Hydrogels for Cardiac Tissue Engineering
Abstract
: Cardiac tissue regeneration is an integrated process involving both cells and supporting matrix. Cardiomyocytes and stem cells are utilized to regenerate cardiac tissue. Hydrogels, because of their tissue-like properties, have been used as supporting matrices to deliver cells into infarcted cardiac muscle. Bioactive and biocompatible hydrogels mimicking biochemical and biomechanical microenvironments in native tissue are needed for successful cardiac tissue regeneration. These hydrogels not only retain cells in the infarcted area, but also provide support for restoring myocardial wall stress and cell survival and functioning. Many hydrogels, including natural polymer hydrogels, synthetic polymer hydrogels, and natural/synthetic hybrid hydrogels are employed for cardiac tissue engineering. In this review, types of hydrogels used for cardiac tissue engineering are briefly introduced. Their advantages and disadvantages are discussed. Furthermore, strategies for cardiac regeneration using hydrogels are reviewed.1. Introduction
The heart is a fascinating engineering marvel of nature. In the heart, cardiac muscle contraction pumps nutrient- and oxygen-rich blood to supply the entire body, including the cardiac muscle itself. Coronary arteries (CA) supply blood for the cardiac muscle [1]. CA narrowing or clogging reduces the blood supply and causes heart cell death within minutes, leading to myocardial infarction (MI). This initiates a decrease in heart function. After MI, the death of muscle cells triggers a remodeling cascade. The infarcted area is gradually filled with collagen-containing scar tissue to withstand the higher pressure during the contraction cycle (systole). As the scar tissue becomes thinner, the heart function further decreases; this finally becomes congestive heart failure (CHF) [2].
Current clinical intervention for MI is mainly concentrated on coronary reperfusion, which aims to reintroduce oxygen in the infarcted heart to lower cell death. Reperfusion therapy, however, is a conservative method that does not involve new cardiac muscle regeneration because adult cardiomyocytes are non-regenerative. Delivering cells into the heart for cardiac regeneration is accepted to be the ultimate therapeutic approach. These cells rebuild the muscle and integrate with the native heart, leading to an increase in heart function. Many clinical trials are ongoing to deliver stem cells into the heart. A commonly used delivery approach is to suspend cells in a buffer and inject into the heart. However, the injected cells have failed to show a satisfied engraftment rate [3]. One of the possible reasons is the harsh environment at the infarcted area. The blockage of the coronary artery causes very low oxygen and nutrient levels. Meanwhile, phagocytosis releases apoptotic cytokines and cell toxic reactive oxygen species (ROS). All of these factors limit the survival of the delivered cells. The engraftment rate may be improved by an approach called regenerative cardiac tissue engineering, in which cells are delivered within a supporting matrix (scaffold or hydrogel). The supporting matrix prevents cell loss during the injection, and provides an environment protecting cells from attack by the harsh environment in the MI heart. In addition, it acts as a mechanical support to alleviate the elevated wall stress caused by the loss of normal cardiac muscle cells, leading to cardiac function improvement. This approach is also versatile, as biomolecules can be readily incorporated into the matrix to build a cell-friendly environment for better regeneration.
Cardiac tissue engineering mainly uses two classes of materials: cardiac scaffold patches and hydrogels. This review mainly discusses hydrogels, which are water-insoluble, crosslinked polymer matrices with high water content (>30%).
2. Physiology of Cardiac Muscles
The heart wall is composed of the endocardium, myocardium, and epicardium layers [4]. The myocardium is the major layer, providing strength in the systole and diastole. The myocardium has a unique structure (Figure 1) in which multiple layers made of extracellular matrix (ECM) and highly oriented cells are packed together. The ECM is considered to be a major factor directing the alignment of cardiomyocytes [5].
Besides its anisotropic structure, the cardiac muscle exhibits unique biomechanical behavior. It is strong but ductile in the systole phase, while it is more elastomeric in the diastole phase. The Frank-Starling relationship describes the mechanical response of cardiac muscle during a full pump cycle (Figure 2). This unique biomechanical behavior allows cardiomyocytes in the heart to function normally. Many studies show that cardiomyocytes behave poorly on a matrix stiffer than cardiac muscle [6,7]. Therefore, for cardiac tissue regeneration, mimicking cardiac muscle's native mechanical environment is important.
The heart also exhibits a unique electrophysiological property. A continuous integrated electrical conductive network is built inside the heart to direct the spontaneous heart cycle. Lack of connection to the existing cardiac electrical network induces arrhythmia, and reduces heart function.
3. Pathology of MI: The Remodeling Process
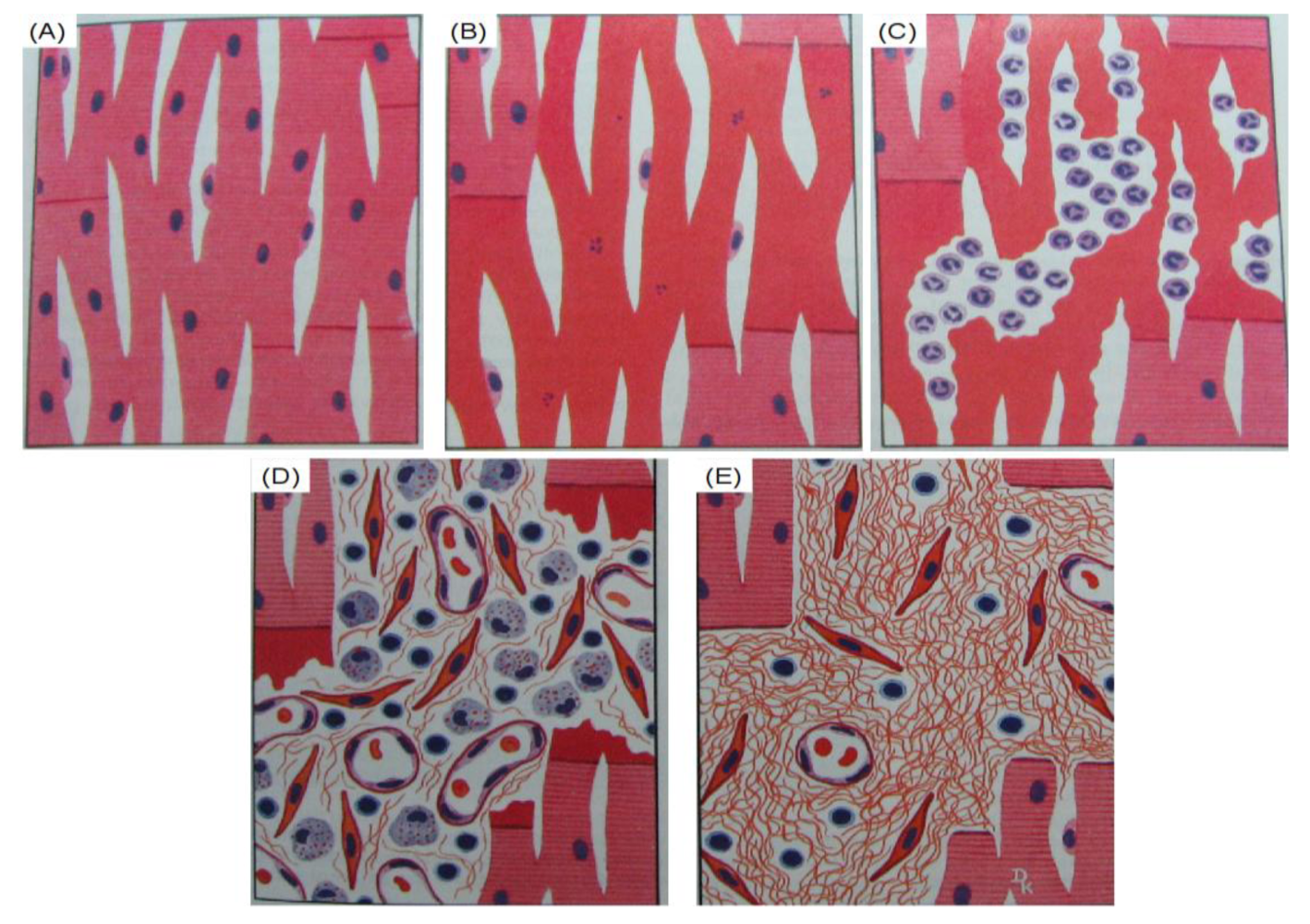
For acute MI, if the blood supply resumes within 30 min, the injured myocardium can revert into normal state, although a depression in contractility may persist for hours (Figure 3(A)). After 30 min, the myocardium undergoes an irreversible necrosis process, beginning with swollen mitochondria, broken nuclei, and disruptions in the cell membrane, and disordered tissue structure. Within 12–18 h, proteins in the cytosol start to leak out and the dead myocardial fibers form “wavy fiber” under the stretching of the systole (Figure 3(B)). Within 2 days, polymorphonuclear leukocytes are attracted to the infarcted area and accumulate in the periphery of the infarcted sites. The neutrophils start to infiltrate until they fill the infarcted area. The phagocytosis of dead cardiomyocytes can then be clearly seen in the infarcted area (Figure 3(C)). At day 5, fibroblasts start to proliferate and collagen deposition begins (Figure 3(D)). After 3 weeks, the inflammation process withdraws and collagen deposition continues. In the fourth week, a dense fibrous scar tissue is formed, and the remodeling of the MI is nearly finished [12] (Figure 3(E)). The gradual maturation of the scar tissue will further induce heart wall dilation to compensate for the lost heart function by accommodating a larger volume of blood.
