2.1. Compartment Fire Experiments
Although comprehensive datasets for intermediate to large-scale experiments on individual fuels conducted over a range of equivalence ratios are limited, sets of usable published data have been identified for six different fuel materials. Details of these tests are reported in the original publications cited, with the test conditions summarised in
Table 1. For each material tested in the compartment fires, tests were also carried out in the SSTF on the same polymers or chemically-similar materials with the same empirical formulas. The apparatus and experimental methods used by different authors for the compartment fires differed somewhat, but in each case, the test materials were combusted under flaming conditions over a range of measured equivalence ratios. The first two materials, polyamide 6.6 (PA66) and polypropylene (PP), were burned in floor pans in an ISO 9705 room by Blomqvist and Lonnermark [
15]. The room ventilation was controlled by varying the area of an opening in the upper doorway. The next two materials, wood and polymethylmethacrylate (PMMA), reported by Gottuk and Lattimer [
9], were also burned on a room floor, but controlled ventilation was provided from below via a distribution plenum. The effluent plume passed out of the room via a vent near the ceiling level. The results from these experiments are compared to those of Beyler [
22] for wood (Ponderosa pine) and PMMA. For these experiments, the fires were burned in the open laboratory, and an upper layer was formed in a cylindrical hood and plenum system. The mass of air entrained into the plume, and hence, the equivalence ratio, was controlled by varying the height of the collecting hood above the fire. The final two materials, medium-density fibreboard (MDF) and polyisocyanurate foam (PIR), were tested by Purser and Purser [
7,
11] in two different compartment fire configurations. For one set of experiments, an ISO 9705 room was used as for the Blomqvist and Lonnermark experiments, but for these experiments, the ventilation was controlled by varying the width of the sliding door panel. The tests were performed as room corner tests. The ignition source was a crib constructed from the test material and placed in a rear corner, with the same test material used to line the walls. For these experiments, the fires were therefore not confined to a discrete fuel package on the floor, but had the potential to ignite and spread across the wall linings. A second set of experiments was performed on MDF and PIR using a half linear scale ISO 9705 room connected via a variable width doorway opening to a corridor. For these experiments the fuel packages were in the form of cribs placed on the floor in the centre of the room.
For all of the compartment fire and SSTF experiments, the purpose was to measure post-ignition flaming decomposition of the gas-phase pyrolysis products from the different fuels. The methods used for igniting the fires are not described in most cases. Normal practice for such compartment fire experiments is to use a small solid or flammable liquid ignition source, such as methenamine or methylate spirits. For Purser and Purser’s experiments, the cribs were ignited using low density fibreboard sticks soaked in 300 mL methylated spirits. For the SSTF, all specimens auto-ignited in the furnace.
For the SSTF experiments, tests were performed by Blomqvist and Lonnermark on the same PA66 and PP as used for their compartment fires. The results are also compared to SSTF data by Purser et al. [
7,
21] on PA66 and PP sourced separately and also on polyamide 6 (PA6) and polyethylene (PE). For their wood compartment fires, Gottuk and Lattimer used spruce, while Beyler used Ponderosa pine. Scot’s pine (
Pinus sylvestris) was used for the SSTF experiments. Separately-sourced PMMA samples were used for the compartment fires and the SSTF experiments. For Purser and Purser’s work on MDF and PIR, the samples from the same specimens were used for the compartment fires and SSTF experiments.
Table 2 shows the composition of the materials tested in the SSTF. The carbon, hydrogen, nitrogen and chlorine contents were measured by Butterworth Laboratories using in-house elemental microanalysis methods (Butterworth Laboratories Method—BLM) BLM 0G, BLM 5 and BLM 267. The oxygen content where appropriate was calculated by difference. The measured compositions compared well to the empirical formulae for pure polymers, as shown in
Table 2. For polymethylmethacrylate (PMMA), low density polyethylene (LDPE) and polyamide 6.6 (PA66), the measured compositions were within 2%–3% of empirical values, so yield calculations were based on the empirical formulae, which were considered most accurate. For wood, medium-density fibreboard (MDF) and polyisocyanurate (PIR) analysis data are shown. These values were used for SSTF yield calculations.
