Stimuli-Responsive Zinc (II) Coordination Polymers: A Novel Platform for Supramolecular Chromic Smart Tools
Abstract
:1. Introduction
1.1. Brief Historical Background
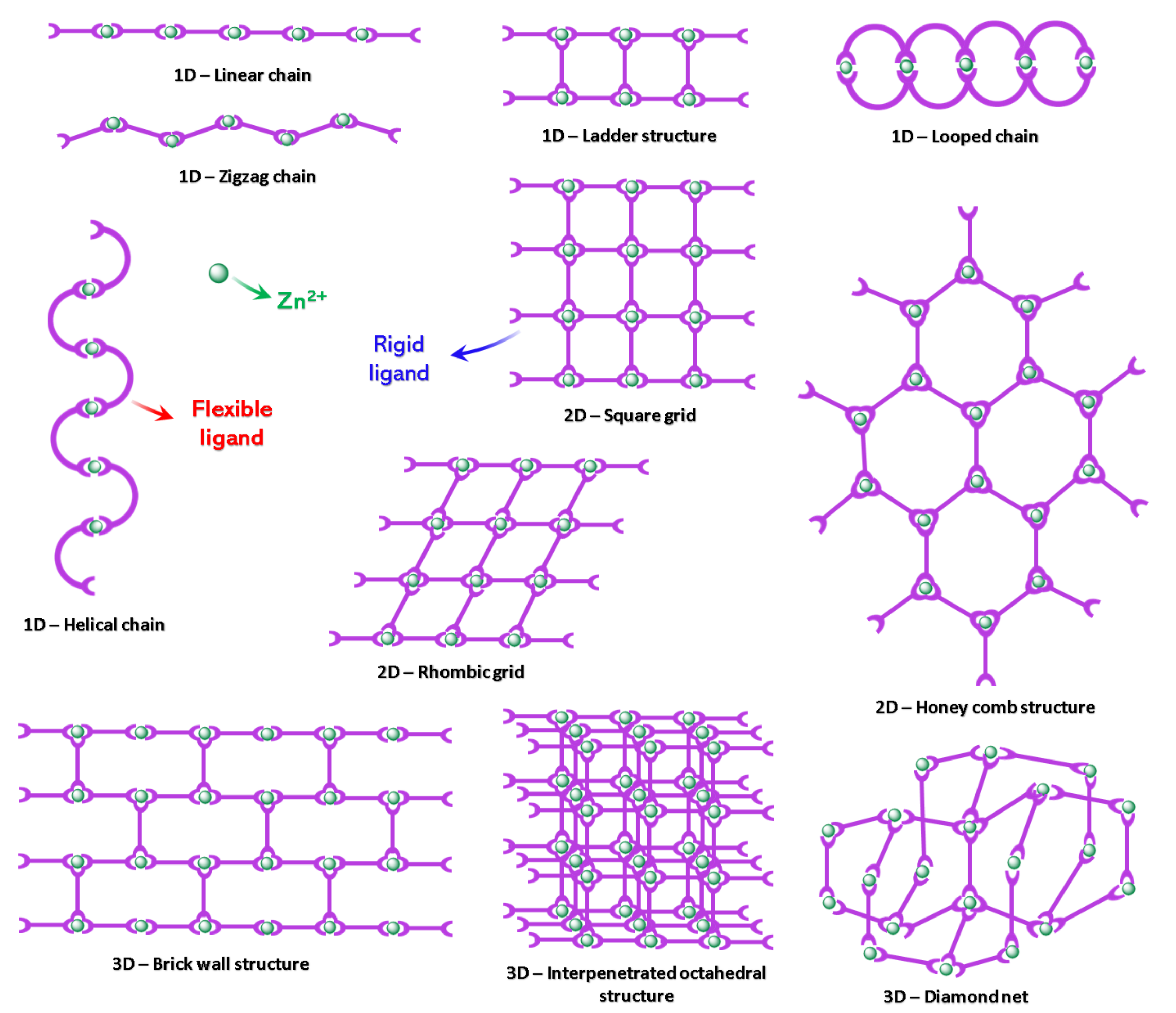
1.2. The Role of the Coordination Bond in the Supramolecular Polymers
1.3. General Considerations about Chromic Stimuli-Responsive CPs
- -
- Thermochromism, which is induced by a change of temperature;
- -
- Mechanochromism, which is induced by a stress in the solid state;
- -
- Electrochromism, which is induced by the gain and loss of electrons;
- -
- Solvatochromism, which is induced by the polarity of different solvents;
- -
- Photochromism, which is induced by light irradiation;
- -
- Chemochromism, which is induced by specific chemical analytes.
1.4. The Charming Peculiarity of Zinc Cation
- (1)
- Zn-CPs for metal cations, oxyanions, small and even harmful molecules;
- (2)
- Zn-CPs responsive to pH and temperature;
- (3)
- Responsive Zn-CPs hydrogels;
- (4)
- Zn-CPs for detection of biological targets;
- (5)
- Multifunctional Zn-CPs for bioimaging.
2. Zn-CPs Responsive toward Metal Cations, Oxyanions, and Small Molecules
2.1. Mono or Multifunctional Metal Cations Sensing
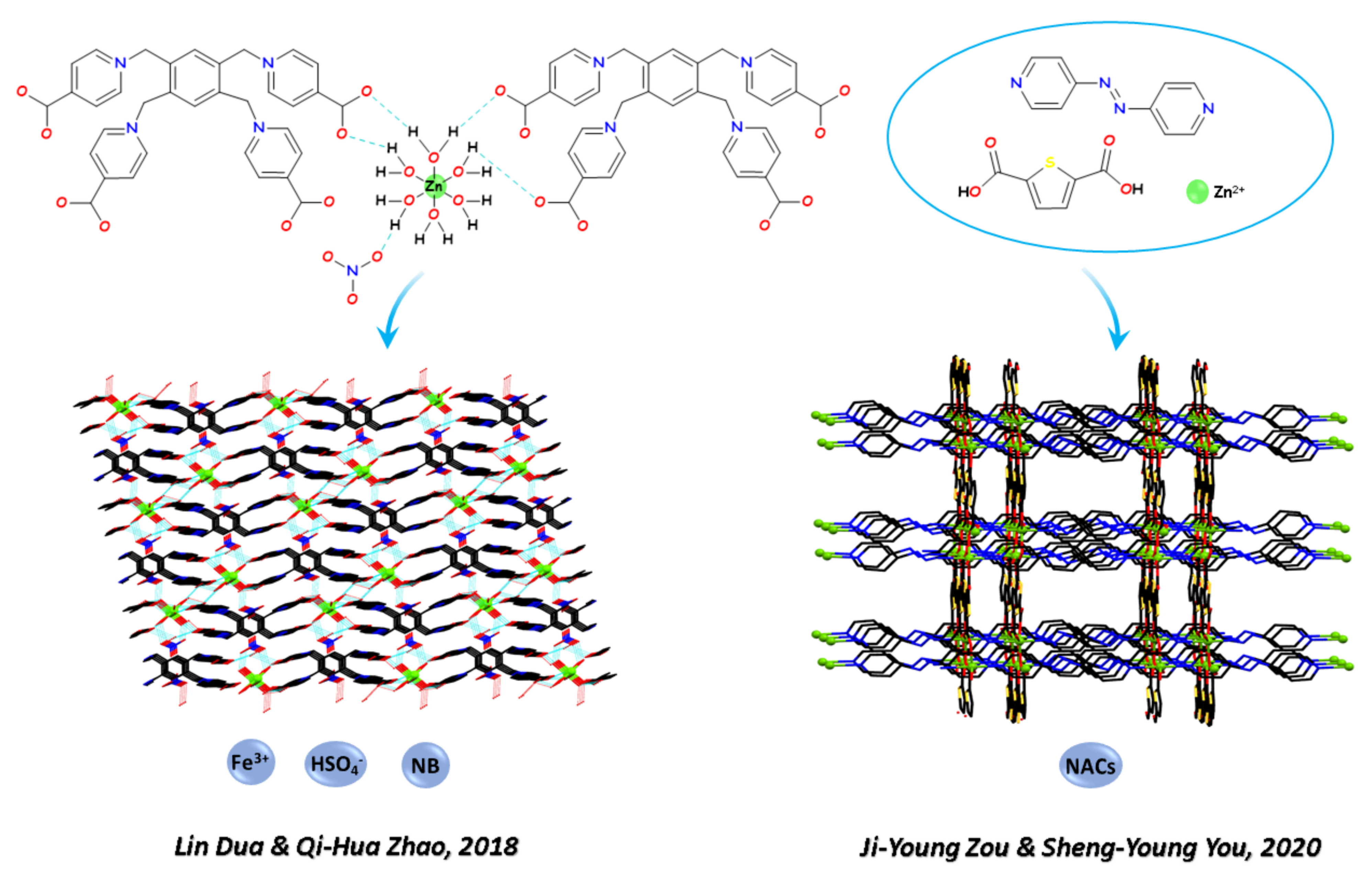
2.2. Multi-Sensing of Metal Cations, Oxyanions, and Nitro Aromatic Compounds
2.3. Multi-Sensing of Biologically Harmful Small Organic Molecules
3. Multi-Mode Zn-CPs Responsive to pH and Temperature
4. Stimuli-Responsive Zn-CP Gels
5. Zn-CPs as Bioenginereed Tools for Theranostic Use
5.1. Zn-LCPs for Sensing and Delivery of Bio-Relevant Species
5.2. Multifunctional Zn-NCPs for Fluorescence Bioimaging
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wise, D.L. Electrical and Optical Polymer Systems: Fundamentals: Methods, and Applications; CRC Press: Boca Raton, FL, USA, 1998. [Google Scholar]
- Batten, S.R.; Neville, S.M.; Turner, D.R. Coordination Polymers: Design, Analysis and Application; Royal Society of Chemistry: London, UK, 2009. [Google Scholar]
- Petrochenkova, N.; Mirochnik, A.; Kulikov, A.; Karasev, V. Structure and fluorescence properties of coordination polymers of europium (III) with pyromellitic acid. Russ. J. Coord. Chem. C/C Koord. Khimiia 1997, 23, 817–819. [Google Scholar]
- Vantomme, G.; Meijer, E.W. The construction of supramolecular systems. Science 2019, 363, 1396–1397. [Google Scholar] [CrossRef] [PubMed]
- de Moraes, M.L.; Caseli, L. Supramolecular Systems. In Nanostructures; William Andrew Publishing: Norwich, NY, USA, 2017; pp. 33–52. [Google Scholar] [CrossRef]
- Elacqua, E.; Ten Brummelhuis, N.; Weck, M. Supramolecular Polymers. In Handbook of Metathesis, 2nd ed.; Romanian Academy, Institute of Organic Chemistry: Bucharest, Romania, 2015; Volume 3-3, pp. 71–92. [Google Scholar]
- Sinha, N.; Hahn, F.E. Metallosupramolecular Architectures Obtained from Poly-N-heterocyclic Carbene Ligands. Acc. Chem. Res. 2017, 50, 2167–2184. [Google Scholar] [CrossRef] [PubMed]
- Lescop, C. Coordination-Driven Syntheses of Compact Supramolecular Metallacycles toward Extended Metallo-organic Stacked Supramolecular Assemblies. Acc. Chem. Res. 2017, 50, 885–894. [Google Scholar] [CrossRef] [PubMed]
- Tiekink, E.R.T. Supramolecular assembly based on “emerging” intermolecular interactions of particular interest to coordination chemists. Coord. Chem. Rev. 2017, 345, 209–228. [Google Scholar] [CrossRef] [Green Version]
- Lusby, P.J. Supramolecular coordination chemistry. Annu. Rep. Prog. Chem. Sec. A 2013, 109, 254–276. [Google Scholar] [CrossRef]
- Zhang, H.; Zou, R.; Zhao, Y. Macrocycle-based metal-organic frameworks. Coord. Chem. Rev. 2015, 292, 74–90. [Google Scholar] [CrossRef]
- Wei, M.; Wan, Y.; Zhang, X. Metal-organic framework-based stimuli-responsive polymers. J. Compos. Sci. 2021, 5, 101. [Google Scholar] [CrossRef]
- Ariga, K.; Mori, T.; Kitao, T.; Uemura, T. Supramolecular Chiral Nanoarchitectonics. Adv. Mater. 2020, 32, 1905657. [Google Scholar] [CrossRef] [PubMed]
- Giliopoulos, D.; Zamboulis, A.; Giannakoudakis, D.; Bikiaris, D.; Triantafyllidis, K. Polymer/metal organic framework (MOF) nanocomposites for biomedical applications. Molecules 2020, 25, 185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, S.; Feng, L.; Wang, K.; Pang, J.; Bosch, M.; Lollar, C.; Sun, Y.; Qin, J.; Yang, X.; Zhang, P.; et al. Stable Metal–Organic Frameworks: Design, Synthesis, and Applications. Adv. Mater. 2018, 30, e1704303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chai, L.Q.; Zhang, X.F.; Tang, L.J. Crystallographic, spectroscopic, TD/DFT calculations and Hirshfeld surface analysis of cadmium(II) coordination polymer containing pyridine ring. J. Mol. Struct. 2021, 1245, 131028. [Google Scholar] [CrossRef]
- Wang, W.Q.; Chen, J.; Wang, S.H.; Wu, S.F. Synthesis, crystal structure and fluorescence property of a Zinc(II) Coordination Polymer with a Theoretical Calculatio. Jiegou Huaxue 2021, 40, 79–84. [Google Scholar] [CrossRef]
- Di Palma, G.; Kirtman, B.; Gentile, F.S.; Platonenko, A.; Ferrari, A.M.; Dovesi, R. The VN2 negatively charged defect in diamond. A quantum mechanical investigation of the EPR response. Carbon 2020, 159, 443–450. [Google Scholar] [CrossRef]
- Ahmed, F.; Ortega-Castro, J.; Frontera, A.; Mir, M.H. Semiconducting properties of pyridyl appended linear dicarboxylate based coordination polymers: Theoretical prediction: Via DFT study. Dalton Trans. 2021, 50, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Gentile, F.S.; Salustro, S.; Desmarais, J.K.; Ferrari, A.M.; D’Arco, P.; Dovesi, R. Vibrational spectroscopy of hydrogens in diamond: A quantum mechanical treatment. Phys. Chem. Chem. Phys. 2018, 20, 11930–11940. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, J.; Lu, Z.; Wu, K.; Ning, G.H.; Li, D. Coinage-metal-based cyclic trinuclear complexes with metal-metal interactions: Theories to experiments and structures to functions. Chem. Rev. 2020, 120, 9675–9742. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.Q.; Luo, Z.D.; Pan, Y.; Kumar Singh, A.; Trivedi, M.; Kumar, A. Recent developments in luminescent coordination polymers: Designing strategies, sensing application and theoretical evidences. Coord. Chem. Rev. 2020, 406, 213145. [Google Scholar] [CrossRef]
- Gentile, F.S.; Pannico, M.; Causà, M.; Mensitieri, G.; Di Palma, G.; Scherillo, G.; Musto, P. Metal defects in HKUST-1 MOF revealed by vibrational spectroscopy: A combined quantum mechanical and experimental study. J. Mater. Chem. A 2020, 8, 10796–10812. [Google Scholar] [CrossRef]
- Nagaoka, M.; Ohta, Y.; Hitomi, H. Theoretical characterization of coordination space: Adsorption state and behavior of small molecules in nanochanneled metal-organic frameworks via electronic state theory, molecular mechanical and Monte Carlo simulation. Coord. Chem. Rev. 2007, 251, 2522–2536. [Google Scholar] [CrossRef]
- Férey, G. Hybrid porous solids: Past, present, future. Chem. Soc. Rev. 2008, 37, 191–214. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Fan, H.T.; Zang, S.Q.; Li, H.Y.; Wang, L.Y. Metal-containing crystalline luminescent thermochromic materials. Coord. Chem. Rev. 2018, 377, 307–329. [Google Scholar] [CrossRef]
- Bünzli, J.C.G. On the design of highly luminescent lanthanide complexes. Coord. Chem. Rev. 2015, 293–294, 19–47. [Google Scholar] [CrossRef]
- Tu, D.; Zheng, W.; Liu, Y.; Zhu, H.; Chen, X. Luminescent biodetection based on lanthanide-doped inorganic nanoprobes. Coord. Chem. Rev. 2014, 273–274, 13–29. [Google Scholar] [CrossRef]
- Zhao, S.N.; Zhang, Y.; Song, S.Y.; Zhang, H.J. Design strategies and applications of charged metal organic frameworks. Coord. Chem. Rev. 2019, 398, 113007. [Google Scholar] [CrossRef]
- Ren, J.; Ledwaba, M.; Musyoka, N.M.; Langmi, H.W.; Mathe, M.; Liao, S.; Pang, W. Structural defects in metal–organic frameworks (MOFs): Formation, detection and control towards practices of interests. Coord. Chem. Rev. 2017, 349, 169–197. [Google Scholar] [CrossRef]
