Synthesis of Novel π-Conjugated Rod-Rod-Rod Triblock Copolymers Containing Poly(3-hexylthiophene) and Polyacetylene Segments by Combination of Quasi-Living GRIM and Living Anionic Polymerization
Abstract
: The first successful synthesis of a new rod-rod-rod triblock copolymer, polyacetylene(PA)-b-poly(3-hexylthiophene)(P3HT)-b-PA could be synthesized by a combination of quasi-living Grignard metathesis (GRIM) and living anionic polymerization. We first confirmed that poly(4-tolyl vinyl sulfoxide) (PTVS), which is a soluble precursor for PA, could be synthesized by living anionic polymerization in THF at −78 °C , initiated with 3-methyl-1,1-diphenylpentyllithium as the initiator in the presence of in situ-generated lithium enolate. The molecular weights (MWs) and polydispersities (PDIs) were well controlled (MW = 5,200–27,000, PDI = 1.10–1.22), respectively. A coil-rod-coil triblock copolymer, PTVS-b-P3HT-b-PTVS, (6,000-12,500-6,000) could also be synthesized, initiated with a P3HT-based difunctional macroinitiator in the presence of lithium enolate. GPC-RALLS and 1H NMR analyses confirmed a high degree of structural homogeneity of PTVS-b-P3HT-b-PTVS. A thermal transformation reaction of the polymer was carried out in the film state at 170 °C for 2 h to afford PA-b-P3HT-b-PA quantitatively, as monitored by TGA and FT-IR analyses. The optical and electronic properties as well as the morphological behavior of the block copolymers were investigated by UV-vis spectroscopy, conductivity measurement, and AFM observation.1. Introduction
Much attention has recently been paid to block copolymers (BCP)s containing π-conjugated stiff-rod segments, because of their unique self-assembled structures in addition to their optical and electronic properties [1,2]. Many potential applications utilizing such materials include batteries, capacitors, light-emitting diodes, transistors, photovoltaics, and so on, by exploiting their functionality. Among them, regioregular P3HT has so far been the best class of balanced high-performance materials as p-type semiconductors in terms of solubility, chemical stability, charge mobility, and commercial availability [3-6]. A well-defined BCP with a P3HT segment, PS-b-oligothiophene(11-mer)-b-PS, was first prepared by a combination of living anionic polymerization and stepwise organic synthesis by Hempenius et al. [7]. Since McCullough et al. discovered the quasi-living Grignard metathesis (GRIM) polymerization system for regioregular P3HT [8], followed by the improvement by Yokozawa et al. [9], various BCPs containing P3HT segments have been synthesized [10-52].
Although there are many examples of rod-coil P3HT-based BCPs [10-40], all conjugated rod-rod P3HT-based BCPs [41-52] are still less focused on and limited only to diblock copolythiophene derivatives, regardless of the expectation of high performance as semiconductors, due to the absence of insulating coil segments. Two exceptional examples of well-defined all conjugated rod-rod BCPs, P3HT-b-poly(2,6-dialkoxyphenylene) [53] and P3HT-b-poly(9,9-dioctylfluorene) [54], were very recently synthesized, comprised of different conjugated backbone structures, based on sequential quasi-living GRIM polymerization. However, these counter blocks, polyphenylenes and polyfluorenes, have large bandgaps and are not suited especially for solar cell application. In addition, the introduction of insulating long alkyl segments should be minimized to maintain charge mobility and conductivity.
Polyacetylene (PA) is the simplest conjugated hydrocarbon polymer without side chains, showing helical rod-like structures [55-57]. It normally displays low bandgaps and high conductivity, particularly after doping [55]. Although PA itself is insoluble, the thermal transformation approach from soluble precursory polymers makes it possible to form a homogeneous PA film obtained by solution casting. Hogen-Esch et al. successfully synthesized well-defined PA by the living anionic polymerization of phenyl vinyl sulfoxide, followed by thermal elimination of sulfenic acid from poly(phenyl vinyl sulfoxide) [58]. They also prepared PA-b-polystyrene (PS) and PA-b-PS-b-PA by a sequential living anionic polymerization technique. Hirao et al. applied this living system to prepare more complex BCPs and star polymers containing PA segments using precursory PTVS [59,60].
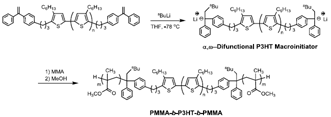
In the course of our study, a facile synthetic method has been developed by a combination of quasi-living GRIM and living anionic polymerization, to synthesize a variety of P3HT-based coil-rod-coil triblock copolymers containing PS, poly(4-vinyltriphenylamine), or PMMA as outer coil segments [16-18]. In the synthesis of PMMA-b-P3HT-b-PMMA [17], we found that an α,ω-difunctional P3HT macroinitiator with a 1,1-diphenylethylene (DPE) anion is efficient for the living anionic polymerization of MMA (Scheme 1). Since a DPE anion can also initiate the living polymerization of tolyl vinyl sulfoxide (TVS) [59,60], well-defined rod-rod-rod PA-b-P3HT-b-P triblock copolymers may possibly be synthesized by using the α,ω-difunctional P3HT macroinitiator for TVS polymerization, followed by thermal transformation (Scheme 2).
