Preparation and Characterization of Poly(ethyl hydrazide) Grafted Oil Palm Empty Fruit Bunch for Removal of Ni(II) Ion in Aqueous Environment
Abstract
:1. Introduction
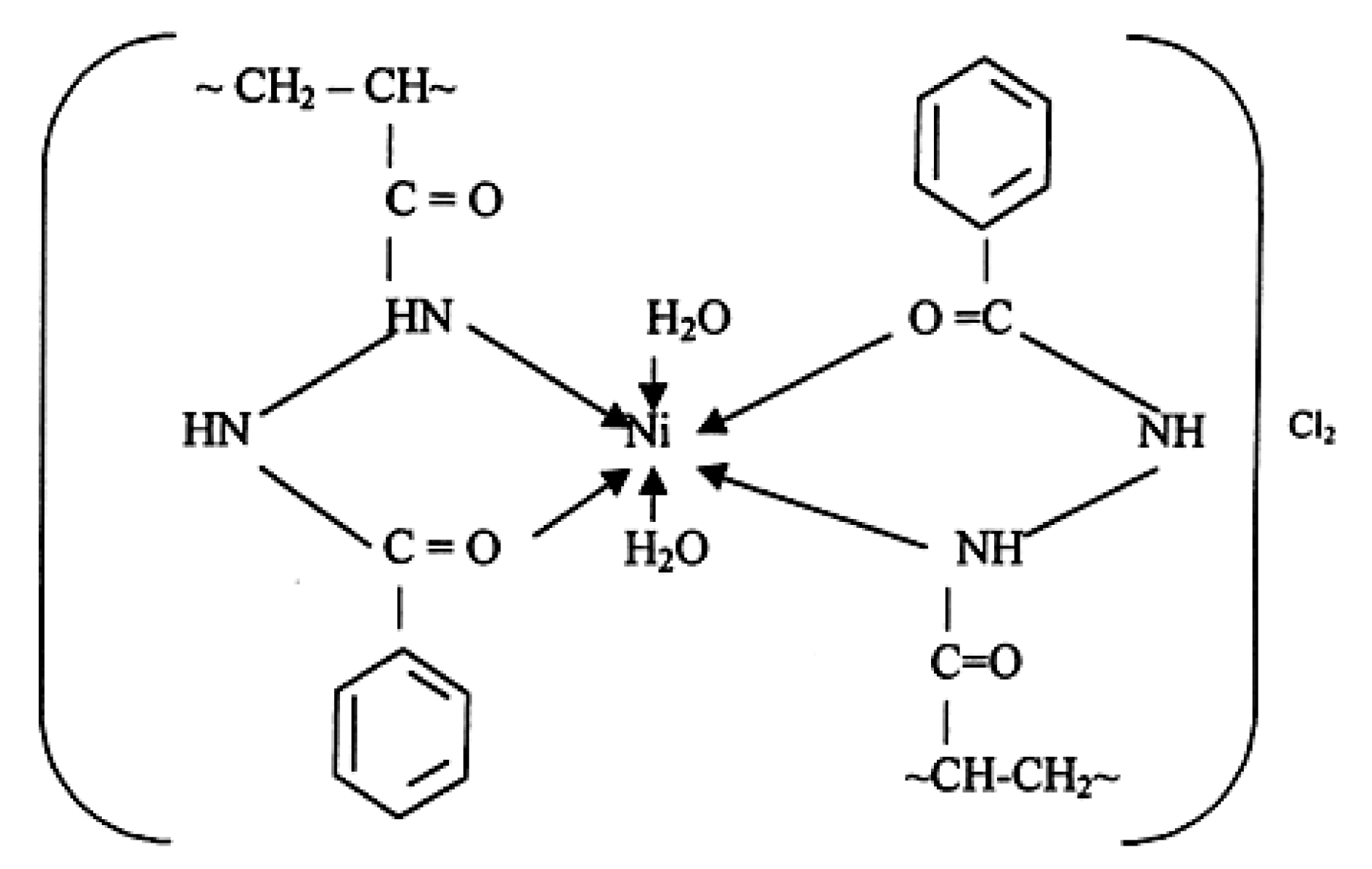
2. Experimental Section
2.1. Preparation of Poly(methyl acrylate) Grafted Oil Palm Empty Fruit Bunch (Pma-g-opefb)
2.2. Preparation of Peh-g-opefb
2.3. FTIR of Peh-g-opefb
2.4. Adsorption Studies
2.4.1. Effect of pH on Ni(II) Adsorption
2.4.2. Effect of Initial Concentration and Temperature on Nickel Ni(II) Adsorption
2.4.3. Kinetic Study
3. Results and Discussion
3.1. Characterization of Peh-g-opefb

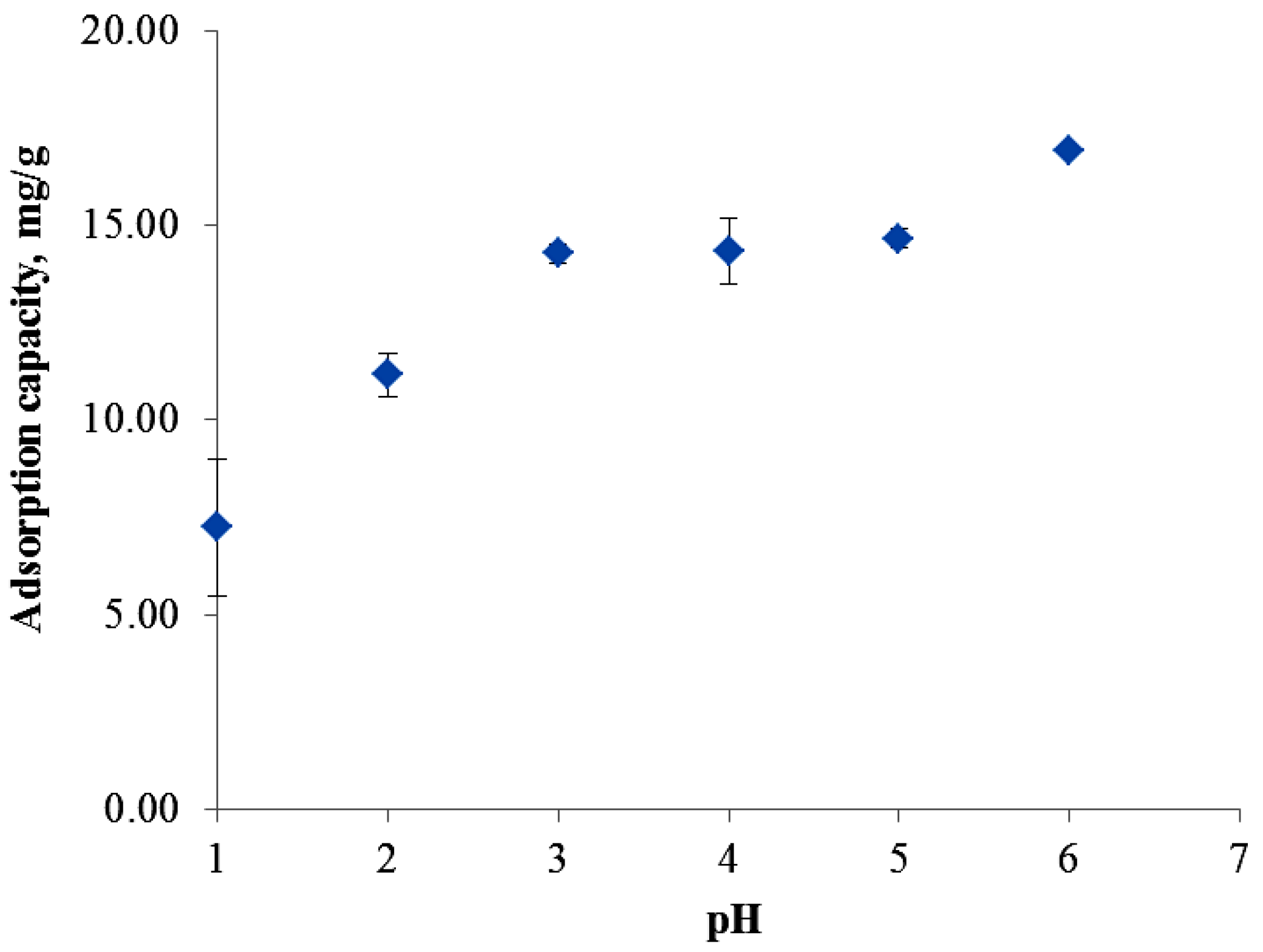
3.2. Effect of pH on Nickel (II) Adsorption
3.3. Effect of Initial Concentration on Ni(II) Adsorption
