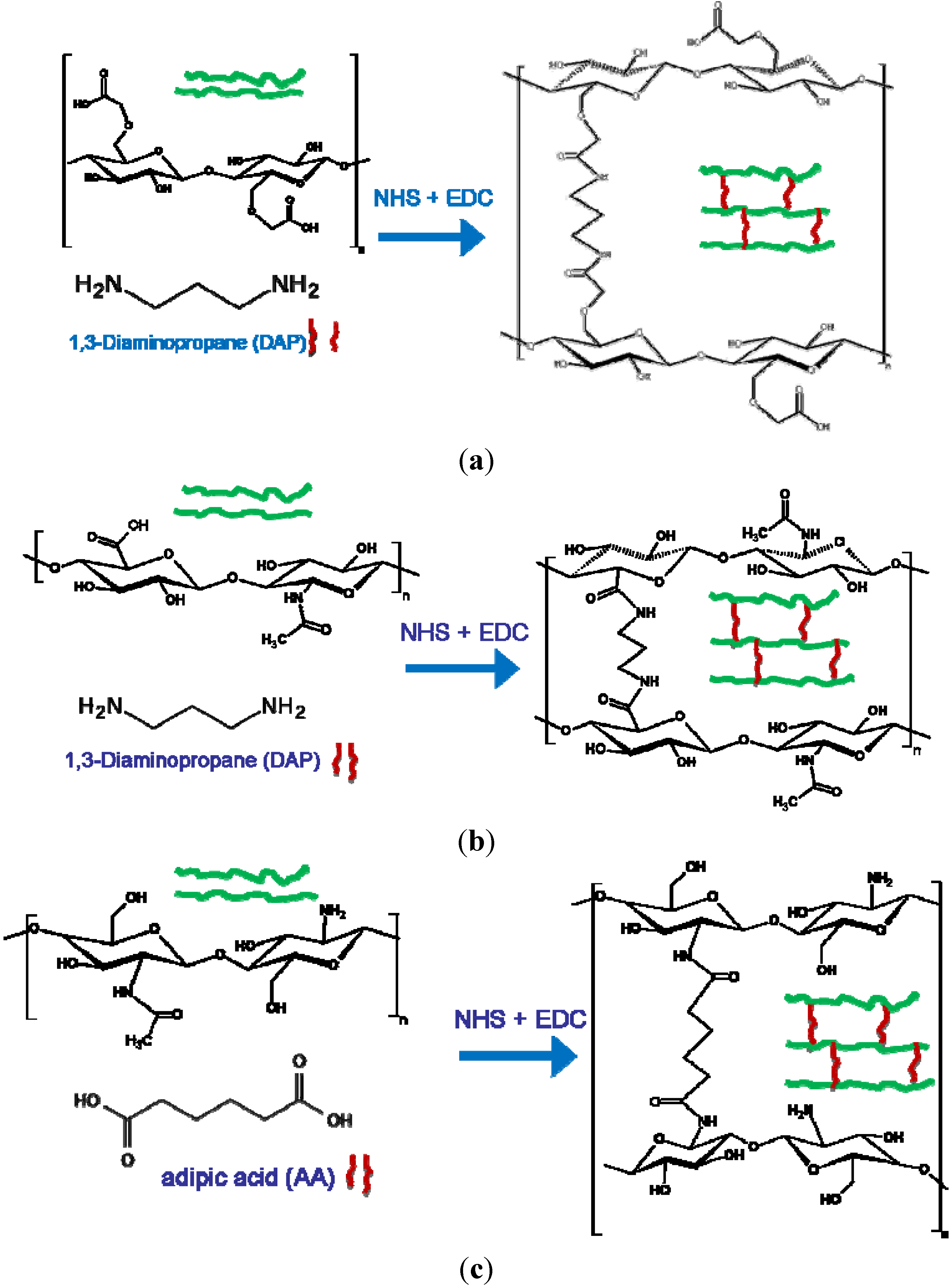
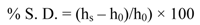2.1. Hydrogel Synthesis
Hydrogel synthesis follows the same chemical route that has been well-documented in carbodiimide chemistry [
7,
25]. Basically, the synthesis of CMC- and Hyal-based hydrogels exploits the formation of an amide bond between the carboxylate groups of the polymer chains and the primary amine groups of 1,3 diaminopropane (DAP), which is added to the polymer aqueous solution as the cross-linking agent of the polymer chain. 1-ethyl-3-[3-(dimethyl-amino)propyl] carbodiimide hydrochloride (EDC) works as the activating agent of the carboxylic groups, and N-hydroxysuccinimide (NHS) as the proton exchanger [
26]. The reaction schemes of CMC and Hyal hydrogel are shown in
Figure 1a,b. The stoichiometry of the reaction can be tuned in order to form hydrogels with different degrees of cross-linking, taking into consideration the number of carboxylic groups in each polysaccharide,
i.e., one carboxylic group per disaccharide unit in Hyal and two in CMC polymer. On the other hand, CHT synthesis (see
Figure 1c) involves the formation of an amide bond between the amine groups of the polymer and the carboxylic groups of the cross-linking agent,
i.e., adipic acid (AA) [
7]. The determination of the degree of cross-linking (defined as the number of polymer groups bound to the cross-linker) is obtained by pontentiometric titration of the free protonable groups remaining after hydrogel formation (carboxylate groups for CMC, Hyal or amine groups for CHT polymer), or by means of
13C nuclear magnetic resonance [
27]. Indeed, even if the molar ratio of polymer, EDC and DAP used for the synthesis of hydrogels, should allow the formation of a hydrogel cross-linked at 100%, the cross-linking degree of CMC, Hyal and CHT hydrogels was 79%, 45% and 44% respectively as determined by potentiometric titrations.