Polyamide 6.6 (PA66) and Polypropylene (PP): For the TOXFIRE project [
15,
17], individual fuels were combusted as discrete pool fire fuel packages in an ISO 9705 room [
23] under different ventilation conditions obtained by varying the size of the room inlet vent. The room had one opening, 0.8 m wide 2.0 m high, centrally located at one end. The ventilation conditions inside the room were controlled by sealing the lower part of the opening with slabs of non-combustible fibreboard, which gave a reduced opening of height 0.89, 0.68, 0.56 and 0.45 m, for the development of under-ventilated conditions. The fuel was put into square pans of 1.2 m
2 for polyamide 6.6 and 1.4 m
2 polypropylene. Four tests were carried out, burning between 55 and 75 kg of PA 6.6 and 60 kg of PP, using a load cell to measure the mass loss. The composition of the effluent and equivalence ratio were measured in the calorimeter hood and in effluent sampled from the upper layer inside the room doorway. A Φ meter was used to determine the equivalence ratio of the fire by adding oxygen to the fire effluent sampled inside the room, passing the mixture over a catalyst at 900 °C, then measuring the resulting oxygen concentration [
18]. Data for two pedigree polymers, polypropylene and polyamide 6.6, have been used for the comparison with the results for the same specimens in the SSTF by Blomqvist and Lonnermark [
15] and for the same polymers (but separately sourced) by Stec et al. [
21,
24] Comparisons have also been made with SSTF results for polyamide 6 [
7,
11], which has the same empirical formula as polyamide 6.6, but a slightly different structure. Data are also shown for the comparison for low density polyethylene (LDPE), which has the same empirical formula as polypropylene [
11], but a slightly different structure.
Wood and PMMA: Data for compartment fire tests on wood and PMMA are reported by Gottuk and Lattimer [
9,
10,
25]. For these experiments, discrete fuel packages were combusted on the floor of a 1.2 × 1.2 × 1.55 m high compartment. Air was supplied at the floor level from below via a plenum and the effluent exhausted via a vent 20 cm below the ceiling. Effluent was sampled in the compartment from the upper layer below the ceiling level. The wood used was spruce. Φ was calculated as the ratio of the measured mass-loss rate of the fuel and the measured air-mass inflow, divided by the stoichiometric fuel:air ratio. Other experiments were carried out by Beyler on Ponderosa pine and PMMA [
22], also using discrete fuel packages (cribs) combusted under a 1.02 m-wide hood. Air entrainment into the fire plume (and hence, the equivalence ratio) was controlled by lowering the hood to different levels. The plume equivalence ratio Φ was determined by measuring the total gas flow into the hood, the measured flow and upper layer composition. For the SSTF, experiments were carried out on separately-sourced PMMA and Scot’s pine wood (
Table 2).
Medium-density fibreboard (MDF) and polyisocyanurate foam (PIR): Specimens of medium-density fibreboard and PIR were combusted as wall linings in an ISO 9705 room at the Building Research Establishment (BRE) under a range of ventilation conditions obtained by setting different door widths [
11,
26]. For the ISO 9705 room experiments, the walls were lined with MDF board or PIR-aluminium foil-faced panels. The standard propane burner was replaced by MDF (16 kg) or PIR cribs (2.07 kg) as the ignition sources, designed to provide a heat release rate of 300 kW. Different ventilation conditions were achieved by reducing the door width from 800 to 400, 280, 200 and 100 mm, resulting in 100%, 50%, 35%, 25% and 12.5% of full ventilation. To obtain yield data, fire parameters were measured in the upper layer inside the ISO room (0.4 m from the ceiling and 1.0 m from the door end of the room). Grab samples were taken using Tedlar bags, bubblers and on glass fibre filters. Data were also captured in the calorimeter duct. Bag samples for the estimation of fuel gases and equivalence ratios were taken as far as possible during periods of steady state combustion. Equivalence ratios were calculated using the oxygen depletion method as for the room-corridor experiments. The equivalence ratio and pyrolyzed fuel mass concentrations were calculated by the secondary combustion of effluent samples with the measurement of total oxygen required for complete combustion and the measurement of total fuel carbon as CO
