Unravelling the High-Pressure Behaviour of Dye-Zeolite L Hybrid Materials
Abstract
:1. Introduction
2. Materials and Methods
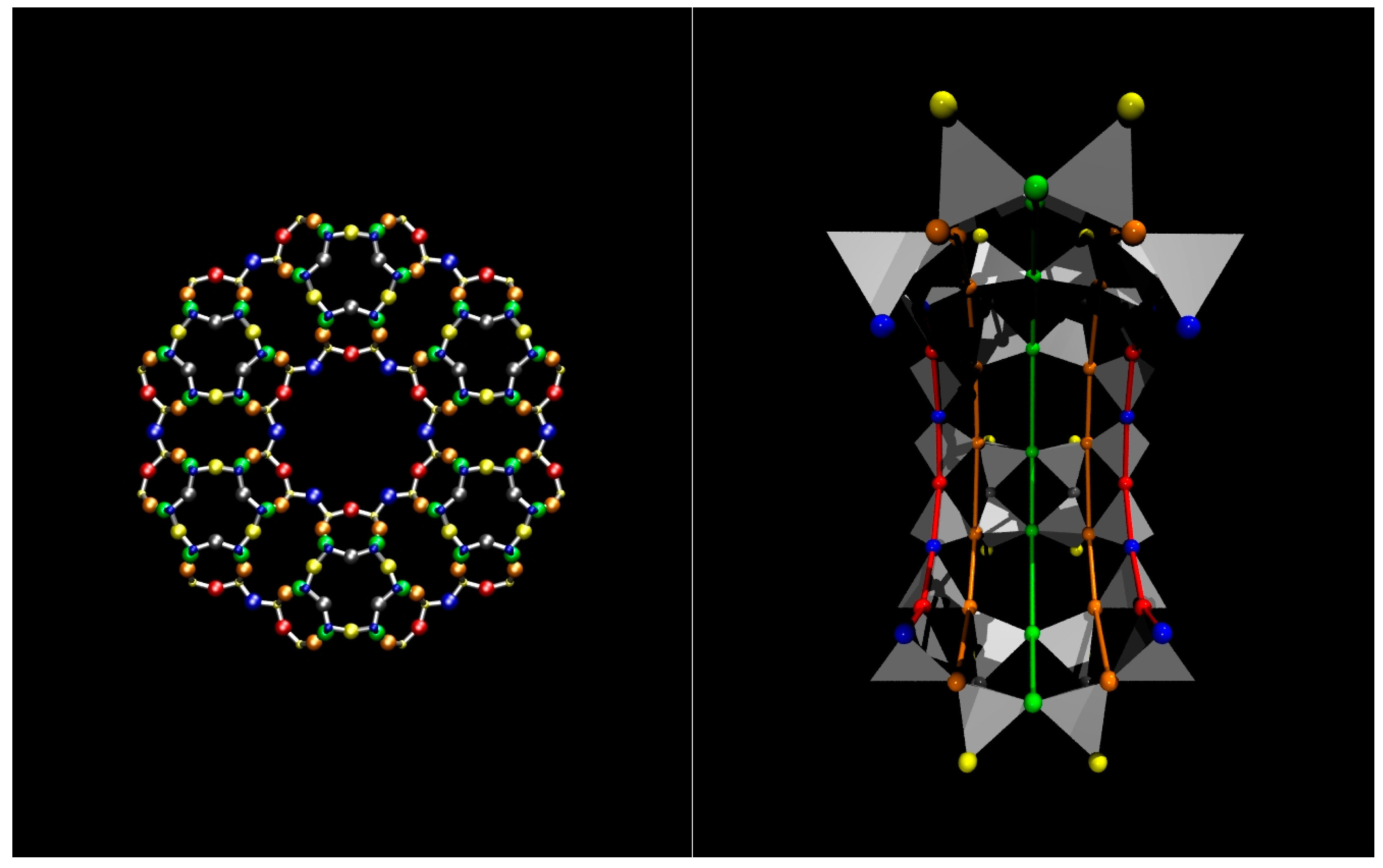
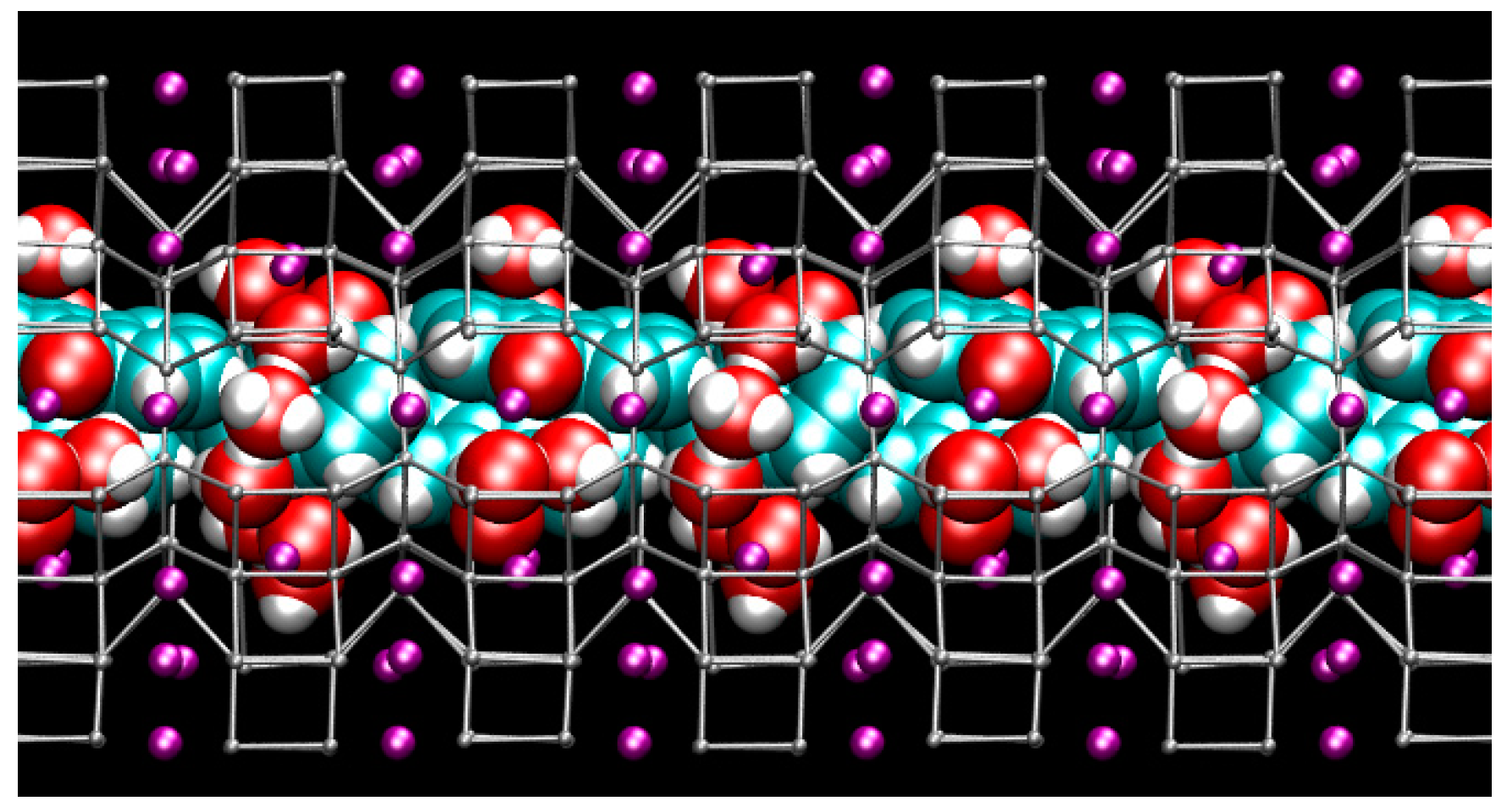
2.1. Zeolite Dye Hybrids: Structural Details at Ambient Conditions
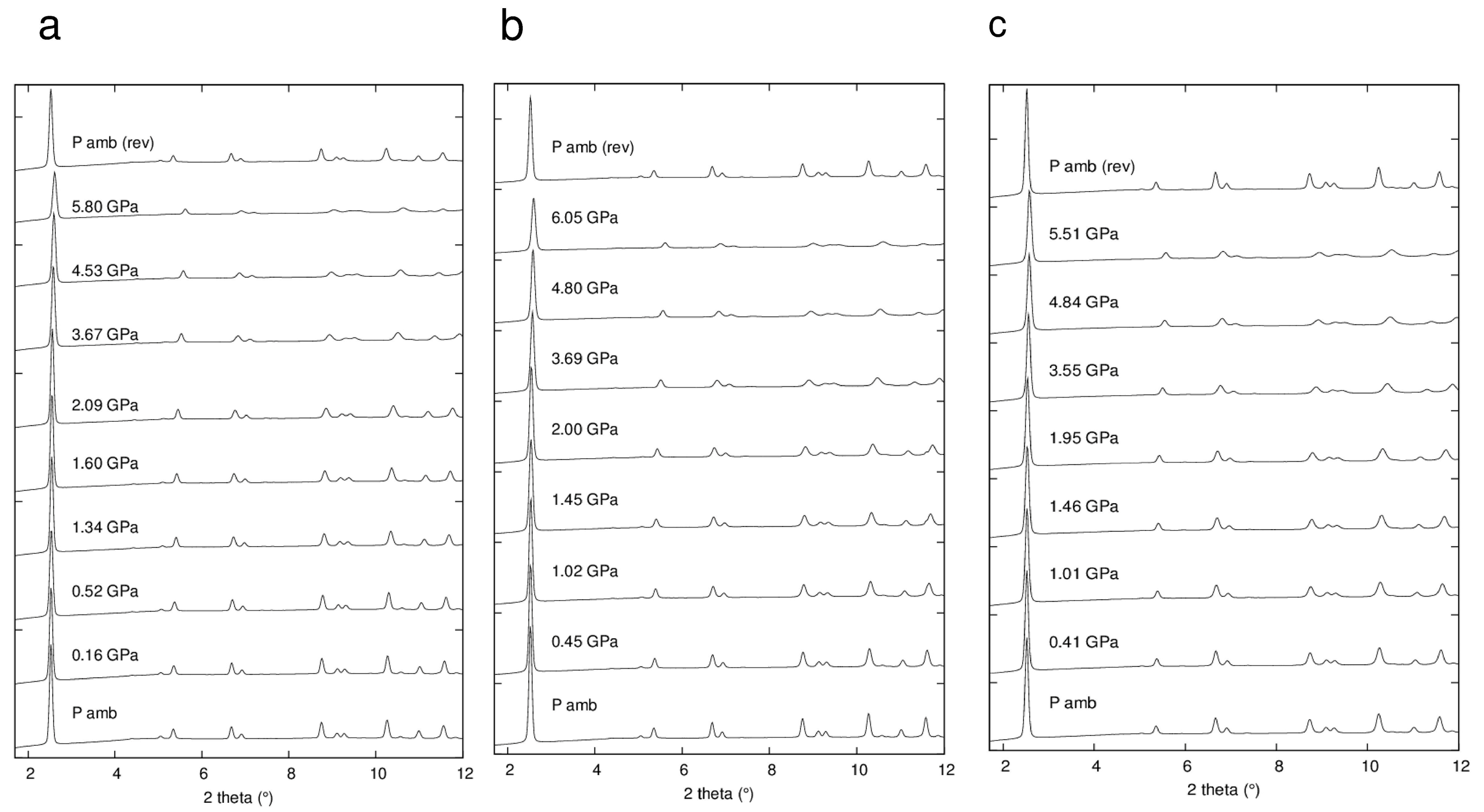
2.2. Synchrotron X-ray Powder Diffraction Experiments
2.3. Theoretical Modelling
3. Results
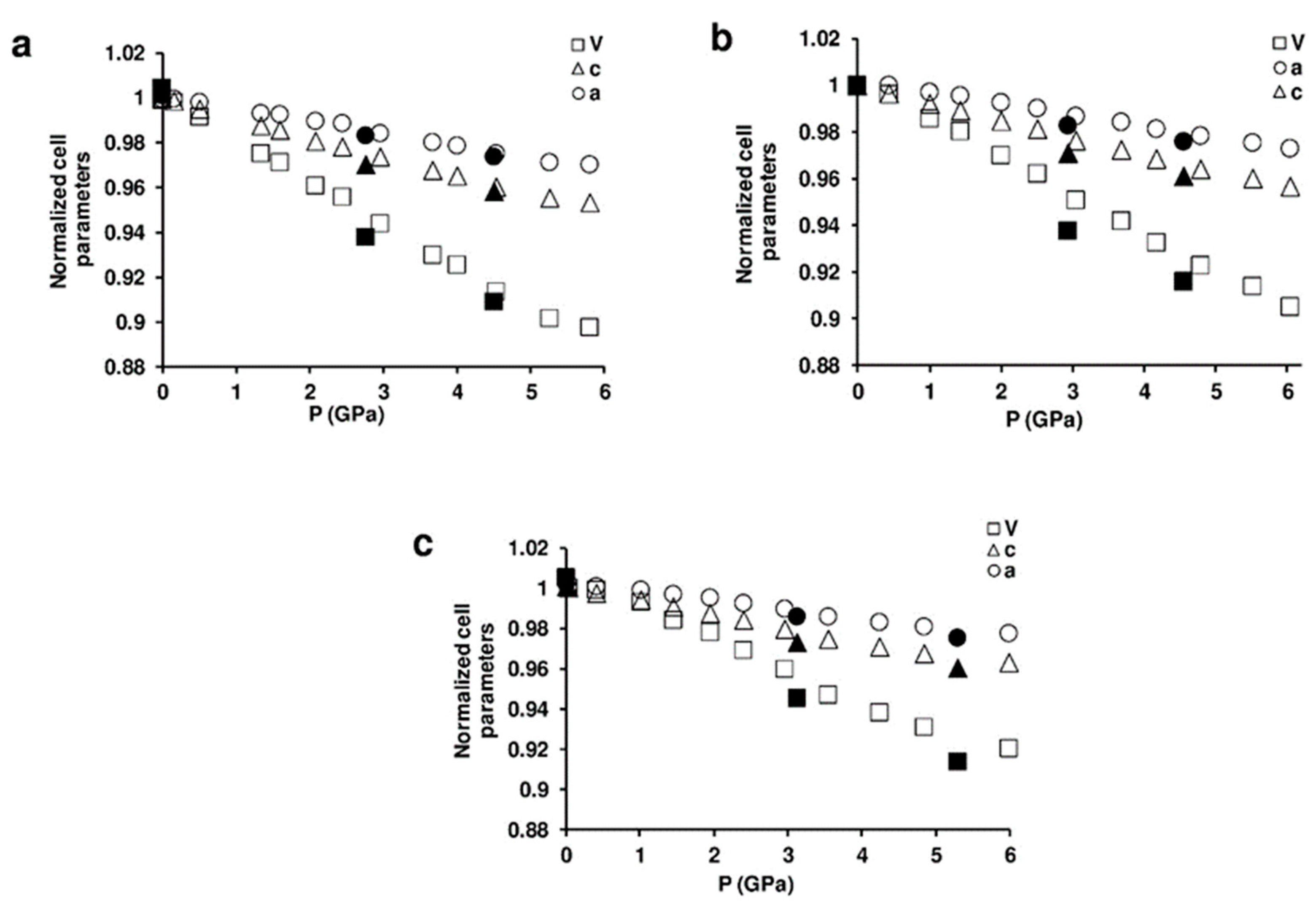
3.1. Structure of the ZL-FL Composites at Different Pressure Conditions
- (i)