Timely reperfusion is crucial for acute MI. A bypass surgery can be done to reestablish blood to the blocked arteries by grafting a short vein (normally from the saphenous veins). However, if the myocardium has already undergone permanent damage, reperfusion cannot regain the lost cardiac muscle due to the non-regenerative nature of adult cardiomyocytes. This leads to a permanent decrease of heart function. The ultimate goal for post-MI therapy is to attenuate the remodeling process and regenerate the new cardiomyocyte-based muscle. This can be achieved by a cell delivery system consisting of a supporting matrix and suitable cells. The function of the supporting matrix is to stop or slow down the remodeling and scar formation process by mechanically preventing the cardiac muscle from dilating [13]. Meanwhile, suitable cells are delivered to replace the dead cardiomyocytes and integrate with the neighboring cardiac tissue. The supporting matrix should also have a cell-friendly environment to support the survival of the delivered cells and facilitate the regeneration process. The properties of an ideal cell delivery system are summarized in Table 1. In the following part of this review, how the hydrogel materials addressing these requirements is discussed.
4. Hydrogels for Cardiac Tissue Engineering
Both natural and synthetic hydrogels are suitable for cardiac tissue engineering because their soft and viscoelastic nature mimic the native tissue. Collagen, gelatin, laminin, matrigel, hyaluronic acid (hyaluronan), alginate, and chitosan are typical natural hydrogels. They have similar or even identical structures to the molecules in biological organisms, thereby reducing the possibility of immune response when implanted in vivo.
Synthetic materials used for cardiac tissue engineering include poly(ethylene glycol) (PEG), polylactide (PLA), polylactide-glycolic acid (PLGA), polycaprolactone (PCL), polyacrylamide (PAAm), and polyurethane (PU). It is easy to tailor the physical and chemical properties of these synthetic polymers, such as modulus, water affinity, and degradation rate, to meet the requirements of cardiac muscle tissue engineering. Therefore, they are considered to be good candidates for tissue engineering. However, potential cytotoxicity is a major concern for synthetic polymers. So far, only PEG, PLA, and PLGA have been approved by the FDA for clinical applications. In fact, many other polymers like PAAm and PU have already been confirmed to be non-toxic in vitro and in vivo. An alternative method is to use natural/synthetic hybrid hydrogels that combine the advantages of both natural and synthetic polymers. This can be achieved by blending or covalently grafting/crosslinking (discussed in Section 4.3).
4.1. Natural Hydrogels
Human tissues and organs are assembled by cells and ECMs [5]. The native ECM components are suitable materials to be used as the delivery vehicles of cells because of their high biocompatibility and biodegradability [14]. In this section, the commonly used natural hydrogels are discussed.
4.1.1. Collagen and Decellularized Matrix
Collagen provides most of the mechanical strength in tissues [1]. There are many types of collagens in the body. Among these, type I collagen is ubiquitously distributed. Because of its excellent biocompatibility, type I collagen has been used in tissue engineering [15-21]. Type I collagen hydrogel can be formed by dissolving the collagen in an acid solution (for example, in 0.3% glacial acetic acid) and then neutralizing the solution to pH = 7.4 [15]. However, the solution-neutralization method is not ideal for cell encapsulation because the initial acid condition may cause cell death. Additionally, injection of acidic solution in an infarcted heart may provoke the local inflammatory reaction. Recently, a method of forming collagen gel via incubating the neutralized solution in physiological conditions has been reported [16]. However, the slow gelation time of this gel may be a problem when used for cardiac tissue engineering, since the encapsulated cells may be flushed away before the gel is formed.
Alternatively, a collagen-containing decellularized matrix can be used. It is prepared by decellularizing native tissues like bladder, pericardium, and heart [17]. Mirsadraee et al. decellularized the pericardium and found that the chemical compositions of the native and acellular pericardium are not significantly different [18,19]. In addition, no cytotoxicity was observed for human dermal fibroblasts seeded on the acellular sample. Further study on foreign body reaction showed that the acellular pericardium provoked minor macrophage response [20]. These results demonstrate the feasibility of using acellular ECM as a potential scaffold for cardiac tissue engineering. Recently, a groundbreaking study by Ott et al. showed that a decellularized rat heart can be used as a template to make a functional heart by recellularizing it with cardiomyocytes, endothelial cells (ECs), and smooth muscle cells (SMCs) [21].
A soluble ECM hydrogel capable of thermally gelling at body temperature can be obtained by an additional digestion process to the above decellularized matrix. The advantage of using acellular ECM hydrogel is that it may be delivered into the heart by a minimally invasive cardiac surgery. Singelyn et al. generated injectable porcine myocardium hydrogel [22] (Figure 4). In vitro tests showed that the gel supported the survival of cardiomyocytes, and the migration of ECs and SMCs. Further in vivo study demonstrated the infiltration of ECs and SMCs into hydrogel. Additionally, vascularization was enhanced within the hydrogel. However, the in vivo study was conducted on a normal rat heart instead of an MI heart. An MI model is needed to verify these findings.
There are disadvantages concerning the decellularized ECM for cardiac tissue engineering. First, any remaining cells or immunogenic proteins will provoke immune rejection. No existing methods can guarantee that the decellularized ECM will be free of immunogenic proteins. Second, the long gelation time may cause the injected gel being flushed away shortly after injection. Rapid gelation (less than one minute) is necessary to retain the delivered material and cells after injection.
