Structures and Synthesis of Zwitterionic Polymers
Abstract
:1. Introduction
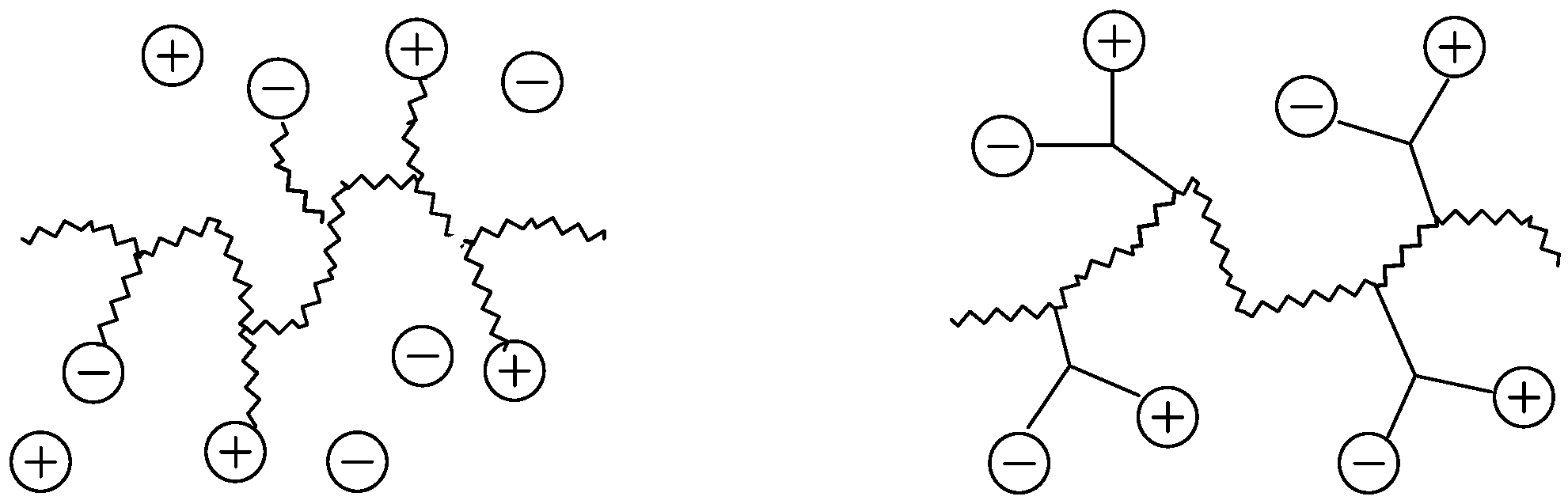

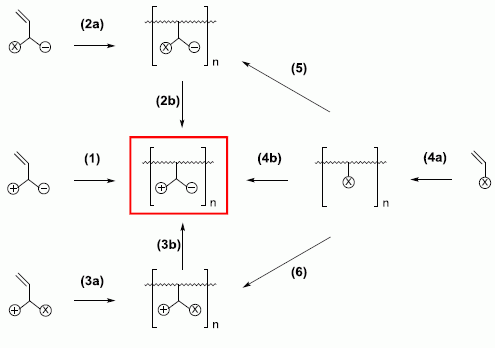
2. General Structures of Zwitterionic Polymers
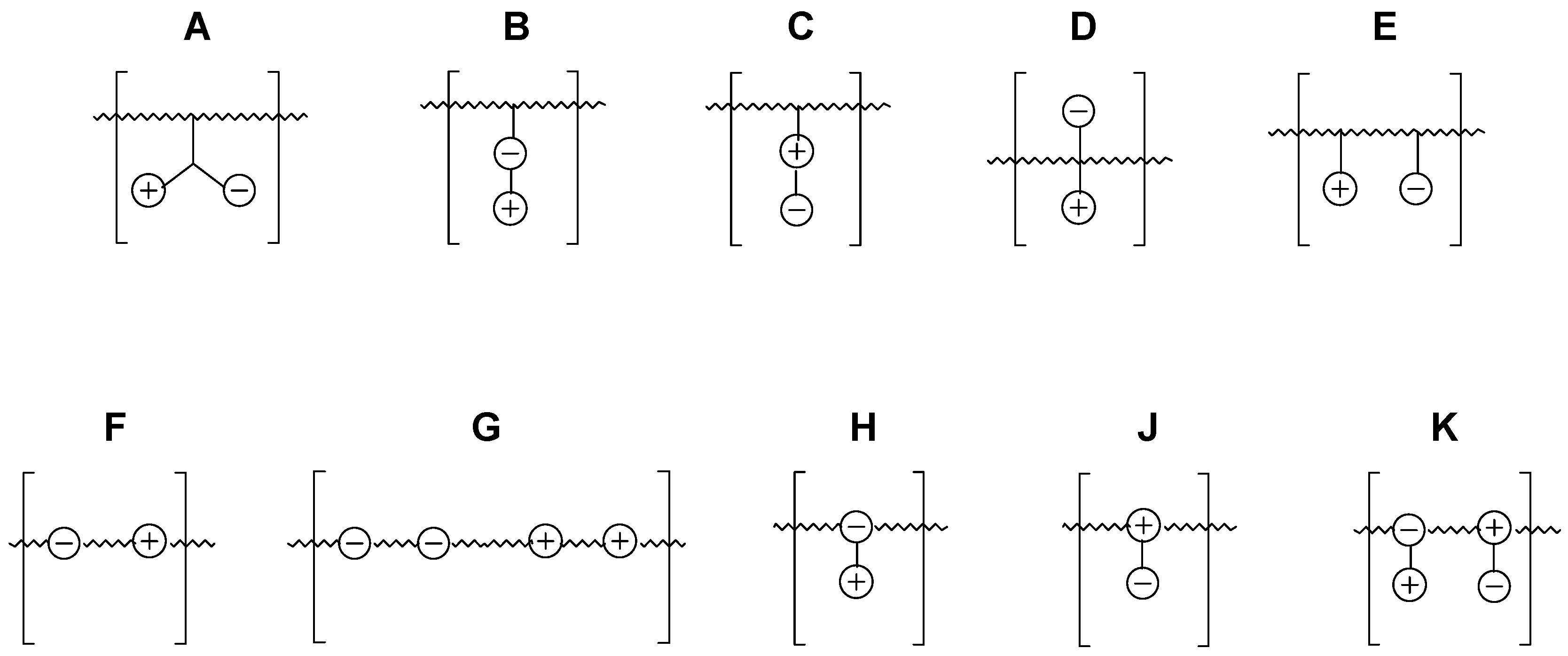
2.1. Possible Polymer Architectures
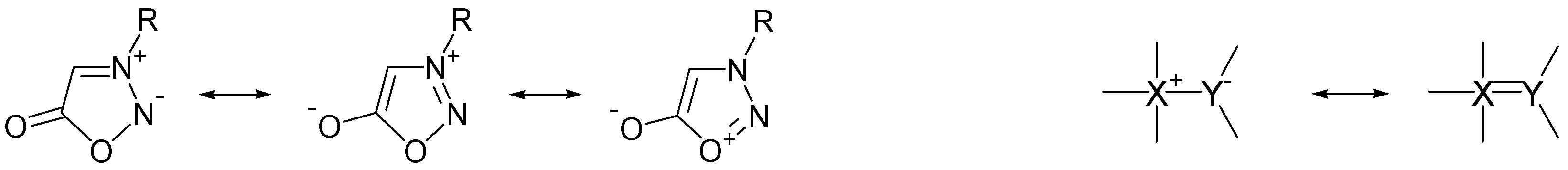
2.2. Zwitterionic Moieties
2.2.1. General Requirements for Useful Zwitterionic Moieties
2.2.2. Zwitterionic Moieties Realized in Polyzwitterions and Their Characteristic Features
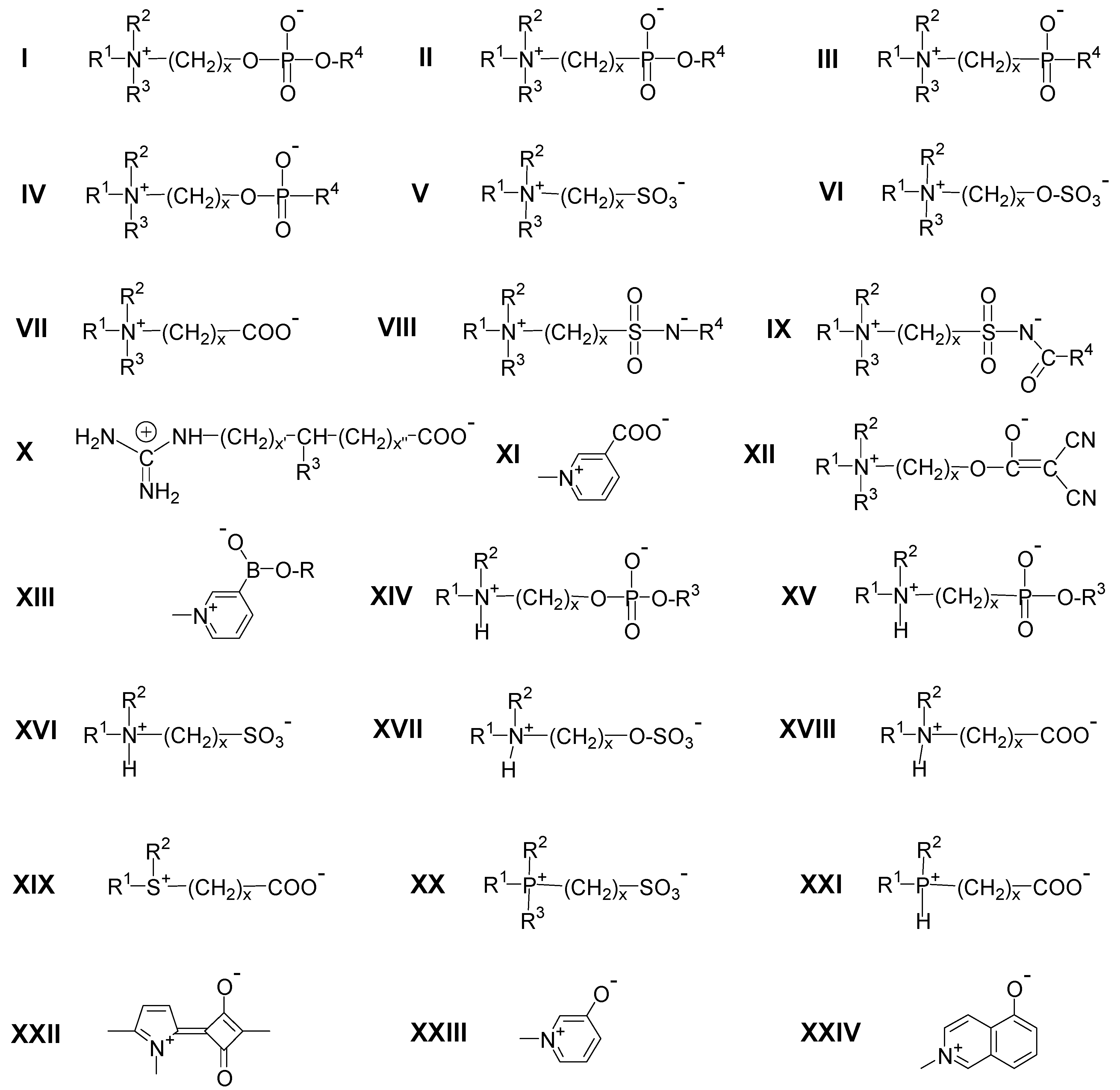
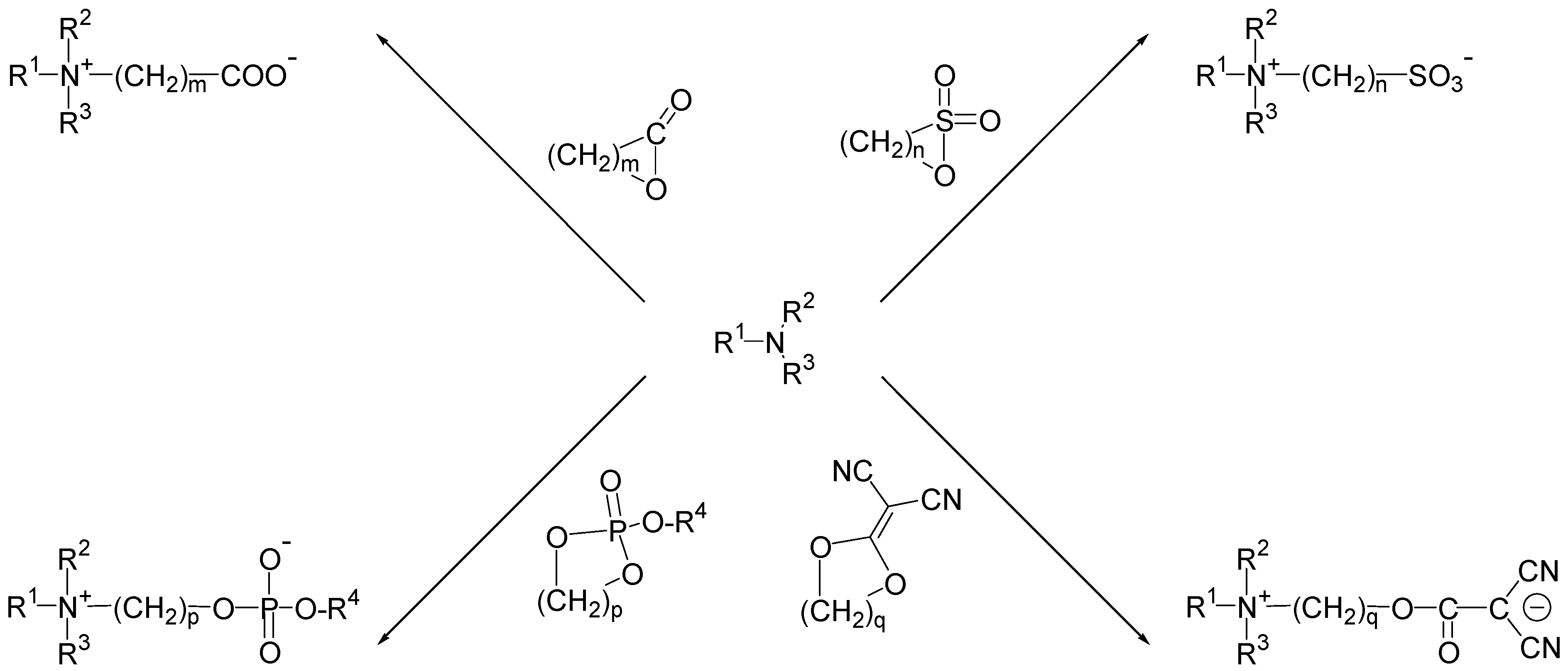
2.3. Synthetic Strategies towards Zwitterionic Moieties: Scope and Limitations

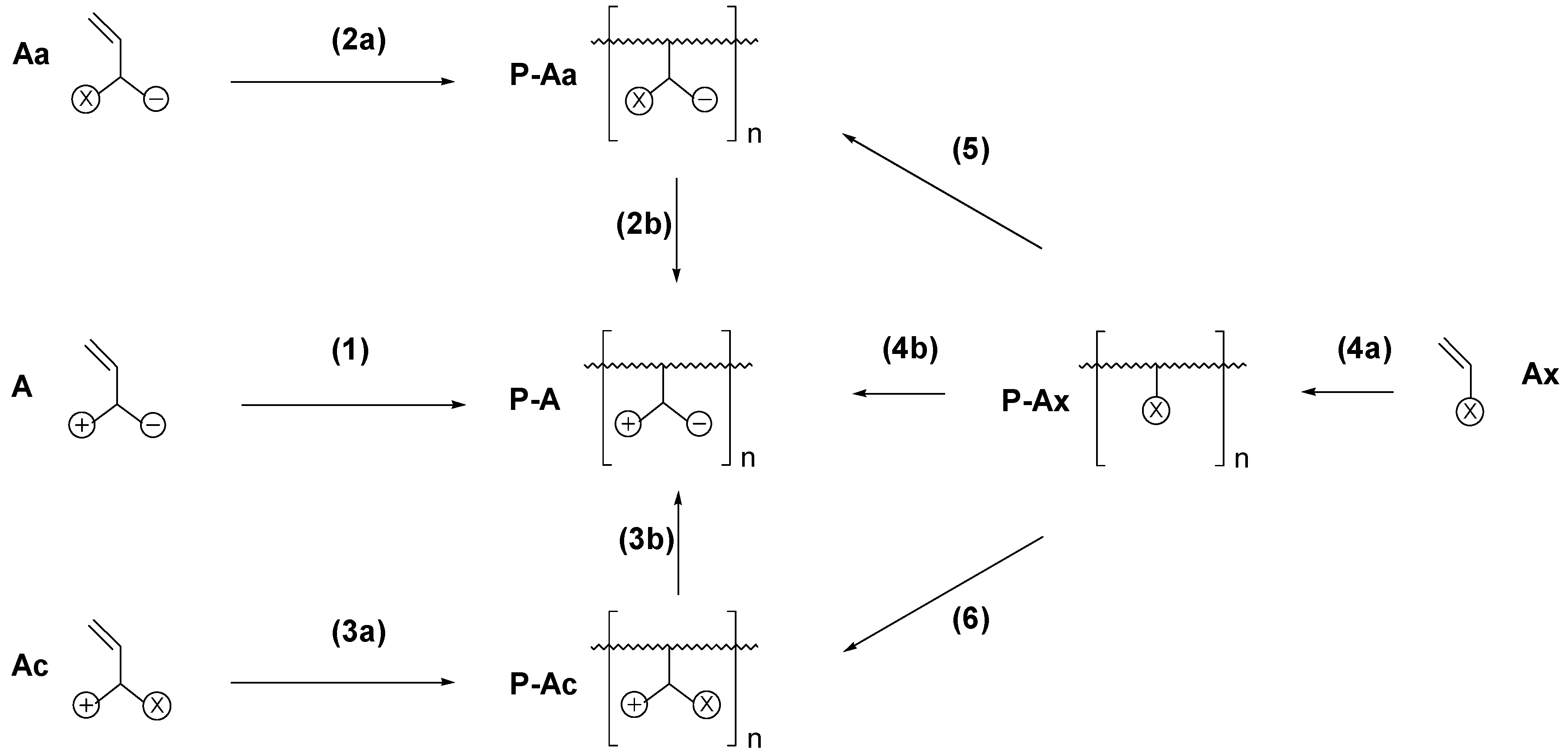
3. Synthesis of Polyzwitterions
3.1. Synthesis from Zwitterionic Monomers
3.1.1. Synthesis by Chain Growth Polymerizations
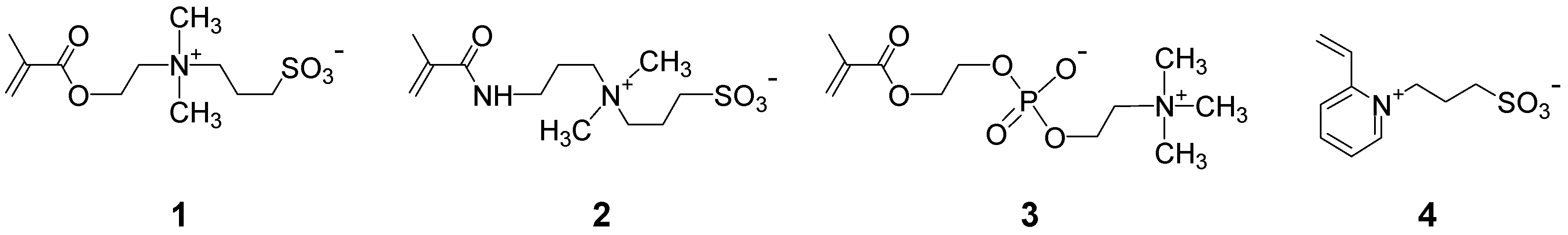
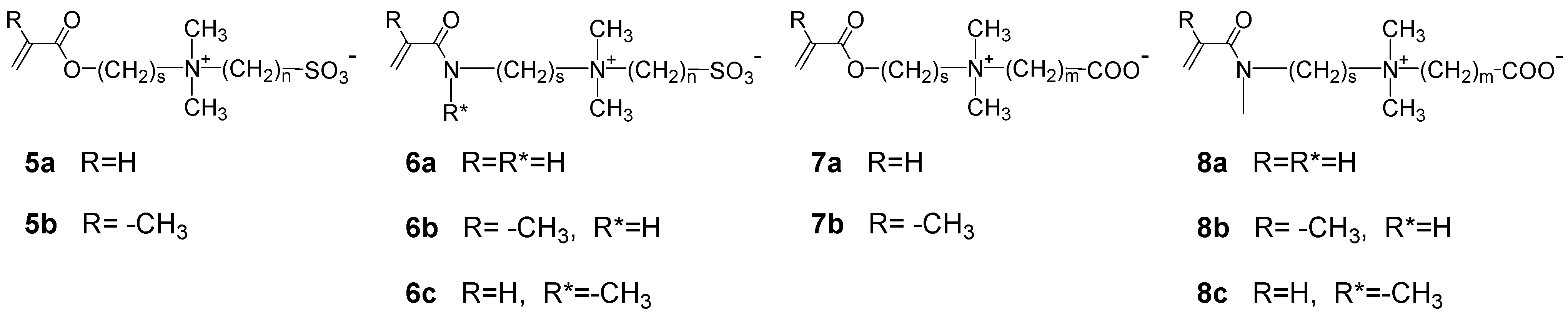
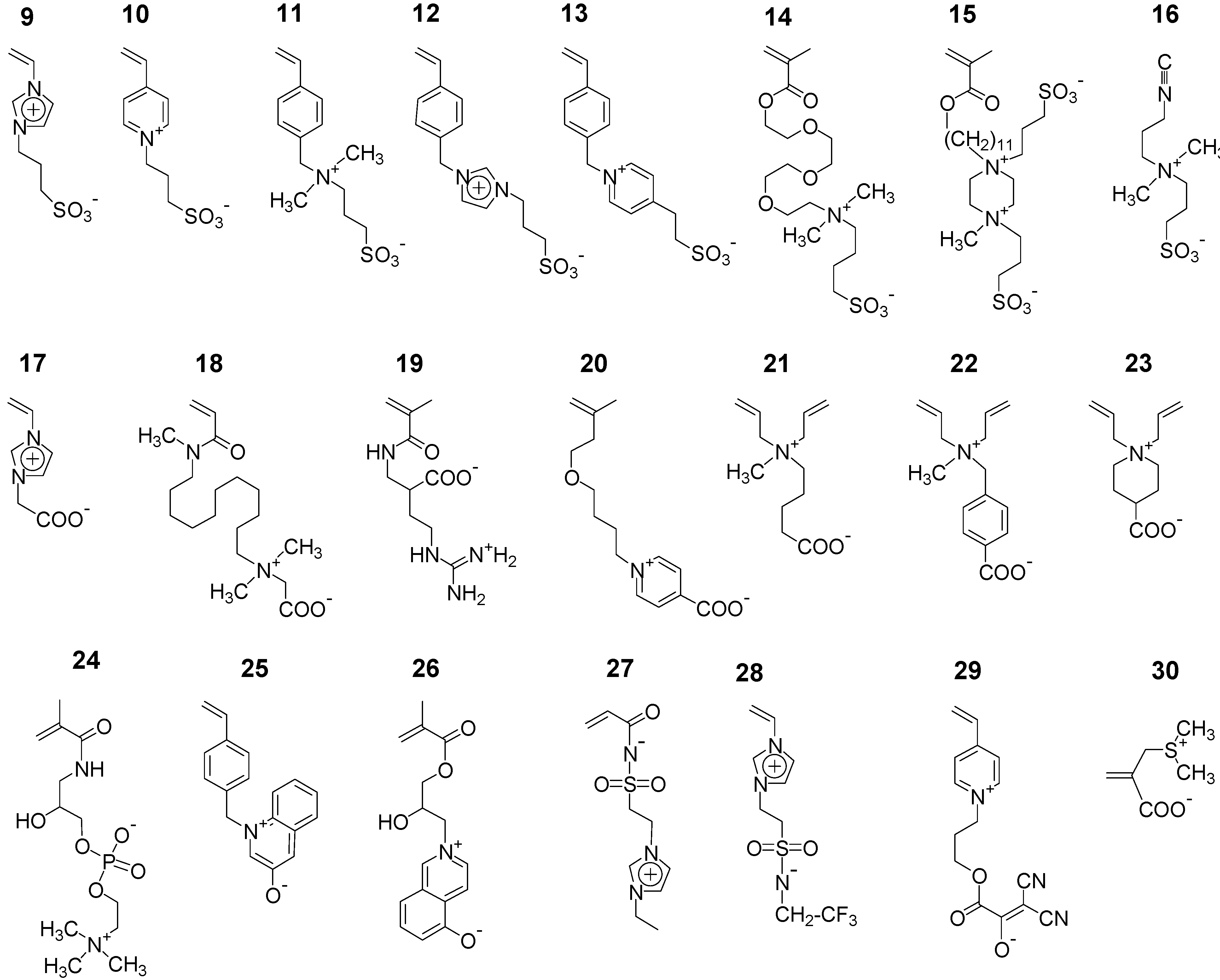
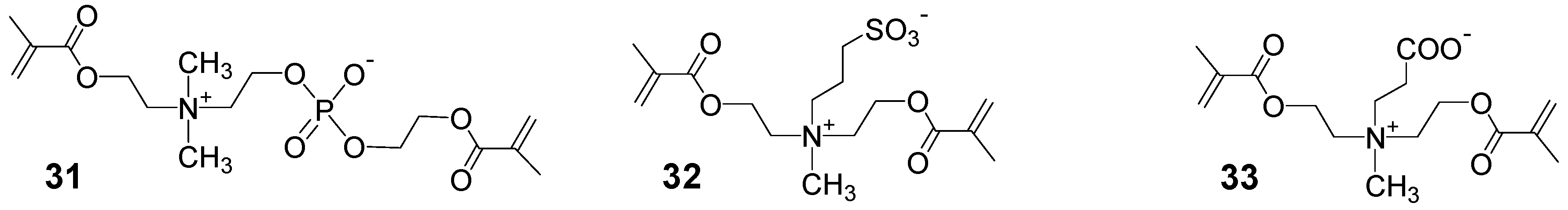

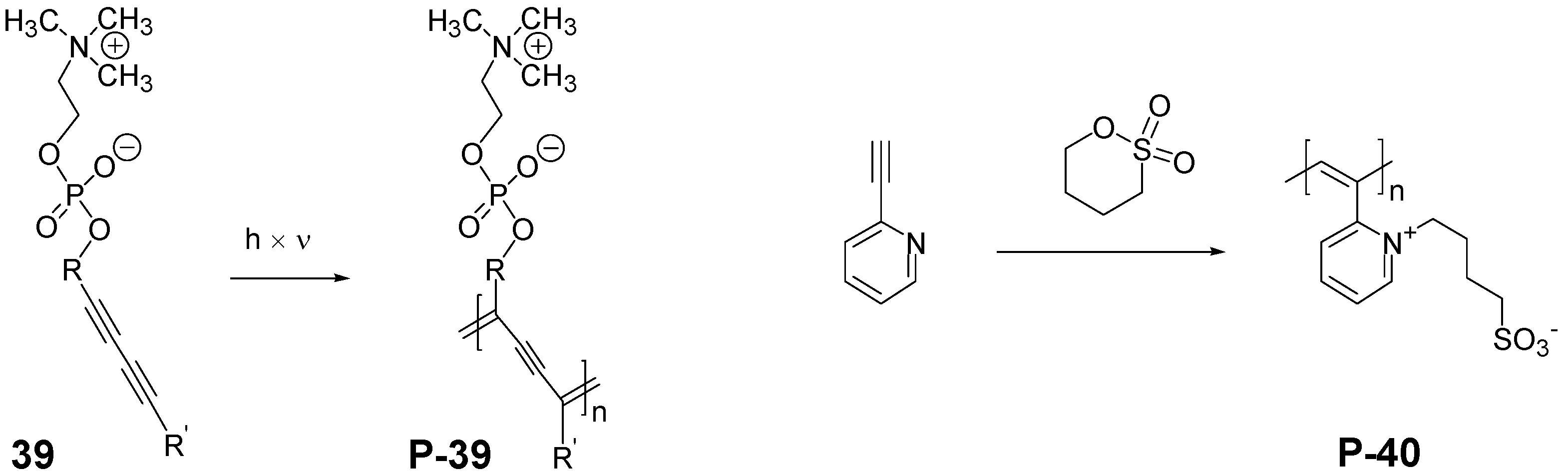
3.1.2. Synthesis by Step Growth Polymerizations
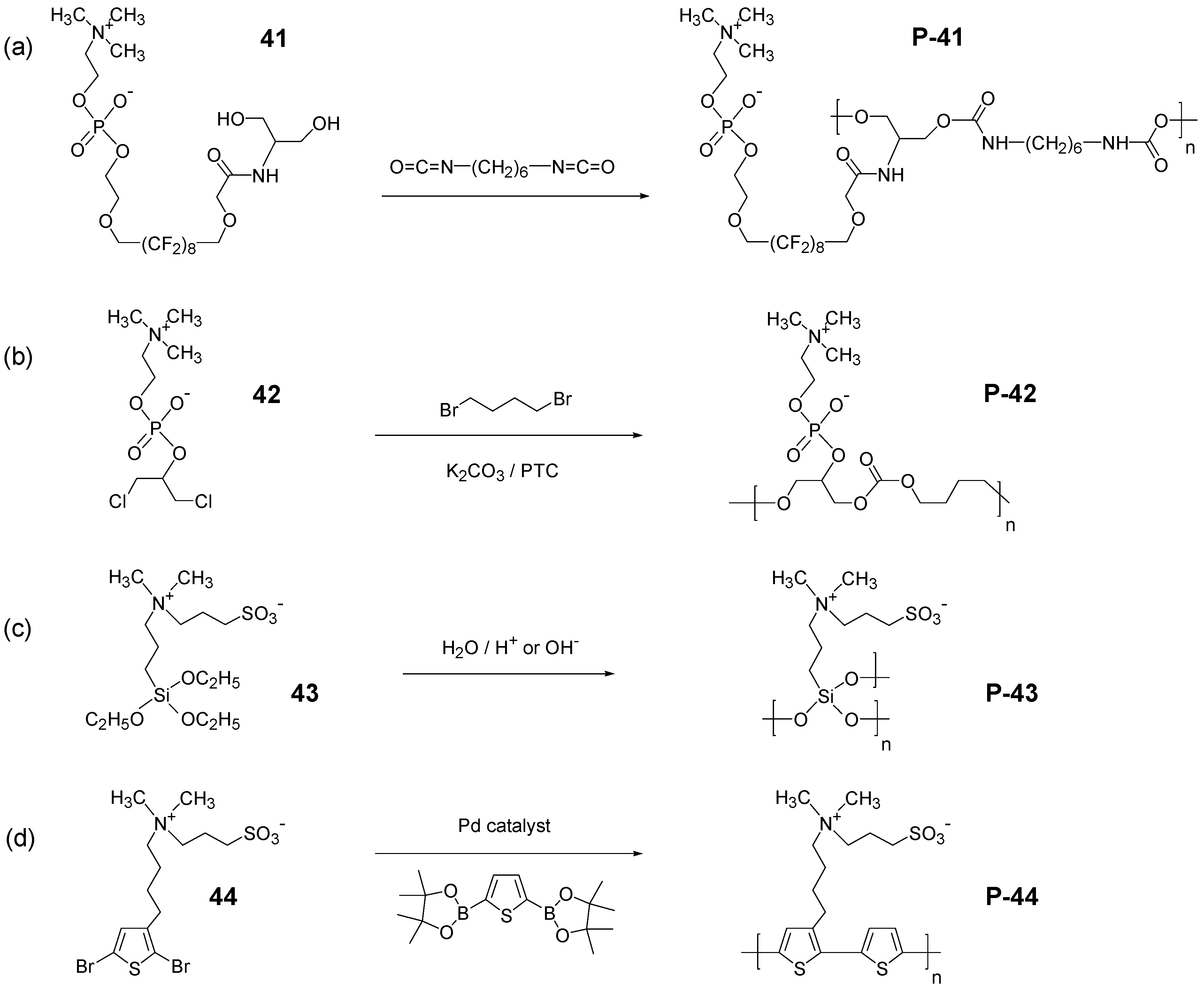
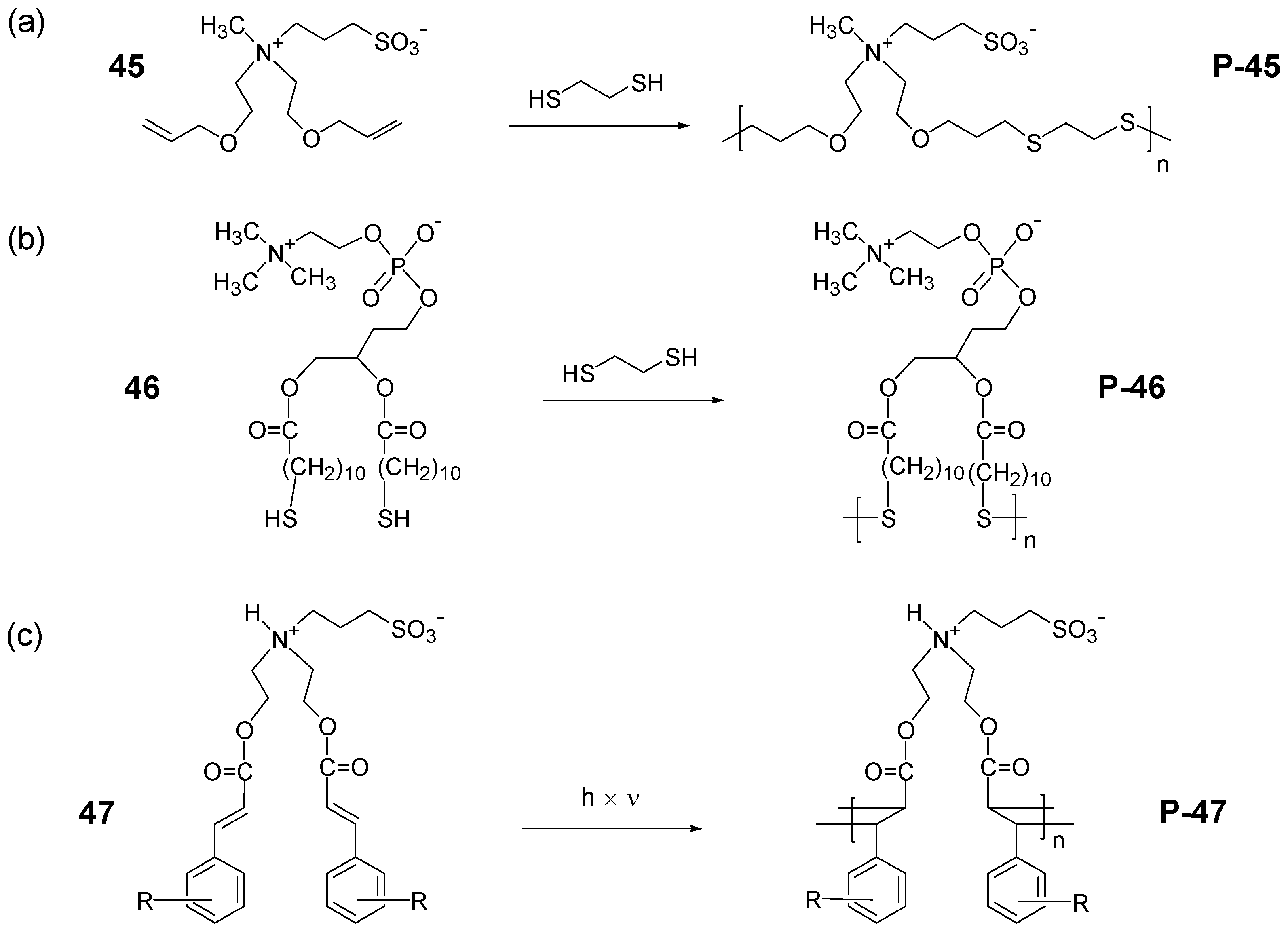
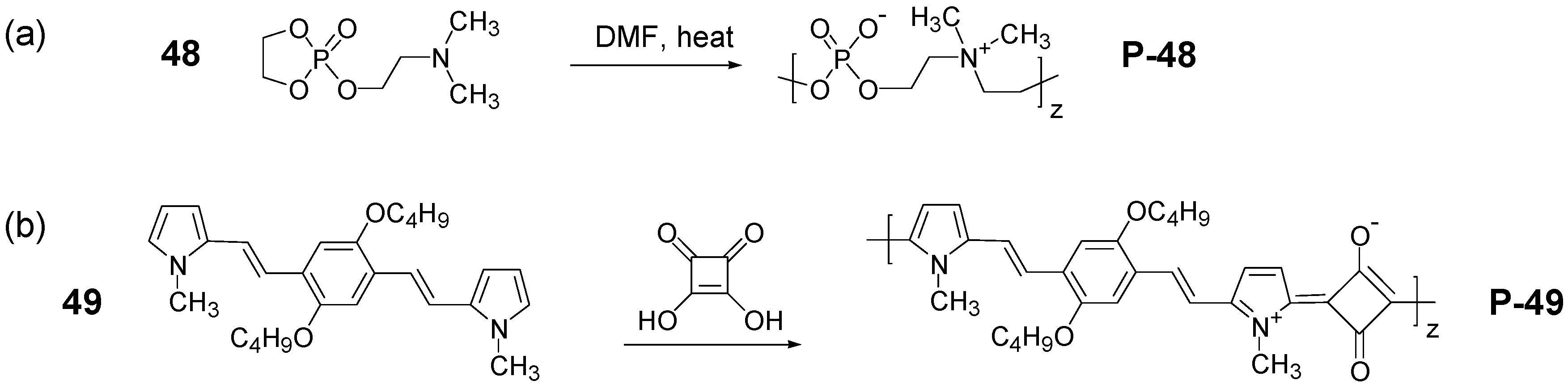
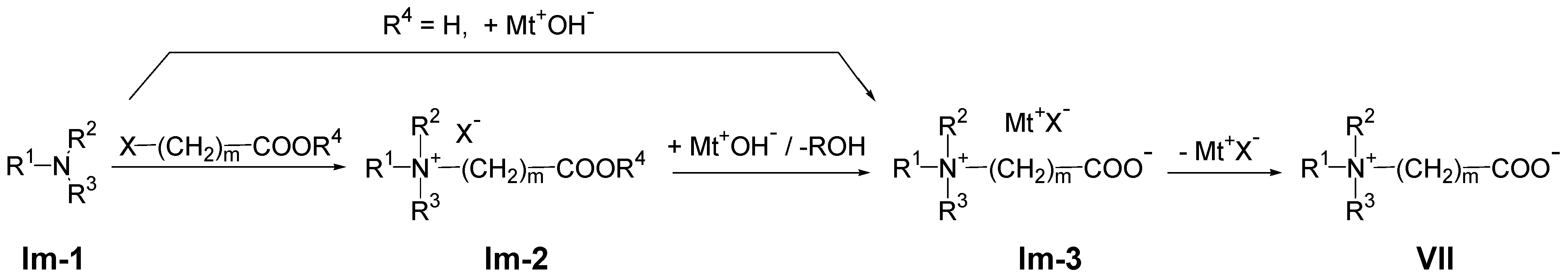
3.2. Synthesis of Polyzwitterions by Post-Polymerization Modification
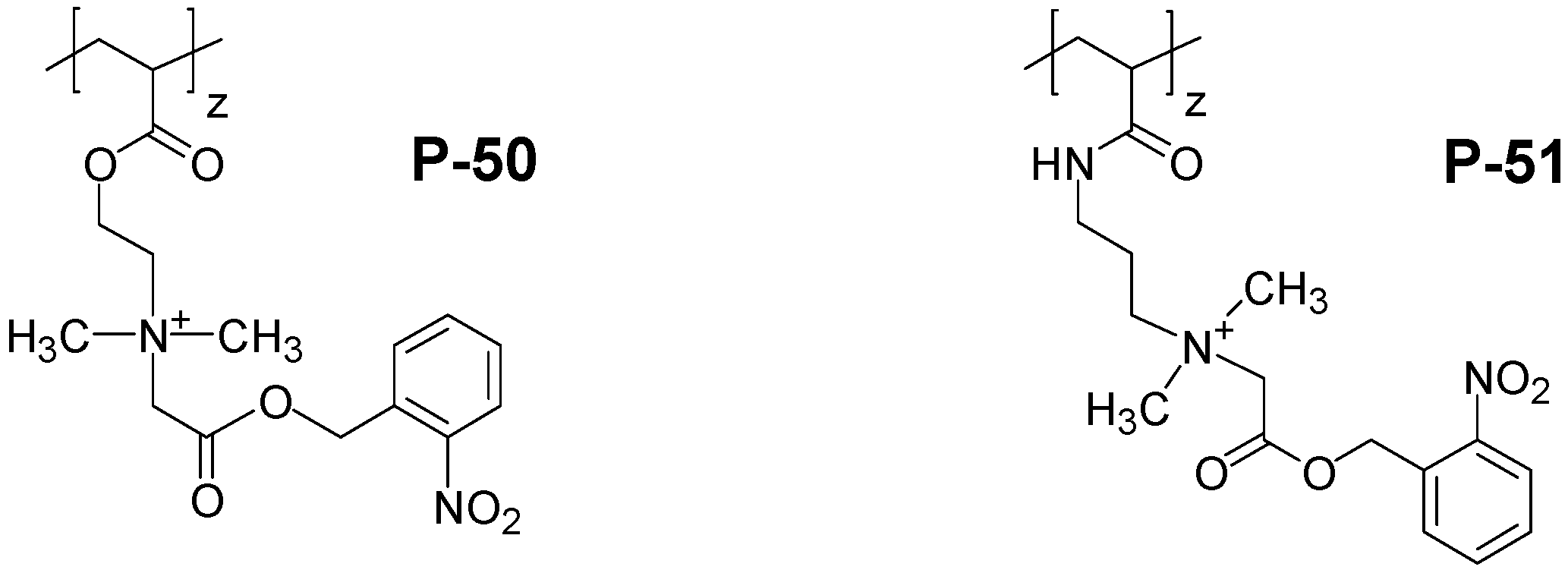
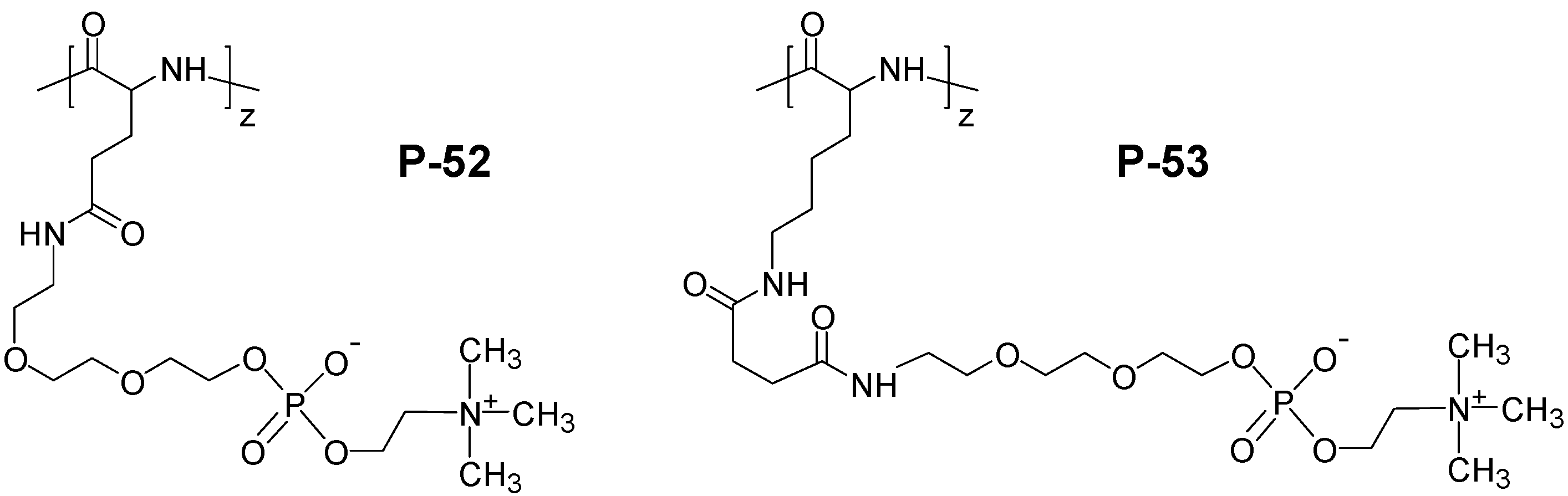
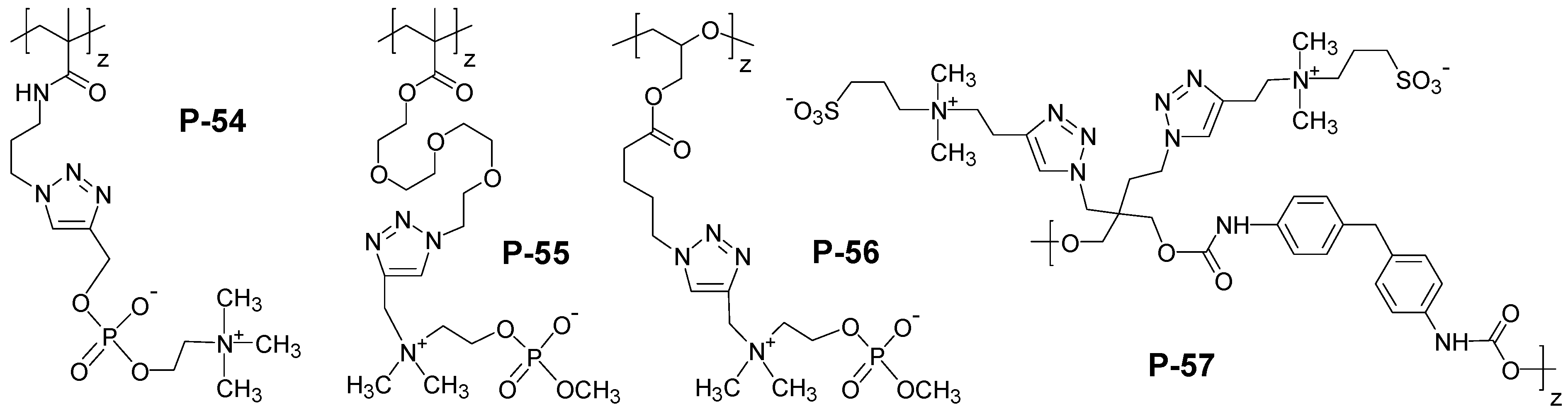
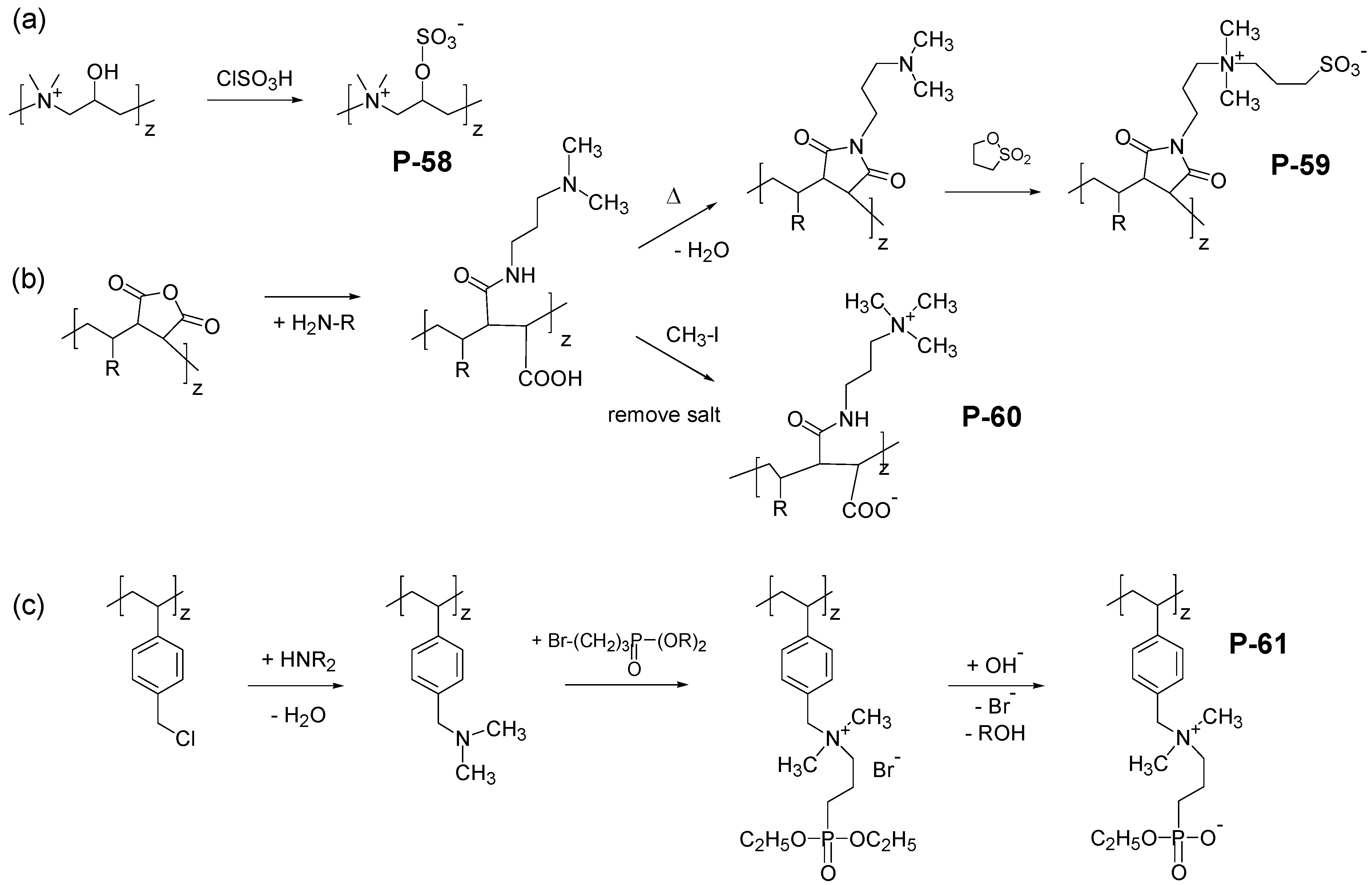

4. Special Aspects in the Chemistry of Polyzwitterions
4.1. Stability of Zwitterionic Moieties in Aqueous Media
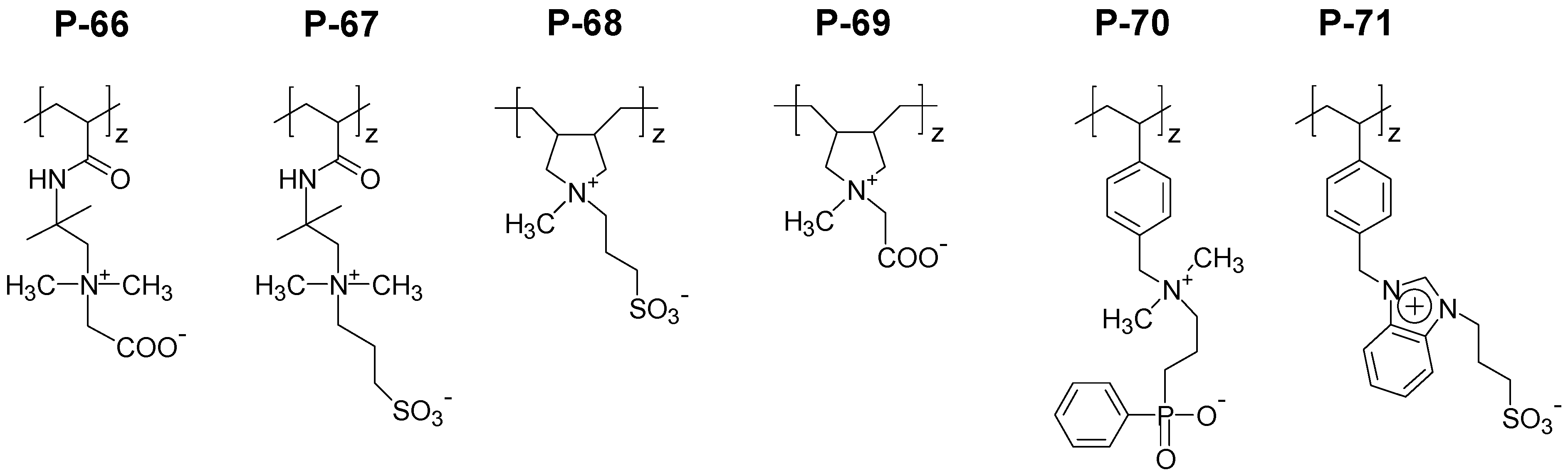
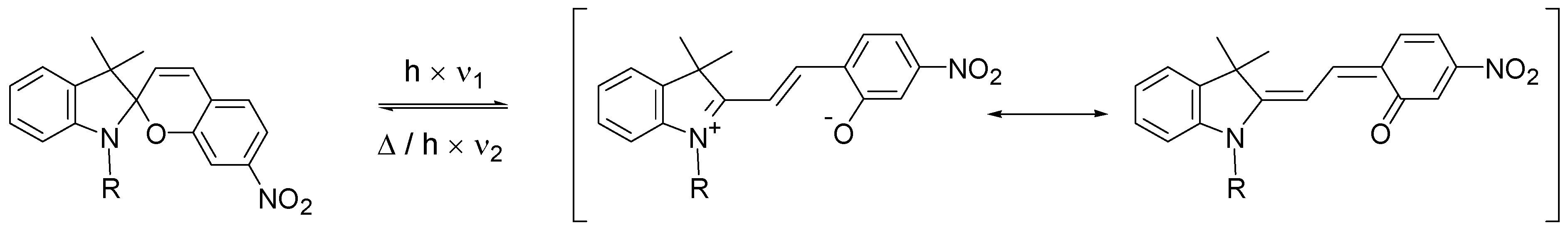
4.2. Responsive Polyzwitterions
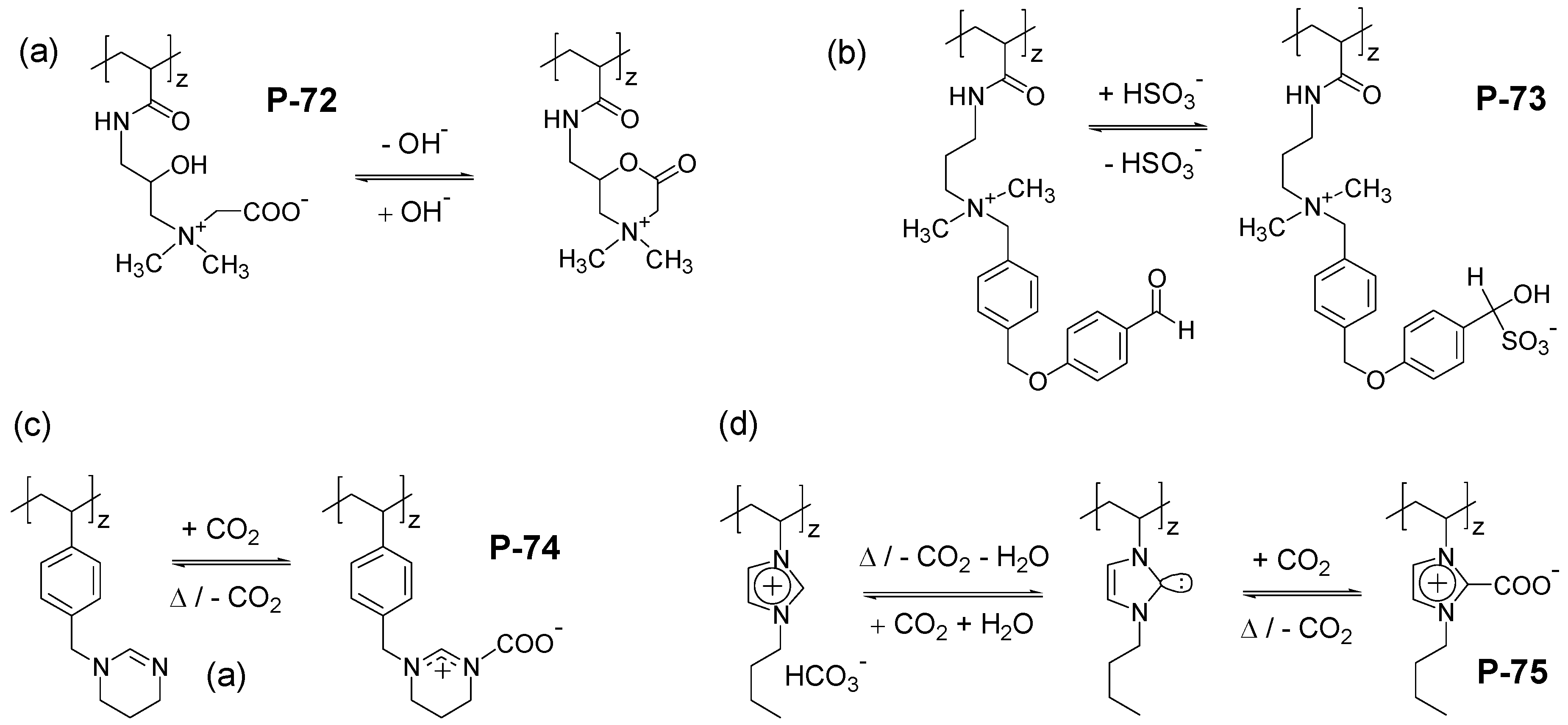
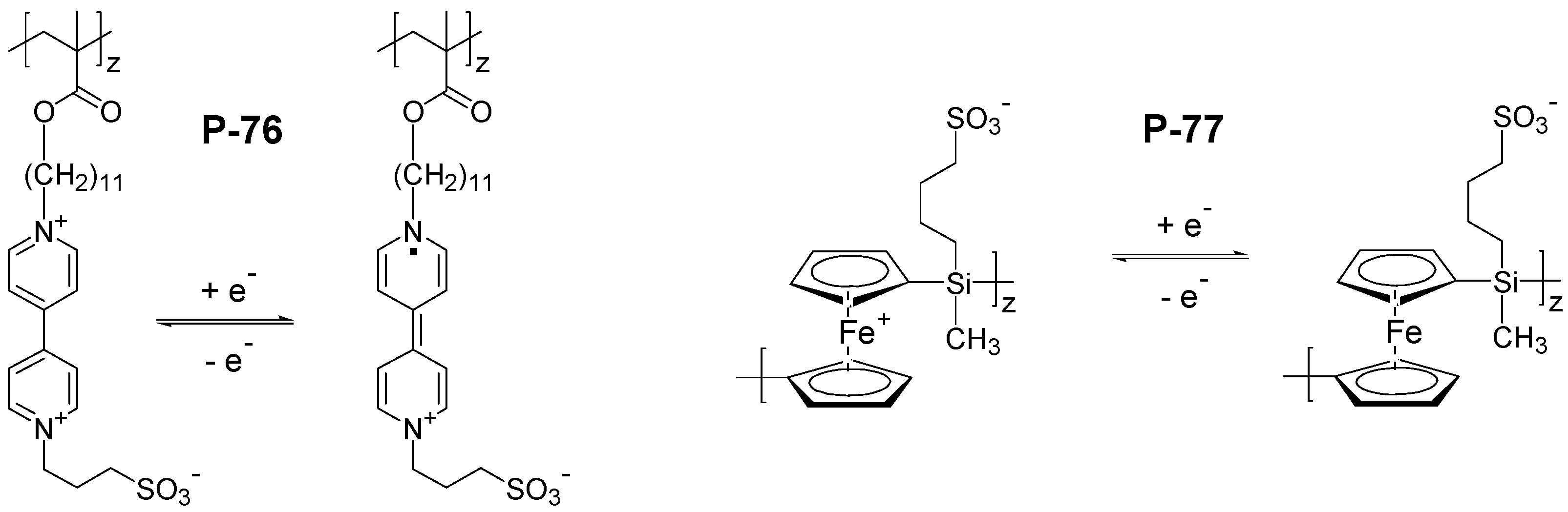
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Hess, M.; Jones, R.G.; Kahovec, J.; Kitayama, T.; Kratochvíl, P.; Kubisa, P.; Mormann, W.; Stepto, R.F.T.; Tabak, D.; Vohlídal, J.; et al. Terminology of polymers containing ionizable or ionic groups and of polymers containing ions. Pure Appl. Chem. 2006, 78, 2067–2074. [Google Scholar]
- Salamone, J.C.; Rice, W.C. Polyampholytes. In Encyclopedia of Polymer Science and Technologie, 2nd ed.; Mark, H.F., Bikales, N.M., Overberger, C.G., Menges, G., Eds.; Wiley-Interscience: New York, NY, USA, 1988; Volume 11, pp. 514–530. [Google Scholar]
- Galin, J.C. Polyzwitterions. In Polymer Materials Encyclopedia; Salamone, J.C., Ed.; CRC Press: Boca Raton, FL, USA, 1996; Volume 9, pp. 7189–7201. [Google Scholar]
- Lowe, A.B.; McCormick, C.L. Synthesis and solution properties of zwitterionic polymers. Chem. Rev. 2002, 102, 4177–4189. [Google Scholar] [CrossRef]
- Kudaibergenov, S.; Jaeger, W.; Laschewsky, A. Polymeric betaines: synthesis, characterization and application. Adv. Polym. Sci. 2006, 201, 157–224. [Google Scholar] [CrossRef]
- Zurick, K.M.; Bernards, M. Recent biomedical advances with polyampholyte polymers. J. Appl. Polym. Sci. 2014, 131, 40069:1–40069:9. [Google Scholar]
- Laughlin, R.G. Fundamentals of the zwitterionic hydrophilic group. Langmuir 1991, 7, 842–847. [Google Scholar] [CrossRef]
- Laughlin, R.G. HLB, from a thermodynamic perspective. J. Soc. Cosmet. Chem. 1981, 32, 371–392. [Google Scholar]
- Ladenheim, H.; Morawetz, H. A new type of polyampholyte: Poly(4-vinyl pyridine betaine). J. Polym. Sci. 1957, 26, 251–254. [Google Scholar] [CrossRef]
- Hart, R.; Timmermann, M. New polyampholytes: The polysulfobetaines. J. Polym. Sci., Part A: Polym. Chem. 1958, 28, 638–640. [Google Scholar]
- Perlmann, G.E.; Katchalski, E. Conformation of poly-L-methionine and some of its derivatives in solution. J. Am. Chem. Soc. 1962, 84, 452–457. [Google Scholar] [CrossRef]
- Yang, C.Z.; Hwang, K.K.S.; Cooper, S.L. Morphology and properties of polybutadiene- and polyether-polyurethane zwitterionomers. Makromol. Chem. 1983, 184, 651–668. [Google Scholar] [CrossRef]
- Brown, R.H.; Duncan, A.J.; Choi, J.H.; Park, J.K.; Wu, T.; Leo, D.J.; Winey, K.I.; Moore, R.B.; Long, T.E. Effect of ionic liquid on mechanical properties and morphology of zwitterionic copolymer membranes. Macromol. 2010, 43, 790–796. [Google Scholar] [CrossRef]
- Wu, T.; Beyer, F.L.; Brown, R.H.; Moore, R.B.; Long, T.E. Influence of zwitterions on thermomechanical properties and morphology of acrylic copolymers: Implications for electroactive applications. Macromol. 2011, 44, 8056–8063. [Google Scholar] [CrossRef]
- Bazuin, C.G.; Zheng, Y.L.; Muller, R.; Galin, J.C. Random ethyl acrylate-sulphonatopropylbetaine copolymers: 2. Dynamic mechanical properties. Polymer 1989, 30, 654–661. [Google Scholar] [CrossRef]
- Bahr, U.; Wieden, H.; Rinkler, H.A.; Nischk, G. Synthese neuer ungesättigter Comonomerer und ihre Copolymerisation mit Acrylnitril. Makromol. Chem. 1972, 161, 1–47. [Google Scholar] [CrossRef]
- Bonte, N.; Laschewsky, A.; Mayer, B.; Vermylen, V. Homogeneous mixtures of polybetaines with low molecular weight salts. Macromol. Symp. 1996, 102, 273–280. [Google Scholar]
- Bonte, N.; Laschewsky, A.; Vermylen, V. Hybrid materials made from polymeric betaines and low molar mass salts. Macromol. Symp. 1997, 117, 195–206. [Google Scholar] [CrossRef]
- Schulz, D.N.; Peiffer, D.G.; Agarwal, P.K.; Larabee, J.; Kaladas, J.J.; Soni, L.; Handwerker, B.; Garner, R.T. Phase behaviour and solution properties of sulphobetaine polymers. Polymer 1986, 27, 1734–1742. [Google Scholar]
- Johnston, D.S.; Sanghera, S.; Pons, M.; Chapman, D. Phospholipid polymers—Synthesis and spectral characteristics. Biochim. Biophys. Acta 1980, 602, 57–69. [Google Scholar] [CrossRef]
- Nakayama, T.; Li, Y.J. Phospholipid polymers. Prog. Polym. Sci. 1999, 24, 143–181. [Google Scholar] [CrossRef]
- Hub, H.H.; Hupfer, B.; Koch, H.; Ringsdorf, H. Polymerizable phospholipid analogues—New stable biomembrane and cell models. Angew. Chem. Int. Ed. 1980, 19, 938–940. [Google Scholar] [CrossRef]
- Ahlers, M.; Müller, W.; Reichert, A.; Ringsdorf, H.; Venzmer, J. Specific interactions of proteins with functional lipid monolayers—Ways of simulating biomembrane processes. Angew. Chem. Int. Ed. 1990, 29, 1269–1285. [Google Scholar] [CrossRef]
- O'Brien, D.F.; Whitesides, T.H.; Klingbiel, R.T. The photopolymerization of lipid-diacetylenes in bimolecular-layer membranes. J. Polym. Sci., Part C: Polym. Lett. Ed. 1981, 19, 95–101. [Google Scholar] [CrossRef]
- Regen, S.L.; Yamaguchi, K.; Samuel, N.K.P.; Singh, M. Polymerized-depolymerized vesicles. A reversible phosphatidylcholine-based membrane. J. Am. Chem. Soc. 1983, 105, 6354–6355. [Google Scholar] [CrossRef]
- Ruoslahti, E.; Pierschbacher, M.D. Arg-Gly-Asp: A versatile cell recognition signal. Cell 1986, 44, 517–518. [Google Scholar]
- Jiang, S.; Cao, Z. Ultralow-fouling, functionalizable, and hydrolyzable zwitterionic materials and their derivatives for biological applications. Adv. Mater. 2010, 22, 920–932. [Google Scholar] [CrossRef]
- Grainger, D.W. All charged up about implanted biomaterials. Nat. Biotechnol. 2013, 31, 507–509. [Google Scholar] [CrossRef]
- Mueller, A.; O'Brien, D.F. Supramolecular materials via polymerization of mesophases of hydrated amphiphiles. Chem. Rev. 2002, 102, 727–758. [Google Scholar] [CrossRef]
- Puri, A.; Blumenthal, R. Polymeric lipid assemblies as novel theranostic tools. Acc. Chem. Res. 2011, 44, 1071–1079. [Google Scholar] [CrossRef]
- Zhang, H.; Joubert, J.R.; Saavedra, S.S. Membranes from polymerizable lipids. Adv. Polym. Sci. 2010, 224, 1–42. [Google Scholar]
- Cashion, M.P.; Long, T.E. Biomimetic design and performance of polymerizable lipids. Acc. Chem. Res. 2009, 42, 1016–1025. [Google Scholar] [CrossRef]
- Chen, M.; Briscoe, W.H.; Armes, S.P.; Klein, J. Lubrication at physiological pressures by polyzwitterionic brushes. Science 2009, 323, 1698–1701. [Google Scholar] [CrossRef]
- Klein, J. Polymers in living systems: from biological lubrication to tissue engineering and biomedical devices. Polym. Adv. Tech. 2012, 23, 729–735. [Google Scholar] [CrossRef]
- Chen, M.; Briscoe, W.H.; Armes, S.P.; Cohen, H.; Klein, J. Polyzwitterionic brushes: Extreme lubrication by design. Eur. Polym. J. 2011, 47, 511–523. [Google Scholar] [CrossRef]
- Estephan, Z.G.; Schlenoff, P.S.; Schlenoff, J.B. Zwitteration as an alternative to PEGylation. Langmuir 2011, 27, 6794–6800. [Google Scholar] [CrossRef]
- Lin, P.; Lin, C.W.; Mansour, R.; Gu, F. Improving biocompatibility by surface modification techniques on implantable bioelectronics. Biosens. Bioelectron. 2013, 47, 451–460. [Google Scholar] [CrossRef]
- Binazadeh, M.; Kabiri, M.; Unsworth, L.D. Poly(ethylene glycol) and Poly(carboxy betaine) based nonfouling architectures: Review and current efforts. ACS Symp. Ser. 2012, 1120, 621–643. [Google Scholar]
- Cao, Z.; Jiang, S. Super-hydrophilic zwitterionic poly(carboxybetaine) and amphiphilic non-ionic poly(ethylene glycol) for stealth nanoparticles. Nano Today 2012, 7, 404–413. [Google Scholar] [CrossRef]
- Lopez-Donaire, M.L.; Santeree, J.P. Surface modifying oligomers used to functionalize polymeric surfaces: Consideration of blood contact applications. J. Appl. Polym. Sci. 2014, 131, 40328:1–40328:15. [Google Scholar]
- Schlenoff, J.B. Zwitteration: Coating surfaces with zwitterionic functionality to reduce nonspecific adsorption. Langmuir 2014, in press. [Google Scholar]
- Mary, P.; Bendejacq, D.D.; Labeau, M.P.; Dupuis, P. Reconciling low- and high-salt solution Behavior of sulfobetaine polyzwitterions. J. Phys. Chem. B 2007, 111, 7767–7777. [Google Scholar] [CrossRef]
- Monroy Soto, V.M.; Galin, J.C. Poly(sulphopropylbetaines): 2. Dilute solution properties. Polymer 1984, 25, 254–262. [Google Scholar] [CrossRef]
- Salamone, J.C.; Volksen, W.; Olson, A.P.; Israel, S.C. Aqueous solution properties of a poly(vinyl imidazolium sulphobetaine). Polymer 1978, 19, 1157–1162. [Google Scholar] [CrossRef]
- Köberle, P.; Laschewsky, A.; Lomax, T.D. Interactions of a zwitterionic polysoap and its cationic analog with inorganic salts. Macromol. Chem. Rapid Commun. 1991, 12, 427–433. [Google Scholar] [CrossRef]
- Arotçaréna, M.; Heise, B.; Ishaya, S.; Laschewsky, A. Switching the inside and the outside of aggregates of water-soluble block copolymers with double thermoresponsivity. J. Am. Chem. Soc. 2002, 124, 3787–3793. [Google Scholar]
- Maeda, Y.; Mochiduki, H.; Ikeda, I. Hydration changes during thermosensitive association of a block copolymer consisting of LCST and UCST blocks. Macromol. Rapid Commun. 2004, 25, 1330–1334. [Google Scholar] [CrossRef]
- Flores, J.D.; Xu, X.; Treat, N.J.; McCormick, C.L. Reversible “self-locked” micelles from a zwitterion-containing triblock copolymer. Macromol. 2009, 42, 4941–4945. [Google Scholar] [CrossRef]
- Wischerhoff, E.; Badi, N.; Laschewsky, A.; Lutz, J.F. Smart polymer surfaces: Concepts and applications in biosciences. Adv. Polym. Sci. 2011, 240, 1–33. [Google Scholar] [CrossRef]
- Roy, D.; Brooks, W.L.A.; Sumerlin, B.S. New directions in thermoresponsive polymers. Chem. Soc. Rev. 2013, 42, 7214–7243. [Google Scholar] [CrossRef]
- Cohen Stuart, M.A.; Huck, W.T.S.; Genzer, J.; Müller, M.; Ober, C.; Stamm, M.; Sukhorukov, G.B.; Szleifer, I.; Tsukruk, V.V.; Urban, M.; et al. Emerging applications of stimuli-responsive polymer materials. Nat. Mater. 2010, 9, 101–113. [Google Scholar] [CrossRef]
- Singh Paresh, K.; Singh Vinay, K.; Singh, M. Zwitterionic polyelectrolytes: A Review. e-Polym. 2007, 7, 335–368. [Google Scholar]
- Tarannum, N.; Singh, M. Advances in synthesis and applications of sulfo and carbo analogues of polybetaines: A Review. Rev. Adv. Sci. Eng. 2013, 2, 1–22. [Google Scholar]
- Armitage, B.A.; Bennett, D.E.; Lamparski, H.G.; O'Brien, D.F. Polymerization and domain formation in lipid assemblies. Adv. Polym. Sci. 1996, 126, 54–84. [Google Scholar]
- Xuan, F.; Liu, J. Preparation, characterization and application of zwitterionic polymers and membranes: current developments and perspective. Polym. Int. 2009, 58, 1350–1361. [Google Scholar] [CrossRef]
- Monroy Soto, V.M.; Galin, J.C. Poly(sulphopropylbetaines): 1. Synthesis and characterization. Polymer 1984, 25, 121–128. [Google Scholar] [CrossRef]
- Laschewsky, A.; Zerbe, I. Polymerizable and polymeric zwitterionic surfactants: 1. Synthesis and bulk properties polymer. Polymer 1991, 32, 2070–2080. [Google Scholar] [CrossRef]
- Huglin, M.B.; Radwan, M.A. Properties of poly[N-2-(methyacryloyloxy)ethyl-N,N-dimethyl-N-3-sulfopropylammonium betaine] in dilute solution. Macromol. Chem. 1991, 192, 2433–2445. [Google Scholar] [CrossRef]
- Strehmel, V.; Wetzel, H.; Laschewsky, A.; Moldenhauer, E.; Klein, T. Influence of imidazolium based ionic liquids on the synthesis of amphiphilic copolymers based on n-butylmethacrylate and a zwitterionic methacrylate. Polym. Adv. Tech. 2008, 19, 1383–1390. [Google Scholar]
- Strehmel, V.; Laschewsky, A.; Wetzel, H. Homopolymerization of a highly polar zwitterionic methacrylate in ionic liquids and its copolymerization with a non-polar methacrylate. e-Polym 2006, 6, 131–140. [Google Scholar]