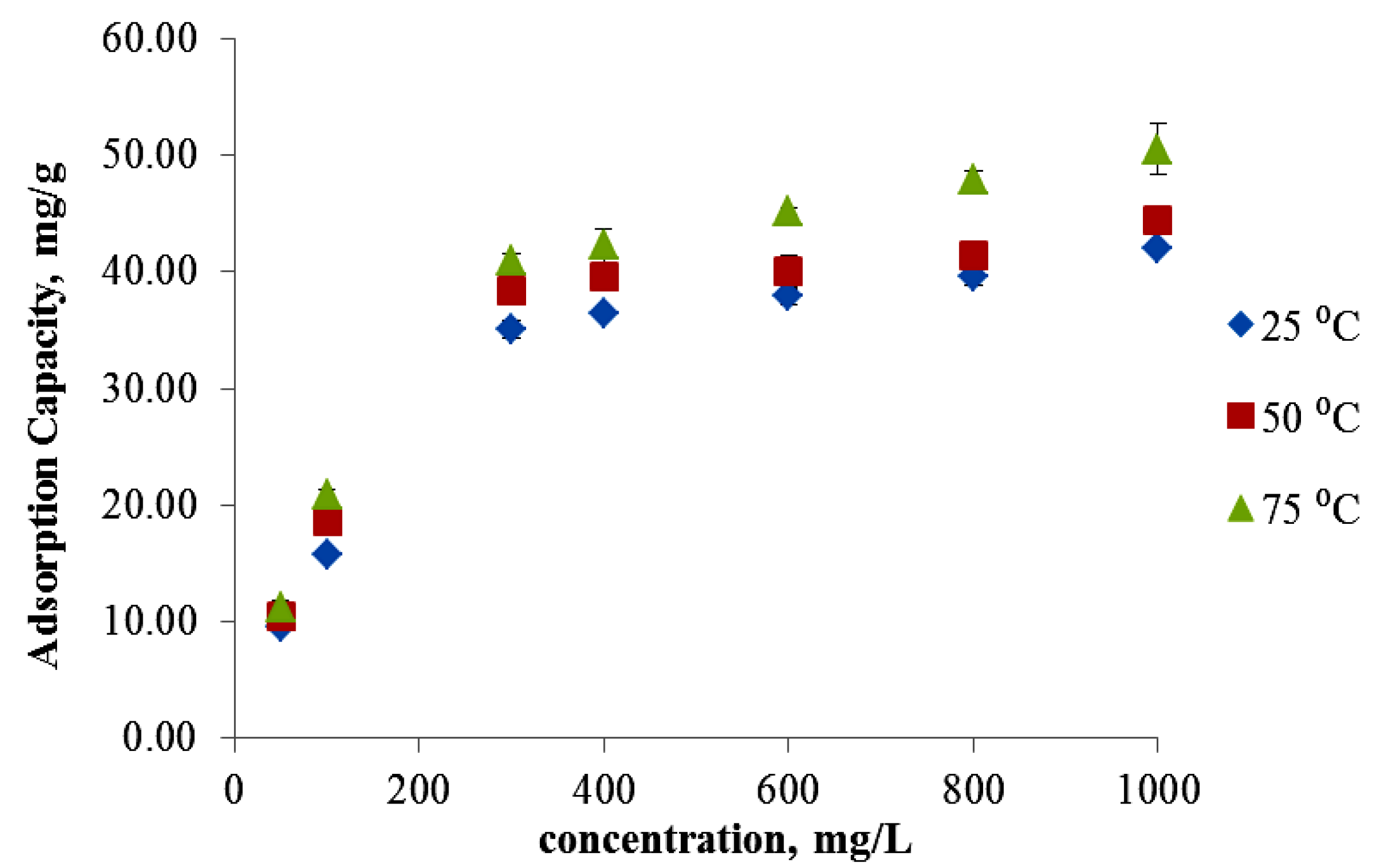
3.4. Effect of Temperature Ni(II) Adsorption
3.5. Adsorption Isotherm
| Temperature (°C) | Langmuir isotherm | Freundlich isotherm | Temkin isotherm | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| qmax(mg∙g−1) | b (L∙mg−1) | R2 | RL | KF [mg/g(L/mg)1/n] | n | R2 | lnA | b | R2 | |
| 25 | 42.19 | 0.0415 | 0.9981 | 0.1087 | 9.86 | 4.3975 | 0.9619 | 1.6643 | 5.0065 | 0.9395 |
| 50 | 44.44 | 0.0661 | 0.9959 | 0.0717 | 16.12 | 6.2461 | 0.9888 | 4.7376 | 3.8720 | 0.9862 |
| 75 | 51.02 | 0.0545 | 0.9936 | 0.0790 | 22.12 | 7.8678 | 0.9330 | 7.2172 | 3.5543 | 0.9731 |
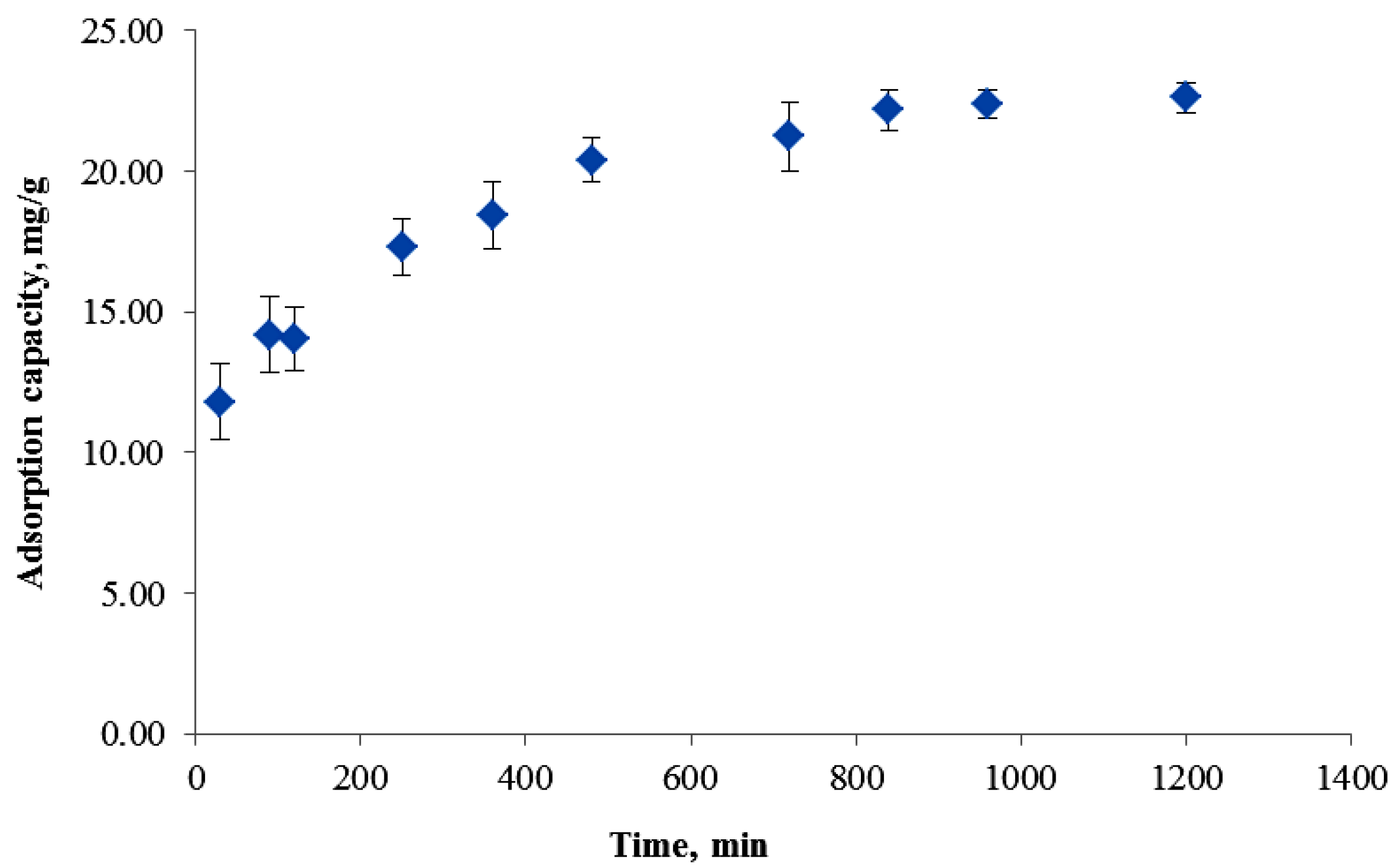
3.6. Adsorption Kinetic
3.7. Adsorption Thermodynamic
| Temperature (K) | Enthalpy, ∆ Hθ (kJ∙mol−1) | Entropy, ∆ Sθ (J∙mol−1∙K−1) | Free energy, −∆ Gθ (kJ∙mol−1) |
