The Impact of Biochar Application on Soil Properties and Plant Growth of Pot Grown Lettuce (Lactuca sativa) and Cabbage (Brassica chinensis)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Growing Substrates
| Treatment | Name | Biochar (g kg−1) (Mg ha−1*) | Lake sediment (g kg−1) | Compost (g kg−1) | Liquid compost |
|---|---|---|---|---|---|
| 0 | Non-fertilized soil without biochar | 0 | 0 | 0 | No |
| B50 | Non-fertilized soil with biochar | 50.24 (80) | 0 | 0 | No |
| CS | Fertilized soil without biochar | 0 | 12.56 | 25.12 | Yes |
| CS + B25 | Fertilized soil with a low dose of biochar | 25.12 (40) | 12.56 | 25.12 | Yes |
| CS + B50 | Fertilized soil with a medium dose of biochar | 50.24 (80) | 12.56 | 25.12 | Yes |
| CS + B150 | Fertilized soil with a high dose of biochar | 150.00 (167) | 12.56 | 25.12 | Yes |
2.2. Pot Experiment
2.3. Analysis
| Properties/Analyte | Unit | Soil (0) | Soil plus local fertilizers (“CS”) | Biochar |
|---|---|---|---|---|
| Ph | pH unit | 4.77 | 5.39 | 7.79 |
| Total C | % dry weight | 0.48 | 0.35 | 28.7 |
| Total N | % dry weight | 0.30 | 0.15 | 0.65 |
| Total S | % dry weight | 0.00 | 0.00 | <0.03 |
| Exchangeable K | cmol + kg−1 | 1.02 | 1.73 | 36.4 |
| Exchangeable Na | cmol + kg−1 | 11.38 | 11.98 | 1.5 |
| Exchangeable Ca | cmol + kg−1 | 4.58 | 11.11 | 12.4 |
| Exchangeable Mg | cmol + kg−1 | 1.70 | 3.09 | 12.8 |
| CEC | cmol + kg−1 | 18.44 | 24.24 | 44.5 |
| LOI | % | 2.02 | 6.80 | - |
| Al | mg kg−1 | 8929 | 9019 | 92.0 |
| As | mg kg−1 | <1.53 | <1.63 | <1.79 |
| Be | mg kg−1 | <3.06 | <3.26 | <3.59 |
| Cd | mg kg−1 | <0.31 | <0.33 | <0.36 |
| Cr | mg kg−1 | 11.0 | 10.9 | <1.44 |
| Cu | mg kg−1 | 2.02 | 2.25 | 8.15 |
| Fe | mg kg−1 | 4485 | 4554 | 65.6 |
| Pb | mg kg−1 | 5.10 | 5.39 | 2.62 |
| Mn | mg kg−1 | 26.3 | 53.4 | 135 |
| Hg | mg kg−1 | <1.53 | <1.63 | <1.79 |
| Ni | mg kg−1 | 3.48 | 3.92 | <1.08 |
| Si | mg kg−1 | 79.3 | 92.9 | 66.0 |
| Ti | mg kg−1 | 42.4 | 42.9 | 1.79 |
| Zn | mg kg−1 | 6.08 | 7.15 | 11.7 |
| V | mg kg−1 | 19.0 | 17.9 | <1.79 |
| Ba | mg kg−1 | 14.9 | 15.4 | 19.3 |
| Na | mg kg−1 | 42.3 | 48.5 | 76.1 |
| Ca | mg kg−1 | 85.4 | 259 | 609 |
| Mg | mg kg−1 | 165 | 226 | 162 |
