1. Introduction
In recent years, the increasing shortage of resources and the consequential sustainable awareness has led to a change in mindset of chemists towards bio-based and renewable resources. The development of enzymatic polymer synthesis [
1], multifunctional high performance fibers [
2], membranes for water purification [
3], transparent substrates for use in electronics from wood [
4,
5] , amino-acid based ionic liquids [
6], and asymmetric building blocks [
7] are results of this rethinking process. Another approach towards sustainability involves the utilization of bio-based by-products. Glycerol is a readily available, major by-product in the production of biodiesel, and thus used as a starting material for the synthesis of monomers, such as epichlorohydrin and glycidol [
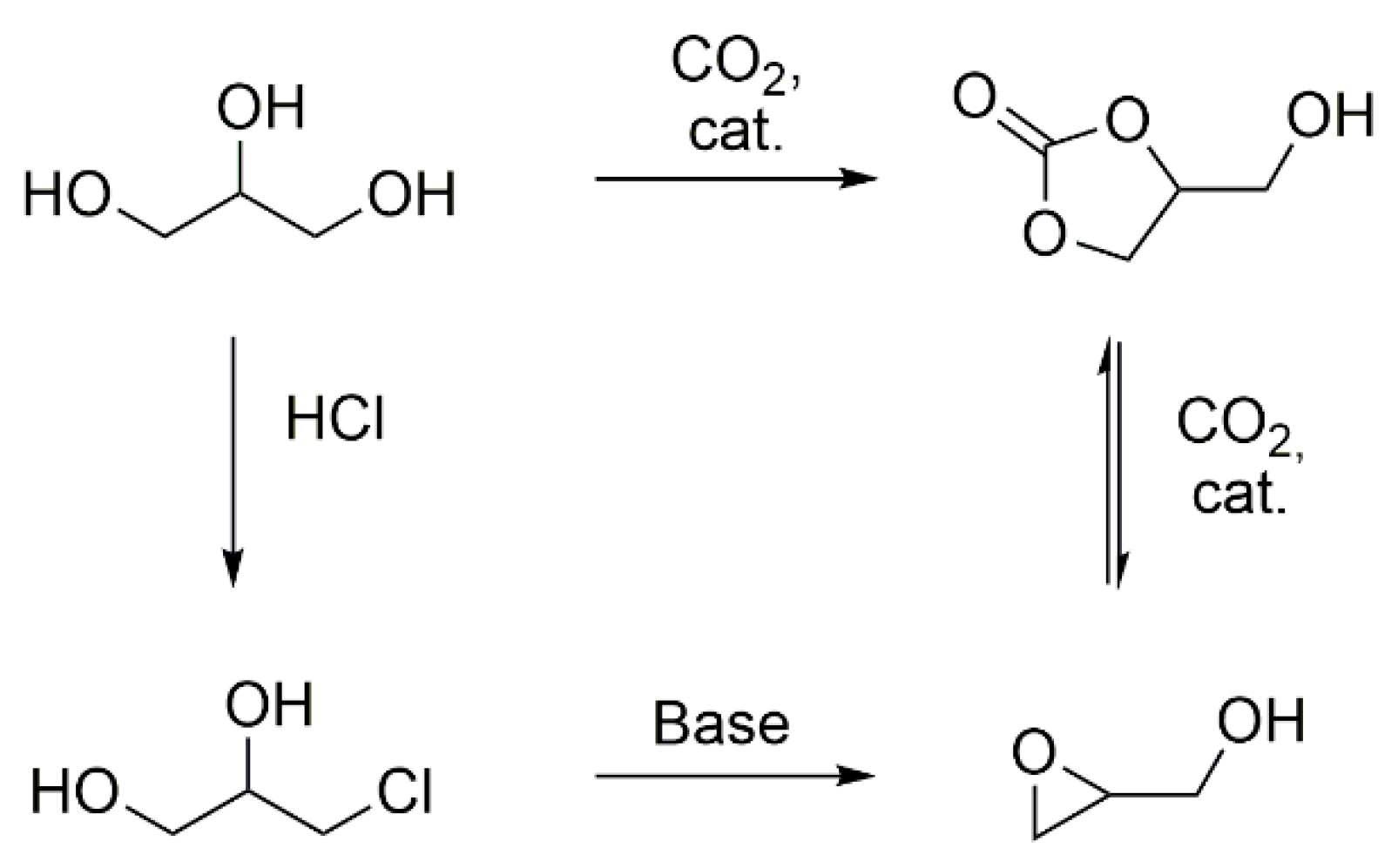
8], or building blocks like glycerol carbonate (
Scheme 1) [
9].
Glycidol acts as the monomer in the synthesis of linear, branched and star-like polyglycidols [
10,
11,
12]. Polyglycidol is soluble in aqueous media, shows no toxicity towards cells and is licensed by the Food and Drug Administration (FDA) [
13,
14]. It is a highly functionalized polymer with a hydroxyl group in every repeating unit, allowing for various further modifications [
15,
16]. Hans et al. presented the application of polyglycidol, as a multifunctional macroinitiator, for the ring-opening polymerization of ɛ-caprolactone [
17,
18]. The reaction was carried out by chemical and enzymatic catalysis, leading to densely and loosely grafted polyglycidols, respectively. The loosely grafted polymers showed enhanced hydrolytic degradation of the poly(caprolactone) side chains allocated to the free hydroxyl groups of the polyglycidol backbone.
The hydrolytic degradation of poly(caprolactone) was further increased by functionalization of the polyglycidol macroinitiator with phosphonate/phosphonic acid groups [
19,
20]. Polyglycidol was functionalized in a Michael-type addition with diethylvinyl phosphonate and subsequent dealkylation of the pendant phosphonate groups [
21]. Additionally, multifunctional polyglycidols carrying phosphonic acid and acrylate moieties were examined as UV-active adhesion promoters for a hydrogel coating on stainless steel wires [
22].
Recently, we developed a synthetic strategy for the post-polymerization functionalization of polyglycidol with pendant phosphate groups by reaction with diethyl chlorophosphate [
23]. The diethyl phosphate moieties were subsequently (mono-)dealkylated. This method allowed for the tailoring of the concentration of pendant phosphate/phosphoric acid groups introduced into the polyglycidol.
