Enhancement Corrosion Resistance of (γ-Glycidyloxypropyl)-Silsesquioxane-Titanium Dioxide Films and Its Validation by Gas Molecule Diffusion Coefficients Using Molecular Dynamics (MD) Simulation
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Preparation of Films
2.3. Electrochemical Measurements [8]
3. Computational Methodology

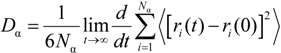
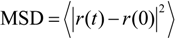
4. Results and Discussion
4.1. Corrosion Resistance of Modified Hybrid Films
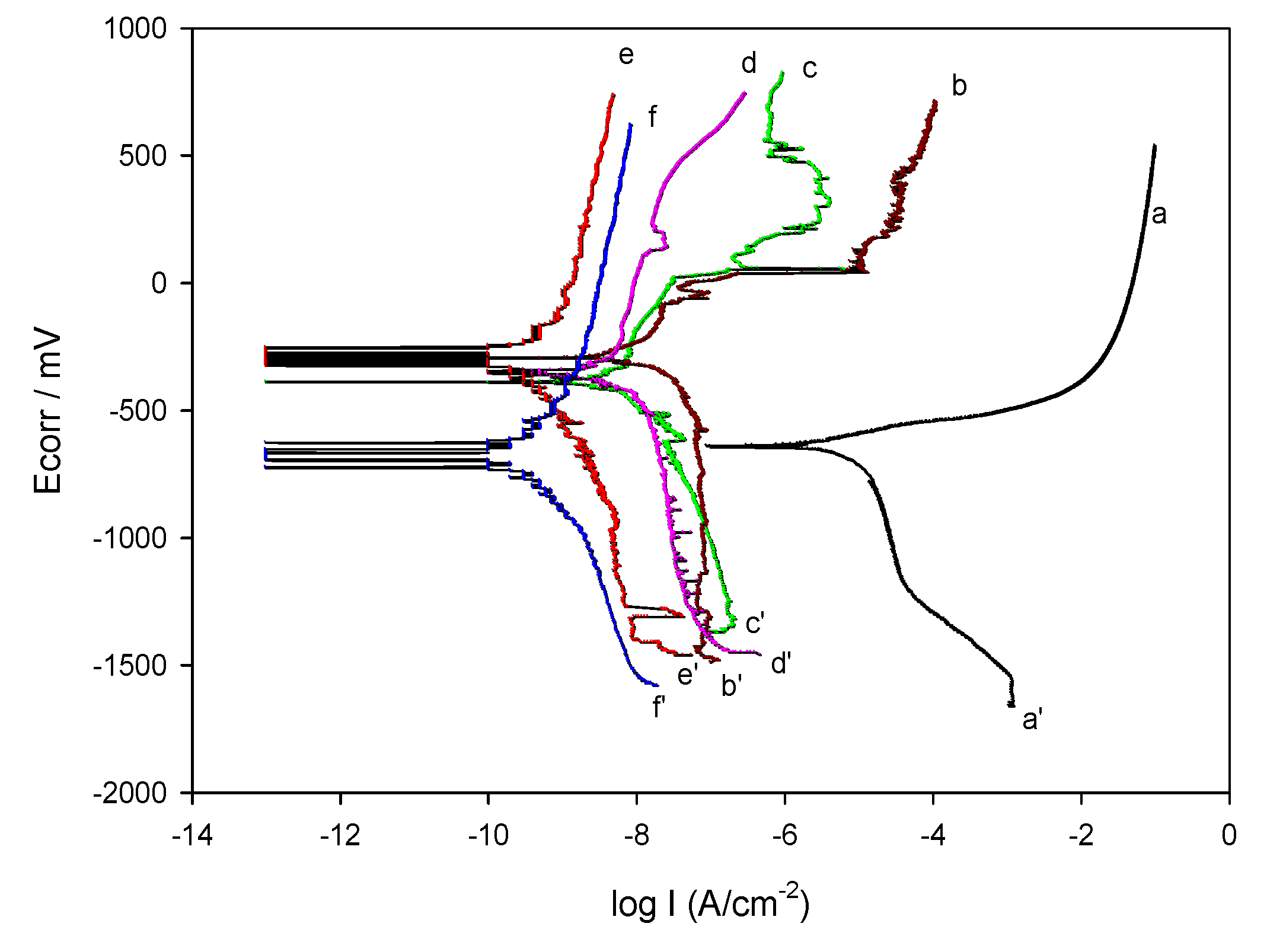
4.2. Correlation between the Film Structure and Anticorrosion Properties

4.3. Verification of Enhancement Corrosion Resistance by SDC Using MD Simulation
| Cells | Penetrant | Slope | SDC (10−8·cm2·s−1) |
|---|---|---|---|
| f-GS | H2O | 5.27 | 0.88 |
| NO2 | 3.99 | 0.66 | |
| SO2 | 2.81 | 0.47 | |
| f-GSTT5% | H2O | 4.26 | 0.71 |
| NO2 | 2.93 | 0.49 | |
| SO2 | 2.04 | 0.34 | |
| H2O | 3.76 | 0.63 | |
| f-GSTT10% | NO2 | 2.52 | 0.42 |
| SO2 | 1.76 | 0.29 | |
| H2O | 3.23 | 0.54 | |
| f-GSTT15% | NO2 | 2.18 | 0.36 |
| SO2 | 1.53 | 0.26 | |
| H2O | 2.66 | 0.44 | |
| f-GSTT20% | NO2 | 1.81 | 0.30 |
| SO2 | 1.26 | 0.21 | |
| H2O | 2.05 | 0.34 | |
| f-GSTT25% | NO2 | 1.37 | 0.23 |
| SO2 | 1.06 | 0.18 |

5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Zhang, X.; Hu, L.; Sun, D. Nanoindentation and nanoscratch profiles of hybrid films based on (γ-methacrylpropyl)trimethoxysilane and tetraethoxysilane. Acta Mater. 2006, 54, 5469–5475. [Google Scholar] [CrossRef]
- Xue, Y.H.; Liu, Y.; Lu, F.; Qu, J.; Chen, H.; Dai, L.M. Functionalization of graphene oxide with polyhedral oligomeric silsesquioxane (POSS) for multifunctional applications. J. Phys. Chem. Lett. 2012, 3, 1607–1612. [Google Scholar] [CrossRef]
- Wang, H.; Lin, D.; Wang, D.; Hu, L.; Huang, Y.; Liu, L.; Loy, D.A. Computational and experimental determinations of the UV adsorption of polyvinylsilsesquioxane-silica and titanium dioxide hybrids. Bio-Med. Mater. Eng. 2014, 24, 651–657. [Google Scholar]
