1. Introduction
In recent years, due to diminishing crude oil reserves and worsening environmental pollution, more and more attention has been paid to the synthesis of polymers derived from renewable resources to replace the petroleum-based ones. However, their thermal or mechanical properties are still subject to improvement when compared with the petroleum-based engineering plastics like poly(ethylene terephthalate) (PET) and polycarbonate (PC) [
1,
2,
3]. The lack of aromatic or rigid segments in their molecular architecture should be responsible for the relatively low performance. Therefore, exploring the new bio-based platform chemicals with unique structures and then developing polymeric materials with a higher performance–price ratio is always a matter of importance.
2,5-furandicarboxylic acid (FDCA) is exactly the promising bio-based platform chemical, which can be derived from cellulose or hemicellulose. It has been selected as one of the top 12 value-added chemicals derived from biomass by the US Department of Energy. It is also referred to as a “sleeping giant” by DuPont and DSM due to its great potential as a bio-based substitute for terephthalate (TPA) [
4]. In fact, as early as 1940s, the FDCA-based polymers had been firstly reported in Celanese’s work. However, due to the difficulty of obtaining FDCA with high purity and its unacceptable price, hardly no work about polymers derived from FDCA has been reported since. Nowadays, with the rapid development of biotechnology and chemical industry, the large scale industrialized production of FDCA is being realized. Therefore, more and more polymeric materials—Such as polyesters, [
5,
6,
7,
8,
9] polyamides, [
10,
11,
12] polyurethanes [
13,
14], and epoxy resins [
15]—Have been synthesized from FDCA rather than TPA. Among them, the FDCA-based polyesters—Including poly (ethylene 2,5-furandicarboxylate) (PEF) [
8,
16], poly(propylene 2,5-furandicarboxylate) (PPF) [
17], and poly(butylene 2,5-furandicarboxylate) (PBF) [
18,
19,
20,
21]—Might be the most popular stars, which demonstrates similar mechanical and thermal properties as well as better barrier properties when compared with the petroleum-based counterparts (PET/PPT/PBT) [
22,
23]. As we know, the total world production of PET, PPT, and PBT was more than 50 Mt per year, if they could be replaced by the bio-based PEF/PPF/PBF or their copolyesters, great progress towards the sustainable development of polymer industry could be achieved. Therefore, the investigation of the synthesis and modification of PEF, PPF, and PBF has become a hot topic in both the industrial and academic communities.
Up to now, synthesis strategies and basic thermal-physic properties of PEF/PPF/PBF have been widely investigated [
5,
6,
7,
8,
16,
17,
18,
19,
20,
21]. However, being fresh members in the family of polyesters, their properties are still subject to improvement in order to meet the various demands from industry [
24,
25]. For example, the crystallization behaviors of PEF/PPF/PBF were well studied [
17,
18,
26]. Results demonstrated that they were all semi-crystalline polyesters with medium high glass transition temperature. In addition, their crystallizabilities were much poorer when compared with those of PET/PPT/PBT, which would severely limit their fields of application, especially when the highly transparent and heat-resistant properties were required. Up to now, a large quantity of chemical compounds—including isosorbide and isoidide [
27], 1,4-cyclohexanedimethanol (CHDM) [
28], terephthalic acid [
29] and different aliphatic diols [
27,
30]—Have been tried to modify PEF/PPF/PBF. For example, Gomes et al. [
27] synthesized several kinds of FDCA-based copolyesters from isosorbide and isoidide. Results indicated that by using these cyclic diols, amorphous polymers with higher
Tg could be obtained. However, their reported molecular weight was too low to be useful. 1,4-cyclohexanedimethanol (CHDM) was also employed to modify PEF and results showed that after the introduction of CHDM, the toughness of PEF could be significantly increased without sacrificing its
Tg and barrier properties in a great degree. Unfortunately, these copolyesters were still semi-crystalline polymers and high transparency was difficult to achieve [
28]. In one word, completely amorphous FDCA-based copolyesters with increased
Tg, higher transparency as well as good barrier properties and mechanical properties are still targets to be achieved, especially when they are used as packaging materials.