- Chen, X.; McRae, S.; Samanta, D.; Emrick, T. Polymer-protein conjugation in ionic liquids. Macromol. 2010, 43, 6261–6263. [Google Scholar] [CrossRef]
- Ostermayer, B.; Albrecht, O.; Vogt, W. Polymerizable lipid analogs of diacetylenic phosphonic acids. Synthesis, spreading behavior and polymerization at the gas-water interface. Chem. Phys. Lipids 1986, 41, 265–291. [Google Scholar] [CrossRef]
- Hamaide, T.; Germanaud, L.; Perchec, P.L. New polymeric phosphonato-, phosphinato- and carboxybetaïnes, 1. Syntheses and characterization by IR spectroscopy. Macromol. Chem. Phys. 1986, 187, 1097–1107. [Google Scholar] [CrossRef]
- Al-Hamouz, O.C.S.; Ali, S.A. pH-responsive polyphosphonates using Butler's cyclopolymerization. J. Polym. Sci. Part A: Polym. Chem. 2012, 50, 3580–3591. [Google Scholar] [CrossRef]
- Buchweitz, K. Synthese und Charakterisierung neuartiger Polysulfobetaine. Ph.D. Thesis, Technische Universität Berlin, Technische Universität Berlin (Germany), 2000. [Google Scholar]
- Pujol-Fortin, M.L.; Galin, J.C. Poly(ammonium alkoxydicyanoethenolates) as new hydrophobic and highly dipolar. Macromol. 1991, 24, 4523–4530. [Google Scholar] [CrossRef]
- Grassl, B.; Meurer, B.; Scheer, M.; Galin, J.C. Segmented poly(tetramethylene oxide) zwitterionomers and their homologous ionenes. 2. Phase separation through DSC and solid state 1H-NMR spectroscopy. Macromol 1997, 30, 236–245. [Google Scholar] [CrossRef]
- Bieglé, A.; Mathis, A.; Galin, J.C. Towards highly functionalized and semi-rigid polyzwitterions, 1. Poly(dizwitterionic methacrylates): Synthesis and specific properties. Macromol. Chem. Phys. 1999, 200, 1393–1406. [Google Scholar] [CrossRef]
- Bieglé, A.; Mathis, A.; Galin, J.C. Towards highly functionalized and semi-rigid polyzwitterions, 2. Poly(zwitterionic isocyanides): Synthesis and specific properties. Macromol. Chem. Phys. 2000, 201, 113–125. [Google Scholar] [CrossRef]
- Xue, C.; Cai, F.; Liu, H. Ultrasensitive fluorescent responses of water-soluble, zwitterionic, boronic acid-bearing, regioregular head-to-tail polythiophene to biological species. Chem. Eur. J. 2008, 14, 1648–1653. [Google Scholar] [CrossRef]
- Görtz, V.; Ritter, H. New photosensitive polymers: Synthesis and free radical polymerization of oxypyridinium and oxyisoquinolinium functionalized methacrylate and styrene derivatives. Macromol. 2002, 35, 4258–4265. [Google Scholar] [CrossRef]
- Yoshizawa, M.; Hirao, M.; Ito-Akita, K.; Ohno, H. Ion conduction in zwitterionic-type molten salts and their polymers. J. Mater. Chem. 2001, 11, 1057–1062. [Google Scholar] [CrossRef]
- Weers, J.G.; Rathman, J.F.; Axe, F.U.; Crichlow, C.A.; Foland, L.D.; Scheuing, D.R.; Wiersema, R.J.; Zielske, A.G. Effect of the intramolecular charge separation distance on the solution properties of betaines and sulfobetaines. Langmuir 1991, 7, 854–867. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, H.; Wang, J. Effects of alkyl chain length and solvents on thermodynamic dissociation constants of the ionic liquids with one carboxyl group in the alkyl chain of imidazolium cations. J. Phys. Chem. B 2014, 118, 4630–4635. [Google Scholar] [CrossRef]
- Wiederkehr, N.A.; Kalyanasundaram, K.; Graetzel, M.; Viscardi, G.; Savarino, P.; Barni, E. Micellization properties of zwitterionic surfactants derived from nicotinic acid in aqueous solutions. Langmuir 1991, 7, 23–29. [Google Scholar] [CrossRef]
- Izumrudov, V.A.; Domashenko, N.I.; Zhiryakova, M.V.; Davydova, O.V. Interpolyelectrolyte reactions in solutions of polycarboxybetaines, 2: Influence of alkyl spacer in the betaine moieties on complexing with polyanions. J. Phys. Chem. B 2005, 119, 17391–17399. [Google Scholar]
- Bohrisch, J.; Schimmel, T.; Engelhardt, H.; Jaeger, W. Charge interaction of synthetic polycarboxybetaines in bulk and solution. Macromol. 2002, 35, 4143–4149. [Google Scholar] [CrossRef]
- Schwarz, S.; Jaeger, W.; Bratskaya, S.; Bohrisch, J.; Schimmel, T.; Mende, M.; Oelmann, M.; Boyko, V. Formation of polyelectrolyte complexes in a polycarboxybetaine/weak polyanion system. Colloid Surf. A 2006, 276, 65–71. [Google Scholar] [CrossRef]
- Herrmann, K.W. Micellar properties of some zwitterionic surfactants. J. Coll. Interf. Sci. 1966, 22, 352–359. [Google Scholar] [CrossRef]
- Tarannum, N.; Singh, M. Synthesis and characterization of zwitterionic organogels based on Schiff base chemistry. J. Appl. Polym. Sci. 2010, 118, 2821–2832. [Google Scholar] [CrossRef]
- Pluzhnov, S.K.; Lents, R.V.; Topchiev, D.A.; Kabanov, V.A. Synthesis of new ionic monomers from α-bromomethacrylic acid. Russ. Chem. Bull. 1982, 31, 176–177. [Google Scholar] [CrossRef]
- Nilsson, P.G.; Lindman, B.; Laughlin, R.G. The upper consolute boundary in zwitterionic surfactant-water systems. J. Phys. Chem. 1984, 88, 6357–6362. [Google Scholar] [CrossRef]
- Avci, D.; Mathias, L.J. Synthesis and photopolymerizations of phosphate-containing acrylate/(di)methacrylate monomers from 3-(acryloyloxy)-2-hydroxypropyl methacrylate. Polym. Bull. 2005, 54, 11–19. [Google Scholar] [CrossRef]
- Garg, G.; Chauhan, G.S.; Gupta, R.; Ahn, J.H. Anion effects on anti-microbial activity of poly[1-vinyl-3-(2-sulfoethyl imidazolium betaine)]. J. Coll. Interf. Sci. 2010, 344, 90–96. [Google Scholar] [CrossRef]
- Sonnenschein, L.; Seubert, A. Synthesis of a series of monomeric styrene sulfobetaine precursors. Tetrahedr. Lett. 2011, 52, 1101–1104. [Google Scholar] [CrossRef]
- Bonte, N.; Laschewsky, A. Zwitterionic polymers with carbobetaine moieties. Polymer 1996, 37, 2011–2019. [Google Scholar] [CrossRef]
- Favresse, P.; Laschewsky, A. New poly(carbobetaines) made from zwitterionic diallylammonium monomers. Macromol. Chem. Phys. 1999, 200, 887–895. [Google Scholar] [CrossRef]
- Favresse, P.; Laschewsky, A. Synthesis and investigation of new amphiphilic poly(carbobetaine)s made from diallylammonium monomers. Polymer 2001, 42, 2755–2766. [Google Scholar] [CrossRef]
- Chevalier, Y.; Storet, Y.; Pourchet, S.; Le Perchec, P. Tensioactive properties of zwitterionic carboxybetaine amphiphiles. Langmuir 1991, 7, 848–853. [Google Scholar] [CrossRef]
- Wielema, T.A.; Engberts, J.B. Zwitterionic polymers—II. Synthesis of a novel series of poly(vinylbetaines) and the effect of the polymeric structure on the solubility behaviour in water. Eur. Polym. J. 1990, 26, 415–421. [Google Scholar] [CrossRef]
- Wielema, T.A.; Engberts, J.B. Zwitterionic polymers—I. Synthesis of a novel series of poly(vinylsulphobetaines). Effect of structure of polymer on solubility in water. Eur. Polym. J. 1987, 23, 947–950. [Google Scholar] [CrossRef]
- Barboiu, V.; Holerca, M.N.; Streba, E.; Luca, C. Reactions on polymers with amine groups. III. Description of the addition reaction of pyridine and imidazole compounds with α,β-unsaturated monocarboxylic acids. J. Polym. Sci., Part A: Polym. Chem. 1996, 34, 261–270. [Google Scholar]
- Avci, D.; Mathias, L.J. Synthesis and cyclopolymerization of novel allyl-acrylate quaternary ammonium salts. J. Polym. Sci., Part A: Polym. Chem. 1999, 37, 901–907. [Google Scholar] [CrossRef]
- Avci, D.; Lemopulo, K.; Mathias, L.J. Cyclocopolymerization of allyl-acrylate quaternary ammonium salts with diallyldimethylammonium chloride. J. Polym. Sci., Part A: Polym. Chem. 2001, 39, 640–649. [Google Scholar] [CrossRef]
- Barboiu, V.; Streba, E.; Luca, C.; Radu, I.; Grigoriu, G.E. Reactions on polymers with amine groups. V. Addition of pyridine and imidazole groups with acetylenecarboxylic acids. J. Polym. Sci., Part A: Polym. Chem. 1998, 36, 1615–1623. [Google Scholar] [CrossRef]
- Xing, C.; Liu, L.; Shi, Z.; Li, Y.; Wang, S. Synthesis of zwitterionic water-soluble oligofluorenes with good light-harvesting ability. Adv. Funct. Mater. 2010, 20, 2175–2180. [Google Scholar] [CrossRef]
- Duan, C.; Wang, L.; Zhang, K.; Guan, X.; Huang, F. Conjugated zwitterionic polyelectrolytes and their neutral precursor as electron injection layer for high-performance polymer light-emitting diodes. Adv. Mater. 2011, 23, 1665–1669. [Google Scholar] [CrossRef]
- Ji, E.; Whitten, D.G.; Schanze, K.S. pH-dependent optical properties of a poly(phenylene ethynylene) conjugated polyampholyte. Langmuir 2011, 27, 1565–1568. [Google Scholar] [CrossRef]
- Scherf, U. Counterion pinning in conjugated polyelectrolytes for applications in organic electronics. Angew. Chem. Int. Ed. 2011, 50, 5016–5017. [Google Scholar] [CrossRef]
- Fang, J.; Wallikewitz, B.H.; Gao, F.; Tu, G.; Müller, C.; Pace, G.; Friend, R.H.; Huck, W.T.S. Conjugated zwitterionic polyelectrolyte as the charge injection layer for high-performance polymer light-emitting diodes. J. Am. Chem. Soc. 2011, 133, 683–685. [Google Scholar]
- Maeda, T.; Tsukamoto, T.; Seto, A.; Yagi, S.; Nakazumi, H. Synthesis and characterization of squaraine-based conjugated polymers with phenylene linkers for bulk heterojunction solar cells. Macromol. Chem. Phys. 2012, 213, 2590–2597. [Google Scholar] [CrossRef]
- Duan, C.; Zhang, K.; Guan, X.; Zhong, C.; Xie, H.; Huang, F.; Chen, J.; Peng, J.; Cao, Y. Conjugated zwitterionic polyelectrolyte-based interface modification materials for high performance polymer optoelectronic devices. Chem. Sci. 2013, 4, 1298–1307. [Google Scholar] [CrossRef]
- Kumar, A.; Pace, G.; Bakulin, A.A.; Fang, J.; Ho, P.K.H.; Huck, W.T.S.; Friend, R.H.; Greenham, N.C. Donor–acceptor interface modification by zwitterionic conjugated polyelectrolytes in polymer photovoltaics. Energy Environ. Sci. 2013, 6, 1589–1596. [Google Scholar] [CrossRef]
- Liu, F.; Page, Z.A.; Duzhko, V.V.; Russell, T.P.; Emrick, T. Conjugated polymeric zwitterions as efficient interlayers in organic solar cells. Adv. Mater. 2013, 25, 6868–6873. [Google Scholar] [CrossRef]
- Page, Z.A.; Duzhko, V.V.; Emrick, T. Conjugated thiophene-containing polymer zwitterions: direct synthesis and thin film electronic properties. Macromol. 2013, 46, 344–351. [Google Scholar] [CrossRef]
- Laschewsky, A. Recent trends in the synthesis of polyelectrolytes. Curr. Opin. Coll. Interf. Sci. 2012, 17, 56–63. [Google Scholar] [CrossRef]
- Matyjaszewski, K.; Müller, A.H.E. Controlled and Living Polymerizations. From Mechanisms to Applications; Wiley-VCH: Weinheim, Germany, 2009. [Google Scholar]
- Jenkins, A.D.; Jones, R.G.; Moad, G. Terminology for reversible-deactivation radical polymerization previously called “controlled” or “living” radical polymerization. Pure Appl. Chem. 2010, 82, 483–491. [Google Scholar]
- Matyjaszewski, K.; Davis, T.P. Handbook of Radical Polymerization; John Wiley and Sons, Inc.: Hoboken, NJ, USA, 2002. [Google Scholar]
- Barner-Kowollik, C. Handbook of RAFT Polymerization; Wiley-VCH: Weinheim, Germany, 2008. [Google Scholar]
- Grubbs, R.H. Olefin-metathesis catalysts for the preparation of molecules and materials (Nobel Lecture). Angew. Chem. Int. Ed. 2006, 45, 3760–3765. [Google Scholar] [CrossRef]
- Buchmeiser, M.R. Polymer-supported well-defined metathesis catalysts. Chem. Rev. 2008, 109, 303–321. [Google Scholar] [CrossRef]
- Bielawski, C.W.; Grubbs, R.H. Living ring-opening metathesis polymerization. Prog. Polym. Sci. 2007, 32, 1–29. [Google Scholar] [CrossRef]
- Wang, Z.; Ma, G.; Zhang, J.; Lin, W.; Ji, F.; Bernards, M.T.; Chen, S. Development of zwitterionic polymer-based doxorubicin conjugates: Tuning the surface charge to prolong the circulation and reduce toxicity. Langmuir 2014, 30, 3764–3774. [Google Scholar] [CrossRef]
- Liu, Y.X.; Kang, E.T.; Neoh, K.G.; Tan, K.L.; Huang, C.C.; Liaw, D.J. Lamination of polytetrafluoroethylene films via surface thermal graft copolymerization with ionic and zwitterionic monomers. J. Appl. Polym. Sci. 1999, 74, 816–824. [Google Scholar] [CrossRef]
- Xue, W.; Huglin, M.B.; Russell, A.T. Unusual behaviour of crosslinked and linear forms of a zwitterionic polymer in aqueous alkali. Macromol. Rapid Commun. 1999, 20, 239–243. [Google Scholar] [CrossRef]
- Xue, W.; Champ, S.; Huglin, M.B. Thermoreversible swelling behaviour of hydrogels based on N-isopropylacrylamide with a zwitterionic comonomer. Eur. Polym. J. 2001, 37, 869–875. [Google Scholar] [CrossRef]
- Xue, W.; Huglin, M.B.; Liao, B. Observations on the swelling characteristics of the zwitterionic hydrogel of poly(1–3-sulfopropyl)-2-vinyl-pyridinium-betaine hydrogel. Eur. Polym. J. 2006, 42, 3015–3023. [Google Scholar] [CrossRef]
- Xue, W.; Huglin, M.B.; Liao, B.; Jones, T.G.J. Swelling behaviour of crosslinked hydrogels based on (2-hydroxyethyl methacrylate) with a zwitterionic comonomer (1-3-sulfopropyl-2-vinyl-pyridinium-betaine). Eur. Polym. J. 2007, 43, 915–927. [Google Scholar] [CrossRef]
- Mertoglu, M.; Laschewsky, A.; Skrabania, K.; Wieland, C. New water soluble agents for reversible addition-fragmentation chain transfer polymerization and their application in aqueous solutions. Macromol. 2005, 38, 3601–3614. [Google Scholar] [CrossRef]
- Laschewsky, A.; Zerbe, I. Polymerizable and polymeric zwitterionic surfactants: 2. Surface activity and aggregation behaviour in aqueous systems. Polymer 1991, 32, 2081–2086. [Google Scholar] [CrossRef]