Figure 1.
Reaction scheme of (a) Carboxy Methyl Cellulose (CMC) hydrogel; (b) Hyaluronan (Hyal) hydrogel; and (c) Chitosan (CHT) hydrogel.
Figure 1.
Reaction scheme of (a) Carboxy Methyl Cellulose (CMC) hydrogel; (b) Hyaluronan (Hyal) hydrogel; and (c) Chitosan (CHT) hydrogel.
2.2. Rheological Characterization and Swelling Degree
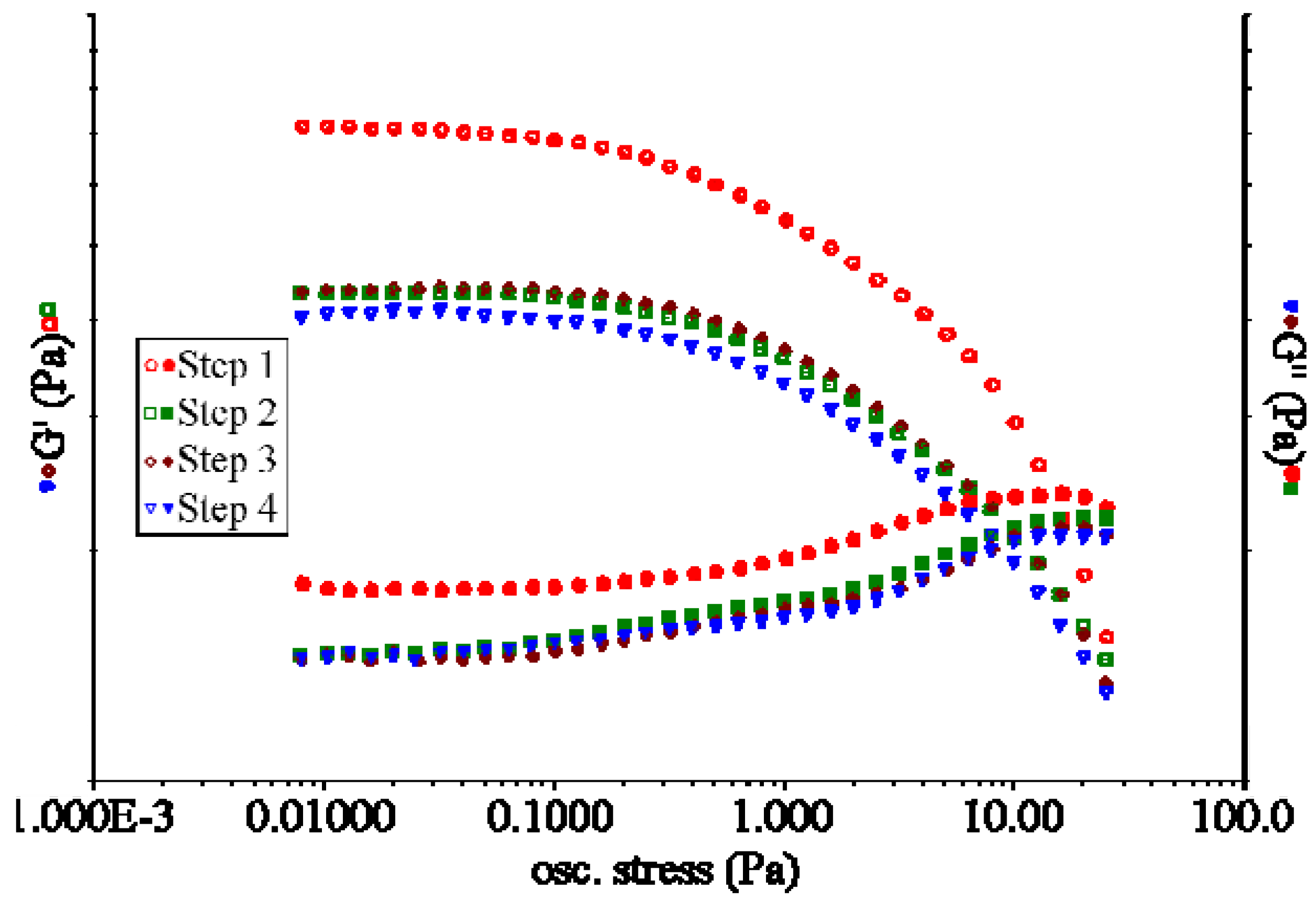
In order to obtain information about the mechanical properties of the polymer network we subjected the hydrogel to a shear stress applied by a rheometer. The typical trend of G’ and G”
vs. oscillation stress for these polysaccharide hydrogels is plotted in
Figure 2. For low values of oscillation stress, the storage modulus (G’) is higher than the viscous one (G”) indicating the gel-like nature of the material. Above the cross-point (
i.e., the oscillation stress at which G’ and G’’ assume the same value), the viscous modulus is higher than the storage modulus, which is characteristic of a liquid. This implies a sol-gel transition of the material [
20]. The behavior of G’ and G” was studied by increasing and decreasing the applied stress cyclically on the same hydrogel sample. When the oscillation stress is decreased (step 2
Figure 2), G’ and G’’ show always lower values than those found during the increase in oscillation stress (step 1
Figure 2). At the end of step 2, the G’ and G” values are lower than those of the native hydrogel at the first step. By repeating the same sequence of steps several times (that is, increasing and decreasing the oscillation stress on the same sample, step 3 and step 4
Figure 2), G’ and G” values follow the same trend and maintain the same values as those determined in step 2. This result indicates a sort of “recovery” effect of the G’ and G” values once the material is subjected to further cycles of stresses [
7,
26]. In the case of squeezed hydrogels (
Figure 3(a–c)), both the storage modulus G’ and the loss modulus G’’ values are reduced due to the passage from the syringe with respect to those of the native hydrogels (