4.1.2. Fibrin Gel
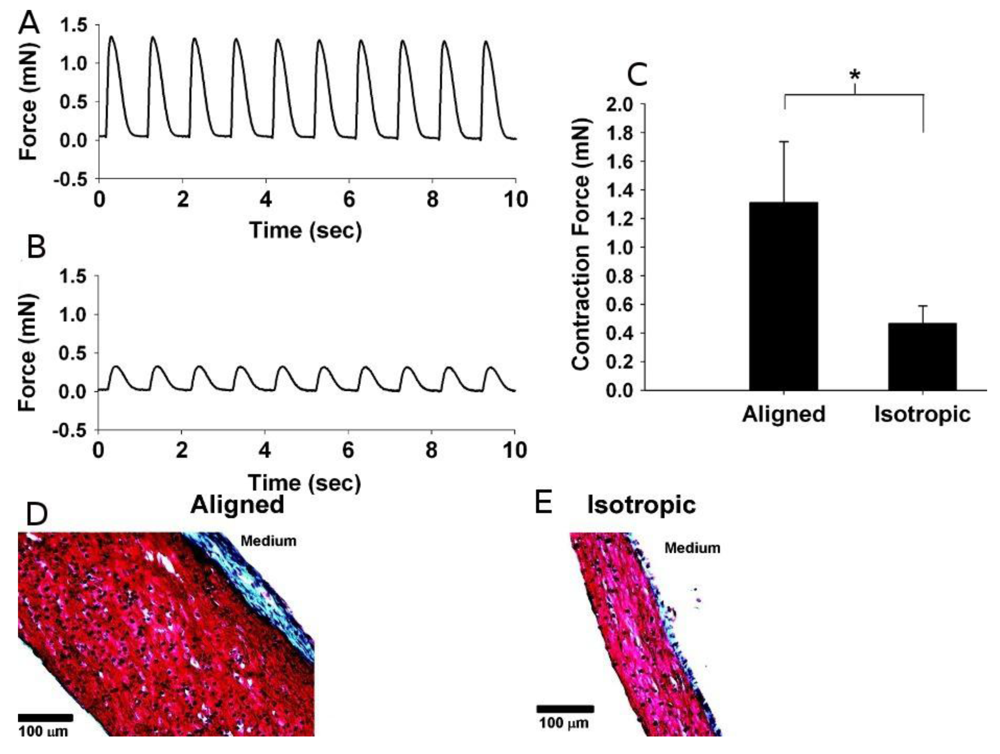
Fibrin is formed during the haemostatic coagulation process by combining fibrinogen and thrombin under the catalysis of calcium ions [23]. Fibrin gel is biodegradable and nontoxic. Therefore, it is suitable for tissue engineering. Birla et al. fabricated hollow fibrin gel tubes populated with neonatal cardiomyocytes and implanted into the femoral artery of adult rats [24]. After 3 weeks of implantation, the fibrin gel/cardiomyocytes construct formed a mature cardiac tissue with a dense capillary network. It possessed all normal cardiac functions, including contractility under electric stimulation and synchronous pacing with an outside electric signal. Huang et al. embedded rat cardiomyocytes within fibrin gel and found that their contractility can be retained up to two months with normal pacing ability [25-27]. The embedded cardiomyocytes showed aligned morphology. Black et al. cultured the fibrin/cardiomyocytes constructs under circumferential/radial contraction condition (“isotropic” group) and circumferential/radial/axial contraction condition (“aligned” group), respectively [28]. Although the “isotropic” group had a high collagen content and cell density, the mechanical contract response under electrical pacing stimulation was significantly less than that in the “aligned” group (Figure 5(A–C)). This difference can be explained by the different contents of connexin 43 (CX43) protein under different conditions.
4.1.3. Matrigel
Matrigel, composed of basement membrane proteins as well as growth factors, is an ECM-mimicking hydrogel produced by mouse Engelbreth-Holm-Swarm tumors [29]. It closely resembles the native ECM with a similar composition and assembling structure. Thus, it has been considered as a cytocompatible gel. In addition, Matrigel shows the ability of faster vascularization compared to other natural hydrogels. These properties make Matrigel a potential candidate for cardiac tissue engineering. Injection of cells with Matrigel into the heart significantly increased the cell retention [30], indicating that the viscous Matrigel solution can increase the cell retention in the infarcted area. Copland et al. used a Matrigel plug as a delivery carrier to deliver genetically modified human mesenchymal stromal cells (MSCs) into an infarcted heart, and showed that MSC survival was significantly enhanced under the ischemic and apoptotic environment [31]. Matrigel can also be combined with other natural materials to improve cell proliferation and angiogenesis in vivo. Giraud et al. demonstrated that Matrigel/collagen hydrogel significantly improved heart function after implantation into acute MI rat hearts [32]. The implantation of Matrigel/collagen hydrogel and H9C2 cardiomyoblasts in an acute rat MI model was found to significantly increase cell engraftment rate [33]. Matrigel was also combined with fibrin gel to encapsulate cardiomyocytes. The cardiomyocytes maintained normal function throughout the entire experiment time (10 days) [34].
Matrigel and fibrin gel are both naturally derived ECMs. They are free of cytotoxic issues. However, fibrin gel and Matrigel are still far from ideal for cardiac tissue engineering. For fibrin gel, the relatively slow gelation rate and lack of sufficient mechanical strength are major challenges. Slow gelation time, as discussed above, causes a loss of delivered cells and low cell retention during the injection. Low mechanical strength leads to the possible breakdown of gels during heart contraction and relaxation. In addition, fibrin gel has a fast degradation (fibrinolysis) rate. Normally, in the fibrin gel culture medium, a chemical like aprotinin is used to inhibit fibrinolysis. However, it is possibly toxic to the cells [35]. Without aprotinin, a high degradation rate results in a rapid loss of the supporting matrix before the maturation of the delivered cells and the establishment of angiogenesis. This interrupts the regeneration process. For Matrigel, a biosafety concern exists, since it is derived from tumors.
4.2. Synthetic Macromolecules for Cardiac Tissue Engineering
4.2.1. Poly(ethylene glycol)
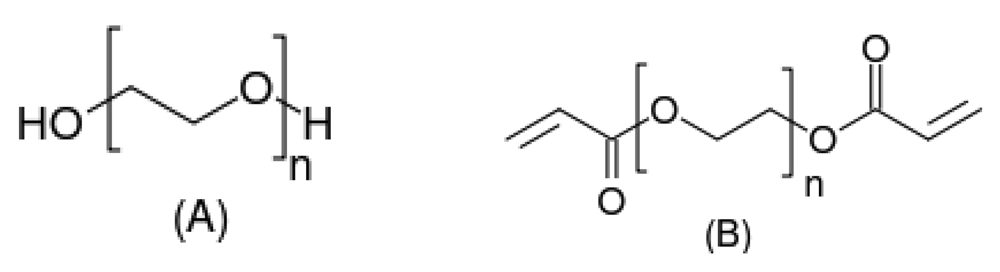
Poly(ethylene glycol) (PEG, Figure 6(A)) is a water soluble polymer synthesized by the ring-opening polymerization of ethylene oxide [36]. It is biocompatible and has been approved by the FDA. PEG hydrogel is typically made by the polymerization of diacrylate-modified PEG (Figure 6(B)) via photo-polymerization under ultraviolet (UV) irradiation. PEG gel has been widely used as a supporting matrix in almost every field of tissue engineering (nerve, cartilage, liver, pancreas, bladder, skin) because its low protein adsorption and inert surface reduce the inflammation after implantation. However, low protein affinity is not beneficial for cell adhesion. Methods like conjugating cell adhesive peptides or proteins and incorporating growth factors are used to increase cell adhesion [6,37]. PEG hydrogel has been used to study cardiomyocyte-matrix interactions in a three-dimensional (3-D) environment. For example, it was found that Arg-Gly-Asp (RGD) peptide modification largely increased the viability of encapsulated cardiomyocytes [38]. Kraehenbuehl et al. constructed a series of PEG hydrogels with different moduli by varying crosslinking density [7]. Embryonic stem cells (ESCs) were encapsulated inside. The results showed that hydrogels with lower moduli (soft) induced the ESCs differentiation into cardiomyocyte lineage. The differentiated cells exhibited a cardiac-like function.
PEG hydrogel can also be formed by PEG-cyclodextrin interaction. This could avoid a potential toxic issue associated with the commonly employed photo-polymerization method. Wang et al. used a PEG–PCL–PEG triblock copolymer mixed with α-cyclodextrin to encapsulate bone marrow mesenchymal stem cells and delivered to the rabbit MI site [39]. The retention and survival of the delivered MSCs were significantly increased compared to the control group (cells suspended in saline). Dense vessel networks at the injection sites and the reduction of infarcted areas were observed.