- Tu, D.; Zheng, W.; Huang, P.; Chen, X. Europium-activated luminescent nanoprobes: From fundamentals to bioapplications. Coord. Chem. Rev. 2019, 378, 104–120. [Google Scholar] [CrossRef]
- Lustig, W.P.; Li, J. Luminescent metal–organic frameworks and coordination polymers as alternative phosphors for energy efficient lighting devices. Coord. Chem. Rev. 2018, 373, 116–147. [Google Scholar] [CrossRef]
- McKeown, N.B.; Budd, P.M.; Book, D. Microporous polymers as potential hydrogen storage materials. Macromol. Rapid Commun. 2007, 28, 995–1002. [Google Scholar] [CrossRef]
- Suh, M.P.; Park, H.J.; Prasad, T.K.; Lim, D.W. Hydrogen storage in metal-organic frameworks. Chem. Rev. 2012, 112, 782–835. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.; Xue, M.; Zhu, G. Metal-organic framework membranes: From synthesis to separation application. Chem. Soc. Rev. 2014, 43, 6116–6140. [Google Scholar] [CrossRef]
- Ma, L.; Abney, C.; Lin, W. Enantioselective catalysis with homochiral metal-organic frameworks. Chem. Soc. Rev. 2009, 38, 1248–1256. [Google Scholar] [CrossRef]
- Li, J.R.; Kuppler, R.J.; Zhou, H.C. Selective gas adsorption and separation in metal-organic frameworks. Chem. Soc. Rev. 2009, 38, 1477–1504. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Qin, J.; Liu, H.; Yao, X.; Liu, M. QSPR study on the melting points of a diverse set of potential ionic liquids by projection pursuit regression. QSAR Comb. Sci. 2009, 28, 1237–1244. [Google Scholar] [CrossRef]
- Yang, X.; Ma, L.F.; Yan, D. Facile synthesis of 1D organic-inorganic perovskite micro-belts with high water stability for sensing and photonic applications. Chem. Sci. 2019, 10, 4567–4572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feng, X.; Feng, Y.Q.; Liu, L.; Wang, L.Y.; Song, H.L.; Ng, S.W. A series of Zn-4f heterometallic coordination polymers and a zinc complex containing a flexible mixed donor dicarboxylate ligand. Dalton Trans. 2013, 42, 7741–7754. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, D.; Hu, Z.; Li, J. Luminescent metal-organic frameworks as explosive sensors. Dalton Trans. 2014, 43, 10668–10685. [Google Scholar] [CrossRef]
- Hu, Z.; Deibert, B.J.; Li, J. Luminescent metal-organic frameworks for chemical sensing and explosive detection. Chem. Soc. Rev. 2014, 43, 5815–5840. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.; Wang, W.; Hu, Z.; Wang, G.; Uvdal, K. Coordination polymers for energy transfer: Preparations, properties, sensing applications, and perspectives. Coord. Chem. Rev. 2015, 284, 206–235. [Google Scholar] [CrossRef]
- Wang, L.; Han, Y.; Feng, X.; Zhou, J.; Qi, P.; Wang, B. Metal-organic frameworks for energy storage: Batteries and supercapacitors. Coord. Chem. Rev. 2016, 307, 361–381. [Google Scholar] [CrossRef]
- Yang, D.; Ma, P.; Hou, Z.; Cheng, Z.; Li, C.; Lin, J. Current advances in lanthanide ion (Ln3+)-based upconversion nanomaterials for drug delivery. Chem. Soc. Rev. 2015, 44, 1416–1448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Landi, G.; Fahrner, W.R.; Concilio, S.; Sessa, L.; Neitzert, H.C. Electrical hole transport properties of an ambipolar organic compound with Zn-Atoms on a crystalline silicon heterostructure. IEEE J. Electron Devices Soc. 2014, 2, 179–181. [Google Scholar] [CrossRef]
- Liu, J.; Chen, L.; Cui, H.; Zhang, J.; Zhang, L.; Su, C.Y. Applications of metal-organic frameworks in heterogeneous supramolecular catalysis. Chem. Soc. Rev. 2014, 43, 6011–6061. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, Y.; Li, B.; O’Keeffe, M.; Chen, B. Multifunctional metal-organic frameworks constructed from meta-benzenedicarboxylate units. Chem. Soc. Rev. 2014, 43, 5618–5656. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Deng, D.S.; Ma, L.F.; Ji, B.M.; Wang, L.Y. A new copper-based metal-organic framework as a promising heterogeneous catalyst for chemo- and regio-selective enamination of β-ketoesters. Chem. Commun. 2013, 49, 10299–10301. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.Y.; Nie, H.J.; Li, J.M.; Shi, Z.F. Highly selective sensing of Fe3+/Hg2+ and proton conduction using two fluorescent Zn(ii) coordination polymers. Dalton Trans. 2020, 49, 11129–11141. [Google Scholar] [CrossRef] [PubMed]
- Sarker, M.; An, H.J.; Yoo, D.K.; Jhung, S.H. Nitrogen-doped porous carbon from ionic liquid@Al-metal-organic framework: A prominent adsorbent for purification of both aqueous and non-aqueous solutions. Chem. Eng. J. 2018, 338, 107–116. [Google Scholar] [CrossRef]
- Lin, R.B.; Xiang, S.; Xing, H.; Zhou, W.; Chen, B. Exploration of porous metal–organic frameworks for gas separation and purification. Coord. Chem. Rev. 2019, 378, 87–103. [Google Scholar] [CrossRef]
- Horcajada, P.; Gref, R.; Baati, T.; Allan, P.K.; Maurin, G.; Couvreur, P.; Férey, G.; Morris, R.E.; Serre, C. Metal-organic frameworks in biomedicine. Chem. Rev. 2012, 112, 1232–1268. [Google Scholar] [CrossRef]
- Giménez-Marqués, M.; Hidalgo, T.; Serre, C.; Horcajada, P. Nanostructured metal-organic frameworks and their bio-related applications. Coord. Chem. Rev. 2016, 307, 342–360. [Google Scholar] [CrossRef]
- Della Rocca, J.; Liu, D.; Lin, W. Nanoscale metal-organic frameworks for biomedical imaging and drug delivery. Acc. Chem. Res. 2011, 44, 957–968. [Google Scholar] [CrossRef] [Green Version]
- Begum, S.; Hassan, Z.; Bräse, S.; Wöll, C.; Tsotsalas, M. Metal-Organic Framework-Templated Biomaterials: Recent Progress in Synthesis, Functionalization, and Applications. Acc. Chem. Res. 2019, 52, 1598–1610. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Liu, Z.; Li, F. Upconversion nanophosphors for small-animal imaging. Chem. Soc. Rev. 2012, 41, 1323–1349. [Google Scholar] [CrossRef] [PubMed]
- Allendorf, M.D.; Bauer, C.A.; Bhakta, R.K.; Houk, R.J.T. Luminescent metal-organic frameworks. Chem. Soc. Rev. 2009, 38, 1330–1352. [Google Scholar] [CrossRef]
- Wei, M.; Gao, Y.; Li, X.; Serpe, M.J. Stimuli-responsive polymers and their applications. Polym. Chem. 2017, 8, 127–143. [Google Scholar] [CrossRef] [Green Version]
- Han, T.; Wang, X.; Wang, D.; Tang, B.Z. Functional Polymer Systems with Aggregation-Induced Emission and Stimuli Responses. Top. Curr. Chem. 2021, 379, 7. [Google Scholar] [CrossRef]
- Roviello, A.; Borbone, F.; Carella, A.; Diana, R.; Roviello, G.; Panunzi, B.; Ambrosio, A.; Maddalena, P. High quantum yield photoluminescence of new polyamides containing oligo-PPV amino derivatives and related oligomers. J. Polym. Sci. Part A Polym. Chem. 2009, 47, 2677–2689. [Google Scholar] [CrossRef]
- Crapnell, R.D.; Hudson, A.; Foster, C.W.; Eersels, K.; van Grinsven, B.; Cleij, T.J.; Banks, C.E.; Peeters, M. Recent advances in electrosynthesized molecularly imprinted polymer sensing platforms for bioanalyte detection. Sensors 2019, 19, 1204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Girolamo Del Mauro, A.; Diana, R.; Grimaldi, I.A.; Loffredo, F.; Morvillo, P.; Villani, F.; Minarini, C. Polymer solar cells with inkjet-printed doped-PEDOT: PSS anode. Polym. Compos. 2013, 34, 1493–1499. [Google Scholar] [CrossRef]
- Hu, R.; Yang, X.; Qin, A.; Tang, B.Z. AIE polymers in sensing, imaging and theranostic applications. Mater. Chem. Front. 2021, 5, 4073–4088. [Google Scholar] [CrossRef]
- Morvillo, P.; Grimaldi, I.A.; Diana, R.; Loffredo, F.; Villani, F. Study of the microstructure of inkjet-printed P3HT:PCBM blend for photovoltaic applications. J. Mater. Sci. 2013, 48, 2920–2927. [Google Scholar] [CrossRef]
- Kanižaj, L.; Šenjug, P.; Pajic, D.; Pavic, L.; Molcanov, K.; Juric, M. Magnetic and electrical behaviors of the homo-and heterometallic 1d and 3d coordination polymers based on the partial decomposition of the [Cr(C2O4)3]3− building block. Materials 2020, 13, 5341. [Google Scholar] [CrossRef] [PubMed]
- Panunzi, B.; Diana, R.; Concilio, S.; Sessa, L.; Shikler, R.; Nabha, S.; Tuzi, A.; Caruso, U.; Piotto, S. Solid-state highly efficient dr mono and poly-dicyano-phenylenevinylene fluorophores. Molecules 2018, 23, 1505. [Google Scholar] [CrossRef] [PubMed]