Herein we describe the first successful synthesis of PA-b-P3HT-b-PA which has never been reported before, by a combination of quasi-living GRIM and living anionic polymerization based on DPE chemistry. The combination of P3HT and PA segments is intriguing because of the attractive features of the PA counter block, such as the absence of insulating side chains, high conductivity, and efficient light harvesting. The formation of self-assembling nanostructures of PA-b-P3HT-b-PA and the possible creation of nanoporous films are also described.
2. Experimental Section
2.1. Materials
All reagents were purchased from Sigma-Aldrich Corp. (>99%) and used as received unless otherwise stated. Tetrahydrofuran (THF, 99.5%, Wako Chemicals Co., Ltd.) was refluxed over sodium benzophenone under nitrogen for 2 h, then distilled just before use. THF for the solvent of living anionic polymerization was refluxed over Na wire, then distilled from lithium aluminum hydride. It was finally distilled from sodium naphthalenide on a high vacuum line. 1,1-Diphenylethylene (DPE) was distilled from calcium hydride and 1,1-diphenylhexylthium under reduced pressure. α,ω-DPE difunctional P3HT (DPE-P3HT-DPE) [16] and TVS [59] were synthesized and purified according to the previous papers.
2.2. Living Anionic Polymerization of TVS
Living anionic polymerization of TVS was performed by using a standard high vacuum technique. In a typical experiment, sec-BuLi (2.0 mmol) was added to a THF solution (5.0 mL) of DPE (0.100 mmol) at −78 °C, and the solution was warmed to room temperature and kept for 1 h. After the solution was again cooled to −78 °C, a THF solution of TVS (0.50 g, 3.0 mmol) was added with vigorous stirring. After 8 h, the solution was poured into a mixture of methanol/water (90 mL/10 mL) to precipitate the polymer. The polymer was purified by reprecipitation using THF and methanol/water, followed by freeze-drying from its absolute benzene solution to afford PTVS as a white solid (0.48 g, 92%).
2.3. Synthesis of PTVS-b-P3HT-b-PTVS
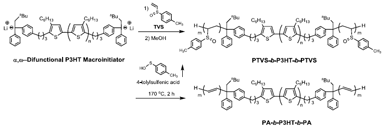
PTVS-b-P3HT-b-PTVS was synthesized by using a standard high vacuum technique. To a THF solution (50.0 mL) of DPE-P3HT-DPE (Mn = 12,500, PDI = 1.10, r.r. = 97%, 0.100 g, 0.008 mmol) sec-BuLi (0.320 mmol) was added at −78 °C, and the solution was warmed to room temperature and kept for 1 h. After the solution was cooled to −78 °C, a THF solution of TVS (0.628 mmol) was added with vigorous stirring. After 24 h, the solution was poured into a mixture of methanol/water (90 mL/10 mL) to precipitate the polymer. The polymer was purified by fractional precipitation using ethyl acetate, followed by freeze-drying from its absolute benzene solution to afford PTVS-b-P3HT-b-PTVS as a purple solid (0.120 g, 61%).
2.4. Synthesis of PA-b-P3HT-b-PA
PTVS-b-P3HT-b-PTVS (1.0 μm thickness) was obtained by spin coating from a chloroform solution (10 mg/mL) onto a glass substrate. The film was dried under vacuum and then heated at 170 °C for 2 h. After cooling down to room temperature and purging with nitrogen, a dark purple PA-b-P3HT-b-PA film was obtained.
2.5. Measurements
Molecular weights (MWs) and polydispersity indices (PDIs) were measured with a HPLC system equipped with a pump, refractometer (RI), absorbance detector (UV, λ = 254 nm), online RALLS detector (λ = 670 nm), sample processor, and polystyrene gel columns connected in the following series: 650, 200, and 75 Å. THF was used as a carrier solvent at a flow rate of 1.0 mL/min at room temperature. 1H NMR was recorded on a Bruker DPX (300 MHz) in chloroform-d calibrated to tetramethylsilane as an internal standard (δH 0.00). TGA was performed on a Seiko EXSTAR 6000 TG/DTA 6300 thermal analyzer at a heating rate of 10 °C/min. FT-IR spectra were measured by a Horiba FT-120 Fourier transform spectrophotometer. UV-vis absorption spectra of a polymer solution in chloroform and the thin film were recorded on a Jasco FP-750 spectrometer over a wavelength range of 250–800 nm. The films were obtained by spin coating from a toluene solution, followed by drying under vacuum. AFM phase images were taken with a SII-NT SPA 400 operating in a tapping mode. Electric conductivity was measured by the typical four point probe technique using a cast film from a toluene solution (5 mg/mL) onto a glass substrate.
3. Results and Discussion
3.1. Living Anionic Polymerization of TVS
As mentioned in the Introduction, Hogen-Esch et al. demonstrated that the polymerization of phenyl vinyl sulfoxide proceeded in a living manner to afford polymers with controlled MWs and relatively low PDIs in the range of 1.2 to 1.4 [58]. They also succeeded in a quantitative transformation reaction into PA simply by thermal treatment. By this success, poly(phenyl vinyl sulfoxide) obtained by living anionic polymerization becomes an advantageous PA precursor, soluble in organic solvents and well-controlled in chain length. Recently, Hirao reported that the use of a DPE anion as the initiator in the presence of a 10 ∼ 20 fold excess of lithium halides (LiCl or LiBr) is effective to further reduce the PDI value (PDI = 1.07–1.2) of a similar polymer, PTVS, which could also be thermally converted to PA in 100% yield [59,60]. However, the addition of a large amount of salts in polymerization media requires a thorough drying process of the salts. In a macroinitiator system, this becomes quite a serious problem, because even a small amount of contamination from salts affords a mixture of a deactivated homopolymer with the expected BCP. Therefore, a new living system has to be developed, especially suited for the macroinitiator system.