4.2.2. Poly(2-hydroxyethyl methacrylate)
Poly(2-hydroxyethyl methacrylate) (PHEMA) is a hydrophilic polymer with pendant hydroxyl groups (Figure 7). PHEMA hydrogel has been used in cardiac tissue engineering. Walker et al. implanted poly(ethylene terephthalate) (PET) mesh reinforced PHEMA gel into the canine epicardium [40,41]. No significant fibrosis or thickening was observed 12 months after implantation. However, there was trace calcification on the gel after 9 and 12 months of implantation, raising the concern of biocompatibility of the PET/PHEMA constructs over a long timeframe.
4.2.3. Polyacrylamide and Its Derivatives
The polyacrylamide family is an amide derivative of poly(acrylic acid) (Figure 8). Polyacrylamide gels, because of their peptide/protein mimicking amide structure, are also candidates for cardiac tissue engineering. Similar to PEG hydrogels, crosslinking is needed to fabricate polyacrylamide hydrogels [7]. What makes polyacrylamide gel different from other gels mentioned above is its thermal sol-gel transition. Such a transition allows aqueous polyacrylamide solution to be liquid when the temperature is low; it then solidifies to form a gel at body temperature. The detailed mechanism of its thermosensitivity is still under debate. A widely accepted theory is that hydrogen bonds between amide groups and water molecules are dissociated and the pendant amide groups tend to collapse together as the temperature increases. Thus, the water molecules are repelled and a hydrophobic solid gel is formed [14]. Regardless of the mechanism, this thermosensitive property has been used to design novel injectable hydrogels that can be delivered in vivo in a liquid form through a needle and then turn into solid gels after contact with the warm tissue.
Poly(N-isopropylacrylamide) (PNIPAAm) is a typical thermosensitive polymer with a thermal transition temperature (LCST) of 32 °C. Its aqueous solution is in liquid state at room temperature while forming hydrogel at 37 °C. Okano et al. utilized this property to generate cell sheets for tissue engineering. Tissue culture plates were coated with PNIPAAm and neonatal cardiomyocytes were cultured on top at 37 °C [42]. After cells reached confluence, the culture plate was cooled down to room temperature and cells were lifted to form a monolayer cell sheet. The cardiomyocyte monolayers were stacked together and implanted into the infarcted myocardium. The recovery of heart function was observed 4 weeks after implantation. PNIPAAm based hydrogel was also used to encapsulate cells. A gelatin grafted PNIPAAm hydrogel was employed to encapsulate rat cardiac cells [43]. The best contractility was obtained with a seeding density of 50 million/mL. Guan et al. developed a family of protein conjugated, biodegradable PNIPAAm hydrogels based on NIPAAm, acrylic acid, acrylic N-succinimide ester and HEMA-poly(trimethylene carbonate) [44]. The hydrogels changed their LCSTs from room temperature before degradation to above 40 °C after degradation. The degradation products are therefore soluble in body fluid. The hydrogels and their degradation products were non-cytotoxic. Fujimoto et al. synthesized similar hydrogels and injected into infarcted hearts and an improvement of heart function was observed [45]. Guan et al. further functionalized the developed PNIPAAm hydrogels with growth factor [46] and antioxidants [47]. It was found that functionalization significantly enhanced MSC growth within the hydrogels. It is expected that the functionalized hydrogels will provide a suitable environment for cells delivered into heart to survive and function.
Many synthetic polymers are used to make hydrogels for cardiac tissue engineering. The advantage of synthetic polymers is their versatility in controlling physical and biochemical properties, including stiffness, water content and cell adhesion. They are easy to make and cost-effective compared to the natural polymers. However, biocompatibility and possible inflammatory reaction may be an issue. To combine the advantages of both natural and synthetic hydrogels, the approach that blends synthetic and natural hydrogels (gelatin, chitosan, fibrin and hyaluronan) seems to be a good alternative.
4.3. Natural/Synthetic Hybrid Hydrogels for Myocardial Tissue Engineering
In general, natural materials exhibit better biocompatibility and cell affinity than synthetic polymers, but are less versatile in terms of tailoring properties like mechanical strength, water content, and degradation rate. Combining natural and synthetic hydrogels to make a blend seems to be a good solution when it comes to capitalizing on the advantages of both. For example, the biocompatibility of synthetic polymers can be improved by introducing collagen, fibronectin, laminin, or fibrinogen. Natural/synthetic hydrogels can also be used to modulate cell phenotype. Putnam et al. introduced fibrinogen into PEG gel and cultured SMCs in the gel [48]. The cells showed a spindle-like shape as compared to the round shape normally observed in purely synthetic PEG gels. In addition, synthetic hydrogels can greatly enhance the function of natural hydrogel. Zhang et al. PEGylated fibrin hydrogel and loaded it with stromal derived factor (SDF) [49]. It was found that incorporation of PEG prolonged the half-life of the encapsulated growth factor.
Introduction of naturally derived peptide sequences into synthetic hydrogels is another approach to fabricate natural/synthetic hydrogels. The peptide sequences can be cell adhesive motifs, enzyme-sensitive segments, and anti-inflammatory segments. RGD is a peptide sequence widely used to improve cell adhesion and biocompatibility [8,37,50-54]. The PEG gel containing RGD sequence has a significantly higher cell adhesion compared to the pure PEG gel [50] (Figure 9). The enzyme-sensitive peptides are used to adjust the degradation of the gel matrix to match the rate of the new ECM deposition during the tissue maturation. For example, a matrix metalloproteinase (MMP)-sensitive peptide was introduced to hyaluronic acid hydrogel and it was found that the gel had a faster degradation rate than the control (non-MMP-sensitive peptide incorporated gel) [52]. Kraehenbuehl et al. used a PEG gel that contains MMP-sensitive peptide to culture P19 embryonic stem cells, and found that MMP-mediated degradation facilitated stem cell differentiation into cardioprogenitor cells [7].
In summary, blending natural and synthetic hydrogels represents a feasible approach to design hydrogels with controllable mechanical and biochemical properties without compromising biocompatibility and biodegradability. Recent progresses in bioconjugation chemistry provide the new possibility for functionalizing a synthetic matrix with naturally derived proteins or peptides [56]. The natural/synthetic hybrid hydrogels can be a future direction in hydrogel design for cardiac tissue engineering.
5. Application of Hydrogels for Cardiac Tissue Engineering and Challenges
Hydrogels for cardiac therapy can be used in two forms, i.e., hydrogel only and hydrogel with cells [3,57-59]. The application of each form for cardiac tissue engineering is discussed in this section.
5.1. Hydrogel-Based Therapy
From a pathology point of view, collagen deposition is the last step of remodeling after MI. If myocardium hypotension cannot be relieved, further ventricle dilation will occur. This will lead to CHF. To break this process, it has been proposed to inject a supporting gel into the infarcted heart to release the elevated wall stress. Injection of PNIPAAm-based copolymers has been shown to attenuate ventricular dilation [45]. This suggests that hydrogel-only therapy can be effective in delaying the cascade that leads to CHF by providing sufficient mechanical support to the infarcted area. However, lack of cells makes this therapy only “passive,” unable to regenerate new myocardium.
5.2. Cell/Hydrogel Delivery Strategy
A promising strategy for cardiac tissue engineering lies in hydrogel-based cell therapy. The viscous hydrogel holds the cells in the target place during injection. The gel itself provides mechanical support to the weakened heart wall [13,22,60,61]. Meanwhile, the gel environment may allow the delivered cells to survive and differentiate into cardiomyocytes to regenerate cardiac muscle. To facilitate cell survival, growth, and differentiation, biochemicals can be co-delivered with hydrogels.
5.2.1. Cells Available for Delivery
The ideal cell type for cardiac tissue engineering is cardiomyocytes. Fetal and neonatal cardiomyocytes have been tested for this purpose [62,63]. However, the implanted cardiomyocytes were unable to integrate with the native myocardium and caused arrhythmias [64]. In addition, the source for human fetal or neonatal cardiomyocytes is a concern. Skeletal myoblasts have also been tested. These cells are fatigue-resistant and have a great tolerance to the ischemic environment [65]. However, the arrhythmia issue persists [66].
