Human Defensins: Potential Tools for Clinical Applications
Abstract
:1. Introduction
2. Antimicrobial Activity

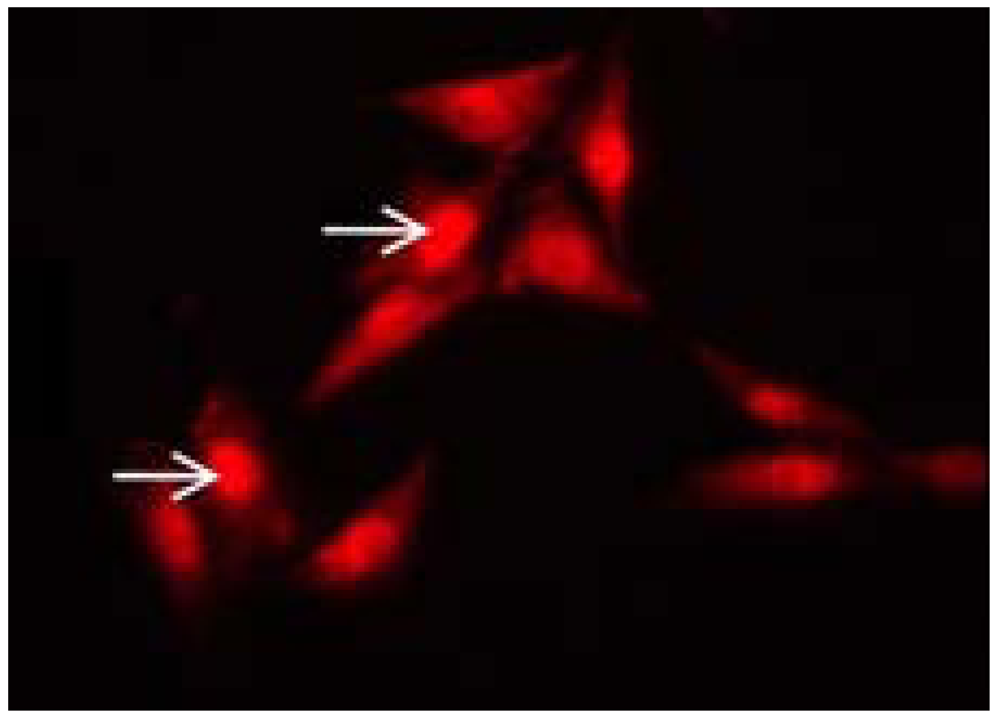
3. Immunomodulatory and Chemotactic Effects
3. Tissue Reorganization and Wound Healing
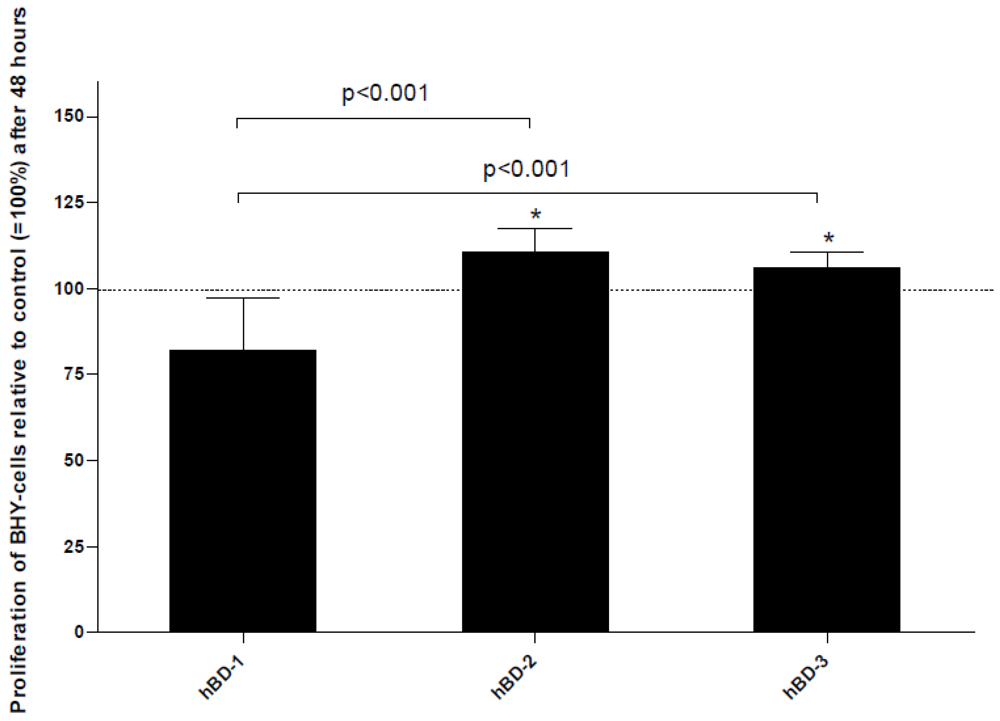
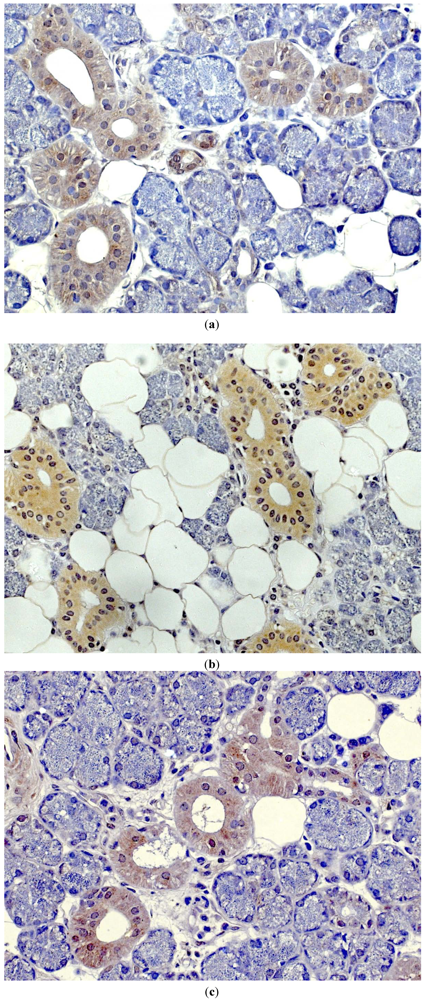
4. Cancer
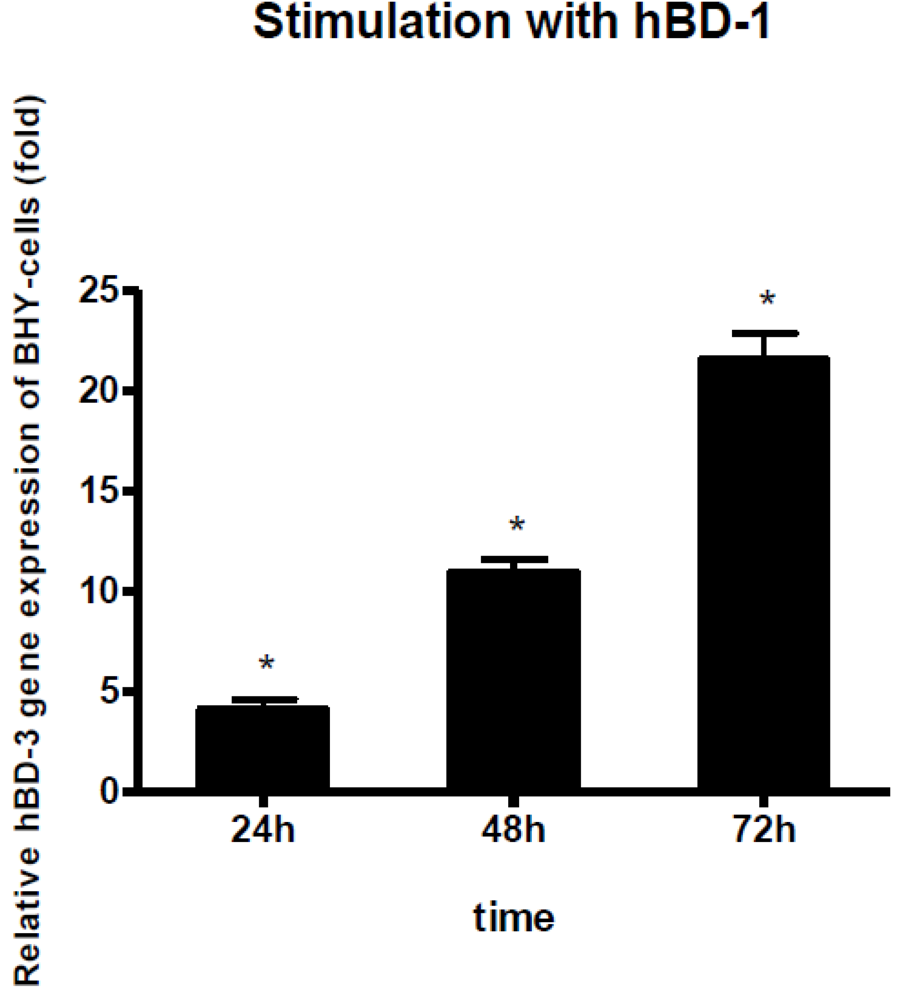
5. Concluding Remarks
Acknowledgements
References
- Ganz, T. Defensins: Antimicrobial peptides of innate immunity. Nat. Rev. Immunol. 2003, 3, 710–720. [Google Scholar] [CrossRef]
- Lehrer, R.I. Primate defensins. Nat. Rev. Microbiol. 2004, 2, 727–738. [Google Scholar]
- Brogden, K.A. Antimicrobial peptides: Pore formers or metabolic inhibitors in bacteria? Nat. Rev. Microbiol. 2005, 3, 238–250. [Google Scholar] [CrossRef]
- Hazlett, L.; Wu, M. Defensins in innate immunity. Cell Tissue Res. 2011, 343, 175–188. [Google Scholar] [CrossRef]
- Mathews, M.; Jia, H.P.; Guthmiller, J.M.; Losh, G.; Graham, S.; Johnson, G.K.; Tack, B.F.; McCray, P.B. Production of β-defensin antimicrobial peptides by the oral mucosa and salivary glands. Infect. Immun. 1999, 67, 2740–2745. [Google Scholar]
- Abiko, Y.; Saitoh, M.; Nishimura, M.; Yamazaki, M.; Sawamura, D.; Kaku, T. Role of β-defensins in oral epithelial health and disease. Med. Mol. Morphol. 2007, 40, 179–184. [Google Scholar] [CrossRef]
- Wenghoefer, M.; Pantelis, A.; Dommisch, H.; Götz, W.; Reich, R.; Bergé, S.; Martini, M.; Allam, J.P.; Jepsen, S.; Merkelbach-Bruse, S.; Fischer, HP.; Novak, N.; Winter, J. Nuclear hBD-1 accumulation in malignant salivary gland tumors. BMC Cancer 2008, 8. [Google Scholar]
- Pantelis, A.; Wenghoefer, M.; Haas, S.; Merkelbach-Bruse, S.; Pantelis, D.; Jepsen, S.; Bootz, F.; Winter, J. Down regulation and nuclear localization of human beta-defensin-1 in pleomorphic adenomas of salivary glands. Oral Oncol. 2009, 45, 526–530. [Google Scholar] [CrossRef]
- Warnke, P.H.; Springer, I.N.; Russo, P.A.J.; Wiltfang, J.; Essig, H.; Kosmahl, M.; Sherry, E.; Acil, Y. Innate immunity in human bone. Bone 2006, 38, 400–408. [Google Scholar]
- Bensch, K.W.; Raida, M.; Mägert, H.J.; Schulz-Knappe, P.; Forssmann, W.G. hBD-1: A novel beta-defensin from human plasma. FEBS Lett. 1995, 368, 331–335. [Google Scholar] [CrossRef]
- Harder, J.; Bartels, J.; Christophers, E.; Schröder, J.M. A peptide antibiotic from human skin. Nature 1997, 387, 861. [Google Scholar]
- Harder, J.; Bartels, J.; Christophers, E.; Schröder, J.M. Isolation and characterization of human β-defensin-3, a novel human inducible peptide antibiotic. J. Biol. Chem. 2001, 276, 5707–5713. [Google Scholar]
- Stockmann, P.; Wehrhan, F.; Schwarz-Furlan, S.; Stelzle, F.; Trabert, S.; Neukam, F.W.; Nkenke, E. Increased human defensine levels hint at an inflammatory etiology of bisphosphonate-associated osteonecrosis of the jaw: An immunohistological study. J. Transl. Med. 2011, 9. [Google Scholar]
- Presicce, P.; Giannelli, S.; Taddeo, A.; Villa, M.L.; Della Bella, S. Human defensins activate monocyte-derived dendritic cells, promote the production of proinflammatory cytokines, and up-regulate the surface expression of CD91. J. Leukoc. Biol. 2009, 86, 941–948. [Google Scholar] [CrossRef]