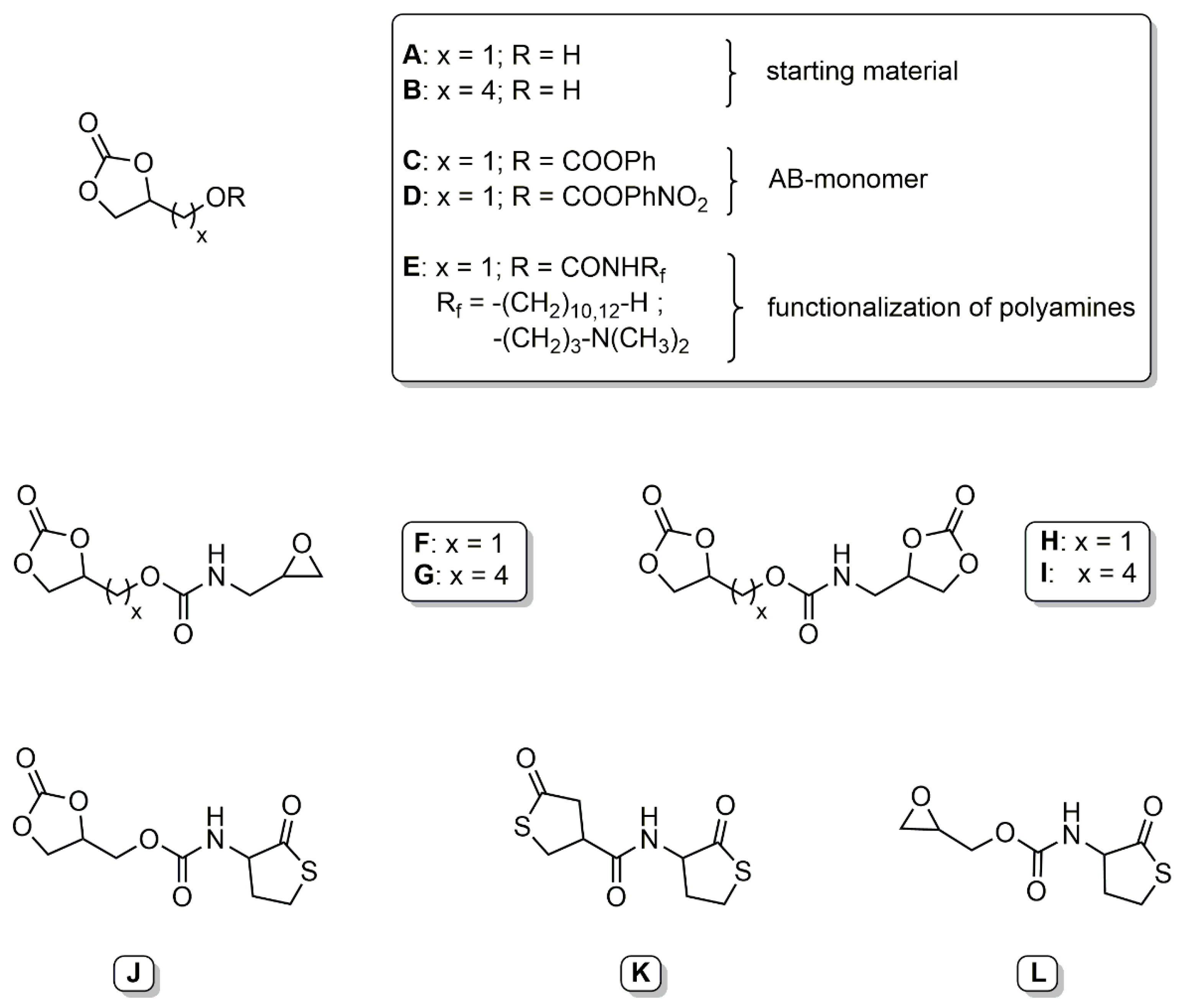
Glycerol carbonate has been used as starting material in the synthesis of various bifunctional couplers. Ubaghs et al. developed a (2-oxo-1,3-dioxolan-4-yl)methyl phenyl carbonate (
Scheme 2,
C) as an AB-type monomer for the isocyanate-free synthesis of poly(urethane)s with pendant hydroxyl groups [
24]. This carbonate was used later by Pasquier et al. as a coupling agent to link functional poly- or monodisperse building blocks [
25]. These couplers are selectively reacted with an amine at the phenyl carbonate moiety at room temperature, followed by ring-opening at the ethylene carbonate at 60 °C. The glycerol carbonate was functionalized with various cationic and hydrophobic amines and linked to a hyperbranched poly(ethylene imine) to determine the antimicrobial properties in aqueous solution and as surface coatings (
Scheme 2,
E) [
25,
26]. Fricke et al. further developed this concept by synthesizing more reactive 4-nitrophenyl-carbonate couplers (
Scheme 2,
D). These couplers were used to introduce a cyclic carbonate moiety to various amino acid derivatives and glucosamine [
27].
He et al. presented two new types of bifunctional couplers based on glycerol carbonate [
28]. The first series of couplers carried a five-membered cyclic carbonate and an epoxy group (
Scheme 2,
F and
G). These coupling agents showed a low selectivity towards primary amines and a very high selectivity towards secondary amines. Introduction of an alkyl chain as a spacer shifted the selectivity towards the epoxide. The second series of couplers carried two five-membered cyclic carbonate rings (
Scheme 2,
H and
I). The carbonate groups showed different reactivity based on the distance of the carbonate from the linking functional group.
Recently, we presented various homocysteine thiolactone-functional couplers. The homocysteine thiolactone moiety has been thoroughly investigated in regard to its reactivity with amines and subsequent thiol-ene reactions [
29,
30]. A bifunctional coupler with an ethylene carbonate and a thiolactone functionality was synthesized starting from either (2-oxo-1,3-dioxolan-4-yl)methyl phenyl carbonate or glycerol chloroformate (
Scheme 2,
J) [
31,
32]. Both functional groups were selectively addressed by an amine, dependent on the temperature, allowing the addition of two different amines. The released thiol was further reacted with methyl acrylate in a Michael-type addition to give the corresponding thioether. The released hydroxyl groups were addressed with an acyl halide. Additionally, a (bis)thiolactone coupler was prepared by reaction of γ-thiobutyrolactone β-acid chloride with homocysteine thiolactone (
Scheme 2,
K) [
33]. The (bis)thiolactone showed no regioselective ring-opening of the thiolactone under the specific reaction conditions. Reaction with two different diamines gave thiol-functional polyamides. A novel epoxy thiolactone coupler was presented as a versatile tool for polymeric materials (
Scheme 2,
L) [
34]. The coupler was used in multicomponent reactions to link up to four different buildings blocks and was converted with amines to poly(thioether urethane)s. Functional gels were generated by either thiol-epoxy polymerization under basic conditions or multicomponent reaction with a triacrylate.
Herein we present two different strategies for the introduction of homoserine lactone into polymeric materials: (i) Synthesis of a thiolactone-lactone coupler; determination of the selectivity of both rings towards amines and use of the coupler for the preparation of polymeric building blocks; (ii) Functionalization of polyglycidol with homoserine lactone; ring-opening of the lactone with an amine and investigation of further modification possibilities.