- In the ZL/0.5FL at 2 GPa, both the diameters (O1–O1 and O2–O2) of the 12 MR shorten. The shortening of the 12 MR O1–O1 diameter is reflected in the lengthening of the O1–O1 diameter of 8 MR channel (ǁ [001]), which becomes more elliptical. Upon pressure release, the original values of the 12 MR diameters are almost regained (remaining slightly smaller than those observed at Pamb), while the 8 MR ones are strongly lengthened with respect to the original values, in accordance with the increase of a parameter (see Table 1 and Table S1).
- (ii)
- In the ZL/1FL sample, O2–O2 (12 MR) diameter decreases with pressure while O1–O1 increases—probably due to the presence of a larger numbers of FL molecules in the channels with respect to the ZL/0.5FL system. The O1–O1 (12 MR) increase is balanced by the decrease of O1–O1 diameter of the 8 MR channel running alongside (ǁ [001]): as a consequence, 8 MR becomes less elliptical. Upon pressure release, the 12 MR opening remains slightly smaller than that observed at Pamb. The significant enlargement of the 8 MR leads to an overall value for a parameter comparable with that at Pamb.
- (iii)
- The 8 MR window (8 MR ‖ [001]) (O1–O1, O6–O6), parallel to the c axis, becomes more circular at 2 GPa in both the samples. Once the pressure is released the starting values are regained.