Table 1). However, by repeating the same cycle of stress several times on the squeezed hydrogels, as done for the native ones (see
Figure 2), G’ and G” values remain the same. This does not occur for a native hydrogel (see step 1 and step 2
Figure 2). The squeezing process induces the same effect on the hydrogel as the stress applied by the rheometer. The lower values of G’ and G” of a squeezed hydrogel with respect to its native sample indicate that the hydrogel network is softened by the application of a shear stress such as squeezing through a syringe or by the stress applied by a rheometer. However, the application of an additional stress does not lead to further alterations of the hydrogel’s mechanical properties. Hence, the squeezed hydrogels behave in the same manner as native hydrogels after the application of a stress applied by a rheometer.
Figure 2.
Typical trend of G’ and G” values vs. oscillation stress during sweep test performed on a polysaccharide-based hydrogel. The figure shows the G’ and G” value vs. oscillation stress for each step of the test.
Figure 2.
Typical trend of G’ and G” values vs. oscillation stress during sweep test performed on a polysaccharide-based hydrogel. The figure shows the G’ and G” value vs. oscillation stress for each step of the test.
Figure 3.
Stress sweep test performed on (a) CMC hydrogel; and (b) Hyal hydrogel; (c) CHT hydrogels, native and after being squeezed through a syringe. The figure shows the G’ and G” values vs. oscillation stress for each step of the test.
Figure 3.
Stress sweep test performed on (a) CMC hydrogel; and (b) Hyal hydrogel; (c) CHT hydrogels, native and after being squeezed through a syringe. The figure shows the G’ and G” values vs. oscillation stress for each step of the test.
Table 1.
Storage modulus (G’) and elastic modulus (G”) of polysaccharide hydrogels in native and after being squeezed. Data are reported as mean of three values ± standard error.
Table 1.
