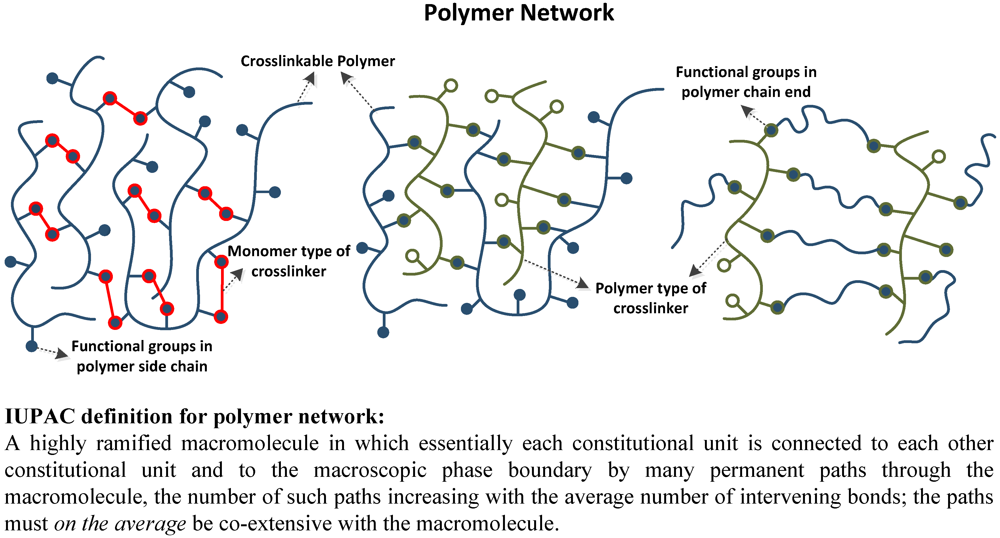
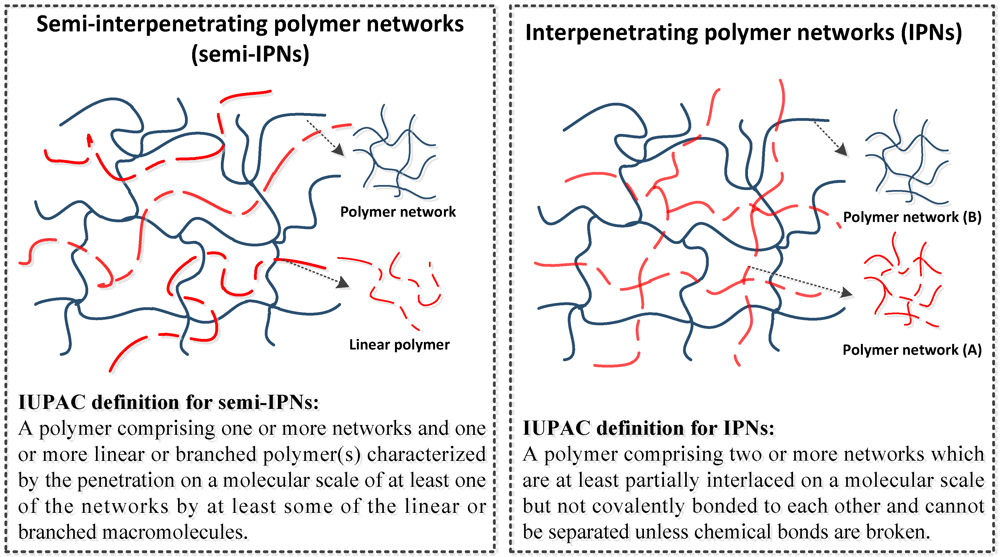
Crosslinked polymers are defined as polymers comprising one or more networks that are interlaced on a molecular scale and covalently, or non-covalently, bonded with each other. The mobility of the polymer chains leads to great variability that can turn a liquid polymer into a ‘solid’ or ‘gel’. The properties of the polymer, such as its: chemical resistance, swellability, permeability, thermal stability, and mechanical properties, depend strongly on its architecture, and density. Due to the desirability of being able to selectively introduce these features in a controlled manner, crosslinked polymers are increasingly being used as engineering materials to provide for a variety of requirements in different applications.
Figure 6.
Schematic examples of different types of common polymer network.