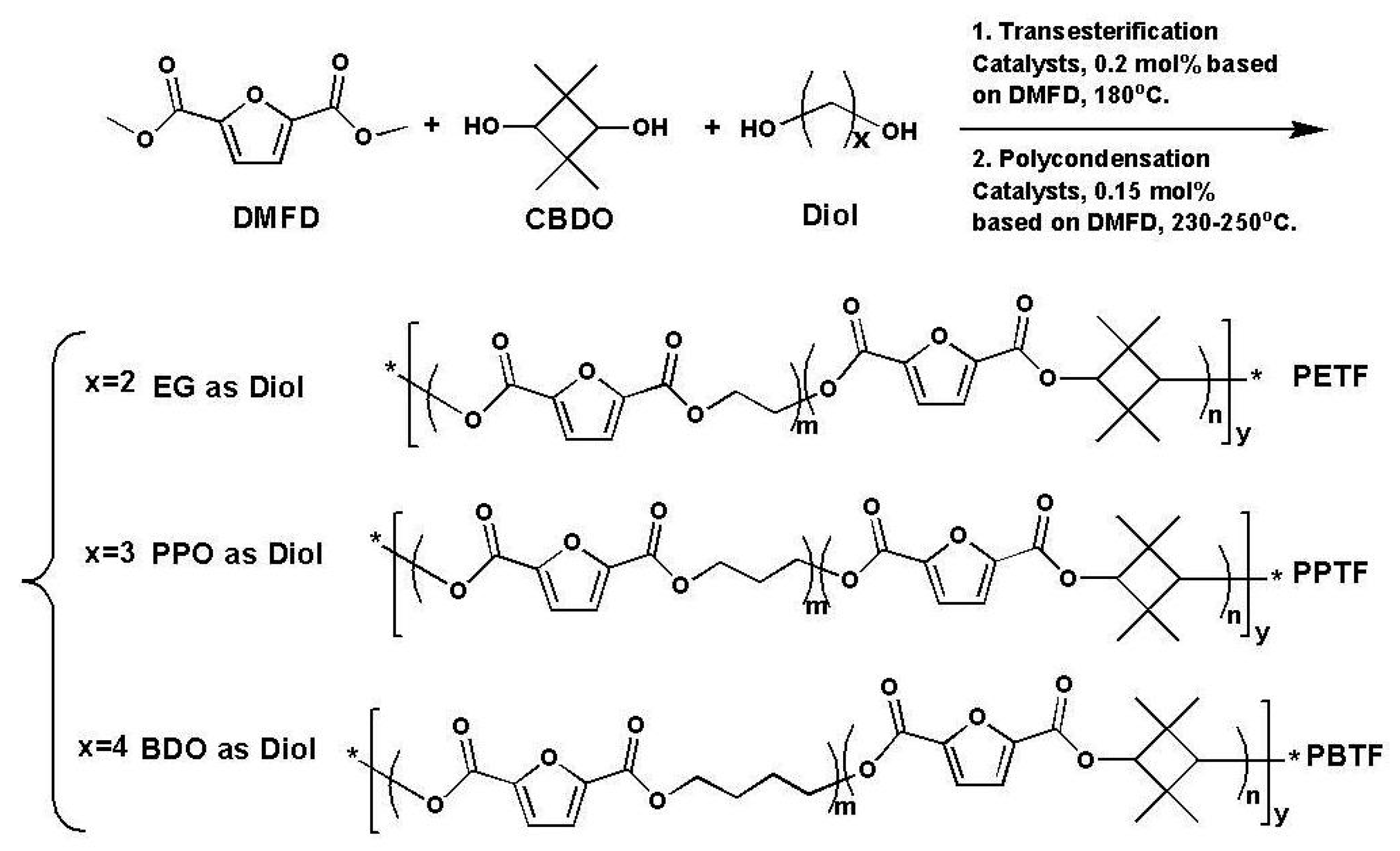
To the best of our knowledge, selecting an ideal cyclic compound as the co-monomer is a well-recognized method to strengthen polyesters without sacrificing their thermal properties. 2,2,4,4-tetramethyl-1,3-cyclobutanediol (CBDO) is one of the unique cyclic aliphatic diols which has been tried as a replacement of bisphenol-A (BPA) to enhance the properties of several polyesters. For instance, Eastman Chemical Company has developed several kinds of CBDO-containing copolyesters, including Tritan
TM, and demonstrated that the introduction of CBDO would lead to higher
Tg, excellent hydrolytic stabilities, and good toughness. Recently, Beall [
31] synthesized an amorphous CBDO-based terephthalate copolyester, which showed much higher
Tg and good toughness. Zhang [
32] prepared a series of multiblock copolyesters from high-
Tg polyester precursors containing CBDO segments. Inspired by the previous publications, in this work, CBDO was employed to modify the FDCA-based polyesters including PEF, PPF, as well as PBF, and a series of copolyesters, such as poly(ethylene-