|---|---|---|---|
| 298 | 51.38 | 195.31 | 6.83 |
| 323 | 11.71 | ||
| 348 | 16.60 |
4. Conclusions
Acknowledgements
Conflict of Interest
References
- Keane, M.A. The removal of copper and nickel from aqueous solution using Y zeolite ion exchangers. Colloids Surf. A 1998, 138, 11–20. [Google Scholar] [CrossRef]
- Ozturk, A. Removal of nickel from aqueous solution by the bacterium Bacillus thuringiensis. J. Hazard. Mater. 2007, 147, 518–523. [Google Scholar] [CrossRef]
- Kasprzak, K.S.; Sunderman, F.W., Jr.; Salnikow, K. Review: Nickel carcinogenesis. Mutat. Res. 2003, 533, 67–97. [Google Scholar] [CrossRef]
- Aloma, I.; Martin-Lara, M.A.; Rodriguez, I.L.; Balzquez, G.; Calero, M. Removal of nickel (II) ions from aqueous solutions by biosorption on sugarcane bagasse. J. Taiwan Inst. Chem. Eng. 2012, 43, 275–281. [Google Scholar] [CrossRef]
- Denkhaus, E.; Salnikow, K. Nickel essentiality, toxicity, and carcinogenicity. Crit. Rev. Oncol. Hematol. 2002, 42, 35–56. [Google Scholar]
- Trombetta, D.; Mondello, M.R.; Cimino, F.; Cristani, M.; Pergolizzi, S.; Saija, A. Toxic effect of nickel in an in vitro model of human oral epithelium. Toxicol. Lett. 2005, 159, 219–225. [Google Scholar] [CrossRef]
- Padmavathy, V. Biosorption of nickel (II) ions by baker’s yeast: Kinetic, thermodynamic and desorption studies. Bioresour. Technol. 2008, 99, 3100–3109. [Google Scholar] [CrossRef]
- Argun, M.E.; Dursun, S.; Ozdemir, C.; Karatas, M. Heavy metal adsorption by modified oak sawdust: Thermodynamics and kinetics. J. Hazard. Mater. 2007, 141, 77–85. [Google Scholar] [CrossRef]
- Abia, A.A.; Asuquo, E.D. Kinetics of Cd2+ and Cr3+ sorption from aqueous solutions using mercaptoacetic acid modified and unmodified oil palm fruit fibre (Elaeis guineensis) adsorbents. Tsinghua Sci. Technol. 2007, 12, 485–492. [Google Scholar] [CrossRef]
- Kobya, M.; Demirbas, E.; Senturk, E.; Ince, M. Adsorption of heavy metal ions from aqueous solutions by activated carbon prepared from apricot stone. Bioresour. Technol. 2005, 96, 1518–1521. [Google Scholar] [CrossRef]
- Pakiam, R. Palm Oil Output in Malaysia to Gain in 2011. Available online: http://www.bloomberg.com/news/2011-02-24/palm-oil-output-in-malaysia-to-gain-as-weather-boosts-yields-group-says.html (4th August 2011).