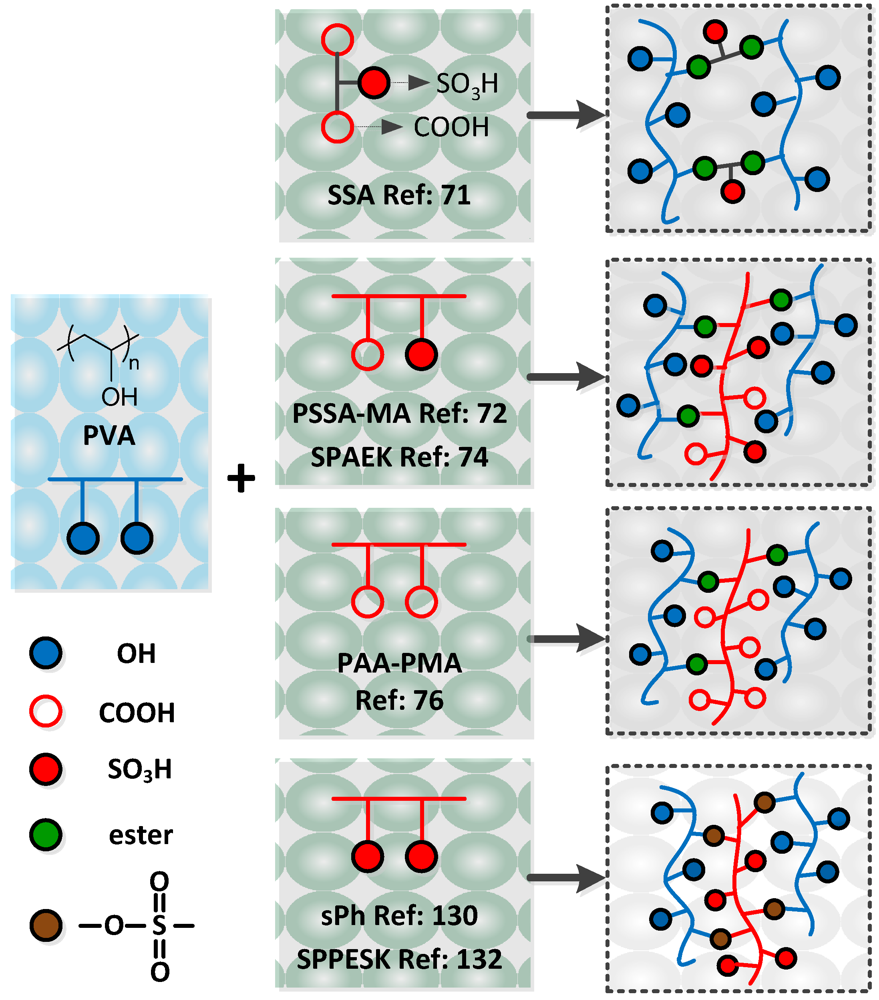
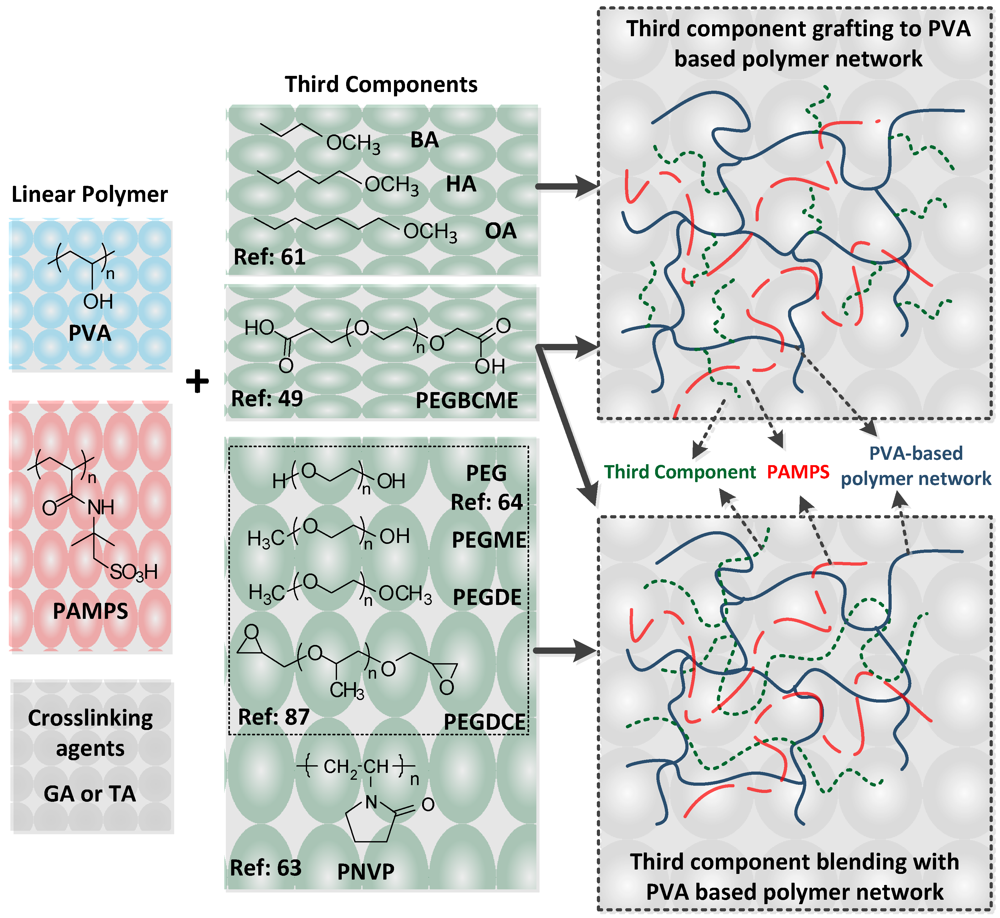
Water soluble polymers (WSPs) have been introduced into crosslinked membranes due to their high flexibility and hydrophilicity. WSPs are expected to serve as better crosslinkers than either hydrophobic alkylic or aromatic crosslinkers. The essential differences are reflected in the production of a finer network morphology, enhanced toughness and strength, improved water absorption and retention, and better proton conduction. Currently there are a number of reports highlighting the successful introduction of WSP into crosslinked membranes that show promising properties for fuel cell applications. In this section, we describe the preparation of crosslinked membranes based on WSPs using chemical crosslinking, excluding membranes synthesized by using IPNs and semi-IPNs strategies. As these latter two network structures have their own characteristics, crosslinked membranes based on IPNs and semi-IPN architectures will be described a following section.