co-2,2,4,4-tetramethyl-1,3-cyclobutanediol 2,5-furandicarboxylate) (PETF), poly(propylene-
co-2,2,4,4-tetramethyl-1,3-cyclobutanediol 2,5-furandicarboxylate) (PPTF), and poly(tetramethylene-
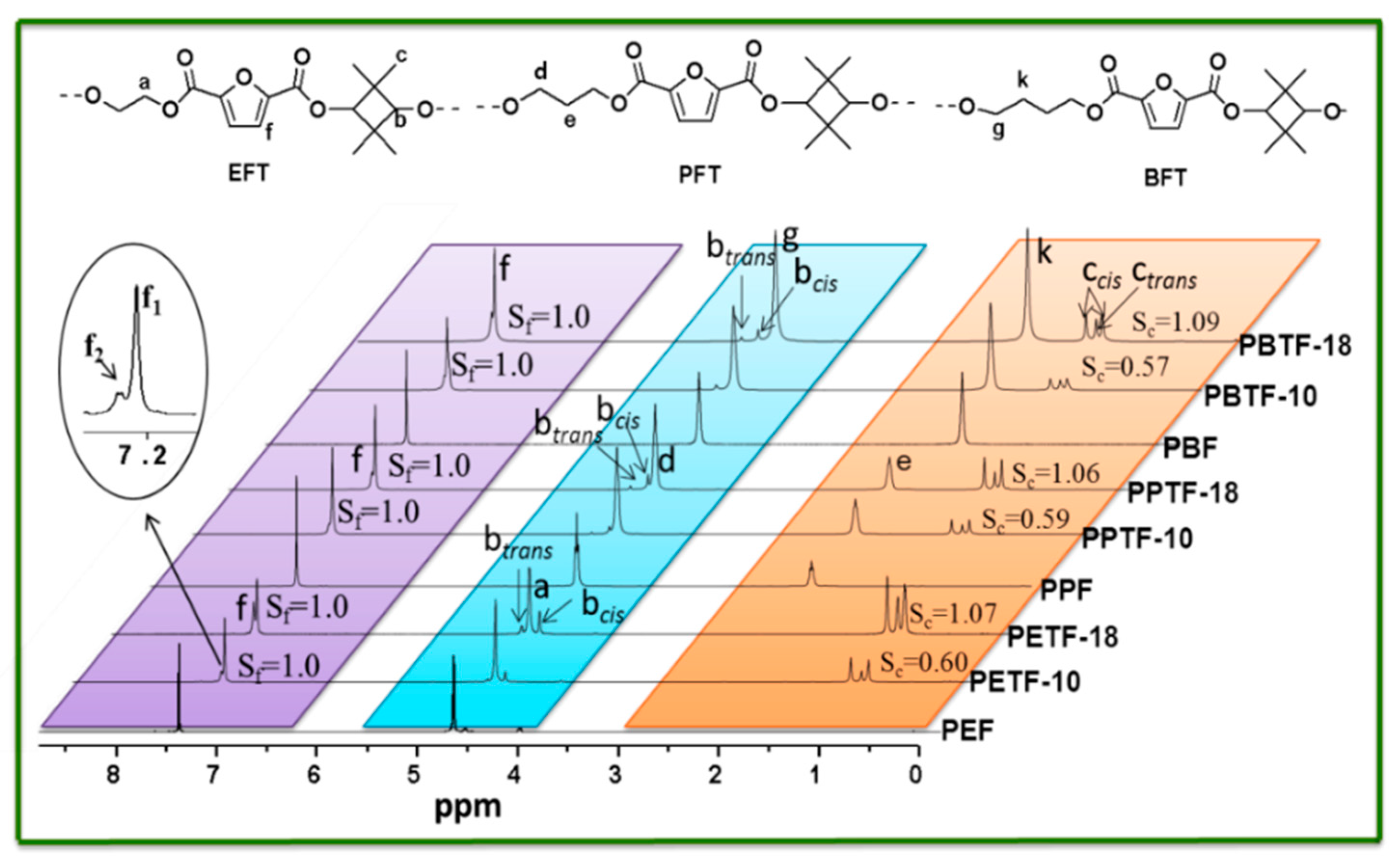
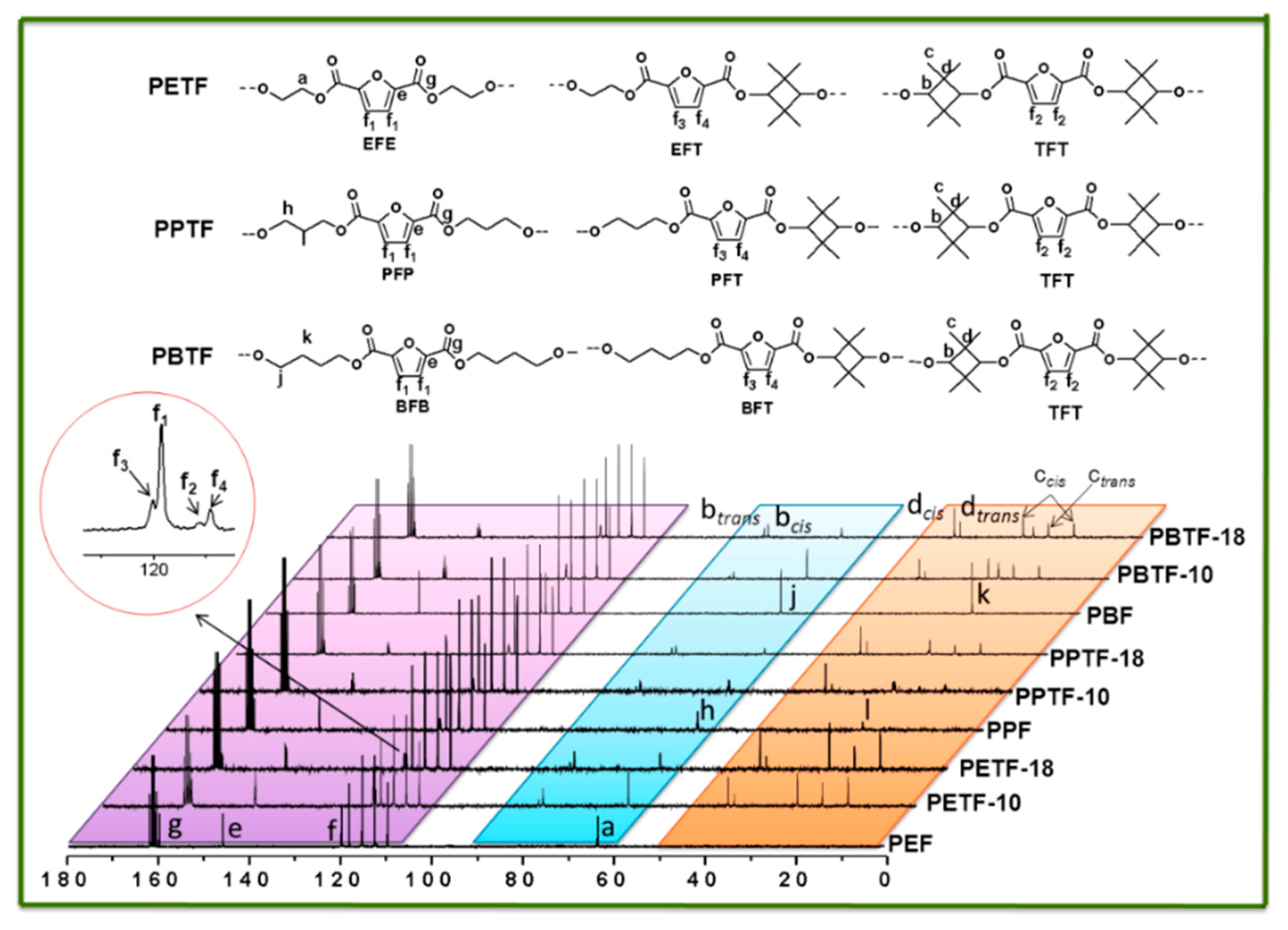
co-2,2,4,4-tetramethyl-1,3-cyclobutanediol 2,5-furandicarboxylate) (PBTF) with high molecular weight were synthesized. Their chemical structures and compositions were determined by
1H NMR and
13C NMR. The crystallization behaviors, mechanical properties, and optical and barrier properties of the copolyesters were investigated by DSC, TGA, tensile testing, UV–visible spectrophotometer and gas permeability tester. The objective of this paper was to provide us FDCA-based copolyesters with increased
Tg and good mechanical properties as well as better transparency without sacrificing their barrier properties severely. Based on which, the bio-based substitute for PET, PPT, and PBT with better comprehensive performance could be developed and used as packaging materials.
2. Materials and Methods
2.1. Materials