Stem cells, including adult stem cells, ESCs, and induced pluripotent stem cells (iPSCs) are possible sources. These stem cells can proliferate and differentiate into cardiomyocytes. For example, bone marrow- and adipose-derived MSCs were reported to differentiate into cardiomyocyte lineage through treatment them with dimethyl sulfoxide and 5-azacytidine (5-aza) for 24 hours in vitro [67], or through delivery them into the heart in vivo [68]. ESCs cannot be used directly because they cause teratoma formation [69], but they can be differentiated into cardiomyocytes and then used [69-72]. iPSCs have recently been invented with a review to replace ESCs, which are involved in ethical controversy. The iPSCs are generated by transfecting four genes into skin fibroblasts [73,74]. They can be induced to differentiate into cardiomyocytes [75].
Despite the success of using stem cells in cardiac therapy, some challenges still remain. First, strategies that precisely control stem cell differentiation into a specific lineage in vitro and in vivo need further exploring. Current approaches are either toxic to cells (for example, 5-aza to MSC and cell signaling inhibitor to ESCs) or have potential biosafety concerns (like genetic modification). Recent progresses suggest that cells' surrounding microenvironment directs stem cell differentiation. The microenvironment is a small surrounding space that cells can feel and to which they respond. It includes matrix's global properties like biomechanics, hydrophilicity, and cell adhesion affinity, as well as localized properties like neighbored conjugated bioactive molecules (signal niches) and 3-D patterns [76]. Discussing the detailed relation between microenvironment and stem cell differentiation is beyond the scope of this review, but the design of a microenvironment regulating stem cell differentiation into cardiac lineage is a very interesting topic from a materials perspective.
Second, a better cell source for cardiac tissue engineering is in demand. The ability of MSCs to differentiate into cardiomyocytes is still debated. ESCs involve ethical problems. iPSCs represent a good alternative to ESCs. However, both the virus-involved transfection process and the potential oncological issue have limited their progression to clinical trials at present. Therefore, new cell sources still need to be sought. Recently, a new source of cardiac stem cells has been discovered from heart biopsy [77-79]. These cells originate from the patient's own small heart apex biopsy, and can proliferate quickly in vitro. The in vivo studies showed that they are able to differentiate into cardiomyocytes.
5.2.2. Choosing Appropriate Hydrogel Matrix for Cardiac Tissue Engineering
Precisely controlling stem cell differentiation is critical for cardiac tissue engineering. As mentioned above, stem cell differentiation may be mediated by its surrounding microenvironment. It is hypothesized that a native myocardium-mimicking microenvironment will facilitate stem cells differentiation into cardiac lineage. Thus, tuning hydrogel properties to mimic the structure and properties of the native heart appears to be a feasible approach for cardiac tissue engineering.
5.2.3. Aligned vs. Isotropic
As shown in Section 2 and Figure 1, cardiomyocytes are highly aligned in the heart. This unique structure affords myocardium with unique physical and electrical properties. Mimicking the alignment structure of myocardium may facilitate cardiac tissue development. A study of cardiomyocytes on the PEG hydrogel surface demonstrated that cells on the patterned surface can align during culture [6]. A larger contractile force (in the aligned direction) and a directional conductive current were observed in the aligned direction. This indicates that cardiomyocytes are more similar to their native phenotype and function if assembled on an aligned structure. These results are based on two-dimensional (2-D) culture. However, achieving alignment of cardiomyocytes in a 3-D hydrogel is challenging [80,81].
To achieve 3-D alignment, a possible approach is to apply mechanical stimulation to induce cell arrangement. An elastic hydrogel is likely needed to withstand the mechanical cycling. The highly extensible PNIPAAm-based hydrogels developed in our laboratory may be good candidates for this approach [44,46,47].
5.2.4. Soft vs. Stiff
As discussed above, the fate of stem cells depends on the surrounding microenvironment. Biomechanics represent one of the determining factors of a microenvironment. The relationship between stem cell fate and substrate matrix was first reported by Engler et al. [82]. Naive MSCs can differentiate into brain, muscle, and bone lineages simply by varying modulus of the substrate. ESCs show a similar response to the matrix modulus. They can differentiate into cardiac lineage by using the gel with Young's modulus of 300 Pa [9]. Adult cardiomyocytes also respond to the matrix modulus. Cardiomyocytes on the soft polyacrylamide showed an upregulated cardiac specific protein Tropinin I expression and a higher response in electrical stimulation than on stiff hydrogel [7]. Thus, a successful material design should consider matrix modulus. Currently, tuning gel modulus is mostly accomplished by varying the crosslinking density. However, this might also change other physical properties like degradation rate, water content, and cell affinity. A better design is needed to independently tune modulus.
5.2.5. Functionalization of Hydrogels
The cardiac ischemic environment is a harsh environment lacking nutrients/oxygen and rich in apoptotic species (e.g., superoxide) (Section 3). Suitable biomolecules can be co-delivered with the hydrogel to neutralize the harsh environment and protect the delivered cells. Prosurvival growth factors can be used to address the nutrient/oxygen supply issue, while antioxidants can be used to protect cells from cytotoxic superoxide. However, these substances are highly unstable in vivo. Therefore, they need to be protected and gradually released from the hydrogels. Chemical conjugation [46] and physical entrapping [47] have been shown to be good approaches.
6. Conclusions
Cardiomyocytes and stem cells are utilized to regenerate cardiac tissue and restore heart functions after MI. However, the injection of cells directly into the infarcted area involves the problems of low cell retention and engraftment rate. A proper supporting matrix can hold cells at the infarcted area initially and further provide support for cell survival and functioning. Hydrogels, because of their native tissue-like properties, are widely used as the cell delivery carriers and supporting matrices for cardiac tissue regeneration. A bioactive, biocompatible hydrogels mimicking native tissue biochemical and biomechanical environment are needed for successful cardiac tissue regeneration.





| Categories | Assessments |
|---|---|
| Cells | Survive in the harsh infarcted area |
| Protected from neutrophils attacks | |
| Potential to be cardiomyocytes | |
| Integrate with neighboring cardiac tissue | |
| Easy to isolate and expand in vitro | |
| Non-immunogenic | |
| … | |
| Matrix | Sufficient strength to reinforce weakened heart wall |
| Cell-friendly micro-environment for survival and differentiation | |
| Cardiac mimicking mechanics | |
| Nontoxic | |
| Degradable | |
| … |
| Category | Name | Properties | Concerns |
|---|---|---|---|
| Natural materials | Collagen and decellular ECM | Biocompatible, biodegradable, | weak strength, immune rejection, slow gelation |
| Fibrin gel | Biodegradable, biocompatible, availability | Slow gelation and fast degradation in vivo | |
| Matrigel | Closely resemble native ECM structure | Potentially carcinogenic concerns | |
| … | |||
| Synthetic materials | PEG | Bio-inert, biocompatible, FDA approved | Low cell adhesion, non-injectable and toxic concerns of small crosslinkers, not degradable |
| PHEMA | Biocompatible and available for functionalization | Modulus mismatch, non-degradable | |
| Polyamides | Fast gelation time, injectibility, versatile for chemical modification | Pure polyamides are not degradable, non-elastic | |
| … |
Acknowledgments
This work was supported by National Science Foundation (DMR1006734) and Institute for Materials Research at the Ohio State University.