- Metroke, T.L.; Kachurina, O.M.; Knobbe, E.T. Spectroscopic and corrosion resistance characterization of GLYMO TEOS ormosil coatings for aluminium alloy corrosion inhibition. Progr. Org. Coat. 2002, 44, 295–305. [Google Scholar] [CrossRef]
- Eisenberg, P.; Erra-Balsells, R.; Ishikawa, Y.; Lucas, J.C.; Mauri, A.N.; Nonami, H.; Riccardi, C.C.; Williams, R.J.J. Cagelike precursors of high-molar-mass silsesquioxanes formed by the hydrolytic condensation of trialkoxysilanes. Macromolecules 2000, 33, 1940–1947. [Google Scholar] [CrossRef]
- Wang, D.; You, H.; Hu, L. Study of three–dimensional configurations of (γ-methacryloxypropyl) silsesquioxanes by ultraviolet laser matrix–assisted desorption/ionization time-of-flight mass spectrometry and quantum chemical calculation. Rapid Commun. Mass Spectrom. 2011, 25, 1652–1660. [Google Scholar] [CrossRef]
- Liu, Y.; Sun, D.; You, H.; Chung, J.S. Corrosion resistance properties of organic-inorganic hybrid coatings on 2024 aluminum alloy. Appl. Surf. Sci. 2005, 246, 82–89. [Google Scholar] [CrossRef]
- Wang, D.; Chen, X.; Zhang, X.; Liu, Y.; Hu, L. Enhancement corrosion resistance of (γ-methacryloxypropyl) silsesquioxane hybrid films and its validation by gas-molecule diffusion coefficients using MD simulation. J. Sol-Gel Sci. Tech. 2009, 49, 293–300. [Google Scholar] [CrossRef]
- Behera, D.; Banthia, A.K. BisGMA/TiO2 organic-inorganic hybrid nanocomposite. Polym. Plast. Technol. Eng. 2007, 46, 1181–1186. [Google Scholar] [CrossRef]
- Hu, L.; Hu, Y.; You, H. Nanoscratchprofiles of SSO Film Based on (g-methacrylpropyl)-Trimethoxysilane Modified with Titanium Tetrabutoxide. In Proceedings of the Abstracts of Papers of 245th National Spring Meeting of the American Chemical Society (ACS), New Orleans, LA, USA, 7–11 April 2013.