- Köberle, P.; Laschewsky, A.; van den Boogaard, D. Self-organization of hydrophobized polyzwitterions. Polymer 1992, 33, 4029–4039. [Google Scholar] [CrossRef]
- Anton, P.; Laschewsky, A. Solubilization by polysoaps. Colloid Polym. Sci. 1994, 272, 1118–1128. [Google Scholar] [CrossRef]
- Köberle, P.; Laschewsky, A. Hydrophobically modified zwitterionic polymers: Synthesis, bulk properties, and miscibility with inorganic salts. Macromol. 1994, 27, 2165–2179. [Google Scholar] [CrossRef]
- Köberle, P.; Laschewsky, A. Hybrid materials from organic polymers and inorganic salts. Macromol. Symp. 1994, 88, 165–175. [Google Scholar] [CrossRef]
- Laschewsky, A. Oligoethyleneoxide spacer groups in polymerizable surfactants. Colloid Polym. Sci. 1991, 269, 785–794. [Google Scholar] [CrossRef]
- Laschewsky, A.; Touillaux, R.; Hendlinger, P.; Vierengel, A. Characterization of sulfobetaine monomers by nuclear magnetic resonance spectroscopy: A note. Polymer 1995, 36, 3045–3049. [Google Scholar] [CrossRef]
- Cao, Z.; Brault, N.; Xue, H.; Keefe, A.; Jiang, S. Manipulating sticky and non-sticky properties in a single material. Angew. Chem. Int. Ed. 2011, 50, 6102–6104. [Google Scholar]
- Zhang, Z.; Vaisocherovà, H.; Cheng, G.; Yang, W.; Xue, H.; Jiang, S. Nonfouling behavior of polycarboxybetaine-grafted surfaces: Structural and environmental effects. BioMacromol. 2008, 9, 2686–2692. [Google Scholar] [CrossRef]
- Abraham, S.; Unsworth, L.D. Multi-functional initiator and poly(carboxybetaine methacrylamides) for building biocompatible surfaces using “nitroxide mediated free radical polymerization” strategies. J. Polym. Sci. Part A: Polym. Chem. 2011, 49, 1051–1060. [Google Scholar] [CrossRef]
- Hupfer, B.; Ringsdorf, H.; Schupp, H. Polyreactions in oriented systems, 21. Polymeric phospholipid monolayers. Macromol. Chem. 1981, 182, 247–253. [Google Scholar] [CrossRef]
- Dorn, K.; Klingbiel, R.T.; Specht, D.P.; Tyminski, P.N.; Ringsdorf, H.; O'Brien, D.F. Permeability characteristics of polymeric bilayer membranes from methacryloyl and butadiene lipids. J. Am. Chem. Soc. 1984, 106, 1627–1633. [Google Scholar] [CrossRef]
- Tyminski, P.N.; Ponticello, I.S.; O'Brien, D.F. Polymerizable dienoyl lipids as spectroscopic bilayer membrane probes. J. Am. Chem. Soc. 1987, 109, 6541–6542. [Google Scholar]
- Anton, P.; Laschewsky, A. Zwitterionic polysoaps with reduced density of surfactant side groups. Macromol. Chem. 1993, 194, 601–624. [Google Scholar] [CrossRef]
- Subramaniam, V.; D’Ambruoso, G.D.; Hall, H.K.; Wysocki, R.J.; Brown, M.F.; Saavedra, S.S. Reconstitution of rhodopsin into polymerizable planar supported lipid bilayers: Influence of dienoyl monomer structure on photoactivation. Langmuir 2008, 24, 11067–11075. [Google Scholar] [CrossRef]
- Heitz, B.A.; Xu, J.; Hall, H.K.; Aspinwall, C.A.; Saavedra, S.S. Enhanced long-term stability for single ion channel recordings using suspended poly(lipid) bilayers. J. Am. Chem. Soc. 2009, 131, 6662–6663. [Google Scholar]
- Joubert, J.R.; Smith, K.A.; Johnson, E.; Keogh, J.P.; Wysocki, V.H.; Gale, B.K.; Conboy, J.C.; Saavedra, S.S. Stable, ligand-doped, poly(bis-SorbPC) lipid bilayer arrays for protein binding and detection. ACS Appl. Mater. Interf. 2009, 1, 1310–1315. [Google Scholar] [CrossRef]
- Muhandiramlage, T.P.; Cheng, Z.; Roberts, D.L.; Keogh, J.P.; Hall, H.K.; Aspinwall, C.A. Determination of pore sizes and relative porosity in porous nanoshell architectures using dextran retention with single monomer resolution and proton permeation. Anal. Chem. 2012, 84, 9754–9761. [Google Scholar]
- Heitz, B.A.; Xu, J.; Jones, I.W.; Keogh, J.P.; Comi, T.J.; Hall, H.K.; Aspinwall, C.A.; Saavedra, S.S. Polymerized planar suspended lipid bilayers for single ion channel recordings: Comparison of several dienoyl lipids. Langmuir 2011, 27, 1882–1890. [Google Scholar] [CrossRef]
- Ali, M.M.; Perzanowski, H.P.; Ali, S.A. Polymerization of functionalized diallyl quaternary ammonium salt to poly(ampholyte–electrolyte). Polymer 2000, 41, 5591–5600. [Google Scholar] [CrossRef]
- Ali, S.A.; Al-Muallem, H.A.; Mazumder, M.A.J. Synthesis and solution properties of a new sulfobetaine/sulfur dioxide copolymer and its use in aqueous two-phase polymer systems. Polymer 2003, 44, 1671–1679. [Google Scholar]
- Ali, S.A.; Mazumder, M.A.J.; Al-Muallem, H.A. Synthesis and solution properties of a new pH-responsive polymer containing amino propanesulfonic acid residues. J. Polym. Sci. Part A: Polym. Chem. 2003, 41, 172–184. [Google Scholar] [CrossRef]
- Thomas, D.B.; Vasilieva, Y.A.; Armentrout, R.S.; McCormick, C.L. Synthesis, characterization, and aqueous solution behavior of electrolyte- and pH-responsive carboxybetaine-containing cyclocopolymer. Macromol. 2003, 36, 9710–9715. [Google Scholar] [CrossRef]
- Umar, Y.; Abu-Sharkh, B.F.; Ali, S.A. The effects of zwitterionic and anionic charge densities in polymer chains on the viscosity behavior of a pH-responsive hydrophobically modified ionic polymer. J. Appl. Polym. Sci. 2005, 98, 1404–1411. [Google Scholar] [CrossRef]
- Umar, Y.; Abu-Sharkh, B.F.; Ali, S.A. The effects of charge densities on the associative properties of a pH-responsive hydrophobically modified sulfobetaine/sulfur dioxide terpolymer. Polymer 2005, 43, 10709–10717. [Google Scholar]
- Abu-Thabit, N.Y.; Kazi, I.W.; Al-Muallem, H.A.; Ali, S.A. Phosphonobetaine/sulfur dioxide copolymer by Butler’s cyclopolymerization process. Eur. Polym. J. 2011, 47, 1113–1123. [Google Scholar] [CrossRef]
- Abu-Thabit, N.Y.; Al-Muallem, H.A.; Ali, S.A. The pH-responsive cycloterpolymers of diallyldimethylammonium chloride, 3-(N,N-diallylammonio)propanesulfonate, and sulfur dioxide. J. Appl. Polym. Sci. 2011, 120, 3662–3672. [Google Scholar] [CrossRef]
- Lezov, A.V.; Vlasov, P.S.; Lezov, A.A.; Domnina, N.S.; Polushina, G.E. Molecular properties of poly(carboxybetaine) in solutions with different ionic strengths and pH values. Polym. Sci., Ser. A 2011, 53, 1012–1018. [Google Scholar] [CrossRef]
- Lezov, A.A.; Vlasov, P.S.; Polushina, G.E.; Lezov, A.V. Effect of chemical structure and charge distribution on behavior of polyzwitterions in solution. Macromol. Symp. 2012, 316, 17–24. [Google Scholar] [CrossRef]
- Ali, S.A.; Haladu, S.A. Cyclopolymerization protocol for the synthesis of a poly(zwitterion-alt-sulfur dioxide) to investigate the polyzwitterion-to-poly(anion-zwitterion) transition. J. Appl. Polym. Sci. 2013, 129, 1394–1404. [Google Scholar] [CrossRef]
- Haladu, S.A.; Ali, S.A.A. pH-responsive cyclopolymer having phospho- and sulfopropyl pendents in the same repeating unit: synthesis, characterization, and its application as an antiscalant. J. Polym. Sci., Part A: Polym. Chem. 2013, 51, 5130–5142. [Google Scholar] [CrossRef]
- Haladu, S.A.; Ali, S.A. Cyclopolymerization protocol for the synthesis of a new poly(electrolyte-zwitterion) containing quaternary nitrogen, carboxylate, and sulfonate functionalities. Eur. Polym. J. 2013, 49, 1591–1600. [Google Scholar] [CrossRef]
- Ali, S.A.; Kazi, I.W.; Rahman, F. Synthesis of a diallylammonio propanephosphonate-alt-(sulfur dioxide) copolymer and its evaluation as an antiscalant in desalination plants. Polym. Int. 2014, 63, 616–625. [Google Scholar]
- Oishi, T.; Yoshimura, Y.; Yamasaki, H.; Onimura, K. Synthesis and polymerization of methacrylate bearing a phosphorylcholine, analogous moiety. Polym. Bull. 2001, 47, 121–126. [Google Scholar] [CrossRef]
- Åbele, S.; Sjöberg, M.; Hamaide, T.; Zicmanis, A.; Guyot, A. Reactive surfactants in heterophase polymerization. 10. Characterization of the surface activity of new polymerizable surfactants derived from maleic anhydride. Langmuir 1997, 13, 176–181. [Google Scholar] [CrossRef]
- Åbele, S.; Zicmanis, A.; Graillat, C.; Guyot, A. Cationic and zwitterionic polymerizable surfactants quaternary ammonium dialkyl maleates. 2. Emulsion polymerization of styrene and butyl acrylate. Langmuir 1999, 15, 1045–1051. [Google Scholar] [CrossRef]
- Favresse, P.; Laschewsky, A.; Emmermann, C.; Gros, L.; Linsner, A. Synthesis and free radical copolymerisation of new zwitterionic monomers: Amphiphilic carbobetaines based on isobutylene. Eur. Polym. J. 2001, 37, 877–885. [Google Scholar] [CrossRef]
- Anton, P.; Laschewsky, A. Polysoaps via alternating olefin/SO2 copolymers. Macromol Rapid Commun. 1991, 12, 189–196. [Google Scholar] [CrossRef]
- Sonnenschein, L.; Seubert, A. Separation of inorganic anions using a series of sulfobetaine exchangers. J. Chromatogr. A 2011, 1218, 1185–1194. [Google Scholar] [CrossRef]
- Salamone, J.C.; Watterson, A.C.; Hsu, T.D.; Tsai, C.C.; Mahmud, M.U.; Wisniewski, A.W.; Israel, S.C. Polymerization of vinylpyridinium salts. X. Copolymerization studies of cationicanionic monomer pairs. J. Polym. Sci.: Polym. Symp. 1978, 64, 229–243. [Google Scholar]
- Carr, L.; Cheng, G.; Xue, H.; Jiang, S. Engineering the polymer backbone to strengthen nonfouling sulfobetaine hydrogels. Langmuir 2010, 26, 14793–14798. [Google Scholar] [CrossRef]
- Gui, Z.; Qian, J.; An, Q.; Xu, H.; Zhao, Q. Synthesis, characterization and flocculation performance of zwitterionic copolymer of acrylamide and 4-vinylpyridine propylsulfobetaine. Eur. Polym. J. 2009, 45, 1403–1411. [Google Scholar] [CrossRef]
- Donovan, M.S.; Sumerlin, B.S.; Lowe, A.B.; McCormick, C.L. Controlled/“living” polymerization of sulfobetaine monomers directly in aqueous media via RAFT. Macromol. 2002, 35, 8663–8666. [Google Scholar]
- Liu, P.S.; Chen, Q.; Liu, X.; Yuan, B.; Wu, S.S.; Shen, J.; Lin, S.C. Grafting of zwitterion from cellulose membranes via ATRP for improving blood compatibility. BioMacromol. 2009, 10, 2809–2816. [Google Scholar] [CrossRef]
- Liu, P.S.; Chen, Q.; Wu, S.S.; Shen, J.; Lin, S.C. Surface modification of cellulose membranes with zwitterionic polymers for resistance to protein adsorption and platelet adhesion. J. Membr. Sci. 2010, 350, 387–394. [Google Scholar] [CrossRef]
- Mertoglu, M.; Garnier, S.; Laschewsky, A.; Skrabania, K.; Storsberg, J. Stimuli responsive amphiphilic block copolymers for aqueous media synthesised via reversible addition fragmentation chain transfer polymerisation (RAFT). Polymer 2005, 46, 7726–7740. [Google Scholar] [CrossRef]
- Wang, D.; Wu, T.; Wan, X.; Wang, X.; Liu, S. Purely salt-responsive micelle formation and inversion based on a novel schizophrenic sulfobetaine block copolymer: structure and kinetics of micellization. Langmuir 2007, 23, 11866–11874. [Google Scholar] [CrossRef]
- Vasantha, V.A.; Jana, S.; Parthiban, A.; Vancso, J.G. Water swelling, brine soluble imidazole based zwitterionic polymers—Synthesis and study of reversible UCST behaviour and gel-sol transitions. Chem. Commun. 2014, 50, 46–48. [Google Scholar] [CrossRef]
- Rodríguez-Hidalgo, M.d.R.; Soto-Figueroa, C.; Vicente, L. Mesoscopic study of salt-responsive polymeric micelles: structural inversion mechanisms via sequential addition of inorganic salts. Soft Matter 2013, 9, 5762–5770. [Google Scholar] [CrossRef]
- Cardoso, J.; Vázquez, G.; García, P.R.; González, I. Morphology, dielectric and thermal properties of poly(sulfobutylbetaine)/montmorillonite (PMBS-4/MMT) nanocomposites as solid polymer electrolytes. Macromol. Symp. 2013, 325/326, 156–167. [Google Scholar] [CrossRef]
- Cardoso, J.; Montiel, R.; Huanosta-Tera, A. Microscopic features of methacrylate polymers with zwitterionic pendant groups determined by various techniques. J. Polym. Sci. Part B: Polym. Phys. 2005, 43, 1152–1160. [Google Scholar] [CrossRef]
- Bardts, M.; Ritter, H. Novel copolymers showing interactions of amidinium-carboxylate groups in water. J. Polym. Res. 2011, 18, 151–155. [Google Scholar] [CrossRef]
- Kim, Y.; Binauld, S.; Stenzel, M.H. Zwitterionic guanidine-based oligomers mimicking cell-penetrating peptides as a nontoxic alternative to cationic polymers to enhance the cellular uptake of micelles. BioMacromol. 2012, 13, 3418–3426. [Google Scholar] [CrossRef]
- Al-Muallem, H.A. Polyzwitterion-to-polyampholyte transition using pH-responsive cycloterpolymers of diallyldimethylammonium chloride, (N,N-diallylammonio)methanecarboxylate and sulfur dioxide. J. Appl. Polym. Sci. 2012, 125, 1959–1969. [Google Scholar] [CrossRef]
- Sugiyama, K.; Ohga, K. Surface modified poly(methyl methacrylate) with 1-methyl- 2-methacrylamidoethyl phosphorylcholine moiety. Macromol. Chem. Phys. 1999, 200, 1439–1445. [Google Scholar] [CrossRef]
- Goda, T.; Watanabe, J.; Takai, M.; Ishihara, K. Water structure and improved mechanical properties of phospholipid polymer hydrogel with phosphorylcholine centered intermolecular cross-linker. Polymer 2006, 47, 1390–1396. [Google Scholar] [CrossRef]
- Goda, T.; Furukawa, H.; Gong, J.P.; Ishihara, K. Relaxation modes in chemically cross-linked poly(2-methacryloyloxyethyl phosphorylcholine) hydrogels. Soft Matter 2013, 9, 2166–2171. [Google Scholar] [CrossRef]