To prepare the α,ω-difunctional P3HT macroinitiator, if a precursory DPE difunctional P3HT (DPE-P3HT-DPE) is reacted with a large excess of sec-butyllithium (sec-BuLi) toward the DPE moieties, the expected macroinitiator can be obtained by an addition reaction of sec-BuLi with the terminal DPE moieties, leaving an excess of sec-BuLi. However, sec-BuLi can selectively decompose with a THF solvent into ethylene gas and lithium enolate by elevating the temperature to 25 °C (Scheme 3) [17]. It should be noted that stabler initiating DPE anions survive under this condition and the generated ethylene and lithium enolate are inert in the living polymerization [17]. We expected that the in situ-generated lithium enolate would function to lower the PDI of PTVS instead of the lithium halides.
A model homopolymerization of TVS was carried out in THF initiated by 3-methyl-1,1-diphenylpentyllithium in the presence of lithium enolate, derived from DPE and a large excess of sec-BuLi (Scheme 4). The results are summarized in Table 1. The molecular weight ranges from 5,200 to 27,000 with low PDI (1.10–1.22), as expected. The representative SEC profile is shown in Figure 1. In a blank experiment, the PDI increases to around 1.5 without lithium enolate. Consequently, the in situ-generated salt, lithium enolate, is found to be effective to obtain PTVS with controlled MWs and low PDIs.
3.2. Synthesis of PTVS-b-P3HT-b-PTVS
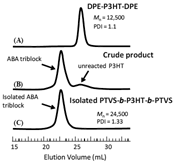
To synthesize PTVS-b-P3HT-b-PTVS, precursory DPE difunctional P3HT (DPE-P3HT-DPE, Mn = 12,500, PDI = 1.10, r.r. = 97%) was reacted with a 20-fold excess of sec-butyllithium (sec-BuLi) toward the DPE moieties in THF at −78 °C for 10 min. The excess sec-BuLi decomposed with THF by warming to room temperature and keeping for 1 h, resulting in in situ generation of excess lithium enolate. The system was then cooled back to −78 °C. TVS was next added at −78 °C to start the living anionic polymerization, followed by quenching with degassed methanol (Scheme 2). As shown in the SEC chromatograms (Figure 2), the peak top for the precursory DPE-P3HT-DPE obviously shifts to a higher molecular weight region after the polymerization. Unfortunately, there is a minor shoulder corresponding to unreacted DPE-P3HT-DPE, probably because of incomplete initiation from the partially aggregated macroinitiator in THF at −78 °C , similar to the case in the synthesis of PMMA-b-P3HT-b-PMMA [17]. Eventually, it was successfully eliminated from the mixture by fractional precipitation using ethyl acetate. The isolated PTVS-b-P3HT-b-PTVS possesses a unimodal sharp peak (Figure 2). The Mn value of PTVS-b-P3HT-b-PTVS, determined by SEC-RALLS (24,500), is in good agreement with the calculated one (22,300). The PDI value determined by SEC is acceptable (1.33).
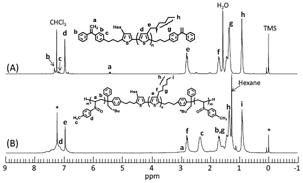
Moreover, 1H NMR spectroscopy of PTVS-b-P3HT-b-PTVS confirms the absence of a signal for the vinyl protons of the DPE moieties at 5.4 ppm, as well as a reasonable composition of two segments being almost consistent with that targeted (Figure 3(B)). Thus, the expected PTVS-b-P3HT-b-PTVS (6,000-12,500-6,000) could be successfully synthesized. It should be noted that the DPE chemistry employed here is quite elegant and efficient for designing a wide range of well-defined BCPs, because the 1,1-diphenylalkyl anion is able to undergo living anionic polymerization of many other kinds of monomers, acrylates, acryl amides, vinylpyridines, epoxides, propylene sulfide, and so on.
3.3. Synthesis of PA-b-P3HT-b-PA
The transformation from PTVS-b-P3HT-b-PTVS into PA-b-P3HT-b-PA was performed simply by thermal treatment at 170 °C for 2 h under vacuum (Scheme 2). Figure 4(A) shows the TGA curve of PTVS-b-P3HT-b-PTVS. The first weight-loss from 120 to 250 °C may be ascribed to the elimination of 4-tolylsulfenic acid from the PTVS segments, as confirmed by the weight-loss behavior of a PTVS homopolymer (Figure 4(B)). The weight loss percentage at temperatures in the range of 120 to 250 °C was nearly consistent with that assumed by complete elimination. The second weight-loss was seen from 350 to 470 °C, corresponding to the degradation of the PA-b-P3HT-b-PA segments.