4.1. Crosslinked Poly (vinyl alcohol) Based Membranes
Poly (vinyl alcohol) (PVA) has often been selected to serve as the polymer matrix in crosslinked membranes due to its high differential selectivity for water over methanol or alcohol, its excellent film forming capacities and its possession of dense reactive chemical functions favorable for crosslinking [
57,
58]. A major disadvantage of PVA as a membrane is its poor mechanical stability. To overcome this drawback, a crosslinking strategy has been employed to fabricate a DMFC membrane. However, the PVA membranes are poor proton conductors as compared with Nafion
®; hence it is necessary to combine it with a monomer, oligomer or polymer that contains negatively charged ions (carboxylic and/or sulfonic acid groups), such as sulfosuccinic acid (SSA) [
71], poly (styrene sulfonic acid-
co-maleic acid) (PSSA-PMA) [
72], poly (acrylic acid-
co-maleic acid) (PAA-PMA) [
76], p-sulfonate phenolic (s-Ph) [
130], sulfonated polyhedral oligosilsesquioxane (sPOSS) [
131], sulfonated poly (phthalazinone ether sulfone ketone) (SPPESK) [
132], sulfonated poly (arylene ether ketone) (SPAKE) [
74] or 4-formyl-1,3-benzenedisulfonic acid disodium salt (DSDSBA) [
70] (
Table 3; 1–9).
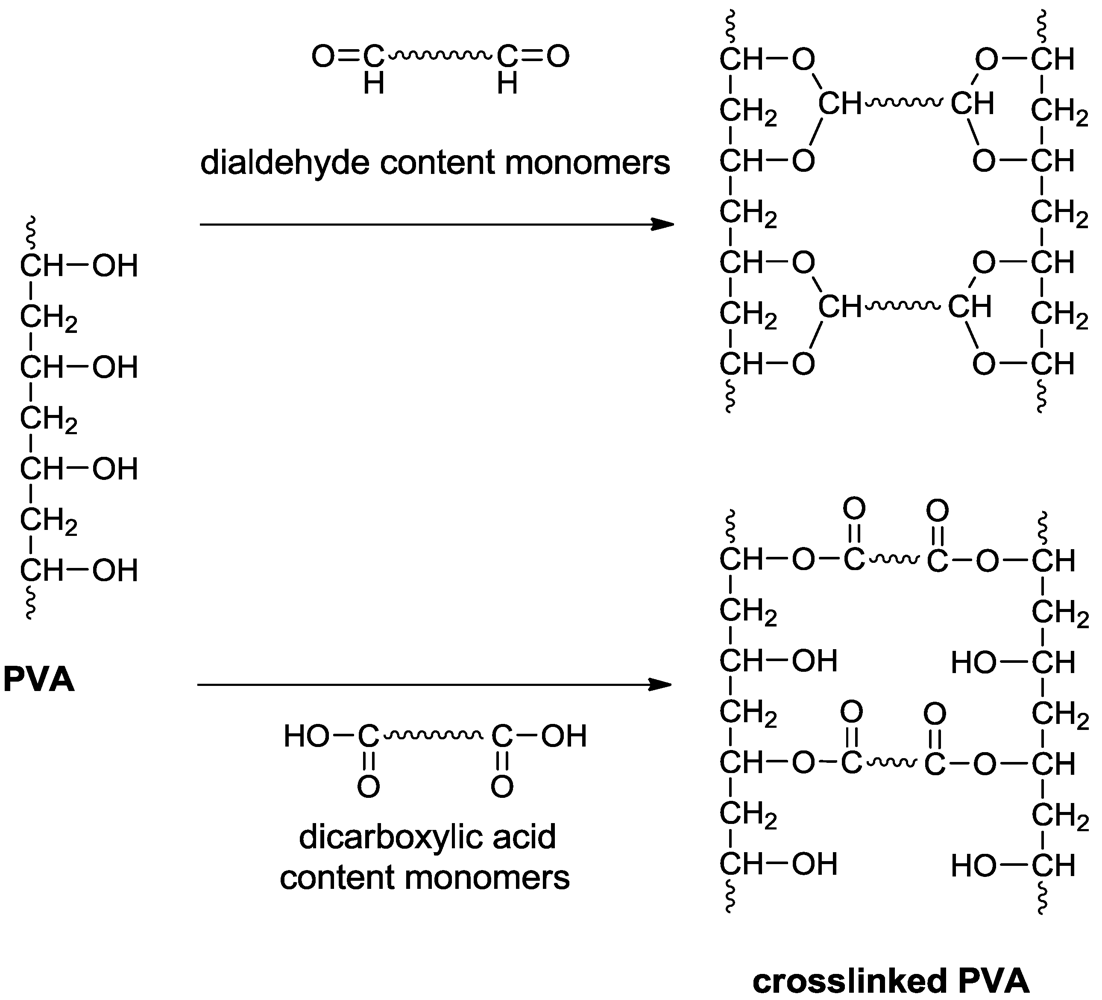
Figure 7 illustrates the preparation of PVA-based crosslinked membranes.