- Kraus, D.; Deschner, J.; Jäger, A.; Wenghoefer, M.; Bayer, S.; Jepsen, S.; Allam, J.; Novak, N.; Meyer, R.; Winter, J. Human β-defensins differently affect proliferation, differentiation, and mineralization of osteoblast-like MG63 cells. J. Cell. Physiol. 2011. [Google Scholar]
- Winter, J.; Pantelis, A.; Reich, R.; Martini, M.; Kraus, D.; Jepsen, S.; Allam, J.P.; Novak, N.; Wenghoefer, M. Human beta-defensin-1, -2, and -3 exhibit opposite effects on oral squamous cell carcinoma cell proliferation. Cancer Invest. 2011, 29, 196–201. [Google Scholar]
- Yang, D.; Chertov, O.; Bykovskaia, S.N.; Chen, Q.; Buffo, M.J.; Shogan, J.; Anderson, M.; Schröder, J.M.; Wang, J.M.; Howard, O.M.; Oppenheim, J.J. Beta-defensins: Linking innate and adaptive immunity through dendritic and T cell CCR6. Science 1999, 286, 525–528. [Google Scholar]
- Biragyn, A.; Ruffini, P.A.; Leifer, C.A.; Klyushnenkova, E.; Shakhov, A.; Chertov, O.; Shirakawa, A.K.; Farber, J.M.; Segal, D.M.; Oppenheim, J.J.; Kwak, L.W. Toll-like receptor 4-dependent activation of dendritic cells by beta-defensin 2. Science 2002, 298, 1025–1029. [Google Scholar]
- Aarbiou, J.; Verhoosel, R.M.; van Wetering, S.; De Boer, W.I.; van Krieken, J.H.; Litvinov, S.V.; Rabe, K.F.; Hiemstra, P.S. Neutrophil defensins enhance lung epithelial wound closure and mucin gene expression in vitro. Am. J. Respir. Cell Mol. Biol. 2004, 30, 193–201. [Google Scholar]
- Niyonsaba, F.; Ushio, H.; Nakano, N.; Ng, W.; Sayama, K.; Hashimoto, K.; Nagaoka, I.; Okumura, K.; Ogawa, H. Antimicrobial peptides human beta-defensins stimulate epidermal keratinocyte migration, proliferation and production of proinflammatory cytokines and chemokines. J. Invest. Dermatol. 2007, 127, 594–604. [Google Scholar]
- Tollner, T.L.; Venners, S.A.; Hollox, E.J.; Yudin, A.I.; Liu, X.; Tang, G.; Xing, H.; Kays, R.J.; Lau, T.; Overstreet, J.W.; Xu, X.; Bevins, C.L.; Cherr, G.N. A common mutation in the defensin DEFB126 causes impaired sperm function and subfertility. Sci. Transl. Med. 2011, 3. [Google Scholar]
- Ganz, T.; Selsted, M.E.; Szklarek, D.; Harwig, S.S.L.; Daher, K.; Bainton, D.F.; Lehrer, R.I. Defensins. J. Clin. Invest. 1985, 76, 1427–1435. [Google Scholar]
- Selsted, M.E.; Miller, S.I.; Henschen, A.H.; Quellette, A.J. Enteric defensins: Antibiotic peptide components of intestinal host defense. J. Cell Biol. 1992, 118, 929–936. [Google Scholar]
- Jones, D.E.; Bevins, C.L. Defensin-6 mRNA in human Paneth cells: Implication for antimicrobial peptides in host defense of the human bowel. FEBS Lett. 1993, 315, 187–192. [Google Scholar]
- Ericksen, B.; Wu, Z.; Lu, W.; Lehrer, R.I. Antibacterial activity and specificity of the six human alpha-defensins. Antimicrob. Agents Chemother. 2005, 49, 269–275. [Google Scholar] [CrossRef]
- Sharma, S.; Verma, I.; Khuller, GK. Antibacterial activity of human neutrophil peptide 1 against Mycobacterium tuberculosis H37Rv: In vitro and ex vivo study. Eur. Respir. J. 2000, 16, 112–117. [Google Scholar]
