Controlled Photoradical Polymerization Mediated by 2,2,6,6-Tetramethylpiperidine-1-Oxyl
Abstract
:1. Introduction
2. Results and Discussion
2.1. Molecular Weight Control of Polymers
| MTEMPO/AMDV | BAI/MTEMPO | Time (h) | Conv. (%) | Mn a | Mw/Mn a |
|---|---|---|---|---|---|
| – b | – | 1 | 85 | 33,000 | 6.94 |
| – | – c | 1 | 87 | 32,300 | 5.86 |
| 1.1 | – | 31 | 40 | 11,600 | 1.47 |
| 1.1 | 0.5 | 3 | 68 | 16,200 | 1.66 |
| 1.1 | 0.75 | 2.5 | 88 | 19,800 | 2.88 |
| 1.1 | 1.5 | 2 | 95 | 20,600 | 3.16 |
| 1.4 | 0.5 | 9 | 68 | 13,600 | 1.41 |
| 2.0 | 0.5 | 12 | 68 | 10,600 | 1.55 |
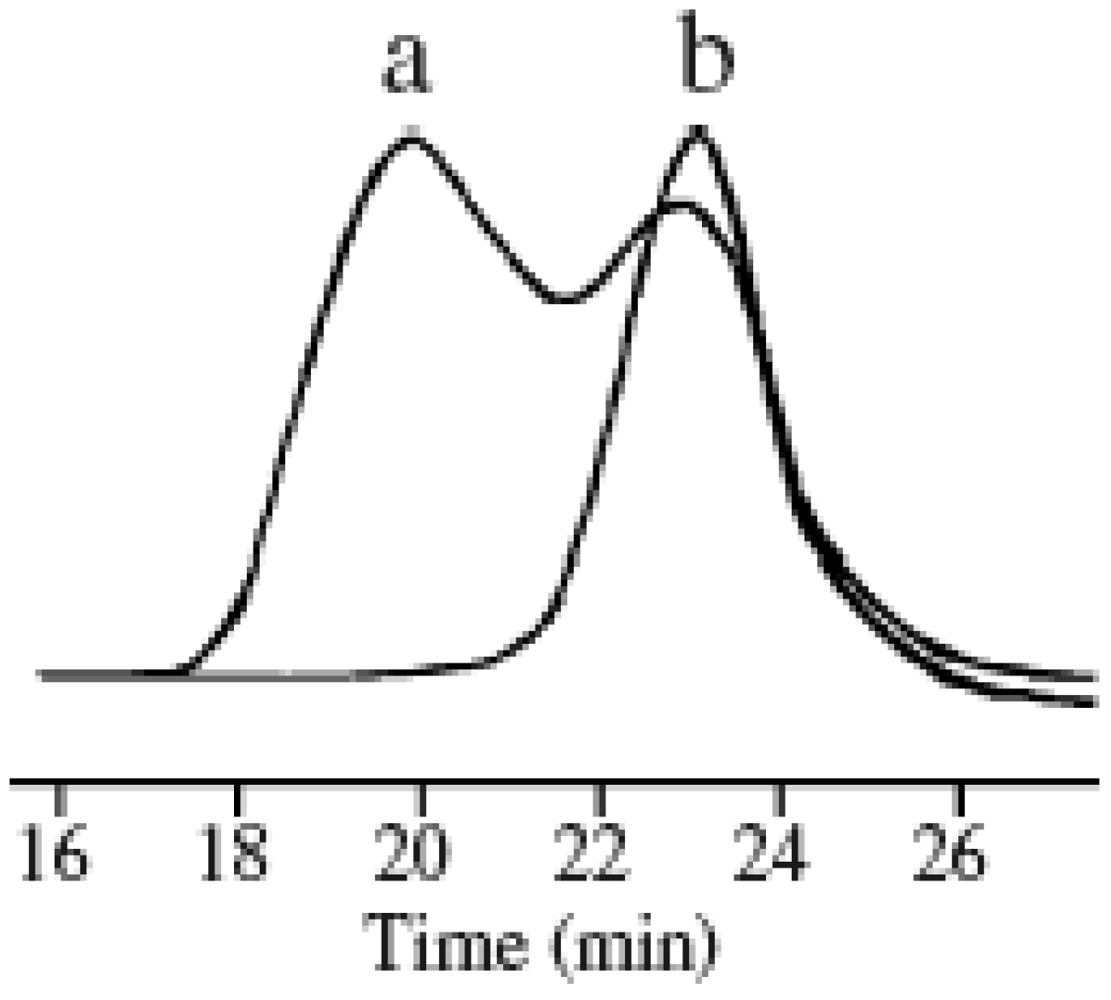
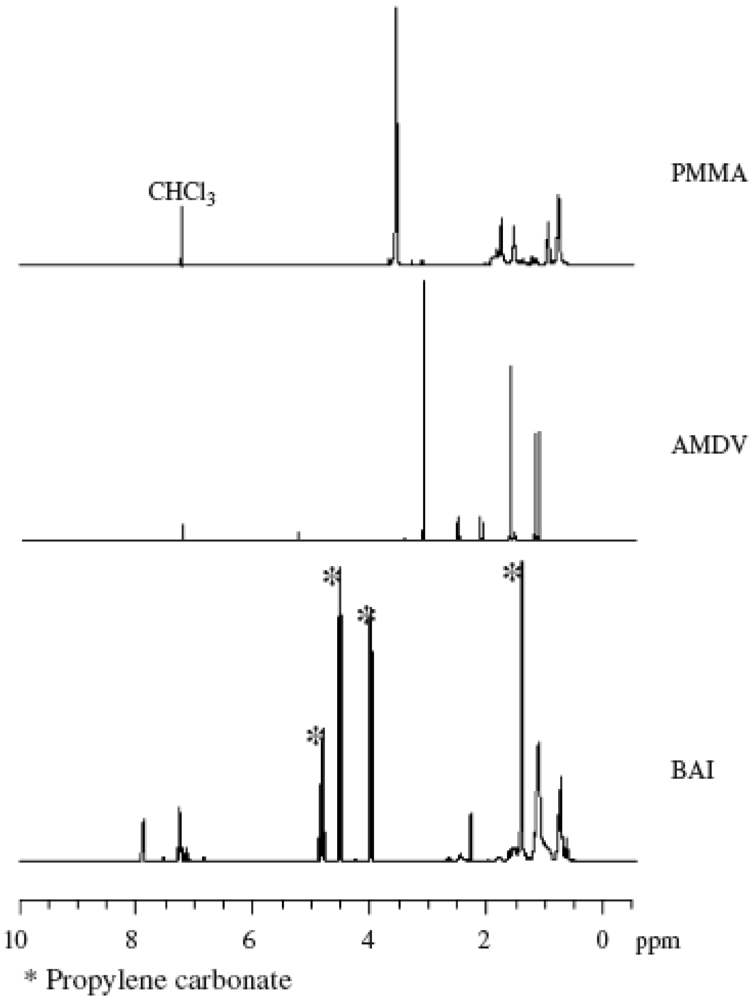
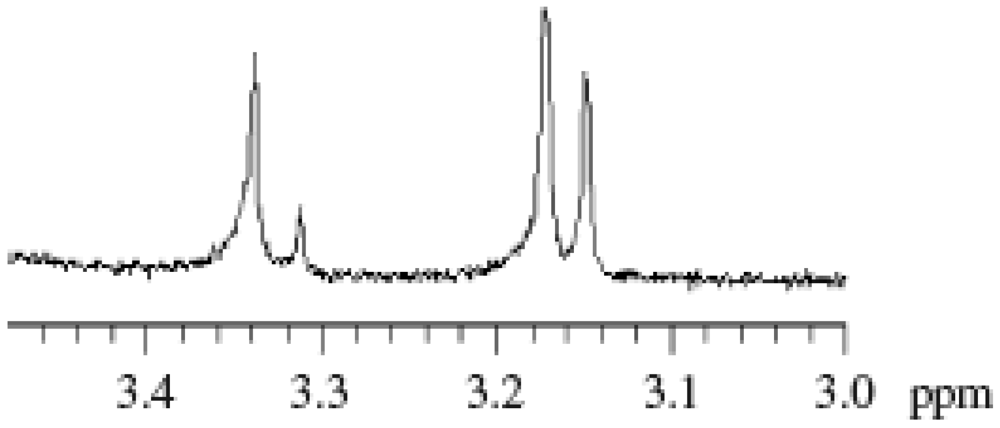

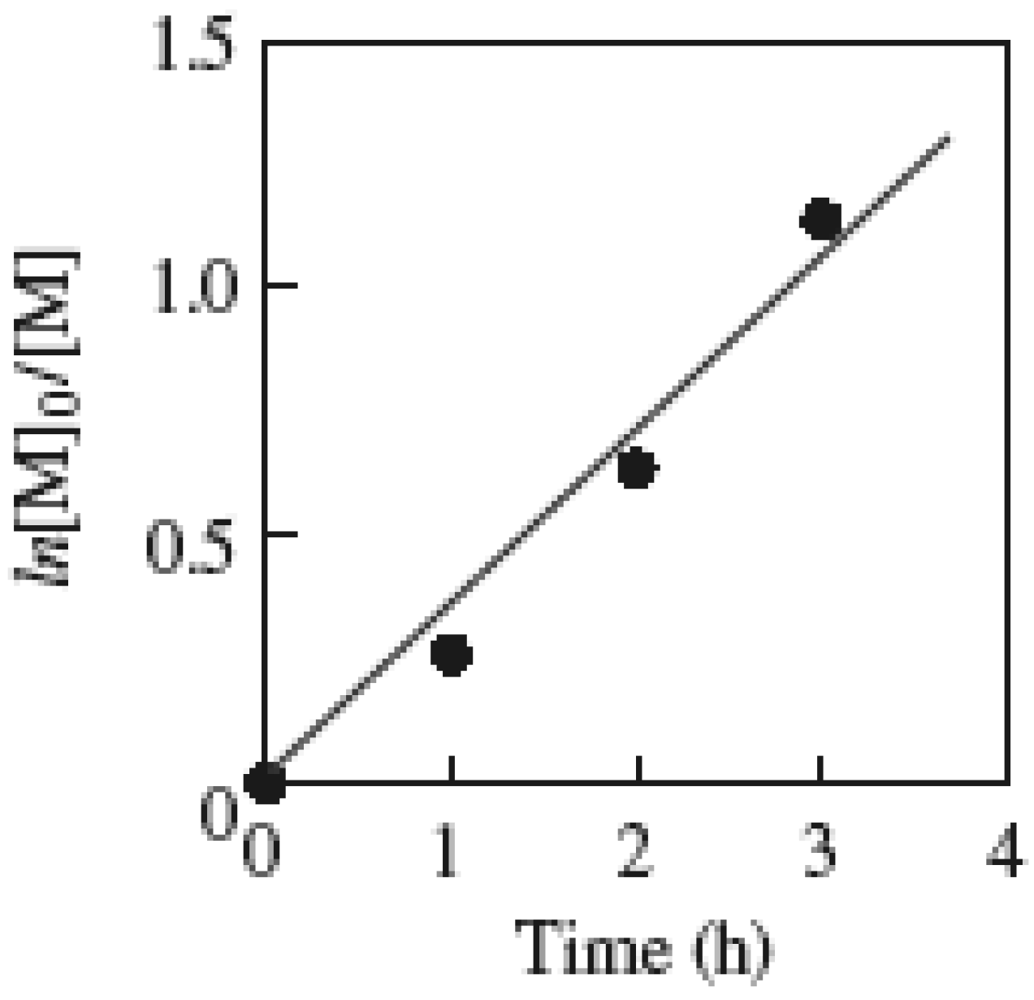
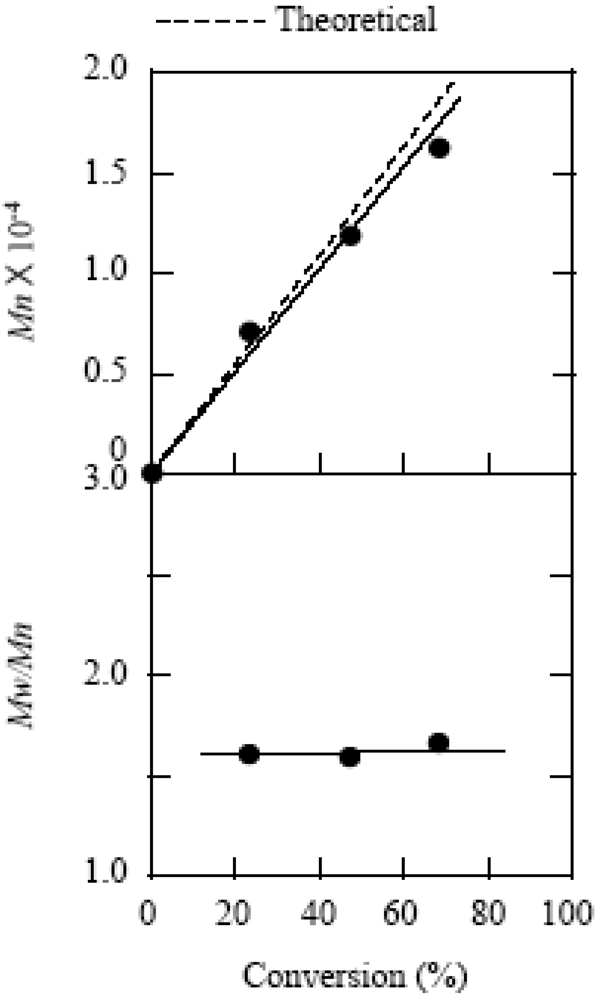
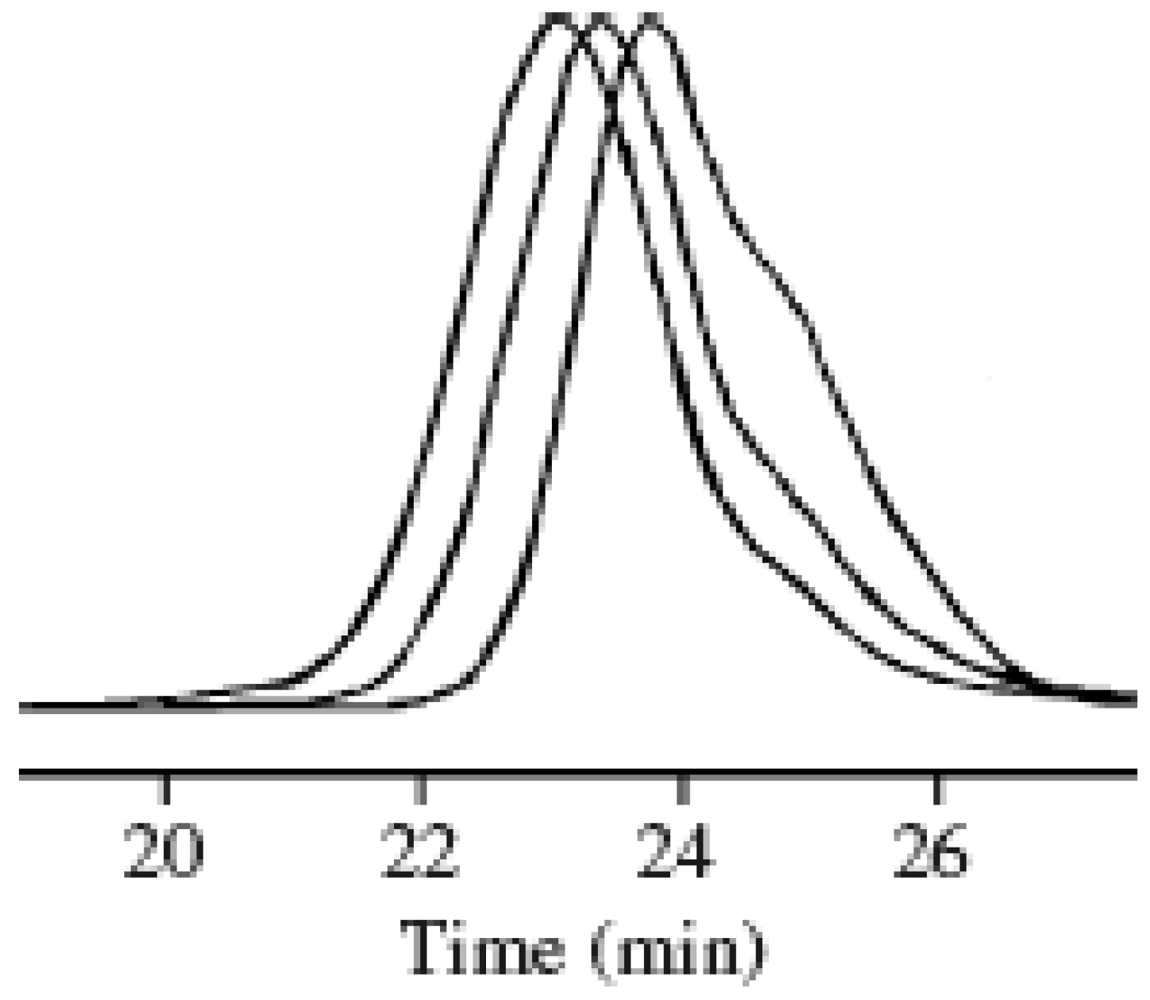
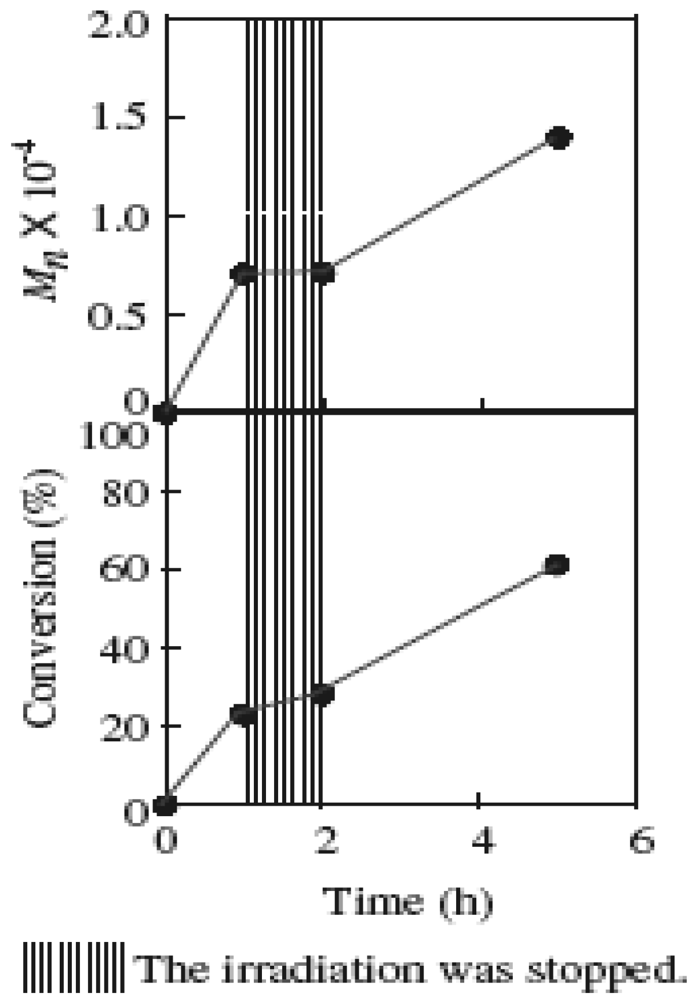
2.2. Effect of Initiators
| Initiators | MTEMPO/Initiator | Conv. (%) | Mn b | Mw/Mn b | [P] (mM) | IE |
|---|---|---|---|---|---|---|
| AIBN | 1 | 95 | 39,700 | 3.37 | 22.4 | 0.131 |
| 2 | 36 | 15,300 | 1.68 | 22.0 | 0.129 | |
| V-59 | 1 | 97 | 31,600 | 3.37 | 28.8 | 0.173 |
| 2 | 46 | 15,700 | 1.66 | 27.4 | 0.165 | |
| V-65 | 1 | 96 | 18,000 | 2.33 | 50.0 | 0.296 |
| 2 | 62 | 9,410 | 1.53 | 61.7 | 0.365 | |
| V-40 | 1 | 98 | 38,200 | 3.24 | 24.0 | 0.140 |
| 2 | 67 | 26,300 | 1.65 | 23.9 | 0.139 | |
| r-AMDV | 1 | 60 | 7,640 | 1.52 | 73.5 | 0.436 |
| 2 | 37 | 3,530 | 1.28 | 98.2 | 0.582 | |
| m-AMDV | 1 | 68 | 10,100 | 1.50 | 63.3 | 0.375 |
| 2 | 40 | 4,580 | 1.34 | 81.8 | 0.485 | |
| V-601 | 1 | 84 | 26,300 | 1.99 | 29.9 | 0.181 |
| 2 | 35 | 10,700 | 1.68 | 30.6 | 0.185 | |
| VAm-110 | 1 | 25 | 16,600 | 1.53 | 14.1 | 0.0847 |

| Initiators | λmax (nm) | Absorbance | ε | T1/2 (10 h) |
|---|---|---|---|---|
| AIBN | 345 | 0.176 | 12.3 | 65 |
| V-59 | 348 | 0.226 | 15.8 | 67 |
| V-65 | 348 | 0.291 | 20.4 | 51 |
| V-40 | 350 | 0.236 | 16.5 | 88 |
| r-AMDV | 348
253 | 0.404
0.050 | 28.3
3.50 | 30 |
| m-AMDV | 341
253 | 0.245
0.078 | 17.2
5.47 | 30 |
| V-601 | 363
253 | 0.273
0.164 | 19.1
11.5 | 66 |
| VAm-110 | 376
258 | 0.455
2.392 | 31.9
167.7 | 110b |
2.3. Mechanisms
| [CMTMP]0 (mM) | [BAI]0 (mM) | BAI/CMTMP | Time (h) | Conv. (%) | Mn a | Mw/Mn a | [P] (mM) | I.E. |
|---|---|---|---|---|---|---|---|---|
| 47.2 | 0 | 0 | 24 | 2 | − | − | − | − |
| 47.2 | 5.24 | 0.11 | 5 | 32 | 27,300 | 1.68 | 11.0 | 0.23 |
| 47.2 | 11.8 | 0.25 | 5 | 51 | 29,300 | 1.66 | 13.7 | 0.29 |
| 47.2 | 24.9 | 0.53 | 5 | 50 | 22,600 | 1.69 | 17.4 | 0.37 |
| 47.2 | 47.2 | 1.0 | 5 | 55 | 16,800 | 1.78 | 30.6 | 0.65 |
| 47.2 | 47.2 | 1.0 | 10 | 94 | 25,700 | 2.30 | 34.2 | 0.73 |
| 31.5 | 31.5 | 1.0 | 11 | 82 | 33,400 | 1.88 | 23.0 | 0.73 |
| 94.4 | 94.4 | 1.0 | 3.5 | 92 | 17,300 | 2.67 | 49.8 | 0.53 |
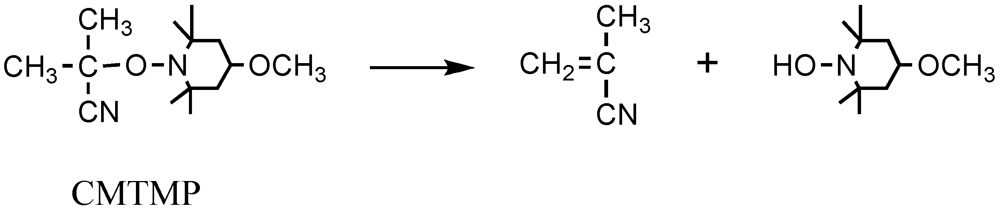
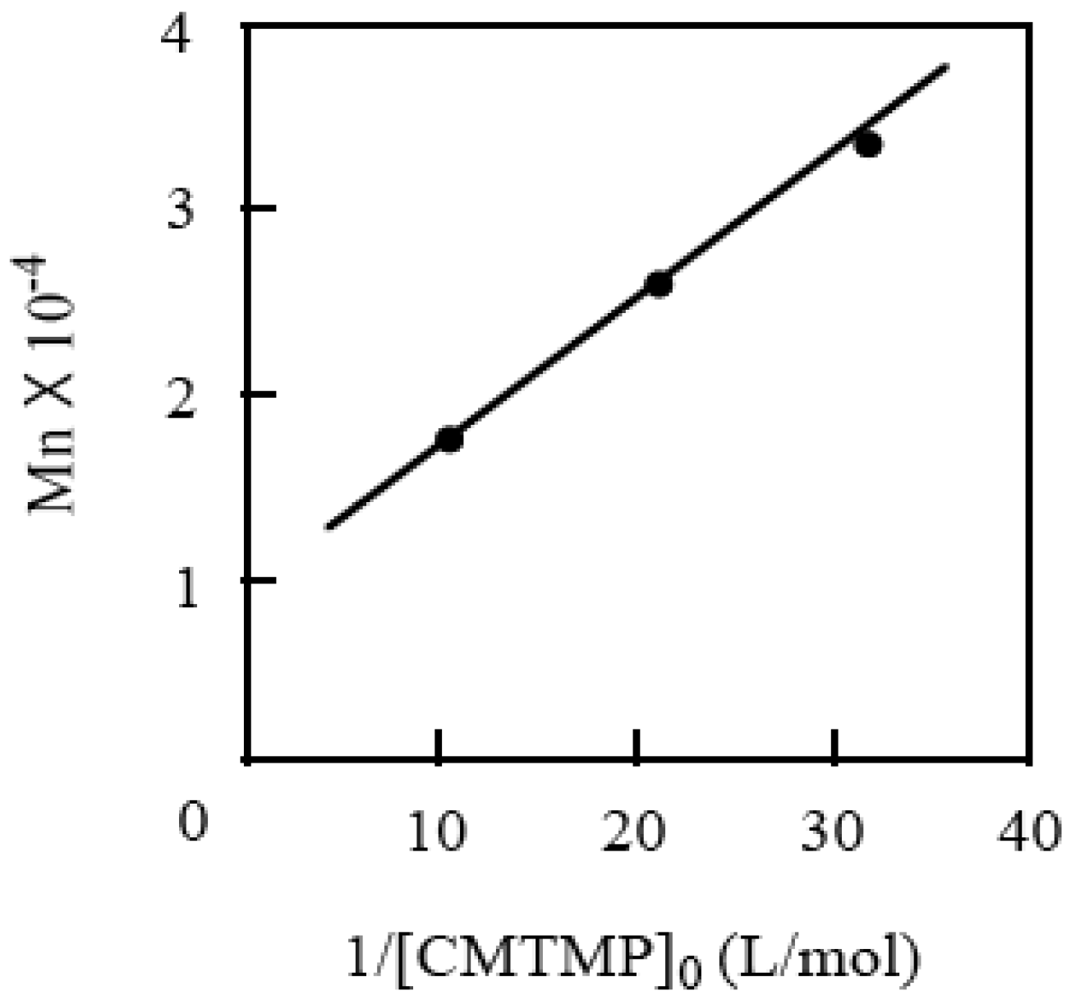
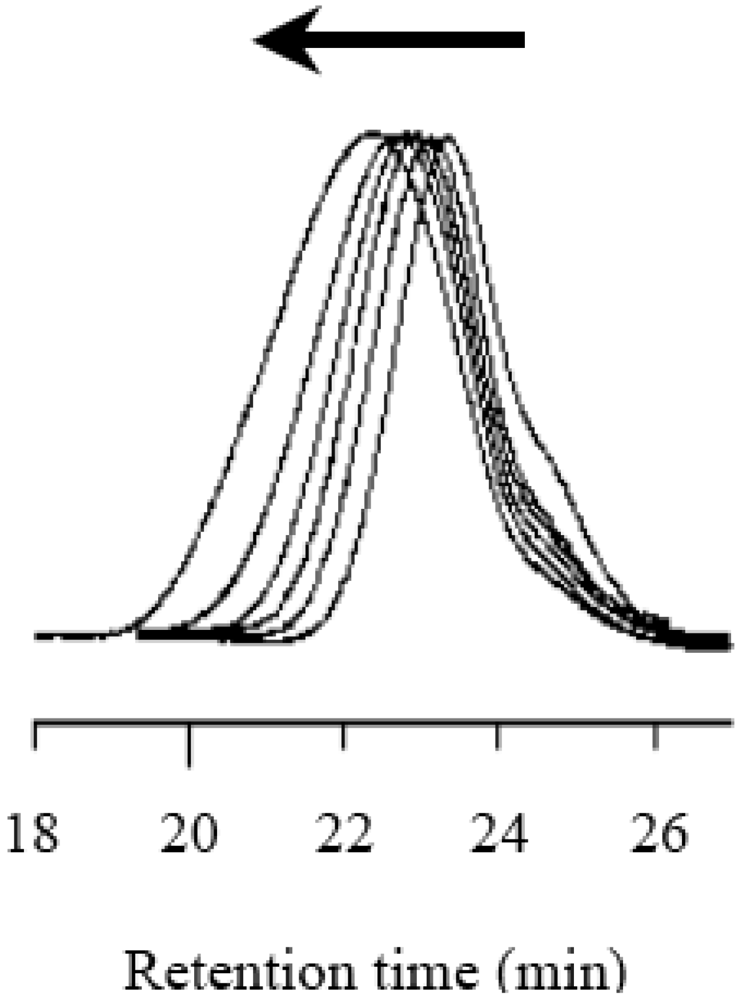
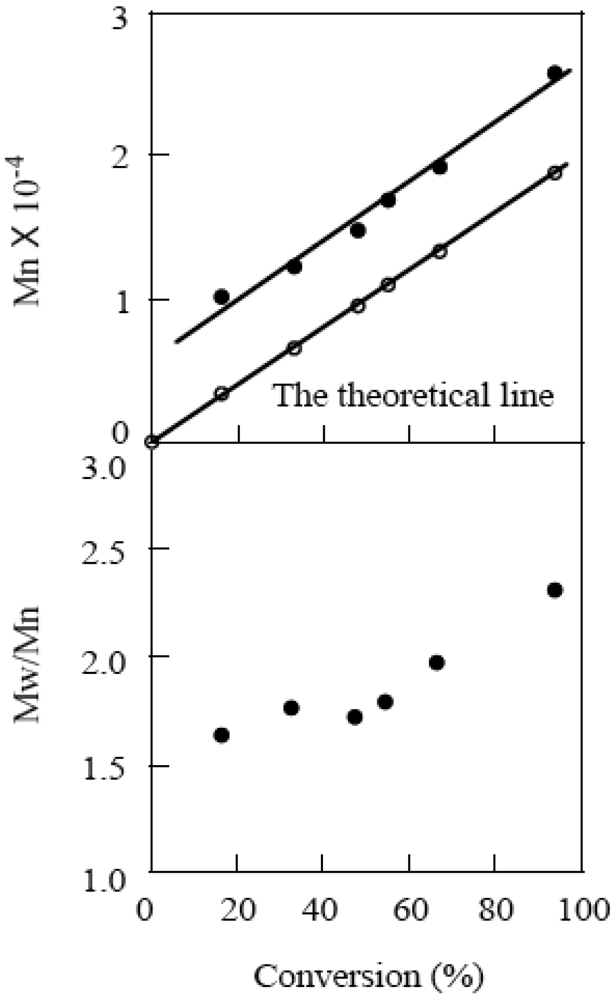
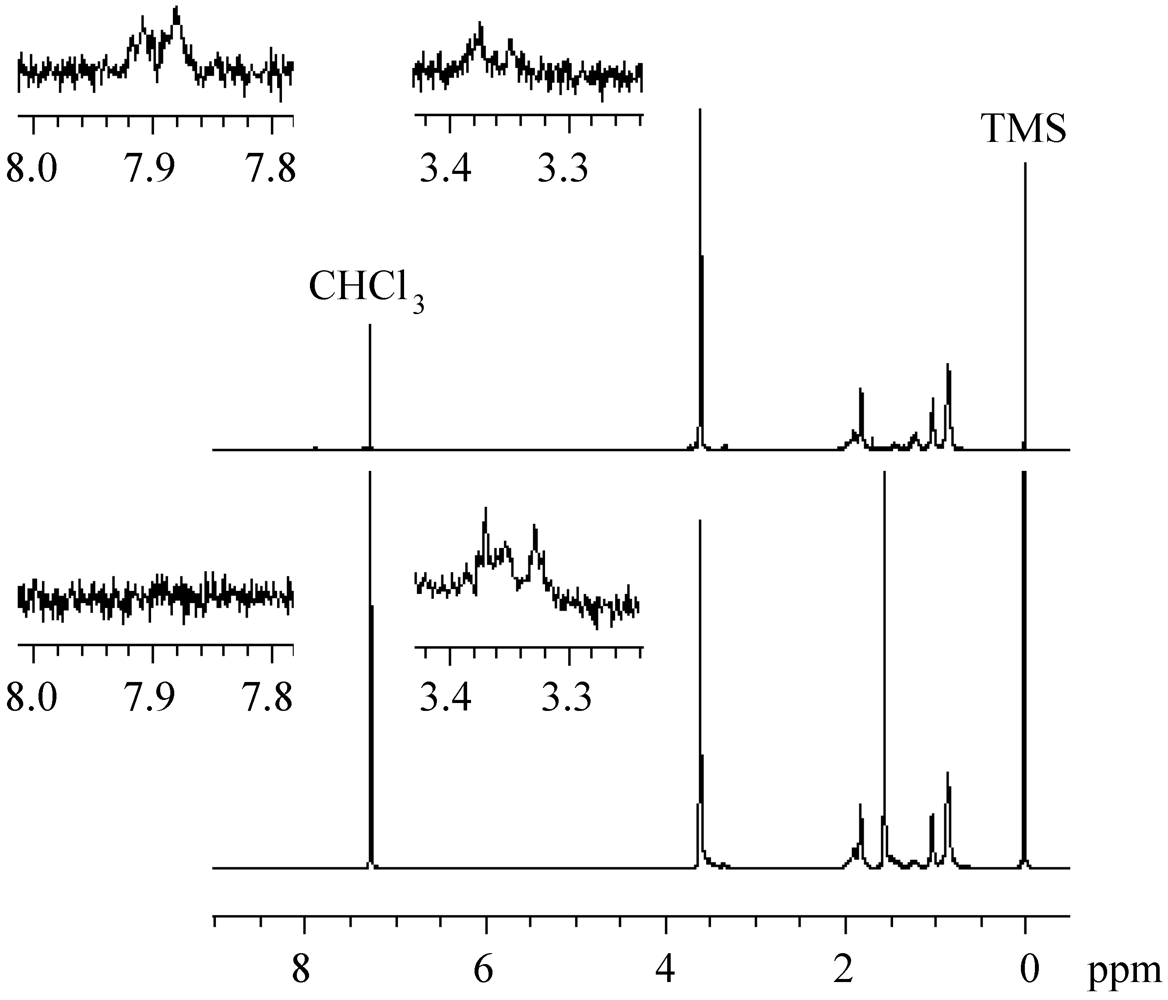