| K | mg kg−1 | 315 | 428 | 595 |
| Sr | mg kg−1 | 2.20 | 2.74 | 1.87 |
| B | mg kg−1 | <1.49 | <1.54 | 5.83 |
| USEPA 16 PAHs* | mg kg−1 | - | - | 14.6 |
| Clay (<0.002 mm) | % | 16 | 17 | - |
| Fine silt (0.002–0.02 mm) | % | 18 | 22 | - |
| Coarse silt (0.02–0.05 mm) | % | 5 | 5 | - |
| Fine sand (0.05–0.2 mm) | % | 20 | 26 | - |
| Coarse sand (0.2–2 mm) | % | 41 | 30 | - |
3. Results and Discussion
3.1. Soil and Biochar Characterisation

3.2. Plant Growth Indicators
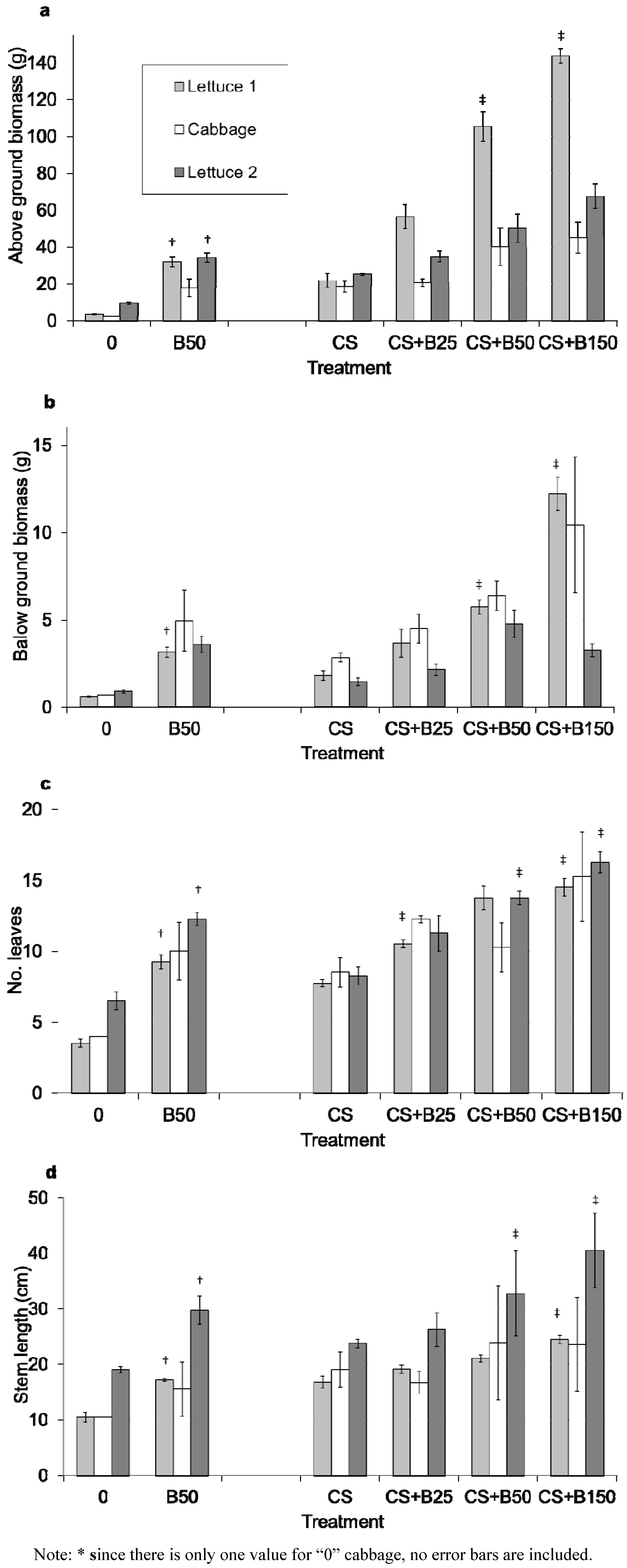
| Crop | Above ground biomass (% above baseline) | Baseline | Biochar additions |
|---|---|---|---|
| Lettuce | 903 | Non-fertilized soil without biochar (0) | Non-fertilized soil with 50 g kg−1 biochar (B50) |
| Lettuce | 259 | Fertilized soil without biochar (CS) | Fertilized soil with 25 g kg−1 biochar (CS + B25) |
| Lettuce | 483 | Fertilized soil with 50 g kg−1 biochar (CS + B50) | |
| Lettuce | 658 | Fertilized soil with 150 g kg−1 biochar (CS + B150) | |
| Cabbage | 750 | Non-fertilized soil without biochar (0) | Non-fertilized soil with 50 g kg−1 biochar (B50) |
| Cabbage | 111 | Fertilized soil without biochar (CS) | Fertilized soil with 25 g kg−1 biochar (CS + B25) |
| Cabbage | 214 | Fertilized soil with 50 g kg−1 biochar (CS + B50) | |
| Cabbage | 241 | Fertilized soil with 150 g kg−1 biochar (CS + B150) | |
| Lettuce | 363 | Non-fertilized soil without biochar (0) | Non-fertilized soil with 50 g kg−1 biochar (B50) |
| Lettuce | 139 | Fertilized soil without biochar (CS) | Fertilized soil with 25 g kg−1 biochar (CS + B25) |
| Lettuce | 200 | Fertilized soil with 50 g kg−1 biochar (CS + B50) | |
| Lettuce | 268 | Fertilized soil with 150 g kg−1 biochar (CS + B150) |
4. Conclusions
Acknowledgments
References
- Shackley, S.; Carter, S.; Knowles, T.; Middelink, E.; Haefele, S.; Sohi, S.; Cross, A.; Haszeldine, S. Sustainable gasification-biochar systems? A case study of rice-husk gasification in Cambodia Part I: Context, chemical properties, environmental and health and safety issues. Energy Policy 2012, 42, 49–58. [Google Scholar]
- Shackley, S.; Carter, S.; Sims, K.; Sohi, S. Expert perceptions of the role of biochar as a carbon abatement option with ancillary agronomic and soil-related benefits. Energy Environ. 2010, 22, 167–187. [Google Scholar]