2. Materials and Methods
2.1. Materials
Thioacetic acid (96%, Sigma Aldrich Chemie GmbH, Steinheim, Germany), itaconic acid (99%, Alfa Aesar, Karlsruhe, Germany), thionyl chloride (99.7%, Thermo Fisher Acros Organics, Geel, Belgium), hexylamine (99%, Sigma Aldrich Chemie GmbH, Steinheim, Germany), methyl acrylate (99%, Sigma Aldrich Chemie GmbH, Steinheim, Germany), poly(ethylene glycol) diamine (M = 368.47 g·mol−1, Iris Biotech GmbH, Marktredwitz, Germany), potassium tert-butoxide (1 M solution in THF, Sigma Aldrich Chemie GmbH, Steinheim, Germany), diglyme (≥99%, extra dry, over molecular sieves, Thermo Fisher Acros Organics, Geel, Belgium), pyridine (99.5%, extra dry, over molecular sieves, Thermo Fisher Acros Organics, Geel, Belgium), 4-nitrophenyl chloroformate (>98%, TCI), 3-(dimethylamino)-1-propylamine (99%, Thermo Fisher Acros Organics, Geel, Belgium), DL-homoserine lactone hydrobromide (99%, Sigma Aldrich Chemie GmbH, Steinheim, Germany), 4-dimethylaminopyridine (>98%, Sigma Aldrich Chemie GmbH, Steinheim, Germany), triethylamine (≥99.5%, anhydrous, Sigma Aldrich Chemie GmbH, Steinheim, Germany), methyl iodide (>99%, Sigma Aldrich Chemie GmbH, Steinheim, Germany), tetrahydrofuran (99.8%, stabilizer free, extra dry, Thermo Fisher Acros Organics, Geel, Belgium), chloroform (99.9%, extra dry, over molecular sieves, Thermo Fisher Acros Organics, Geel, Belgium), N,N-dimethylformamide (99.8%, extra dry, over molecular sieves, Thermo Fisher Acros Organics, Geel, Belgium), methanol (≥99.8%, p.a., Th. Geyer CHEMSOLUTE®, Renningen, Germany) and dichloromethane (≥99.8%, anhydrous, Sigma Aldrich Chemie GmbH, Steinheim, Germany) were used as received.
3-Phenyl-1-propanol (99%, Sigma Aldrich Chemie GmbH, Steinheim, Germany) was stirred with calcium hydride for 24 h and then distilled. Ethoxyethyl glycidyl ether (EEGE) was synthesized from 2,3-epoxypropan-1-ol (glycidol) and ethyl vinyl ether according to Fitton et al. [
35], purified by distillation, and stored under a nitrogen atmosphere over a molecular sieve (3 Å).
Water-sensitive reactions were carried out in a nitrogen atmosphere. Nitrogen (Linde 5.0) was passed over a molecular sieve (4 Å) and finely distributed potassium on aluminum oxide.
2.2. Measurements
1H NMR and 13C NMR spectra were recorded on a Bruker DPX-400 FT-NMR spectrometer (Bruker Corporation, Billerica, MA, USA) at 400 and 101 MHz, respectively. Deuterated dimethyl sulfoxide (DMSO-d6), deuterated dimethylformamide (DMF-d7) and deuterium oxide (D2O) were used as solvents. The residual solvent signal was used as the internal standard. Coupling constants Jxy are given in Hz.
Molecular weights (Mn,SEC) and molecular weight distributions (Đ) were determined by size exclusion chromatography (SEC). SEC analyses were carried out with DMF (HPLC grade, VWR) as the eluent. SEC was performed using an Agilent 1100 system (Agilent Technologies, Santa Clara, CA, USA) equipped with a dual RI-/Visco detector (ETA-2020, WGE Dr. Bures GmbH & Co KG, Dallgow-Doeberitz, Germany). The eluent contained 1 g·L−1 LiBr (≥99%, Aldrich). The sample solvent contained traces of distilled water as the internal standard. One pre-column (8 mm × 50 mm) and four GRAM gel columns (8 mm × 300 mm, Polymer Standards Service) were applied at a flow rate of 1.0 mL·min−1 at 40 °C. The diameter of the gel particles measured 10 µm, the nominal pore widths were 30, 100, 1000 and 3000 Å. Calibration was achieved using narrowly distributed poly(methyl methacrylate) standards (Polymer Standards Service). Results were evaluated using the PSS WinGPC UniChrom software (Version 8.1.1, PSS Polymer Standards Service GmbH, Mainz, Germany).
FTIR spectra were recorded on a Thermo Nicolet Nexus 470 FTIR spectrometer at 25 °C. The samples were recorded between KBr disks and scanned over a range of 400–4000 cm−1.