2.4. Effects of Photo-Acid Generators
2.4.1. Triarylsulfonium Salts
| [MTEMPO]0 (mM) | [tBuS] 0 (mM) | MTEMPO/AMDV | tBuS/MTEMPO | Time (h) | Conv. (%) | Mn b | Mw/Mnb | [P] (mM) |
|---|---|---|---|---|---|---|---|---|
| 48.3 | − | 1.06 | − | 31 | 40 | 11,600 | 1.47 | 32.3 |
| 48.3 | 12.8 | 1.06 | 0.265 | 9 | 63 | 13,200 | 1.46 | 44.8 |
| 48.3 | 23.5 | 1.06 | 0.487 | 9 | 63 | 11,100 | 1.46 | 53.0 |
| 48.3 | 34.1 | 1.06 | 0.706 | 9 | 64 | 10,200 | 1.44 | 59.0 |
| 48.3 | 47.0 | 1.06 | 0.973 | 6 | 77 | 15,700 | 1.58 | 46.0 |
| 48.3 | 70.4 | 1.06 | 1.46 | 6 | 71 | 11,100 | 1.45 | 60.1 |
| 80.5 | 66.2 | 1.77 | 0.822 | 23 | 74 | 9,350 | 1.51 | 74.1 |
| 91.3 | 91.8 | 2.01 | 1.01 | 23 | 82 | 7,790 | 1.70 | 98.5 |
| 91.3 | 47.0 | 2.01 | 0.515 | 14 | 26 | 4,670 | 1.36 | 52.1 |
| 21.5 | 10.7 | 0.473 | 0.498 | 5 | 71 | 18,300 | 1.76 | 36.4 |
| 48.3 | 25.6 | − c | 0.530 | 6 | 0 | − | − | − |
| − | 94.4 | − c | − | 3 | 73 | 417,000 | 19.6 | 1.64 |

| Run no. | R1 | R2 | Solubility b | Conv. (%) | Mn c | Mw/Mn c | [P] (mM) | IE |
|---|---|---|---|---|---|---|---|---|
| 1 |  | I | 63 | 12,200 | 1.42 | 48.2 | 0.531 | |
| 2 | S | 52 | 10,200 | 1.39 | 47.8 | 0.527 | ||
| 3 | S | 58 | 11,000 | 1.43 | 49.5 | 0.545 | ||
| 4 | I | 62 | 11,000 | 1.50 | 52.9 | 0.582 | ||
| 5 | S | 46 | 9,810 | 1.45 | 43.9 | 0.483 | ||
| 6 | S | 80 | 427,000 d 14,000 | 3.64 1.70 | 40.1 | 0.442 | ||
| 7 | S | 66 | 12,200 | 1.45 | 51.0 | 0.562 | ||
| 8 | S | 58 | 12,600 | 1.44 | 43.0 | 0.474 | ||
| 9 | S | 64 | 11,400 | 1.45 | 52.3 | 0.577 | ||
| 10 | I | 53 | 11,000 | 1.44 | 45.0 | 0.496 | ||
| 11 | S | 70 | 12,100 | 1.49 | 54.0 | 0.594 | ||
| 12 | I | 62 | 10,100 | 3.14 | 57.5 | 0.634 | ||
| 13 | I | 73 | 13,200 | 2.40 | 51.9 | 0.571 | ||
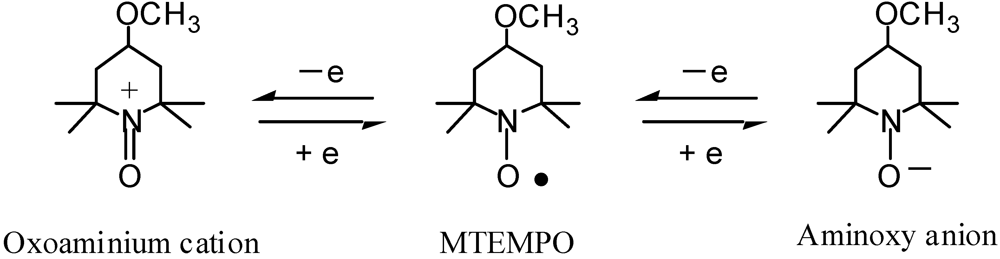
2.4.2. An Iron-Arene Complex
| r-AMDV (μmol) | MTEMPO (μmol) | BzCpFeII (μmol) | Time (h) | Conv. (%) | Mn a | Mw/Mn a |
|---|---|---|---|---|---|---|
| 45.4 | 48.3 | 48.3 | 3 | 73 | 17,500 | 1.54 |
| 45.4 | 48.3 | 48.3 | 4 | 66 | 16,700 | 1.58 |
| 45.4 | 48.3 | 96.5 | 7 | 46 | 11,500 | 1.47 |
| − | − | 48.3 | 3 | 6 | 90,500 | 3.09 |
| − | 48.3 | 48.3 | 3 | 20 | 48,300 | 2.45 |
| 45.4 | − | 48.3 | 1 | 80 | 15,100 | 7.21 |

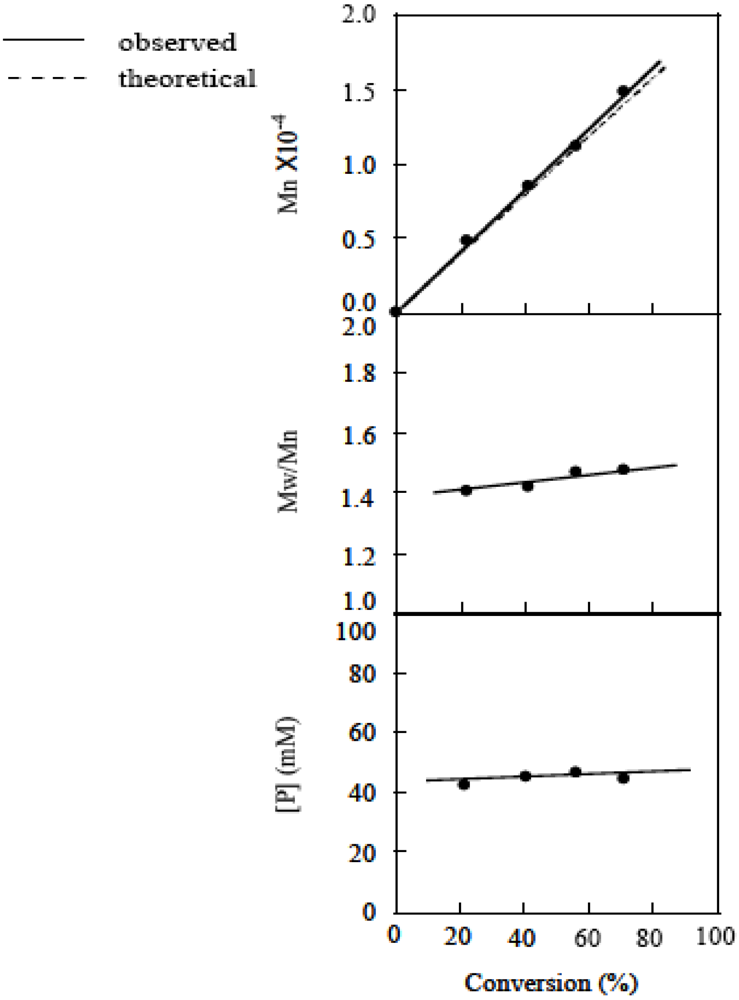
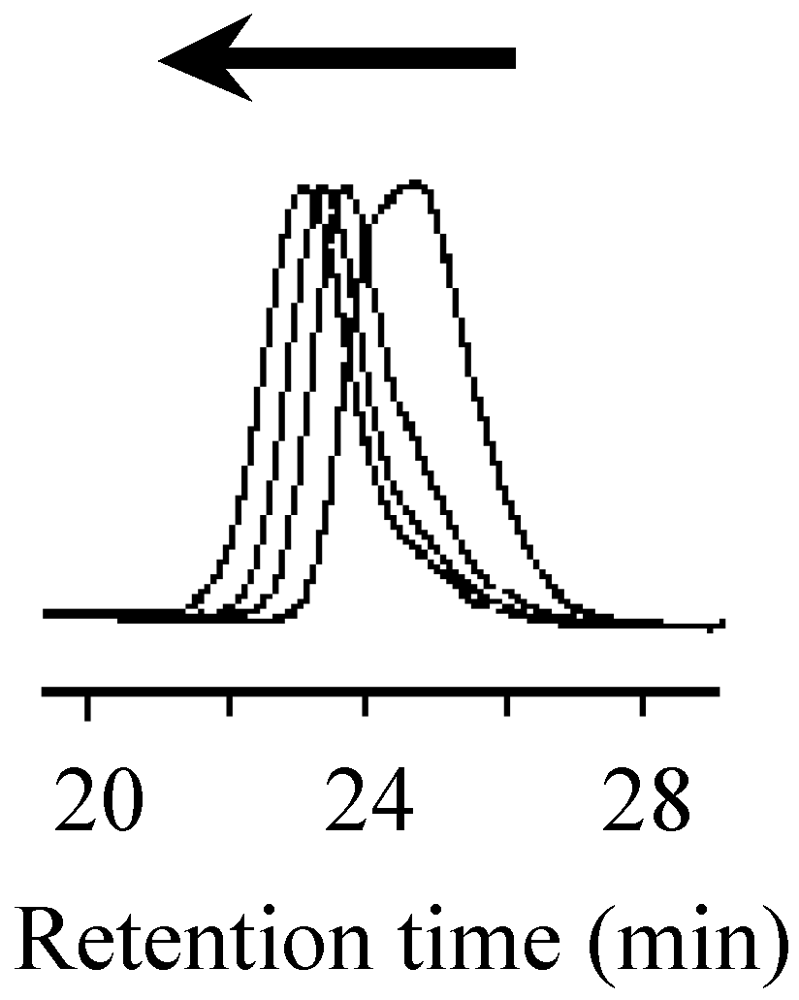
2.5. Stability of the Growing Polymer Chain End
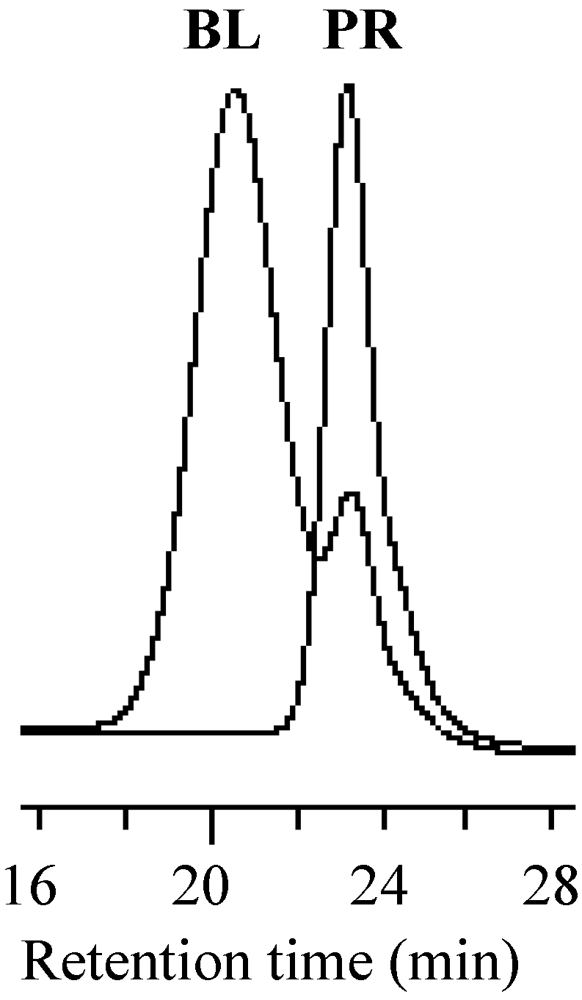

2.6. Solution Polymerization
| MTEMPO/r-AMDV | tBuS/MTEMPO | Conv. (%) | Mn a | Mw/Mn a |
|---|---|---|---|---|
| 1.06 | 0.53 | 80 | 7,930 | 1.72 |
| 2.13 | 0.53 | 67 | 10,100 | 1.67 |
| 2.13 | 1.02 | 97 | 11,700 | 1.78 |

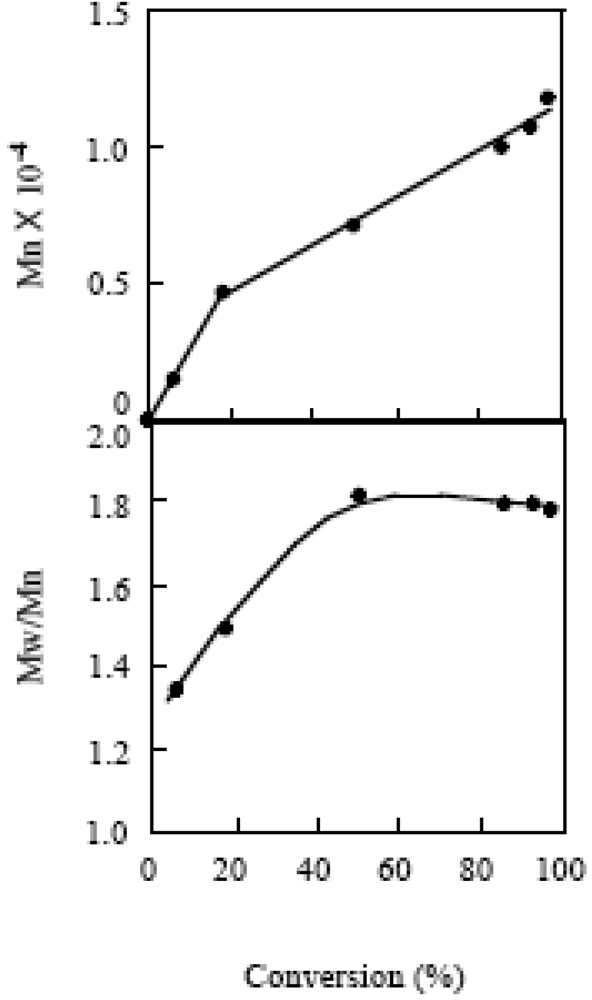
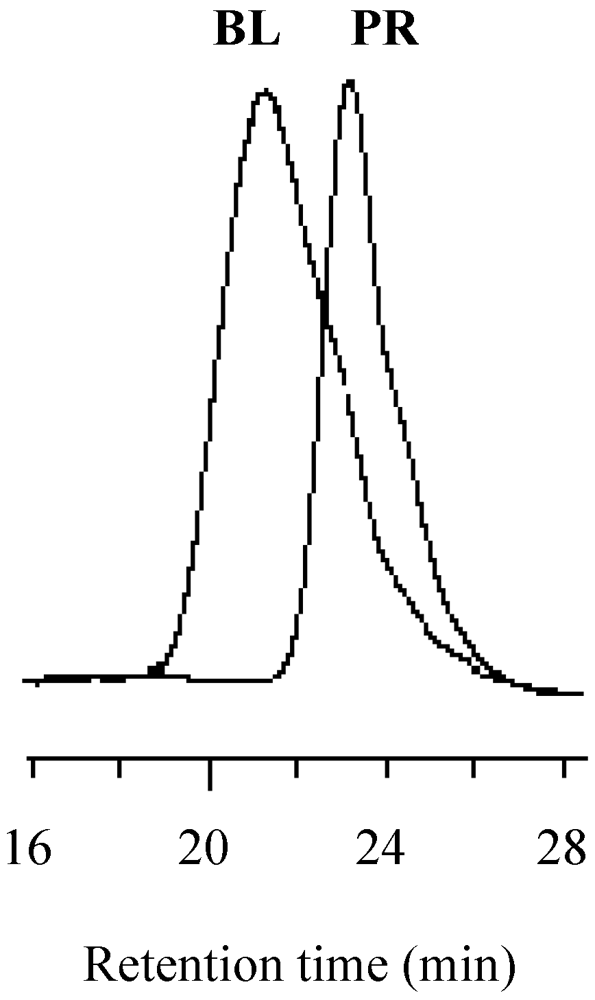
2.7. Block Copolymerization Using a TEMPO Macromediator
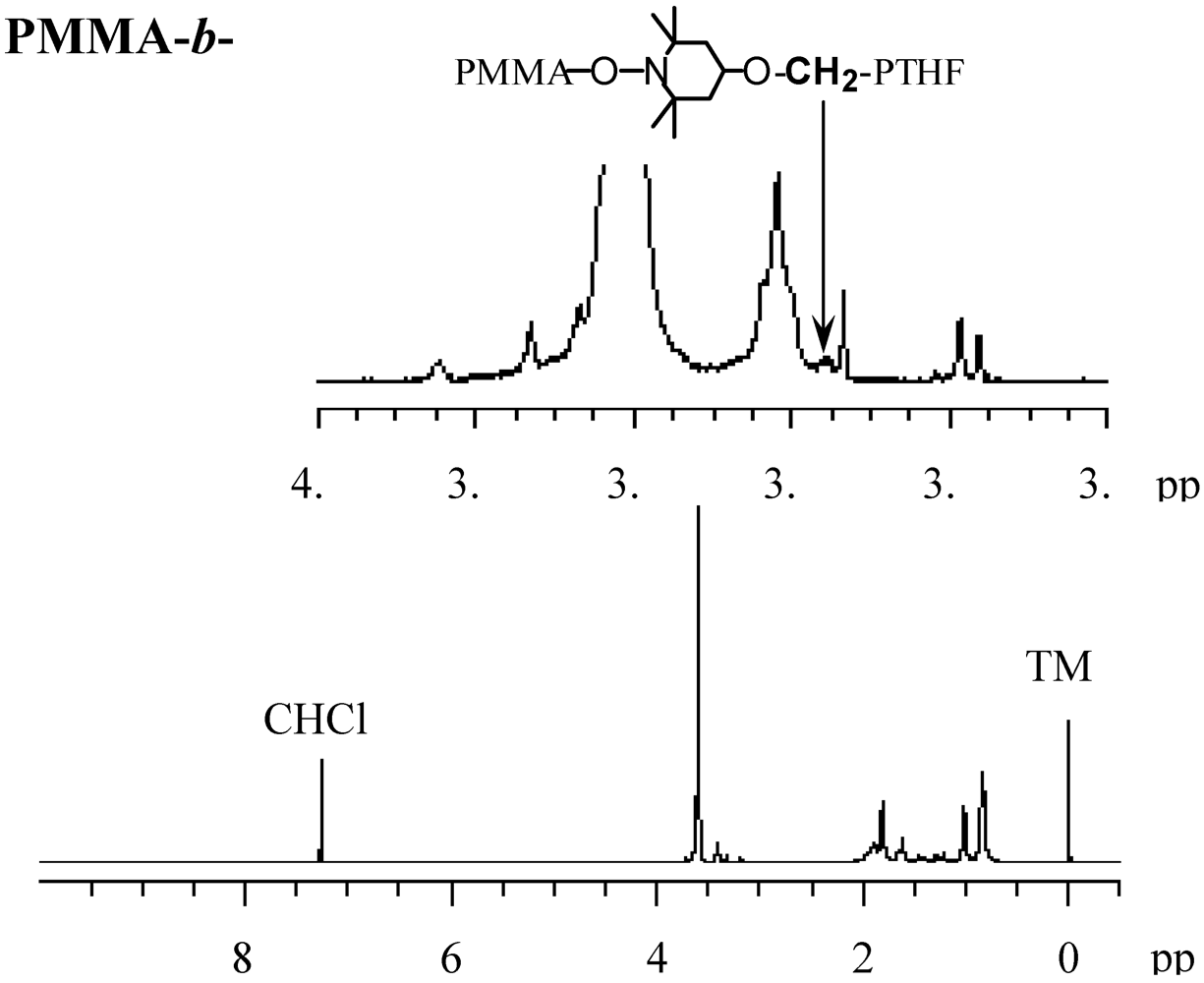
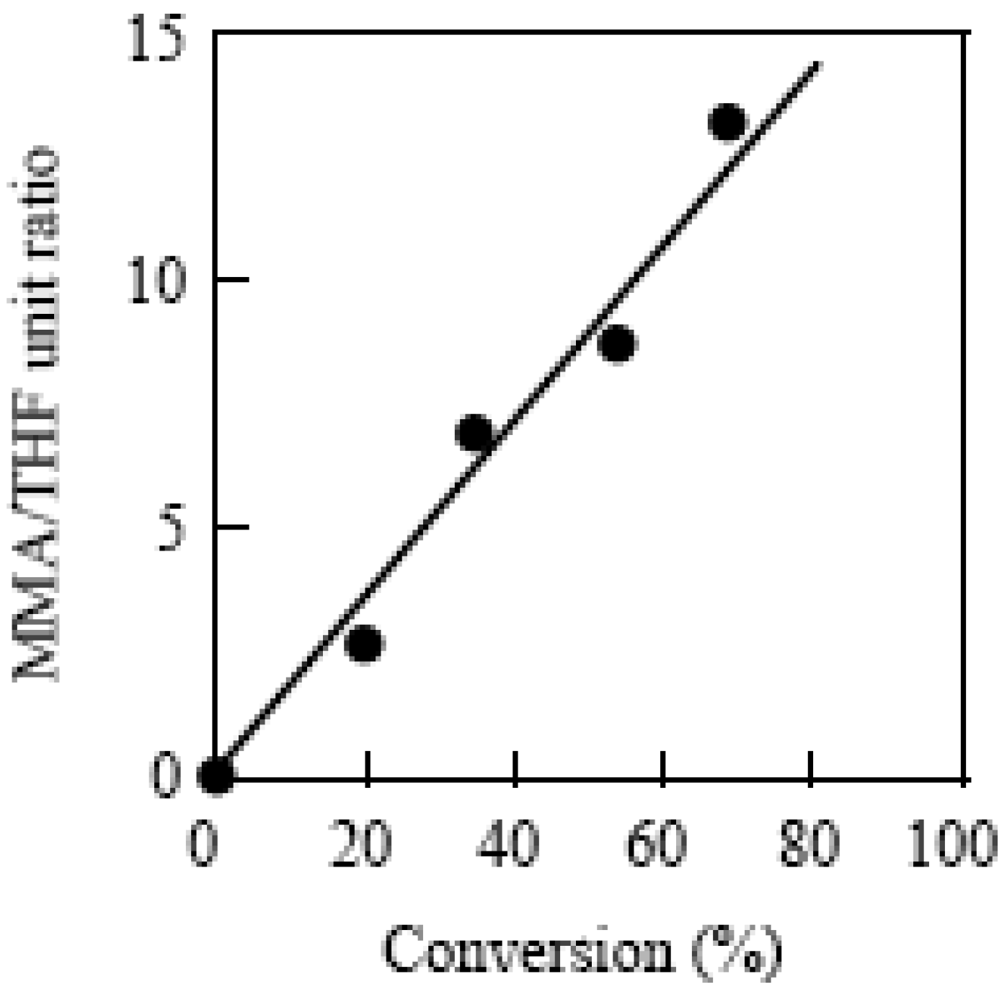
2.8. Photo Dispersion Polymerization

| r-AMDV (μmol) | MTEMPO (μmol) | tBuS (μmol) | PVP (wt%) | Conv. (%) | Mn a | Mw/Mn a | Morphology b | Dn b (μm) | Dw/Dn b |