- Mrinalini, M.; Prasanthkumar, S. Recent Advances on Stimuli-Responsive Smart Materials and their Applications. ChemPlusChem 2019, 84, 1103–1121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhattarai, D.P.; Pant, B.; Acharya, J.; Park, M.; Ojha, G.P. Recent Progress in Metal-Organic Framework-Derived Nanostructures in the Removal of Volatile Organic Compounds. Molecules 2021, 26, 4948. [Google Scholar] [CrossRef]
- Solórzano, R.; Suárez-García, S.; Novio, F.; Lorenzo, J.; Alibés, R.; Busqué, F.; Ruiz-Molina, D. Nanoscale coordination polymers for medicine and sensors. In Advances in Inorganic Chemistry; Academic Press Inc.: Cambridge, MA, USA, 2020; Volume 76, pp. 3–31. [Google Scholar]
- Weng, W.; Benjamin Beck, J.; Jamieson, A.M.; Rowan, S.J. Understanding the mechanism of gelation and stimuli-responsive nature of a class of metallo-supramolecular gels. J. Am. Chem. Soc. 2006, 128, 11663–11672. [Google Scholar] [CrossRef] [PubMed]
- Li, X.N.; Tu, Z.M.; Li, L.; Wang, Z.H.; Zhang, H. A novel viologen-based coordination polymer with multi-stimuli responsive chromic properties: Photochromism, thermochromism, chemochromism and electrochromism. Dalton Trans. 2020, 49, 3228–3233. [Google Scholar] [CrossRef]
- Higuchi, M. Stimuli-responsive metallo-supramolecular polymer films: Design, synthesis and device fabrication. J. Mater. Chem. C 2014, 2, 9331–9341. [Google Scholar] [CrossRef]
- Shifrina, Z.B.; Matveeva, V.G.; Bronstein, L.M. Role of Polymer Structures in Catalysis by Transition Metal and Metal Oxide Nanoparticle Composites. Chem. Rev. 2020, 120, 1350–1396. [Google Scholar] [CrossRef]
- Ruan, C.; Zeng, K.; Grimes, C.A. A mass-sensitive pH sensor based on a stimuli-responsive polymer. Anal. Chim. Acta 2003, 497, 123–131. [Google Scholar] [CrossRef]
- Aida, T.; Meijer, E.W.; Stupp, S.I. Functional supramolecular polymers. Science 2012, 335, 813–817. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Greef, T.F.A.; Meijer, E.W. Materials science: Supramolecular polymers. Nature 2008, 453, 171–173. [Google Scholar] [CrossRef] [Green Version]
- Diana, R.; Panunzi, B.; Shikler, R.; Nabha, S.; Caruso, U. Highly efficient dicyano-phenylenevinylene fluorophore as polymer dopant or zinc-driven self-assembling building block. Inorg. Chem. Commun. 2019, 104, 145–149. [Google Scholar] [CrossRef]
- Yang, L.; Tan, X.; Wang, Z.; Zhang, X. Supramolecular Polymers: Historical Development, Preparation, Characterization, and Functions. Chem. Rev. 2015, 115, 7196–7239. [Google Scholar] [CrossRef] [PubMed]
- Di Costanzo, L.; Panunzi, B. Visual pH sensors: From a chemical perspective to new bioen-gineered materials. Molecules 2021, 26, 2952. [Google Scholar] [CrossRef] [PubMed]
- Quintard, A.; Rodriguez, J. Iron cyclopentadienone complexes: Discovery, properties, and catalytic reactivity. Angew. Chem. Int. Ed. 2014, 53, 4044–4055. [Google Scholar] [CrossRef] [PubMed]
- Abu-Dief, A.M.; Mohamed, I.M. A review on versatile applications of transition metal complexes incorporating Schiff bases. Beni-Suef Univ. J. Basic Appl. Sci. 2015, 4, 119–133. [Google Scholar] [CrossRef] [Green Version]
- Caruso, U.; Panunzi, B.; Roviello, A.; Tuzi, A. Fluorescent metallopolymers with Zn(II) in a Schiff base/phenoxide coordination environment. Inorg. Chem. Commun. 2013, 29, 138–140. [Google Scholar] [CrossRef]
- Caruso, U.; Panunzi, B.; Roviello, A.; Tingoli, M.; Tuzi, A. Two aminobenzothiazole derivatives for Pd(II) and Zn(II) coordination: Synthesis, characterization and solid state fluorescence. Inorg. Chem. Commun. 2011, 14, 46–48. [Google Scholar] [CrossRef]
- Umezawa, K.; Citterio, D. New Trends in Near-Infrared Fluorophores for Bioimaging. Suzuki, K. Anal. Sci. 2014, 30, 1348–2246. [Google Scholar] [CrossRef] [Green Version]
- Ma, Y.; Zeng, Y.; Liang, H.; Ho, C.L.; Zhao, Q.; Huang, W.; Wong, W.Y. A water-soluble tetraphenylethene based probe for luminescent carbon dioxide detection and its biological application. J. Mater. Chem. C 2015, 3, 11850–11856. [Google Scholar] [CrossRef]
- Kowada, T.; Maeda, H.; Kikuchi, K. BODIPY-based probes for the fluorescence imaging of biomolecules in living cells. Chem. Soc. Rev. 2015, 44, 4953–4972. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Zheng, Y.; Zhao, X.; Wang, L.; Ma, S.; Han, X.; Xu, B.; Tian, W.; Gao, H. Highly Efficient Far Red/Near-Infrared Solid Fluorophores: Aggregation-Induced Emission, Intramolecular Charge Transfer, Twisted Molecular Conformation, and Bioimaging Applications. Angew. Chem. Int. Ed. 2016, 55, 155–159. [Google Scholar] [CrossRef]
- Leng, S.; Qiao, Q.L.; Gao, Y.; Miao, L.; Deng, W.G.; Xu, Z.C. SNAP-tag fluorogenic probes for wash free protein labeling. Chin. Chem. Lett. 2017, 28, 1911–1915. [Google Scholar] [CrossRef]
- Xu, Z.; Chen, J.; Hu, L.L.; Tan, Y.; Liu, S.H.; Yin, J. Recent advances in formaldehyde-responsive fluorescent probes. Chin. Chem. Lett. 2017, 28, 1935–1942. [Google Scholar] [CrossRef]
- Chen, Y.; Wei, T.; Zhang, Z.; Zhang, W.; Lv, J.; Chen, T.; Chi, B.; Wang, F.; Chen, X. A mitochondria-targeted fluorescent probe for ratiometric detection of hypochlorite in living cells. Chin. Chem. Lett. 2017, 28, 1957–1960. [Google Scholar] [CrossRef]
- Sun, H.; Liu, S.; Lin, W.; Zhang, K.Y.; Lv, W.; Huang, X.; Huo, F.; Yang, H.; Jenkins, G.; Zhao, Q.; et al. Smart responsive phosphorescent materials for data recording and security protection. Nat. Commun. 2014, 5, 3601. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bünzli, J.C.G.; Eliseeva, S.V. Intriguing aspects of lanthanide luminescence. Chem. Sci. 2013, 4, 1939–1949. [Google Scholar] [CrossRef]
- Diana, R.; Panunzi, B.; Shikler, R.; Nabha, S.; Caruso, U. A symmetrical azo-based fluorophore and the derived salen multipurpose framework for emissive layers. Inorg. Chem. Commun. 2019, 104, 186–189. [Google Scholar] [CrossRef]
- Diana, R.; Panunzi, B.; Tuzi, A.; Caruso, U. Two tridentate pyridinyl-hydrazone zinc(II) complexes as fluorophores for blue emitting layers. J. Mol. Struct. 2019, 1197, 672–680. [Google Scholar] [CrossRef]
- Ariga, K.; Nishikawa, M.; Mori, T.; Takeya, J.; Shrestha, L.K.; Hill, J.P. Self-assembly as a key player for materials nanoarchitectonics. Sci. Technol. Adv. Mater. 2019, 20, 51–95. [Google Scholar] [CrossRef] [Green Version]
- Roy, S.; Chakraborty, C. Interfacial Coordination Nanosheet Based on Nonconjugated Three-Arm Terpyridine: A Highly Color-Efficient Electrochromic Material to Converge Fast Switching with Long Optical Memory. ACS Appl. Mater. Interfaces 2020, 12, 35181–35192. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.Y.; Shu, X.; Zeng, Y.; Liu, Q.Y.; Du, Z.Y.; He, C.T.; Zhang, W.X.; Chen, X.M. Molecule-based nonlinear optical switch with highly tunable on-off temperature using a dual solid solution approach. Nat. Commun. 2020, 11, 2752. [Google Scholar] [CrossRef] [PubMed]
- Pointel, Y.; Daiguebonne, C.; Suffren, Y.; Le Natur, F.; Freslon, S.; Calvez, G.; Bernot, K.; Jacob, D.; Guillou, O. Colloidal suspensions of highly luminescent lanthanide-based coordination polymer molecular alloys for ink-jet printing and tagging of technical liquids. Inorg. Chem. Front. 2021, 8, 2125–2135. [Google Scholar] [CrossRef]
- Mueller, U.; Schubert, M.; Teich, F.; Puetter, H.; Schierle-Arndt, K.; Pastré, J. Metal-organic frameworks—Prospective industrial applications. J. Mater. Chem. 2006, 16, 626–636. [Google Scholar] [CrossRef]
- Heine, J.; Müller-Buschbaum, K. Engineering metal-based luminescence in coordination polymers and metal-organic frameworks. Chem. Soc. Rev. 2013, 42, 9232–9242. [Google Scholar] [CrossRef] [PubMed]
- Diana, R.; Panunzi, B. The role of zinc(II) ion in fluorescence tuning of tridentate pincers: A review. Molecules 2020, 25, 4984. [Google Scholar] [CrossRef]
- Mu, T.; Chen, S.; Zhang, Y.; Meng, F.; Guo, P.; Chen, H.; Liu, X. Fluorescence polarization technique: A new method for vegetable oils classification. Anal. Methods 2015, 7, 5175–5179. [Google Scholar] [CrossRef]