The FT-IR spectrum of PTVS-b-P3HT-b-PTVS (Figure 5(A)) showed a strong absorption band at 1,045 cm−1, assignable to S=O stretching, which completely disappeared after thermal treatment. Instead, an absorption band at 1,005 cm−1, attributed to trans-vinylene out-of-plane deformation from PA main chains, newly appeared (Figure 5(B)). In addition, the polymer became insoluble in common organic solvents, while precursory PTVS-b-P3HT-b-PTVS was well soluble in toluene, chloroform, and THF.
These analytical results clearly confirmed the complete conversion from PTVS to PA chains by quantitatively eliminating 4-tolylsulfenic acid in PTVS-b-P3HT-b-PTVS.
3.4. Optical and Electronic Properties
The optical property of PTVS-b-P3HT-b-PTVS was studied by UV-vis spectroscopy. The solution-state UV-vis spectrum of PTVS-b-P3HT-b-PTVS in chloroform showed maximum absorption (λmax) for the π–π* transition of the P3HT inner segment at around 442 nm (Figure 6 (yellow line)). In the film state, the λmax value of the polymer was bathochromically shifted to around 565 nm compared with that in the solution state (pink line). In addition, the polymer showed a shoulder at around 610 nm related to vibronic absorption, similar to the pristine P3HT film (purple line). This observation indicates a high degree of ordering in the polymer films even if PTVS is covalently linked to P3HT. After thermal transformation, the absorbance was somewhat red-shifted to obtain an off-set value at a longer wavelength (∼700 nm, green line)). This indicates the successful conversion from PTVS to PA segments in the PTVS-b-P3HT-b-PTVS film. Interestingly, even after generating a highly rigid-rod PA segment, the shoulder at around 610 nm remains intact, indicative of highly ordered P3HT packing structures. Therefore, the thermal transformation reaction seems not to destroy the self-assembled structures, derived from precursory PTVS-b-P3HT-b-PTVS in the film state.
Electrical conductivity of PA-b-P3HT-b-PA was then measured by using a typical four-point probe technique. After iodine doping, relatively high conductivity of 0.126 S/cm was obtained, while overflow resistance was obtained for an undoped sample of PA-b-P3HT-b-PA. In order to improve the conductivity to more than 10 S/cm, a film-forming condition to obtain nanofibril or nanowire structures may be required [61].
3.5. AFM Images of Block Copolymers
To gain insight into the morphology, a thin film of PTVS-b-P3HT-b-PTVS was prepared by drop casting from a toluene solution onto a mica substrate at room temperature. The film thickness was estimated to be 100 nm by Dektak3. Figure 7(A) and Figure 7(B) show tapping-mode AFM height and phase images of the PTVS-b-P3HT-b-PTVS thin film surface, respectively, after solvent annealing with toluene vapor at room temperature for 3 h. Although a periodic microphase-separated morphology is not observed, continuous nanofibril structures are observed at 10–20 nm sizes in whole areas, without macrophase separation or large aggregation. Figure 7(C) and Figure 7(D) show tapping-mode AFM height and phase images of the PA-b-P3HT-b-PA thin film obtained by a thermal transformation reaction of PTVS-b-P3HT-b-PTVS on a mica substrate. As can be seen, there is almost no difference in morphology before and after the transformation, which might be coincident with the observation by UV-vis spectroscopy showing a high degree of ordering in P3HT packing even after the thermal transformation. In contrast, the roughness determined by height images significantly changed after the transformation from 6.5 to 17.6 nm. This result suggests that the PA-b-P3HT-b-PA film possesses discrete pits [22] in the matrix on a nano-scale, probably generated by eliminating ca. 85 vol% of a large leaving group, 4-tolylsulfenic acid, from PTVS domains, which are anchored by rigid and crystalline P3HT domains in the film state. Such semiconducting nanoporous films are quite interesting materials, especially for solar cell device applications [62], after back-fill of n-type semiconductors into nanoporous matrices, possibly achieving large p/n interface areas with controlled morphological domain widths which correspond to exciton diffusion lengths.
4. Conclusions
Living anionic polymerization of TVS in THF at −78 °C , initiated with a DPE anion as the initiator, has been achieved in the presence of in situ-generated lithium enolate. The MWs of PTVS was controlled from 5,200 to 27,000 with relatively low PDI (1.10–1.22). Based on these results, PTVS-b-P3HT-b-PTVS (6,000-12,500-6,000) could be successfully synthesized using a P3HT-based difunctional macroinitiator with two DPE anions at both chain ends in the presence of lithium enolate. The high degree of structural homogeneity of the BCP was confirmed by GPC-RALLS and 1H NMR. The thermal transformation from PTVS-b-P3HT-b-PTVS to PA-b-P3HT-b-PA was quantitative, as monitored by TGA and FT-IR analyses. The synthesized rod-rod-rod type triblock copolymer, PA-b-P3HT-b-PA, has never been reported before. The optical and electronic properties of BCPs were investigated by UV-vis spectroscopy and conductivity measurement, respectively. Finally, tapping mode AFM images of PTVS-b-P3HT-b-PTVS and PA-b-P3HT-b-PA thin film surfaces showed continuous nanofibril structures. In particular, AFM height images of those surfaces suggest the creation of nanoporous structures in the PA-b-P3HT-b-PA film.