- Zub, V.Y.; Bugaeva, P.V.; Strizhakova, N.G.; Maletin, Y.A. Complexes of some 3d-metal salts with N,N-dimethylhydrazide of 4-nitrobenzoic acid. Russ. J. Coord. Chem. 2004, 30, 747–751. [Google Scholar] [CrossRef]
- Gad, A.M.; El-Dissouky, A.; Mansour, E.M.; El-Maghraby, A. Thermal stability of poly acryloyl benzoic hydrazide and its complexes with some transition metals. Polym. Degrad. Stab. 2000, 68, 153–158. [Google Scholar] [CrossRef]
- Haron, M.J.; Tiansin, M.; Ibrahim, N.A.; Kassim, A.; Wan Yunus, W.M.Z. Sorption of Cu(II) by polyhydroxamic acid chelating exchanger prepared from polymethyl acrylate grafted oil palm empty fruit bunch. Bioresources 2009, 4, 1305–1318. [Google Scholar]
- Raju, G.; Ratnam, C.T.; Ibrahim, N.A.; Ab Rahman, M.Z.; Wan Yunus, W.M.Z. Graft copolymerization of methyl acrylate onto oil palm empty fruit bunch (OPEFB) fiber. Polym. Plast. Technol. Eng. 2007, 46, 949–955. [Google Scholar] [CrossRef]
- Chang, X.; Su, Z.; Luo, X.; Zhan, G. Synthesis of poly(acrylamidrazone-hydrazide) chelating fiber and application of enrichment—Separation for traces of indium, tin, chromium, vanadium and titanium from solution samples. Talanta 1993, 40, 527–532. [Google Scholar] [CrossRef]
- Bekheit, M.M.; Nawar, N.; Addison, A.W.; Abdel-Latif, D.A.; Monier, M. Preparation and characterization of chitosan-grafted-poly(2-amino-4,5-pentamethylene-thiophene-3-carboxylic acid N′-acryloyl-hydrazide) chelating resin for removal of Cu(II), Co(II) and Ni(II) metal ions from aqueous solutions. Int. J. Biol. Macromol. 2011, 48, 558–565. [Google Scholar] [CrossRef]
- Liu, R.X.; Zhang, B.W.; Tang, H.X. Synthesis and characterization of poly(acrylaminophosphonic-carboxyl-hydrazide) chelating fibre. React. Funct. Polym. 1999, 39, 71–81. [Google Scholar] [CrossRef]
- Dai, J.; Yan, H.; Yang, H.; Cheng, R. Simple method for preparation of chitosan/poly(acrylic acid) blending hydrogel beads and adsorption of copper (II) from aqueous solutions. Chem. Eng. J. 2010, 165, 240–249. [Google Scholar] [CrossRef]
- Wan Ngah, W.S.; Hanafiah, M.A.K.M. Biosorption of copper ions from dilute aqueous solutions on base treated rubber (Hevea brasiliensis) leaves powder: Kinetics, isotherm, and biosorption mechanisms. J. Environ. Sci. 2008, 20, 1168–1176. [Google Scholar] [CrossRef]
- Hasar, H. Adsorption of nickel (II) from aqueous solution onto activated carbon prepared from almond husk. J. Hazard. Mater. 2003, B97, 49–57. [Google Scholar] [CrossRef]
- Kamari, A.; Wan Ngah, W.S. Isotherm, kinetic and thermodynamic studies of lead and copper uptake by H2SO4 modified chitosan. Colloids Surf. B 2009, 73, 257–266. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Minocha, A.K. Biosorption optimization of nickel removal from water using Punica granatum peel waste. Colloids Surf. B 2010, 76, 544–548. [Google Scholar]
- Fil, B.A.; Boncukcuoglu, R.; Yilmaz, A.E.; Bayar, S. Adsorption of Ni(II) on ion exchange resin: Kinetics, equilibrium and thermodynamic studies. Korean J. Chem. Eng. 2012, 1–7. [Google Scholar]