- Liu, L.L.; Yu, C.X.; Sun, J.; Meng, P.P.; Ma, F.J.; Du, J.M.; Ma, L.F. Three coordination polymers constructed from various polynuclear clusters spaced by 2,2′-azodibenzoic acid: Syntheses and fluorescent properties. Dalton Trans. 2014, 43, 2915–2924. [Google Scholar] [CrossRef]
- Diana, R.; Panunzi, B. Zinc (Ii) and aiegens: The “clip approach” for a novel fluorophore family. A review. Molecules 2021, 26, 4176. [Google Scholar] [CrossRef] [PubMed]
- Alam, P.; Climent, C.; Alemany, P.; Laskar, I.R. “Aggregation-induced emission” of transition metal compounds: Design, mechanistic insights, and applications. J. Photochem. Photobiol. C Photochem. Rev. 2019, 41, 100317. [Google Scholar] [CrossRef]
- Castellano, F.N.; Pomestchenko, I.E.; Shikhova, E.; Hua, F.; Muro, M.L.; Rajapakse, N. Photophysics in bipyridyl and terpyridyl platinum(II) acetylides. Coord. Chem. Rev. 2006, 250, 1819–1828. [Google Scholar] [CrossRef]
- Lamansky, S.; Djurovich, P.; Murphy, D.; Abdel-Razzaq, F.; Lee, H.E.; Adachi, C.; Burrows, P.E.; Forrest, S.R.; Thompson, M.E. Highly phosphorescent bis-cyclometalated iridium complexes: Synthesis, photophysical characterization, and use in organic light emitting diodes. J. Am. Chem. Soc. 2001, 123, 4304–4312. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Xu, Y.; Wan, Y.; Fan, W.; Si, Z. Aggregation-Induced Phosphorescent Emission from ReI Complexes: Synthesis and Property Studies. Eur. J. Inorg. Chem. 2016, 2016, 1340–1347. [Google Scholar] [CrossRef]
- You, Y.; Nam, W. Photofunctional triplet excited states of cyclometalated Ir(iii) complexes: Beyond electroluminescence. Chem. Soc. Rev. 2012, 41, 7061–7084. [Google Scholar] [CrossRef]
- Caruso, U.; Diana, R.; Panunzi, B.; Roviello, A.; Tingoli, M.; Tuzi, A. Facile synthesis of new Pd(II) and Cu(II) based metallomesogens from ligands containing thiophene rings. Inorg. Chem. Commun. 2009, 12, 1135–1138. [Google Scholar] [CrossRef]
- Hsieh, W.H.; Wan, C.F.; Liao, D.J.; Wu, A.T. A turn-on Schiff base fluorescence sensor for zinc ion. Tetrahedron Lett. 2012, 53, 5848–5851. [Google Scholar] [CrossRef]
- Nugent, J.W.; Lee, H.; Lee, H.S.; Reibenspies, J.H.; Hancock, R.D. The effect of π contacts between metal ions and fluorophores on the fluorescence of PET sensors: Implications for sensor design for cations and anions. Inorg. Chem. 2014, 53, 9014–9026. [Google Scholar] [CrossRef] [PubMed]
- Kwok, R.T.K.; Leung, C.W.T.; Lam, J.W.Y.; Tang, B.Z. Biosensing by luminogens with aggregation-induced emission characteristics. Chem. Soc. Rev. 2015, 44, 4228–4238. [Google Scholar] [CrossRef]
- Diana, R.; Panunzi, B.; Concilio, S.; Marrafino, F.; Shikler, R.; Caruso, T.; Caruso, U. The effect of bulky substituents on two π-conjugated mesogenic fluorophores. Their organic polymers and zinc-bridged luminescent networks. Polymers 2019, 11, 1379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chua, M.H.; Zhou, H.; Zhu, Q.; Tang, B.Z.; Xu, J.W. Recent advances in cation sensing using aggregation-induced emission. Mater. Chem. Front. 2021, 5, 659–708. [Google Scholar] [CrossRef]
- Nugent, J.W.; Lee, H.; Lee, H.S.; Reibenspies, J.H.; Hancock, R.D. Mechanism of chelation enhanced fluorescence in complexes of cadmium(ii), and a possible new type of anion sensor. Chem. Commun. 2013, 49, 9749–9751. [Google Scholar] [CrossRef] [PubMed]
- Panunzi, B.; Diana, R.; Caruso, U. A highly efficient white luminescent zinc (II) based metallopolymer by RGB approach. Polymers 2019, 11, 1712. [Google Scholar] [CrossRef] [Green Version]
- Kumar, V.; Kumar, A.; Diwan, U.; Upadhyay, K.K. A Zn2+-responsive highly sensitive fluorescent probe and 1D coordination polymer based on a coumarin platform. Dalton Trans. 2013, 42, 13078–13083. [Google Scholar] [CrossRef] [PubMed]
- Patra, L.; Das, S.; Gharami, S.; Aich, K.; Mondal, T.K. A new multi-analyte fluorogenic sensor for efficient detection of Al3+ and Zn2+ ions based on ESIPT and CHEF features. New J.Chem. 2018, 42, 19076–19082. [Google Scholar] [CrossRef]
- Mei, J.; Leung, N.L.C.; Kwok, R.T.K.; Lam, J.W.Y.; Tang, B.Z. Aggregation-Induced Emission: Together We Shine, United We Soar! Chem. Rev. 2015, 115, 11718–11940. [Google Scholar] [CrossRef]
- Chen, J.; Law, C.C.W.; Lam, J.W.Y.; Dong, Y.; Lo, S.M.F.; Williams, I.D.; Zhu, D.; Tang, B.Z. Synthesis, light emission, nanoaggregation, and restricted intramolecular rotation of 1,1-substituted 2,3,4,5-tetraphenylsiloles. Chem. Mater. 2003, 15, 1535–1546. [Google Scholar] [CrossRef]
- Leung, N.L.C.; Xie, N.; Yuan, W.; Liu, Y.; Wu, Q.; Peng, Q.; Miao, Q.; Lam, J.W.Y.; Tang, B.Z. Restriction of intramolecular motions: The general mechanism behind aggregation-induced emission. Chem. A Eur. J. 2014, 20, 15349–15353. [Google Scholar] [CrossRef]
- Diana, R.; Caruso, U.; Di Costanzo, L.; Bakayoko, G.; Panunzi, B. A novel DR/NIR T-shaped aiegen: Synthesis and X-ray crystal structure study. Crystals 2020, 10, 269. [Google Scholar] [CrossRef] [Green Version]
- MacRae, C.F.; Sovago, I.; Cottrell, S.J.; Galek, P.T.A.; McCabe, P.; Pidcock, E.; Platings, M.; Shields, G.P.; Stevens, J.S.; Towler, M.; et al. Mercury 4.0: From visualization to analysis, design and prediction. J. Appl. Crystallogr. 2020, 53, 226–235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borbone, F.; Caruso, U.; Palma, S.D.; Fusco, S.; Nabha, S.; Panunzi, B.; Shikler, R. High solid state photoluminescence quantum yields and effective color tuning in polyvinylpyridine based zinc(II) metallopolymers. Macromol. Chem. Phys. 2015, 216, 1516–1522. [Google Scholar] [CrossRef]
- Parmar, B.; Bisht, K.K.; Rachuri, Y.; Suresh, E. Zn(ii)/Cd(ii) based mixed ligand coordination polymers as fluorosensors for aqueous phase detection of hazardous pollutants. Inorg. Chem. Front. 2020, 7, 1082–1107. [Google Scholar] [CrossRef]
- Park, M.K.; Lim, K.S.; Park, J.H.; Song, J.H.; Kang, D.W.; Lee, W.R.; Hong, C.S. Two- and three-dimensional Zn(II) coordination polymers constructed from mixed ligand systems: Interpenetration, structural transformation and sensing behavior. CrystEngComm 2016, 18, 4349–4358. [Google Scholar] [CrossRef]
- Gu, T.Y.; Dai, M.; Young, D.J.; Ren, Z.G.; Lang, J.P. Luminescent Zn(II) Coordination Polymers for Highly Selective Sensing of Cr(III) and Cr(VI) in Water. Inorg. Chem. 2017, 56, 4668–4678. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.; Xu, J.; Wang, S.; Wang, P.; Gao, L.; Chai, J.; Shen, L.; Chen, X.; Fan, Y.; Wang, L. Layer-structured coordination polymers based on 5-(1H-tetrazol-5-yl)isophthalic acid: Structure, sensitization of lanthanide(III) cations and small-molecule sensing. CrystEngComm 2016, 18, 7126–7134. [Google Scholar] [CrossRef]
- Li, W.X.; Li, H.X.; Li, H.Y.; Chen, M.M.; Shi, Y.X.; Lang, J.P. 1,4-Bis(2-(pyridin-4-yl)vinyl)naphthalene and Its Zinc(II) Coordination Polymers: Synthesis, Structural Characterization, and Selective Luminescent Sensing of Mercury(II) Ion. Cryst. Growth Des. 2017, 17, 3948–3959. [Google Scholar] [CrossRef]
- Fan, C.; Wang, L.; Xu, C.; Wu, R.; Li, N.; Zhang, D.; Zhang, X.; Bi, S.; Fan, Y. Synthesis, structure diversity, and dye adsorption and luminescent sensing properties of Zinc (II) coordination polymers based on 1,3,5-tris(1-imidazolyl)benzene and 1,3-bis(1-imidazolyl)toluene. J. Solid State Chem. 2020, 288, 121445. [Google Scholar] [CrossRef]
- Roy, E.; Nagar, A.; Chaudhary, S.; Pal, S. Advanced Properties and Applications of AIEgens-Inspired Smart Materials. Ind. Eng. Chem. Res. 2020, 59, 10721–10736. [Google Scholar] [CrossRef]
- Fan, C.; Zhang, X.; Li, N.; Xu, C.; Wu, R.; Zhu, B.; Zhang, G.; Bi, S.; Fan, Y. Zn-MOFs based luminescent sensors for selective and highly sensitive detection of Fe3+ and tetracycline antibiotic. J. Pharm. Biomed. Anal. 2020, 188, 113444. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xie, S.; Hu, Y.; Xiang, J.; Wang, L.; Li, R.; Chen, M.; Wang, F.; Liu, Q.; Chen, X. AIEgen modulated per-functionalized flower-like IRMOF-3 frameworks with tunable light emission and excellent sensing properties. Chem. Commun. 2021, 57, 2392–2395. [Google Scholar] [CrossRef] [PubMed]