| [lithium enolate]/[DPE] | DPnb | Mn (g/mol) | PDI | ||
|---|---|---|---|---|---|
| calcdb | GPC-RALLS | 1H NMRc | |||
| 20 | 30 | 5,220 | 5,200 | 5,320 | 1.10 |
| 17 | 56 | 9,690 | 9,860 | 8,690d | 1.18 |
| 19 | 150 | 25,200 | 27,000 | - | 1.22 |
| 0 | 60 | 10,200 | 8,600 | 9,300 | 1.54 |
aYields of polymers are always more than 99%bCalculated from feed ratio of initiator and monomer.cCalculated from signal intensities assignable to 6 methyl protons of sec-butyl group (0.6–0.8 ppm) and aromatic protons of main chains (7.0–8.0 ppm).d1,1-Bis(4-trimethylsilylphenyl)ethylene was used instead of DPE for more accurate calculation of MWs by 1H NMR spectrum, showing a sharp signal assignable to trimethylsilyl terminal units at 0.2–0.3 ppm. The MWs were calculated from the signal intensities assignable to 18 protons of the trimethylsilyl group (0.2–0.3 ppm) and aromatic protons of the main chains (7.0–8.0 ppm).
Acknowledgments
We would like to thank the Japan Society for the Promotion of Science (JSPS), KAKENHI (20850014), as well as the Noguchi institute, Noguchi foundation, for supporting this work.
References
- Scherf, U.; Gutacker, A.; Koenen, N. All-conjugated block copolymers. Acc. Chem. Res. 2008, 41, 1086–1097. [Google Scholar]
- Lee, M.; Cho, B.K.; Zin, W.C. Supramolecular structures from rod-coil block copolymers. Chem. Rev. 2001, 101, 3869–3. [Google Scholar]
- Di, C.-A.; Yu, C.; Liu, Y.-Q.; Zhu, D.-B. High-performance organic field-effect transistors: Molecular design, device fabrication, and physical properties. J. Phys. Chem. B 2007, 111, 14083–14096. [Google Scholar]
- Kim, J.; Lee, Y.K.; Coates, N.E.; Moses, D.; Nguyen, T.-Q.; Dante, M.; Heeger, A.J. Efficient tandem polymer solar cells fabricated by all-solution processing. Science 2007, 317, 222–225. [Google Scholar]
- Thompson, B.C.; Fréchet, J.M.J. Polymer-fullerene composite solar cells. Angew. Chem. Int. Ed. 2008, 47, 58–77. [Google Scholar]
- Wei, Q.; Nishizawa, T.; Tajima, K.; Hashimoto, K. Self-organized buffer layers in organic solar cells. Adv. Mater. 2008, 20, 2211–2216. [Google Scholar]
- Hempenius, M.A.; Langeveld-Voss, B.M.W.; van Haare, J.A.E.H.; Janssen, R.A.J.; Sheiko, S.S.; Spatz, J.P.; Moeller, M.; Meijer, E.W. Polystyrene-oligothiophene-polystyrene triblock copolymer. J. Am. Chem. Soc. 1998, 120, 2798–2804. [Google Scholar]
- Iovu, M.C.; Sheina, E.E.; Gil, R.R.; McCullough, R.D. Experimental evidence for the quasi-“living” nature of the Grignard metathesis method for the synthesis of regioregular poly(3-alkylthiophenes). Macromolecules 2005, 38, 8649–8656. [Google Scholar]
- Miyakoshi, R.; Yokoyama, A.; Yokozawa, T. Catalyst-transfer polycondensation. Mechanism of Ni-catalyzed chain-growth polymerization leading to well-defined poly(3-hexylthiophene). J. Am. Chem. Soc. 2005, 127, 17542–17547. [Google Scholar]
- Jeffries-El, M.; Sauvé, G.; McCullough, R.D. In-situ end-group functionalization of regioregular poly(3-alkylthiophene) using the Grignard metathesis polymerization method. Adv. Mater. 2004, 16, 1017–1019. [Google Scholar]
- Jeffries-El, M.; Sauvé, G.; McCullough, R.D. Facile synthesis of end-functionalized regioregular poly(3-alkylthiophene)s via modified Grignard metathesis reaction. Macromolecules 2005, 38, 10346–10352. [Google Scholar]
- Iovu, M.C.; Jeffries-EL, M.E.; Sheina, E.; Cooper, J.R.; McCullough, R.D. Regioregular poly(3-alkylthiophene) conducting block copolymers. Polymer 2005, 46, 8582–8586. [Google Scholar]
- Radano, C.P.; Scherman, O. A.; Stingelin-Stutzmann, N.; Mueller, C.; Breiby, D.W.; Smith, P.; Janssen, R.A.J.; Meijer, E.W. Crystalline-crystalline block copolymers of regioregular poly(3-hexylthiophene) and polyethylene by ring-opening metathesis polymerization. J. Am. Chem. Soc. 2005, 127, 12502–12503. [Google Scholar]
- Iovu, M.C.; Craley, C.R.; Jeffries-EL, M.; Krankowski, A.B.; Zhang, R.; Kowalewski, T.; McCullough, R.D. Conducting regioregular polythiophene block copolymer nanofibrils synthesized by reversible addition fragmentation chain transfer polymerization (RAFT) and nitroxide mediated polymerization (NMP). Macromolecules 2007, 40, 4733–4735. [Google Scholar]