Figure 7.
Scheme illustrating the preparation of PVA-based crosslinked membranes.
Figure 7.
Scheme illustrating the preparation of PVA-based crosslinked membranes.
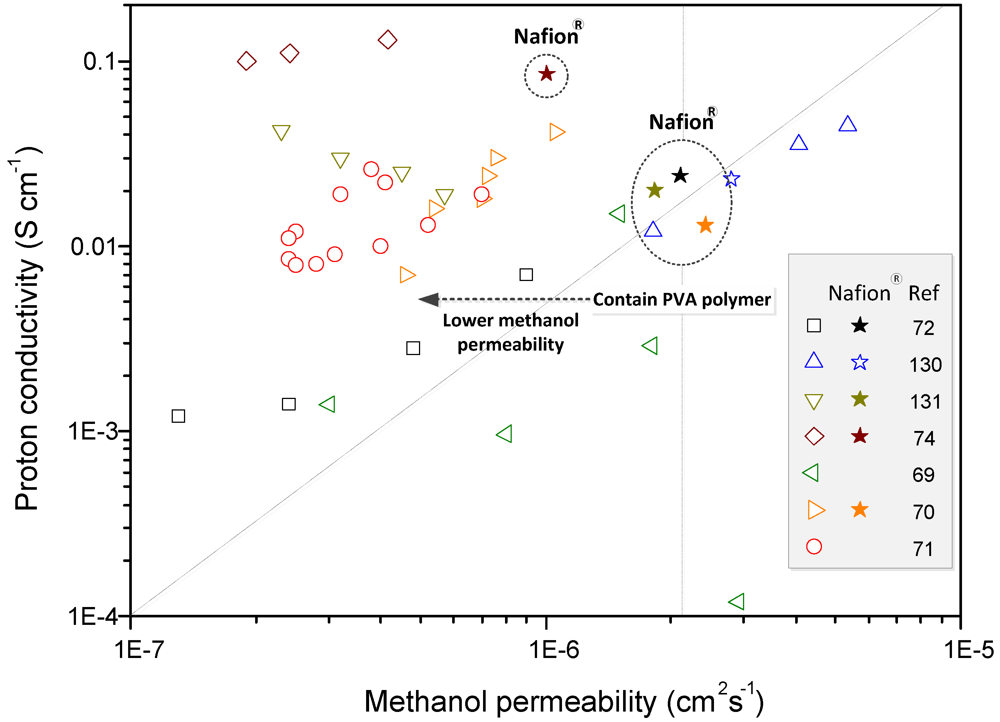
Crosslinked PVA membranes have been prepared using various amounts (5–30 wt%) of sulfosuccinic acid (SSA) as the crosslinking agent and also by varying the crosslinking conditions to achieve desirable proton conductive properties for fuel cell applications [
71] (
Table 4; 1). The results obtained showed that the degree of crosslinking in the PVA/SSA membranes significantly affects their water retention properties, proton conductivities and methanol permeabilities. As the amount of SSA in the PVA/SSA membrane was reduced to less than 17 wt%, the proton conductivity and methanol permeability decreased due to the dominant effect of crosslinking on proton and methanol transport. Above this, the concentration-effect of the sulfonic acid groups contained in the SSA dominates any effects due to crosslinking on the proton’s conductivity and methanol permeability. Thus, crosslinking agents containing sulfonic acid groups can play a decisive role in proton conduction (–SO
3−H
+) and at the same time act as a barrier to methanol transport. Similar results have also been obtained for PVA-based crosslinked membranes by using poly (styrene sulfonic acid-co-maleic acid) (PSSA-PMA) as both crosslinker and as a donor of the hydrophilic group (carboxylic and/or sulfonic acid groups) [
72] (
Table 4; 2). The results show that proton and methanol transport decreased with an increase in the PSSA-PMA content. The effect of crosslinking may play a more dominant role than does an increase in the number of ionic exchange sites. For the two systems above (PVA/SSA and PSSA-PMA), the proton conductivities and the methanol permeabilities of all the membranes were in the ranges 10
−3 to 10
−2 S∙cm
−1 and 10
−7 to 10
−6 cm
2∙s
−1 (in the temperature range 25–90 °C), depending on the crosslinking conditions. The same authors have also mixed other kinds of crosslinkers, e.g., poly (acrylic acid-
co-maleic acid) (PAA-PMA) with PVA to form crosslinked membranes and investigated the aging effect on swelling time, morphology and proton conductivity [
76] (
Table 4; 3). It was found that swelling loosened the polymer’s structure, resulting in an increase in the free volume capable of containing free water molecules. Swelling of the membrane may provide space for the transport of protons and thereby increase the mobility of protonic charge carriers, leading to slightly increased proton conductivities. Although the proton conductivities of these crosslinked PVA/PMA membranes are known to increase, no report of their methanol permeability has yet appeared.