- Lei, Z.; Zhou, J.; Liang, M.; Xiao, Y.; Liu, Z. Aggregation-Induced Emission of Au/Ag Alloy Nanoclusters for Fluorescence Detection of Inorganic Pyrophosphate and Pyrophosphatase Activity. Front. Bioeng. Biotechnol. 2021, 8, 628181. [Google Scholar] [CrossRef] [PubMed]
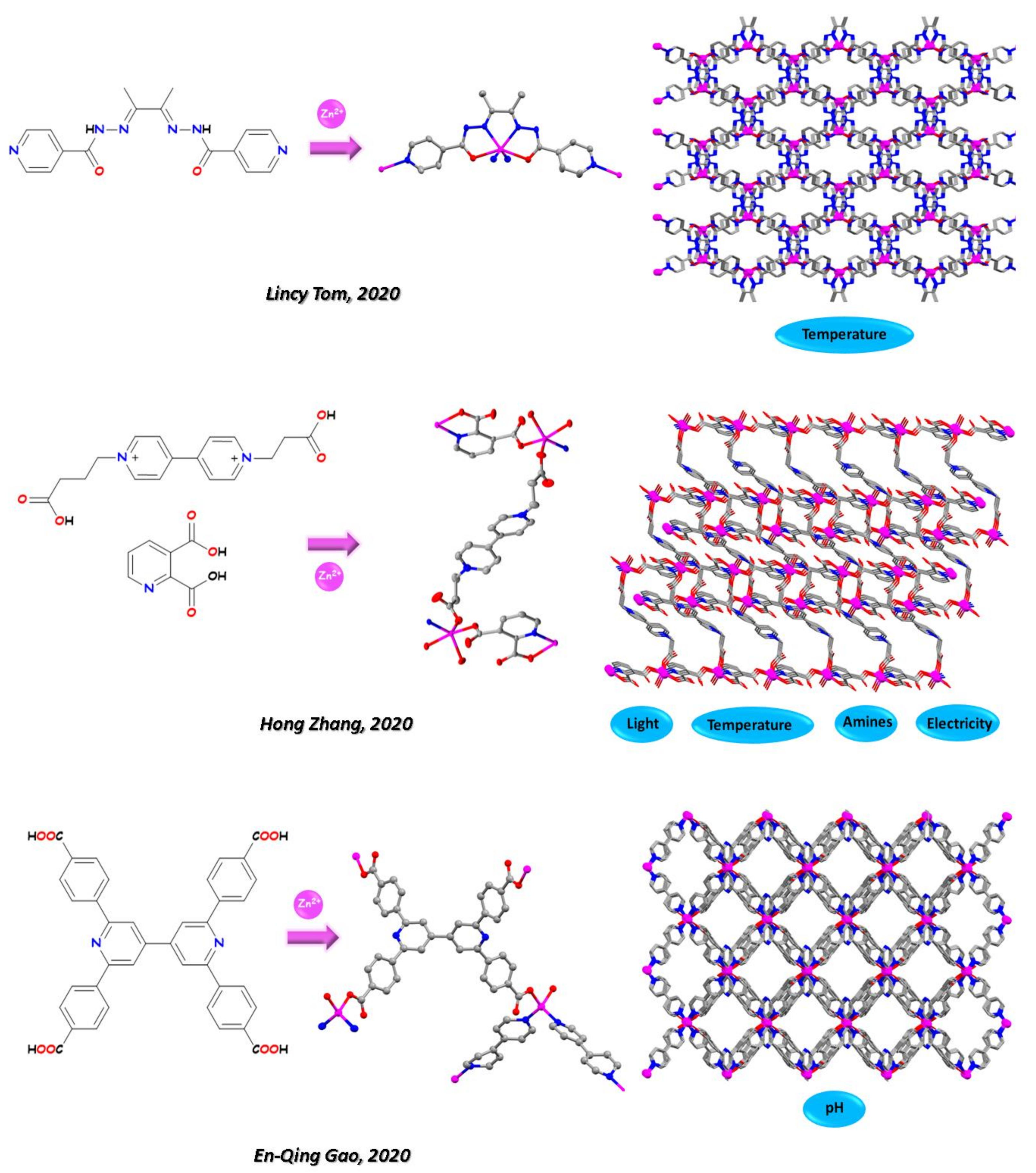
- Yang, S.L.; Liu, W.S.; Li, G.; Bu, R.; Li, P.; Gao, E.Q. A pH-Sensing Fluorescent Metal-Organic Framework: PH-Triggered Fluorescence Transition and Detection of Mycotoxin. Inorg. Chem. 2020, 59, 15421–15429. [Google Scholar] [CrossRef]
- Wang, Y.; Huang, R.; Zhang, J.; Cheng, G.; Yang, H. Lanthanide(Tb3+, Eu3+)-functionalized a new one dimensional Zn-MOF composite as luminescent probe for highly selectively sensing Fe3+. Polyhedron 2018, 148, 178–183. [Google Scholar] [CrossRef]
- Ren, G.; Li, Z.; Li, M.; Liang, Z.; Yang, W.; Qiu, P.; Pan, Q.; Zhu, G. A hexanuclear cluster based metal-organic framework for Fe3+ sensing. Inorg. Chem. Commun. 2018, 91, 108–111. [Google Scholar] [CrossRef]
- Lv, R.; Li, H.; Su, J.; Fu, X.; Yang, B.; Gu, W.; Liu, X. Zinc Metal-Organic Framework for Selective Detection and Differentiation of Fe(III) and Cr(VI) Ions in Aqueous Solution. Inorg. Chem. 2017, 56, 12348–12356. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.N.; Fan, T.T.; Wang, Q.S.; Han, H.L.; Li, X. Zn/Cd/Cu- frameworks constructed by 3,3′-diphenyldicarboxylate and 1,4-bis(1,2,4-triazol-1-yl)butane: Syntheses, structure, luminescence and luminescence sensing for metal ion in aqueous medium. J. Solid State Chem. 2018, 258, 744–752. [Google Scholar] [CrossRef]
- Farahani, Y.D.; Safarifard, V. Highly selective detection of Fe3+, Cd2+ and CH2Cl2 based on a fluorescent Zn-MOF with azine-decorated pores. J. Solid State Chem. 2019, 275, 131–140. [Google Scholar] [CrossRef]
- Levina, A.; Lay, P.A. Mechanistic studies of relevance to the biological activities of chromium. Coord. Chem. Rev. 2005, 249, 281–298. [Google Scholar] [CrossRef]
- Ostrowski, S.R.; Wilbur, S.; Chou, C.H.S.J.; Pohl, H.R.; Stevens, Y.W.; Allred, P.M.; Roney, N.; Fay, M.; Tylenda, C.A. Agency for Toxic Substances and Disease Registry’s 1997 priority list of hazardous substances. Latent effects—Carcinogenesis, neurotoxicology, and developmental deficits in humans and animals. Toxicol. Ind. Health 1999, 15, 602–644. [Google Scholar] [CrossRef] [PubMed]
- Zhitkovich, A. Chromium in drinking water: Sources, metabolism, and cancer risks. Chem. Res. Toxicol. 2011, 24, 1617–1629. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.Q.; Blatov, V.A.; Lv, X.X.; Yang, C.H.; Qian, L.L.; Li, K.; Li, B.L.; Wu, B. Construction of five zinc coordination polymers with 4-substituted bis(trizole) and multicarboxylate ligands: Syntheses, structures and properties. Polyhedron 2018, 155, 223–231. [Google Scholar] [CrossRef]
- Wang, X.; Feng, J.; Zhao, Y.D.; Zhang, X.F.; Yan, T.; Wang, Q.; Du, L.; Zhao, Q.H. Flexible coordination polymers based on zwitterion ligands and d10 metal ions for selective sensing of nitrobenzene, Fe3+ and HSO4−. Inorg. Chem. Commun. 2018, 98, 80–86. [Google Scholar] [CrossRef]
- Mai, Z.; Luo, X.; Lei, H. Ionothermal Synthesis of Zn(II) Coordination Polymers with Fluorescent Sensing and Selective Dye Adsorption Properties. J. Inorg. Organomet. Polym. Mater. 2019, 29, 1746–1754. [Google Scholar] [CrossRef]
- Wang, H.; Gao, T.; Zhang, Y. Synthesis of two 3D supramoleculars and their fluorescent sensing for nitroaromatic compounds/Fe3+ ions in aqueous medium. Inorg. Chem. Commun. 2020, 122, 108293. [Google Scholar] [CrossRef]
- Chi, J.; Zhong, B.; Li, Y.; Shao, P.; Liu, G.; Gao, Q.; Chen, B. Uncoordinated-substituents-induced zinc(II) coordination polymers exhibiting multifunctional fluorescent sensing activity for cations, anions and organochlorine pesticides. Z. Anorg. Allg. Chem. 2021, 647, 1284–1293. [Google Scholar] [CrossRef]
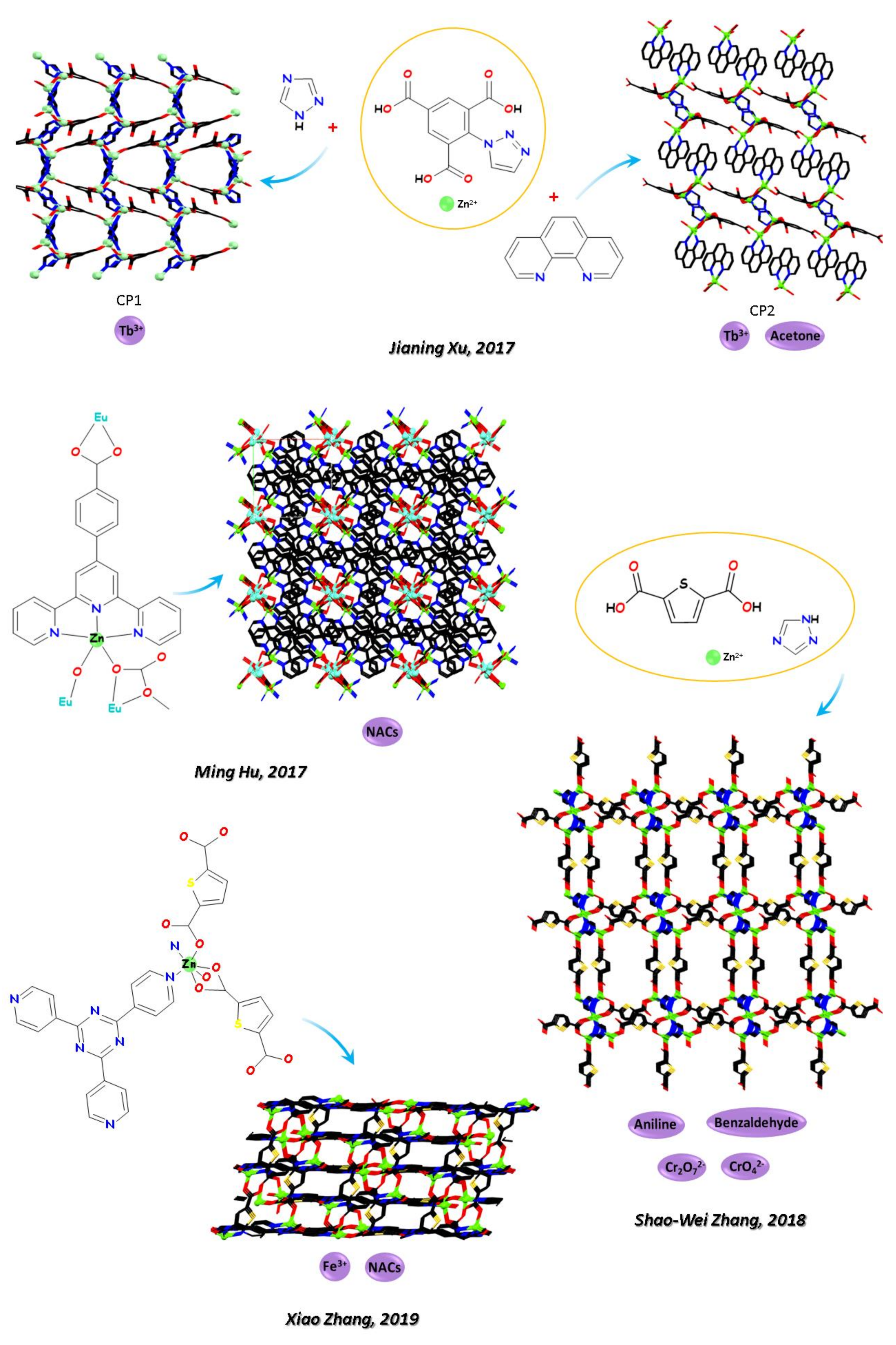
- Jia, J.; Wang, P.; Chai, J.; Ma, B.; Sun, J.; Chen, X.; Fan, Y.; Wang, L.; Xu, J. Construction of new zinc(II) coordination polymers by 1-(triazol-1-yl)-2,4,6-benzenetricarboxylate ligand for sensitizing lanthanide(III) ions and sensing small molecules. J. Solid State Chem. 2017, 253, 430–437. [Google Scholar] [CrossRef]
- Li, L.; Zou, J.Y.; You, S.Y. A luminescent pillar-layer Zn(II) metal–organic framework for the ultrasensitive detection of nitroaniline. Inorg. Chim. Acta 2020, 509, 119703. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, C.; Liu, X.; Gao, P.; Hu, M. Series of chiral interpenetrating 3d–4f heterometallic MOFs: Luminescent sensors and magnetic properties. J. Solid State Chem. 2017, 253, 360–366. [Google Scholar] [CrossRef]
- Lu, L.; He, J.; Wang, J.; Wu, W.P.; Li, B.; Singh, A.; Kumar, A.; Qin, X. Luminescent sensing and photocatalytic degradation in a new 3D Zn(II)-based highly luminescent metal−organic framework. J. Mol. Struct. 2019, 1179, 612–617. [Google Scholar] [CrossRef]
- Chen, Z.; Mi, X.; Wang, S.; Lu, J.; Li, Y.; Li, D.; Dou, J. Two novel penetrating coordination polymers based on flexible S-containing dicarboxylate acid with sensing properties towards Fe3+ and Cr2O72− ions. J. Solid State Chem. 2018, 261, 75–85. [Google Scholar] [CrossRef]
- Xiao, Z.Z.; Han, L.J.; Wang, Z.J.; Zheng, H.G. Three Zn(II)-based MOFs for luminescence sensing of Fe3+ and Cr2O72− ions. Dalton Trans. 2018, 47, 3298–3302. [Google Scholar] [CrossRef]
- Yin, J.C.; Li, N.; Qian, B.B.; Yu, M.H.; Chang, Z.; Bu, X.H. Highly stable Zn-MOF with Lewis basic nitrogen sites for selective sensing of Fe3+ and Cr2O72− ions in aqueous systems. J. Coord. Chem. 2020, 73, 2718–2727. [Google Scholar] [CrossRef]
- Gao, L.; Zhang, J.; Zhai, L.; Liang, J.; Liang, J.; Niu, X.; Hu, T. Fluorescent sensing properties of Cd (II)/Zn (II) metal–organic frameworks based on 3,5-di (2′,5′-dicarboxylphenyl) benozoic acid. Polyhedron 2019, 164, 90–95. [Google Scholar] [CrossRef]
- Zhuang, X.; Zhang, X.; Zhang, N.; Wang, Y.; Zhao, L.; Yang, Q. Novel multifunctional Zn metal–organic framework fluorescent probe demonstrating unique sensitivity and selectivity for detection of PA and Fe3+ ions in water solution. Cryst. Growth Des. 2019, 19, 5729–5736. [Google Scholar] [CrossRef]
- Yang, H.; Qi, D.; Chen, Z.; Cao, M.; Deng, Y.; Liu, Z.; Shao, C.; Yang, L. A Zn-based metal–organic framework as bifunctional chemosensor for the detection of nitrobenzene and Fe3+. J. Solid State Chem. 2021, 296, 121970. [Google Scholar] [CrossRef]
- Li, J.M.; Han, Y.H.; Wang, A.R.; He, K.H.; Shi, Z.F. A new 3D four-fold interpenetrated dia-like luminescent Zn(ii)-based metal-organic framework: The sensitive detection of Fe3+, Cr2O72−, and CrO42− in water, and nitrobenzene in ethanol _auth 1,2*Xu*T.-Y.*Tian-Yang. New J.Chem. 2020, 44, 4011–4022. [Google Scholar] [CrossRef]
- Zou, J.-Y.; Li, L.; You, S.-Y.; Cui, H.-M.; Liu, Y.-W.; Chen, K.-H.; Chen, Y.-H.; Cui, J.-Z.; Zhang, S.-W. Sensitive luminescent probes of aniline, benzaldehyde and Cr (VI) based on a zinc (II) metal-organic framework and its lanthanide (III) post-functionalizations. Dye. Pigment. 2018, 159, 429–438. [Google Scholar] [CrossRef]
- Gupta, V.K.; Suhas. Application of low-cost adsorbents for dye removal—A review. J. Environ. Manag. 2009, 90, 2313–2342. [Google Scholar] [CrossRef] [PubMed]
- Lv, B.; Wang, X.; Hu, H.M.; Zhao, Y.F.; Yang, M.L.; Xue, G. Synthesis, structure and luminescent sensor of zinc coordination polymers based on a new functionalized bipyridyl carboxylate ligand. Inorganica Chim. Acta 2016, 453, 771–778. [Google Scholar] [CrossRef]
- Ma, J.; Xu, N.; Liu, Y.; Wang, Y.; Li, H.; Liu, G.; Wang, X.; Li, J. A Stable 3D Zn-Coordination Polymer Sensor Based on Dual Luminescent Ligands for Efficient Detection of Multiple Analytes under Acid or Alkaline Environment. Inorg. Chem. 2020, 59, 15495–15503. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Ren, S.; Sun, B.; Jia, S. Optimization protocols and improved strategies for metal-organic frameworks for immobilizing enzymes: Current development and future challenges. Coord. Chem. Rev. 2018, 370, 22–41. [Google Scholar] [CrossRef]