- Sharma, S.; Verma, I.; Khuller, GK. Therapeutic potential of human neutrophil peptide 1 against experimental tuberculosis. Antimicrob. Agents Chemother. 2001, 45, 639–640. [Google Scholar] [CrossRef]
- Verma, C.; Seebah, S.; Low, S.M.; Zhou, L.; Liu, S.P.; Li, J.; Beuerman, R.W. Defensins: Antimicrobial peptides for therapeutic development. Biotechnol. J. 2007, 2, 1353–1359. [Google Scholar]
- Hazrati, E.; Galen, B.; Lu, W.; Wang, W.; Ouyang, Y.; Keller, M.J.; Lehrer, R.I.; Herold, B.C. Human alpha- and beta-defensins block multiple steps in herpes simplex virus infection. J. Immunol. 2006, 177, 8658–8666. [Google Scholar]
- Wang, W.; Owen, S.M.; Rudolph, D.L.; Cole, A.M.; Hong, T.; Waring, A.J.; Lal, R.B.; Lehrer, R.I. Activity of alpha- and theta-defensins against primary isolates of HIV-1. J. Immunol. 2004, 173, 515–520. [Google Scholar]
- Mackewicz, C.E.; Yuan, J.; Tran, P.; Diaz, L.; Mack, E.; Selsted, M.E.; Levy, J.A. Alpha-defensins can have anti-HIV activity but are not CD8 cell anti-HIV factors. AIDS 2003, 26, F23–F32, Erratum in: AIDS 2003, 26, F31.. [Google Scholar]
- Chang, T.L.; Vargas, J., Jr.; DelPortillo, A.; Klotman, M.E. Dual role of alpha-defensin-1 in anti-HIV-1 innate immunity. J. Clin. Invest. 2005, 115, 765–773. [Google Scholar]
- Yasin, B.; Wang, W.; Pang, M.; Cheshenko, N.; Hong, T.; Waring, A.J.; Herold, B.C.; Wagar, E.A.; Lehrer, R.I. Theta defensins protect cells from infection by herpes simplex virus by inhibiting viral adhesion and entry. J. Virol. 2004, 78, 5147–5156. [Google Scholar]
- McDermott, A.M. Defensins and other antimicrobial peptides at the ocular surface. Ocul. Surf. 2004, 2, 229–247. [Google Scholar]
- Valore, E.V.; Ganz, T. Posttranslational processing of defensins in immature human myeloid cells. Blood 1992, 79, 1538–1544. [Google Scholar]
- Schröder, J.M.; Harder, J. Human β-defensin-2. Int. J. Cell Biol. 1999, 31, 645–651. [Google Scholar] [CrossRef]
- García, J.R.; Krause, A.; Schulz, S.; Rodríguez-Jiménez, F.J.; Klüver, E.; Adermann, K.; Forssmann, U.; Frimpong-Boateng, A.; Bals, R.; Forssmann, W.G. Human beta-defensin 4: A novel inducible peptide with a specific salt-sensitive spectrum of antimicrobial activity. FASEB J. 2001, 15, 1819–1821. [Google Scholar]
- Feng, Z.; Jiang, B.; Chandra, J.; Ghannoum, M.; Nelson, S.; Weinberg, A. Human beta-defensins: Differential activity against Candidal species and regulation by Candida albicans. J. Dent. Res. 2005, 84, 445–450. [Google Scholar]
- Joly, S.; Organ, C.C.; Johnson, G.K.; McCray, P.B.; Guthmiller, J.M. Correlation between beta-defensin expression and induction profiles in gingival keratinocytes. Mol. Immunol. 2005, 42, 1073–1084. [Google Scholar]
- Quiñones-Mateu, M.E.; Lederman, M.M.; Feng, Z.; Chakraborty, B.; Weber, J.; Rangel, H.R.; Marotta, M.L.; Mirza, M.; Jiang, B.; Kiser, P.; Medvik, K.; Sieg, S.F.; Weinberg, A. Human epithelial beta-defensins 2 and 3 inhibit HIV-1 replication. AIDS 2003, 17, F39–F48. [Google Scholar]