- (iv)
- The O3–O5–O3 and O5–O3–O5 angle variations indicate that the D6R slightly increases its ditrigonal distortion in both samples. Upon pressure release the Pamb features are almost recovered.
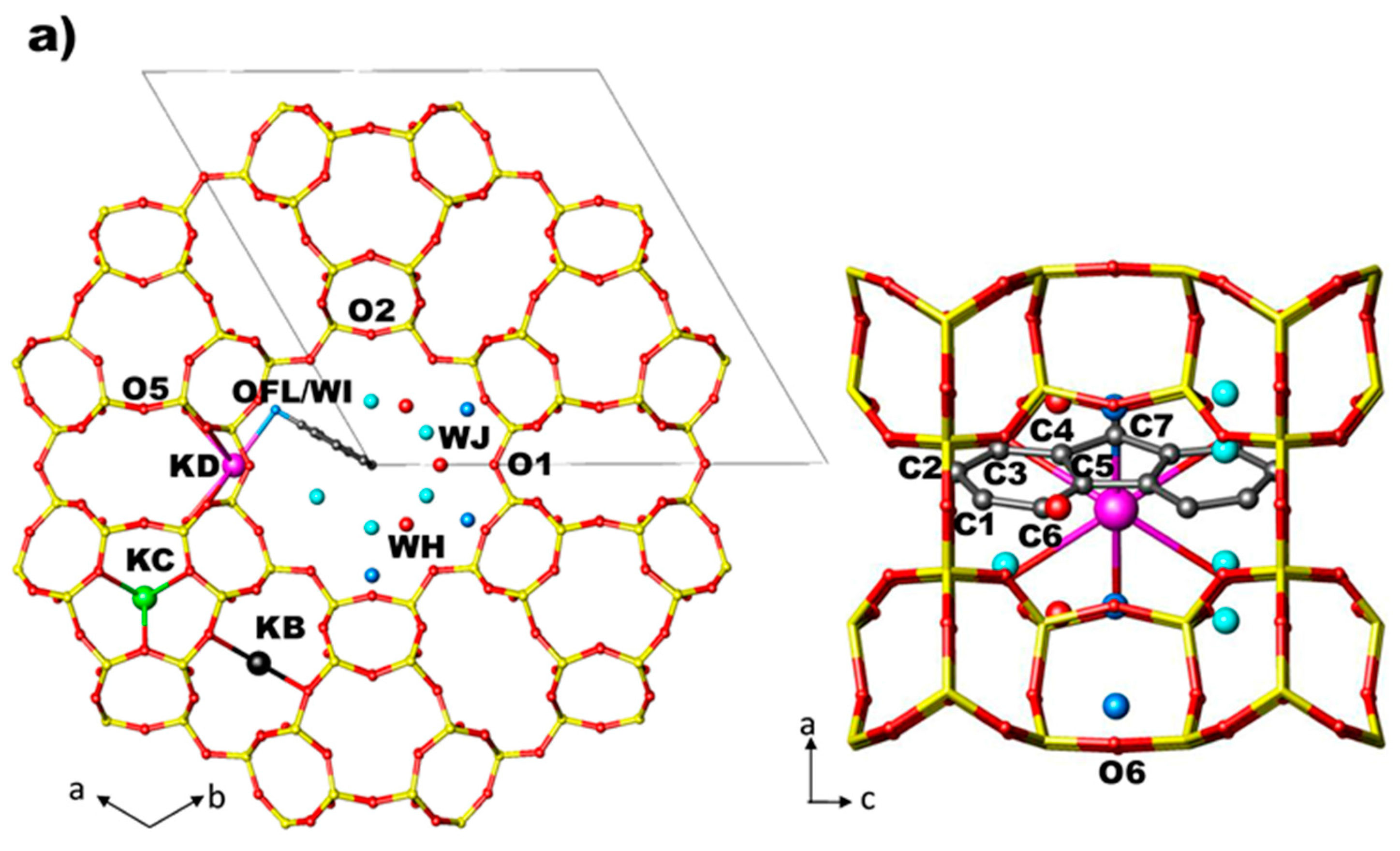
- (i)
- site KB—in the centre of the cancrinite cage—is fully occupied and coordinated to six framework oxygen atoms O3;
- (ii)
- site KC—in the centre of the 8 MR channel—is fully occupied and coordinated to four oxygen atoms O5;
- (iii)