References
- Berne, R.M.; Levy, M.N. Physiology; Mosby: New York, NY, USA, 1998; p. 1160. [Google Scholar]
- Wang, F.; Guan, J. Cellular cardiomyoplasty and cardiac tissue engineering for myocardial therapy. Adv. Drug Deliv. Rev. 2010, 62, 784–797. [Google Scholar]
- Hofmann, M.; Wollert, K.C.; Meyer, G.P.; Menke, A.; Arseniev, L.; Hertenstein, B.; Ganser, A.; Knapp, W.H.; Drexler, H. Monitoring of bone marrow cell homing into the infarcted human myocardium. Circulation 2005, 111, 2198–2202. [Google Scholar]
- Kierszenbaum, A.L. Histology and Cell Biology; Elsevier Health Sciences: New York, NY, USA, 2002; p. 648. [Google Scholar]
- Kim, D.; Kim, P.; Song, I.; Cha, J.M.; Lee, S.H.; Kim, B.; Suh, K.Y. Guided three-dimensional growth of functional cardiomyocytes on polyethylene glycol nanostructures. Langmuir 2006, 22, 5419–5426. [Google Scholar]
- Bhana, B.; Iyer, R.K.; Chen, W.L.K.; Zhao, R.; Sider, K.L.; Likhitpanichkul, M.; Simmons, C.A.; Radisic, M. Influence of substrate stiffness on the phenotype of heart cells. Biotechnol. Bioeng. 2010, 105, 1148–1160. [Google Scholar]
- Kraehenbuehl, T.P.; Zammaretti, P.; der Vlies, A.J.V.; Schoenmakers, R.G.; Lutolf, M.P.; Jaconi, M.E.; Hubbell, J.A. Three-dimensional extracellular matrix-directed cardioprogenitor differentiation: Systematic modulation of a synthetic cell-responsive PEG-hydrogel. Biomaterials 2008, 29, 2757–2766. [Google Scholar]
- Berne, R.M.; Levy, M.N. Physiology; Mosby Year Book: New York, NY, USA, 1993; p. 405. [Google Scholar]
- Fung, Y.C. Biomechanics: Mechanical Properties of Living Tissues; Springer: Berlin, Germany, 1993; p. 591. [Google Scholar]
- Wilson, T.E.; Brothers, R.M.; Tollund, C.; Dawson, E.A.; Nissen, P.; Yoshiga, C.C.; Jons, C.; Secher, N.H.; Crandall, C.G. Effect of thermal stress on Frank-Starling relations in humans. J Physiol. 2009, 587, 3383–3392. [Google Scholar]
- Levine, B.D.; Lane, L.D.; Buckey, J.C.; Friedman, D.B.; Blomqvist, C.G. Left ventricular pressure-volume and Frank-Starling relations in endurance athletes. Implications for orthostatic tolerance and exercise performance. Circulation 1991, 84, 1016–1023. [Google Scholar]
- Rubin, R.; Strayer, D.S.; Rubin, E. Rubin's Pathology; Lippincott Williams & Wilkins: Hagerstown, MD, USA, 2008; p. 1368. [Google Scholar]
- Wu, J.; Zeng, F.; Huang, X.P.; Chung, J.C.Y.; Konecny, F.; Weisel, R.D.; Li, R.K. Infarct stabilization and cardiac repair with a VEGF-conjugated, injectable hydrogel. Biomaterials 2010. [Google Scholar] [CrossRef]
- Ratner, B.D. Biomaterials Science; Academic Press: San Diego, CA, USA, 2004; p. 879. [Google Scholar]
- Xu, T.; Molnar, P.; Gregory, C.; Das, M.; Boland, T.; Hickman, J.J. Electrophysiological characterization of embryonic hippocampal neurons cultured in a 3D collagen hydrogel. Biomaterials 2009, 30, 4377–4383. [Google Scholar]
- Yang, Y.; Motte, S.; Kaufman, L.J. Pore size variable type I collagen gels and their interaction with glioma cells. Biomaterials 2010, 31, 5678–5688. [Google Scholar]
- Flynn, L.E. The use of decellularized adipose tissue to provide an inductive microenvironment for the adipogenic differentiation of human adipose-derived stem cells. Biomaterials 2010, 31, 4715–4724. [Google Scholar]
- Mirsadraee, S.; Wilcox, H.E.; Korossis, S.A.; Kearney, J.N.; Watterson, K.G.; Fisher, J.; Ingham, E. Development and characterization of an acellular human pericardial matrix for tissue engineering. Tissue Eng. 2006, 12, 763–773. [Google Scholar]
- Mirsadraee, S.; Wilcox, H.E.; Watterson, K.G.; Kearney, J.N.; Hunt, J.; Fisher, J.; Ingham, E. Biocompatibility of acellular human pericardium. J. Surg. Res. 2007, 143, 407–414. [Google Scholar]
- Ariganello, M.B.; Labow, R.S.; Lee, J.M. In vitro response of monocyte-derived macrophages to a decellularized pericardial biomaterial. J. Biomed. Mater. Res. A 2010, 93, 280–288. [Google Scholar]
- Ott, H.C.; Matthiesen, T.S.; Goh, S.; Black, L.D.; Kren, S.M.; Netoff, T.I.; Taylor, D.A. Perfusiondecellularized matrix: Using nature's platform to engineer a bioartificial heart. Nat. Med. 2008, 14, 213–221. [Google Scholar]
- Singelyn, J.M.; DeQuach, J.A.; Seif-Naraghi, S.B.; Littlefield, R.B.; Schup-Magoffin, P.J.; Christman, K.L. Naturally derived myocardial matrix as an injectable scaffold for cardiac tissue engineering. Biomaterials 2009, 30, 5409–5416. [Google Scholar]
- Shaikh, F.M.; Callanan, A.; Kavanagh, E.G.; Burke, P.E.; Grace, P.A.; McGloughlin, T.M. Fibrin: A natural biodegradable scaffold in vascular tissue engineering. Cells Tissues Organs 2008, 188, 333–346. [Google Scholar]
- Birla, R.K.; Borschel, G.H.; Dennis, R.G.; Brown, D.L. Myocardial engineering in vivo: Formation and characterization of contractile, vascularized three-dimensional cardiac tissue. Tissue Eng. 2005, 11, 803–813. [Google Scholar]
- Hecker, L.; Khait, L.; Radnoti, D.; Birla, R. Development of a microperfusion system for the culture of bioengineered heart muscle. ASAIO J. 2008, 54, 284–294. [Google Scholar]
- Huang, Y.; Khait, L.; Birla, R.K. Contractile three-dimensional bioengineered heart muscle for myocardial regeneration. J. Biomed. Mater. Res. A 2007, 80, 719–731. [Google Scholar]
- Birla, R.K.; Huang, Y.C.; Dennis, R.G. Development of a novel bioreactor for the mechanical loading of tissue-engineered heart muscle. Tissue Eng. 2007, 13, 2239–2248. [Google Scholar]
- Black, L.D.; Meyers, J.D.; Weinbaum, J.S.; Shvelidze, Y.A.; Tranquillo, R.T. Cell-induced alignment augments twitch force in fibrin gel-based engineered myocardium via gap junction modification. Tissue Eng. A 2009, 15, 3099–3108. [Google Scholar]
- Arnaoutova, I.; George, J.; Kleinman, H.K.; Benton, G. The endothelial cell tube formation assay on basement membrane turns 20: State of the science and the art. Angiogenesis 2009, 12, 267–274. [Google Scholar]
- Anderl, J.N.; Robey, T.E.; Stayton, P.S.; Murry, C.E. Retention and biodistribution of microspheres injected into ischemic myocardium. J. Biomed. Mater. Res. A 2009, 88, 704–710. [Google Scholar]
- Copland, I.B.; Jolicoeur, E.M.; Gillis, M.; Cuerquis, J.; Eliopoulos, N.; Annabi, B.; Calderone, A.; Tanguay, J.; Ducharme, A.; Galipeau, J. Coupling erythropoietin secretion to mesenchymal stromal cells enhances their regenerative properties. Cardiovasc. Res. 2008, 79, 405–415. [Google Scholar]