ESI mass spectra were recorded on a Finnigan SSQ 7000 spectrometer (Thermo Scientific (Finnigan Corporation), Waltham, MA, USA) and HRMS spectra on a Thermo Scientific LTQ Orbitrap XL spectrometer (Thermo Scientific, Waltham, MA, USA).
NALDI-TOF mass spectrometry was performed on a Bruker ultrafleXtreme (Bruker Corporation, Billerica, MA, USA) equipped with a 337 nm smartbeam laser in the reflective mode. Tetrahydrofuran (THF) solutions of analyte (20 μL of 10 mg·mL−1) were prepared, 2 μL thereof were applied on the sample plate. Laser shots (6000) with 24% up to 60% laser power were collected. The laser repetition rate was 1000 Hz.
Elemental analysis was performed on an Elementar Vario EL (Elementar Analysensysteme GmbH, Langenselbold, Germany). Results are reported in weight percent.
Dialysis was performed in methanol using Biotech CE Tubing (MWCO: 100–500 D, 3.1 mL·cm−1, Spectrum Laboratories, Inc., Rancho Dominguez, CA, USA). The membrane was washed for 15 min in water before use to remove the sodium azide solution.
2.3. Synthesis of the Thiolactone-Homoserine Lactone Coupler (1)
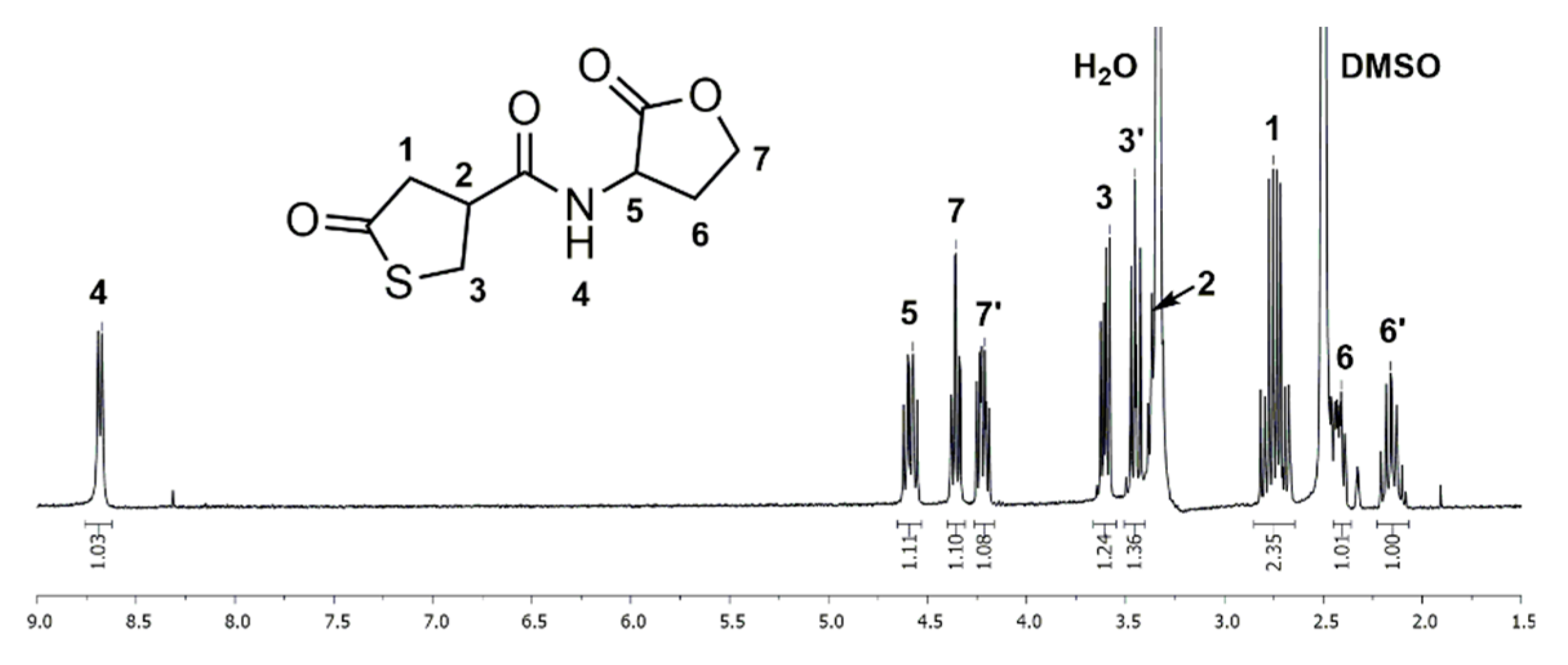
To an ice-cold mixture of γ-thiobutyrolactone β-acid chloride (1.788 g, 10.86 mmol) and DL-homoserine lactone hydrobromide (2.000 g, 10.86 mmol), pyridine (8.74 mL) was slowly added and stirred for 16 h at room temperature. Excess pyridine was removed under reduced pressure. The crude product was purified by flash column chromatography on silica gel using DCM/MeOH (95:5) as the eluent. The coupler 1 was obtained as a slightly yellow solid (1.80 g, 82%). Rf = 0.15, Tm = 157.0 °C. 1H NMR (400 MHz, DMSO-d6): δ = 2.10–2.22 (m, 1H, NCHCH2), 2.38–2.46 (m, 1H, NCHCH2), 2.65–2.84 (m, 2H, SC=OCH2), 3.31–3.40 (m, 1H, SCH2CH), 3.41–3.51 (m, 1H, SCH2CH), 3.57–3.67 (m, 1H, SCH2CH), 4.17–4.26 (m, 1H, OCH2), 4.31–4.39 (m, 1H, OCH2), 4.54–4.64 (m, 1H, NCH), 8.68 (d, J = 7.8 Hz, 1H, NH) ppm. 13C NMR (100 MHz, DMSO-d6): δ = 28.1 (NCHCH2), 34.8 (SCH2), 42.8 (SCH2CH), 43.5 (SC=OCH2), 48.2 (NCH), 65.3 (OCH2), 170.7 (NC=O), 175.0 (OC=O), 206.5 (SC=O) ppm. IR (KBr): υmax = 3371 (m), 3312 (s), 3083 (w), 2929 (w), 1790 (m), 1771 (s), 1700 (s), 1652 (vs), 1551 (s), 1372 (w), 1284 (w) 1161 (m), 1022 (m), 985 (w), 654 (w). m/z for C9H11NO4S: measured: (M + Cl)− 264.017/269.012, (M + Cl)− calculated: 264.01. Elemental analysis: C9H11NO4S (M = 229.25 g·mol−1), measured: C: 46.83, H: 4.82, N: 5.73, calculated: C: 47.15, H: 4.84, N: 6.11.
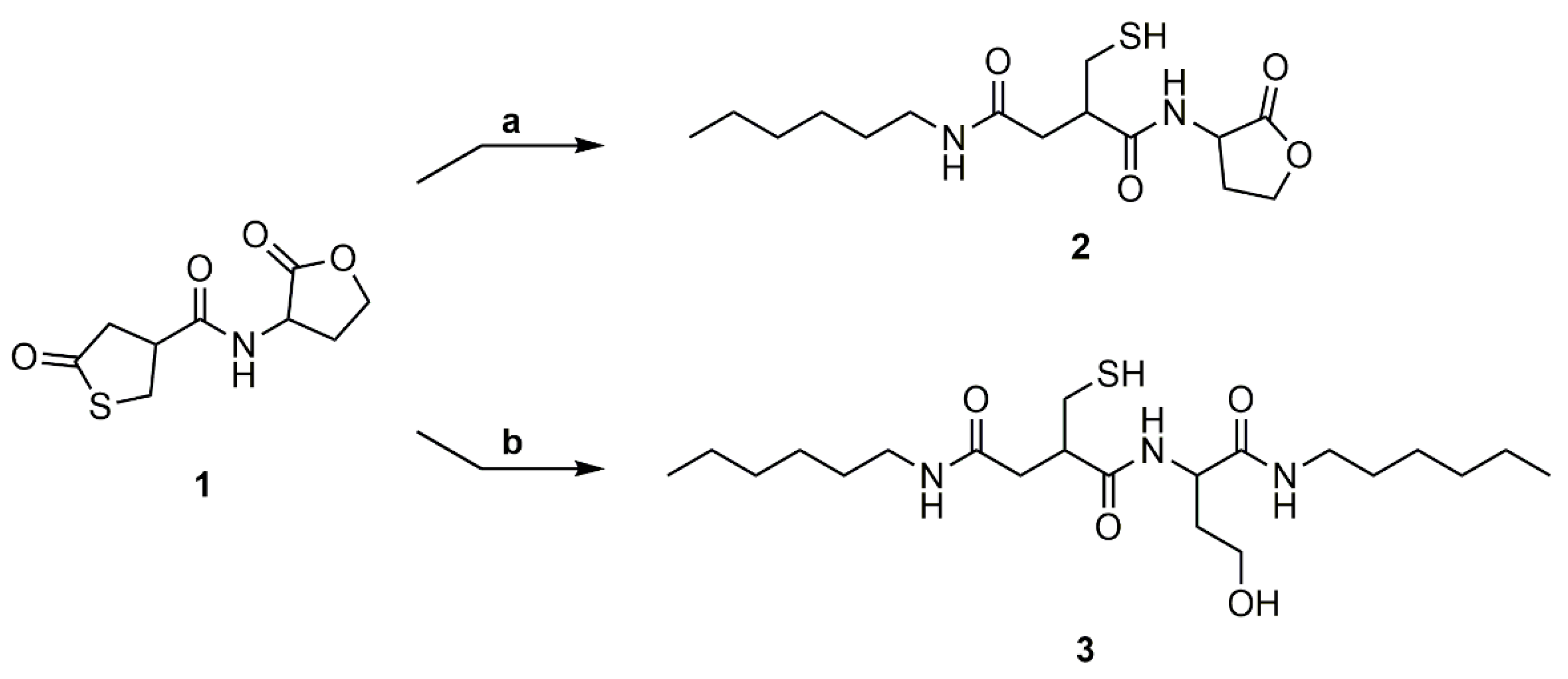
2.4. Conversion of Homoserine Lactone with Hexylamine in a 1:1 Molar Ratio
Hexylamine (115 μL, 0.877 mmol) was added to a stirred solution of the coupler 1 (201 mg, 0.877 mmol) in dry DMF (1.75 mL, 0.5 M) at 0 °C. The reaction mixture was allowed to warm slowly to room temperature and stirred at room temperature for 16 h. The solvent was evaporated under reduced pressure to give the crude product. Purification by flash column chromatography on silica gel using DCM/MeOH (95:5) as the eluent gave the addition product 2 (240 mg, 83%) as a waxy solid. Rf = 0.19. 1H NMR (400 MHz, DMSO-d6): δ = 0.85 (t, J = 6.3 Hz, 3H, CH3CH2CH2), 1.18–1.30 (m, 6H, CH3CH2CH2CH2CH2), 1.31–1.41 (m, 2H, CH2CH2NH), 2.06–2.44 (m, 4H, CH2CHCH2SH, NHCHCH2), 2.53–2.65 (m, 2H, CH2CHCH2SH) 2.75–2.85 (m, 1H, CH2CHCH2SH), 2.95–3.09 (m, 2H, CH2CH2NH), 4.14–4.25 (m, 1H, NHCHCH2CH2), 4.28–4.40 (m, 1H, NHCHCH2CH2), 4.41–4.62 (m, 1H, NHCHCH2), 7.79–7.91 (m, 1H, NH), 8.44–8.61 (m, 1H, NH) ppm. 13C NMR (100 MHz, DMSO-d6): δ = 13.9 (CH3CH2CH2), 22.1 (CH2CHCH2SH), 25.9 (CH3CH2CH2), 26.1 (CH3CH2CH2CH2CH2), 28.1 (CH3CH2CH2CH2CH2), 29.0 (NHCHCH2), 31.0 (CH3CH2CH2CH2), 37.0 (CH2CH2NH), 38.6 (CH2CHCH2SH), 45.1 (CH2CHCH2SH), 54.9 (NHCHCH2), 65.3 (NHCHCH2CH2), 169.7 (NHC=O), 172.7 (C=ONH), 175.3 (OC=O) ppm. m/z for C15H26N2O4S: measured: (M + Na)+ 353.128, calculated: (M + Na)+ 353.15.