- Kiritoshi, Y.; Ishihara, K. Synthesis of hydrophilic cross-linker having phosphorylcholine-like linkage for improvement of hydrogel properties. Polymer 2004, 45, 7499–7504. [Google Scholar] [CrossRef]
- Kasák, P.; Kroneková, Z.; Krupa, I.; Lacík, I. Zwitterionic hydrogels crosslinked with novel zwitterionic crosslinkers: Synthesis and characterization. Polymer 2011, 52, 3011–3020. [Google Scholar] [CrossRef]
- Carr, L.R.; Zhou, Y.; Krause, J.E.; Xue, H.; Jiang, S. Uniform zwitterionic polymer hydrogels with a nonfouling and functionalizable crosslinker using photopolymerization. Biomater. 2011, 32, 6893–6899. [Google Scholar] [CrossRef]
- Zhang, L.; Cao, Z.; Bai, T.; Carr, L.; Ella-Menye, J.R.; Irvin, C.; Ratner, B.D.; Jiang, S. Zwitterionic hydrogels implanted in mice resist the foreign-body reaction. Nat. Biotechn. 2013, 31, 553–557. [Google Scholar] [CrossRef]
- Lobb, E.J.; Ma, I.; Billingham, N.C.; Armes, S.P.; Lewis, A.L. Facile synthesis of well-defined, biocompatible phosphorylcholine-based methacrylate copolymers via atom transfer radical polymerization at 20 °C. J. Am. Chem. Soc. 2001, 123, 7913–7914. [Google Scholar] [CrossRef]
- McCormick, C.; Lowe, A.B. Aqueous RAFT polymerization: Recent developments in synthesis of functional water-soluble (co)polymers with controlled structures. Acc. Chem. Res. 2004, 37, 312–325. [Google Scholar] [CrossRef]
- Miyamoto, D.; Watanabe, J.; Ishihara, K. Highly stabilized papain conjugated with water-soluble phospholipid polymer chain having a reacting terminal group. J. Appl. Polym. Sci. 2004, 91, 827–832. [Google Scholar] [CrossRef]
- Ding, W.; Lv, C.; Sun, Y.; Liu, X.; Yu, T.; Qu, G.; Luan, H. Synthesis of zwitterionic polymer by SET-LRP at room temperature in aqueous. J. Polym. Sci. Part A: Polym. Chem. 2011, 49, 432–440. [Google Scholar] [CrossRef]
- Terayama, Y.; Kikuchi, M.; Kobayashi, M.; Takahara, A. Well-defined poly(sulfobetaine) brushes prepared by surface-initiated ATRP using a fluoroalcohol and ionic liquids as the solvents. Macromol. 2011, 44, 104–111. [Google Scholar] [CrossRef]
- Rodriguez-Emmenegger, C.; Schmidt, B.V.K.J.; Sedlakova, Z.; Šubr, V.; Alles, A.B.; Brynda, E.; Barner-Kowollik, C. Low Temperature aqueous living/controlled (RAFT) polymerization of carboxybetaine methacrylamide up to high molecular weights. Macromol. Rapid Commun. 2011, 32, 958–965. [Google Scholar] [CrossRef]
- Willcock, H.; Lu, A.; Hansell, C.F.; Chapman, E.; Collins, I.R.; O'Reilly, R.K. One-pot synthesis of responsive sulfobetaine nanoparticles by RAFT polymerisation: the effect of branching on the UCST cloud point. Polym. Chem. 2014, 5, 1023–1030. [Google Scholar] [CrossRef]
- Bhuchar, N.; Deng, Z.; Ishihara, K.; Narain, R. Detailed study of the reversible addition–fragmentation chain transfer polymerization and co-polymerization of 2-methacryloyloxyethyl phosphorylcholine. Polym. Chem. 2011, 2, 632–639. [Google Scholar] [CrossRef]
- Cai, Y.; Armes, S.P. Synthesis of well-defined Y-shaped zwitterionic block copolymers via atom-transfer radical polymerization. Macromol. 2004, 38, 271–279. [Google Scholar] [CrossRef]
- Xu, J.P.; Ji, J.; Chen, W.D.; Shen, J.C. Novel biomimetic surfactant: Synthesis and micellar characteristics. Macromol. Biosci. 2005, 5, 164–171. [Google Scholar] [CrossRef]
- Chen, X.; McRae, S.; Parelkar, S.; Emrick, T. Polymeric phosphorylcholine−camptothecin conjugates prepared by controlled free radical polymerization and click chemistry. Bioconj. Chem. 2009, 20, 2331–2341. [Google Scholar] [CrossRef]
- Yu, H.Y.; Kang, Y.; Liu, Y.; Mi, B. Grafting polyzwitterions onto polyamide by click chemistry and nucleophilic substitution on nitrogen: A novel approach to enhance membrane fouling resistance. J. Membr. Sci. 2014, 449, 50–57. [Google Scholar] [CrossRef]
- Murou, M.; Kitano, H.; Fujita, M.; Maeda, M.; Saruwatari, Y. Self-association of zwitterionic polymer–lipid conjugates in water as examined by scattering measurements. J. Coll. Interf. Sci. 2013, 390, 47–53. [Google Scholar] [CrossRef]
- Matsuura, K.; Ohno, K.; Kagaya, S.; Kitano, H. Carboxybetaine polymer-protected gold nanoparticles: High dispersion stability and resistance against non-specific adsorption of proteins. Macromol. Chem. Phys. 2007, 208, 862–873. [Google Scholar] [CrossRef]
- Samanta, D.; McRae, S.; Cooper, B.; Hu, Y.; Emrick, T.; Pratt, J.; Charles, S.A. End-functionalized phosphorylcholine methacrylates and their use in protein conjugation. BioMacromol. 2008, 9, 2891–2897. [Google Scholar] [CrossRef]
- Thompson, K.L.; Bannister, I.; Armes, S.P.; Lewis, A.L. Preparation of biocompatible sterically stabilized latexes using well-defined poly(2-(methacryloyloxy)ethyl phosphorylcholine) macromonomers. Langmuir 2009, 26, 4693–4702. [Google Scholar]
- Chen, X.; Parelkar, S.S.; Henchey, E.; Schneider, S.; Emrick, T. PolyMPC–doxorubicin prodrugs. Bioconj. Chem. 2012, 23, 1753–1763. [Google Scholar] [CrossRef]
- Li, G.; Xue, H.; Cheng, G.; Chen, S.; Zhang, F.; Jiang, S. Ultralow fouling zwitterionic polymers grafted from surfaces covered with an initiator via an adhesive mussel mimetic linkage. J. Phys. Chem. B 2008, 112, 15269–15274. [Google Scholar] [CrossRef]
- Kuang, J.; Messersmith, P.B. Universal surface-initiated polymerization of antifouling zwitterionic brushes using a mussel-mimetic peptide initiator. Langmuir 2012, 28, 7258–7266. [Google Scholar] [CrossRef]
- McRae, S.; Chen, X.; Kratz, K.; Samanta, D.; Henchey, E.; Schneider, S.; Emrick, T. Pentafluorophenyl ester-functionalized phosphorylcholine polymers: Preparation of linear, two-arm, and grafted polymer-protein conjugates. BioMacromol. 2012, 13, 2099–2109. [Google Scholar] [CrossRef]
- Chang, Y.; Shih, Y.J.; Lai, C.J.; Kung, H.H.; Jiang, S. Blood-inert surfaces via ion-pair anchoring of zwitterionic copolymer brushes in human whole blood. Adv. Funct. Mater. 2013, 23, 1100–1110. [Google Scholar] [CrossRef]
- Chen, X.; Lawrence, J.; Parelkar, S.; Emrick, T. Novel zwitterionic copolymers with dihydrolipoic acid: Synthesis and preparation of nonfouling nanorods. Macromol. 2013, 46, 119–127. [Google Scholar] [CrossRef]
- Tu, Q.; Wang, J.C.; Liu, R.; Zhang, Y.; Xu, J.; Liu, J.; Yuan, M.S.; Liu, W.; Wang, J. Synthesis of polyethylene glycol- and sulfobetaine-conjugated zwitterionic poly(l-lactide) and assay of its antifouling properties. Coll. Surf. B 2013, 102, 331–340. [Google Scholar] [CrossRef]
- Wang, W.; Nan, W.; Sun, L.; Liu, W. A systemic gene vector constructed by zwitterionic polymer modified low molecular weight PEI. React. Funct. Polym. 2013, 73, 993–1000. [Google Scholar] [CrossRef]
- Chien, H.W.; Tsai, C.C.; Tsai, W.B.; Wang, M.J.; Kuo, W.H.; Wei, T.C.; Huang, S.T. Surface conjugation of zwitterionic polymers to inhibit cell adhesion and protein adsorption. Coll. Surf. B 2013, 107, 152–159. [Google Scholar] [CrossRef]
- Lewis, A.; Tang, Y.; Brocchini, S.; Choi, J.W.; Godwin, A. Poly(2-methacryloyloxyethyl phosphorylcholine) for protein conjugation. Bioconj. Chem. 2008, 19, 2144–2155. [Google Scholar] [CrossRef]
- Zhai, S.; Ma, Y.; Chen, Y.; Li, D.; Cao, J.; Liu, Y.; Cai, M.; Xie, X.; Chen, Y.; Luo, X. Synthesis of an amphiphilic block copolymer containing zwitterionic sulfobetaine as a novel pH-sensitive drug carrier. Polym. Chem. 2014, 5, 1285–1297. [Google Scholar] [CrossRef]
- Li, Y.; Armes, S.P.; Jin, X.; Zhu, S. Direct synthesis of well-defined quaternized homopolymers and diblock copolymers via ATRP in protic media. Macromol. 2003, 36, 8268–8275. [Google Scholar] [CrossRef]
- Du, J.; Tang, Y.; Lewis, A.L.; Armes, S.P. pH-sensitive vesicles based on a biocompatible zwitterionic diblock copolymer. J. Am. Chem. Soc. 2005, 127, 17982–17983. [Google Scholar]
- Sugihara, S.; Sugihara, K.; Armes, S.P.; Ahmad, H.; Lewis, A.L. Synthesis of biomimetic poly(2-(methacryloyloxy)ethyl phosphorylcholine) nanolatexes via atom transfer radical dispersion polymerization in alcohol/water mixtures. Macromol. 2010, 43, 6321–6329. [Google Scholar] [CrossRef]
- Osawa, K.; Imae, T.; Ujihara, M.; Harada, A.; Ochi, K.; Ishihara, K.; Yusa, S.I. Preparation of amphiphilic diblock copolymers with pendant hydrophilic phosphorylcholine and hydrophobic dendron groups and their self-association behavior in water. J. Polym. Sci. Part A: Polym. Chem. 2013, 51, 4923–4931. [Google Scholar] [CrossRef]
- Yang, J.; Lv, J.; Behl, M.; Lendlein, A.; Yang, D.; Zhang, L.; Shi, C.; Guo, J.; Feng, Y. Functionalization of polycarbonate surfaces by grafting PEG and zwitterionic polymers with a multicomb structure. Macromol. Biosci. 2013, 13, 1681–1688. [Google Scholar] [CrossRef]
- Inoue, Y.; Watanabe, J.; Takai, M.; Yusa, S.I.; Ishihara, K. Synthesis of sequence-controlled copolymers from extremely polar and apolar monomers by living radical polymerization and their phase-separated structures. J. Polym. Sci. Part A: Polym. Chem. 2005, 43, 6073–6083. [Google Scholar] [CrossRef]
- Li, C.; Buurma, N.J.; Haq, I.; Turner, C.; Armes, S.P.; Castelletto, V.; Hamley, I.W.; Lewis, A.L. Synthesis and characterization of biocompatible, thermoresponsive ABC and ABA triblock copolymer gelators. Langmuir 2005, 21, 11026–11033. [Google Scholar] [CrossRef]
- Hsiue, G.H.; Lo, C.L.; Cheng, C.H.; Lin, C.P.; Huang, C.K.; Chen, H.H. Preparation and characterization of poly(2-methacryloyloxyethyl phosphorylcholine)-block-poly(D,L-lactide) polymer nanoparticles. J. Polym. Sci., Part A: Polym. Chem. 2007, 45, 688–698. [Google Scholar] [CrossRef]
- Dai, F.; Wang, P.; Wang, Y.; Tang, L.; Yang, J.; Liu, W.; Li, H.; Wang, G. Double thermoresponsive polybetaine-based ABA triblock copolymers with capability to condense DNA. Polymer 2008, 49, 5322–5328. [Google Scholar] [CrossRef]
- Madsen, J.; Armes, S.P.; Bertal, K.; Lomas, H.; MacNeil, S.; Lewis, A.L. Biocompatible wound dressings based on chemically degradable triblock copolymer hydrogels. BioMacromol. 2008, 9, 2265–2275. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, Z.; Pan, F.; Waigh, T.A.; Lu, J.R. Plasmid DNA complexation with phosphorylcholine diblock copolymers and its effect on cell transfection. Langmuir 2008, 24, 6881–6888. [Google Scholar]
- Cristiano, C.M.Z.; Soldi, V.; Li, C.; Armes, S.P.; Rochas, C.; Pignot-Paintrand, I.; Borsali, R. Thermo-responsive copolymers based on Poly(N-isopropylacrylamide) and Poly[2-(methacryloyloxy)ethyl phosphorylcholine]: Light scattering and microscopy experiments. Macromol. Chem. Phys. 2009, 210, 1726–1733. [Google Scholar] [CrossRef]
- Du, J.; Armes, S.P. Preparation of biocompatible zwitterionic block copolymer vesicles by direct dissolution in water and subsequent silicification within their membranes. Langmuir 2009, 25, 9564–9570. [Google Scholar] [CrossRef]
- Madsen, J.; Armes, S.P.; Bertal, K.; MacNeil, S.; Lewis, A.L. Preparation and aqueous solution properties of thermoresponsive biocompatible AB diblock copolymers. BioMacromol. 2009, 10, 1875–1887. [Google Scholar]
- Yu, B.; Lowe, A.B.; Ishihara, K. RAFT synthesis and stimulus-induced self-assembly in water of copolymers based on the biocompatible monomer 2-(methacryloyloxy)ethyl phosphorylcholine. BioMacromol. 2009, 10, 950–958. [Google Scholar] [CrossRef]
- Liu, G.Y.; Lv, L.P.; Chen, C.J.; Hu, X.F.; Ji, J. Biocompatible Poly(D,L-lactide)-block-Poly(2-methacryloyloxyethylphosphorylcholine) micelles for drug delivery. Macromol. Chem. Phys. 2011, 212, 643–651. [Google Scholar] [CrossRef]
- Liu, G.Y.; Lv, L.P.; Chen, C.J.; Liu, X.S.; Hu, X.F.; Ji, J. Biocompatible and biodegradable polymersomes for pH-triggered drug release. Soft Matter 2011, 7, 6629–6636. [Google Scholar] [CrossRef]
- Madsen, J.; Warren, N.J.; Armes, S.P.; Lewis, A.L. Synthesis of rhodamine 6G-based compounds for the ATRP synthesis of fluorescently labeled biocompatible polymers. BioMacromol. 2011, 12, 2225–2234. [Google Scholar] [CrossRef]
- Sugihara, S.; Armes, S.P.; Blanazs, A.; Lewis, A.L. Non-spherical morphologies from cross-linked biomimetic diblock copolymers using RAFT aqueous dispersion polymerization. Soft Matter 2011, 7, 10787–10793. [Google Scholar] [CrossRef]
- Yoshikawa, H.Y.; Rossetti, F.F.; Kaufmann, S.; Kaindl, T.; Madsen, J.; Engel, U.; Lewis, A.L.; Armes, S.P.; Tanaka, M. Quantitative evaluation of mechanosensing of cells on dynamically tunable hydrogels. J. Am. Chem. Soc. 2011, 133, 1367–1374. [Google Scholar] [CrossRef]
- Du, J.; Fan, L.; Liu, Q. pH-sensitive block copolymer vesicles with variable trigger points for drug delivery. Macromol. 2012, 45, 8275–8283. [Google Scholar] [CrossRef]
- Shih, Y.J.; Chang, Y.; Deratani, A.; Quemener, D. “Schizophrenic” hemocompatible copolymers via switchable thermoresponsive transition of nonionic/zwitterionic block self-assembly in human blood. BioMacromol. 2012, 13, 2849–2858. [Google Scholar] [CrossRef]
- Nakai, K.; Nishiuchi, M.; Inoue, M.; Ishihara, K.; Sanada, Y.; Sakurai, K.; Yusa, S.I. Preparation and characterization of polyion complex micelles with phosphobetaine shells. Langmuir 2013, 29, 9651–9661. [Google Scholar] [CrossRef]