- Dai, C.-A.; Yen, W.-C.; Lee, Y.-H.; Ho, C.-C.; Su, W.-F. Facile synthesis of well-defined block copolymers containing regioregular poly(3-hexylthiophene) via anionic macroinitiation method and their self-assembly behavior. J. Am. Chem. Soc. 2007, 129, 11036–11038. [Google Scholar]
- Higashihara, T.; Ohshimizu, K.; Hirao, A.; Ueda, M. Facile synthesis of ABA triblock copolymer containing regioregular poly(3-hexylthiophene) and polystyrene segments via linking reaction of poly(styryl)lithium. Macromolecules 2008, 41, 9505–9507. [Google Scholar]
- Higashihara, T.; Ueda, M. Synthesis and characterization of a novel coil-rod-coil triblock copolymers comprised of regioregular poly(3-hexylthiophene) and poly(methyl methacrylate) segments. React. Funct. Polym. 2009, 69, 457–462. [Google Scholar]
- Higashihara, T.; Ueda, M. Living anionic polymerization of 4-vinyltriphenylamine for synthesis of novel block copolymers containing low-polydisperse poly(4-vinyltriphenylamine) and regioregular poly(3-hexylthiophene) segments. Macromolecules 2009, 42, 8794–8800. [Google Scholar]
- Higashihara, T.; Takahashi, A.; Tajima, S.; Jin, S.; Rho, Y.; Ree, M.; Ueda, M. Synthesis of block copolymers consisting of poly(3-hexylthiophene) and polystyrene segments through ionic interaction and their self-assembly behavior. Polym. J. 2010, 42, 43–50. [Google Scholar]
- Takahashi, A.; Rho, Y.; Higashihara, T.; Ahn, B.; Ree, M.; Ueda, M. Preparation of nanoporous poly(3-hexylthiophene) films based on a template system of block copolymers via ionic interaction. Macromolecules 2010, 43, 4843–4852. [Google Scholar]
- Tsai, J.-H.; Lai, Y.-C.; Higashihara, T.; Lin, C.-J.; Ueda, M.; Chen, W.-C. Enhancement of P3HT/PCBM photovoltaic efficiency using the surfactant of triblock copolymer containing poly(3-hexylthiophene) and poly(4-vinyltriphenylamine) segments. Macromolecules 2010, 43, 6085–6091. [Google Scholar]
- Boudouris, B.W.; Frisbie, C.D.; Hillmyer, M.A. Nanoporous poly(3-alkylthiophene) thin films generated from block copolymer templates. Macromolecules 2008, 41, 67–75. [Google Scholar]
- Boudouris, B.W.; Frisbie, C.D.; Hillmyer, M.A. Polylactide-polythiophene-polylactide triblock copolymers. Macromolecules 2010, 43, 3566–3569. [Google Scholar]
- Urien, M.; Erothu, H.; Cloutet, E.; Hiorns, R.C.; Vignau, L.; Cramail, H. Poly(3-hexylthiophene) based block copolymers prepared by Click chemistry. Macromolecules 2008, 41, 7033–7040. [Google Scholar]
- Richard, F.; Brochon, C.; Leclerc, N.; Eckhardt, D.; Heiser, T.; Hadziioannou, G. Design of a linear poly(3-hexylthiophene)/fullerene-based donor-acceptor rod-coil block copolymer. Macromol. Rapid Commun. 2008, 29, 885–891. [Google Scholar]
- Sommer, M.; Lang, A.S.; Thelakkat, M. Crystalline-crystalline donor-acceptor block copolymers. Angew. Chem., Int. Ed. 2008, 47, 7901–7904. [Google Scholar]
- Craley, C.R.; Zhang, R.; Kowalewski, T.; McCullough, R.D.; Stefan, M. Regioregular poly(3-hexylthiophene) in a novel conducting amphiphilic block copolymer. Macromol. Rapid Commun. 2009, 30, 11–16. [Google Scholar]
- Zou, J.; Khondaker, S.I.; Huo, Q.; Zhai, L. A general strategy to disperse and functionalize carbon nanotubes using conjugated block copolymers. Adv. Funct. Mater. 2009, 19, 479–483. [Google Scholar]
- Lee, J.U.; Cirpan, A.; Emrick, T.; Russell, T.P.; Jo, W.H. Synthesis and photophysical property of well-defined donor-acceptor diblock copolymer based on regioregular poly(3-hexylthiophene) and fullerene. J. Mater. Chem. 2009, 19, 1483–1489. [Google Scholar]
- Yang, C.; Lee, J.K.; Heeger, A.J.; Wudl, F. Well-defined donor-acceptor rod-coil diblock copolymers based on P3HT containing C60: The morphology and role as a surfactant in bulk-heterojunction solar cells. J. Mater. Chem. 2009, 19, 5416–5423. [Google Scholar]
- Zhang, Q.; Cirpan, A.; Russell, T.P.; Emrick, T. Donor-acceptor poly(thiophene-block-perylene diimide) copolymers: Synthesis and solar cell fabrication. Macromolecules 2009, 42, 1079–1082. [Google Scholar]
- Tao, Y.; McCulloch, B.; Kim, S.; Segalman, R.A. The relationship between morphology and performance of donor-acceptor rod-coil block copolymer solar cells. Soft Matter 2009, 5, 4219–4230. [Google Scholar]