- Giraud, M.; Ayuni, E.; Cook, S.; Siepe, M.; Carrel, T.P.; Tevaearai, H.T. Hydrogel-based engineered skeletal muscle grafts normalize heart function early after myocardial infarction. Artif. Org. 2008, 32, 692–700. [Google Scholar]
- Kutschka, I.; Chen, I.Y.; Kofidis, T.; Arai, T.; von Degenfeld, G.; Sheikh, A.Y.; Hendry, S.L.; Pearl, J.; Hoyt, G.; Sista, R.; Yang, P.C.; Blau, H.M.; Gambhir, S.S.; Robbins, R.C. Collagen matrices enhance survival of transplanted cardiomyoblasts and contribute to functional improvement of ischemic rat hearts. Circulation 2006, 114, I167–173. [Google Scholar]
- Hansen, A.; Eder, A.; Bnstrup, M.; Flato, M.; Mewe, M.; Schaaf, S.; Aksehirlioglu, B.; Schwrer, A.; Uebeler, J.; Eschenhagen, T. Development of a drug screening platform based on engineered heart tissue. Circ. Res. 2010, 107, 35–44. [Google Scholar]
- Cholewinski, E.; Dietrich, M.; Flanagan, T.C.; Schmitz-Rode, T.; Jockenhoevel, S. Tranexamic acid—an alternative to aprotinin in fibrin-based cardiovascular tissue engineering. Tissue Eng. A 2009, 15, 3645–3653. [Google Scholar]
- Odian, G.G. Principles of Polymerization; John Wiley and Sons: New York, NY, USA, 2004; p. 838. [Google Scholar]
- Motlagh, D.; Senyo, S.E.; Desai, T.A.; Russell, B. Microtextured substrata alter gene expression, protein localization and the shape of cardiac myocytes. Biomaterials 2003, 24, 2463–2476. [Google Scholar]
- Jongpaiboonkit, L.; King, W.J.; Lyons, G.E.; Paguirigan, A.L.; Warrick, J.W.; Beebe, D.J.; Murphy, W.L. An adaptable hydrogel array format for 3-dimensional cell culture and analysis. Biomaterials 2008, 29, 3346–3356. [Google Scholar]
- Wang, T.; Jiang, X.; Tang, Q.; Li, X.; Lin, T.; Wu, D.; Zhang, X.; Okello, E. Bone marrow stem cells implantation with alpha-cyclodextrin/MPEG-PCL-MPEG hydrogel improves cardiac function after myocardial infarction. Acta Biomater. 2009, 5, 2939–2944. [Google Scholar]
- Allder, M.A.; Guilbeau, E.J.; Brandon, T.A.; Walker, A.S.; Koeneman, J.B.; Fisk, R.L. A hydrogel pericardial patch. ASAIO Trans. Am. Soc. Artif. Internal Organs 1990, 36, M572–M574. [Google Scholar]
- Walker, A.S.; Blue, M.A.; Brandon, T.A.; Emmanual, J.; Guilbeau, E.J. Performance of a hydrogel composite pericardial substitute after long-term implant studies. ASAIO J. 1992, 38, M550–M554. [Google Scholar]
- Miyagawa, S.; Sawa, Y.; Sakakida, S.; Taketani, S.; Kondoh, H.; Memon, I.A.; Imanishi, Y.; Shimizu, T.; Okano, T.; Matsuda, H. Tissue cardiomyoplasty using bioengineered contractile cardiomyocytes sheets to repair damaged myocardium: Their integration with recipient myocardium. Transplantation 2005, 80, 1586–1595. [Google Scholar]
- Naito, H.; Takewa, Y.; Mizuno, T.; Ohya, S.; Nakayama, Y.; Tatsumi, E.; Kitamura, S.; Takano, H.; Taniguchi, S.; Taenaka, Y. Three-dimensional cardiac tissue engineering using a thermoresponsive artificial extracellular matrix. ASAIO J. 2004, 50, 344–348. [Google Scholar]
- Guan, J.; Hong, Y.; Ma, Z.; Wagner, W.R. Protein-reactive, thermoresponsive copolymers with high flexibility and biodegradability. Biomacromolecules 2008, 9, 1283–1292. [Google Scholar]
- Fujimoto, K.L.; Ma, Z.; Nelson, D.M.; Hashizume, R.; Guan, J.; Tobita, K.; Wagner, W.R. Synthesis, characterization and therapeutic efficacy of a biodegradable, thermoresponsive hydrogel designed for application in chronic infarcted myocardium. Biomaterials 2009, 30, 4357–4368. [Google Scholar]
- Wang, F.; Li, Z.; Khan, M.; Tamama, K.; Kuppusamy, P.; Wagner, W.R.; Sen, C.K.; Guan, J. Injectable, rapid gelling and highly flexible hydrogel composites as growth factor and cell carriers. Acta Biomater. 2010, 6, 1978–1991. [Google Scholar]
- Li, Z.; Wang, F.; Roy, S.; Sen, C.K.; Guan, J. Injectable, highly flexible, and thermosensitive hydrogels capable of delivering superoxide dismutase. Biomacromolecules 2009, 10, 3306–3316. [Google Scholar]
- Kim, P.D.; Peyton, S.R.; Van Strien, A.J.; Putnam, A.J. The influence of ascorbic acid, TGF-beta1, and cell-mediated remodeling on the bulk mechanical properties of 3-D PEG-fibrinogen constructs. Biomaterials 2009, 30, 3854–3864. [Google Scholar]
- Zhang, G.; Nakamura, Y.; Wang, X.; Hu, Q.; Suggs, L.J.; Zhang, J. Controlled release of stromal cell-derived factor-1 alpha in situ increases c-kit+ cell homing to the infarcted heart. Tissue Eng. 2007, 13, 2063–2071. [Google Scholar]
- Miller, J.S.; Shen, C.J.; Legant, W.R.; Baranski, J.D.; Blakely, B.L.; Chen, C.S. Bioactive hydrogels made from step-growth derived PEG-peptide macromers. Biomaterials 2010, 31, 3736–3743. [Google Scholar]
- LaNasa, S.M.; Bryant, S.J. Influence of ECM proteins and their analogs on cells cultured on 2-D hydrogels for cardiac muscle tissue engineering. Acta Biomater. 2009, 5, 2929–2938. [Google Scholar]
- Poon, Y.F.; Cao, Y.; Zhu, Y.; Judeh, Z.M.A.; Chan-Park, M.B. Addition of beta-malic acidcontaining poly(ethylene glycol) dimethacrylate to form biodegradable and biocompatible hydrogels. Biomacromolecules 2009, 10, 2043–2052. [Google Scholar]
- Kim, J.; Park, Y.; Tae, G.; Lee, K.B.; Hwang, S.J.; Kim, I.S.; Noh, I.; Sun, K. Synthesis and characterization of matrix metalloprotease sensitive-low molecular weight hyaluronic acid based hydrogels. J. Mater. Sci. Mater. Med. 2008, 19, 3311–3318. [Google Scholar]
- Peyton, S.R.; Raub, C.B.; Keschrumrus, V.P.; Putnam, A.J. The use of poly(ethylene glycol) hydrogels to investigate the impact of ECM chemistry and mechanics on smooth muscle cells. Biomaterials 2006, 27, 4881–4893. [Google Scholar]
- DeLong, S.A.; Gobin, A.S.; West, J.L. Covalent immobilization of RGDS on hydrogel surfaces to direct cell alignment and migration. J. Control. Release 2005, 109, 139–148. [Google Scholar]
- Lin, C.; Metters, A.T.; Anseth, K.S. Functional PEG-peptide hydrogels to modulate local inflammation inducedby the pro-inflammatory cytokine TNF[alpha]. Biomaterials 2009, 30, 4907–4914. [Google Scholar]
- Christman, K.L.; Lee, R.J. Biomaterials for the treatment of myocardial infarction. J. Am. Coll. Cardiol. 2006, 48, 907–913. [Google Scholar]