Other crosslinkers containing proton sources (sulfonated phenolic resin (s-Ph)) have been incorporated into PVA to from crosslinked membranes by covalently linking the sulfonated groups of the phenolic resin and the hydroxyl groups of the PVA at 110 °C (
Table 4; 4). This approach sacrifices a certain amount of the sulfonated groups, leading to a slight reduction of proton conductivity. However, the residual conductivity is still significant (10
−2 S∙cm
−1). The reduced methanol permeability (10
−7 cm
2∙s
−1) was due to an increase in the s-Ph concentration with an increasing curing temperature, leading to a reduction of methanol uptake as well as to the membrane’s higher selectivity towards water. The conductivity/permeability ratios (selectivity) of the crosslinked PVA/s-Ph 40/60 composition (40:60 by weight) membranes showed the highest value (0.97 × 10
−4 S∙cm
−3∙s
−1), which is higher than that determined for Nafion
® 117. Sulfonated polyhedral oligosilsesquioxane (sPOSS) has also been incorporated into PVA and crosslinked by ethylene diaminetetraacetic dianhydride (EDTAD) to produce PVA-based crosslinked membranes [
131] (
Table 4; 5). The resulting crosslinked PVA/sPOSS membranes showed that proton conductivity increased and methanol permeability decreased with increasing sPOSS content. Incorporating 20 wt% sPOSS, led to a proton conductivity of about 2 × 10
−2 S∙cm
−1, which is comparable to Nafion
® 117: increasing the sPOSS further to 50 wt% led to a conductivity of 0.042 S∙cm
−1. Additionally, the methanol permeability of PVA/sPOSS membranes decreases significantly with increases in the sPOSS content rendering it lower than Nafion
® 117. sPOSS cages, as crosslinkers and sources of proton, can provide ion conduction paths while the hydrophobic portions of the same sPOSS cages combine to form a barrier to methanol permeation while contributing to the improved thermal stability of the membrane.
Table 4.
Characteristics of various crosslinked membranes that incorporate water soluble polymers.
Table 4.
Characteristics of various crosslinked membranes that incorporate water soluble polymers.
| Line | Polymer | Acid Monomer /Polymer | Crosslinker | Ionic Partner (wt%) | Proton Conductivity (mS∙cm−1)/Measurement Condition | Methanol Permeability × 108 (cm2∙s−1) | References |
|---|
| 1 | PVA | SSA | SSA | 5–30 | ~1–10 | 25–50 °C | 10–100 | [71] |
| 2 | PVA | PSSA-PMA | PSSA-PMA | 5–11 | ~1–10 | 25–90 °C | 10–100 | [72] |
| 3 | PVA | PAA-PMA | PAA-PMA | 7 | ~1–10 | 30 and 50 °C | - | [76] |
| 4 | PVA | sPh | sPh | 20–80 | 12–86 | - | 20.9–181 | [130] |
| 5 | PVA | sPOSS | EDTAD | 20–50 | ~10–50 | 25 °C | 10–100 | [131] |
| 6 | PVA | SPPESK | PVA | 60–85 | ~5–23 | 60 °C | - | [132] |
| 7 | PVA | SPAEK | PVA | 85–100 | 100–220 | 30–80 °C | 19.0–41.6 | [74] |
| 8 | sPVA | - | GA | 100 | 1–15 | RT | 30–290 | [69] |
| 9 | PVA | DSDSBA | GA | ~16–64 | 3–41 | 25 °C | 41–105 | [70] |
| 10 | PVA | PS-
b-PHEA-b-PSSA and SSA | SSA | 10–100 | 18–44 | 25 °C | - | [73] |
| 11 | PEG | SPI | PEG | 75–100 | 10–120 | 30–90 °C | - | [47] |
| 12 | PEG | (PMA-alt-PS)-
co-PAMPS | PEG | 60–80 | 0.9–68 | 30–90 °C | - | [48] |
| 13 | PEG | SPAEK | PEG | 70–100 | ~60–180 | 20–80 °C | - | [51] |
| 14 | PEG | SPEEK | PEG | 60–75 | ~0.4–100 | 100 °C, 40–100% RH | - | [50] |
| CDM | 67–75 | ~0.4–100 | 100 °C, 60–100% RH |
| 15 | sSBS | - | - | - | 23 | 25 °C | 8.1 | [133] |
| 16 | NaSS-4VP | NaSS-4VP | DBD | 75–92 | ~61–223 | 30–70 °C, 90 % RH | 21–646 | [134] |
| 17 | CS | SSA | SSA and GA | 0–16 | ~10–120 | 25–90 °C | ~60–120 | [39] |
Sulfonated aromatic polymers, such as sulfonated poly (phthalazinone ether sulfone ketone) (SPPESK) [
132] and sulfonated poly (arylene ether ketone) bearing carboxylic acid groups (SPAEK-C) [
74], have been used with PVA to modify the membrane’s properties by covalent crosslinking. Gu
et al. [
132] investigated the effect of using PVA as the crosslinker in comparison with a small molecule crosslinker comprising glycol and glycerol: the results indicated that the crosslinking of SPPSK with PVA requires a relatively lower crosslinking temperature and has a much higher stability in water (
Table 4; 6). The swelling ratio and proton conductivity of the SPPESK/PVA crosslinked membrane decreases rapidly with increasing PVA content, because of the decreasing IEC and the increasing crosslinker density. When SPPESK with a high degree of sulfonation (DS: 140 %) is incorporated with a lower content of PVA (<30 wt%) the resulting membranes have a higher proton conductivity than stable pristine SPPESK (DS: 81%). The highest proton conductivity (2.00 × 10
−2 S∙cm
−1) was obtained with SPPESK/PVA 85/15 (85:15 by weight) which is almost twice as that (1.03 × 10