- Shackley, S.; Carter, S.; Knowles, T.; Middelink, E.; Haefele, S.; Haszeldine, S. Sustainable gasification-biochar systems? A case-study of rice-husk gasification in Cambodia, Part II: Field trial results, carbon abatement, economic assessment and conclusions. Energy Policy 2012, 41, 618–623. [Google Scholar] [CrossRef]
- Sokchea, H.; Borin, K.; Preston, T. Effect of biochar from rice husks (combusted in a downdraft gasifier or a paddy rice dryer) on production of rice fertilized with biodigester effluent or urea. Livest. Res. Rural Dev. 2013, 25. Article No. 4.. Available online: http://www.lrrd.org/lrrd25/1/ sokc25004.htm (accessed on 23 January 2013). [Google Scholar]
- Lehmann, J.; Gaunt, J.; Rondon, M. Bio-char sequestration in terrestrial ecosystems—A review. Mitig. Adapt. Strategies Glob. Chang. 2006, 11, 403–427. [Google Scholar]
- Asai, H.; Samson, B.K.; Haefele, S.M.; Songyikhangs, K.; Homma, K.; Kiyono, Y.; Inoue, Y.; Shiraiwa, T.; Horie, T. Biochar amendment techniques for upland rice production in Northern Laos. 1. Soil physical properties, leaf SPAD and grain yield. Field Crops Res. 2009, 111, 81–84. [Google Scholar]
- Hammond, J.; Shackley, S.; Prendergast-Miller, M.; Cook, J.; Buckingham, S.; Pappa, V. Biochar Field Testing in the UK: Outcomes and Implications for Use. Carbon Manag. 2013, in press. [Google Scholar]
- Rice Production in Cambodia; Nesbitt, H.J. (Ed.) International Rice Research Institute: Manilla, Philippines, 1997; pp. 15–29.
- Shackley, S.; Sohi, S.P. An Assessment of the Benefits and Issues Associated with the Application of Biochar to Soil; Report to the Department for Environment, Food and Rural Affairs and the Department of Energy and Climate Change: London, UK, 2010; pp. 14–132. [Google Scholar]
- International Biochar Initiative (IBI), Standardized Product Definition and Product Testing Guidelines for Biochar that is Used in Soil, International Biochar Initiative 2012. Available online: http://www.biochar-international.org/sites/default/files/guidelines_for_biochar_that_is_used_in_ soil_final.pdf (accessed on 21 January 2013).