Storage modulus (G’) and elastic modulus (G”) of polysaccharide hydrogels in native and after being squeezed. Data are reported as mean of three values ± standard error.
| Hydrogel type | Status | G’ (Pa) | G” (Pa) |
|---|
| CMC | Native | 550 ± 30 | 25 ± 1 |
| Squeezed | 240 ± 20 | 20 ± 2 |
| HYAL | Native | 970 ± 25 | 65 ± 10 |
| Squeezed | 340 ± 20 | 45 ± 5 |
| CHT | Native | 4350 ± 650 | 165 ± 45 |
| Squeezed | 3460 ± 150 | 230 ± 15 |
Infrared spectra and nuclear magnetic resonance (N.M.R.) of the native hydrogel and that of the same hydrogel once stressed appear to be overlapped, indicating that the hydrogel’s chemistry is maintained. Therefore passing through a syringe or being subjected to a stress applied by a rheometer does not alter the chemical structure or affect the degree of cross-linking of the hydrogel [
26]. We can say that the application of a stress induces a change in the hydrogel’s matrix without affecting its chemistry. Therefore, the involvement of different factors such as the amount of water contained within the hydrogel and its arrangement as well as the polymer chains disposal have been considered.
The amount of water contained in the hydrogels was determined by measurements of swelling in NaCl 0.1 M solution at pH 7.4 (see
Table 2). All the native hydrogels uptake the same amount of water at their maximum swelling degree within the experimental errors even if the structure of the starting polymers and the cross-linking of the hydrogels are different from each other. Concerning the swelling degree of squeezed hydrogels, the value of Hyal is slightly higher than that of CMC. CHT shows the lowest swelling degree (see
Figure 4). This trend can be explained by considering the pKa of polysaccharides. The CHT hydrogel, which contains free amine groups, is partially charged at pH 7.4, as the pKa of CHT is between 6.5 and 7.5, depending on the degree of deacetylation [
28]. The NH
3+ groups, which are highly hydrophilic, interact with the remaining free NH
2, collapsing the polymeric chains and hindering the solvation of the hydrogel. In fact, the swelling degree of CHT is significantly higher (~1000) in NaCl 0.1 M at pH 4, when all the free –NH
2 groups are protonated and able to repel each other. The CMC and Hyal hydrogels are both negatively charged at pH 7.4 because of the presence of free, completely unprotonated COO
− (the pKa of both Hyal and CMC is ~4.0–4.5) [
29]. The presence of negatively charged groups increases the electrostatic repulsions between the polymer chains and thus the amount of water that can enter the hydrogel.
Table 2.
Swelling degree of native and squeezed hydrogels in NaCl 0.1 M solution. The reported data represent the average of three samples ± standard error.
Table 2.
Swelling degree of native and squeezed hydrogels in NaCl 0.1 M solution. The reported data represent the average of three samples ± standard error.
| Hydrogel type | Status | Swelling degree % |
|---|
| CMC | Native | 640 ± 50 |
| Squeezed | 1235 ± 125 |
| HYAL | Native | 720 ± 25 |
| Squeezed | 1300 ± 150 |
| CHT | Native | 650 ± 80 |
| Squeezed | 830 ± 125 |
Figure 4.
Swelling degree of different squeezed polysaccharide-based hydrogels in 0.1 M NaCl solution at pH 7.4. Data are reported as mean of three values ± standard error.
Figure 4.
Swelling degree of different squeezed polysaccharide-based hydrogels in 0.1 M NaCl solution at pH 7.4. Data are reported as mean of three values ± standard error.
The total amount of water within hydrogels in NaCl 0.1M is higher for the squeezed hydrogels than for the native ones (see
Table 2). Under the application of a shear stress the polymer chains which are entangled during the cross-linking reaction unroll and align along the direction of the stress [
30,
31]. Hence, this necessarily induces a new disposal of water molecules within the hydrogel network. Bound water molecules move to a semi-bound state (see FT-IR analyses). This movement of water molecules determines a decrease of the mechanical properties of the hydrogel. The change in the arrangement of water molecules seems to be irreversible. In fact, by dipping a squeezed hydrogel in water for several days, it does not regain the original mechanical properties before being squeezed. The passage of water molecules from bound state to a semi bound state make the material also capable to uptake a higher amount of water because the higher mobility of water molecules allow them to form a higher number of bonds with other free water molecules.
2.3. FT-IR Analysis: The Role of Water
The role of water and water interactions inside the hydrogel were investigated by means of FT-IR analyses of the hydrogel in both swollen and dried forms. A typical IR spectrum of CMC hydrogel in dried state is shown in
Figure 5, with main peak assignment listed in
Table 3. In a first approximation the spectrum is composed of four regions [
32,
33]:
1. An intense band in the region between 950 and 1200 cm−1 corresponding to the –C–O, C–CH stretching and CO and C–O–C bending of the glycoside ring.
2. A region around 1300 and 1450 cm−1 where bands related to C–CH and O–CH stretching and –CO stretching are observed together with a peak at 1408 cm−1 beloning to the symmetric stretching of the free carboxylate groups of the polymer not involved in the hydrogel formations.
