Dye-sensitized Solar Cells with New One-Dimensional Halide-Bridged Cu(I)–Ni(II) Heterometal Coordination Polymers Containing Hexamethylene Dithiocarbamate Ligand
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Synthesis of [CuI2NiIIBr2(Hm-dtc)2(CH3CN)2]n (1a)
2.3. Synthesis of [CuI2NiIII2(Hm-dtc)2(CH3CN)2]n (1b)
2.4. Crystal Structure Determination
2.5. Fabrication of DSSCs
2.6. Physical Measurements
3. Results and Discussion
3.1. Description of Crystal Structures
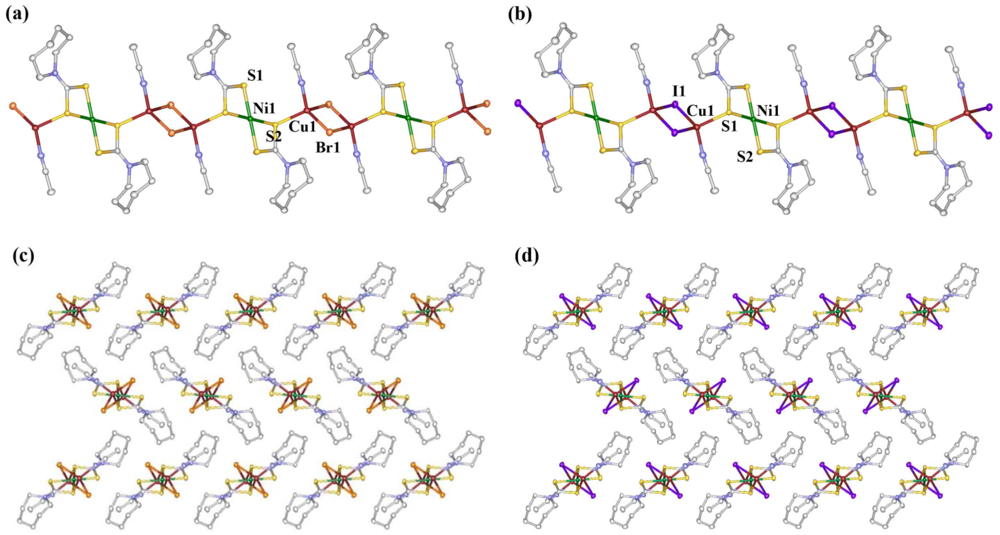
3.2. Determination of HOMO and LUMO Levels of 1a and 1b
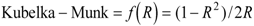
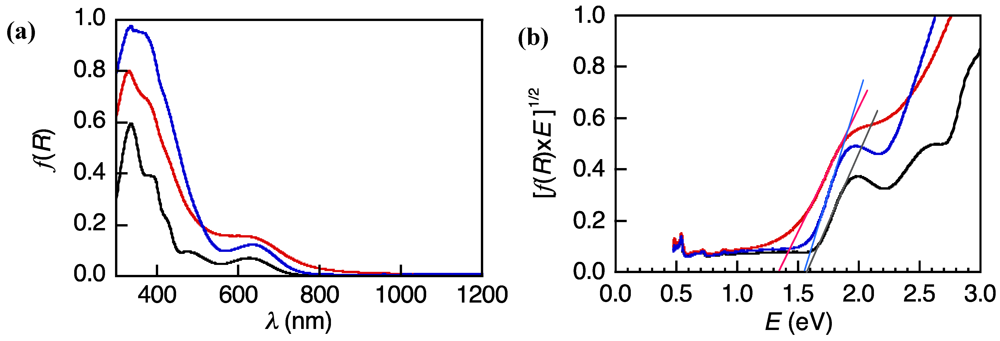

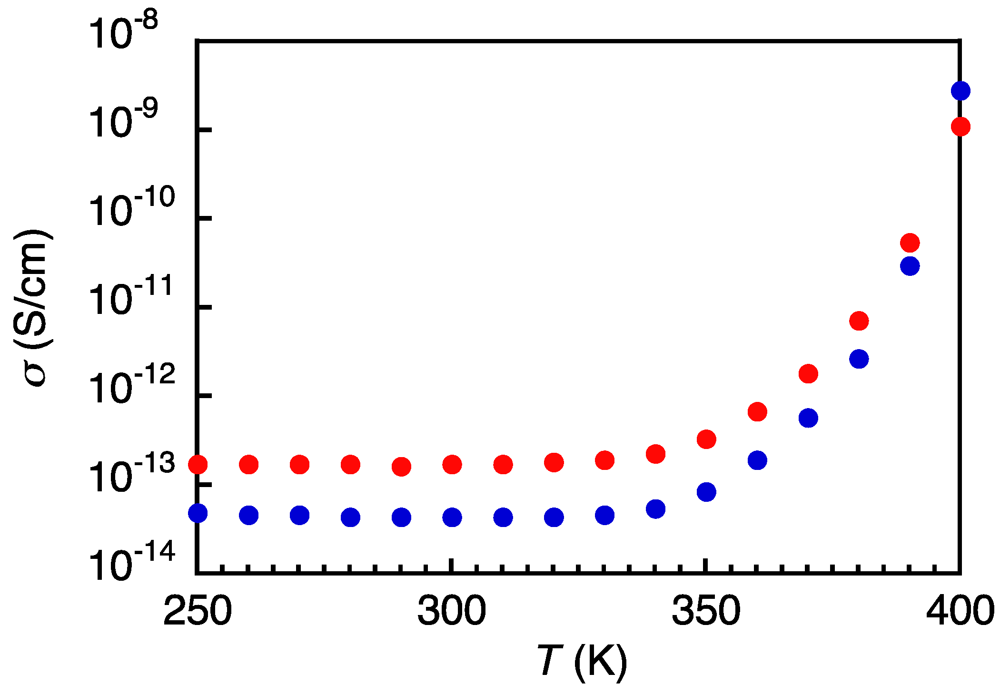
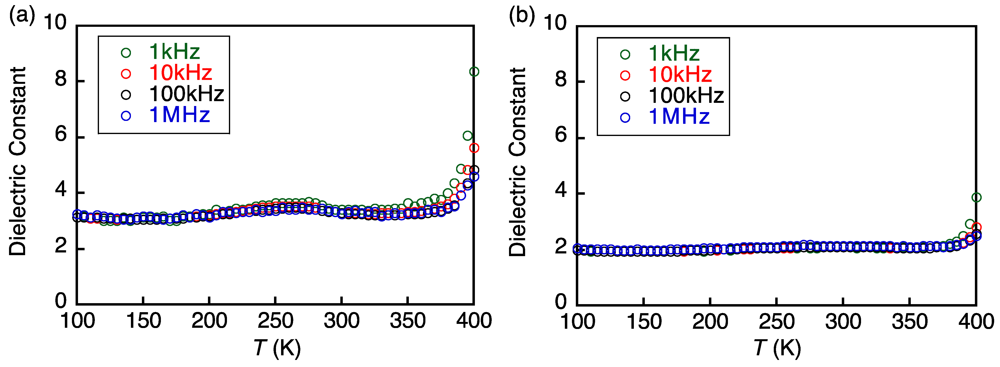
3.3. Electroconducting and Dielectric Properties of 1a and 1b
3.4. Photovoltaic Performance of DSSCs with Coordination Polymers 1a, 1b, 2a, 2b and 3
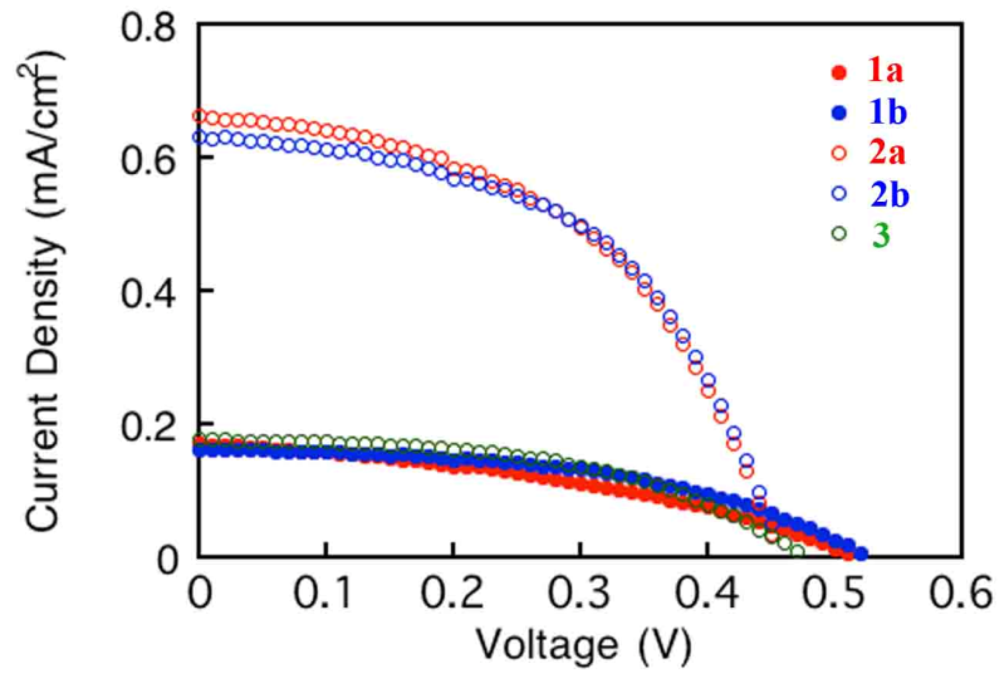
| Complex | JSC (mA/cm) | VOC (V) | FF | PCE (%) |
|---|---|---|---|---|
| 1a | 0.171 | 0.519 | 0.387 | 0.034 |
| 1b | 0.163 | 0.527 | 0.489 | 0.041 |
| 2a | 0.663 | 0.457 | 0.492 | 0.149 |
| 2b | 0.633 | 0.460 | 0.519 | 0.151 |
| 3 | 0.177 | 0.478 | 0.492 | 0.041 |
4. Conclusions
Acknowledgments
References and Notes
- Kahn, O. Molecular Magnetism; VCH: New York, NY, USA, 1993. [Google Scholar]
- Ferlay, S.; Mallah, T.; Ouahes, R.; Veillet, P.; Verdaguer, M. A room-temperature organometallic magnet based on Prussian blue. Nature 1995, 378, 701–703. [Google Scholar]
- Sato, O.; Iyoda, T.; Fujishima, A.; Hashimoto, K. Photoinduced magnetization of a cobalt-iron cyanide. Science 1996, 272, 704–705. [Google Scholar]