- European Biochar Certificate (EBC) Guidelines for a Sustainable Production of Biochar, Version 4.5; Delinat Institut and Biochar Science Network: Ayent, Switzerland, 2013. Available online: http://www.european-biochar.org/biochar/media/doc/1358641517626.pdf (accessed on 21 January 2013).
- Sohi, S.P. Appendix 1. Analysis of scientific studies published on the function of char, its quantification, and its stability in soil. In An Assessment of the Benefits and Issues Associated with the Application of Biochar to Soil; Shackley, S., Sohi, S.P., Eds.; A Report to the Department for Environment, Food and Rural Affairs and the Department of Energy and Climate Change: London, UK, 2010; pp. 1–4. [Google Scholar]
- Jeffery, S.; Verheijen, F.G.A.; van der Velde, M.; Bastos, A.C. A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agric. Ecosyst. Environ. 2011, 144, 175–187. [Google Scholar] [CrossRef]
- Verheijen, F.G.A.; Jeffery, S.; Bastos, A.C.; van der Velde, M.; Diafas, I. Biochar Application to Soils—A Critical Scientific Review of Effects on Soil Properties, Processes and Functions; Office for the Official Publications of the European Communities: Luxembourg, 2009; EUR 24099 EN; p. 61. [Google Scholar]
- Haefele, S.; Konboon, Y.; Wongboon, W.; Amarante, S.; Maarifat, A.; Pfeiffer, E.; Knoblauch, C. Effects and fate of biochar from rice residues in rice-based systems. Field Crops Res. 2011, 121, 430–441. [Google Scholar] [CrossRef]
- Shackley, S.; Sohi, S.; Ibarrola, R.; Hammond, J.; Mašek, O.; Brownsort, P.; Haszeldine, S. Biochar as a Tool for Climate Change Mitigation and Soil Management. In Encyclopedia of Sustainability Science and Technology; Meyers, R., Ed.; Springer: New York, NY, USA, 2012; pp. 183–205. [Google Scholar]
- Lehmann, J. Bio-energy in the black. Front. Ecol. Environ. 2007, 5, 381–387. [Google Scholar] [CrossRef]
- Glaser, B.; Lehmann, J.; Zech, W. Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal—A review. Biol. Fertil. Soils 2002, 35, 219–230. [Google Scholar] [CrossRef]
- Chan, K.Y.; Van Zwieten, L.; Meszaros, I.; Downie, A.; Joseph, S. Agronomic values of greenwaste biochar as a soil amendment. Aust. J. Soil Res. 2007, 45, 629–634. [Google Scholar] [CrossRef]
- Saarnio, S.; Heimonen, K.; Kettunen, R. Biochar addition indirectly affects N2O emissions via soil moisture and plant N uptake. Soil Biol. Biochem. 2013, 58, 99–106. [Google Scholar] [CrossRef]
- Zavalloni, C.; Alberti, G.; Biasiol, S.; Vedove, G.D.; Fornasier, F.; Liu, J.; Peressotti, A. Microbial mineralization of biochar and wheat straw mixture in soil: A short-term study. Appl. Soil Ecol. 2011, 50, 45–51. [Google Scholar] [CrossRef]
- Case, S.J.; McNamara, N.P.; Reay, D.S.; Whitaker, J. The effect of biochar addition on N2O and CO2 emissions from a sandy loam soil—The role of soil aeration. Soil Biol. Biochem. 2012, 51, 125–134. [Google Scholar] [CrossRef]
- FAO. Food and Agriculture organization of the UN World soil resources online. Land and Water development division. WRB Map of World Soil Resources, 2002 updates. Available online: http://www.fao.org/ag/agl/agll/wrb/soilres.stm (accessed on 1 July 2010).