- Alemseghed, M.G.; Gowrisanker, S.; Servello, J.; Stefan, M.C. Synthesis of di-block copolymers containing regioregular poly(3-hexylthiophene) and poly(tetrahydrofuran) by a combination of Grignard metathesis and cationic polymerizations. Macromol. Chem. Phys. 2009, 210, 2007–2014. [Google Scholar]
- Alemseghed, M.G.; Servello, J.; Hundt, N.; Sista, P.; Biewer, M.C.; Stefan, M.C. Amphiphilic block copolymers containing regioregular poly(3-hexylthiophene) and poly(2-ethyl-2-oxazoline). Macromol. Chem. Phys. 2010, 211, 1291–1297. [Google Scholar]
- Moon, H.C.; Anthonysamy, A.; Lee, Y.; Kim, J.K. Facile synthesis of well-defined coil-rod-coil block copolymer composed of regioregular poly(3-hexylthiophene) via anionic coupling reaction. Macromolecules 2010, 43, 1747–1752. [Google Scholar]
- Lim, H.; Huang, K.-T.; Su, W.-F.; Chao, C.-Y. Facile syntheses, morphologies, and optical absorptions of P3HT coil-rod-coil triblock copolymers. J. Polym. Sci. A Polym. Chem. 2010, 48, 3311–3322. [Google Scholar]
- Kaul, E.; Senkovskyy, V.; Tkachov, R.; Bocharova, V.; Komber, H.; Stamm, M.; Kiriy, A. Synthesis of a bifunctional initiator for controlled Kumada catalyst-transfer polycondensation/nitroxide-mediated polymerization and preparation of poly(3-hexylthiophene)-polystyrene block copolymer therefrom. Macromolecules 2010, 43, 77–81. [Google Scholar]
- Surin, M.; Coulembier, O.; Tran, K.; De Winter, J.; Leclere, P.; Gerbaux, P.; Lazzaroni, R.; Dubois, P. Regioregular poly(3-hexylthiophene)-poly(ε-caprolactone) block copolymers: Controlled synthesis, microscopic morphology, and charge transport properties. Org. Elect. 2010, 11, 767–774. [Google Scholar]
- Park, S.-J.; Kang, S.-G.; Fryd, M.; Saven, J.G.; Park, S.-J. Highly tunable photoluminescent properties of amphiphilic conjugated block copolymers. J. Am. Chem. Soc. 2010, 132, 9931–9933. [Google Scholar]
- Wu, Z.-Q.; Ono, R.J.; Chen, Z.; Bielawski, C.W. Synthesis of poly(3-alkylthiophene)-block-poly(arylisocyanide): Two sequential, mechanistically distinct polymerizations using a single catalyst. J. Am. Chem. Soc. 2010, 132, 14000–14001. [Google Scholar]
- Yokozawa, T.; Adachi, I.; Miyakoshi, R.; Yokoyama, A. Catalyst-transfer condensation polymerization for the synthesis of well-defined polythiophene with hydrophilic side chain and of diblock copolythiophene with hydrophilic and hydrophobic side chains. High Perform. Polym. 2007, 19, 684–699. [Google Scholar]
- Ohshimizu, K.; Ueda, M. Well-controlled synthesis of block copolythiophenes. Macromolecules 2008, 41, 5289–5294. [Google Scholar]
- Chueh, C.C.; Higashihara, T.; Tsai, J.-H.; Ueda, M.; Chen, W.-C. All-conjugated diblock copolymer of poly(3-hexylthiophene)-block-poly(3-phenoxymethylthiophene) for field-effect transistor and photovoltaic applications. Org. Electron. 2009, 10, 1541–1548. [Google Scholar]
- Zhang, Y.; Tajima, K.; Hirota, K.; Hashimoto, K. Synthesis of all-conjugated diblock copolymers by quasi-living polymerization and observation of their microphase separation. J. Am. Chem. Soc. 2008, 130, 7812–7813. [Google Scholar]
- Benanti, T.L.; Kalaydjian, A.; Venkataraman, D. Protocols for efficient postpolymerization functionalization of regioregular polythiophenes. Macromolecules 2008, 41, 8312–8315. [Google Scholar]
- Ouhib, F.; Khoukh, A.; Ledeuil, J.-B.; Martinez, H.; Desbrires, J.; Dagron-Lartigau, C. Diblock and random donor/acceptor “double cable” polythiophene copolymers via the GRIM method. Macromolecules 2008, 41, 9736–9743. [Google Scholar]
- van den Bergh, K.; Huybrechts, J.; Verbiest, T.; Koeckelberghs, G. Transfer of supramolecular chirality in block copoly(thiophene)s. Chem. Eur. J. 2008, 14, 9122–9125. [Google Scholar]
- Ouhib, F.; Hiorns, R.C.; de Bettignies, R.; Bailly, S.; Desbrières, J.; Dagron-Lartigau, C. Photovoltaic cells based on polythiophenes carrying lateral phenyl groups. Thin Sol. Films 2008, 516, 7199–7204. [Google Scholar]
- Wu, P.-T.; Ren, G.; Li, C.; Mezzenga, R.; Jenekhe, S.A. Crystalline diblock conjugated copolymers: Synthesis, self-assembly, and microphase separation of poly(3-butylthiophene)-b-poly(3-octylthiophene). Macromolecules 2009, 42, 2317–2320. [Google Scholar]