- Amo-Ochoa, P.; Castillo, O.; Alexandre, S.S.; Welte, L.; de Pablo, P.J.; Rodriguez-Tapiador, I.; Gomez-Herrero, J.; Zamora, F. Synthesis of designed conductive one-dimensional coordination polymers of Ni(II) with 6-mercaptopurine and 6-thioguanine. Inorg. Chem. 2009, 48, 7931–7936. [Google Scholar] [CrossRef]
- Ichikawa, S.; Kimura, S.; Takahashi, K.; Mori, H.; Yoshida, G.; Manabe, Y.; Matsuda, M.; Tajima, H.; Yamaura, J.-I. Intrinsic carrier doping in antiferromagnetically interacted supramolecular copper complexes with (pyrazino)tetrathiafulvalene (pyra-TTF) as the ligand, [CuIICl2(pyra-TTF)] and (pyra-TTF)2[CuI3Cl4(pyra-TTF)]. Inorg. Chem. 2008, 47, 4140–4145. [Google Scholar] [CrossRef]
- Kishida, H.; Ito, T.; Nakamura, A.; Takaishi, S.; Yamashita, M. Current oscillation originating from negative differential resistance in one-dimensional halogen-bridged nickel compounds. J. Appl. Phys. 2009, 106, 016106:1–016106:3. [Google Scholar]
- Mitsumi, M.; Murase, T.; Kishida, H.; Yoshinari, T.; Ozawa, Y.; Toriumi, K.; Sonoyama, T.; Kitagawa, H.; Mitani, T. Metallic behavior and periodical valence ordering in a MMX chain compound, Pt2(EtCS2)4I. J. Am. Chem. Soc. 2001, 123, 11179–11192. [Google Scholar]
- Miyasaka, H.; Motokawa, N.; Matsunaga, S.; Yamashita, M.; Sugimoto, K.; Mori, T.; Toyota, N.; Dunbar, K.R. Control of charge transfer in a series of Ru2(II,II)/TCNQ two-dimensional networks by tuning the electron affinity of TCNQ units: A route to synergistic magnetic/conducting materials. J. Am. Chem. Soc. 2010, 132, 1532–1544. [Google Scholar]
- Otsubo, K.; Kobayashi, A.; Kitagawa, H.; Hedo, M.; Uwatoko, Y.; Sagayama, H.; Wakabayashi, Y.; Sawa, H. Most stable metallic phase of the mixed-valence MMX-chain, Pt2(dtp)4I (dtp = C2H5CS2−), in purely d-electronic conductors based on the transition-metal complex. J. Am. Chem. Soc. 2006, 128, 8140–8141. [Google Scholar]