|---|---|---|---|---|---|---|---|---|---|
| 45.4 | – | – | 45 | 70 | 78,900 | 3.26 | nonspecific | – | – |
| 45.4 | – | 23.5 | 45 | 34 | 57,600 | 3.64 | nonspecific | – | – |
| – | – | 23.5 | 45 | 69 | 175,000 | 2.50 | nonspecific | – | – |
| 45.4 | 48.3 | 23.5 | 30 | 20 | 18,200 | 1.62 | spherical | 3.87 | 2.80 |
| 45.4 | 48.3 | 23.5 | 45 | 23 | 17,700 | 1.61 | spherical | 5.66 | 1.27 |
| 45.4 | 48.3 | 23.5 | 60 | 37 | 20,500 | 1.65 | spherical | 3.99 | 1.06 |
| 45.4 | 48.3 | 23.5 | 75 | 28 | 18,000 | 1.64 | spherical | 4.14 | 1.04 |

3. Conclusions
4. Experimental Section
References
- Odian, G. Radical chain polymerization. In Principles of Polymerization, 4th ed; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2004. [Google Scholar]
- Otsu, T. Organic polysulfides as polymerization initiators. J. Polym. Sci. 1956, 21, 559–561. [Google Scholar]
- Moad, G.; Rizzardo, E.; Thang, S.H. Toward living radical polymerization. Accounts Chem. Res. 2008, 41, 1133–1142. [Google Scholar]
- Braunecker, W.A.; Matyjaszewski, K. Controlled/living radical polymerization: Features, developments, and perspectives. Prog. Polym. Sci. 2007, 32, 93–146. [Google Scholar] [CrossRef]
- Uegaki, H.; Kotani, Y.; Kamigaito, M.; Sawamoto, M. Nickel-mediated living radical polymerization of methyl methacrylate. Macromolecules 1997, 30, 2249–2253. [Google Scholar]
- Wayland, B.B.; Poszmik, G.; Mukerjee, S.L. Living radical polymerization of acrylates by organocobalt porphyrin complexes. J. Am. Chem. Soc. 1994, 116, 7943–7944. [Google Scholar]
- Ando, T.; Kamigaito, M.; Sawamoto, M.; Higashimura, T. Iron(II) chloride complex for living radical polymerization of methyl methacrylate. Macromolecules 1995, 30, 4507–4510. [Google Scholar]
- Kato, M.; Kamigaito, M.; Sawamoto, M.; Higashimura, T. Polymerization of methyl methacrylate with the carbon tetrachloride/dichlorotris-(triphenylphosphine)ruthenium(II)/methylaluminum bis(2,6-di-tert-butylphenoxide) initiating system: Possibility of living radical polymerization. Macromolecules 1995, 28, 1721–1723. [Google Scholar] [CrossRef]
- Percec, V.; Barboiu, B.; Neumann, A.; Ronda, J.C.; Zhao, M. Metal-catalyzed “living” radical polymerization of styrene initiated with arenesulfonyl chlorides. From heterogeneous to homogeneous catalysis. Macromolecules 1996, 29, 3665–3668. [Google Scholar] [CrossRef]
- Lecmote, P.; Drapier, I.; Dubois, P.; Teyssie, P.; Jerome, R. Controlled radical polymerization of methyl methacrylate in the presence of palladium acetate, triphenylphosphine, and carbon tetrachloride. Macromolecules 1997, 30, 7631–7633. [Google Scholar] [CrossRef]
- Kotani, Y.; Kamigaito, M.; Sawamoto, M. Re(V)-mediated living radical polymerization of styrene: ReO2I(PPh3)2/R−I initiating systems. Macromolecules 1999, 32, 2420–2424. [Google Scholar] [CrossRef]
- Oka, M.; Tatemoto, M. Vinylidene fluoride–Hexafluoropropylene copolymer having terminal iodines. In Contemporary Topics in Polymer Science; Plenum Press: New York, NY, USA, 1984. [Google Scholar]