−2 S∙cm
−1) of a pristine SPPEKSK membrane (DS: 81%). More recently, Zhao
et al. [
74] have also used PVA as a crosslinker incorporated into sulfonated poly (arylene ether ketone) bearing carboxylic acid groups (SPAEK-C) using thermal esterification to form a crosslinked membrane (
Table 4; 7). Similar results were observed in SPAEK-C/PVA crosslinked membranes–that is the proton conductivities of these membranes decreased with increasing amounts of PVA due to the lower water uptake and lower IEC. From the relationship between the relative proton conductivity decrease (from 1 to 0.63%) and the relative water uptake decrease (from 1 to 0.27%), it is interesting to find that decreases in the ratios of both proton conductivity and IEC are small with respect to the decrease in the uptake of water. This result indicates that the crosslinked structure of SPAEK-C and PVA can significantly decrease water uptake while maintaining high proton conductivity. Additionally, the methanol permeabilities of SPAEK-C/PVA membranes gradually decreased (from 4.16 × 10
−7 to 1.9 × 10
−7 cm
2∙s
−1) when the PVA content increased (from 0 to 15 wt%), and were all lower than that of Nafion
® 115 (10.05 × 10
−7 cm
2∙s
−1) under the same measurement conditions. The SPPAEK-C/PVA crosslinked membranes with PVA (15 wt%) possess better selectivity, lower water uptakes and adequate proton conductivity,
i.e., nearly five times higher than Nafion
® 115. Both sulfonated aromatic polymers used PVA as a crosslinker, showing that these crosslinked membranes are promising proton conducting materials and have potential applications in DMFCs.
Another approach is to select PVA as the PEM polymer matrix and modify it with negatively charged ions to facilitate proton transfer. In order to improve the mechanical and thermal stabilities of PVA-based membranes, glutaraldehyde (GA) was used as the crosslinking agent to form PVA network membranes. Thus, Shen
et al. [
69] have used PVA as the polymer matrix for PEMs while the protogenic sulfonic acid groups have been introduced by chlorosulfonic acid through a condensation reaction to form sulfonated PVA (sPVA) (
Table 4; 8). The crosslinked sPVA membranes were obtained by an aldol condensation between the OH groups of PVA and the aldehyde groups of GA. Crosslinked sPVA membranes with a high DS show higher conductivity (1.4 × 10
−3 S∙cm
−1) and lower methanol permeability (0.3 × 10
−6 cm
2∙s
−1) than membranes with a low DS (proton conductivity: 0.96 × 10
−3 S∙cm
−1; methanol permeability: 0.8 × 10
−6 cm
2∙s
−1); this is quite different to other sulfonated polymer or perfluorinated ionomer membranes where the methanol permeability increases with conductivity. The authors believe the effect is due to the quite different microstructures and morphologies in these membranes. The same group have also prepared crosslinked s-PVA based membranes by introducing the 4-formyl-1, 3-benzenedisulfonic acid disodium salt (DSDSBA) into the PVA chains act as an ion carrier while using GA as a crosslinker to form the sPVA network [
70] (
Table 4; 9). The combination of a flexible backbone, with short and rigid side chains and dense acid functionalities improved the proton conduction in these sPVA based crosslinked membranes. It was found that by increasing the DSDSBA content up to about 20 wt%, proton the conductivities of these membranes (>1.4 × 10
−2 S∙cm
−1) were higher than that of Nafion
® 117 (1.3 × 10
−2 S∙cm
−1); however, the methanol permeability of these membranes (<0.78 × 10
−6 cm
2∙s
−1) is significantly low when compared to Nafion
® 117 (2.43 × 10
−6 cm
2∙s
−1).These results can be attributed to the well connected system of narrow water channels that facilitated both a lower methanol permeation and the high mobility of the protonic charge carriers.
Finally, proton conducting crosslinked membranes have also been prepared using polymer blends of poly (styrene-b-hydroxyethyl acrylate-b-styrene sulfonic acid) (PS-
b-PHEA-
b-PSSA) and PVA (
Table 4; 10). The middle PHEA triblock copolymer was crosslinked with PVA by an esterification reaction using sulfosuccinic acid (SSA) as a crosslinking agent. Similar results were also observed in the IEC values (from 1.56 to 0.61 meq∙g
−1) and proton conductivity (4.4 × 10
−2 to 1.8 × 10
−2 S∙cm
−1), which continuously decreased with an increasing PVA content (from 33 to 66 wt%). In contrast, the mechanical properties increased from 15.0 to 38.5 MPa with increases in the PVA content, due to the decrease of water uptake (from 87.0 to 44.3%). Operating in an H
2/O
2 polymer electrolyte membrane fuel cell (PEMFC), the crosslinked membrane containing PVA 50 wt% had a power density of 230 mW∙cm
−2 at 70 °C and 100 RH %.