3. An intense group of bands that extends from 1500 to 1700 cm−1 corresponding to the amide I, amide II and free carboxylate groups of the CMC polymer.
4. A broad and intense band in the region between 2500 and 3600 cm−1 which is composed of several bands ascribed to the –OH stretching of the polymer glycoside ring, and the –OH stretching of water free and involved in hydrogel bonds.
Figure 5.
Infrared spectrum of native CMC hydrogel in the region between 4000 and 750 cm−1.
Figure 5.
Infrared spectrum of native CMC hydrogel in the region between 4000 and 750 cm−1.
Table 3.
Peak wavenumbers (cm−1) and assignments for CMC hydrogel.
Table 3.
Peak wavenumbers (cm−1) and assignments for CMC hydrogel.
| Wavenumber (cm−1) | Assignment |
|---|
| 3264 | water molecules –OH |
| 2923 | –C–C–C–H stretch |
| 1640 | amide –C=O stretch |
| 1582 | carboxylate symm. stretch |
| 1408 | carboxylate asymm. stretch |
| 1320 | C–CH and O–CH stretch |
| 1245 | –CO stretch |
| 1106 | C–C C–O stretch |
| 1060 | C–O–C bend |
Our attention was focused on the spectral region between 1500 and 1800 cm
−1. In order to determine the components contributing to the band envelopes, we performed a curve-fitting analysis of the spectra of the swollen hydrogel in the region between 1500 and 1800 cm
−1 (see
Figure 6). The assignment of peak components for the CMC hydrogel in its native state and after squeezing is reported in
Table 4. The main difference regards the position of the peak ascribed to the carboxylate asymmetric stretching, which shifts from 1578 cm
−1 (spectrum of native hydrogel) to 1590 cm
−1 (spectrum of squeezed hydrogel). The shift to higher wavenumbers is attributed to the loss of some hydrogen bonds involving the free carboxylate groups of the polymer chains [
34]. No significant shifts were observed in the position of amide I in the spectrum of native and squeezed CMC hydrogel in their swollen state. However, once the hydrogel is left to dry, the amide I peak shifts to higher wavenumbers compared with the position of the same peak for the hydrogel in the swollen state (from ~1631 cm
−1 to ~1645 cm
−1). This indicates that in the dried state, additional interactions between amide groups are involved [
34,
35].
Figure 6.
Curve-fitting analysis of the spectrum of swollen CMC hydrogel in native state. The components contributing to the peak were fitted using a mixture of Lorentzian and Gaussian line shapes. The component frequency, the full width, half, maximum and peak areas were optimized in the fitting procedure. The thick line represents the overall fit to four components.
Figure 6.
Curve-fitting analysis of the spectrum of swollen CMC hydrogel in native state. The components contributing to the peak were fitted using a mixture of Lorentzian and Gaussian line shapes. The component frequency, the full width, half, maximum and peak areas were optimized in the fitting procedure. The thick line represents the overall fit to four components.
Table 4.
Assignment of bands of spectra for CMC hydrogels (a) native and (b) squeezed in swollen state present in the region between 1700 and 1500 cm−1.
Table 4.
Assignment of bands of spectra for CMC hydrogels (a) native and (b) squeezed in swollen state present in the region between 1700 and 1500 cm−1.
| CMC hydrogelswollen state | Main peak wavenumber (cm−1) | Wavenumber (cm−1) | Assignment |
|---|
| native | 1631 | 1675 | water molecules –OH bend |
| 1631 | amide –C=O stretch. |
| 1578 | carboxylate asymmetry stretch. |
| 1550 | 1550 | amide –N–H bend |
| squeezed | 1632 | 1675 | water molecules -OH bend |
| 1631 | amide –C=O stretch. |
| 1590 | carboxylate asymm. stretch. |
| 1552 | 1552 | amide –N–H bend |
The same behavior was found for Hyal hydrogels. The typical IR spectrum of Hyal hydrogel in dried state is reported in
Figure 7, with the main peak assignments listed in
Table 5. As made for CMC hydrogel, a curve-fitting analysis of the swollen hydrogel spectra in the region between 1500 and 1800 cm
−1 was carried out. The asymmetric stretching of free carboxylate groups shifted from 1608 cm
−1 in the spectrum of native swollen hydrogel to 1616 cm
−1 in the spectrum of squeezed hydrogel. The shift of the carboxylate groups is again attributed to the presence of hydrogen bonds involving the carboxylate groups. The higher amount of hydrogen bonds [
36] in the native hydrogels could be responsible for the higher storage modulus (G’) and viscous modulus (G”) values of the native CMC and Hyal hydrogels in comparison with the same squeezed hydrogels.
Figure 7.