- Lacroix-Desmazes, P.; Severac, R.; Boutevin, B. Reverse iodine transfer polymerization of methyl acrylate and n-butyl acrylate. Macromolecules 2005, 38, 6299–6309. [Google Scholar] [CrossRef]
- Pouget, E.; Tonnar, J.; Eloy, C.; Lacroix-Desmazes, P.; Boutevin, B. Synthesis of poly(styrene)-b-poly(dimethylsiloxane)-b-poly(styrene) triblock copolymers by iodine transfer polymerization in miniemulsion. Macromolecules 2006, 39, 6009–6016. [Google Scholar] [CrossRef]
- Hawker, C.J.; Bosman, A.W.; Harth, E. New polymer synthesis by nitroxide mediated living radical polymerizations. Chem. Rev. 2001, 101, 3661–3688. [Google Scholar]
- Sciannamea, V.; Jérôme, R.; Detrembleur, C. In situ nitroxide-mediated radical polymerization (NMP) processes: Their understanding and optimization. Chem. Rev. 2008, 108, 1104–1126. [Google Scholar] [CrossRef]
- Yousi, Z.; Jian, L.; Rongchuan, Z.; Jianliang, Y.; Lizong, D.; Lansun, Z. Synthesis of block copolymer from dissimilar vinyl monomer by stable free radical polymerization. Macromolecules 2000, 33, 4745–4749. [Google Scholar]
- Devonport, W.; Michalak, L.; Malmstrom, E.; Mate, M.; Kurdi, B.; Hawker, C.J.; Barclay, G.G.; Sinta, R. “Living” free-radical pin the absence of initiators: Controlled autopolymerization. Macromolecules 1997, 30, 1929–1934. [Google Scholar]
- Mardare, D.; Matyjaszewski, K. “Living” radical polymerization of vinyl acetate. Macromolecules 1994, 27, 645–649. [Google Scholar] [CrossRef]
- Granel, C.; Jerime, R.; Teyssie, P.; Jasieczek, C.B.; Shooter, A.J.; Haddleton, D.M.; Hastings, J.J.; Gigmes, D.; Grimaldi, S.; Tordo, P.; Greszta, D.; Matyjaszewski, K. Investigation of methyl methacrylate and vinyl acetate polymerization promoted by Al(iBu)3/2,2’-bipyridine and Al(iBu)3/2,2’-bipyridine/TEMPO complexes. Macromolecules 1998, 31, 7133–7141. [Google Scholar]
- Matyjaszewski, K.; Gaynor, S.; Greszta, D.; Mardare, D.; Shigemoto, T. “Living” and controlled radical polymerization. J. Phys. Org. Chem. 1995, 8, 306–315. [Google Scholar] [CrossRef]
- Hawker, C.J.; Barclay, G.G.; Orellana, A.; Dao, J.; Devonport, W. Initiating systems for nitroxide-mediated “living” free radical polymerizations: Synthesis and evaluation. Macromolrcules 1996, 29, 5245–5254. [Google Scholar]
- Dao, J.; Benoit, D.; Hawker, C.J. A versatile and efficient synthesis of alkoxyamine LFR initiators via manganese based asymmetric epoxidation catalysts. J. Polym. Sci. A Polym. Chem. 1998, 36, 2161–2167. [Google Scholar] [CrossRef]
- Banoit, D.; Chaplinski, V.; Braslau, R.; Hawker, C.J. Development of a universal alkoxyamine for “living” free radical polymerizations. J. Am. Chem. Soc. 1999, 121, 3904–3920. [Google Scholar]
- Veregin, R.P.N.; Georges, M.K.; Hamer, G.K.; Kazmaier, P.M. mechanism of living free radical polymerizations with narrow polydispersity: Electron spin resonance and kinetic studies. Macromolecules 1995, 28, 4391–4398. [Google Scholar] [CrossRef]