- Hwang, D.K.; Moon, J.H.; Shul, Y.G.; Jung, K.T.; Kim, D.H.; Lee, D.W. Scratch resistant and transparent UV-protective coating on polycarbonate. J. Sol-Gel Sci. Technol. 2003, 26, 783–787. [Google Scholar] [CrossRef]
- Schmidt, H.K.; Mennig, M.; Nonninger, R.; Oliveira, P.W.; Schirra, H. Organic-inorganic hybrid materials processing and applications. Mater. Res. Soc. Symp. Proc. 1999, 576, 395–407. [Google Scholar] [CrossRef]
- Liu, Q.L.; Huang, Y. Structure-related diffusion in poly(methyl methacrylate)/polyhedral oligomeric silsesquioxanes. J. Phys. Chem. B 2006, 110, 17375–17382. [Google Scholar] [CrossRef]
- Karayiannis, N.C.; Mavrantzas, V.G.; Theodorou, D.N. Detailed atomistic simulation of the segmental dynamics and barrier properties of amorphous poly(ethylene terephthalate) and poly(ethylene isophthalate). Macromolecules 2004, 37, 2978–2995. [Google Scholar] [CrossRef]
- Meunier, M. Diffusion coefficients of small gas molecules in amorphous cis-1,4-polybutadiene estimated by molecular dynamics simulations. J. Chem. Phys. 2005, 123, 134906:1–134906:7. [Google Scholar]
- Charati, S.G.; Stern, S.A. Diffusion of gases in silicone polymers: Molecular dynamics simulations. Macromolecules 1998, 31, 5529–5535. [Google Scholar] [CrossRef]
- Wang, D.; Zhu, P.; Wei, S.; Hu, L. Influence of molecular structure of POSS on gas-molecule diffusion coefficients using molecular dynamic simulation. Mater. Sci. For. 2011, 689, 114–121. [Google Scholar]
- Xie, G.; Wang, P.; Hu, L. Enhancement corrosion resistance of (γ-glycidyloxypropyl)-silsesquioxane hybrid films and its validation by gas-molecule diffusion coefficients using MD simulation. Mater. Sci. For. 2009, 610–613, 190–197. [Google Scholar]
- Kuznetsova, A.; Yates, J.T., Jr.; Zhou, G.; Yang, J.C.; Chen, X. Making a superior oxide corrosion passivation layer on Al using ozone. Langmuir 2001, 17, 2146–2152. [Google Scholar] [CrossRef]
- Morita, R.; Azuma, K.; Inoue, S.; Miyano, R.; Takikawa, H.; Kobayashi, A.; Fujiwara, E.; Uchida, H.; Yatsuzuka, M. Corrosion resistance of TiN coatings produced by various dry processes. Surf. Coat. Technol. 2001, 136, 207–210. [Google Scholar] [CrossRef]
- Jung, H.; Alfantazi, A. An electrochemical impedance spectroscopy and polarization study of nanocrystalline Co and Co–P alloy in 0.1 M H2SO4 solution. Electrochim. Acta 2006, 51, 1806–1814. [Google Scholar] [CrossRef]
- Sayed, S.Y.; El-Deab, M.S.; El-Anadouli, B.E.; Ateya, B.G. Synergistic effects of benzotriazole and copper ions on the electrochemical impedance spectroscopy and corrosion behavior of iron in sulfuric acid. J. Phys. Chem. B 2003, 107, 5575–5585. [Google Scholar]
- Li, W.; Luo, J. Electric properties and pitting susceptibility of passive films formed on iron in chromate solution. Electrochem. Commun. 1999, 1, 349–353. [Google Scholar] [CrossRef]
- Spellane, P. A DC electrochemical method for studying the inhibition of metal corrosion by chromate containing paint. Progr. Org. Coat. 1999, 35, 277–282. [Google Scholar] [CrossRef]
- Yeh, J.; Liou, S.; Lin, C.; Yu, C.; Chang, Y.; Lee, K. Anticorrosively enhanced PMMA-clay nanocomposite materials with quaternary alkylphosphonium salt as an intercalating agent. Chem. Mater. 2002, 14, 154–161. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, H.; Liu, L.; Huang, Y.; Wang, D.; Hu, L.; Loy, D.A. Enhancement Corrosion Resistance of (γ-Glycidyloxypropyl)-Silsesquioxane-Titanium Dioxide Films and Its Validation by Gas Molecule Diffusion Coefficients Using Molecular Dynamics (MD) Simulation. Polymers 2014, 6, 300-310. https://doi.org/10.3390/polym6020300
Wang H, Liu L, Huang Y, Wang D, Hu L, Loy DA. Enhancement Corrosion Resistance of (γ-Glycidyloxypropyl)-Silsesquioxane-Titanium Dioxide Films and Its Validation by Gas Molecule Diffusion Coefficients Using Molecular Dynamics (MD) Simulation. Polymers. 2014; 6(2):300-310. https://doi.org/10.3390/polym6020300
Chicago/Turabian StyleWang, Haiyan, Li Liu, Yudong Huang, Di Wang, Lijiang Hu, and Douglas A. Loy. 2014. "Enhancement Corrosion Resistance of (γ-Glycidyloxypropyl)-Silsesquioxane-Titanium Dioxide Films and Its Validation by Gas Molecule Diffusion Coefficients Using Molecular Dynamics (MD) Simulation" Polymers 6, no. 2: 300-310. https://doi.org/10.3390/polym6020300