- Wang, H.; Xu, F.; Li, D.; Liu, X.; Jin, Q.; Ji, J. Bioinspired phospholipid polymer prodrug as a pH-responsive drug delivery system for cancer therapy. Polym. Chem. 2013, 4, 2001–2010. [Google Scholar]
- Wang, X.; Jiang, G.; Wang, Y.; Wang, R.; Sun, X.; Hu, R.; Xi, X.; Zhou, Y.; Wang, S.; Wang, T. Preparation of biomimetic zwitterionic core cross-linked nanocarriers for control release of bioactive agents. J. Macromol. Sci. A 2013, 50, 644–652. [Google Scholar] [CrossRef]
- Cummings, C.; Murata, H.; Koepsel, R.; Russell, A.J. Dramatically increased pH and temperature stability of chymotrypsin using dual block polymer-based protein engineering. BioMacromol. 2014, 15, 763–771. [Google Scholar] [CrossRef]
- Fukuda, K.; Enomoto, R.; Ishihara, K.; Morishima, Y.; Yusa, S.I. Thermo-responsive and biocompatible diblock copolymers prepared via reversible addition-fragmentation chain transfer (RAFT) radical polymerization. Polymers 2014, 6, 846–859. [Google Scholar] [CrossRef]
- Kuang, W.; Zhao, X. Synthesis, characterization, and properties of novel hydrophobically associating fluorinated copolymers for DNA delivery. React. Funct. Polym. 2013, 73, 703–709. [Google Scholar] [CrossRef]
- Edlund, U.; Rodriguez-Emmenegger, C.; Brynda, E.; Albersson, A.C. Self-assembling zwitterionic carboxybetaine copolymers via aqueous SET-LRP from hemicellulose multi-site initiators. Polym. Chem. 2012, 3, 2920–2927. [Google Scholar] [CrossRef]
- Deng, Z.; Bouchékif, H.; Babooram, K.; Housni, A.; Choytun, N.; Narain, R. Facile synthesis of controlled-structure primary amine-based methacrylamide polymers via the reversible addition-fragmentation chain transfer process. J. Polym. Sci., Part A: Polym. Chem. 2008, 46, 4984–4996. [Google Scholar] [CrossRef]
- Sugihara, S.; Armes, S.P.; Lewis, A.L. One-pot synthesis of biomimetic shell cross-linked micelles and nanocages by ATRP in alcohol/water mixtures. Angew. Chem. Int. Ed. 2010, 49, 3500–3503. [Google Scholar] [CrossRef]
- Yusa, S.I.; Fukuda, K.; Yamamoto, T.; Ishihara, K.; Morishima, Y. Synthesis of well-defined amphiphilic block copolymers having phospholipid polymer sequences as a novel biocompatible polymer micelle reagent. BioMacromol. 2005, 6, 663–670. [Google Scholar] [CrossRef]
- Gawel, K.; Szczubialka, K.; Zapotoczny, S.; Nowakowska, M. Zwitterionically modified hydroxypropylcellulose for biomedical applications. Eur. Polym. J. 2010, 46, 1475–1479. [Google Scholar] [CrossRef]
- Li, J.H.; Li, M.Z.; Miao, J.; Wang, J.B.; Shao, X.S.; Zhang, Q.Q. Improved surface property of PVDF membrane with amphiphilic zwitterionic copolymer as membrane additive. Appl. Surf. Sci. 2012, 258, 6398–6405. [Google Scholar] [CrossRef]
- Jin, S.; Zhou, N.; Xu, D.; Shen, J. Synthesis and characterization of poly(2-methacryloyloxyethyl phosphorylcholine) onto graphene oxide. Polym. Adv. Tech. 2013, 24, 685–691. [Google Scholar] [CrossRef]
- Tian, M.; Wang, J.; Zhang, E.; Li, J.; Duan, C.; Yao, F. Synthesis of agarose-graft-poly[3-dimethyl (methacryloyloxyethyl) ammonium propanesulfonate] Zwitterionic graft copolymers via ATRP and their thermally-induced aggregation behavior in aqueous media. Langmuir 2013, 29, 8076–8085. [Google Scholar] [CrossRef]
- Iwasaki, Y.; Akiyoshi, K. Synthesis and characterization of amphiphilic polyphosphates with hydrophilic graft chains and cholesteryl groups as nanocarriers. BioMacromol. 2006, 7, 1433–1438. [Google Scholar] [CrossRef]
- Zeng, R.; Xu, S.; Cheng, J.; Cai, Z.; Pi, P.; Wen, X. Thermoresponsive/low-fouling zwitterionic hydrogel for controlled drug release. J. Appl. Polym. Sci. 2014, 131, 39816:1–39816:7. [Google Scholar]
- Wang, X.; Chen, X.; Xing, L.; Mao, C.; Yu, H.; Shen, J. Blood compatibility of a new zwitterionic bare metal stent with hyperbranched polymer brushes. J. Mater. Chem. B 2013, 1, 5036–5044. [Google Scholar] [CrossRef]
- Bersani, S.; Salmaso, S.; Mastrotto, F.; Ravazzolo, E.; Semenzato, A.; Caliceti, P. Star-like oligo-arginyl-maltotriosyl derivatives as novel cell-penetrating enhancers for the intracellular delivery of colloidal therapeutic systems. Bioconj. Chem. 2012, 23, 1415–1425. [Google Scholar] [CrossRef]
- Wang, X.; Sun, X.; Jiang, G.; Wang, R.; Hu, R.; Xi, X.; Zhou, Y.; Wang, S.; Wang, T. Synthesis of biomimetic hyperbranched zwitterionic polymers as targeting drug delivery carriers. J. Appl. Polym. Sci. 2013, 128, 3289–3294. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, S.; Chang, Y.; Jiang, S. Surface grafted sulfobetaine polymers via atom transfer radical polymerization as superlow fouling coatings. J. Phys. Chem. B 2006, 110, 10799–10804. [Google Scholar] [CrossRef]
- Zhang, Z.; Chao, T.; Chen, S.; Jiang, S. Superlow fouling sulfobetaine and carboxybetaine polymers on glass slides. Langmuir 2006, 22, 10072–10077. [Google Scholar] [CrossRef]
- Narain, R.; Housni, A.; Lane, L. Modification of carboxyl-functionalized single-walled carbon nanotubes with biocompatible, water-soluble phosphorylcholine and sugar-based polymers: Bioinspired nanorods. J. Polym. Sci. Part A: Polym. Chem. 2006, 44, 6558–6568. [Google Scholar] [CrossRef]
- Azzaroni, O.; Brown, A.A.; Huck, W.T.S. UCST wetting transitions of polyzwitterionic brushes driven by self-association. Angew. Chem. Int. Ed. 2006, 45, 1770–1774. [Google Scholar] [CrossRef]
- Matsuda, Y.; Kobayashi, M.; Annaka, M.; Ishihara, K.; Takahara, A. Dimensions of a free linear polymer and polymer immobilized on silica nanoparticles of a zwitterionic polymer in aqueous solutions with various ionic strengths. Langmuir 2008, 24, 8772–8778. [Google Scholar] [CrossRef]
- Cheng, G.; Li, G.; Xue, H.; Chen, S.; Bryers, J.D.; Jiang, S. Zwitterionic carboxybetaine polymer surfaces and their resistance to long-term biofilm formation. Biomater. 2009, 30, 5234–5240. [Google Scholar] [CrossRef]
- Jia, G.; Cao, Z.; Xue, H.; Xu, Y.; Jiang, S. Novel zwitterionic-polymer-coated silica nanoparticles. Langmuir 2009, 25, 3196–3199. [Google Scholar] [CrossRef]
- Gao, C.; Li, G.; Xue, H.; Yang, W.; Zhang, F.; Jiang, S. Functionalizable and ultra-low fouling zwitterionic surfaces via adhesive mussel mimetic linkages. Biomater. 2010, 31, 1486–1492. [Google Scholar] [CrossRef]
- Inoue, Y.; Ishihara, K. Reduction of protein adsorption on well-characterized polymer brush layers with varying chemical structures. Coll. Surf. B 2010, 81, 350–357. [Google Scholar] [CrossRef]
- Zhao, Y.H.; Wee, K.H.; Bai, R. Highly hydrophilic and low-protein-fouling polypropylene membrane prepared by surface modification with sulfobetaine-based zwitterionic polymer through a combined surface polymerization method. J. Membr. Sci. 2010, 362, 326–333. [Google Scholar] [CrossRef]
- Kitano, H.; Suzuki, H.; Kondo, T.; Sasaki, K.; Iwanaga, S.; Nakamura, M.; Ohno, K.; Saruwatari, Y. Image printing on the surface of anti-biofouling zwitterionic polymer brushes by ion beam irradiation. Macromol. Biosci. 2011, 11, 557–564. [Google Scholar] [CrossRef]
- Li, L.; Marchant, R.E.; Dubnisheva, A.; Roy, S.; Fissell, W.H. Anti-biofouling sulfobetaine polymer thin films on silicon and silicon nanopore membranes. J. Biomater. Sci. Polym. Ed. 2011, 22, 91–106. [Google Scholar]
- Nguyen, A.T.; Baggerman, J.; Paulusse, J.M.J.; van Rijn, C.J.M.; Zuilhof, H. Stable protein-repellent zwitterionic polymer brushes grafted from silicon nitride. Langmuir 2011, 27, 2587–2594. [Google Scholar]
- Suzuki, H.; Murou, M.; Kitano, H.; Ohno, K.; Saruwatari, Y. Silica particles coated with zwitterionic polymer brush: Formation of colloidal crystals and anti-biofouling properties in aqueous medium. Coll. Surf. B 2011, 84, 111–116. [Google Scholar] [CrossRef]
- Hu, H.; Wang, X.B.; Xu, S.L.; Yang, W.T.; Xu, F.J.; Shen, J.; Mao, C. Preparation and evaluation of well-defined hemocompatible layered double hydroxide-poly(sulfobetaine) nanohybrids. J. Mater. Chem. 2012, 22, 15362–15369. [Google Scholar]
- Kondo, T.; Nomura, K.; Murou, M.; Gemmei-Ide, M.; Kitano, H.; Noguchi, H.; Uosaki, K.; Ohno, K.; Saruwatari, Y. Structure of water in the vicinity of a zwitterionic polymer brush as examined by sum frequency generation method. Coll. Surf. B 2012, 100, 126–132. [Google Scholar] [CrossRef]
- Li, Q.; Bi, Q.Y.; Zhou, B.; Wang, X.L. Zwitterionic sulfobetaine-grafted poly(vinylidene fluoride) membrane surface with stably anti-protein-fouling performance via a two-step surface polymerization. Appl. Surf. Sci. 2012, 258, 4707–4717. [Google Scholar] [CrossRef]
- Li, Q.; Zhou, B.; Bi, Q.Y.; Wang, X.L. Surface modification of PVDF membranes with sulfobetaine polymers for a stably anti-protein-fouling performance. J. Appl. Polym. Sci. 2012, 125, 4015–4027. [Google Scholar] [CrossRef]
- Li, Y.; Giesbers, M.; Gerth, M.; Zuilhof, H. Generic Top-functionalization of patterned antifouling zwitterionic polymers on indium tin oxide. Langmuir 2012, 28, 12509–12517. [Google Scholar] [CrossRef]
- Lu, C.Y.; Zhou, N.L.; Xiao, Y.H.; Tang, Y.D.; Jin, S.X.; Wu, Y.; Zhang, J.; Shen, J. Surface grafting density analysis of high anti-clotting PU-Si-g-P(MPC) films. Appl. Surf. Sci. 2012, 258, 3920–3926. [Google Scholar] [CrossRef]
- Nguyen, A.T.; Baggerman, J.; Paulusse, J.M.J.; Zuilhof, H.; van Rijn, C.J.M. Bioconjugation of protein-repellent zwitterionic polymer brushes grafted from silicon nitride. Langmuir 2012, 28, 604–610. [Google Scholar] [CrossRef]
- Santonicola, M.G.; Memesa, M.; Meszynska, A.; Ma, Y.; Vancso, G.J. Surface-grafted zwitterionic polymers as platforms for functional supported phospholipid membranes. Soft Matter 2012, 8, 1556–1562. [Google Scholar] [CrossRef]
- Yang, W.J.; Neoh, K.G.; Kang, E.T.; Lee, S.S.C.; Teo, S.L.M.; Rittschof, D. Functional polymer brushes via surface-initiated atom transfer radical graft polymerization for combating marine biofouling. Biofouling 2012, 28, 895–912. [Google Scholar] [CrossRef]
- Liu, P.; Chen, Q.; Yuan, B.; Chen, M.; Wu, S.; Lin, S.; Shen, J. Facile surface modification of silicone rubber with zwitterionic polymers for improving blood compatibility. Mater. Sci. Eng. C 2013, 33, 3865–3874. [Google Scholar] [CrossRef]
- Yue, W.W.; Li, H.J.; Xiang, T.; Qin, H.; Sun, S.D.; Zhao, C.S. Grafting of zwitterion from polysulfone membrane via surface-initiated ATRP with enhanced antifouling property and biocompatibility. J. Membr. Sci. 2013, 446, 79–91. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, S.; Jiang, S. Dual-functional biomimetic materials: Nonfouling poly(carboxybetaine) with active functional groups for protein immobilization. BioMacromol. 2006, 7, 3311–3315. [Google Scholar] [CrossRef]
- Cho, W.K.; Kong, B.; Choi, I.S. Highly efficient non-biofouling coating of zwitterionic polymers: Poly((3-(methacryloylamino)propyl)-dimethyl(3-sulfopropyl)ammonium hydroxide). Langmuir 2007, 23, 5678–5682. [Google Scholar]
- Chang, Y.; Liao, S.C.; Higuchi, A.; Ruaan, R.C.; Chu, C.W.; Chen, W.Y. A highly stable nonbiofouling surface with well-packed grafted zwitterionic polysulfobetaine for plasma protein repulsion. Langmuir 2008, 24, 5453–5458. [Google Scholar]
- Cheng, N.; Brown, A.A.; Azzaroni, O.; Huck, W.T.S. Thickness-dependent properties of polyzwitterionic brushes. Macromol. 2008, 41, 6317–6321. [Google Scholar] [CrossRef]
- Ladd, J.; Zhang, Z.; Chen, S.; Hower, J.C.; Jiang, S. Zwitterionic polymers exhibiting high resistance to nonspecific protein adsorption from human serum and plasma. BioMacromol. 2008, 9, 1357–1361. [Google Scholar] [CrossRef]
- Yang, W.; Chen, S.; Cheng, G.; Vaisocherová, H.; Xue, H.; Li, W.; Zhang, J.; Jiang, S. Film thickness dependence of protein adsorption from blood serum and plasma onto poly(sulfobetaine)-grafted surfaces. Langmuir 2008, 24, 9211–9214. [Google Scholar]
- Chiang, Y.C.; Chang, Y.; Higuchi, A.; Chen, W.Y.; Ruaan, R.C. Sulfobetaine-grafted poly(vinylidene fluoride) ultrafiltration membranes exhibit excellent antifouling property. J. Membr. Sci. 2009, 339, 151–159. [Google Scholar] [CrossRef]
- Yang, W.; Xue, H.; Li, W.; Zhang, J.; Jiang, S. Pursuing “zero” protein adsorption of poly(carboxybetaine) from undiluted blood serum and plasma. Langmuir 2009, 25, 11911–11916. [Google Scholar] [CrossRef]
- Zhang, Z.; Finlay, J.A.; Wang, L.; Gao, Y.; Callow, J.A.; Callow, M.E.; Jiang, S. Polysulfobetaine-grafted surfaces as environmentally benign ultralow fouling marine coatings. Langmuir 2009, 25, 13516–13521. [Google Scholar] [CrossRef]
- Kitano, H.; Suzuki, H.; Matsuura, K.; Ohno, K. Molecular recognition at the exterior surface of a zwitterionic telomer brush. Langmuir 2010, 26, 6767–6774. [Google Scholar] [CrossRef]
- Zhao, C.; Li, L.; Zheng, J. Achieving highly effective nonfouling performance for surface-grafted poly(HPMA) via atom-transfer radical polymerization. Langmuir 2010, 26, 17375–17382. [Google Scholar] [CrossRef]
- Mi, L.; Giarmarco, M.M.; Shao, Q.; Jiang, S. Divalent cation-mediated polysaccharide interactions with zwitterionic surfaces. Biomater. 2012, 33, 2001–2006. [Google Scholar] [CrossRef]