- site KD—near the wall of the main 12 MR channel—is partially occupied and coordinated to six oxygen atoms (O4, O6), two water molecules (WH and WI) and to the oxygen atom of FL molecule (OFL).
- (i)
- the distances between KB, KC and the coordinating framework oxygen atoms (Table S3 and Ref. [56]) decrease as a consequence of the shape modifications of both 8 MR channel aperture and D6R. The distances between the cation in the main channel (KD) and O4 and O6 decrease as well. All these effects are more marked in the ZL/1FL system.
- (ii)
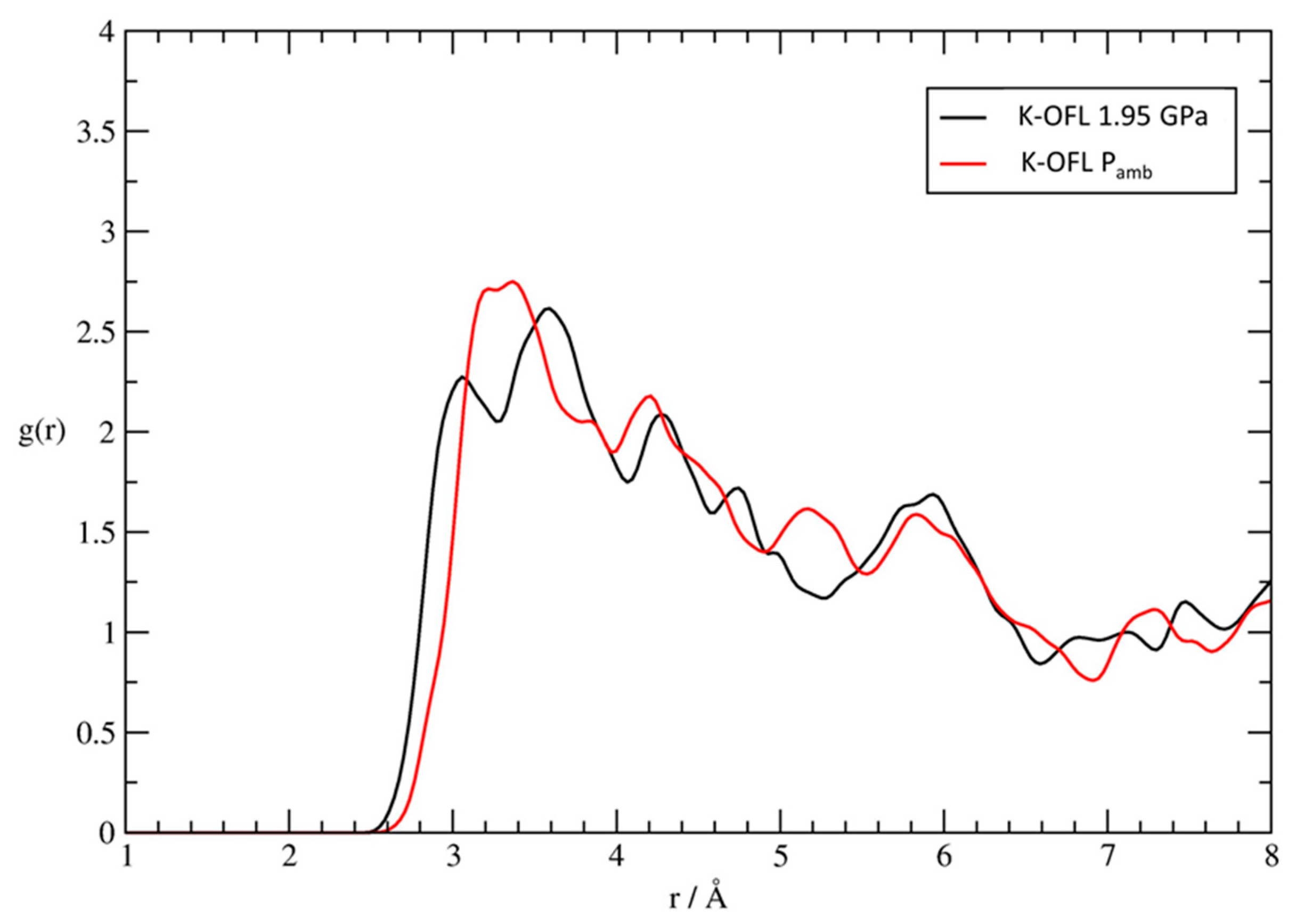
- OFL–KD distance decreases in ZL/0.5FL and remains almost constant in ZL/1.0FL.