- Tadokoro, M.; Yasuzuka, S.; Nakamura, M.; Shinoda, T.; Tatenuma, T.; Mitsumi, M.; Ozawa, Y.; Toriumi, S.; Tatenuma, T.; Mitsumi, M.; Ozawa, Y.; Toriumi, K.; Yoshino, H.; Shiomi, D.; Sato, K.; Takui, T.; Mori, T.; Murata, K. A high-conductivity crystal containing a copper(I) coordination polymer bridged by the organic acceptor TANC. Angew. Chem. Int. Ed. 2006, 45, 5144–5147. [Google Scholar]
- Zhong, J.C.; Misaki, Y.; Munakata, M.; Kuroda-Sowa, T.; Maekawa, M.; Suenaga, Y.; Konaka, H. Silver(I) coordination polymer of 2,5-Bis- (4',5'-bis(methylthio)-1',3'-dithiol-2'-ylidene)- 1,3,4,6-tetrathiapentalene (TTM-TTP) and its highly conductive iodine derivative. Inorg. Chem. 2001, 40, 7096–7098. [Google Scholar] [CrossRef]
- Okubo, T.; Tanaka, N.; Kim, K.H.; Yone, H.; Maekawa, M.; Kuroda-Sowa, T. Magnetic and conducting properties of new halide-bridged mixed-valence CuI-CuII 1D coordination polymers including a hexamethylene dithiocarbamate ligand. Inorg. Chem. 2010, 49, 3700–3702. [Google Scholar] [CrossRef]
- Okubo, T.; Tanaka, N.; Kim, K.H.; Anma, H.; Seki, S.; Saeki, A.; Maekawa, M.; Kuroda-Sowa, T. Crystal structure and carrier transport properties of a new 3D mixed-valence Cu(I)-Cu(II) coordination polymer including pyrrolidine dithiocarbamate ligand. Dalton Trans. 2011, 40, 2218–2224. [Google Scholar] [CrossRef]
- Kim, K.H.; Ueta, T.; Okubo, T.; Hayami, S.; Anma, H.; Kato, K.; Shimizu, T.; Fujimori, J.; Maekawa, M.; Kuroda-Sowa, T. Synthesis and conducting properties of a new mixed-valence Cu(I)-Cu(II) 1-D coordination polymer bridged by morpholine dithiocarbamate. Chem. Lett. 2011, 40, 1184–1186. [Google Scholar] [CrossRef]