- Hu, R.; Li, G.; Jiang, Y.; Zhang, Y.; Zou, J.J.; Wang, L.; Zhang, X. Silver-zwitterion organic-inorganic nanocomposite with antimicrobial and antiadhesive capabilities. Langmuir 2013, 29, 3773–3779. [Google Scholar] [CrossRef]
- Kobayashi, M.; Takahara, A. Environmentally friendly repeatable adhesion using a sulfobetaine-type polyzwitterion brush. Polym. Chem. 2013, 4, 4987–4992. [Google Scholar] [CrossRef]
- Kim, Y.; An, S.; Kim, S.; Park, J.H.; Son, H.A.; Kim, H.T.; Suh, K.D.; Kim, J.W. Synthesis and characterization of TiO2 nanoparticles chemically coated with zwitterionic polymer brushes. Polymer 2013, 54, 5609–5614. [Google Scholar]
- Liu, P.; Chen, Q.; Yuan, B.; Chen, M.; Wu, S.; Lin, S.; Shen, J. Facile surface modification of silicone rubber with zwitterionic polymers for improving blood compatibility. Mater. Sci. Eng. C 2013, 33, 3865–3874. [Google Scholar] [CrossRef]
- Chen, L.; Thérien-Aubin, H.; Wong, M.C.Y.; Hoek, E.M.V.; Ober, C.K. Improved antifouling properties of polymer membranes using a ‘layer-by-layer’ mediated method. J. Mater. Chem. B 2013, 1, 5651–5658. [Google Scholar]
- Quintana, R.; Gosa, M.; Janczewski, D.; Kutnyanszky, E.; Vancso, G.J. Enhanced stability of low fouling zwitterionic polymer brushes in seawater with diblock architecture. Langmuir 2013, 29, 10859–10867. [Google Scholar] [CrossRef]
- Song, L.; Zhao, J.; Luan, S.; Ma, J.; Liu, J.; Xu, X.; Yin, J. Fabrication of a detection platform with boronic-acid-containing zwitterionic polymer brush. ACS Appl. Mater. Interf. 2013, 5, 13207–13215. [Google Scholar] [CrossRef]
- Wei, Q.; Cai, M.; Zhou, F.; Liu, W. Dramatically tuning friction using responsive polyelectrolyte brushes. Macromol. 2013, 46, 9368–9379. [Google Scholar] [CrossRef]
- Zhang, Z.; Morse, A.J.; Armes, S.P.; Lewis, A.L.; Geoghegan, M.; Leggett, G.J. Nanoscale contact mechanics of biocompatible polyzwitterionic brushes. Langmuir 2013, 29, 10684–10692. [Google Scholar] [CrossRef]
- Dong, Z.; Mao, J.; Wang, D.; Yang, M.; Wang, W.; Bo, S.; Ji, X. Tunable dual-thermoresponsive phase behavior of zwitterionic polysulfobetaine copolymers containing poly(N,N-dimethylaminoethyl methacrylate)-grafted silica nanoparticles in aqueous solution. Macromol. Chem. Phys. 2014, 215, 111–120. [Google Scholar] [CrossRef]
- Sin, M.C.; Sun, Y.M.; Chang, Y. Zwitterionic-based stainless steel with well-defined polysulfobetaine brushes for general bioadhesive control. ACS Appl. Mater. Interf. 2014, 6, 861–873. [Google Scholar] [CrossRef]
- Dong, Z.; Mao, J.; Yang, M.; Wang, D.; Bo, S.; Ji, X. Phase Behavior of poly(sulfobetaine methacrylate)-grafted silica nanoparticles and their stability in protein solutions. Langmuir 2011, 27, 15282–15291. [Google Scholar] [CrossRef]
- Rodriguez Emmenegger, C.; Brynda, E.; Riedel, T.; Sedlakova, Z.; Houska, M.; Alles, A.B. Interaction of blood plasma with antifouling surfaces. Langmuir 2009, 25, 6328–6333. [Google Scholar]
- Brault, N.D.; White, A.D.; Taylor, A.D.; Yu, Q.; Jiang, S. Directly functionalizable surface platform for protein arrays in undiluted human blood plasma. Anal. Chem. 2013, 85, 1447–1453. [Google Scholar] [CrossRef]
- Polzer, F.; Heigl, J.; Schneider, C.; Ballauff, M.; Borisov, O.V. Synthesis and analysis of zwitterionic spherical polyelectrolyte brushes in aqueous solution. Macromol. 2011, 44, 1654–1660. [Google Scholar]
- Yang, Q.; Ulbricht, M. Novel membrane adsorbers with grafted zwitterionic polymers synthesized by surface-initiated ATRP and their salt-modulated permeability and protein binding properties. Chem. Mater. 2012, 24, 2943–2951. [Google Scholar] [CrossRef]
- Kobayashi, M.; Terayama, Y.; Kikuchi, M.; Takahara, A. Chain dimensions and surface characterization of superhydrophilic polymer brushes with zwitterion side groups. Soft Matter 2013, 9, 5138–5148. [Google Scholar] [CrossRef]
- Krause, J.E.; Brault, N.D.; Li, Y.; Xue, H.; Zhou, Y.; Jiang, S. Photoiniferter-mediated polymerization of zwitterionic carboxybetaine monomers for low-fouling and functionalizable surface coatings. Macromol. 2011, 44, 9213–9220. [Google Scholar] [CrossRef]
- Huang, C.J.; Li, Y.; Krause, J.B.; Brault, N.D.; Jiang, S. Internal architecture of zwitterionic polymer brushes regulates nonfouling properties. Macromol. Rapid Commun. 2012, 33, 1003–1007. [Google Scholar] [CrossRef]
- Liu, Q.; Singh, A.; Liu, L. Amino acid-based zwitterionic poly(serine methacrylate) as an antifouling material. BioMacromol. 2013, 14, 226–231. [Google Scholar] [CrossRef]
- You, Y.Z.; Hong, C.Y.; Pan, C.Y. Directly growing ionic polymers on multi-welled carbon nanotubes via surface RAFT polymerization. Nanotechn. 2006, 17, 2350–2354. [Google Scholar]
- Sun, J.T.; Yu, Z.Q.; Hong, C.Y.; Pan, C.Y. Biocompatible zwitterionic sulfobetaine copolymer-coated mesoporous silica nanoparticles for temperature-responsive drug release. Macromol. Rapid Commun. 2012, 33, 811–818. [Google Scholar] [CrossRef]
- Yameen, B.; Rodriguez-Emmenegger, C.; Ahmed, I.; Preuss, C.M.; Dürr, C.J.; Zydziak, N.; Trouillet, V.; Fruk, L.; Barner-Kowollik, C. A facile one-pot route to poly(carboxybetaine acrylamide) functionalized SWCNTs. Chem. Commun. 2013, 49, 6734–6736. [Google Scholar] [CrossRef]
- Yuan, J.; Huang, X.; Li, P.; Li, L.; Shen, J. Surface-initiated RAFT polymerization of sulfobetaine from cellulose membranes to improve hemocompatibility and antibiofouling property. Polym. Chem. 2013, 41, 5074–5085. [Google Scholar]
- Huang, G.; Xiong, Z.; Qin, H.; Zhu, J.; Sun, Z.; Zhang, Y.; Peng, X.; Ou, J.; Zou, H. Synthesis of zwitterionic polymer brushes hybrid silica nanoparticles via controlled polymerization for highly efficient enrichment of glycopeptides. Anal. Chim. Acta 2014, 809, 61–68. [Google Scholar]
- Abraham, S.; So, A.; Unsworth, L.D. Poly(carboxybetaine methacrylamide)-modified nanoparticles: A model system for studying the effect of chain chemistry on film properties, adsorbed protein conformation, and clot formation kinetics. BioMacromol. 2011, 12, 3567–3580. [Google Scholar] [CrossRef]
- Kitano, H.; Kawasaki, A.; Kawasaki, H.; Morokoshi, S. Resistance of zwitterionic telomers accumulated on metal surfaces against nonspecific adsorption of proteins. J. Coll. Interf. Sci. 2005, 282, 340–348. [Google Scholar] [CrossRef]
- Zhang, L.; Xue, H.; Gao, C.; Carr, L.; Wang, J.; Chu, B.; Jiang, S. Imaging and cell targeting characteristics of magnetic nanoparticles modified by a functionalizable zwitterionic polymer with adhesive 3,4-dihydroxyphenyl-L-alanine linkages. Biomater. 2010, 31, 6582–6588. [Google Scholar] [CrossRef]
- Rahaman, M.S.; Thérien-Aubin, H.; Ben-Sasson, M.; Ober, C.K.; Nielsen, M.; Elimelech, M. Control of biofouling on reverse osmosis polyamide membranes modified with biocidal nanoparticles and antifouling polymer brushes. J. Mater. Chem. B 2014, 2, 1724–1732. [Google Scholar] [CrossRef]
- Li, M.; Neoh, K.G.; Xu, L.Q.; Wang, R.; Kang, E.T.; Lau, T.; Olszyna, D.P.; Chiong, E. Surface modification of silicone for biomedical applications requiring long-term antibacterial, antifouling, and hemocompatible properties. Langmuir 2012, 28, 16408–16422. [Google Scholar] [CrossRef]
- McRae Page, S.; Parelkar, S.; Gerasimenko, A.; Shin, D.Y.; Peyton, S.R.; Emrick, T. Promoting cell adhesion on slippery phosphorylcholine hydrogel surfaces. J. Mater. Chem. B 2014, 2, 620–624. [Google Scholar]
- Zhang, J.; Yuan, J.; Yuan, Y.; Zang, X.; Shen, J.; Lin, S. Platelet adhesive resistance of segmented polyurethane film surface-grafted with vinyl benzyl sulfo monomer of ammonium zwitterions. Biomater. 2003, 24, 4223–4231. [Google Scholar] [CrossRef]
- Zhang, J.; Yuan, J.; Yuan, Y.; Shen, J.; Lin, S. Chemical modification of cellulose membranes with sulfo ammonium zwitterionic vinyl monomer to improve hemocompatibility. Coll. Surf. B 2003, 30, 249–257. [Google Scholar] [CrossRef]
- Ishihara, K.; Iwasaki, Y.; Ebihara, S.; Shindo, Y.; Nakabayashi, N. Photoinduced graft polymerization of 2-methacryloyloxyethyl phosphorylcholine on polyethylene membrane surface for obtaining blood cell adhesion resistance. Coll. Surf. B 2000, 18, 325–335. [Google Scholar] [CrossRef]
- Arasawa, H.; Odawara, C.; Yokoyama, R.; Saitoh, H.; Yamauchi, T.; Tsubokawa, N. Grafting of zwitterion-type polymers onto silica gel surface and their properties. React. Funct. Polym. 2004, 61, 153–161. [Google Scholar] [CrossRef]
- Moro, T.; Takatori, Y.; Ishihara, K.; Konno, T.; Takigawa, Y.; Matsushita, T.; Chung, U.I.; Nakamura, K.; Kawaguchi, H. Surface grafting of artificial joints with a biocompatible polymer for preventing periprosthetic osteolysis. Nat. Mater. 2004, 3, 829–836. [Google Scholar] [CrossRef]
- Shan, B.; Yan, H.; Shen, J.; Lin, S. Ozone-induced grafting of a sulfoammonium zwitterionic polymer onto low-density polyethylene film for improving hemocompatibility. J. Appl. Polym. Sci. 2006, 101, 3697–3703. [Google Scholar] [CrossRef]
- Yang, C.; Sun, K.; Liu, J.; Wang, H.; Cao, Y. Zwitterionic sulfobetaine-modified non-woven fabric for blood filtration. Polym. Int. 2010, 59, 1296–1302. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, S.; Wang, G. The preparation of antifouling ultrafiltration membrane by surface grafting zwitterionic polymer onto poly(arylene ether sulfone) containing hydroxyl groups membrane. Desalination 2013, 316, 127–136. [Google Scholar] [CrossRef]
- Sun, J.; Zeng, F.; Jian, H.; Wu, S. Grafting zwitterionic polymer chains onto PEI as a convenient strategy to enhance gene delivery performance. Polym. Chem. 2013, 4, 5810–5818. [Google Scholar] [CrossRef]
- Zhou, J.; Yuan, J.; Zang, X.; Shen, J.; Lin, S. Platelet adhesion and protein adsorption on silicone rubber surface by ozone-induced grafted polymerization with carboxybetaine monomer. Coll. Surf. B 2005, 41, 55–62. [Google Scholar] [CrossRef]
- Jiang, W.; Fischer, G.; Girmay, Y.; Irgum, K. Zwitterionic stationary phase with covalently bonded phosphorylcholine type polymer grafts and its applicability to separation of peptides in the hydrophilic interaction liquid chromatography mode. J. Chromatogr. A 2006, 1127, 82–91. [Google Scholar] [CrossRef]
- Shan, B.; Yan, H.; Shen, J.; Lin, S. Ozone-induced grafting of a sulfoammonium zwitterionic polymer onto low-density polyethylene film for improving hemocompatibility. J. Appl. Polym. Sci. 2006, 101, 3697–3703. [Google Scholar] [CrossRef]
- Yu, H.; Cao, Y.; Kang, G.; Liu, J.; Li, M.; Yuan, Q. Enhancing antifouling property of polysulfone ultrafiltration membrane by grafting zwitterionic copolymer via UV-initiated polymerization. J. Membr. Sci. 2009, 342, 6–13. [Google Scholar] [CrossRef]
- Chang, Y.; Higuchi, A.; Shih, Y.J.; Li, P.T.; Chen, W.Y.; Tsai, E.M.; Hsiue, G.H. Bioadhesive control of plasma proteins and blood cells from umbilical cord blood onto the interface grafted with zwitterionic polymer brushes. Langmuir 2012, 28, 4309–4317. [Google Scholar] [CrossRef]
- Chen, S.H.; Chang, Y.; Lee, K.R.; Wei, T.C.; Higuchi, A.; Ho, F.M.; Tsou, C.C.; Ho, H.T.; Lai, J.Y. Hemocompatible control of sulfobetaine-grafted polypropylene fibrous membranes in human whole blood via plasma-induced surface zwitterionization. Langmuir 2012, 28, 17733–17742. [Google Scholar]
- Li, M.Z.; Li, J.H.; Shao, X.S.; Miao, J.; Wang, J.B.; Zhang, Q.Q.; Xu, X.P. Grafting zwitterionic brush on the surface of PVDF membrane using physisorbed free radical grafting technique. J. Membr. Sci. 2012, 405, 141–148. [Google Scholar]
- Razi, F.; Sawada, I.; Ohmukai, Y.; Maruyama, T.; Matsuyama, H. The improvement of antibiofouling efficiency of polyethersulfone membrane by functionalization with zwitterionic monomers. J. Membr. Sci. 2012, 401/402, 292–299. [Google Scholar] [CrossRef]
- Shao, Z.; Yang, Y.; Lee, H.; Kim, J.W.; Osuji, C.O. Synthesis and suspension rheology of titania nanoparticles grafted with zwitterionic polymer brushes. J. Coll. Interf. Sci. 2012, 386, 135–140. [Google Scholar] [CrossRef]
- Xu, Y.; Takai, M.; Ishihara, K. Suppression of protein adsorption on a charged phospholipid polymer interface. BioMacromol. 2008, 10, 267–274. [Google Scholar]
- Konno, T.; Watanabe, J.; Ishihara, K. Conjugation of enzymes on polymer nanoparticles covered with phosphorylcholine groups. BioMacromol. 2004, 5, 342–347. [Google Scholar] [CrossRef]
- Higuchi, A.; Hashiba, H.; Hayashi, R.; Yoon Boo, O.; Hattori, M.; Hara, M. Chemically modified polysulfone hollow fibers with zwitterionic sulfoalkylbetaine group having improved blood compatibility. ACS Symp. Ser. 2004, 876, 366–382. [Google Scholar] [CrossRef]
- Heinz, B.S.; Laschewsky, A.; Rekaï, E.D.; Wischerhoff, E.; Zacher, T. Grafting of functionalized water soluble polymers on gold surfaces: Stable stimuli-responsive thin hydrogel films exhibiting a LCST or a UCST. ACS Symp. Ser. 2001, 780, 162–180. [Google Scholar]
- Li, G.; Cheng, G.; Xue, H.; Chen, S.; Zhang, F.; Jiang, S. Ultra low fouling zwitterionic polymers with a biomimetic adhesive group. Biomater. 2008, 29, 4592–4597. [Google Scholar] [CrossRef]