Infrared spectrum of native Hyal hydrogel in the region between 4000 and 750 cm−1.
Figure 7.
Infrared spectrum of native Hyal hydrogel in the region between 4000 and 750 cm−1.
Table 5.
Peak wavenumbers (cm−1) and assignments for Hyal hydrogel.
Table 5.
Peak wavenumbers (cm−1) and assignments for Hyal hydrogel.
| Wavenumber (cm−1) | Assignment |
|---|
| 3310 | water molecules –OH |
| 2930 | –C–C–C–H stretch |
| 2875 | –C–C–C–H stretch |
| 1640 | amide –C=O stretch |
| 1610 | carboxylate asymm. stretch |
| 1560 | amide N–H bend |
| 1410 | carboxylate symm. stretch |
| 1375 | C–CH and O–CH stretch |
| 1281 | –C–O stretch |
| 1146 | C–C C–O stretch |
| 1046 | C–O–C bend |
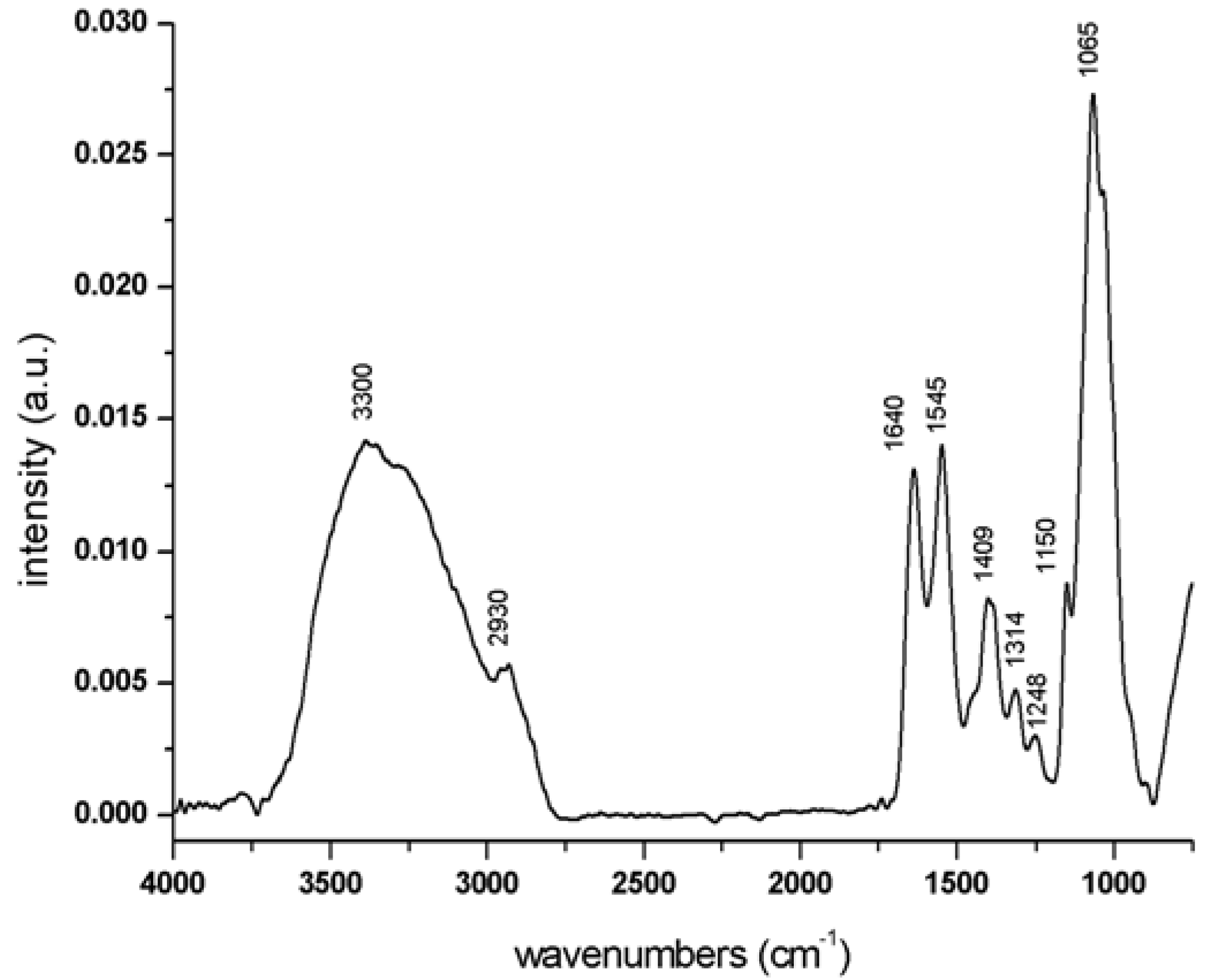
The spectrum of CHT hydrogel is shown in
Figure 8 and the assignment of the main peaks is shown in
Table 6. As discussed above for CMC hydrogel, the entire spectrum can be divided into four regions: An intense band in the region between 950 and 1200 cm
−1 corresponding to –C–O, C–CH stretching and CO and C–O–C bending of the glycosidic ring; a region around 1300 and 1450 cm
−1 where typical chitosan polymer bands related to C–CH and O–CH stretching and –CO stretching are found; and an intense group of bands that extends from 1500 to 1700 cm
−1 corresponding to the amide I and amide II peaks ascribable to the two amide groups of CHT hydrogel, one belonging to chitin polymer and the other related to the one formed during the cross-linking reaction. The last region, which is composed of several bands ascribed to the –OH stretching of the polymer glycosidic ring, –OH stretching of water molecules (both free and involved in hydrogen bonds) and different types of amine group stretching, is between 2500 and 3600 cm
−1 [
37,
38]. The spectrum of the swollen squeezed hydrogel was compared to that of the same hydrogel before squeezing in the region between 2600 and 3600 cm
−1 (see
Table 7) after application of the same curve-fitting procedure used for CMC and Hyal-based hydrogels. A shift of 12 cm
−1 to lower wavenumbers was noted for the peaks at 3510 cm
−1 and 3312 cm
−1 ascribable to the stretching of free NH
2 groups and the stretching of free NH