2. Compartment fire temperatures were measured with thermocouple trees inside the room.
In other experiments, the same materials were combusted as a discrete crib in a half linear scale ISO room corridor rig [
7,
11,
26]. The “room” had linear dimensions half those of the ISO 9705 calorimeter (
Table 3;
Figure 1). The corridor (4.8 m long) had a horizontal partition along its length to limit mixing of incoming air with outgoing fuel-rich fire effluent and prevent secondary combustion.
The rig was constructed from heat-resistant mineral board (“Supalux”) panels on a wooden frame. To minimise heat losses it was lined internally with ceramic blanket (room ceiling) and externally with PIR foam panels. A full height (1.2 m) adjustable sliding panel, which opened to a maximum of 30 cm in width for maximum ventilation conditions, was inserted between the room and corridor. MDF board and PIR foam cribs (mass 4–5 kg) were placed in the centre of the room on a layer of bricks to allow ventilation at the base. The MDF cribs used 120 sticks 25 × 2 × 1.22 cm in 12 layers 29.5 cm high. The PIR cribs were of 159 sticks 60 × 3.8 × 3.8 cm in 16 layers 63 cm high. Ventilation was controlled by varying the “door” panel width between 2.5 and 30 cm in 14 experiments. Effluent samples were taken from a point 12 cm below the ceiling and 20 cm from the back and side walls of the fire room and from the upper corridor (
Figure 1). Samples were taken continuously for CO
2, CO and O
2, in the fire room and corridor, with bubbler samples for HCN and grab samples (in Tedlar bags) taken during periods of steady state combustion for the measurement of total hydrocarbons and total airborne fuel. The total airborne fuel and total oxygen demand were measured by passing a bag sample through the secondary oxidising furnace (diluting the sample with additional air if necessary). This was also used for the calculation of the equivalence ratio (Φ) under which atmosphere samples were formed according to Equation (2):
where:
O2 supplied (%) = ambient concentration (assumed to be 20.95%) and
O2 required (%) = the total oxygen depletion required for complete combustion of the fully-oxidized fuel products released in the furnace.