2.5. Conversion of Homoserine Lactone with Hexylamine in a 1:2 Molar Ratio
Hexylamine (235 μL, 1.79 mmol) was added to a stirred solution of the coupler 1 (205 mg, 0.894 mmol) in dry DMF (1.79 mL, 0.5 M) at 40 °C. The reaction mixture was stirred at 40 °C for 20 h. The solvent was evaporated under reduced pressure to give the crude product. Purification by flash column chromatography on silica gel using DCM/MeOH (95:5) as the eluent gave the addition product 3 (159 mg, 34%) as a waxy solid. Rf = 0.24. 1H NMR (400 MHz, DMSO-d6): δ = 0.85 (t, J = 6.8 Hz, 6H, CH3CH2CH2), 1.14–1.30 (m, 12H, CH3CH2CH2CH2CH2), 1.31–1.47 (m, 4H, CH2CH2NH), 1.54–1.72 (m, CHCH2CH2OH), 1.78–2.05 (m, 1H, CHCH2CH2OH), 2.16 (t, J = 8.0 Hz, 1H, SH) 2.23–2.44 (m, 2H, CH2CHCH2SH), 2.52–2.63 (m, 2H, CH2CHCH2SH), 2.76–2.86 (m, 1H, CH2CHCH2SH), 2.92–3.14 (m, 4H, CH2CH2NH), 3.37–3.52 (m, 2H, CHCH2CH2OH), 4.14–4.24 (m, 1H, CHCH2CH2OH), 4.38–4.49 (m, 1H, OH), 7.79–8.08 (m, 3H, NH) ppm. 13C NMR (100 MHz, DMSO-d6): δ = 13.9 (CH3CH2CH2), 22.0 (CH2CHCH2SH), 25.9 (CH3CH2CH2), 29.0 (CH3CH2CH2CH2CH2), 30.9 (CH3CH2CH2CH2), 34.3 (CH3CH2CH2CH2CH2), 34.8 (CH2CH2NH), 36.9 (CHCH2CH2OH), 38.5 (CH2CHCH2SH), 50.1 (CH2CHCH2SH), 54.6 (CHCH2CH2OH), 57.8 (CHCH2CH2OH), 171.5 (CHC=ONHCH2), 172.5 (NHC=OCH2), 173.0 (CHC=ONHCH) ppm. m/z for C21H45N3O4S: measured: (M + Na)+ 454.268, calculated: (M + Na)+ 454.27.
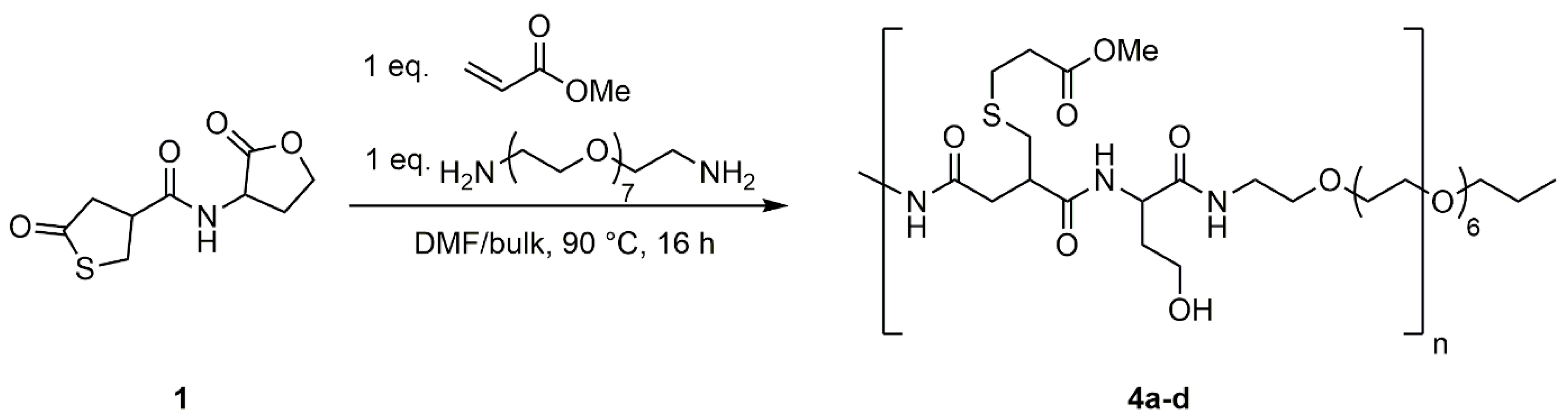
2.6. General Procedure for the Polyaddition of PEG-Diamine to Homoserine Lactone in the Presence of Methyl Acrylate
Methyl acrylate (1 eq.) and PEG-diamine (1 eq.) were added to a solution of coupler
1 (1 eq.) in DMF. The solution was stirred for 16 h at 90 °C. The solvent was removed under reduced pressure and the crude product was obtained as a yellow oil.