- Tuergong, M.; Nizamidin, P.; Yimit, A.; Simayi, R. In situ growth and optical gas adsorption performance of Zn (II) metal–organic framework membranes at room temperature. Analyst 2019, 144, 4887–4896. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.H.; Yan, B. A dye@MOF crystalline probe serving as a platform for ratiometric sensing of trichloroacetic acid (TCA), a carcinogen metabolite in human urine. CrystEngComm 2019, 21, 4637–4643. [Google Scholar] [CrossRef]
- Guo, M.; Liu, S.; Guo, H.; Sun, Y.; Guo, X.; Deng, R. The mixed-ligand strategy to assemble a microporous anionic metal-organic framework: Ln3+ post-functionalization, sensors and selective adsorption of dyes. Dalton Trans. 2017, 46, 14988–14994. [Google Scholar] [CrossRef] [PubMed]
- Qu, T.-G.; Hao, X.-M.; Wang, H.; Cui, X.-G.; Chen, F.; Wu, Y.-B.; Yang, D.; Zhang, M.; Guo, W.-L. A luminescent 2D zinc (II) metal–organic framework for selective sensing of Fe (III) ions and adsorption of organic dyes. Polyhedron 2018, 156, 208–217. [Google Scholar] [CrossRef]
- Zhao, C.; Zhao, L.; Meng, L.; Liu, X. A Zn-MOF with 8-fold interpenetrating structure constructed with N,N′-bis (4-carbozylbenzyl)-4-aminotoluene ligands, sensors and selective adsorption of dyes. J. Solid State Chem. 2019, 274, 86–91. [Google Scholar] [CrossRef]
- Mukhopadhyay, A.; Jindal, S.; Savitha, G.; Moorthy, J.N. Temperature-Dependent Emission and Turn-Off Fluorescence Sensing of Hazardous “Quat” Herbicides in Water by a Zn-MOF Based on a Semi-Rigid Dibenzochrysene Tetraacetic Acid Linker. Inorg. Chem. 2020, 59, 6202–6213. [Google Scholar] [CrossRef]
- Fan, L.; Wang, F.; Zhao, D.; Peng, Y.; Deng, Y.; Luo, Y.; Zhang, X. A self-penetrating and chemically stable zinc (ii)-organic framework as multi-responsive chemo-sensor to detect pesticide and antibiotics in water. Appl. Organomet. Chem. 2020, 34, e5960. [Google Scholar] [CrossRef]
- Qin, X.; Yang, W.; Yang, Y.; Gu, D.; Guo, D.; Pan, Q. A zinc metal–organic framework for concurrent adsorption and detection of uranium. Inorg. Chem. 2020, 59, 9857–9865. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Ma, W.; Yu, P.; Mao, L. Colorimetric and Fluorescent Dual Mode Sensing of Alcoholic Strength in Spirit Samples with Stimuli-Responsive Infinite Coordination Polymers. Anal. Chem. 2015, 87, 6958–6965. [Google Scholar] [CrossRef]
- Panunzi, B.; Diana, R.; Concilio, S.; Sessa, L.; Tuzi, A.; Piotto, S.; Caruso, U. Fluorescence pH-dependent sensing of Zn(II) by a tripodal ligand. A comparative X-ray and DFT study. J. Lumin. 2019, 212, 200–206. [Google Scholar] [CrossRef]
- Peng, F.; Yang, H.; Hernandez, A.; Schier, D.E.; Feng, P.; Bu, X. Bimetallic Rod-Packing Metal–Organic Framework Combining Two Charged Forms of 2-Hydroxyterephthalic Acid. Chem. A Eur. J. 2020, 26, 11146–11149. [Google Scholar] [CrossRef]
- Diana, R.; Panunzi, B.; Tuzi, A.; Piotto, S.; Concilio, S.; Caruso, U. An amphiphilic pyridinoyl-hydrazone probe for colorimetric and fluorescence pH sensing. Molecules 2019, 24, 3833. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Wang, Y.; Zhang, X.-S.; Sheng, Y.-S.; Li, W.; Yang, A.-A.; Luan, J.; Liu, H.; Wang, Z. A novel 3D Zn-coordination polymer based on a multiresponsive fluorescent sensor demonstrating outstanding sensitivities and selectivities for the efficient detection of multiple analytes. Dalton Trans. 2021. [Google Scholar] [CrossRef]
- Ning, Y.; Wang, X.; Sheng, K.; Yang, L.; Han, W.; Xiao, C.; Li, J.; Zhang, Y.; Wu, S. A novel colorimetric and fluorescence turn-on pH sensor with a notably large Stokes shift for its application. New J.Chem. 2018, 42, 14510–14516. [Google Scholar] [CrossRef]
- Diana, R.; Caruso, U.; Tuzi, A.; Panunzi, B. A highly water-soluble fluorescent and colorimetric pH probe. Crystals 2020, 10, 83. [Google Scholar] [CrossRef] [Green Version]
- Venkatesan, M.; Veeramuthu, L.; Liang, F.C.; Chen, W.C.; Cho, C.J.; Chen, C.W.; Chen, J.Y.; Yan, Y.; Chang, S.H.; Kuo, C.C. Evolution of electrospun nanofibers fluorescent and colorimetric sensors for environmental toxicants, pH, temperature, and cancer cells—A review with insights on applications. Chem. Eng. J. 2020, 397, 125431. [Google Scholar] [CrossRef]
- Concilio, S.; Ferrentino, I.; Sessa, L.; Massa, A.; Iannelli, P.; Diana, R.; Panunzi, B.; Rella, A.; Piotto, S. A novel fluorescent solvatochromic probe for lipid bilayers. Supramol. Chem. 2017, 29, 887–895. [Google Scholar] [CrossRef]
- Jiang, Z.; Chen, J.; Du, J.; Liu, T.; Li, X.; Yu, H.; Zhang, Q.; Hu, Y.; Liu, F.; Yu, X.; et al. Ratiometric optical sensor for high-resolution imaging of pH with low cross-talk. Appl. Opt. 2018, 57, 9922–9928. [Google Scholar] [CrossRef] [PubMed]
- Bruchez, M., Jr.; Moronne, M.; Gin, P.; Weiss, S.; Alivisatos, A.P. Semiconductor nanocrystals as fluorescent biological labels. Science 1998, 281, 2013–2016. [Google Scholar] [CrossRef] [Green Version]
- Algar, W.R.; Krull, U.J. Luminescence and stability of aqueous thioalkyl acid capped CdSe/ZnS quantum dots correlated to ligand ionization. ChemPhysChem 2007, 8, 561–568. [Google Scholar] [CrossRef]
- Chen, J.; Song, J.L.; Sun, X.W.; Deng, W.Q.; Jiang, C.Y.; Lei, W.; Huang, J.H.; Liu, R.S. An oleic acid-capped CdSe quantum-dot sensitized solar cell. Appl. Phys. Lett. 2009, 94, 153115. [Google Scholar] [CrossRef]
- Pletnev, S.; Shcherbo, D.; Chudakov, D.M.; Pletneva, N.; Merzlyak, E.M.; Wlodawer, A.; Dauter, Z.; Pletnev, V. A crystallographic study of bright far-red fluorescent protein mKate reveals pH-induced cis-trans isomerization of the chromophore. J. Biol. Chem. 2008, 283, 28980–28987. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goedhart, J.; Von Stetten, D.; Noirclerc-Savoye, M.; Lelimousin, M.; Joosen, L.; Hink, M.A.; Van Weeren, L.; Gadella, T.W.; Royant, A. Structure-guided evolution of cyan fluorescent proteins towards a quantum yield of 93%. Nat. Commun. 2012, 3, 751. [Google Scholar] [CrossRef] [PubMed]
- Adam, V.; Nienhaus, K.; Bourgeois, D.; Nienhaus, G.U. Structural basis of enhanced photoconversion yield in green fluorescent protein-like protein Dendra2. Biochemistry 2009, 48, 4905–4915. [Google Scholar] [CrossRef]
- De Meulenaere, E.; Nguyen Bich, N.; de Wergifosse, M.; Van Hecke, K.; Van Meervelt, L.; Vanderleyden, J.; Champagne, B.; Clays, K. 4061−4069 Downloaded via UNIV OF NAPLES FEDERICO II on. J. Am. Chem. Soc. 2013, 135. [Google Scholar] [CrossRef]
- Piatkevich, K.D.; Malashkevich, V.N.; Almo, S.C.; Verkhusha, V.V. Engineering ESPT pathways based on structural analysis of LSSmKate red fluorescent proteins with large stokes shift. J. Am. Chem. Soc. 2010, 132, 10762–10770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pakhomov, A.A.; Martynov, V.I.; Orsa, A.N.; Bondarenko, A.A.; Chertkova, R.V.; Lukyanov, K.A.; Petrenko, A.G.; Deyev, I.E. Fluorescent protein Dendra2 as a ratiometric genetically encoded pH-sensor. Biochem. Biophys. Res. Commun. 2017, 493, 1518–1521. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Zhang, Y.; Pan, M.; Song, Y.; Bai, L.; Miao, Y.; Huang, Y.; Zhu, X.; Song, C.P. Dynamic imaging of cellular pH and redox homeostasis with a genetically encoded dual-functional biosensor, pHaROS, in yeast. J. Biol. Chem. 2019, 294, 15768–15780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, W.; Lu, W.; Xie, J.; Song, L.; Sa, R.; Song, J.L. Thermochromic Behavior of Azobenzene-based Coordination Polymer with Reversible Breathing Process. ChemistrySelect 2019, 4, 3222–3227. [Google Scholar] [CrossRef]
- Tom, L.; Kurup, M.R.P. A reversible thermo-responsive 2D Zn(ii) coordination polymer as a potential self-referenced luminescent thermometer. J. Mater. Chem. C 2020, 8, 2525–2532. [Google Scholar] [CrossRef]
- Zhao, D.; Yue, D.; Jiang, K.; Cui, Y.; Zhang, Q.; Yang, Y.; Qian, G. Ratiometric dual-emitting MOF⊃dye thermometers with a tunable operating range and sensitivity. J. Mater. Chem. C 2017, 5, 1607–1613. [Google Scholar] [CrossRef]