The yield calculations were based on measurements obtained during the steady state flaming plateau for each fire test. Specimens of the same MDI and PIR were tested in the SSTF.
2.2. Steady State Tube Furnace Experiments
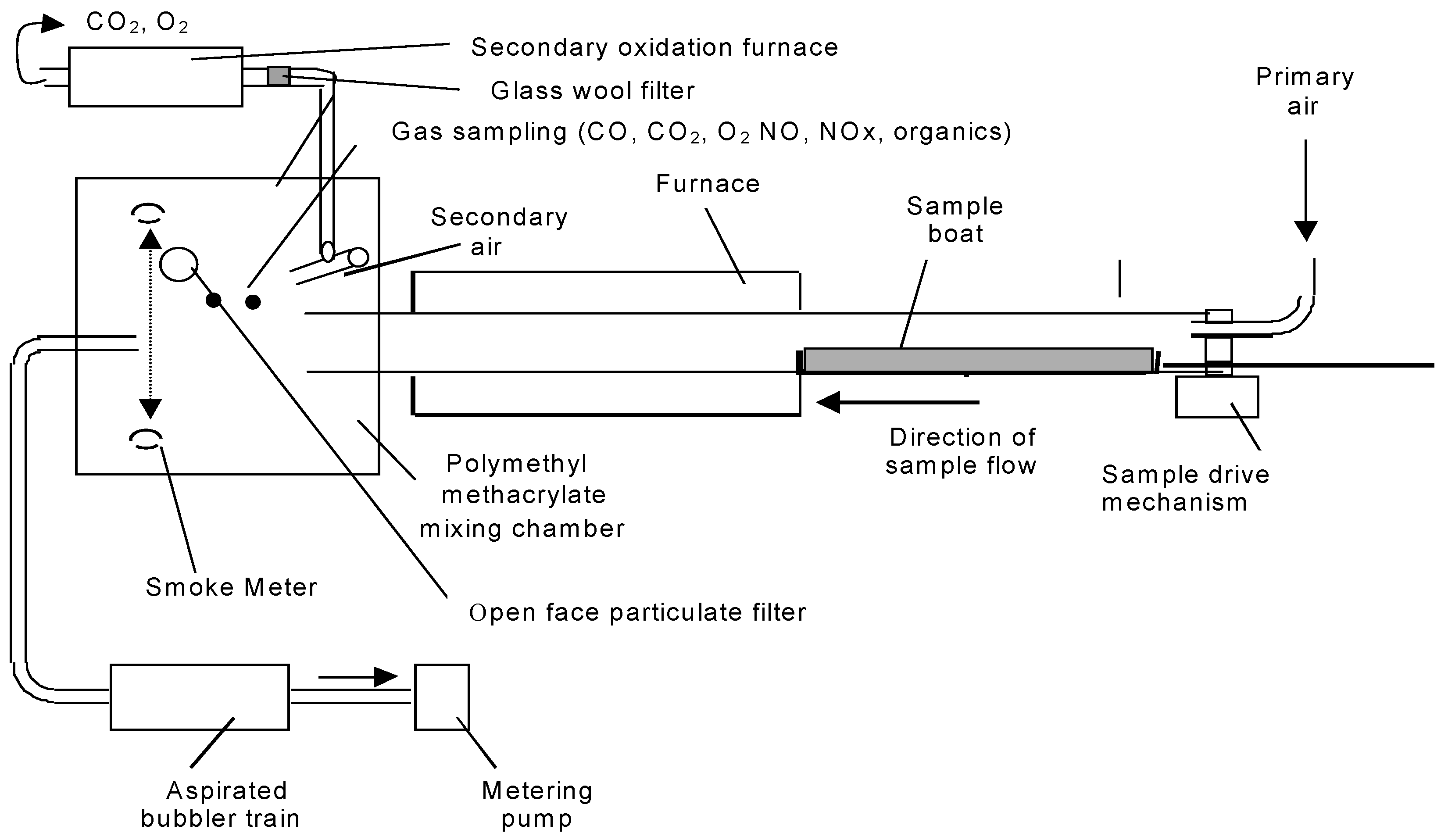
The tube furnace apparatus (
Figure 2) and experimental method were carried essentially as described in ISO TS19700 [
2]. A specimen of material in strip form was introduced into the tube furnace at a constant rate. A current of air was passed through the furnace over the specimen to support flaming combustion. The effluent was expelled from the tube furnace into the mixing and measurement chamber, where it was diluted with secondary air, then exhausted to waste. The decomposition conditions in the furnace were set using different combinations of temperature and air flow in separate runs, to model the decomposition conditions in a range of stages and types of fires as required. The aim for each experiment was to obtain stable (steady state) flaming conditions for up to 12 min (minimum of five min) during a 20-min experimental run to enable multiple measurements to be made of the composition of the effluent in the mixing and measurement chamber. The specimen was weighed before and after the test to enable the calculation of the fuel mass charge and mass loss rate. From the data obtained, it was possible to calculate the yields of each toxic product and the actual equivalence ratio under which the combustion was carried out. This then enabled the relationships between toxic product yields and equivalence ratio to be investigated.
For these experiments, a test specimen mass of 25–50 mg/mm was used, and air-flow rates were varied between 2 and 13 L/min in order to cover the range of combustion conditions (equivalence ratios) required. For each test run, the specimen was spread evenly along the furnace boat in granular or pellet form or as short segments. Experiments were conducted under constant steady flaming conditions (verified by observation) at a furnace temperature of 650 °C. For PIR, it was necessary to increase the furnace temperature to 700 °C in order to obtain steady flaming.