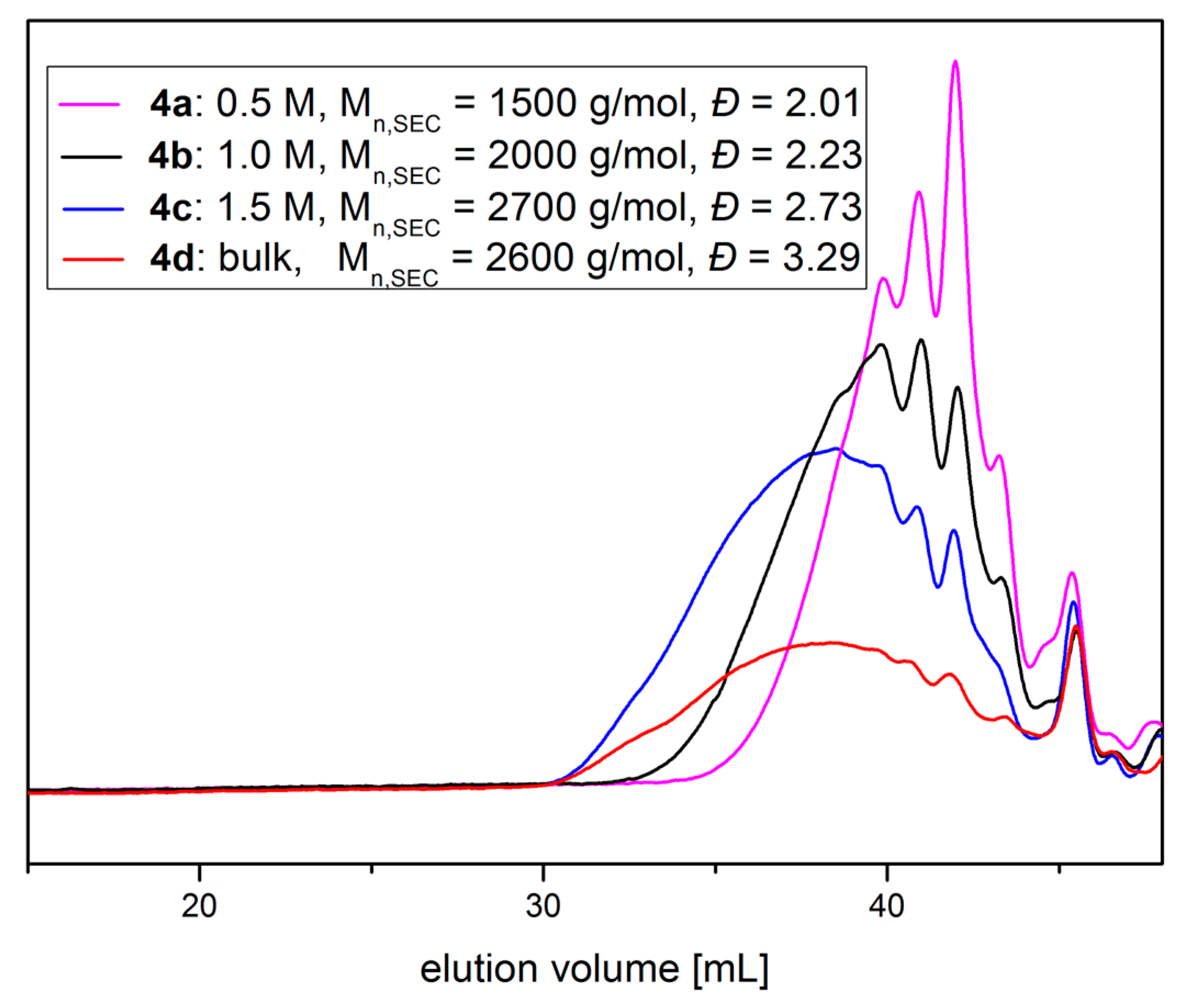
4a–
c were synthesized using DMF solutions of different concentrations.
4d was synthesized in bulk. Detailed information can be found in the supporting information (
Table S3). As the polyaddition procedure does not require any purification of the product, the product can be considered as obtained in quantitative yield.
1H NMR (400 MHz, DMSO-d6): δ = 1.54–1.91 (m, NHCHCH2CH2OH), 2.06–2.75 (m, CH2CHCH2SCH2CH2), 2.97–3.10 (m, CH2CHCH2SCH2CH2), 3.12–3.27 (m, CH2CHCH2SCH2CH2), 3.28–3.43 (m, C=ONHCH2CH2OCH2), 3.44–3.57 (m, NHCH2CH2OCH2CH2OCH2), 3.58–3.72 (m, NHCHCH2CH2OH, SCH2CH2C=OOCH3), 4.12–4.57 (m, NHCHCH2CH2OH), 7.66–8.67 (m, NH) ppm.
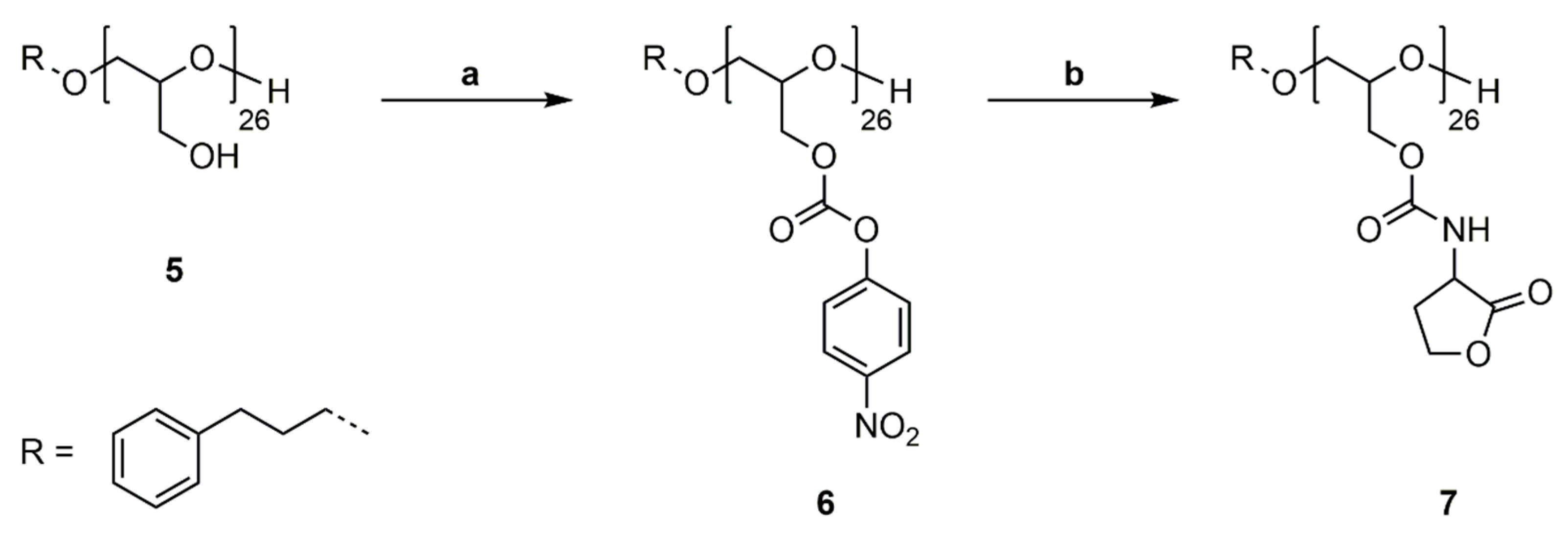
2.7. Synthesis of Poly(Glycidyl-4-Nitrophenylcarbonate)26 (P(GNPC)26) (6)
PG26 (5) (0.990 g, 13.36 mmol OH) was dissolved in pyridine (9.28 mL). 4-Nitrophenyl chloroformate (2.963 g, 14.70 mmol) was dissolved in DCM (9.0 mL) and added to the polymer solution via syringe pump in 30 min at 0 °C. The solution was stirred for 20 h at room temperature. The crude product was washed with water (30 mL), 1 M HCl (aq.) (3 × 30 mL) and saturated NaCl solution (aq., 30 mL). The organic phase was separated, dried over Na2SO4 and precipitated in cold MeOH. The solvent was removed under reduced pressure and polymer 6 was obtained as a colorless solid (2.922 g, 91%). 1H-NMR (400 MHz, DMSO-d6): δ = 1.69–1.83 (m, ArCH2CH2), 2.54–2.61 (m, ArCH2CH2), 3.68–3.72 (m, ArCH2CH2CH2, OCH2CH(CH2OC=OOArNO2)O), 4,26–4.42 (d, CH2OC=OOArNO2), 7.12–7.21 (m, ArCH2CH2), 7.41 (s, CH2OC=OOArNO2), 8.17 (s, CH2OC=OOArNO2) ppm. 13C NMR (100 MHz, DMSO-d6): δ = 30.8 (ArCH2CH2), 31.6 (ArCH2), 68.3 (OCH2CH(CH2OC=OOArNO2)O), 76.4 (OCH2CH(CH2OC=OOArNO2)O), 76.5 (ArCH2CH2CH2), 76.6 (OCH2CH(CH2OC=OOArNO2)O), 122.3 (CH2OC=OOArNO2), 125.2 (CH2OC=OOArNO2), 125.7 (ArCH2), 128.2 (ArCH2), 128.3 (ArCH2), 141.6 (ArCH2), 145.0 (CH2OC=OOArNO2), 152.0 (OC=OO), 155.1 (CH2OC=OOArNO2) ppm.