- Xie, Y.-M.; Liu, J.-H.; Wu, X.-Y.; Zhao, Z.-G.; Zhang, Q.-S.; Wang, F.; Chen, S.-C.; Lu, C.-Z. New ferroelectric and nonlinear optical porous coordination polymer constructed from a rare (CuBr)∞ castellated chain. Cryst. Growth Des. 2008, 8, 3914–3916. [Google Scholar] [CrossRef]
- Ye, Q.; Song, Y.-M.; Wang, G.-X.; Chen, K.; Fu, D.-W.; Chan, P.W.H.; Zhu, J.-S.; Huang, S.D.; Xiong, R.-G. Ferroelectric metal-organic framework with a high dielectric constant. J. Am. Chem. Soc. 2006, 128, 6554–6555. [Google Scholar]
- Zhang, W.; Xiong, R.-G.; Huang, S.D. 3D framework containing Cu4Br4 cubane as connecting node with strong ferroelectricity. J. Am. Chem. Soc. 2008, 130, 10468–10469. [Google Scholar] [CrossRef]
- Kitagawa, S.; Kitaura, R.; Noro, S.-I. Functional porous coordination polymers. Angew. Chem.Int.Ed. Engl. 2004, 43, 2334–2375. [Google Scholar] [CrossRef]
- Noro, S.-I. Rational synthesis and characterization of porous Cu(II) coordination polymers. Phys. Chem. Chem. Phys. 2010, 12, 2519–2531. [Google Scholar] [CrossRef]
- Yaghi, O.M.; O'Keeffe, M.; Ockwig, N.W.; Chae, H.K.; Eddaoudi, M.; Kim, J. Reticular synthesis and the design of new materials. Nature 2003, 423, 705–714. [Google Scholar] [CrossRef]
- Cho, S.-H.; Ma, B.; Nguyen, S.T.; Hupp, J.T.; Albrecht-Schmitt, T.E. A metal-organic framework material that functions as an enantioselective catalyst for olefin epoxidation. Chem. Commun. 2006, 2563–2565. [Google Scholar]
- Farrusseng, D.; Aguado, S.; Pinel, C. Metal-organic frameworks: Opportunities for catalysis. Angew. Chem. Int. Ed. 2009, 48, 7502–7513. [Google Scholar] [CrossRef]