- Wang, S.; Gong, M.; Han, X.; Zhao, D.; Liu, J.; Lu, Y.; Li, C.; Chen, B. Embedding Red Emitters in the NbO-Type Metal-Organic Frameworks for Highly Sensitive Luminescence Thermometry over Tunable Temperature Range. ACS Appl. Mater. Interfaces 2021, 13, 11078–11088. [Google Scholar] [CrossRef] [PubMed]
- Xing, K.; Fan, R.; Wang, F.; Nie, H.; Du, X.; Gai, S.; Wang, P.; Yang, Y. Dual-stimulus-triggered programmable drug release and luminescent ratiometric pH sensing from chemically stable biocompatible zinc metal–Organic framework. ACS Appl. Mater. Interfaces 2018, 10, 22746–22756. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.H.; Lee, J.H.; Silverman, J.R.; John, G. Coordination polymer gels with important environmental and biological applications. Chem. Soc. Rev. 2013, 42, 924–936. [Google Scholar] [CrossRef] [PubMed]
- Inadomi, T.; Ikeda, S.; Okumura, Y.; Kikuchi, H.; Miyamoto, N. Photo-induced anomalous deformation of poly(N-isopropylacrylamide) gel hybridized with an inorganic nanosheet liquid crystal aligned by electric field. Macromol. Rapid Commun. 2014, 35, 1741–1746. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Liang, G.; Feng, B.; Wang, G.; Wu, N.; Deng, Y.; Elzatahry, A.A.; Alghamdi, A.; Zhao, Y.; Wei, J. Facile synthesis of metal-polyphenol-formaldehyde coordination polymer colloidal nanoparticles with sub-50 nm for T1-weighted magnetic resonance imaging. Chin. Chem. Lett. 2021, 32, 842–848. [Google Scholar] [CrossRef]
- Shao, L.; Yang, J.; Hua, B. A dual-responsive cross-linked supramolecular polymer network gel: Hierarchical supramolecular self-assembly driven by pillararene-based molecular recognition and metal-ligand interactions. Polym. Chem. 2018, 9, 1293–1297. [Google Scholar] [CrossRef]
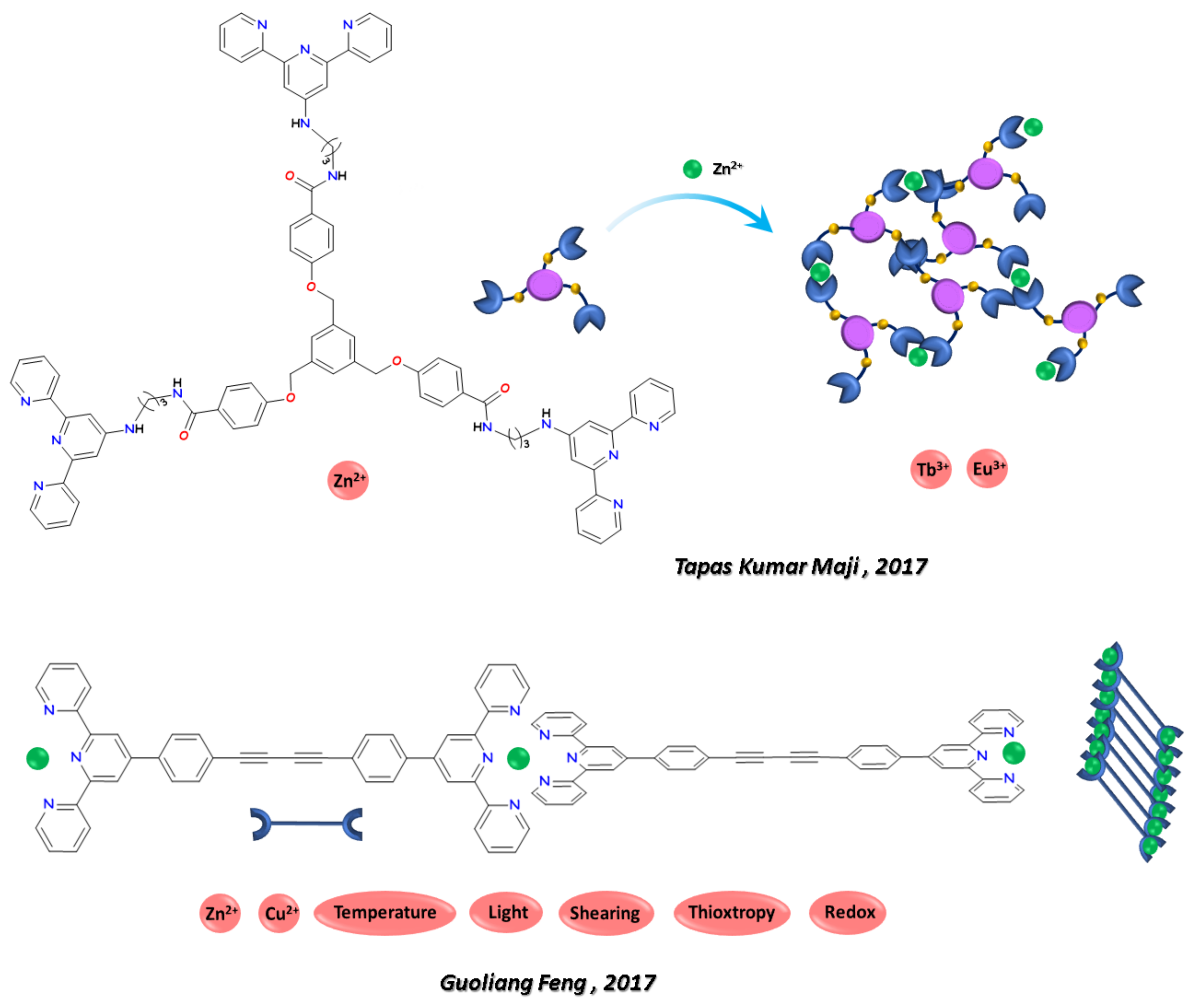
- Sutar, P.; Maji, T.K. Coordination Polymer Gels with Modular Nanomorphologies, Tunable Emissions, and Stimuli-Responsive Behavior Based on an Amphiphilic Tripodal Gelator. Inorg. Chem. 2017, 56, 9417–9425. [Google Scholar] [CrossRef]
- Yu, X.; Wang, Z.; Li, Y.; Geng, L.; Ren, J.; Feng, G. Fluorescent and Electrochemical Supramolecular Coordination Polymer Hydrogels Formed from Ion-Tuned Self-Assembly of Small Bis-Terpyridine Monomer. Inorg. Chem. 2017, 56, 7512–7518. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; He, L.; Chan, Y.T. Stimuli-Responsive Supramolecular Gels Constructed by Hierarchical Self-Assembly Based on Metal-Ligand Coordination and Host–Guest Recognition. Macromol. Rapid Commun. 2018, 39, e1800465. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Ma, Z.; Gong, S. Multi-stimuli-responsive self-healing metallo-supramolecular polymer nanocomposites. J. Mater. Chem. A 2016, 4, 3324–3334. [Google Scholar] [CrossRef]
- Chen, H.; Jiang, J.; Chen, Z.; Du, B.; Dai, C.; Wang, L.; Yin, Q.; Liu, Y.; Weng, G. Dynamic coordination of metal-alanine to control the multi-stimuli responsiveness of self-powered polymer hydrogels. J. Mater. Chem. A 2021, 9, 16594–16604. [Google Scholar] [CrossRef]
- Huo, Y.; He, Z.; Wang, C.; Zhang, L.; Xuan, Q.; Wei, S.; Wang, Y.; Pan, D.; Dong, B.; Wei, R.; et al. The recent progress of synergistic supramolecular polymers: Preparation, properties and applications. Chem. Commun. 2021, 57, 1413–1429. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Zhu, C.; Yuan, H.; Liu, L.; Lv, F.; Wang, S. Conjugated polymer nanoparticles: Preparation, properties, functionalization and biological applications. Chem. Soc. Rev. 2013, 42, 6620–6633. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Amin, D.; Latona, J.; Heber-Katz, E.; Messersmith, P.B. Supramolecular Polymer Hydrogels for Drug-Induced Tissue Regeneration. ACS Nano 2019, 13, 5493–5501. [Google Scholar] [CrossRef]
- Senapati, S.; Mahanta, A.K.; Kumar, S.; Maiti, P. Controlled drug delivery vehicles for cancer treatment and their performance. Signal Transduct. Target. Ther. 2018, 3, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mao, W.; Liao, Y.; Ma, D. A supramolecular assembly mediated by host-guest interactions for improved chemo-photodynamic combination therapy. Chem. Commun. 2020, 56, 4192–4195. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.S.; Liu, Y. Supramolecular chemistry of p-Sulfonatocalix[n]arenes and its biological applications. Acc. Chem. Res. 2014, 47, 1925–1934. [Google Scholar] [CrossRef]
- Webber, M.J.; Appel, E.A.; Meijer, E.W.; Langer, R. Supramolecular biomaterials. Nat. Mater. 2015, 15, 13–26. [Google Scholar] [CrossRef]
- Shi, X.L.; Chen, W.Y.; Zhang, T.; Zou, J.; Chen, Z.G. Fiber-based thermoelectrics for solid, portable, and wearable electronics. Energy Environ. Sci. 2021, 14, 729–764. [Google Scholar] [CrossRef]
- Yang, P.; Zhu, F.; Zhang, Z.; Cheng, Y.; Wang, Z.; Li, Y. Stimuli-responsive polydopamine-based smart materials. Chem. Soc. Rev. 2021, 50, 8319–8343. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Yang, L.; Shi, S.; Wang, T.; Duan, G.; Liu, X.; Li, Y. Flexible Polydopamine Bioelectronics. Adv. Funct. Mater. 2021, 31, 2103391. [Google Scholar] [CrossRef]
- Lago, A.B.; Pino-Cuevas, A.; Carballo, R.; Vázquez-López, E.M. A new metal-organic polymeric system capable of stimuli-responsive controlled release of the drug ibuprofen. Dalton Trans. 2016, 45, 1614–1621. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, X.Y.; Dong, W.W.; Han, H.M.; Zhao, J.; Li, D.S. A water-stable Zn (II) coordination polymer as fluorescent sensor for selective and sensitive detection of antibiotics and Fe3+. J. Solid State Chem. 2021, 296, 122032. [Google Scholar] [CrossRef]
- Sun, A.; Yang, Y.; Liu, Y.; Ding, L.; Duan, P.; Yang, W.; Pan, Q. A Zinc Coordination Polymer Sensor for Selective and Sensitive Detection of Doxycycline Based on Fluorescence Enhancement. Cryst. Growth Des. 2021, 21, 4971–4978. [Google Scholar] [CrossRef]
- Chen, W.; Kong, S.; Lu, M.; Chen, F.; Cai, W.; Du, L.; Wang, J.; Wu, C. Comparison of different zinc precursors for the construction of zeolitic imidazolate framework-8 artificial shells on living cells. Soft Matter 2019, 16, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Yao, S.; Chi, J.; Wang, Y.; Zhao, Y.; Luo, Y.; Wang, Y. Zn-MOF Encapsulated Antibacterial and Degradable Microneedles Array for Promoting Wound Healing. Adv. Healthc. Mater. 2021, 10, 2100056. [Google Scholar] [CrossRef] [PubMed]
- El-ghamry, M.A.; El-Shafiy, H. Synthesis and structural characterization of nano-sized metal complexes of 3-(1-methyl-4-hydroxy-2-oxo-1,2-dihydroquinolin-3-yl)-2-nitro-3-oxopropanoic acid. XRD, thermal, 3D modeling, and antitumor activity studies. Appl. Organomet. Chem. 2021, 35, e6206. [Google Scholar] [CrossRef]
- Niu, S.; Zhang, Q.; Zang, Y.; Hou, F. A fluorescent Zn(II)-containing coordination polymer for antibiotic sensing and treatment activity on periodontal tissue inflammation after orthodontics by inhibiting the growth of Porphyromonas gingivalis. J. Polym. Res. 2020, 27, 58. [Google Scholar] [CrossRef]
- Guo, P.; Liu, M.; Shi, L. A Zn-based coordination polymer as a luminescent sensor for simple and sensitive detecting of sulfonamides antibiotics and nitroaromatic. J. Solid State Chem. 2020, 286, 121247. [Google Scholar] [CrossRef]
- Li, X.; Zhao, W.J.; Zhang, L.; Yu, D.P.; Shang, B.B.; Zhou, X. A new luminescent Zn(II) coordination polymer: Selective detection of TNP and enhancement activity on sepsis by regulating the miR-16 expression in immune cells. Mon. Chem. 2020, 151, 1069–1076. [Google Scholar] [CrossRef]
- Erxleben, A. Structures and properties of Zn(II) coordination polymers. Coord. Chem. Rev. 2003, 246, 203–228. [Google Scholar] [CrossRef]
- Borbone, F.; Caruso, U.; Diana, R.; Panunzi, B.; Roviello, A.; Tingoli, M.; Tuzi, A. Second order nonlinear optical networks with excellent poling stability from a new trifunctional thiophene based chromophore. Org. Electron. 2009, 10, 53–60. [Google Scholar] [CrossRef]