- Brault, N.D.; Gao, C.; Xue, H.; Piliarik, M.; Homola, J.; Jiang, S.; Yu, Q. Ultra-low fouling and functionalizable zwitterionic coatings grafted onto SiO2 via a biomimetic adhesive group for sensing and detection in complex media. Biosens. Bioelectron. 2010, 25, 2276–2282. [Google Scholar] [CrossRef]
- Sakuragi, M.; Tsuzuki, S.; Obuse, S.; Wada, A.; Matoba, K.; Kubo, I.; Ito, Y. A photoimmobilizable sulfobetaine-based polymer for a nonbiofouling surface. Mater. Sci. Eng. C 2010, 30, 316–322. [Google Scholar] [CrossRef]
- Yang, Y.F.; Li, Y.; Li, Q.L.; Wan, L.S.; Xu, Z.K. Surface hydrophilization of microporous polypropylene membrane by grafting zwitterionic polymer for anti-biofouling. J. Membr. Sci. 2010, 362, 255–264. [Google Scholar] [CrossRef]
- Chakrabarty, T.; Kumar, M.; Shahi, V.K. pH responsive hybrid zwitterionomer for protein separation: Smart nanostructured adsorbent. Ind. Eng. Chem. Res. 2012, 51, 3015–3022. [Google Scholar] [CrossRef]
- Suzuki, H.; Li, L.; Nakaji-Hirabayashi, T.; Kitano, H.; Ohno, K.; Matsuoka, K.; Saruwatari, Y. Carboxymethylbetaine copolymer layer covalently fixed to a glass substrate. Coll. Surf. B 2012, 94, 107–113. [Google Scholar] [CrossRef]
- Gong, Y.K.; Liu, L.P.; Messersmith, P.B. Doubly biomimetic catecholic phosphorylcholine copolymer: a platform strategy for fabricating antifouling surfaces. Macromol. Biosci. 2012, 12, 979–985. [Google Scholar] [CrossRef]
- Jiang, Y.; Rongbing, B.; Ling, T.; Jian, S.; Sicong, L. Blood compatibility of polyurethane surface grafted copolymerization with sulfobetaine monomer. Coll. Surf. B 2004, 36, 27–33. [Google Scholar] [CrossRef]
- Choi, D.C.; Kim, S.H.; Lee, J.H.; Cho, H.N.; Choi, S.K. A new class of conjugated ionic polyacetylenes. 3. Cyclopolymerization of alkyldipropargyl(4-sulfobutyl)ammonium betaines by transition metal catalysts. Macromol. 1997, 30, 176–181. [Google Scholar] [CrossRef]
- Colak, S.; Tew, G.N. Synthesis and solution properties of norbornene based polybetaines. Macromol. 2008, 41, 8436–8440. [Google Scholar] [CrossRef]
- Rankin, D.A.; Lowe, A.B. New well-defined polymeric betaines: First report detailing the synthesis and ROMP of salt-responsive sulfopropylbetaine- and carboxyethylbetaine-exo-7-oxanorbornene monomers. Macromol. 2008, 41, 614–622. [Google Scholar] [CrossRef]
- Colak, S.; Nelson, C.F.; Nüsslein, K.; Tew, G.N. Hydrophilic modifications of an amphiphilic polynorbornene and the effects on its hemolytic and Antibacterial Activity. BioMacromol. 2009, 10, 353–359. [Google Scholar] [CrossRef]
- Kratz, K.; Breitenkamp, K.; Hule, R.; Pochan, D.; Emrick, T. PC-Polyolefins: Synthesis and assembly behavior in water. Macromol. 2009, 42, 3227–3229. [Google Scholar] [CrossRef]
- Kratz, K.; Xie, W.; Lee, A.; Freeman, B.D.; Emrick, T. Phosphorylcholine-substituted ROMP polyolefin coatings provide fouling resistance to membrane materials. Macromol. Mater. Eng. 2011, 296, 1142–1148. [Google Scholar] [CrossRef]
- Maddikeri, R.R.; Colak, S.; Gido, S.P.; Tew, G.N. Zwitterionic polymersomes in an ionic liquid: room temperature TEM characterization. BioMacromol. 2011, 12, 3412–3417. [Google Scholar] [CrossRef]
- Colak, S.; Tew, G.N. Amphiphilic polybetaines: The effect of side-chain hydrophobicity on protein adsorption. BioMacromol. 2012, 13, 1233–1239. [Google Scholar] [CrossRef]
- Leng, C.; Gibney, K.A.; Liu, Y.; Tew, G.N.; Chen, Z. In situ probing the surface restructuring of antibiofouling amphiphilic polybetaines in water. ACS Macro. Lett. 2013, 2, 1011–1015. [Google Scholar] [CrossRef]
- Colak, S.; Tew, G.N. Dual-functional ROMP-based betaines: Effect of hydrophilicity and backbone structure on nonfouling properties. Langmuir 2012, 28, 666–675. [Google Scholar] [CrossRef]
- Charrier, A.; Mischki, T.; Lopinski, G.P. Direct stabilization of a phospholipid monolayer on H-terminated silicon. Langmuir 2009, 26, 2538–2543. [Google Scholar] [CrossRef]
- Ma, G.; Cheng, Q. Vesicular polydiacetylene sensor for colorimetric signaling of bacterial pore-forming toxin. Langmuir 2005, 21, 6123–6126. [Google Scholar] [CrossRef]
- Shenoy, D.K.; Barger, W.R.; Singh, A.; Panchal, R.G.; Misakian, M.; Stanford, V.M.; Kasianowicz, J.J. Functional reconstitution of protein ion channels into planar polymerizable phospholipid membranes. Nano Lett. 2005, 5, 1181–1185. [Google Scholar] [CrossRef]
- Morigaki, K.; Schönherr, H.; Okazaki, T. Polymerization of diacetylene phospholipid bilayers on solid substrate: Influence of the film deposition temperature. Langmuir 2007, 23, 12254–12260. [Google Scholar] [CrossRef]
- Okazaki, T.; Inaba, T.; Tatsu, Y.; Tero, R.; Urisu, T.; Morigaki, K. Polymerized lipid bilayers on a solid substrate: Morphologies and obstruction of lateral diffusion. Langmuir 2008, 25, 345–351. [Google Scholar]
- Guo, C.; Liu, S.; Jiang, C.; Li, W.; Dai, Z.; Fritz, H.; Wu, X. A promising drug controlled-release system based on diacetylene/phospholipid polymerized vesicles. Langmuir 2009, 25, 13114–13119. [Google Scholar] [CrossRef]
- Okazaki, T.; Tatsu, Y.; Morigaki, K. Phase separation of lipid microdomains controlled by polymerized lipid bilayer matrices. Langmuir 2010, 26, 4126–4129. [Google Scholar] [CrossRef]
- Puri, A.; Jang, H.; Yavlovich, A.; Masood, M.A.; Veenstra, T.D.; Luna, C.; Aranda-Espinoza, H.; Nussinov, R.; Blumenthal, R. Material properties of matrix lipids determine the conformation and intermolecular reactivity of diacetylenic phosphatidylcholine in the lipid bilayer. Langmuir 2011, 27, 15120–15128. [Google Scholar] [CrossRef]
- Morigaki, K.; Kimura, S.; Okada, K.; Kawasaki, T.; Kawasaki, K. Formation of substrate-supported membranes from mixtures of long- and short-chain phospholipids. Langmuir 2012, 28, 9649–9655. [Google Scholar] [CrossRef]
- Temprana, C.F.; Duarte, E.L.; Femia, A.L.; Alonso Sdel, V.; Lamy, M.T. Structural effect of cationic amphiphiles in diacetylenic photopolymerizable membranes. Chem. Phys. Lipids 2012, 165, 589–600. [Google Scholar] [CrossRef]
- Morigaki, K.; Mizutani, K.; Saito, M.; Okazaki, T.; Nakajima, Y.; Tatsu, Y.; Imaishi, H. Surface functionalization of a polymeric lipid bilayer for coupling a model biological membrane with molecules, cells, and microstructures. Langmuir 2013, 29, 2722–2730. [Google Scholar] [CrossRef]
- Smith, C.E.; Kong, H. Cross-linkable liposomes stabilize a magnetic resonance contrast-enhancing polymeric fastener. Langmuir 2014, 30, 3697–3704. [Google Scholar] [CrossRef]
- Albrecht, O.; Laschewsky, A.; Ringsdorf, H. Investigations of polymerizable multilayers as gas separation membranes. J. Membr. Sci. 1985, 22, 187–197. [Google Scholar] [CrossRef]
- Gal, Y.S.; Lee, W.C.; Jin, S.H.; Lim, K.T.; Park, J.W.; Kim, S.Y. Electrochemical properties of a self-dopable ionic conjugated polymer: Poly[2-ethynyl-N-(4-sulfobutyl)pyridinium betaine]. Mol. Cryst. Liq. Cryst. 2008, 492, 229–236. [Google Scholar]
- Jenkins, I.H.; Kar, A.K.; Lindsell, W.E.; Murray, C.; Preston, P.N.; Wang, C.; Wherrett, B.S. Synthesis and optical characterization of polydiacetylenes containing carboxylic acid, carbamate, phosphonium, and quaternary ammonium functionalities. Macromol. 1996, 29, 6365–6370. [Google Scholar] [CrossRef]
- Hub, H.H.; Hupfer, B.; Koch, H.; Ringsdorf, H. Polymerization of lipid and lysolipid like diacetylenes in monolayers and liposomes. J. Macromol. Sci. A 1981, 15, 701–715. [Google Scholar] [CrossRef]
- Yang, W.; Ella-Menye, J.R.; Liu, S.; Bai, T.; Wang, D.; Yu, Q.; Li, Y.; Jiang, S. Cross-linked carboxybetaine SAMs enable nanoparticles with remarkable stability in complex media. Langmuir 2014, 30, 2522–2529. [Google Scholar] [CrossRef]
- Gal, Y.S.; Jin, S.H.; Park, Y.I.; Park, J.W.; Lyoo, W.S.; Lim, K.T. Synthesis and properties of an ionic polyacetylene by the activated polymerization of 2-ethynylpyridine with the ring-opening of propiolactone. Synth. Met. 2011, 161, 445–449. [Google Scholar] [CrossRef]
- Tan, H.; Liu, J.; Li, J.; Jiang, X.; Xie, X.; Zhong, Y.; Fu, Q. Synthesis and hemocompatibility of biomembrane mimicing poly(carbonate urethane)s containing fluorinated alkyl phosphatidylcholine side groups. BioMacromol. 2006, 7, 2591–2599. [Google Scholar] [CrossRef]
- Yamada, M.; Li, Y.; Nakaya, T. Synthesis and properties of polyurethanes containing phosphatidylcholine analogues in the side chains. J. Macromol. Sci. A 1995, 32, 1235–1242. [Google Scholar] [CrossRef]
- Korematsu, A.; Murakami, T.; Sakurai, I.; Kodama, M.; Nakaya, T. Synthesis, characterization and blood compatibilities of novel segmented polyurethanes containing poly(butadiene) soft segments and phosphatidylcholine analogues in the main chains and long-chain alkyl groups in the side chains. J. Mater. Chem. 1999, 9, 647–653. [Google Scholar] [CrossRef]
- Tan, H.; Sun, T.; Li, J.; Guo, M.; Xie, X.; Zhong, Y.; Fu, Q.; Jiang, L. Novel biomembrane-mimicking polymer surface with environmental responsiveness. Macromol. Rapid Commun. 2005, 26, 1418–1422. [Google Scholar] [CrossRef]
- Iwasaki, Y.; Nakabayashi, N.; Ishihara, K. Synthesis of novel phospholipid polymers by polycondensation. Macromol. Rapid Commun. 2000, 21, 287–290. [Google Scholar] [CrossRef]
- Hankari, S.E.; Motos-Perez, B.; Hesemann, P.; Bouhaouss, A.; Moreau, J.J.E. Periodic mesoporous organosilica from zwitterionic precursors. Chem. Commun. 2011, 47, 6704–6706. [Google Scholar]
- Anton, P.; Laschewsky, A. Synthesis of polymeric surfactants by radical thiol/ene addition reaction. Eur. Polym. J. 1995, 31, 387–394. [Google Scholar] [CrossRef]
- Samuel, N.K.P.; Singh, M.; Yamaguchi, K.; Regen, S.L. Polymerized-depolymerized vesicles. Reversible thiol-disulfide-based phosphatidylcholine membranes. J. Am. Chem. Soc. 1985, 107, 42–47. [Google Scholar] [CrossRef]
- Diem, T.; Czajka, B.; Weber, B.; Regen, S.L. Spontaneous-assembly of phospholipid monolayers via adsorption onto gold. J. Am. Chem. Soc. 1986, 108, 6094–6095. [Google Scholar] [CrossRef]
- Regen, S.L.; Samuel, N.K.P.; Khurana, J.M. Polymerization of macrocyclic phospholipid- and surfactant-based vesicles. J. Am. Chem. Soc. 1985, 107, 5804–5805. [Google Scholar]
- Weber, B.A.; Dodrer, N.; Regen, S.L. Phospholipid membranes from a polymeric phosphatidylcholine. J. Am. Chem. Soc. 1987, 109, 4419–4421. [Google Scholar] [CrossRef]
- Stefely, J.; Markowitz, M.A.; Regen, S.L. Permeability characteristics of lipid bilayers from lipoic acid-derived phosphatidylcholines. Comparison of monomeric, crosslinked and noncrosslinked polymerized membranes. J. Am. Chem. Soc. 1988, 110, 7463–7469. [Google Scholar] [CrossRef]
- Koch, H.; Laschewsky, A.; Ringsdorf, H.; Teng, K. Photodimerization and photopolymerization of amphiphilic cinnamic acid derivatives in oriented monolayers, vesicles and solution. Macromol. Chem. Phys. 1986, 187, 1843–1853. [Google Scholar] [CrossRef]
- Nakaya, T.; Yasuzawa, M.; Imoto, M. Poly (phosphatidylcholine) analogs. Macromol. 1989, 22, 3180–3181. [Google Scholar] [CrossRef]
- Eldo, J.; Ajayaghosh, A. New low band gap polymers: Control of optical and electronic properties in near infrared absorbing π-conjugated polysquaraines. Chem. Mater. 2001, 14, 410–418. [Google Scholar] [CrossRef]
- Theato, P. Synthesis of well-defined polymeric activated esters. J. Polym. Sci., Part A: Polym. Chem. 2008, 46, 6677–6687. [Google Scholar] [CrossRef]
- Bohrisch, J.; Wendler, U.; Jaeger, W. Controlled radical polymerization of 4-vinylpyridine. Macromol. Rapid Commun. 1997, 18, 975–982. [Google Scholar] [CrossRef]
- Bohrisch, J.; Grosche, O.; Wendler, U.; Jaeger, W.; Engelhardt, H. Electroosmotic mobility and aggregation phenomena of model polymers with permanent cationic groups. Macromol. Chem. Phys. 2000, 201, 447–452. [Google Scholar] [CrossRef]
- Izumrudov, V.A.; Zelikin, A.N.; Zhiryakova, M.V.; Jaeger, W.; Bohrisch, J. Interpolyelectrolyte reactions in solutions of polycarboxybetaines. J. Phys. Chem. B 2003, 107, 7982–7986. [Google Scholar] [CrossRef]
- Yaroslavov, A.A.; Sitnikova, T.A.; Rakhnyanskaya, A.A.; Ermakov, Y.A.; Burova, T.V.; Grinberg, V.Y.; Menger, F.M. Contrasting behavior of zwitterionic and cationic polymers bound to anionic liposomes. Langmuir 2007, 23, 7539–7544. [Google Scholar]
- Yaroslavov, A.A.; Sitnikova, T.A.; Rakhnyanskaya, A.A.; Yaroslavova, E.G.; Davydov, D.A.; Burova, T.V.; Grinberg, V.Y.; Shi, L.; Menger, F.M. Biomembrane sensitivity to structural changes in bound polymers. J. Am. Chem. Soc. 2009, 131, 1666–1667. [Google Scholar] [CrossRef]
- Gui, Z.; Du, B.; Qian, J.; An, Q.; Zhao, Q. Construction and deconstruction of multilayer films containing polycarboxybetaine: Effect of pH and ionic strength. J. Coll. Interf. Sci. 2011, 353, 98–106. [Google Scholar] [CrossRef]
- Sinclair, A.; Bai, T.; Carr, L.R.; Ella-Menye, J.R.; Zhang, L.; Jiang, S. Engineering buffering and hydrolytic or photolabile charge shifting in a polycarboxybetaine ester gene delivery platform. BioMacromol. 2013, 14, 1587–1593. [Google Scholar] [CrossRef]
- Cao, Z.; Yu, Q.; Xue, H.; Cheng, G.; Jiang, S. Nanoparticles for drug delivery prepared from amphiphilic PLGA zwitterionic block copolymers with sharp contrast in polarity between two blocks. Angew. Chem. Int. Ed. 2010, 49, 3771–3776. [Google Scholar] [CrossRef]