- Greszta, D.; Matyjaszewski, K. Comments on the paper “living radical polymerization: Kinetic results”. Macromolecules 1996, 29, 5239–5240. [Google Scholar]
- Braslau, R.; Burrill, L.C., II; Siano, M.; Naik, N.; Howden, R.K.; Mahal, L.K. Low-temperature preparations of unimolecular nitroxide initiators for “living” free radical polymerizations. Macromolecules 1997, 30, 6445–6450. [Google Scholar]
- Li, I.Q.; Howell, B.A.; Koster, R.A.; Priddy, D.B. Mono- and dinitroxide styrene polymerization initiators. Macromolecules 1996, 29, 8554–8555. [Google Scholar]
- Mercier, C.L.; Acerbis, S.; Bertin, D.; Chauvin, F.; Gigmes, D.; Guerret, O.; Lansalot, M.; Marque, S.; Moigne, F.L.; Fischer, H.; Tordo, P. Design and use of β-phosphorus nitroxides and alkoxyamines in controlled/”living” free radical polymerizations. Macromol. Symposia 2002, 182, 225–247. [Google Scholar]
- Yoshida, E.; Ishizone, T.; Hirao, A.; Nakahama, S.; Takata, T.; Endo, T. Synthesis of polystyrene having an aminoxy terminal by the reactions of living polystyrene with an oxoaminium salt and with the corresponding nitroxyl radical. Macromolecules 1994, 27, 3119–3124. [Google Scholar]
- Yoshida, E.; Nakamura, K.; Takata, T.; Endo, T. Oxoaminium salts as initiators for cationic polymerization of vinyl monomers. J. Polym. Sci. A Polym. Chem. 1993, 31, 1505–1512. [Google Scholar]
- Pyun, J.; Tang, C.; Kowalewski, T.; Fréchet, J.M.J.; Hawker, C.J. Synthesis and direct visualization of block copolymers composed of different macromolecular architectures. Macromolecules 2005, 38, 2674–2685. [Google Scholar]
- Hawker, C.J.; Hedrick, J.L. Accurate control of chain ends by a novel “living” free-radical polymerization process. Macromolecules 1995, 28, 2993–2995. [Google Scholar]
- Hawker, C.J. Architectural Control in “Living” free radical polymerizations: Preparation of star and graft polymers. Angew. Chem. Int. Ed. Eng. 1995, l34, 1456–1459. [Google Scholar]
- Tsoukatos, T.; Pispas, S.; Hadjichristidis, N. Complex macromolecular architectures by combining TEMPO living free radical and anionic polymerization. Macromolecules 2000, 33, 9504–9511. [Google Scholar]
- Yoshida, E. Photo-living radical polymerization of methyl methacrylate by a nitroxide mediator. Colloid Polym. Sci. 2008, 286, 1663–1666. [Google Scholar]
- Yoshida, E. Photo-living radical polymerization of methyl methacrylate by 2,2,6,6-tetramethylpiperidine-1-oxyl in the presence of a photo-acid generator. Colloid Polym. Sci. 2009, 287, 767–772. [Google Scholar]
- Yoshida, E. Synthesis of poly(methyl methacrylate)-block-poly(tetrahydrofuran) by photo-living radical polymerization using a 2,2,6,6-tetramethylpiperidine-1-oxyl macromediator. Colloid Polym. Sci. 2009, 287, 1417–1424. [Google Scholar] [CrossRef]
- Yoshida, E. Photo-living radical polymerization of methyl methacrylate using alkoxyamine as an initiator. Colloid Polym. Sci. 2010, 288, 7–13. [Google Scholar]