- Sun, L.; Finnegan, C.M.; Kish-Catalone, T.; Blumenthal, R.; Garzino-Demo, P.; La Terra Maggiore, G.M.; Berrone, S.; Kleinman, C.; Wu, Z.; Abdelwahab, S.; Lu, W.; Garzino-Demo, A. Human beta-defensins suppress human immunodeficiency virus infection: Potential role in mucosal protection. J. Virol. 2005, 79, 14318–14329. [Google Scholar]
- Steinberg, D.A.; Hurst, M.A.; Fujii, C.A.; Kung, A.H.; Ho, J.F.; Cheng, C.; Loury, D.J.; Fiddess, J.C. Protegrin-1: A broad-spectrum, rapidly microbicidal peptide with in vivo activity. Antimicrob. Agents Chemother. 1997, 41, 1738–1742. [Google Scholar]
- Ge, Y.D.; MacDonald, D.L.; Holroyd, K.J.; Thornsberry, C.; Wexler, H.; Zasloff, M. In vitro antibacterial properties of pexiganan, an analog of magainin. Antimicrob. Agents Chemother. 1999, 43, 782–788. [Google Scholar]
- Zhang, L.; Parente, J.; Harris, S.M.; Woods, D.E.; Hancock, E.W.; Falla, T.J. Antimicrobial peptide therapeutics for cystic fibrosis. Antimicrob. Agents Chemother. 2005, 49, 2921–2927. [Google Scholar]
- Greenwald, G.I.; Ganz, T. Defensins mediate the microbicidal activity of human neutrophil granule extract against Acinetobacter calcoaceticus. Infect. Immun. 1987, 55, 1365–1368. [Google Scholar]
- Harder, J.; Meyer-Hoffert, U.; Teran, L.M.; Schwichtenberg, L.; Bartels, J.; Maune, S.; Schröder, J.M. Mucoid Pseudomonas aeroginosa, TNF-alpha, and IL-1beta, but not IL-6, induce human beta-defensin-2 in respiratory epithelia. Am. J. Respir. Cell Mol. Biol. 2000, 22, 714–721. [Google Scholar]
- Jia, H.P.; Schutte, B.C.; Schudy, A.; Linzmeier, R.; Guthmiller, J.M.; Johnson, G.K.; Tack, B.F.; Mitros, J.P.; Rosenthal, A.; Ganz, T. Discovery of new human b-defensins using a genomics-based approach. Gene 2001, 263, 211–218. [Google Scholar] [CrossRef]
- Duits, L.A.; Ravensbergen, B.; Rademaker, M.; Hiemstra, P.S.; Nibbering, P.H. Expression of beta-defensin 1 and 2 mRNA by human monocytes, macrophages and dendritic cells. Immunology 2002, 106, 517–525. [Google Scholar]
- Sumikawa, Y.; Asada, H.; Hoshino, K.; Azukizawa, H.; Katayama, I.; Akira, S.; Itami, S. Induction of beta-defensin 3 in keratinocytes stimulated by bacterial lipopeptides through toll-like receptor 2. Microbes Infect. 2006, 8, 1513–1521. [Google Scholar] [CrossRef]
- Chalifour, A.; Jeannin, P.; Gauchat, J.F.; Blaecke, A.; Malissard, M.; N’Guyen, T.; Thieblemont, N.; Delneste, Y. Direct bacterial protein PAMP recognition by human NK cells involves TLRs and triggers alpha-defensin production. Blood 2004, 104, 1778–1783. [Google Scholar]
- Wehkamp, J.; Harder, J.; Weichenthal, M.; Schwab, M.; Schäffeler, E.; Schlee, M.; Herrlinger, K.R.; Stallmach, A.; Noack, F.; Fritz, P.; Schröder, J.M.; Bevins, C.L.; Fellermann, K.; Stange, E.F. NOD2 (CARD15) mutations in Crohn’s disease are associated with diminished mucosal alpha-defensin expression. Gut 2004, 53, 1658–1664. [Google Scholar]
- Lu, Q.; Darveau, R.P.; Samaranayake, L.P.; Wang, C.Y.; Jin, L. Differential modulation of human beta-defensins expression in human gingival epithelia by Porphyromonas gingivalis lipopolysaccharide with tetra- and penta-acylated lipid A structures. Innate Immun. 2009, 15, 325–335. [Google Scholar] [CrossRef]