- Uemura, T.; Kitaura, R.; Ohta, Y.; Nagaoka, M.; Kitagawa, S. Nanochannel-promoted polymerization of substituted acetylenes in porous coordination polymers. Angew. Chem. Int. Ed. 2006, 45, 4112–4116. [Google Scholar] [CrossRef]
- Zou, R.-Q.; Sakurai, H.; Xu, Q. Preparation, adsorption properties, and catalytic activity of three-dimensional porous metal-organic frameworks composed of cubic building blocks and alkali-metal ions. Angew. Chem. Int. Ed. 2006, 45, 2542–2546. [Google Scholar] [CrossRef]
- Enoki, T.; Miyazaki, A. Magnetic TTF-based charge-transfer complexes. Chem. Rev. 2004, 104, 5449–5477. [Google Scholar] [CrossRef]
- Montes, V.A.; Zyryanov, G.V.; Danilov, E.; Agarwal, N.; Palacios, M.A.; Anzenbacher, P. Ultrafast energy transfer in oligofluorene-aluminum bis(8-hydroxyquinoline)acetylacetone coordination polymers. J. Am. Chem. Soc. 2009, 131, 1787–1795. [Google Scholar]
- Kim, K.H.; Okubo, T.; Tanaka, N.; Mimura, N.; Maekawa, M.; Kuroda-Sowa, T. Dye-sensitized solar cells with halide-bridged mixed-valence Cu(I)-Cu(II) coordination polymers with hexamethylenedithiocarbamate ligand. Chem. Lett. 2010, 39, 792–793. [Google Scholar] [CrossRef]
- Tang, C.W. Two-layer organic photovoltaic cell. Appl. Phys. Lett. 1986, 48, 183–185. [Google Scholar] [CrossRef]
- Hiramoto, M.; Fujiwara, H.; Yokoyama, M. Three-layered organic solar cell with a photoactive interlayer of codeposited pigments. Appl. Phys. Lett. 1991, 58, 1062–1064. [Google Scholar] [CrossRef]
- O'Regan, B.; Graetzel, M. A low-cost, high-efficiency solar cell based on dye-sensitized colloidal titanium dioxide films. Nature 1991, 353, 737–740. [Google Scholar] [CrossRef]
- Graetzel, M. Solar energy conversion by dye-sensitized photovoltaic cells. Inorg. Chem. 2005, 44, 6841–6851. [Google Scholar] [CrossRef]
