 1.04 ± 0.23 | ||||||
| Salt-N | 25% ETm | 0.54 ± 0.12 | 0.53 ± 0.11 | 0.79 ± 0.06 | |||||
| 100% ETm | 3.38 ± 0.44 | 1.12 ± 0.16 | 1.40 ± 0.12 | ||||||
| AMF-N | Salt-Y | 25% ETm | 0.40 ± 0.07 | 0.00 ± 0.00 | 0.61 ± 0.23 | ||||
| 100% ETm | 1.61 ± 0.20 | 0.94 ± 0.25 | 1.18 ± 0.15 | ||||||
| Salt-N | 25% ETm | 0.47 ± 0.10 | 0.00 ± 0.00 | 0.47 ± 0.10 | |||||
| 100% ETm | 1.58 ± 0.22 | 0.85 ± 0.17 | 1.34 ± 0.11 | ||||||
| ANOVA | Sign. | LSD | Sign. | LSD | Sign. | LSD | |||
| Genotype (G) | ns | - | ** | 0.148 | * | 0.140 | |||
| Mycorrhizal inoculation (M) | ** | 0.249 | ** | 0.148 | * | 0.140 | |||
| Water salinity (S) | ** | 0.249 | ** | 0.148 | ** | 0.185 | |||
| Water restitution (W) | ** | 0.249 | ** | 0.148 | ** | 0.185 | |||
| G × M | ns | - | ns | - | * | 0.198 | |||
| G × S | ns | - | ns | - | ns | - | |||
| G × W | * | 0.266 | ** | 0.210 | ns | - | |||
| M × S | ** | 0.352 | ** | 0.210 | ns | - | |||
| M × W | ** | 0.352 | * | 0.159 | ns | - | |||
| S × W | ** | 0.352 | ns | - | ns | - | |||
| G × M × S | ns | - | ns | - | ns | - | |||
| G × M × W | ns | - | ns | - | ns | - | |||
| G × S × W | ns | - | ns | - | ns | - | |||
| M × S × W | ns | - | ns | - | ns | - | |||
| G × M × S × W | ns | - | ns | - | ns | - | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caruso, C.; Maucieri, C.; Berruti, A.; Borin, M.; Barbera, A.C. Responses of Different Panicum miliaceum L. Genotypes to Saline and Water Stress in a Marginal Mediterranean Environment. Agronomy 2018, 8, 8. https://doi.org/10.3390/agronomy8010008
Caruso C, Maucieri C, Berruti A, Borin M, Barbera AC. Responses of Different Panicum miliaceum L. Genotypes to Saline and Water Stress in a Marginal Mediterranean Environment. Agronomy. 2018; 8(1):8. https://doi.org/10.3390/agronomy8010008
Chicago/Turabian StyleCaruso, Caterina, Carmelo Maucieri, Andrea Berruti, Maurizio Borin, and Antonio Carlo Barbera. 2018. "Responses of Different Panicum miliaceum L. Genotypes to Saline and Water Stress in a Marginal Mediterranean Environment" Agronomy 8, no. 1: 8. https://doi.org/10.3390/agronomy8010008