- Varoga, D.; Wruck, C.J.; Tohidnezhad, M.; Brandenburg, L.; Paulsen, F.; Mentlein, R.; Seekamp, A.; Besch, L.; Pufe, T. Osteoblasts participate in the innate immunity of the bone by producing human beta defensin-3. Histochem. Cell Biol. 2009, 131, 207–218. [Google Scholar] [CrossRef]
- Scott, M.G.; Vreugdenhil, A.C.; Buurman, W.A.; Hancock, R.E.; Gold, M.R. Cutting edge: Cationic antimicrobial peptides block the binding of lipopolysaccharide (LPS) to LPS binding protein. J. Immunol. 2000, 164, 549–553. [Google Scholar]
- Miles, K.; Clarke, D.J.; Lu, W.; Sibinska, Z.; Beaumont, P.E.; Davidson, D.J.; Barr, T.A.; Campopiano, D.J.; Gray, M. Dying and necrotic neutrophils are anti-inflammatory secondary to the release of alpha-defensins. J. Immunol. 2009, 183, 2122–2132. [Google Scholar]
- Soehnlein, O.; Kai-Larsen, Y.; Frithiof, R.; Sorensen, O.E.; Kenne, E.; Scharffetter-Kochanek, K.; Eriksson, E.E.; Herwald, H.; Agerberth, B.; Lindblom, L. Neutrophil primary granule proteins HBP and HNP1-3 boost bacterial phagocytosis by human and murine macrophages. J. Clin. Invest. 2008, 118, 3491–3502. [Google Scholar] [CrossRef]
- Nagaoka, I.; Niyonsaba, F.; Tsutsumi-Ishii, Y.; Tamura, H.; Hirata, M. Evaluation of the effect of human beta-defensins on neutrophil apoptosis. Int. Immunol. 2008, 20, 543–553. [Google Scholar]
- Oppenheim, J.J.; Yang, D. Alarmins: Chemotatctic activators of immune response. Curr. Opin. Immunol. 2005, 17, 359–365. [Google Scholar]
- Niyonsaba, F.; Iwabuchi, K.; Matsuda, H.; Ogawa, H.; Nagaoka, I. Epithelial cell-derived human beta-defensin-2 acts as a chemotaxin for mast cells through a pertussis toxin-sensitive and phospholipase C-dependent pathway. Int. Immunol. 2002, 14, 421–426. [Google Scholar] [CrossRef]
- Niyonsaba, F.; Ogawa, H.; Nagaoka, I. Human beta-defensin-2 functions as a chemotactic agent for tumor necrosis factor-alpha-treated human neutrophils. Immunology 2004, 111, 273–281. [Google Scholar] [CrossRef]
- Territo, M.C.; Ganz, T.; Selsted, M.E.; Lehrer, R.I. Monocyte-chemotactic activity of defensins from human neutrophils. J. Clin. Invest. 1989, 84, 2017–2020. [Google Scholar]
- Yang, D.; Chen, Q.; Chertov, O.; Oppenheim, J.J. Human neutrophil defensins selectively chemoattract naive T and immature dendritic cells. J. Leukoc. Biol. 2000, 68, 9–14. [Google Scholar]
- Yang, D.; Chertov, O.; Oppenheim, J.J. Participation of mammalian defensins and cathelicidins in anti-microbial immunity: Receptors and activities of human defensins and cathelicidin (LL-37). J. Leukoc. Biol. 2001, 69, 691–697. [Google Scholar]
- Rohrl, J.; Yand, D.; Oppenheim, J.J.; Hehlgans, T. Human β-defensins 2 and 3 and their mouse orthologs induce chemotaxis through interaction with CCR2. J. Immunol. 2010, 184, 6688–6694. [Google Scholar] [CrossRef]
- Funderburg, N.; Lederman, M.M.; Feng, Z.; Drage, M.G.; Jadlowsky, J.; Harding, C.V.; Weinberg, A.; Sieg, S.F. Human -defensin-3 activates professional antigen-presenting cells via Toll-like receptors 1 and 2. Proc. Natl. Acad. Sci. USA 2007, 104, 18631–18635. [Google Scholar]