- Yoshida, E. Nitroxide-mediated photo-living radical polymerization of vinyl acetate. Colloid Polym. Sci. 2010, 288, 73–78. [Google Scholar]
- Yoshida, E. Nitroxide-mediated photo-living radical polymerization of methyl methacrylate using (4-tert-butylphenyl)diphenylsulfonium triflate as a photo-acid generator. Colloid Polym. Sci. 2010, 288, 239–243. [Google Scholar] [CrossRef]
- Yoshida, E. Effect of azoinitiators on nitroxide-mediated photo-living radical polymerization of methyl methacrylate. Colloid Polym. Sci. 2010, 288, 341–345. [Google Scholar]
- Yoshida, E. Effects of initiators and photo-acid generators on nitroxide-mediated photo-living radical polymerization of methyl methacrylate. Colloid Polym. Sci. 2010, 288, 901–905. [Google Scholar]
- Yoshida, E. Stability of growing polymer chain ends for nitroxide-mediated photo-living radical polymerization. Colloid Polym. Sci. 2010, 288, 1027–1030. [Google Scholar]
- Yoshida, E. Nitroxide-mediated photo-living radical polymerization of methyl methacrylate in solution. Colloid Polym. Sci. 2010, 288, 1639–1643. [Google Scholar]
- Yoshida, E. Nitroxide-mediated photo-living radical polymerization of methyl methacrylate in the presence of (η6-benzene)(η5-cyclopentadienyl)FeII hexafluorophosphate. Colloid Polym. Sci. 2010, 288, 1745–1749. [Google Scholar]
- Yoshida, E. Graft copolymerization of methyl methacrylate on polystyrene backbone through nitroxide-mediated photo-living radical polymerization. Colloid Polym. Sci. 2011, 289, 837–841. [Google Scholar]
- Yoshida, E. Nitroxide-mediated photo-controlled/living radical polymerization of ethyl acrylate. Colloid Polym. Sci. 2011, 289, 1127–1132. [Google Scholar]
- Yoshida, E. Nitroxide-mediated photo-controlled/living radical dispersion polymerization of methyl methacrylate. Colloid Polym. Sci. 2011, 289, 1625–1630. [Google Scholar]
- Guillaneuf, Y.; Bertin, D.; Gigmes, D.; Versace, D.; Lalevee, J.; Fouassier, J. Toward nitroxide-mediated photopolymerization. Macromolecules 2010, 43, 2204–2212. [Google Scholar]
- Goto, A.; Scaiano, J.C.; Maretti, L. Photolysis of an alkoxyamine using intramolecular energy transfer from a quinoline antenna—Towards photo-induced living radical polymerization. Photochem. Photobiol. Sci. 2007, 6, 833–853. [Google Scholar]
- Lalevee, J.; Allonas, X.; Fouassier, J.P. A new efficient photoiniferter for living radical photopolymerization. Macromolecules 2006, 39, 8216–8218. [Google Scholar]
- Nemoto, Y.; Nakayama, Y. Optimal irradiation wavelength in iniferter-based photocontrolled radical polymerization. J Polym. Sci. A Polym. Chem. 2008, 46, 4505–4512. [Google Scholar]
- You, Y.; Hong, C.; Bai, R.; Pan, C.; Wang, J. Photo-initiated living free radical polymerization in the presence of dibenzyl trithiocarbonate. Macromol. Chem. Phys. 2002, 203, 477–483. [Google Scholar]
- Ran, R.; Yu, Y.; Wan, T. Photoinitiated RAFT polymerization in the presence of trithiocarbonate. J. Appl. Polym. Sci. 2007, 105, 398–404. [Google Scholar]