3+ groups respectively, in the spectrum of the squeezed swollen CHT hydrogel with respect to that of native CHT hydrogel. In the case of the peak related to the amide N–H stretching, a shift of 8 cm
−1 in position was found (3130 cm
−1 for native hydrogels and 3138 cm
−1 for squeezed hydrogels). Generally, the shift of the stretching of primary amine groups to lower wavenumbers is due to an interaction among such groups leading to the formation of NH
2–NH
3+ clusters [
39]. In the case of CHT hydrogels, such interactions seem to be stronger for the squeezed sample than for the native one (see
Table 1). On the contrary, no significant difference of the positions of the amide I or amide II bands were observed in the spectrum of native and squeezed hydrogels in the swollen or dried state. Furthermore, in the case of the dried hydrogel, the peaks of NH
2 groups, the stretching of free NH
3+ groups and the stretching of NH groups are located at 3440, 3260 and 3100 cm
−1 respectively, all at wavenumbers lower than those of the swollen hydrogel. This effect is again due to the existence of NH
2–NH
3+ interactions, which are more intense when the hydrogel is dried. In contrast, when the hydrogel is in the swollen state, the NH
2–NH
3+ interaction can be mediated by water molecules via hydrogen bonds between water and primary amine groups. A sort of competition between NH
2-water, NH
3+-water and NH
2–NH
3+ occurs, and it seems that the interactions between amine groups and water molecules are greater in the native swollen state than in the squeezed and dried state.
Figure 8.
Infrared spectrum of native CHT hydrogel in the region between 4000 and 750 cm−1.
Figure 8.
Infrared spectrum of native CHT hydrogel in the region between 4000 and 750 cm−1.
Table 6.
Peak wavenumbers (cm−1) and assignments for CHT hydrogel.
Table 6.
Peak wavenumbers (cm−1) and assignments for CHT hydrogel.
| Wavenumber (cm−1) | Assignment |
|---|
| 3300 | water molecules –OH |
| 2930 | –C–C–C–H stretch |
| 1640 | amide –C=O stretch. |
| 1545 | amide N–H stretch |
| 1409 | CH2 bend |
| 1314 | C–CH and O–CH stretch. |
| 1248 | –CO stretch |
| 1150 | C–CH C–O stretch |
| 1066 | C–O–C bend |
Table 7.
Assignment of bands of spectra for CHT hydrogels (a) native; and (b) squeezed, both in swollen state, present in the region between 3000and 3650cm−1
Table 7.