- Bals, R.; Weiner, D.J.; Meegalla, R.L.; Wilson, J.M. Transfer of a cathelicidin peptide antibiotic gene restores bacterial killing in a cystic fibrosis xenograft model J. Clin. Invest. 1999, 103, 1113–1117. [Google Scholar] [CrossRef]
- Jacobsen, F.; Mittler, D.; Hirsch, T.; Gerhards, A.; Lehnhardt, M.; Voss, B.; Steinau, H.U.; Steinstraesser, L. Transient cutaneous adenoviral gene therapy with human host defense peptide hCAP-18/LL-37 is effective fort the treatment of burn wound infections. Gene Ther. 2005, 12, 1494–1502. [Google Scholar]
- Lai, Y.; Gallo, R.L. AMPed up immunity: How antimicrobial peptides have multiple roles in immune defense. Trends Immunol. 2009, 30, 131–141. [Google Scholar] [CrossRef]
- Han, W.; Wang, W.; Mohammed, K.A.; Su, Y. Defensins increase lung fibroblast proliferation and collagen synthesis via β-catenin signaling pathway. FEBS J. 2009, 276, 6603–6614. [Google Scholar] [CrossRef]
- Baroni, A.; Donnarumma, G.; Paoletti, I.; Longanesi-Cattani, I.; Bifulco, K.; Tufano, M.A.; Carriero, M.V. Antimicrobial human beta-defensin-2 stimulates migration, proliferation and tube formation of human umbilical vein endothelial cells. Peptides 2009, 30, 267–272. [Google Scholar]
- Bick, R.J.; Poindexter, B.J.; Buja, L.M.; Lawyer, C.H.; Milner, S.M.; Bhat, S. Nuclear lokalization of hBD-1 in human keratinocytes. J. Burn Wounds 2007, 7, 25–32. [Google Scholar]
- Prado-Montes de Orca, E. Human β-defensin 1: A restless warrior against allergies, infections and cancer. Int. J. Biochem. Cell Biol. 2010, 42, 800–804. [Google Scholar] [CrossRef]
- St John, M.A.; Li, Y.; Zhou, X.; Denny, P.; Ho, C.M.; Montemagno, C.; Shi, W.; Qi, F.; Wu, B.; Sinha, U.; Jordan, R.; Wolinsky, L.; Park, N.H.; Liu, H.; Abemayor, E.; Wong, D.T. Interleukin 6 and interleukin 8 as potential biomarkers for oral cavity and oropharyngeal squamous cell carcinoma. Arch. Otolaryngol. Head Neck Surg. 2004, 130, 929–935. [Google Scholar]
- Michiels, K.; Schutyser, E.; Conings, R.; Lenaerts, J.P.; Put, W.; Nuyts, S.; Delaere, P.; Jacobs, R.; Struyf, S.; Proost, P.; van Damme, J. Carcinoma cell-derived chemokines and their presence in oral fluid. Eur. J. Oral Sci. 2009, 117, 362–368. [Google Scholar] [CrossRef]
- Pitiyage, G.; Tilakaratne, W.M.; Tavassoli, M.; Warnakulasuriya, S. Molecular markers in oral epithelial dysplasia: Review. J. Oral Pathol. Med. 2009, 38, 737–752. [Google Scholar] [CrossRef]
- Porta, C.; Larghi, P.; Rimoldi, M.; Totaro, M.G.; Allavena, P.; Mantovani, A.; Sica, A. Cellular and molecular pathways linking inflammation and cancer. Immunobiology 2009, 214, 761–777. [Google Scholar] [CrossRef]
- Sica, A.; Allavena, P.; Mantovani, A. Cancer related inflammation: The macrophage connection. Cancer Lett. 2008, 267, 204–215. [Google Scholar] [CrossRef]
- Tezal, M.; Sullivan, M.A.; Reid, M.E.; Marshall, J.R.; Hyland, A.; Loree, T.; Lillis, C.; Hauck, L.; Wactawski-Wende, J.; Scannapieco, F.A. Chronic periodontitis and the risk of tongue cancer. Arch. Otolaryngol. Head Neck Surg. 2007, 133, 450–454. [Google Scholar]