- (iii)
- Compression induces the splitting of OFL/WI and C3/WJ sites, which in the Pamb structures of ZL/0.5FL and ZL/1FL samples [56], occupy single sites. After the splitting, WJ increases its distance from the framework O2 atom, approaching WI site (Table S3). After pressure release, the original positions are recovered in the ZL/0.5FL composite, while this does not happen in the ZL/1FL sample.
- (iv)
- At 2 GPa, the shape, orientation and arrangement of the fluorenone molecules in the main channel do not change with respect to ambient pressure.
3.2. Structure of the ZL/1.5FL Composite from First-Principles Molecular Dynamics
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Baerlocher, C.; McCusker, L.B.; Olson, D.H. Atlas of Zeolite Framework Types; Elsevier: Amsterdam, The Netherland, 2007; ISBN 9780444530646. [Google Scholar]
- Baur, W.; Fischer, R.X. Microporous and Other Framework Materials with Zeolite-Type Structures; Springer: Berlin, Germany, 2017; ISBN 9783662542514. [Google Scholar]
- Čejka, J.; Centi, G.; Perez-Pariente, J.; Roth, W.J. Zeolite-based materials for novel catalytic applications: Opportunities, perspectives and open problems. Catal. Today 2012, 179, 2–15. [Google Scholar] [CrossRef]
- Corma, A. Heterogeneous catalysis: Understanding for designing designing for applications. Angew. Chem. Int. Ed. 2016, 55, 6112–6113. [Google Scholar] [CrossRef] [PubMed]
- Seifert, R.; Kunzmann, A.; Calzaferri, G. The yellow color of silver-containing zeolite A. Angew. Chem. Int. Ed. 1998, 37, 1521–1524. [Google Scholar] [CrossRef]
- Baldansuren, A.; Roduner, E. EPR experiments of Ag species supported on NaA. Chem. Phys. Lett. 2009, 473, 135–137. [Google Scholar] [CrossRef]
- Coutiño-Gonzalez, E.; Baekelant, W.; Steele, J.A.; Kim, C.W.; Roeffaers, M.B.J.; Hofkens, J. Silver clusters in zeolites: From self-assembly to ground-breaking luminescent properties. Acc. Chem. Res. 2017, 50, 2353–2361. [Google Scholar] [CrossRef] [PubMed]
- Dong, B.; Retoux, R.; de Waele, V.; Chiodo, S.G.; Mineva, T.; Cardin, J.; Mintova, S. Sodalite cages of EMT zeolite confined neutral molecular-like silver clusters. Microporous Mesoporous Mater. 2017, 244, 74–82. [Google Scholar] [CrossRef]
- Kim, H.S.; Yoon, K.B. Preparation and characterization of CdS and PbS quantum dots in zeolite Y and their applications for nonlinear optical materials and solar cell. Coord. Chem. Rev. 2014, 263–264, 239–256. [Google Scholar] [CrossRef]
- Wang, Y.; Li, H. Luminescent materials of zeolite functionalized with lanthanides. CrystEngComm 2014, 16, 9764–9778. [Google Scholar] [CrossRef]
- Calzaferri, G.; Huber, S.; Maas, H.; Minkowski, C. Host-guest antenna materials. Angew. Chem. Int. Ed. 2003, 42, 3732–3758. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, S. Zeolite photochemistry: Impact of zeolites on photochemistry and feedback from photochemistry to zeolite science. J. Photochem. Photobiol. C Photochem. Rev. 2003, 4, 19–49. [Google Scholar] [CrossRef]
- Alarcos, N.; Cohen, B.; Ziółek, M.; Douhal, A. Photochemistry and photophysics in silica-based materials: Ultrafast and Single molecule spectroscopy observation. Chem. Rev. 2017, 117, 13639–13720. [Google Scholar] [CrossRef] [PubMed]
- Busby, M.; Devaux, A.; Blum, C.; Subramaniam, V.; Calzaferri, G.; De Cola, L. Interactions of perylene bisimide in the one-dimensional channels of zeolite L. J. Phys. Chem. C 2011, 115, 5974–5988. [Google Scholar] [CrossRef]
- Calzaferri, G.; Méallet-Renault, R.; Brühwiler, D.; Pansu, R.; Dolamic, I.; Dienel, T.; Adler, P.; Li, H.; Kunzmann, A. Designing dye-nanochannel antenna hybrid materials for light harvesting, transport and trapping. ChemPhysChem 2011, 12, 580–594. [Google Scholar] [CrossRef] [PubMed]