Assignment of bands of spectra for CHT hydrogels (a) native; and (b) squeezed, both in swollen state, present in the region between 3000and 3650cm−1
| CHT hydrogelswollen state | Main peak wavenumber (cm−1) | Wavenumber (cm−1) | Assignment |
|---|
| native | 3317 | 3620 | –OH bend |
| 3510 | free –NH2 stretch |
| 3312 | free –NH3+ stretch |
| 3130 | 3130 | amide –N–H stretch |
| squeezed | 3300 | 3595 | –OH bend |
| 3475 | Free –NH2 stretch |
| 3300 | free –NH3+ stretch |
| 3138 | 3138 | amide –N–H stretch |
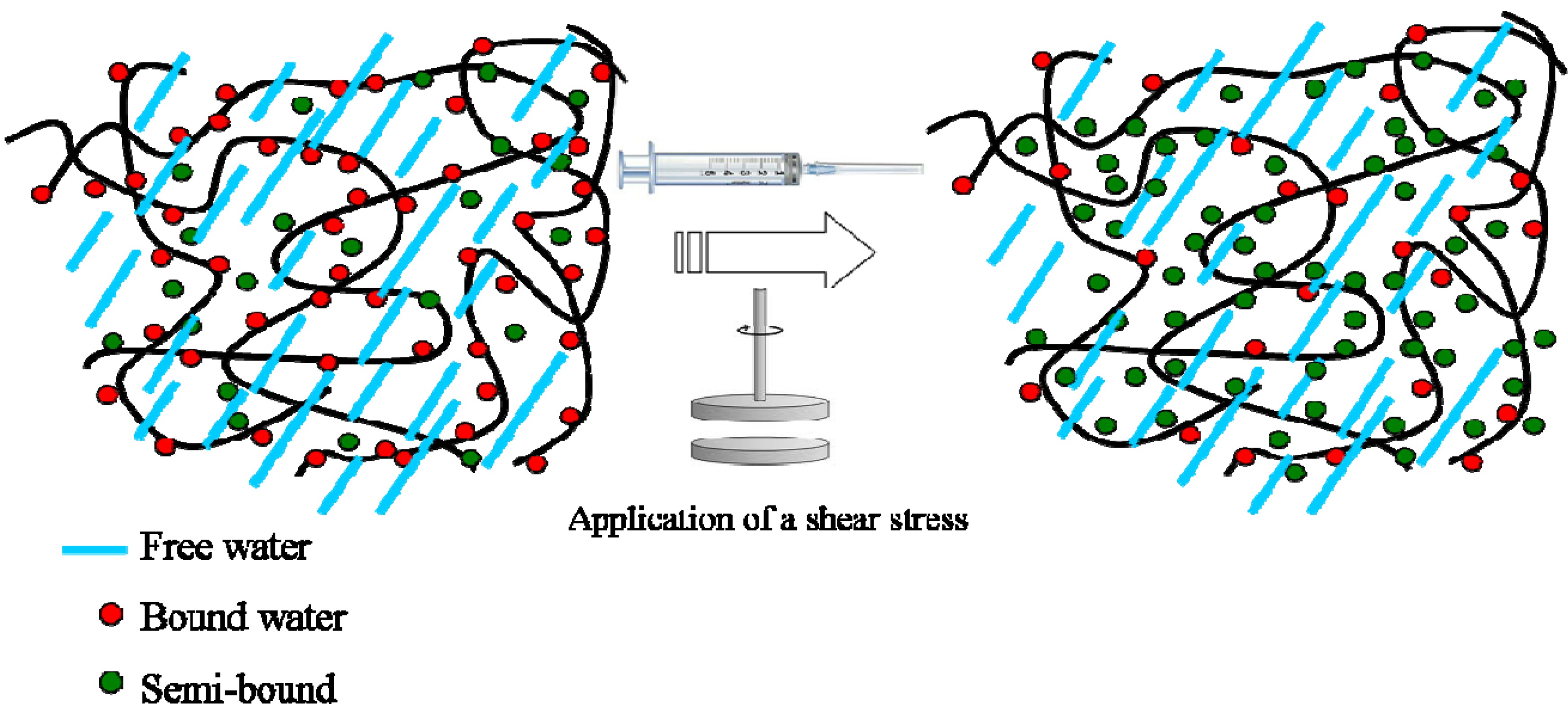
Upon application of a shear stress, such as the passage from a syringe needle, the strength of such physical interactions decreases, making the hydrogel softer. Basically, a different arrangement of water molecules from a bound state to a semi-bound state occurs, leading to a decrease in the mechanical properties of the hydrogels (see
Figure 9). In summary, the presence of water molecules organized near the polymeric chains-so-called bound and semi-bound water-influences the mechanical properties of the hydrogels, making the materials tougher or softer. In the case of native-based hydrogel, polymer-water interactions occurring via hydrogen bonds which are quite strong are responsible for the higher values of G’ and G” as compared with the same squeezed hydrogel.
Figure 9.
Schematic drawing showing the arrangement of water molecules inside a polysaccharide-based hydrogel in the native state and under stress.
Figure 9.
Schematic drawing showing the arrangement of water molecules inside a polysaccharide-based hydrogel in the native state and under stress.