- Tezal, M.; Sullivan, M.A.; Hyland, A.; Marshall, J.R.; Stoler, D.; Reid, M.E.; Loree, T.R.; Rigual, N.R.; Merzianu, M.; Hauck, L.; Lillis, C.; Wactawski-Wende, J.; Scannapieco, F.A. Chronic periodontitis and the incidence of head and neck squamous cell carcinoma. Cancer Epidemiol. Biomarkers Prev. 2009, 18, 2406–2412. [Google Scholar]
- Donald, C.D.; Sun, C.Q.; Lim, S.D.; Macoska, J.; Cohen, C.; Amin, M.B.; Young, A.N.; Ganz, T.A.; Marshall, F.F.; Petros, J.A. Cancer-specific loss of beta-defensin 1 in renal and prostatic carcinomas. Lab. Invest. 2003, 83, 501–505. [Google Scholar]
- Sun, C.Q.; Arnold, R.; Fernandez-Golarz, C.; Parrish, A.B.; Almekinder, T.; He, J.; Ho, S.M.; Svoboda, P.; Pohl, J.; Marshall, F.F.; Petros, J.A. Human beta-defensin-1, a potential chromosome 8p tumor suppressor: Control of transcription and induction of apoptosis in renal cell carcinoma. Cancer Res. 2006, 66, 8542–8549. [Google Scholar]
- Joly, S.; Compton, L.M.; Pujol, C.; Kurago, Z.B.; Guthmiller, J.M. Loss of human beta-defensin 1, 2, and 3 expression in oral squamous cell carcinoma. Oral Microbiol. Immunol. 2009, 24, 353–360. [Google Scholar] [CrossRef]
- Wenghoefer, M.; Pantelis, A.; Dommisch, H.; Reich, R.; Martini, M.; Allam, J.P.; Novak, N.; Bergé, S.; Jepsen, S.; Winter, J. Decreased gene expression of human beta-defensin-1 in the development of squamous cell carcinoma of the oral cavity. Int. J. Oral Maxillofac. Surg. 2008, 37, 660–663. [Google Scholar] [CrossRef]
- Kesting, M.R.; Loeffelbein, D.J.; Hasler, R.J.; Wolff, K.D.; Rittig, A.; Schulte, M.; Hirsch, T.; Wagenpfeil, S.; Jacobsen, F.; Steinstraesser, L. Expression profile of human beta-defensin 3 in oral squamous cell carcinoma. Cancer Invest. 2009, 27, 575–581. [Google Scholar] [CrossRef]
- Wenghoefer, M.; Pantelis, A.; Najafi, T.; Deschner, J.; Allam, J.P.; Novak, N.; Reich, R.; Martini, M.; Bergé, S.; Fischer, HP.; Jepsen, S.; Winter, J. Gene expression of oncogenes, antimicrobial peptides, and cytokines in the development of oral leukoplakia. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2010, 110, 351–356. [Google Scholar]
- Winter, J.; Pantelis, A.; Allam, J.P.; Novak, N.; Reich, R.; Martini, M.; Bergé, S.; Deschner, J.; Jepsen, S.; Wenghoefer, M. High α-defensin and S100A7 expression and missing DOC-1 down-regulation characterize irritation fibromas of the oral cavity and may counteract malignant transformation. J. Craniofac. Surg. 2011, 22, 100–104. [Google Scholar]
- Schaefer, A.S.; Richter, G.M.; Nothnagel, M.; Laine, M.L.; Rühling, A.; Schäfer, C.; Cordes, N.; Noack, B.; Folwaczny, M.; Glas, J.; Dörfer, C.; Dommisch, H.; Groessner-Schreiber, B.; Jepsen, S.; Loos, B.G.; Schreiber, S. A 3' UTR transition within DEFB1 is associated with chronic and aggressive periodontitis. Genes Immun. 2010, 11, 45–54. [Google Scholar]
- Lichtenstein, A.; Ganz, T.; Selsted, M.E.; Lehrer, R.I. In vitro tumor cell cytolysis mediated by peptide defensins of human and rabbit granulocytes. Blood 1986, 68, 1407–1410. [Google Scholar]