- Baruah, S.R.; Kakati, D.K. Photopolymerization of methyl methacrylate by 2,2'-dithiodiethanol: Effect of reaction conditions. J. Appl. Polym. Sci. 2006, 100, 1601–1606. [Google Scholar]
- Ajayaghosh, A.; Francis, R. A xanthate-derived photoinitiator that recognizes and controls the free radical polymerization pathways of methyl methacrylate and styrene. J. Am. Chem. Soc. 1999, 121, 6599–6606. [Google Scholar]
- Yang, W.; Ranby, B. Radical living graft polymerization on the surface of polymeric materials. Macromolecules 1996, 29, 3308–3310. [Google Scholar]
- Tasdelen, M.A.; Durmaz, Y.Y.; Karagoz, B.; Bicak, N.; Yagci, Y. A new photoiniferter/RAFT agent for ambient temperature rapid and well-controlled radical polymerization. J. Polym. Sci. A Polym. Chem. 2008, 46, 3387–3395. [Google Scholar]
- Ajayaghosh, A.; Francis, R. Narrow polydispersed reactive polymers by a photoinitiated free radical polymerization approach. Controlled polymerization of methyl methacrylate. Macromolecules 1998, 31, 1436–1438. [Google Scholar] [CrossRef]
- Yin, H.; Zheng, H.; Lu, L.; Liu, P.; Cai, Y. Highly efficient and well-controlled ambient temperature RAFT polymerization of glycidyl methacrylate under visible light radiation. J. Polym. Sci. A Polym. Chem. 2007, 45, 5091–5102. [Google Scholar]
- Cohen, N.A.; Tillman, E.S.; Thakur, S.; Smith, J.R.; Eckenhoff, W.T.; Pintauer, T. Effect of the ligand in atom transfer radical polymerization reactions initiated by photodimers of 9-bromoanthracene. Macromol. Chem. Phys. 2009, 210, 263–268. [Google Scholar]
- Li, P.; Qiu, K. Cu(S2CNEt2)Cl-catalyzed reverse atom-transfer radical polymerization of vinyl monomers. Macromol. Rapid Commun. 2002, 23, 1124–1129. [Google Scholar]
- Yoshida, E.; Okada, Y. Living radical polymerization of styrene in the presence of 4-hydroxy-2,2,6,6-tetramethylpiperidine-1-oxyl, and radical transformation of the resulting polymers by other radicals. Bull. Chem. Soc. Jpn. 1997, 70, 275–281. [Google Scholar]
- McHale, R.; Aldabbagh, F.; Zetterlund, P.B. The role of excess nitroxide in the SG1 (N-tert-butyl-N-[1-diethlphosphono-(2,2-dimethylpropyl)] nitroxide)-mediated polymerization of methyl methacrylate. J. Polym. Sci. A Polym. Chem. 1999, 45, 2194–2203. [Google Scholar]
- Bovey, F.A.; Tiers, G.V.D. Polymer NSR spectroscopy. II. The high resolution spectra of methyl methacrylate polymers prepared with free radical and anionic initiators. J. Polym. Sci. 1960, 44, 173–182. [Google Scholar] [CrossRef]
- Kita, Y.; Gotanda, K.; Murata, K.; Suemura, M.; Sano, A.; Yamaguchi, T.; Oka, M.; Matsugi, M. Practical radical additions under mild conditions using 2,2'-azobis(2,4-dimethyl-4-methoxyvaleronitrile) [V-70] as an initiator. Org. Process Res. Dev. 1998, 2, 250–254. [Google Scholar]
- Crivello, J.V.; Sangermano, M. Visible and long-wavelength photoinitiated cationic polymerization. J. Polym. Sci. A Polym. Chem. 2001, 39, 343–356. [Google Scholar]