- Gartzia-Rivero, L.; Bañuelos, J.; López-Arbeloa, I. Photoactive nanomaterials inspired by nature: LTL zeolite doped with laser dyes as artificial light harvesting systems. Materials (Basel) 2017, 10, 495. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Martínez, V.; García, R.; Gómez-Hortigüela, L.; Sola Llano, R.; Pérez-Pariente, J.; López-Arbeloa, I. Highly luminescent and optically switchable hybrid material by one-pot encapsulation of dyes into MgAPO-11 unidirectional nanopores. ACS Photonics 2014, 1, 205–211. [Google Scholar] [CrossRef]
- Sola-Llano, R.; Fujita, Y.; Gómez-Hortigüela, L.; Alfayate, A.; Ujii, H.; Fron, E.; Toyouchi, S.; Perez-Pariente, J.; Lopez-Arbeloa, I.; Martinez-Martinez, V. One-directional antenna systems: Energy transfer from monomers to J-Aggregates within 1D nanoporous aluminophosphates. ACS Photonics 2017, 5, 151–157. [Google Scholar] [CrossRef]
- Ramamurthy, V.; Lakshminarasimhan, P.; Grey, C.P.; Johnston, L.J. Energy transfer, proton transfer and electron transfer reactions within zeolites. Chem. Commun. 1998, 2411–2424. [Google Scholar] [CrossRef]
- Calzaferri, G. Nanochannels: Hosts for the supramolecular organization of molecules and complexes. Langmuir 2012, 28, 6216–6231. [Google Scholar] [CrossRef] [PubMed]
- Shim, T.; Lee, M.H.; Kim, D.; Kim, H.S.; Yoon, K.B. Fluorescence Properties of Hemicyanine in the Nanoporous Materials with Varying Pore Sizes. J. Phys. Chem. B 2009, 113, 966–969. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Kim, H.S. Enhancement of fluorescence from one- and two-photon absorption of hemicyanine dyes by confinement in silicalite-1 nanochannels. Microporous Mesoporous Mater. 2017, 243, 69–75. [Google Scholar] [CrossRef]
- Doungmanee, S.; Siritanon, T.; Insuwan, W.; Jungsuttiwong, S.; Rangsriwatananon, K. Multi step energy transfer between three Si_LTL and SiGe_LTL zeolite-loaded dyes. J. Porous Mater. 2017, 1–9. [Google Scholar] [CrossRef]
- Hu, D.D.; Lin, J.; Zhang, Q.; Lu, J.N.; Wang, X.Y.; Wang, Y.W.; Bu, F.; Ding, L.F.; Wang, L.; Wu, T. Multi-Step Host-Guest Energy Transfer Between Inorganic Chalcogenide-Based Semiconductor Zeolite Material and Organic Dye Molecules. Chem. Mater. 2015, 27, 4099–4104. [Google Scholar] [CrossRef]
- Noh, T.H.; Jang, J.; Hong, W.; Lee, H.; Jung, O.-S. Truncated trigonal prismatic tubular crystals consisting of a zeolite L-mimic metal-organic framework. Chem. Commun. 2014, 50, 7451–7454. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, A.Z.; Brühwiler, D.; Ban, T.; Calzaferri, G. Synthesis of zeolite L. Tuning size and morphology. Monatshefte fur Chemie 2005, 136, 77–89. [Google Scholar] [CrossRef]
- Cho, H.S.; Hill, A.R.; Cho, M.; Miyasaka, K.; Jeong, K.; Anderson, M.W.; Kang, J.K.; Terasaki, O. Directing the distribution of potassium cations in zeolite-LTL through crown ether addition. Cryst. Growth Des. 2017, 17, 4516–4521. [Google Scholar] [CrossRef]
- Maas, H.; Calzaferri, G. Trapping energy from and injecting energy into dye-zeolite nanoantennae. Angew. Chem. Int. Ed. 2002, 41, 2284–2288. [Google Scholar] [CrossRef]
- Brühwiler, D.; Calzaferri, G. Selective functionalization of the external surface of zeolite L. C. R. Chim. 2005, 8, 391–398. [Google Scholar] [CrossRef]
- Dieu, L.-Q.; Devaux, A.; López-Duarte, I.; Victoria Martínez-Díaz, M.; Brühwiler, D.; Calzaferri, G.; Torres, T. Novel phthalocyanine-based stopcock for zeolite L. Chem. Commun. 2008, 0, 1187–1189. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Wang, Y.; Li, H.; Calzaferri, G. Luminescence enhancement after adding stoppers to europium(III) nanozeolite L. Angew. Chem. Int. Ed. 2014, 53, 2904–2909. [Google Scholar] [CrossRef] [PubMed]