CBDO with the purity of 99% was purchased from Suzhou Yacoo Chemical Reagent Co., Ltd. (Suzhou, China). Ethanediol (EG, 99%), propanediol (PPD, 99%), and butanediol (BDO, 99%) were purchased from Aladdin Reagent Co. Ltd. (Shanghai, China). Antimony trioxide (III, 99.99%), zinc acetate (99%), trifluoroacetic acid, phenol, and tetrachloroethane were obtained from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). Bio-based 2,5-furandicarboxlic acid (99.9%) was purchased from Chemtarget Technologies Co., Ltd. (Mianyang, China). It is the oxidation product of 5-hydroxymethylfurfural (HMF), which is the important intermediate from fructose to FDCA. All the chemicals were used as received without any purification.
2.2. Synthesis of Dimethyl Furan-2,5-dicarboxylate (DMFD)
The synthesis of DMFD followed the procedures described in previous literature [
28]. Its chemical structure was confirmed by
1H NMR and
13C NMR before the polycondensation.
1H NMR (400 MHz, DMSO-d6, δ, ppm): 7.41 (2H), 3.88 (6H).
13C NMR (400 MHz, DMSO-d6, δ, ppm): 119.6, 146.7 (4C), 158.7 (2C), 52.9 (2C).
2.3. Synthesis of PEF/PPF/PBF and Their Copolyesters
The two-stage melt polycondensation reaction was taken to prepare PEF, PPF, PBF, PETF, PPTF, and PBTF from diol (EG, PPD, BDO), CBDO, and DMFD with the molar ratio of diol/DMFD was 1.6. All the reactions were conducted in a 500 mL three-necked round bottom flask equipped with a mechanical stirrer with torque indicator. The predetermined diol (EG, PPD, or BDO), CBDO, DMFD (0.50 mol) and catalyst (Zinc acetate, 0.2 mol % based on DMFD) were added into the flask. Then the reaction was conducted at 180 °C for 4 h under the protection of nitrogen. After almost all the theoretically produced methanol was distilled out, subsequently the second portion of catalyst was added (antimony trioxide (III), 0.15 mol·% based on DMFD). After that, the reaction temperature was increased to 230–245 °C and pressure of the reaction system was gradually reduced to 10–20 Pa for a certain time until the torque value of the stirrer reached a fixed value to ensure similar viscosity of all the products. Finally, the reaction system was returned to the normal atmospheric pressure by the introduction of N2 and the target product was obtained.