The aim of each experiment was to maintain steady flaming for separate runs over a range of equivalence ratios from Φ = 0.5–2.5 at a furnace temperature of 650 °C (700 °C for PIR) to represent pre-flashover combustion conditions over the range from well-ventilated (ISO 19706 Stage 2) [
14] to under-ventilated (ISO 19706 Stage 3a). Additional experiments included a higher temperature (850 °C) to represent post-flashover under-ventilated combustion conditions (ISO 19706 Stage 3b). Another variation was the use of 10% or 12% oxygen/nitrogen mixtures instead of air. By this means, it was possible to compare yields of CO and other products under the same equivalence ratios, but at different oxygen concentrations. The combustion products were diluted to a standard 50 L/min, and sampled from the mixing and measurement chamber, where other parameters were measured. The combustion products cooled as they passed through the tube beyond the furnace and down to <35 °C as they entered the mixing chamber, preventing further oxidation. Data available include fuel mass loss, heat of combustion, smoke optical density, yields of toxic species (CO, HCN, NO
x particulates, etc.) and O
2 depletion. Data on total airborne unburned fuel content and total unburned hydrocarbons were obtained by further oxidation of the diluted fire effluent using a small secondary oxidizing tube furnace at 900 °C, with measurement of CO, CO
2 and O
2. CO
2 and CO were measured dry by non-dispersive infrared analysers (NDIR) and O
2 by a paramagnetic analyser, from the mixing chamber and secondary furnace. Mixing chamber NO and NO
2 were measured by chemiluminescence, HCN by spectrophotometric analysis, HCl, HBr and SO
2 by ion chromatography of bubbler solutions, smoke particulates by optical density and gravimetrically using glass fibre filters.
Experiments were set up to provide nominal equivalence ratios between 0.5 and 2.5 using appropriate fuel:air ratios calculated from the stoichiometric oxygen demand for complete combustion of the entire material or product under test. After each run, the equivalence ratio actually achieved was calculated from the total oxygen consumed for complete combustion of the airborne fuel products in the secondary furnace (basically, a Φ meter method), summed with that calculated for oxidation of the particulate sample (soot) to give the total oxygen required for the combustion of the airborne fuel during the sample period according to Equation (2). For materials undergoing complete combustion, the actual Φ values obtained were very close to the nominal values, but for materials forming a