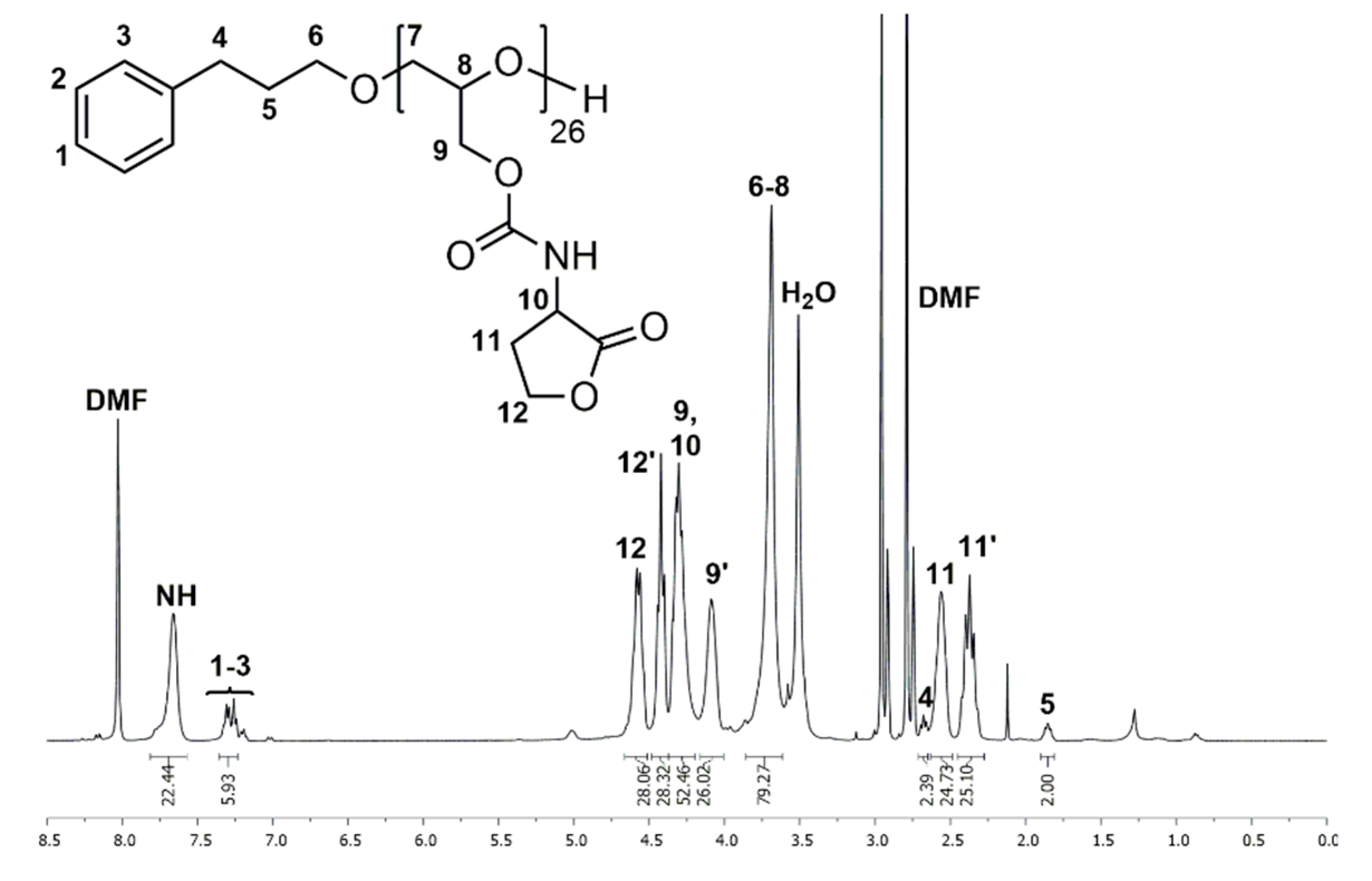
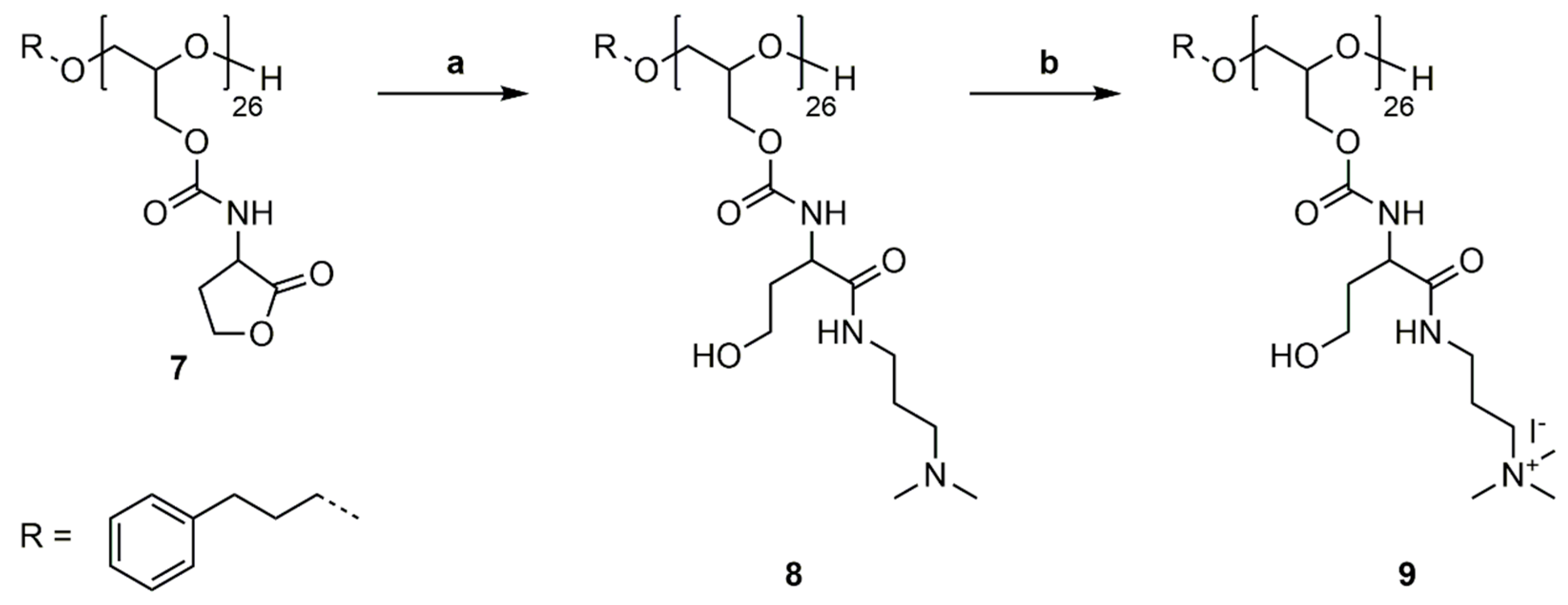
2.8. Synthesis of Poly(Glycidyl Homoserine Lactonylcarbamate)26 (P(GHSL)26) (7)
Polymer 6 (1.000 g, 4.181 mmol OH) was dissolved in DMF (20 mL), and 4-(dimethylamino)pyridine (0.051 g, 0.418 mmol) and DL-homoserine lactone hydrobromide (0.761 g, 4.181 mmol) were added. The mixture was cooled to 0 °C and triethylamine (0.846 g, 8.361 mmol) was added over 1 h via syringe pump. The solution was stirred for 20 h at room temperature. DMF was removed under reduced pressure at 50 °C and the crude product was precipitated in MeOH. Drying under reduced pressure at 50 °C gave polymer 7 as a colorless solid (0.648 g, 77%). 1H NMR (400 MHz, DMF-d7): δ = 1.81–1.90 (m, ArCH2CH2), 2.27–2.45 (m, NHCHCH2), 2.49–2.63 (m, NHCHCH2), 2.65–2.71 (m, ArCH2CH2), 3.61–3.86 (m, ArCH2CH2CH2, OCH2CH(CH2OC=OR)O), 4.09 (s, OCH2CH(CH2OC=OR)O), 4.20–4.37 (m, NHCHCH2, OCH2CH(CH2OC=OR)O), 4.37–4.49 (m, NHCHCH2CH2), 4.51–4.66 (m, NHCHCH2CH2), 7.23–7.36 (m, ArCH2CH2), 7.66 (s, NH) ppm. 13C NMR (100 MHz, DMF-d7): δ = 28.7 (NHCHCH2), 31.6 (ArCH2CH2), 32.2 (ArCH2), 50.3 (NHCHCH2), 65.7 (NHCHCH2CH2), 64.7, 69.5, 78.0 (ArCH2CH2CH2, OCH2CH(CH2OR)O), 128.7 (ArCH2), 128.8 (ArCH2), 156.6 (OC=ONH), 175.8 (NHCHC=OO) ppm.
2.9. Synthesis of P(GHSL,o)26 (8)
Polymer 7 (0.263 g, 1.307 mmol HSL) was dissolved in DMF (4 mL), and 3-(dimethylamino)-1-propylamine (0.339 g, 3.268 mmol) and 4-(dimethylamino)pyridine (0.016 g, 0.131 mmol) were added. The mixture was stirred for 20 h at room temperature. DMF was removed under reduced pressure at 50 °C and the crude product was precipitated in diethyl ether. Dialysis in MeOH gave 8 as a slightly yellow solid (0.373 g, 94%).