The synthetic condition and composition of different polyesters were listed in
Table 1. The different samples were named as PETF-10, PPTF-10, PBTF-10, PETF-18, PPTF-18, and PBTF-18, where the numbers 10 and 18 represent the mole percentage of CBDO in the copolyesters. For example, PPTF-10 stands for the sample in which the mole ratio of CBDO/(CBDO + PPD) in the copolyesters was about 0.1.
2.4. Measurements
The viscosity of synthesized copolyesters was measured by Ubbelohde viscometer in a 25 °C water bath. The diameter of the capillary tube was 0.792 mm. The phenol and tetrachloroethane mixture (1/1,
w/w) was used as the solvent with the concentration of copolyester was 0.125 g/25 mL. The equation [η] = η
sp/
c was used to calculate the intrinsic viscosity [η], where η
sp = (
t1 −
t0)/
t0,
t0 is the elution time of solvent and
t1 the elution time of polyesters solutions [
5].
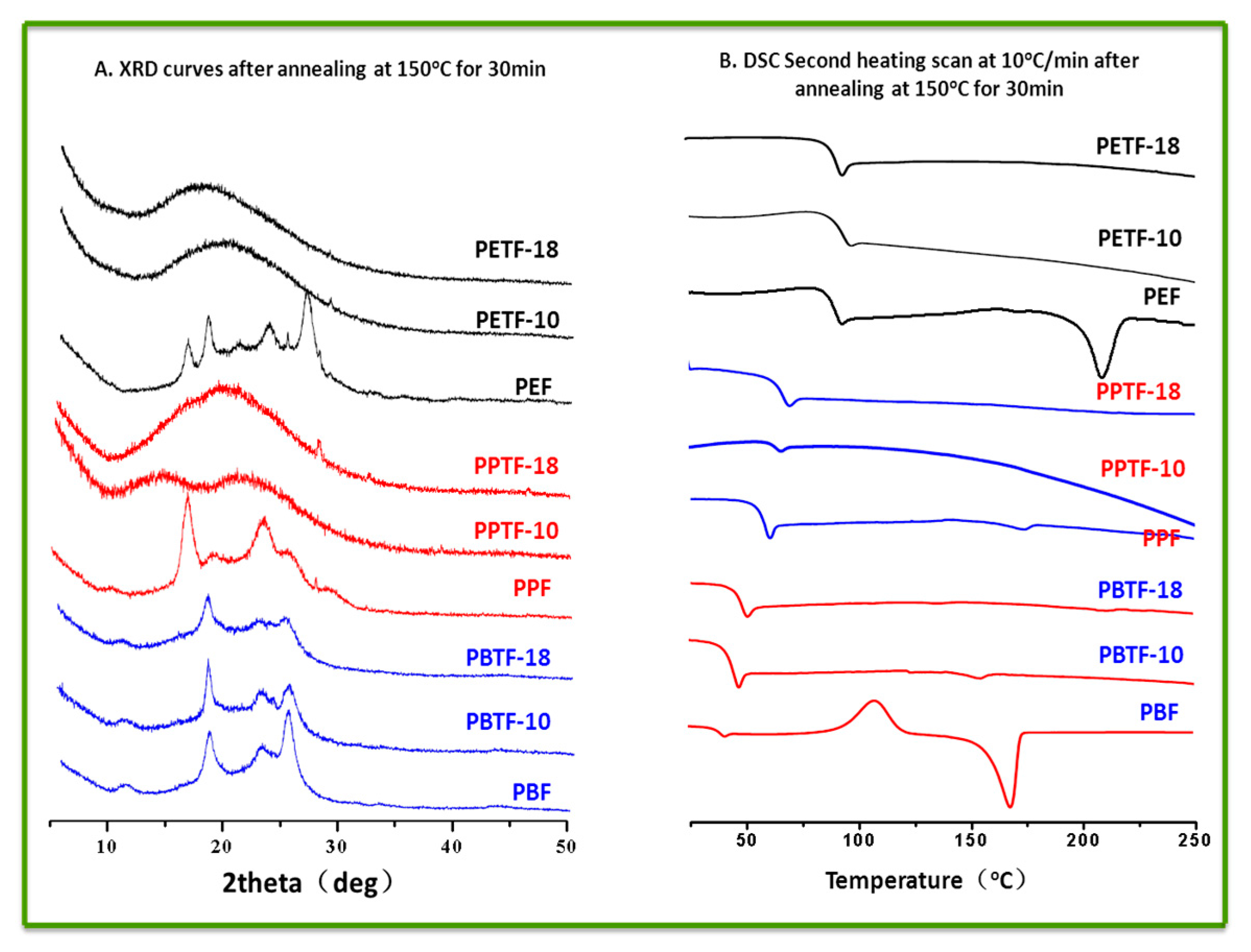
Chemical structures, compositions, and sequence distribution of the synthesized copolyesters were determined by 1H NMR and 13C NMR in CF3COOD using a Bruker AVIII400 NMR spectrometer (Karlsruhe, Germany) at 25 °C. Tetramethysilane (TMS) was used as the internal standard. DSC measurements were performed on a differential scanning calorimeter (Mettler-Toledo DSC I, Zurich, Switzerland) under the nitrogen flow rate of 50 mL/min. Before the measurement, it was calibrated with pure indium and zinc standards to ensure accuracy. A sample of about 5–6 mg was placed in an alumina pan before it was heated to 250 °C with the heating rate of 10 °C/min. All the samples should be maintained at this temperature for 3 min to erase thermal history. Then it was cooled down to room temperature at a rate of 10 °C/min and heated to 250 °C with the same heating rate for the second time. All the DSC curves were recorded for thermal properties analysis. Thermal stability evaluation was conducted using a TGA instrument (Mettler-Toledo TGA/DSC thermogravimetric analysis, Zurich, Switzerland). For each measurement, 6–10 mg sample was placed in a ceramic furnace and the TGA curves under nitrogen atmosphere were recorded from 50 to 800 °C for thermal stability analysis. The X-ray diffraction (XRD) patterns were collected on a Bruker D8 ADVANCE, using Cu-Kα radiation (40 kv, 40 mA) with a scanning range from 5° to 50°. The scanning speed was 0.2°/s. Ultraviolet–visible (UV–vis) spectra of the polyester films were recorded on a Lambda 950 UV/Vis/NIR spectrophotometer (Waltham, MA, USA). Tensile testing was performed on an Instron 5567 tensile machine (Norwood, MA, USA) with a 500 N load cell. The crosshead speed was 10 mm/min. The dumbbell-shaped samples were prepared by hot press-molding at 230–240 °C with the dimension of 20.0 mm in length, 2.0 mm in neck width and 1.0 mm in thickness. Before testing, all the samples were conditioned at 25 °C for over 48 h. For accuracy, at least five specimens were tested for each sample.
The barrier properties were measured by Labthink VAC-V2 gas permeability tester (Jinan, China). The experiment was conducted at 30 °C with the relative humidity (RH) of 50%. The oxygen and carbon dioxide with the purity of 99.99% were used in our experiment. The completely amorphous polyesters films were prepared following the melt-press/quench procedures. The round films with the diameter of 97 mm and the surface area of 38.5 cm2 were used and the testing range is 0.05–50,000 cm3/m2·24 h·0.1 MPa.