- Lapteva, N.; Aldrich, M.; Rollins, L.; Ren, W.; Goltsova, T.; Chen, S.Y.; Huang, X.F. Attraction and activation of dendritic cells at the site of tumor elicits potent antitumor immunity. Mol. Ther. 2009, 17, 1626–1636. [Google Scholar]
- Escher, N.; Spies-Weisshart, B.; Kaatz, M.; Melle, C.; Bleul, A.; Driesch, D.; Wollina, U.; von Eggeling, F. Identification of HNP3 as a tumor marker in CD4+ and CD4- lymphocytes of patients with cutaneous cell lymphoma. Eur. J. Cancer 2006, 42, 249–255. [Google Scholar] [CrossRef]
- Radeva, M.Y.; Jahns, F.; Wilhelm, A.; Glei, M.; Settmacher, U.; Greulich, K.O.; Mothes, H. Defensins alpha 6 (DEFA 6) overexpression threshold of over 60 fold can distinguish between adenoma and fully blown colon carcinoma in individual patients. BMC Cancer 2010, 10. [Google Scholar]
- Zhang, L.; Parente, J.; Harris, S.M.; Woods, D.E.; Hancock, R.E.W.; Falla, T.J. Antimicrobial peptide therapeutics for cystic fibrosis. Antimicrob. Agents Chemother 2005, 49, 2921–2927. [Google Scholar]
- Winder, D.; Gunzburg, W.H.; Erfle, V.; Salmons, B. Expression of antimicrobial peptides has an antitumour effect in human cells. Biochem. Biophys. Res. Commun. 1998, 242, 608–612. [Google Scholar] [CrossRef]
- Papo, N.; Shahar, M.; Eisenbach, L.; Shai, Y. A novel lytic peptide composed of D, L amino acids selectively kills cancer cells in culture and in mice. J. Biol. Chem. 2003, 278, 21018–21023. [Google Scholar]
- Chen, Y.; Xu, X.; Hong, S.; Chen, J.; Liub, N.; Underhill, C.B.; Creswell, K.; Zhang, L. RGD-tachyplesin inhibits tumor growth. Cancer Res. 2001, 61, 2434–2438. [Google Scholar]
- Papo, N.; Shai, Y. Host defense peptides as new weapons in cancer treatment. Cell. Mol. Life Sci. 2005, 62, 784–790. [Google Scholar] [CrossRef]
- Jenssen, H.; Hamill, P.; Hancock, R.E.W. Peptide antimicrobial agents. Clin. Microbiol. Rev. 2006, 19, 491–511. [Google Scholar] [CrossRef]
- Hancock, R.E.W.; Sahl, H.G. Antimicrobial and host defense peptides as new anti-infective therapeutic strategies. Nat. Biotechnol. 2006, 24, 1551–1557. [Google Scholar] [CrossRef]
- Rotem, S.; Mor, A. Antimicobial peptide mimics for improved therapeutic properties. Biochim. Biophys. Acta 1788, 1582–1592. [Google Scholar]
- Findlay, B.; Zhanel, G.G.; Schweizer, F. Cationic amphiphiles, a new generation of antimicrobials inspired by the natural antimicrobial peptide scaffold. Antimicrob. Agents Chemother 2010, 54, 4049–4058. [Google Scholar] [CrossRef]
- Yeung, A.T.Y.; Gellatly, S.L.; Hancock, R.E.W. Multifunctional cationic host defense peptides and their clinical applications. Cell. Mol. Life Sci. 2011, 68, 2161–2176. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Winter, J.; Wenghoefer, M. Human Defensins: Potential Tools for Clinical Applications. Polymers 2012, 4, 691-709. https://doi.org/10.3390/polym4010691
Winter J, Wenghoefer M. Human Defensins: Potential Tools for Clinical Applications. Polymers. 2012; 4(1):691-709. https://doi.org/10.3390/polym4010691
Chicago/Turabian StyleWinter, Jochen, and Matthias Wenghoefer. 2012. "Human Defensins: Potential Tools for Clinical Applications" Polymers 4, no. 1: 691-709. https://doi.org/10.3390/polym4010691