- Bell, R.W.; Seng, V. Rainfed lowland rice-growing soils of Cambodia, Laos, and North-east. Thailand; ACIAR Proceedings No. 116e. In Water in Agriculture; Seng, V., Craswell, E., Fukai, S., Fisher, K., Eds.; Australian Centre for International Agricultural Research(ACIAR): Canberra, Australia, 2004; pp. 161–173. [Google Scholar]
- Mulcahy, D.N.; Mulcahy, D.L.; Dietz, D. Biochar soil amendment increases tomato seedling resistance to drought in sandy soils. J. Arid Environ. 2013, 81, 222–225. [Google Scholar] [CrossRef]
- Hilber, I.; Blum, F.; Leifeld, J.; Schmidt, H.P.; Bucheli, T. Quantitative Determination of PAHs in Biochar: A Prerequisite To Ensure its Quality and Safe Application. J. Agri. Food Chem. 2012, 60, 3042–3050. [Google Scholar]
- Hale, S.; Lehmann, J.; Rutherford, D.; Zimmerman, A.; Bachmann, R.; Shitumbanuma, V.; O’Toole, A.; Sundqvist, K.; Arp, H.; Cornelissen, G. Quantifying the total and bioavailable polycyclic armoatic hydrocarbons and dioxins in biochar. Environ. Sci. Technol. 2012, 46, 2830–2838. [Google Scholar] [CrossRef]
- Somtrakoon, K.; Chouychai, W. Phytotoxicity of single and combined polycyclic aromatic hydrocarbons toward economic crops. Russian J. Plant Physiol. 2013, 60, 139–148. [Google Scholar] [CrossRef]
- Biederman, L.A.; Harpole, W.S. Biochar and its effects on plant productivity and nutrient cycling: A meta-analysis. GCB Bioenergy. 2013, 5, pp. 202–214. Available online: http://onlinelibrary.wiley.com/doi/10.1111/gcbb.12037/full (accessed on 21 January 2013). [CrossRef]
- Food and Agriculture Organization of the UN Ecocrop data sheet—Brassica chinensis. Available online: http://ecocrop.fao.org/ecocrop/srv/en/dataSheet?id=547 (accessed on 27 September 2010).
- Food and Agriculture Organization of the UN Ecocrop data sheet—Lactuca sativa. Available online: http://ecocrop.fao.org/ecocrop/srv/en/dataSheet?id=1313 (accessed on 27 September 2010).
- Wunderground: Siem Reap Airport. Available online: http://www.wunderground.com/ (accessed on 4 July 2010).
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Carter, S.; Shackley, S.; Sohi, S.; Suy, T.B.; Haefele, S. The Impact of Biochar Application on Soil Properties and Plant Growth of Pot Grown Lettuce (Lactuca sativa) and Cabbage (Brassica chinensis). Agronomy 2013, 3, 404-418. https://doi.org/10.3390/agronomy3020404
Carter S, Shackley S, Sohi S, Suy TB, Haefele S. The Impact of Biochar Application on Soil Properties and Plant Growth of Pot Grown Lettuce (Lactuca sativa) and Cabbage (Brassica chinensis). Agronomy. 2013; 3(2):404-418. https://doi.org/10.3390/agronomy3020404
Chicago/Turabian StyleCarter, Sarah, Simon Shackley, Saran Sohi, Tan Boun Suy, and Stephan Haefele. 2013. "The Impact of Biochar Application on Soil Properties and Plant Growth of Pot Grown Lettuce (Lactuca sativa) and Cabbage (Brassica chinensis)" Agronomy 3, no. 2: 404-418. https://doi.org/10.3390/agronomy3020404
APA StyleCarter, S., Shackley, S., Sohi, S., Suy, T. B., & Haefele, S. (2013). The Impact of Biochar Application on Soil Properties and Plant Growth of Pot Grown Lettuce (Lactuca sativa) and Cabbage (Brassica chinensis). Agronomy, 3(2), 404-418. https://doi.org/10.3390/agronomy3020404