4. Conclusions
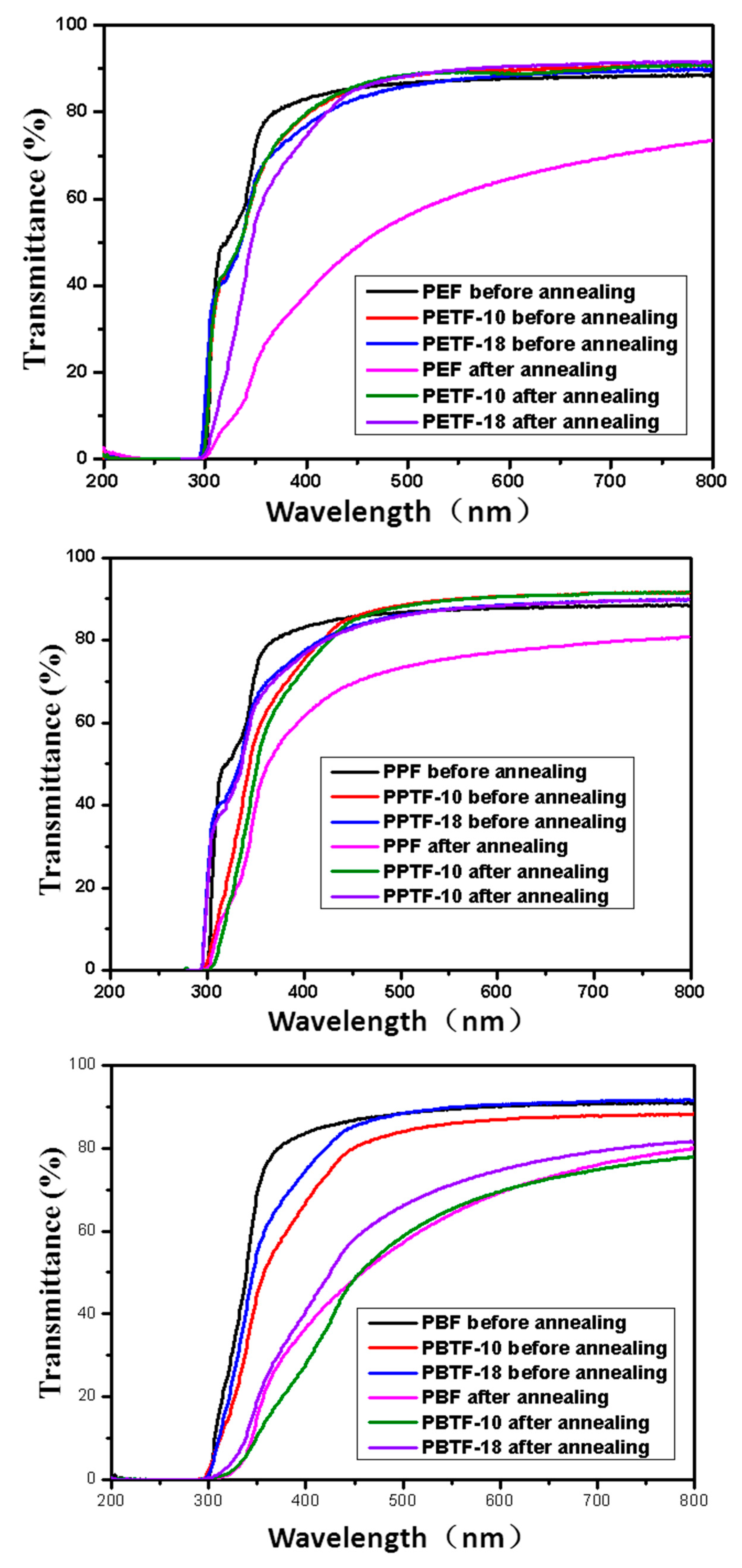
FDCA-based polyesters—including PEF, PPF, as well as PBF—And their copolyesters modified with CBDO were synthesized successfully. All the copolyesters showed the degree of random close to 1, indicating their random structures. The introduction of CBDO units led to the increased glass transition temperature, tensile modulus, and strength as well as decreased crystallizability. When the content of CBDO reached 10%, the semi-crystalline PEF and PPF were became completely amorphous (PETF-10, PETF-18, PPTF-10, and PPTF-18) and then high transparency was easily achieved. Although the barrier properties to O2 and CO2 of PEF, PPF, and PBF were all decreased with the introduction of CBDO, the modified copolyesters containing 18% CBDO still showed better barrier properties to CO2 when compared with PEN, and the ones containing 10% CBDO demonstrated better barrier properties to O2. This paper provided us FDCA-based copolyesters with increased Tg, better mechanical properties, and higher transparency as well as good barrier properties. Based on which, the bio-based substitutes for PET, PPT, and PBT with better performance could be developed, especially when used as packaging materials.