- Wu, P.-T.; Ren, G.; Kim, F.S.; Li, C.; Mezzenga, R.; Jenekhe, S.A. Poly(3-hexylthiophene)-b-poly(3-cyclohexylthiophene): Synthesis, microphase separation, thin film transistors, and photovoltaic applications. J. Polym. Sci. A Polym. Chem. 2010, 48, 614–626. [Google Scholar]
- He, M.; Zhao, L.; Wang, J.; Han, W.; Yang, Y.; Qiu, F.; Lin, Z. Self-assembly of all-conjugated poly(3-alkylthiophene) diblock copolymer nanostructures from mixed selective solvents. ACS Nano 2010, 4, 3241–3247. [Google Scholar]
- Hollinger, J.; Jahnke, A.A.; Coombs, N.; Seferos, D.S. Controlling phase separation and pptical properties in conjugated polymers through selenophene-thiophene copolymerization. J. Am. Chem. Soc. 2010, 132, 8546–8547. [Google Scholar]
- Miyakoshi, R.; Yokoyama, A.; Yokozawa, T. Importance of the order of successive catalyst-transfer condensation polymerization in the synthesis of block copolymers of polythiophene and poly(p-phenylene). Chem. Lett. 2008, 37, 1022–1023. [Google Scholar]
- Javier, A.E.; Varshney, S.R.; McCullough, R.D. Chain-growth synthesis of polyfluorenes with low polydispersities, block bopolymers of fluorene, and end-capped polyfluorenes: Toward new optoelectronic materials. Macromolecules 2010, 43, 3233–3237. [Google Scholar]
- Shirakawa, H.; Louis, E.J.; MacDiarmid, A.G.; Chiang, C.K.; Heeger, A.J. Synthesis of electrically conducting organic polymers: halogen derivatives of polyacetylene, (CH)x. J. Chem. Soc. Chem. Commun. 1977, 16, 578–580. [Google Scholar]
- Simionescu, C.I.; Percec, V. Progress in polyacetylene chemistry. Prog. Polym. Sci. 1982, 8, 133–214. [Google Scholar]
- Akagi, K. Helical polyacetylene: Asymmetric polymerization in a chiral liquid-crystal field. Chem. Rev. 2009, 109, 5354–5401. [Google Scholar]
- Kanga, R.S.; Hogen-Esch, T.E.; Randrianalimanana, E.; Soum, A.; Fontanille, M. Studies of the anionic polymerization of phenylvinylsulfoxide and its copolymer with styrene. Macromolecules 1990, 23, 4235–4240. [Google Scholar]
- Zhao, Y.; Higashihara, T.; Sugiyama, K.; Hirao, A. Synthesis of functionalized asymmetric star polymers containing conductive polyacetylene segments by living anionic polymerization. J. Am. Chem. Soc. 2005, 127, 14158–14159. [Google Scholar]
- Zhao, Y.; Higashihara, T.; Sugiyama, K.; Hirao, A. Synthesis of asymmetric star-branched polymers having two polyacetylene arms by means of living anionic polymerization using 1,1-diphenylethylene derivatives. Macromolecules 2007, 40, 228–238. [Google Scholar]
- Iovu, M.C.; Craley, C.R.; Jeffries-EL, M.; Krankowski, A.B.; Zhang, R.; Kowalewski, T.; McCullough, R.D. Conducting regioregular polythiophene block copolymer nanofibrils synthesized by reversible addition fragmentation chain transfer polymerization (RAFT) and nitroxide mediated polymerization (NMP). Macromolecules 2007, 40, 4733–4735. [Google Scholar]
- Kim, J.S.; Park, Y.; Lee, D.Y.; Lee, J.H.; Park, J.H.; Kim, J.K.; Cho, K. Poly(3-hexylthiophene) nanorods with aligned chain orientation for organic photovoltaics. Adv. Funct. Mater. 2010, 20, 540–545. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Higashihara, T.; Liu, C.-L.; Chen, W.-C.; Ueda, M. Synthesis of Novel π-Conjugated Rod-Rod-Rod Triblock Copolymers Containing Poly(3-hexylthiophene) and Polyacetylene Segments by Combination of Quasi-Living GRIM and Living Anionic Polymerization. Polymers 2011, 3, 236-251. https://doi.org/10.3390/polym3010236
Higashihara T, Liu C-L, Chen W-C, Ueda M. Synthesis of Novel π-Conjugated Rod-Rod-Rod Triblock Copolymers Containing Poly(3-hexylthiophene) and Polyacetylene Segments by Combination of Quasi-Living GRIM and Living Anionic Polymerization. Polymers. 2011; 3(1):236-251. https://doi.org/10.3390/polym3010236
Chicago/Turabian StyleHigashihara, Tomoya, Cheng-Liang Liu, Wen-Chang Chen, and Mitsuru Ueda. 2011. "Synthesis of Novel π-Conjugated Rod-Rod-Rod Triblock Copolymers Containing Poly(3-hexylthiophene) and Polyacetylene Segments by Combination of Quasi-Living GRIM and Living Anionic Polymerization" Polymers 3, no. 1: 236-251. https://doi.org/10.3390/polym3010236