- Reffelmann, T.; Kloner, R.A. Cellular cardiomyoplasty-cardiomyocytes, skeletal myoblasts, or stem cells for regenerating myocardium and treatment of heart failure? Cardiovasc. Res. 2003, 58, 358–368. [Google Scholar]
- Hou, D.; Youssef, E.A.; Brinton, T.J.; Zhang, P.; Rogers, P.; Price, E.T.; Yeung, A.C.; Johnstone, B.H.; Yock, P.G.; March, K.L. Radiolabeled cell distribution after intramyocardial, intracoronary, and interstitial retrograde coronary venous delivery: Implications for current clinical trials. Circulation 2005, 112, 1150–1156. [Google Scholar]
- Jawad, H.; Ali, N.N.; Lyon, A.R.; Chen, Q.Z.; Harding, S.E.; Boccaccini, A.R.J. Myocardial tissue engineering: A review. Tissue Eng. Regen. Med. 2007, 1, 327–342. [Google Scholar]
- Zimmermann, W.H.; Eschenhagen, T. Cardiac tissue engineering for replacement therapy. Heart Fail Rev. 2003, 8, 259–269. [Google Scholar]
- Rubart, M.; Pasumarthi, K.B.S.; Nakajima, H.; Soonpaa, M.H.; Nakajima, H.O.; Field, L.J. Physiological coupling of donor and host cardiomyocytes after cellular transplantation. Circ. Res. 2003, 92, 1217–1224. [Google Scholar]
- Soonpaa, M.H.; Koh, G.Y.; Klug, M.G.; Field, L.J. Formation of nascent intercalated disks between grafted fetal cardiomyocytes and host myocardium. Science 1994, 264, 98–101. [Google Scholar]
- Li, R.K.; Weisel, R.D.; Mickle, D.A.; Jia, Z.Q.; Kim, E.J.; Sakai, T.; Tomita, S.; Schwartz, L.; Iwanochko, M.; Husain, M.; Cusimano, R.J.; Burns, R.J.; Yau, T.M. Autologous porcine heart cell transplantation improved heart function after a myocardial infarction. J. Thorac. Cardiovasc. Surg. 2000, 119, 62–68. [Google Scholar]
- Reinlib, L.; Field, L. Cell transplantation as future therapy for cardiovascular disease?: A workshop of the National Heart, Lung, and Blood Institute. Circulation 2000, 101, E182–E187. [Google Scholar]
- Leobon, B.; Garcin, I.; Menasche, P.; Vilquin, J.; Audinat, E.; Charpak, S. Myoblasts transplanted into rat infarcted myocardium are functionally isolated from their host. Proc. Natl. Acad. Sci. USA 2003, 100, 7808–7811. [Google Scholar]
- Yang, M.; Wang, S.; Chou, N.; Chi, N.; Huang, Y.; Chang, Y.; Shieh, M.; Chung, T. The cardiomyogenic differentiation of rat mesenchymal stem cells on silk fibroin-polysaccharide cardiac patches in vitro. Biomaterials 2009, 30, 3757–3765. [Google Scholar]
- Toma, C.; Pittenger, M.F.; Cahill, K.S.; Byrne, B.J.; Kessler, P.D. Human mesenchymal stem cells differentiate to a cardiomyocyte phenotype in the adult murine heart. Circulation 2002, 105, 93–98. [Google Scholar]
- Heng, B.C.; Haider, H.K.; Sim, E.K.; Cao, T.; Ng, S.C. Strategies for directing the differentiation of stem cells into the cardiomyogenic lineage in vitro. Cardiovasc. Res. 2004, 62, 34–42. [Google Scholar]
- Kehat, I.; Kenyagin-Karsenti, D.; Snir, M.; Segev, H.; Amit, M.; Gepstein, A.; Livne, E.; Binah, O.; Itskovitz-Eldor, J.; Gepstein, L. Human embryonic stem cells can differentiate into myocytes with structural and functional properties of cardiomyocytes. J. Clin. Invest. 2001, 108, 407–414. [Google Scholar]
- Klug, M.G.; Soonpaa, M.H.; Koh, G.Y.; Field, L.J. Genetically selected cardiomyocytes from differentiating embronic stem cells form stable intracardiac grafts. J. Clin. Invest. 1996, 98, 216–224. [Google Scholar]
- Doetschman, T.C.; Eistetter, H.; Katz, M.; Schmidt, W.; Kemler, R. The in vitro development of blastocyst-derived embryonic stem cell lines: Formation of visceral yolk sac, blood islands and myocardium. J. Embryol. Exp. Morphol. 1985, 87, 27–45. [Google Scholar]
- Yu, J.; Vodyanik, M.A.; Smuga-Otto, K.; Antosiewicz-Bourget, J.; Frane, J.L.; Tian, S.; Nie, J.; Jonsdottir, G.A.; Ruotti, V.; Stewart, R.; Slukvin, I.I.; Thomson, J.A. Induced pluripotent stem cell lines derived from human somatic cells. Science 2007, 318, 1917–1920. [Google Scholar]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131, 861–872. [Google Scholar]
- Nelson, T.J.; Martinez-Fernandez, A.; Yamada, S.; Perez-Terzic, C.; Ikeda, Y.; Terzic, A. Repair of acute myocardial infarction by human stemness factors induced pluripotent stem cells. Circulation 2009, 120, 408–416. [Google Scholar]
- Curtis, M.W.; Sharma, S.; Desai, T.A.; Russell, B. Hypertrophy, gene expression, and beating of neonatalcardiac myocytes are affected by microdomain heterogeneityin 3D. Biomed. Microdev. 2010, 12, 1073–85. [Google Scholar]
- Cheng, K.; Li, T.S.; Malliaras, K.; Davis, D.; Zhang, Y.; Marbn, E. Magnetic targeting enhances engraftment and functional benefit of iron-labeled cardiosphere-derived cells in myocardial infarction. Circ. Res. 2010, 106, 1570–1581. [Google Scholar]
- Davis, D.R.; Kizana, E.; Terrovitis, J.; Barth, A.S.; Zhang, Y.; Smith, R.R.; Miake, J.; Marbn, E. Isolation and expansion of functionally-competent cardiac progenitor cells directly from heart biopsies. J. Mol. Cell Cardiol 2010, 49, 312–321. [Google Scholar]
- Davis, D.R.; Zhang, Y.; Smith, R.R.; Cheng, K.; Terrovitis, J.; Malliaras, K.; Li, T.S.; White, A.; Makkar, R.; Marbn, E. Validation of the cardiosphere method to culture cardiac progenitor cells from myocardial tissue. PLoS One 2009, 4, e7195. [Google Scholar]
- Bian, W.; Bursac, N. Engineered skeletal muscle tissue networks with controllable architecture. Biomaterials 2009, 30, 1401–1412. [Google Scholar]
- Bian, W.; Liau, B.; Badie, N.; Bursac, N. Mesoscopic hydrogel molding to control the 3D geometry of bioartificial muscle tissues. Nat. Protoc. 2009, 4, 1522–1534. [Google Scholar]
- Engler, A.J.; Sen, S.; Sweeney, H.L.; Discher, D.E. Matrix elasticity directs stem cell lineage specification. Cell 2006, 126, 677–689. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, Z.; Guan, J. Hydrogels for Cardiac Tissue Engineering. Polymers 2011, 3, 740-761. https://doi.org/10.3390/polym3020740
Li Z, Guan J. Hydrogels for Cardiac Tissue Engineering. Polymers. 2011; 3(2):740-761. https://doi.org/10.3390/polym3020740
Chicago/Turabian StyleLi, Zhenqing, and Jianjun Guan. 2011. "Hydrogels for Cardiac Tissue Engineering" Polymers 3, no. 2: 740-761. https://doi.org/10.3390/polym3020740
APA StyleLi, Z., & Guan, J. (2011). Hydrogels for Cardiac Tissue Engineering. Polymers, 3(2), 740-761. https://doi.org/10.3390/polym3020740