1H NMR (400 MHz, DMSO-d6): δ = 1.41–1.58 (m, NHCH2CH2CH2NMe2), 1.66–1.77 (m, ArCH2CH2, NHCHCH2CH2OH), 2.09 (s, NMe2), 2.18 (m, NHCH2CH2CH2NMe2), 2.55–2.61 (m, ArCH2CH2), 3.03–3.10 (m, NHCH2CH2CH2NMe2), 3.47–3.73 (m, ArCH2CH2CH2, OCH2CH(CH2OR)O, NHCHCH2CH2OH), 3.81–4.25 (m, OCH2CH(CH2OR)O, NHCHCH2CH2OH), 7.14–7.36 (m, ArCH2, NH), 7.81–8.02 (m, NH) ppm. 13C NMR (100 MHz, DMSO-d6): δ = 26.9 (NHCH2CH2CH2NMe2), 31.0 (ArCH2CH2), 31.7 (ArCH2CH2), 35.1 (NHCHCH2CH2OH), 37.2 (NHCH2CH2CH2NMe2), 45.2 (NMe2), 52.2 (NHCHCH2CH2OH), 56.8 (NHCHCH2CH2OH), 57.7 (NHCH2CH2CH2NMe2), 63.8, 68.7, 77.8 (ArCH2CH2CH2, OCH2CH(CH2OR)O), 125.8 (ArCH2), 128.3 (ArCH2), 128.4 (ArCH2), 141.8(ArCH2), 156.0 (OC=ONH), 171.9 (CHC=ONH) ppm.
2.10. Synthesis of P(GHSL,oq)26 (9)
Polymer 8 (0.250 g, 0.824 mmol –NMe2 groups) was dissolved in MeOH (5 mL) and methyl iodide (0.175 g, 1.236 mmol) was added. The solution was stirred under reflux for 20 h. Removal of MeOH and excess methyl iodide under reduced pressure at 50 °C gave polymer 9 as a yellow solid (0.324 g, 88%). 1H NMR (400 MHz, D2O): δ = 1.95–2.08 (m, ArCH2CH2, NHCH2CH2CH2NMe3, NHCHCH2CH2OH), 2.66–2.74 (m, ArCH2CH2), 3.18 (s, NMe3), 3.25–3.47 (m, NHCH2CH2CH2NMe3), 3.72–3.87 (m, ArCH2CH2CH2, OCH2CH(CH2OR)O, NHCHCH2CH2OH), 4.12–4.37 (m, OCH2CH(CH2OR)O, NHCHCH2CH2OH), 7.33–7.42 (m, ArCH2) ppm. 13C NMR (100 MHz, D2O): δ = 22.8 (NHCH2CH2CH2NMe3), 33.8 (NHCHCH2CH2OH), 36.2 (NHCH2CH2CH2NMe3), 52.8 (NHCHCH2CH2OH), 53.2 (NMe3), 58.0 (NHCHCH2CH2OH), 64.2 (NHCH2CH2CH2NMe3), 64.5 (OCH2CH(CH2OR)O), 68.8, 76.6, 77.3 (ArCH2CH2CH2, OCH2CH(CH2OR)O), 128.2 (ArCH2), 128.8 (ArCH2), 141.0 (ArCH2), 157.5 (OC=ONH), 174.7 (CHC=ONH) ppm.