- Ozcan, A.; Ozcan, A.S.; Tunali, S.; Akar, T.; Kiran, I. Determination of the equilibrium, kinetic and thermodynamic parameters of adsorption of copper (II) ions onto seeds of Capsicum annuum. J. Hazard. Mater. 2005, B124, 200–208. [Google Scholar]
- Anidruhan, T.S.; Radhakrishnan, P.G. Chromium (III) removal from water and wastewater using a carboxylate-functionalized cation exchanger prepared from a lignocellulosic residue. J. Colloid Interface Sci. 2007, 316, 268–276. [Google Scholar] [CrossRef]
- Mohamad Ibrahim, M.N.; Wan Ngah, W.S.; Norliyana, M.S.; Wan Daud, W.R.; Rafatullah, M.; Sulaiman, O.; Hashim, R. A novel agricultural waste adsorbent for the removal of lead (II) ions from aqueous solutions. J. Hazard. Mater. 2010, 182, 377–385. [Google Scholar] [CrossRef]
- Shahmohammadi-Kalalagh, S.; Babazadeh, H.; Nazemi, A.H.; Manshouri, M. Isotherm and kinetic studies on adsorption of Pb, Zn and Cu by kaolinite. Casp. J. Environ. Sci. 2011, 9, 243–255. [Google Scholar]
- Reddy, D.H.K.; Seshaiah, K.; Reddy, A.V.R.; Lee, S.M. Optimization of Cd(II), Cu(II) and Ni(II) biosorption by chemically modified Moringa oleifera leaves powder. Carbohydr. Polym. 2012, 88, 1077–1086. [Google Scholar] [CrossRef]
- Ho, Y.S.; McKay, G. The kinetics of sorption of divalent metal ions onto sphagnum moss peat. Water Res. 1999, 34, 735–742. [Google Scholar]
- Reddy, D.H.K.; Ramana, D.K.V.; Seshaiah, K.; Reddy, A.V.R. Biosorption of Ni(II) from aqueous phase by Moringa oleifera bark, a low cost biosorbent. Desalination 2011, 268, 150–157. [Google Scholar] [CrossRef]
- Pimentel, P.M.; Melo, M.A.F.; Melo, D.M.A.; Assuncao, A.L.C.; Henrique, D.M.; Silva, C.N., Jr.; Gonzalez, G. Kinetics and thermodynamics of Cu(II) adsorption on oil shale wastes. Fuel Process. Technol. 2008, 89, 62–67. [Google Scholar] [CrossRef]
- Aksu, Z. Determination of the equilibrium, kinetic and thermodynamic parameters of the batch biosorption of nickel (II) ions onto Chlorella vulgaris. Process Biochem. 2002, 38, 89–99. [Google Scholar] [CrossRef]
- Tong, K.S.; Jain Kassim, M.; Azraa, A. Adsorption of copper ion from its aqueous solution by a novel biosorbent Uncaria gambir: Equilibrium, kinetics, and thermodynamic studies. Chem. Eng. J. 2011, 170, 145–153. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Johari, I.S.b.; Yusof, N.A.; Haron, M.J.; Nor, S.M.M. Preparation and Characterization of Poly(ethyl hydrazide) Grafted Oil Palm Empty Fruit Bunch for Removal of Ni(II) Ion in Aqueous Environment. Polymers 2013, 5, 1056-1067. https://doi.org/10.3390/polym5031056
Johari ISb, Yusof NA, Haron MJ, Nor SMM. Preparation and Characterization of Poly(ethyl hydrazide) Grafted Oil Palm Empty Fruit Bunch for Removal of Ni(II) Ion in Aqueous Environment. Polymers. 2013; 5(3):1056-1067. https://doi.org/10.3390/polym5031056
Chicago/Turabian StyleJohari, Ili Syazana bt, Nor Azah Yusof, Md Jelas Haron, and Siti Mariam Mohd Nor. 2013. "Preparation and Characterization of Poly(ethyl hydrazide) Grafted Oil Palm Empty Fruit Bunch for Removal of Ni(II) Ion in Aqueous Environment" Polymers 5, no. 3: 1056-1067. https://doi.org/10.3390/polym5031056