- Crivello, J.V. The discovery and development of onium salt cationic photoinitiators. J. Polym. Sci. A Polym. Chem. 1999, 37, 4241–4254. [Google Scholar]
- Devoe, R.J.; Sahyunn, M.R.V.; Sepone, D.; Sharmac, D.K. Transient intermediates in the photolysis of iodonium cations. Can. J. Chem. 1987, 65, 2342–2349. [Google Scholar]
- Crivello, J.V.; Lam, J.H. W.; Moore, J.E.; Schroeter, S.H. Triarylsulfonium salts: A new class of photoinitiators for cationic polymerization. J. Radiat. Curing 1978, 5, 2–17. [Google Scholar]
- Crivello, J.V.; Lam, J.H.W. Complex triarylsulfonium salt photoinitiators. I. The identification, characterization, and syntheses of a new class of triarylsulfonium salt photoinitiators. J. Polym. Sci. Polym. Chem. Ed. 1980, 18, 2677–2695. [Google Scholar] [CrossRef]
- Crivello, J.V.; Lam, J.H.W. Photoinitiated cationic polymerization with triarylsulfonium salts. J. Polym. Sci. Polym. Chem. Ed. 1979, 17, 977–999. [Google Scholar]
- Liu, Y.C.; Wu, L.M.; Chen, P. A facile generation of radical cations via the action of nitroxides. Tetrahedron Lett. 1985, 26, 4201–4202. [Google Scholar]
- Meier, K.; Zweifel, H. Photoinitiated cationic polymerization of epoxides with iron-arene complexes. J. Radiat. Curing 1986, 13, 26–32. [Google Scholar]
- Li, I.; Howell, B.A.; Matyjaszewski, K.; Shigemoto, T.; Smith, P.B.; Priddy, D.B. Kinetics of decomposition of 2,2,6,6-tetramethyl-1-(1-phenylethoxy)piperidine and its implications on nitroxyl-mediated styrene polymerization. Macromolecules 1995, 28, 6692–6693. [Google Scholar] [CrossRef]
- Yoshida, E.; Sugita, A. Synthesis of poly(styrene-b-tetrahydrofuran-b-styrene) triblock copolymers by transformation from living cationic into living radical polymerization using 4-hydroxy-2,2,6,6-tetramethylpiperidine-1-oxyl as a transforming agent. J. Polym. Sci. Polym. Chem. Ed. 1998, 36, 2059–2068. [Google Scholar]
- Yoshida, E.; Sugita, A. Synthesis of poly(tetrahydrofuran) with a nitroxyl radical at the chain end, and its application to living radical polymerization. Macromolecules 1996, 29, 6422–6426. [Google Scholar]
- Zetterlund, P.B.; Kagawa, Y.; Okubo, M. Controlled/living radical polymerization in dispersed systems. Chem. Rev. 2008, 108, 3747–3794. [Google Scholar]
- Miyazawa, T.; Endo, T.; Shiihashi, S.; Ogawara, M. Selective oxidation of alcohols by oxoaminium salts (R2N:O+ X-). J. Org. Chem. 1985, 50, 1332–1334. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yoshida, E. Controlled Photoradical Polymerization Mediated by 2,2,6,6-Tetramethylpiperidine-1-Oxyl. Polymers 2012, 4, 1125-1156. https://doi.org/10.3390/polym4021125
Yoshida E. Controlled Photoradical Polymerization Mediated by 2,2,6,6-Tetramethylpiperidine-1-Oxyl. Polymers. 2012; 4(2):1125-1156. https://doi.org/10.3390/polym4021125
Chicago/Turabian StyleYoshida, Eri. 2012. "Controlled Photoradical Polymerization Mediated by 2,2,6,6-Tetramethylpiperidine-1-Oxyl" Polymers 4, no. 2: 1125-1156. https://doi.org/10.3390/polym4021125