carbonaceous char residue, a proportion of the fuel was unavailable for gas phase combustion, so that the achieved Φ values calculated from oxygen depletion were somewhat lower than the nominal values, although very close to nominal values calculated assuming that residues were 100% carbon.
2.3. Data Comparisons
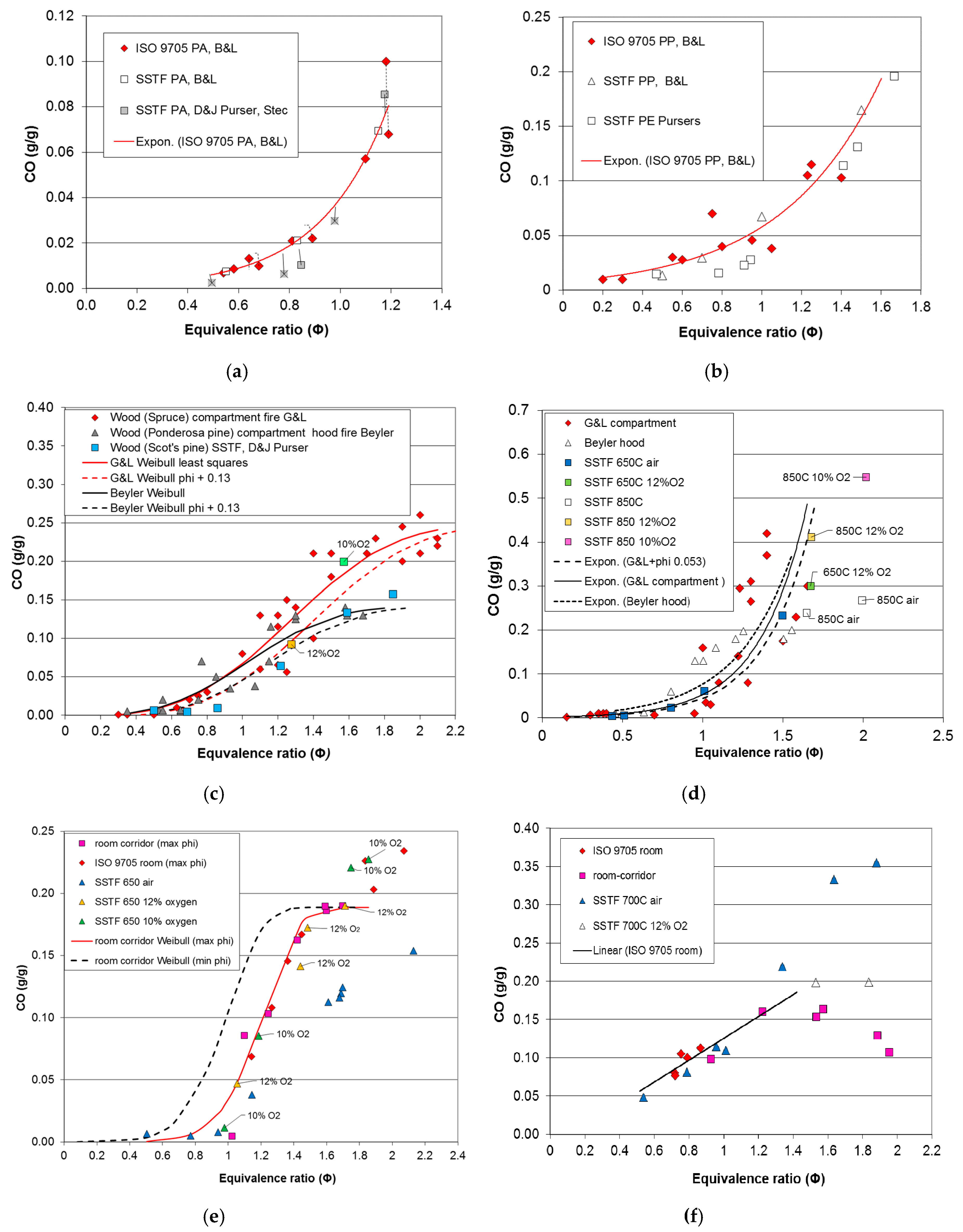
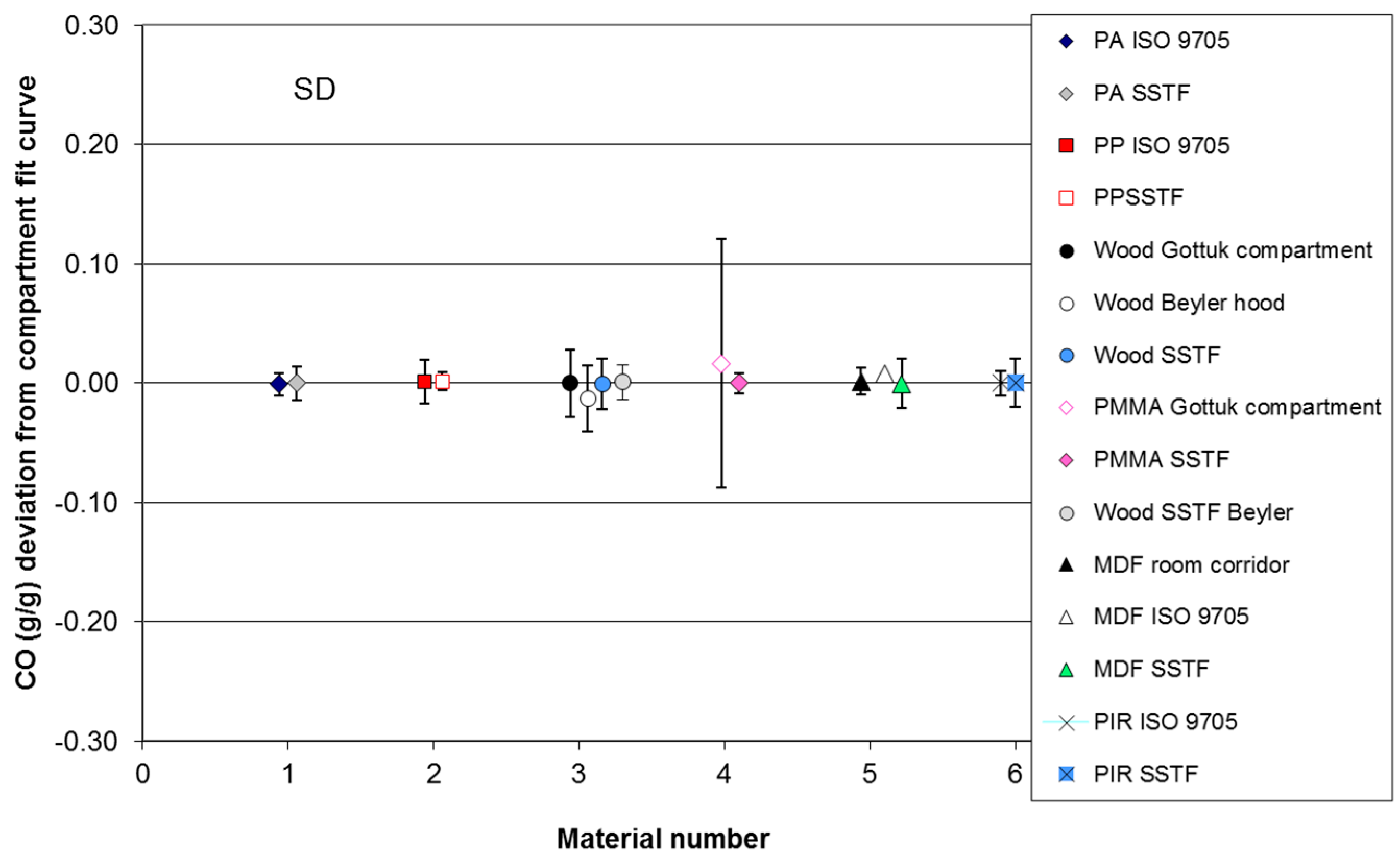
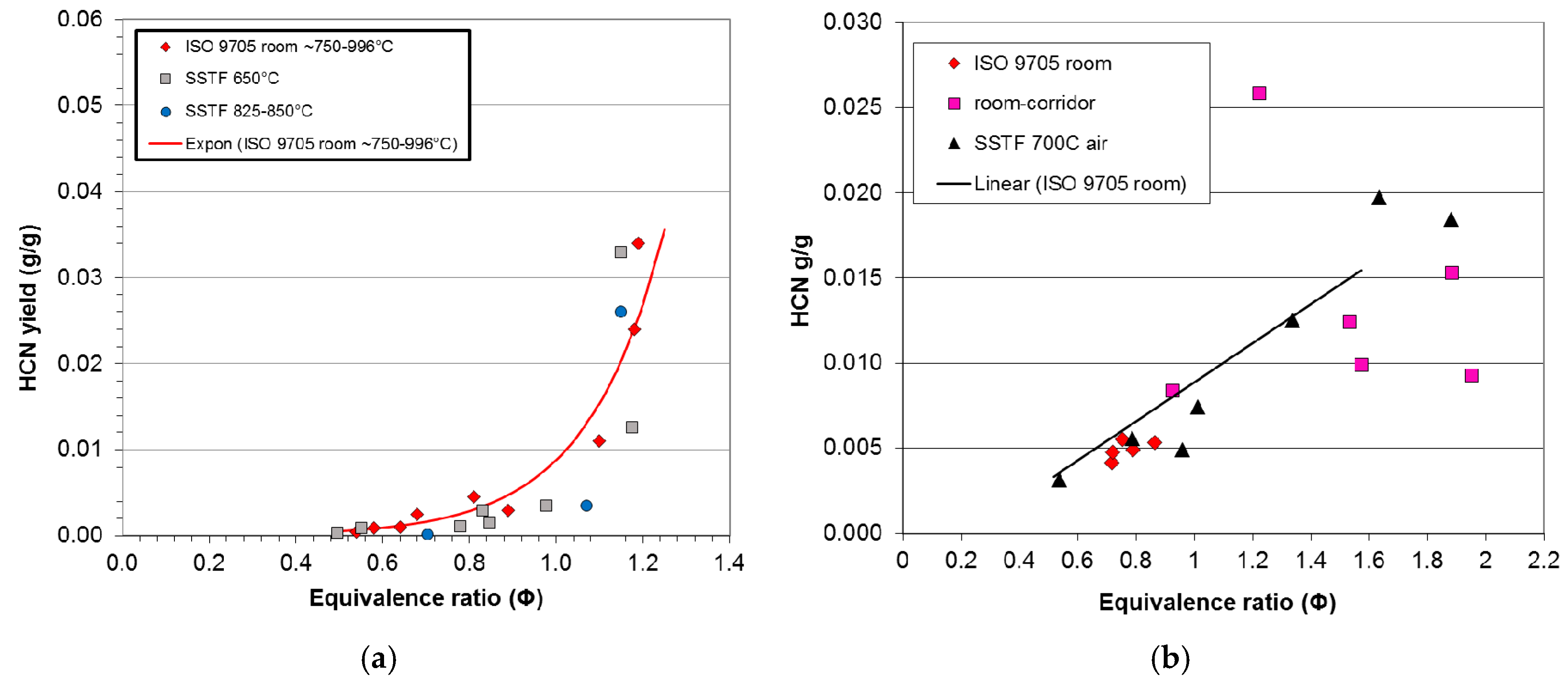
For the compartment fire tests, the data for CO yield and, where appropriate, HCN yield have been plotted as a function of equivalence ratio for each material. A least squares best-fit curve has been fitted to each compartment fire CO dataset. The deviation of each data point for CO yield from the best-fit compartment fire curve for each dataset has been measured and the range, mean and standard deviation of the deviations calculated for each case. The deviation of each SSTF data point from each compartment fire best-fit curve has also been measured, and the range, mean and standard deviation of the deviations from the compartment fire best-fit line have been calculated. For the BRE compartment fire data, the values for equivalence ratio were calculated assuming any upper layer oxygen present together with unburned fuel was a secondary diluent not involved in combustion. For other compartment fire tests, the best-fit curves were right-shifted where appropriate to align with the SSTF curves before measuring the deviations (Φ shift in
Table 4). The ranges, means and standard deviations of the deviations for the compartment fire and SSTF data were then compared. The typical shape of curves for CO yield as a function of equivalence ratio over the Φ range is sigmoid, so that CO yields are generally low below stoichiometry, with a rapid increase around Φ = 1–1.5, which then tends to level off. For curves of this general form, the best fit has been obtained using a Weibull function of the form:
where:
is the yield of product R (g/g), in this case CO, Φ is the equivalence ratio and the values for α, β and constants
k1–3 vary between the individual materials.
For polyamide, polyethylene and PMMA, the compartment fire data did not extend to high enough Φ levels for the curve to flatten off, so that a good fit was obtained with an exponential function.
Sigmoid Weibull functions were used for wood and MDF.
For PIR, a linear expression fitted the ISO 9705 room Φ data plus the lower Φ points of the room-corridor data, so that:
Above Φ ~ 1.3, the behaviour of PIR was more complex, as described. The expressions fitted for each material and constants are listed in
Table 4.