- Li, T.-L.; Lee, Y.-L.; Teng, H. CuInS2 quantum dots coated with CdS as high-performance sensitizers for TiO2 electrodes in photoelectrochemical cells. J. Mater. Chem. 2011, 21, 5089–5098. [Google Scholar] [CrossRef]
- Jian, F.; Wang, Z.; Bai, Z.; You, X.; Fun, H.-K.; Chinnakali, K.; Razak, I.A. The crystal structure, equilibrium and spectroscopic studies of bis(dialkyldithiocarbamate) copper(II) complexes [Cu2(R2dtc)4] (dtc = dithiocarbamate). Polyhedron 1999, 18, 3401–3406. [Google Scholar] [CrossRef]
- Ngo, S.C.; Banger, K.K.; DelaRosa, M.J.; Toscano, P.J.; Welch, J.T. Thermal and structural characterization of a series of homoleptic Cu(II) dialkyldithiocarbamate complexes: Bigger is only marginally better for potential MOCVD performance. Polyhedron 2003, 22, 1575–1583. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. A 2008, A64, 112–122. [Google Scholar] [CrossRef]
- Burla, M.C.; Caliandro, R.; Camalli, M.; Carrozzini, B.; Cascarano, G.L.; de Caro, L.; Giacovazzo, C.; Polidori, G.; Siliqi, D.; Spagna, R. IL MILIONE: A suite of computer programs for crystal structure solution of proteins. J. Appl.Crystallogr. 2007, 40, 609–613. [Google Scholar] [CrossRef]
- Least Squares function minimized (SHELX97): Σw(FO2−FC2)2 where w = Least Squares weights.
- Crystal Structure Analysis Package, Rigaku Corporation (200-2010) Tokyo 196-8666, Japan.
- Crystal data for [Cu2NiBr2(Hm-dtc)2(CH3CN)2]n: fw 776.30, monoclinic, P21/c, a = 10.3600(6) Å, b = 8.1909(5) Å, c = 15.962(2) Å, β = 105.983(3)°, V = 1302.1(2) Å3, Z = 2, Dcalcd = 1.980 g/cm3, 12132 reflections measured, 2966 were unique. R1 = 0.0315 (I > 2.00σ(I)), wR2 = 0.0775 (all data). CCDC: 889235.