4.2. Crosslinked Poly (ethylene glycol) Based Membranes
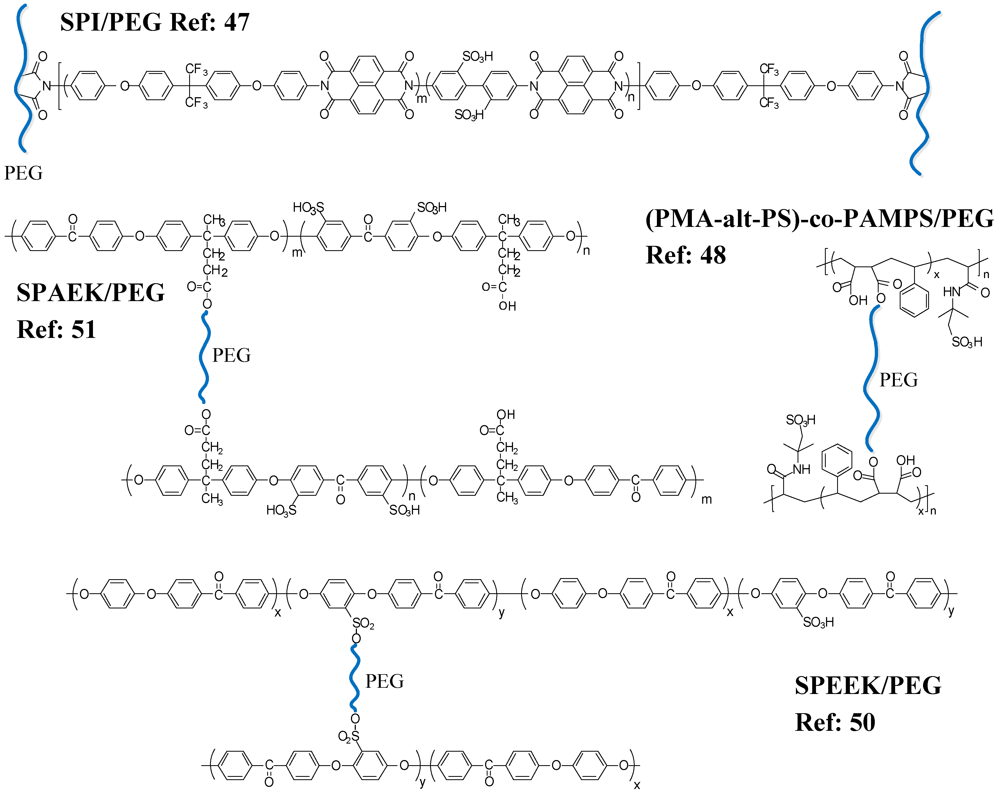
Other WSP polymers, e.g., poly (ethylene glycol) (PEG), have also been employed in the preparation of crosslinked membranes for fuel cell applications. PEG is generally used as a crosslinker and has been introduced into several sulfonated polymer matrices, such as sulfonated polyimide (SPI) [
47], poly [(maleic anhydride)-alt-styrene-
co-(2-acrylamido-2-methyl-1-propanesulfonic acid)] (PMA-alt-PS)-
co-PAMPS [
48], sulfonated poly (arylene ether ketone) (SPAEK) [
51] and sulfonated poly (ether ether ketone) (SPEEK) [
50], to improve the membrane’s mechanical and hydrolytic stabilities while reducing methanol crossover without compromising proton conductivity and brittleness. (
Figure 8 and
Table 4; 11–14) Thus, Yang
et al. [
47] prepared a series of crosslinked SPI/PEG membranes with various ratios of maleic anhydride SPI and PEG diacrylate as PEMs (
Table 4; 11). Their results showed that the water uptake (from 23.8 to 24.3%) and the proton conductivity increased with increasing contents of PEG (from 0 to 25 wt%) despite a decrease in the ion exchange capacity (IEC) (from 1.27 to 1.23 meq∙g
−1) and an increase in the crosslinking density. The effect of the flexible PEG crosslinker on the proton conductivities and hydrolytic stabilities in the crosslinked SPI membrane with 25 wt% content of PEG were more obvious, showing high proton conductivities (>0.1 S∙cm
−1) and good hydrolytic stabilities (>200 h to the time when the film started to disintegrate) at high temperatures (80~90 °C).
Figure 8.
Proposed structure of sulfonated polymer/ Poly (ethylene glycol) (PEG) crosslinked membranes.
Figure 8.
Proposed structure of sulfonated polymer/ Poly (ethylene glycol) (PEG) crosslinked membranes.