- Ramireddy, R.R.; Subrahmanyam, A.V.; Thayumanavan, S. Zwitterionic moieties from the huisgen reaction: A case study with amphiphilic dendritic assemblies. Chem. Eur. J. 2013, 19, 16374–16381. [Google Scholar] [CrossRef]
- Gonsior, N.; Schmitz, S.; Ritter, H. Thermal sensitivity of tert-butyloxycarbonylmethyl-modified polyquats in condensed phase and solubility properties of copolymers with N-isopropylacrylamide. Macromol. Chem. Phys. 2010, 211, 1695–1700. [Google Scholar] [CrossRef]
- Keefe, A.J.; Jiang, S. Poly(zwitterionic)protein conjugates offer increased stability without sacrificing binding affinity or bioactivity. Nat. Chem. 2012, 4, 59–63. [Google Scholar] [CrossRef]
- Sobolciak, P.; Spirek, M.; Katrlik, J.; Gemeiner, P.; Lacik, I.; Kasak, P. Light-switchable polymer from cationic to zwitterionic form: Synthesis, characterization, and interactions with DNA and bacterial cells. Macromol. Rapid Commun. 2013, 34, 635–639. [Google Scholar] [CrossRef]
- Zeng, R.; Fu, H.; Zhao, Y. Synthesis of novel biomimetic zwitterionic phosphorylcholine-bound chitosan derivative. Macromol. Rapid Commun. 2006, 27, 548–552. [Google Scholar] [CrossRef]
- Zeng, R.; Guo, K.; Wang, Z.; Tu, M.; Zhao, J.; Wang, J. Synthesis and self-assembly of biomimetic phosphorylcholine-bound chitosan derivatives. React. Funct. Polym. 2012, 72, 745–751. [Google Scholar] [CrossRef]
- Ali, S.A.; Al-Hamouz, O.C.S. Comparative solution properties of cyclocopolymers having cationic, anionic, zwitterionic and zwitterionic/anionic backbones of similar degree of polymerization. Polymer 2012, 53, 3368–3377. [Google Scholar] [CrossRef]
- Hu, N.; Johnson, L.M.; Pothayee, N.; Pothayee, N.; Lin, Y.; Davis, R.M.; Riffle, J.S. Synthesis of ammonium bisphosphonate monomers and polymers. Polymer 2013, 54, 3188–3197. [Google Scholar] [CrossRef]
- Huang, X.J.; Xu, Z.K.; Wan, L.S.; Wang, Z.G.; Wang, J.L. Novel acrylonitrile-based copolymers containing phospholipid moieties: Synthesis and characterization. Macromol. Biosci. 2005, 5, 322–330. [Google Scholar] [CrossRef]
- Kim, G.; Park, S.; Jung, J.; Heo, K.; Yoon, J.; Kim, H.; Kim, I.J.; Kim, J.R.; Lee, J.I.; Ree, M. Novel brush polymers with phosphorylcholine bristle ends: synthesis, structure, properties, and biocompatibility. Adv. Funct. Mater. 2009, 19, 1631–1644. [Google Scholar]
- Jung, J.; Kim, H.; Ree, M. Self-assembly of novel lipid-mimicking brush polymers in nanoscale thin films. Soft Matter 2014, 10, 701–708. [Google Scholar] [CrossRef]
- Reisch, A.; Voegel, J.C.; Decher, G.; Schaaf, P.; Mésini, P.J. Synthesis of polyelectrolytes bearing phosphorylcholine moieties. Macromol. Rapid Commun. 2007, 28, 2217–2223. [Google Scholar] [CrossRef]
- Kim, G.; Yoo, C.E.; Kim, M.; Kang, H.J.; Park, D.; Lee, M.; Huh, N. Noble polymeric surface conjugated with zwitterionic moieties and antibodies for the isolation of exosomes from human serum. Bioconj. Chem. 2012, 23, 2114–2120. [Google Scholar] [CrossRef]
- Woodfield, P.A.; Zhu, Y.; Pei, Y.; Roth, P.J. Hydrophobically modified sulfobetaine copolymers with tunable aqueous UCST through postpolymerization modification of poly(pentafluorophenyl acrylate). Macromol. 2014, 47, 750–762. [Google Scholar] [CrossRef]
- Ebbesen, M.F.; Schaffert, D.H.; Crowley, M.L.; Oupický, D.; Howard, K.A. Synthesis of click-reactive HPMA copolymers using RAFT polymerization for drug delivery applications. J. Polym. Sci., Part A: Polym. Chem. 2013, 51, 5091–5099. [Google Scholar] [CrossRef]
- Huang, J.; Xu, W. Efficient synthesis of zwitterionic sulfobetaine group functional polyurethanes via “click” reaction. J. Appl. Polym. Sci. 2011, 122, 1251–1257. [Google Scholar] [CrossRef]
- Yu, X.; Liu, Z.; Janzen, J.; Chafeeva, I.; Horte, S.; Chen, W.; Kainthan, R.K.; Kizhakkedathu, J.N.; Brooks, D.E. Polyvalent choline phosphate as a universal biomembrane adhesive. Nat. Mater. 2012, 11, 468–476. [Google Scholar] [CrossRef]
- Yu, X.; Zou, Y.; Horte, S.; Janzen, J.; Kizhakkedathu, J.N.; Brooks, D.E. Thermal reversal of polyvalent choline phosphate, a multivalent universal biomembrane adhesive. BioMacromol. 2013, 14, 2611–2621. [Google Scholar] [CrossRef]
- Yu, X.; Yang, X.; Horte, S.; Kizhakkedathu, J.N.; Brooks, D.E. A thermoreversible poly(Choline Phosphate) based universal biomembrane adhesive. Macromol. Biosci. 2014, 14, 334–339. [Google Scholar] [CrossRef]
- Sun, J.; Zeng, F.; Jian, H.; Wu, S. Conjugation with betaine: A facile and effective approach to significant improvement of gene delivery properties of PEI. BioMacromol. 2013, 14, 728–736. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Z.; Ma, G.; Lin, W.; Chen, S. Reducing the cytotoxity of poly(amidoamine) dendrimers by modification of a single layer of carboxybetaine. Langmuir 2013, 29, 8914–8921. [Google Scholar] [CrossRef]
- Lee, W.F.; Huang, G.Y. Poly(sulfobetaine)s and corresponding cationic polymers: 5. Synthesis and dilute aqueous solution properties of poly(sulfobetaine)s derived from acrylamide-maleic anhydride copolymer. Polymer 1996, 37, 4389–4395. [Google Scholar] [CrossRef]
- Lee, W.F.; Lee, C.H. Poly(sulfobetaine)s and corresponding cationic polymers: 3. Synthesis and dilute aqueous solution properties of poly(sulfobetaine)s derived from styrene-maleic anhydride. Polymer 1997, 38, 971–979. [Google Scholar] [CrossRef]
- Lee, W.F.; Chen, Y.M. Poly(sulfobetaine)s and corresponding cationic polymers. XI. Synthesis and aqueous solution properties of cationic poly(methyl iodide quaternized ethyl vinyl ether/N,N-dimethylaminopropyl maleamidic acid) copolymer. J. Appl. Polym. Sci. 2003, 89, 2261–2269. [Google Scholar] [CrossRef]
- Koetse, M.; Laschewsky, A.; Mayer, B.; Rolland, O.; Wischerhoff, E. Ultrathin coatings by multiple polyelectrolyte adsorption/surface activation (CoMPAS). Macromol. 1998, 31, 9316–9327. [Google Scholar] [CrossRef]
- Laschewsky, A.; Mayer, B.; Wischerhoff, E.; Arys, X.; Jonas, A.; Kaurannen, M.; Persoons, A. A new assembly technique for thin, defined multilayers. Angew. Chem. Int. Ed. 1997, 36, 2788–2791. [Google Scholar] [CrossRef]
- Thünemann, A.; Sander, K.; Jaeger, W.; Dimova, R. Polyampholyte-dressed micelles of fluorinated and hydrogenated dodecanoic acid. Langmuir 2002, 18, 5099–5105. [Google Scholar] [CrossRef]
- Note, C.; Koetz, J.; Wattebled, L.; Laschewsky, A. Effect of a new hydrophobically modified polyampholyte on the formation of inverse microemulsions and the preparation of gold nanoparticles. J. Coll. Interf. Sci. 2007, 308, 162–169. [Google Scholar] [CrossRef]
- Fechner, M.; Kramer, M.; Kleinpeter, E.; Koetz, J. Polyampholyte-modified ionic microemulsions. Colloid Polym. Sci. 2009, 287, 1145–1153. [Google Scholar] [CrossRef]
- Kathmann, E.E.; White, L.A.; McCormick, C.L. Water soluble polymers: 70. Effects of methylene versus propylene spacers in the pH and electrolyte responsiveness of zwitterionic copolymers incorporating carboxybetaine monomers. Polymer 1997, 38, 879–886. [Google Scholar] [CrossRef]
- Johnson, K.M.; Fevola, M.J.; McCormick, C.L. Hydrophobically modified acrylamide-based polybetaines. I. Synthesis, characterization, and stimuli-responsive solution behavior. J. Appl. Polym. Sci. 2004, 92, 647–657. [Google Scholar] [CrossRef]
- McCormick, C.; Armentrout, R.S. The synthesis and solution behavior of zwitterionic cyclocopolymers coantining a novel sulfobetaine monomer. Polym. Prepr. (Am. Chem. Soc., Div. Polym. Chem.) 1998, 39, 617–618. [Google Scholar]
- Kim, M.; Kim, J.C.; Rho, Y.; Jung, J.; Kwon, W.; Kim, H.; Ree, M. Bacterial adherence on self-assembled films of brush polymers bearing zwitterionic sulfobetaine moieties. J. Mater. Chem. 2012, 22, 19418–19428. [Google Scholar] [CrossRef]
- Kaladas, J.J.; Kastrup, R.; Schulz, D.N. Poly(cyclosulfobetaines): Synthesis, characterization, and solution properties. Polym. Prepr. (Am. Chem. Soc., Div. Polym. Chem.) 1998, 39, 619–620. [Google Scholar]
- Ali, S.A.; Umar, Y.; Abu-Sharkh, B.F.; Al-Muallem, H.A. Synthesis and comparative solution properties of single-, twin-, and triple-tailed associating ionic polymers based on diallylammonium salts. J. Polym. Sci., Part A: Polym. Chem. 2006, 44, 5480–5494. [Google Scholar] [CrossRef]
- Hussein, I.A.; Ali, S.K.A.; Suleiman, M.A.; Umar, Y. Rheological behavior of associating ionic polymers based on diallylammonium salts containing single-, twin-, and triple-tailed hydrophobes. Eur. Polym. J. 2010, 46, 1063–1073. [Google Scholar] [CrossRef]
- Ali, S.A.; Al-Muallem, H.A.; Mazumder, M.A.J. Coexistence curves of aqueous two-phase systems of some pH-responsive homo- and copolymers of 3-(diallylammonio)propane-1-sulfonate and urethanized Poly(ethenol) or Poly(oxyethylene). J. Chem. Eng. Data 2013, 58, 2574–2585. [Google Scholar] [CrossRef]
- Kharlampieva, E.; Izumrudov, V.A.; Sukhishvili, S.A. Electrostatic layer-by-layer self-assembly of poly(carboxybetaine)s: role of zwitterions in film growth. Macromol. 2007, 40, 3663–3668. [Google Scholar] [CrossRef]
- Zhang, Z.; Cheng, G.; Carr, L.R.; Vaisocherová, H.; Chen, S.; Jiang, S. The hydrolysis of cationic polycarboxybetaine esters to zwitterionic polycarboxybetaines with controlled properties. Biomater. 2008, 29, 4719–4725. [Google Scholar] [CrossRef]
- Carr, L.R.; Jiang, S. Mediating high levels of gene transfer without cytotoxicity via hydrolytic cationic ester polymers. Biomater. 2010, 31, 4186–4193. [Google Scholar] [CrossRef]
- Arnáiz, E.; Vacas-Córdoba, E.; Galán, M.; Pion, M.; Gómez, R.; Muñoz-Fernández, M.A.Á.; de la Mata, F.J. Synthesis of anionic carbosilane dendrimers via “click chemistry” and their antiviral properties against HIV. J. Polym. Sci., Part A: Polym. Chem. 2014, 52, 1099–1112. [Google Scholar] [CrossRef]
- Endo, T.; Nagai, D.; Monma, T.; Yamaguchi, H.; Ochiai, B. A novel construction of a reversible fixation−release system of carbon dioxide by amidines and their polymers. Macromol. 2004, 37, 2007–2009. [Google Scholar] [CrossRef]
- Coupillaud, P.; Pinaud, J.; Guidolin, N.; Vignolle, J.; Fèvre, M.; Veaudecrenne, E.; Taton, D. Poly(ionic liquid)s based on imidazolium hydrogen carbonate monomer units as recyclable polymer-supported N-heterocyclic carbenes: Use in organocatalysis. J. Polym. Sci., Part A: Polym. Chem. 2013, 51, 4530–4540. [Google Scholar]
- Soll, S.; Zhao, Q.; Weber, J.; Yuan, J. Activated CO2 sorption in mesoporous imidazolium-type poly(ionic liquid)-based polyampholytes. Chem. Mater. 2013, 25, 3003–3010. [Google Scholar] [CrossRef]
- Zhou, H.; Zhang, W.Z.; Wang, Y.M.; Qu, J.P.; Lu, X.B. N-heterocyclic carbene functionalized polymer for reversible fixation-release of CO2. Macromol. 2009, 42, 5419–5421. [Google Scholar] [CrossRef]
- Cao, Z.; Mi, L.; Mendiola, J.; Ella-Menye, J.R.; Zhang, L.; Xue, H.; Jiang, S. Reversibly switching the function of a surface between attacking and defending against bacteria. Angew. Chem. Int. Ed. 2012, 51, 2602–2605. [Google Scholar] [CrossRef]
- Koetse, M.; Laschewsky, A.; Mayer, B.; Rolland, O.; Wischerhoff, E. Ultrathin coatings by multiple polyelectrolyte adsorption/surface activation (CoMPAS). Macromol. 1998, 31, 9316–9327. [Google Scholar] [CrossRef]
- Berkovic, G.; Krongauz, V.; Weiss, V. Spiropyrans and spirooxazines for memories and switches. Chem. Rev. 2000, 100, 1741–1754. [Google Scholar] [CrossRef]
- Anton, P.; Köberle, P.; Laschewsky, A. Structure and properties of zwitterionic polysoaps: Functionalization by redox-switchable moieties. Progr. Colloid Polym. Sci. 1992, 89, 56–59. [Google Scholar] [CrossRef]
- Anton, P.; Heinze, J.; Laschewsky, A. Redox-active monomeric and polymeric surfactants. Langmuir 1993, 9, 77–85. [Google Scholar] [CrossRef]
- Anton, P.; Laschewsky, A.; Ward, M.D. Solubilization control by redox-switching of polysoaps. Polym. Bull. 1995, 34, 331–335. [Google Scholar] [CrossRef]
- Song, J.; Janczewski, D.; Ma, Y.; Hempenius, M.; Xu, J.; Vancso, G.J. Redox-controlled release of molecular payloads from multilayered organometallic polyelectrolyte films. J. Mater. Chem. B 2013, 1, 828–834. [Google Scholar] [CrossRef]
- Song, J.; Jańczewski, D.; Ma, Y.; Hempenius, M.; Xu, J.; Vancso, G.J. Disassembly of redox responsive poly(ferrocenylsilane) multilayers: The effect of blocking layers, supporting electrolyte and polyion molar mass. J. Coll. Interface Sci. 2013, 405, 256–261. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Laschewsky, A. Structures and Synthesis of Zwitterionic Polymers. Polymers 2014, 6, 1544-1601. https://doi.org/10.3390/polym6051544
Laschewsky A. Structures and Synthesis of Zwitterionic Polymers. Polymers. 2014; 6(5):1544-1601. https://doi.org/10.3390/polym6051544
Chicago/Turabian StyleLaschewsky, André. 2014. "Structures and Synthesis of Zwitterionic Polymers" Polymers 6, no. 5: 1544-1601. https://doi.org/10.3390/polym6051544
APA StyleLaschewsky, A. (2014). Structures and Synthesis of Zwitterionic Polymers. Polymers, 6(5), 1544-1601. https://doi.org/10.3390/polym6051544