- Kumar, R.; Mukherjee, A.; Mittal, B.R. Special techniques in PET/computed tomography imaging for evaluation of head and neck cancer. PET Clin. 2016, 11, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Restrepo, C.S.; Tavakoli, S.; Marmol-Velez, A. Contrast-enhanced cardiac magnetic resonance imaging. Magn. Reson. Imaging Clin. N. Am. 2012, 20, 739–760. [Google Scholar] [CrossRef]
- Chan, S.S.; Ntoulia, A.; Khrichenko, D.; Back, S.J.; Tasian, G.E.; Dillman, J.R.; Darge, K. Role of magnetic resonance urography in pediatric renal fusion anomalies. Pediatr. Radiol. 2017, 47, 1707–1720. [Google Scholar] [CrossRef] [PubMed]
- Tonet, E.; Pompei, G.; Faragasso, E.; Cossu, A.; Pavasini, R.; Passarini, G.; Tebaldi, M.; Campo, G. Coronary microvascular dysfunction: Pet, cmr and ct assessment. J. Clin. Med. 2021, 10, 1848. [Google Scholar] [CrossRef] [PubMed]
- Saitou, T.; Kiyomatsu, H.; Imamura, T. Quantitative Morphometry for Osteochondral Tissues Using Second Harmonic Generation Microscopy and Image Texture Information. Sci. Rep. 2018, 8, 2826. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kriech, M.A.; Conboy, J.C. Imaging chirality with surface second harmonic generation microscopy. J. Am. Chem. Soc. 2005, 127, 2834–2835. [Google Scholar] [CrossRef] [PubMed]
- Su, J.; Zhang, J.; Tian, X.; Zhao, M.; Song, T.; Yu, J.; Cui, Y.; Qian, G.; Zhong, H.; Luo, L.; et al. A series of multifunctional coordination polymers based on terpyridine and zinc halide: Second-harmonic generation and two-photon absorption properties and intracellular imaging. J. Mater. Chem. B 2017, 5, 5458–5463. [Google Scholar] [CrossRef]
- Suárez-García, S.; Solórzano, R.; Novio, F.; Alibés, R.; Busqué, F.; Ruiz-Molina, D. Coordination polymers nanoparticles for bioimaging. Coord. Chem. Rev. 2021, 432, 213716. [Google Scholar] [CrossRef]
- Alivisatos, A.P. Semiconductor clusters, nanocrystals, and quantum dots. Science 1996, 271, 933–937. [Google Scholar] [CrossRef] [Green Version]
- Jaiswal, J.K.; Mattoussi, H.; Mauro, J.M.; Simon, S.M. Long-term multiple color imaging of live cells using quantum dot bioconjugates. Nat. Biotechnol. 2002, 21, 47–51. [Google Scholar] [CrossRef] [PubMed]
- El-Sayed, M.A. Small is different: Shape-, size-, and composition-dependent properties of some colloidal semiconductor nanocrystals. Acc. Chem. Res. 2004, 37, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Rosi, N.L.; Mirkin, C.A. Nanostructures in biodiagnostics. Chem. Rev. 2005, 105, 1547–1562. [Google Scholar] [CrossRef]
- Burda, C.; Chen, X.; Narayanan, R.; El-Sayed, M.A. Chemistry and properties of nanocrystals of different shapes. Chem. Rev. 2005, 105, 1025–1102. [Google Scholar] [CrossRef]
- Zhao, D.; Cui, Y.; Yang, Y.; Qian, G. Sensing-functional luminescent metal–organic frameworks. CrystEngComm 2016, 18, 3746–3759. [Google Scholar] [CrossRef]
- Mittal, A.K.; Banerjee, U.C. Current status and future prospects of nanobiomaterials in drug delivery. In Nanobiomaterials in Drug Delivery; Elsevier: Amsterdam, The Netherlands, 2016; pp. 147–170. [Google Scholar]
- Kumar, N.; Roopa; Bhalla, V.; Kumar, M. Beyond zinc coordination: Bioimaging applications of Zn(II)-complexes. Coord. Chem. Rev. 2021, 427, 213550. [Google Scholar] [CrossRef]
- Li, S.; Zou, Q.; Li, Y.; Yuan, C.; Xing, R.; Yan, X. Smart Peptide-Based Supramolecular Photodynamic Metallo-Nanodrugs Designed by Multicomponent Coordination Self-Assembly. J. Am. Chem. Soc. 2018, 140, 10794–10802. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; Sun, L.; Huang, Y.; Wang, Y.; Zhang, M. Bioinspired fluorescent dipeptide nanoparticles for targeted cancer cell imaging and real-time monitoring of drug release. Nat. Nanotechnol. 2016, 11, 388–394. [Google Scholar] [CrossRef]
- Wang, S.; Ouyang, L.; Deng, G.; Deng, Z.; Wang, S. DNA adsorption on nanoscale zeolitic imidazolate framework-8 enabling rational design of a DNA-based nanoprobe for gene detection and regulation in living cells. RSC Adv. 2020, 10, 31012–31021. [Google Scholar] [CrossRef]
- Cai, H.-J.; Shen, T.-T.; Zhang, J.; Shan, C.-F.; Jia, J.-G.; Li, X.; Liu, W.-S.; Tang, Y. A core–shell metal–organic-framework (MOF)-based smart nanocomposite for efficient NIR/H2O2-responsive photodynamic therapy against hypoxic tumor cells. J. Mater. Chem. B 2017, 5, 2390–2394. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Wang, J.; Guo, X.; Liu, X.; Tang, X.; Zhang, H. A new three-dimensional zinc-based metal-organic framework as a fluorescent sensor for detection of cadmium ion and nitrobenzene. J. Colloid Interface Sci. 2018, 513, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.J.; Li, Y.M.; Zheng, K.; Zhang, W.H.; Liu, Y.L.; Yang, H. Construction of a Zn(II)-containing MOF for highly selective detection of picric acid and inhibition of human glioma cell growth. J. Mol. Struct. 2020, 1202, 127359. [Google Scholar] [CrossRef]
- Bhunia, S.; Dutta, B.; Pal, K.; Chandra, A.; Jana, K.; Sinha, C. Ultra-trace level detection of Cu2+ in an aqueous medium by novel Zn(ii)-dicarboxylato-pyridyl coordination polymers and cell imaging with HepG2 cells. New J. Chem. 2021, 45, 13941–13948. [Google Scholar] [CrossRef]
- Wu, Y.Y.; Fan, H.Q.; Chen, H.J.; Jiang, L.X.; Zhang, W.P. A Zn(II)-MOF with Suitable Pore Surroundings for Cyanosilylation Reaction and Protective Effect on Bladder Cancer Cells by Regulating miR-130 and CYLD. J. Inorg. Organomet. Polym. Mater. 2021, 31, 520–527. [Google Scholar] [CrossRef]
- Peng, T.; Jia, P.; Wang, R.; Feng, H.; Han, X.M. A ketone-functionalized Zn-MOF for solvent-free cyanosilylation of aldehyde and treatment activity against osteosarcoma trough increasing Mg63 cells autophagy. J. Coord. Chem. 2020, 73, 2235–2249. [Google Scholar] [CrossRef]
- Xin, X.T.; Cheng, J.Z. A mixed-ligand approach for building a N-rich porous metal-organic framework for drug release and anticancer activity against oral squamous cell carcinoma. J. Coord. Chem. 2018, 71, 3565–3574. [Google Scholar] [CrossRef]
- Ha, L.; Choi, K.M.; Kim, D.P. Interwoven MOF-Coated Janus Cells as a Novel Carrier of Toxic Proteins. ACS Appl. Mater. Interfaces 2021, 13, 18545–18553. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Chang, Z.; Zhang, D.; Binder, K.J.; Shen, S.; Huang, Y.Y.S.; Bai, Y.; Wheatley, A.E.H.; Liu, H. Harnessing surface-functionalized metal-organic frameworks for selective tumor cell capture. Chem. Mater. 2017, 29, 8052–8056. [Google Scholar] [CrossRef]
- Zhang, W.; Ding, X.; Cheng, H.; Yin, C.; Yan, J.; Mou, Z.; Wang, W.; Cui, D.; Fan, C.; Sun, D. Dual-targeted gold nanoprism for recognition of early apoptosis, dual-model imaging and precise cancer photothermal therapy. Theranostics 2019, 9, 5610–5625. [Google Scholar] [CrossRef] [PubMed]
- Caruso, U.; Panunzi, B.; Diana, R.; Concilio, S.; Sessa, L.; Shikler, R.; Nabha, S.; Tuzi, A.; Piotto, S. AIE/ACQ effects in two DR/NIR emitters: A structural and DFT comparative analysis. Molecules 2018, 23, 1947. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wei, Y.; Wang, L.; Huang, J.; Zhao, J.; Yan, Y. Multifunctional Metallo-Organic Vesicles Displaying Aggregation-Induced Emission: Two-Photon Cell-Imaging, Drug Delivery, and Specific Detection of Zinc Ion. ACS Appl. Nano Mater. 2018, 1, 1819–1827. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diana, R.; Caruso, U.; Panunzi, B. Stimuli-Responsive Zinc (II) Coordination Polymers: A Novel Platform for Supramolecular Chromic Smart Tools. Polymers 2021, 13, 3712. https://doi.org/10.3390/polym13213712
Diana R, Caruso U, Panunzi B. Stimuli-Responsive Zinc (II) Coordination Polymers: A Novel Platform for Supramolecular Chromic Smart Tools. Polymers. 2021; 13(21):3712. https://doi.org/10.3390/polym13213712
Chicago/Turabian StyleDiana, Rosita, Ugo Caruso, and Barbara Panunzi. 2021. "Stimuli-Responsive Zinc (II) Coordination Polymers: A Novel Platform for Supramolecular Chromic Smart Tools" Polymers 13, no. 21: 3712. https://doi.org/10.3390/polym13213712
APA StyleDiana, R., Caruso, U., & Panunzi, B. (2021). Stimuli-Responsive Zinc (II) Coordination Polymers: A Novel Platform for Supramolecular Chromic Smart Tools. Polymers, 13(21), 3712. https://doi.org/10.3390/polym13213712