Effects due to the molecular weight and the amount of PEG incorporated on the membrane’s properties include alterations to its mechanical properties, swelling behavior, and proton conductivity; these are discussed in (PMA-alt-PS)-
co-PAMPS [
48] and SPAEK systems [
51]. A series of (PMA-alt-PS)-
co-PAMPS crosslinked membranes using PEG with different molecular weights (M
n = 200, 1450 and 4,000 g∙mol
−1) and amounts (20–40 wt%) have been prepared [
48] (
Table 4; 12). The resulting membrane’ tensile strength slightly increased with decreasing PEG chain lengths–this chain length decrease also has the effect of making the membranes more rigid. These results imply that short PEG crosslinking molecules in the polymer matrix could not provide sufficient flexibility to produce stable membranes when compared to longer PEG crosslinking molecules. With respect to swelling behavior, the water absorption was increased with an increase in the chain length of PEG and a decrease in its content. The highest proton conductivity was obtained when the membranes were prepared with a lower content (20 wt%) of the PEG crosslinker but with a high molecular weight (M
n = 4,000 g∙mol
−1). Similar results have also been observed for a SPAEK/PEG membrane, in which PEG was used as the crosslinker [
51] (
Table 4; 13). In all SPAEK/PEG crosslinked membranes the conductivity decreased with an increasing PEG content. The decrease in water uptake and IEC with increasing PEG content is thought to be the reason for the observed decrease in proton conductivity. Although the introduction of the crosslinker PEG in the membranes generally leads to a decrease in proton conductivity; at higher temperatures (80 °C) a significant increase in proton conductivity and chemical stability is apparent. More recently, Gupta
et al. [
50] have prepared crosslinked membranes based on SPEEK in the presence of varying amounts of PEG and cyclohexane dimethanol (CDM) (
Table 4; 14). PEG is more flexible and hydrophilic than CDM, so this structural difference is reflected clearly in the finer morphology of the network produced, the enhanced storage modulus, the glass transition temperature and the proton conductivity. The highest proton conductivity (5.7 × 10
−2 S∙cm
−1) was achieved in SPEEK/PEG 67/33 (67: 33 by weight) which is comparable to neat SPEEK (5.6 × 10
−2 S∙cm
−1). Results from various sulfonated polymer matrices made using PEG as crosslinker showed that PEG is a very effective crosslinker in the sense that it maintains hydrolytic stability even at higher temperatures with no compromise in proton conductivity.
4.3. Other Crosslinked Membranes Involve Water Soluble Polymer
Other crosslinked membranes that incorporate WSP have been prepared for application in fuel cells. Won
et al. [
133] used a crosslinking approach to control and fix microphase separated sulfonated poly (styrene-b-butadiene-b-styrene) (sSBS) to reduce methanol permeability while maintaining high proton conductivity (
Table 4; 15). The crosslinked sulfonated SBS membranes (scSBS) were prepared by photo-crosslinking and additional sulfonating to obtain well-structured (
i.e., semi-rigid) PEMs. The effect of fixed channels on methanol permeability and proton conductivity were confirmed by comparing the resulting properties of scSBS and non-crosslinkable sulfonated poly (styrene-b-(ethylene-r-butylene)-b-styrene (sSEBS) membranes. The results show that the scSBS membrane (DS = 36.8) exhibits a methanol permeability of 8.1 × 10
−8 cm
2∙s
−1,
i.e., more than 30 times smaller than that of Nafion
® 117 (2.9 × 10
−6 cm
2∙s
−1), and a proton conductivity of 2.3× 10
−2 S∙cm
−1, which is comparable to that of Nafion
® 117 (6.3 × 10
−2 S∙cm
−1) and sSEBS (3.8 × 10
−2 S∙cm
−1). These results indicated that limiting the ionic channel size would severely limit the methanol permeability but would not affect the proton conductivity. Crosslinked membranes composed of zwitterionic molecules with various crosslinking densities have also been fabricated for the development of high durability PEMs (
Table 4; 16). After crosslinking poly (styrene sulfonic acid-
co-vinylpyridine) (NaSS-4VP) membranes with a crosslinking fraction of 90.3 showed excellent hydrolytic stability with only slightly changes in weight (decrease in weight: 1.9%) and proton conductivity (from 7.1 to 5.5 × 10
−2 S∙cm
−1) after 28 days of testing in water at 100 °C: note that the initial NaSS-4VP is a WSP. Moreover, an optimum membrane maintained adequate proton conductivities (>10
−2 S∙cm
−1) after 120 h in a high concentration of Fenton’s reagent, and is thus much stronger than other sulfonated polymers that typically decompose after 24–60 h. The optimum membrane possessed a high selectivity for methanol fuel cells (3.38 × 10
5 S∙cm
−3∙s
−1),
i.e., approximately five times that of Nafion
® 117 (0.71 × 10
5 S∙cm
−3∙s
−1).
With its distinct advantages in forming an excellent alcohol barrier while maintaining acceptable ion conductivity properties, chitosan (CS) has been used for the polymer matrix of PEMs [
39] (
Table 4; 17). Various compositions of sulfosuccinic acid (SSA) and GA were introduced into CS as a binary crosslinking agent to form CS network membranes. It was found that both proton conductivity and methanol permeability were increased with increasing contents of proton sources of SSA. According to selectivity value calculations, the optimum composition of crosslinking agent was chosen as 12 and 2 weight % of SSA and GA. The optimum membrane (CS/SSA/GA 12) showed relatively high proton conductivity (4.5 × 10
−2 S∙cm
−1) and low methanol permeability (9.6 × 10
−7 cm
2∙s
−1). The selectivity of the optimum membrane was approximately 4.7 × 10
4 S∙cm
−3∙s
−1 compared to 4.1 × 10
4 S∙cm
−3∙s
−1 for Nafion
®. The single cell DMFC performance test showed that the CS/SSA/GA 12 membrane had a maximum power density (with 2 M methanol feed at 30 °C) of 17 mW∙cm
−2, while a higher power density 41 mW∙cm
−2 was obtained at 60 °C. From the results, it is apparent that these CS based crosslinked membranes have promising potential for use in DMFC applications.