- Ramalingam, K.; Radha, K.; Aravamudan, G.; Mahadevan, C.; Subramanyam, C.; Seshasayee, M. Structure of bis[N,N-bis(2-hydroxyethyl)dithiocarbamato]nickel(II), C10H20N2NiO4S4. Acta Crystallogr. C 1984, C40, 1838–1839. [Google Scholar]
- Radha, A.; Seshasayee, M.; Aravamudan, G. Structures of bis(piperidine-1-dithiocarbamato)nickel(II) and bis(piperidine-1-dithiocarbamato)copper(II). Acta Crystallogr. C 1988, C44, 1378–1381. [Google Scholar]
- Srinivasan, S.; Ramalingam, K.; Rizzoli, C. Synthesis, NMR and single crystal X-ray structural studies on planar NiS4 and NiS2PN chromophores: Steric and electronic effects. Polyhedron 2012, 33, 60–66. [Google Scholar] [CrossRef]
- Hogarth, G.; Rainford-Brent, E.-J.C.-R.C.R.; Richards, I. Functionalised dithiocarbamate complexes: Synthesis and molecular structures of bis(2-methoxyethyl)dithiocarbamate complexes [M{S2CN(CH2CH2OMe)2}2] (M = Ni, Cu, Zn) and [Cu{S2CN(CH2CH2OMe)2}2][ClO4. Inorg. Chim. Acta 2009, 362, 1361–1364. [Google Scholar] [CrossRef]
- Brese, N.E.; O'Keeffe, M. Bond-valence parameters for solids. Acta Crystallogr. B 1991, B47, 192–197. [Google Scholar]
- Crystal data for [Cu2NiI2(Hm-dtc)2(CH3CN)2]n: fw 870.30, monoclinic, P21/c, a = 10.3943(8) Å, b = 8.2724(6) Å, c = 16.385(2) Å, β = 106.045(3)°, V = 1354.0(2) Å3, Z = 2, Dcalcd = 2.135 g/cm3, 12486 reflections measured, 3060 were unique. R1 = 0.0262 (I > 2.00σ(I)), wR2 = 0.0651 (all data). CCDC: 889238.
- Ivanov, A.V.; Ivakhnenko, E.V.; Forsling, W.; Gerasimenko, A.V.; Bukvetskii, B.V. A comparative study of the structural organization of nickel(II) and copper(II) complexes with dialkyl-substituted and cyclic diethyldithiocarbamate ligands by X-ray crystallography, EPR, and CP/MAS 13C and 15N NMR. Russ. J. Inorg. Chem. 2002, 47, 410–422. [Google Scholar]
- Kubelka, P. New contributions to the optics of intensely light-scattering materials. J. Opt. Soc. Am. 1948, 38, 448–457. [Google Scholar] [CrossRef]
- Nazeeruddin, M.K.; Kay, A.; Rodicio, I.; Humphry-Baker, R.; Mueller, E.; Liska, P.; Vlachopoulos, N.; Graetzel, M. Conversion of light to electricity by cis-X2bis(2,2'-bipyridyl-4,4'-dicarboxylate)ruthenium(II) charge-transfer sensitizers (X = Cl−, Br−, I−, CN−, and SCN−) on nanocrystalline TiO2 electrodes. Am. Chem. Soc. 1993, 115, 6382–6390. [Google Scholar]
- Zhang, S.; Yanagida, M.; Yang, X.; Han, L. Effect of 4-tert-butylpyridine on the quasi-Fermi level of dye-sensitized TiO2 films. Appl. Phys. Express 2011, 4, 042301:1–042301:3. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Okubo, T.; Tanaka, N.; Anma, H.; Kim, K.H.; Maekawa, M.; Kuroda-Sowa, T. Dye-sensitized Solar Cells with New One-Dimensional Halide-Bridged Cu(I)–Ni(II) Heterometal Coordination Polymers Containing Hexamethylene Dithiocarbamate Ligand. Polymers 2012, 4, 1613-1626. https://doi.org/10.3390/polym4031613
Okubo T, Tanaka N, Anma H, Kim KH, Maekawa M, Kuroda-Sowa T. Dye-sensitized Solar Cells with New One-Dimensional Halide-Bridged Cu(I)–Ni(II) Heterometal Coordination Polymers Containing Hexamethylene Dithiocarbamate Ligand. Polymers. 2012; 4(3):1613-1626. https://doi.org/10.3390/polym4031613
Chicago/Turabian StyleOkubo, Takashi, Naoya Tanaka, Haruho Anma, Kyung Ho Kim, Masahiko Maekawa, and Takayoshi Kuroda-Sowa. 2012. "Dye-sensitized Solar Cells with New One-Dimensional Halide-Bridged Cu(I)–Ni(II) Heterometal Coordination Polymers Containing Hexamethylene Dithiocarbamate Ligand" Polymers 4, no. 3: 1613-1626. https://doi.org/10.3390/polym4031613