- Tabacchi, G.; Fois, E.; Calzaferri, G. Structure of Nanochannel Entrances in Stopcock-Functionalized Zeolite L Composites. Angew. Chem. Int. Ed. 2015, 54, 11112–11116. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, R.Q.; Popović, Z.; De Cola, L.; Calzaferri, G. Luminescence quenching by O2 of a Ru2+ complex attached to zeolite L. ChemPhysChem 2006, 7, 1050–1053. [Google Scholar] [CrossRef] [PubMed]
- Beierle, J.M.; Roswanda, R.; Erne, P.M.; Coleman, A.C.; Browne, W.R.; Feringa, B.L. An improved method for site-specific end modification of zeolite L for the formation of zeolite L and gold nanoparticle self-assembled structures. Part. Part. Syst. Charact. 2013, 30, 273–279. [Google Scholar] [CrossRef]
- Ramachandra, S.; Popović, Z.D.; Schuermann, K.C.; Cucinotta, F.; Calzaferri, G.; De Cola, L. Förster resonance energy transfer in quantum dot-dye-loaded zeolite L nanoassemblies. Small 2011, 7, 1488–1494. [Google Scholar] [CrossRef] [PubMed]
- Popović, Z.; Otter, M.; Calzaferri, G.; De Cola, L. Self-Assembling living systems with functional nanomaterials. Angew. Chem. Int. Ed. 2007, 46, 6188–6191. [Google Scholar] [CrossRef] [PubMed]
- Strassert, C.A.; Otter, M.; Albuquerque, R.Q.; Hone, A.; Vida, Y.; Maier, B.; De Cola, L. Photoactive hybrid nanomaterial for targeting, labeling killing antibiotic-resistant bacteria. Angew. Chem. Int. Ed. 2009, 48, 7928–7931. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, H.; Feng, Y.; Zhang, H.; Calzaferri, G.; Ren, T. Orienting zeolite L microcrystals with a functional linker. Angew. Chem. Int. Ed. 2010, 49, 1434–1438. [Google Scholar] [CrossRef] [PubMed]
- Devaux, A.; Calzaferri, G.; Belser, P.; Cao, P.; Brühwiler, D.; Kunzmann, A. Efficient and robust host-guest antenna composite for light harvesting. Chem. Mater. 2014, 26, 6878–6885. [Google Scholar] [CrossRef]
- Brühwiler, D.; Calzaferri, G.; Torres, T.; Ramm, J.H.; Gartmann, N.; Dieu, L.-Q.; López-Duarte, I.; Martínez-Díaz, M.V. Nanochannels for supramolecular organization of luminescent guests. J. Mater. Chem. 2009, 19, 8040–8067. [Google Scholar] [CrossRef]
- Woodtli, P.; Giger, S.; Müller, P.; Sägesser, L.; Zucchetto, N.; Reber, M.J.; Ecker, A.; Brühwiler, D. Indigo in the nanochannels of zeolite L: Towards a new type of colorant. Dyes Pigments 2017, 149, 456–461. [Google Scholar] [CrossRef]
- El-Gindi, J.; Benson, K.; De Cola, L.; Galla, H.J.; Seda Kehr, N. Cell adhesion behavior on enantiomerically functionalized zeolite L monolayers. Angew. Chem. Int. Ed. 2012, 51, 3716–3720. [Google Scholar] [CrossRef] [PubMed]
- Greco, A.; Maggini, L.; De Cola, L.; De Marco, R.; Gentilucci, L. Diagnostic implementation of fast and selective integrin-mediated adhesion of cancer cells on functionalized zeolite L monolayers. Bioconjug. Chem. 2015, 26, 1873–1878. [Google Scholar] [CrossRef] [PubMed]
- Fois, E.; Tabacchi, G.; Calzaferri, G. Interactions, behavior stability of fluorenone inside zeolite nanochannels. J. Phys. Chem. C 2010, 114, 10572–10579. [Google Scholar] [CrossRef] [Green Version]
- Zhou, X.; Wesolowski, T.A.; Tabacchi, G.; Fois, E.; Calzaferri, G.; Devaux, A. First-principles simulation of the absorption bands of fluorenone in zeolite L. Phys. Chem. Chem. Phys. 2013, 15, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Fois, E.; Tabacchi, G.; Calzaferri, G. Orientation and order of xanthene dyes in the one-dimensional channels of zeolite L: Bridging the gap between experimental data and molecular behavior. J. Phys. Chem. C 2012, 116, 16784–16799. [Google Scholar] [CrossRef]
- Viani, L.; Minoia, A.; Cornil, J.; Beljonne, D.; Egelhaaf, H.J.; Gierschner, J. Resonant energy transport in dye-filled monolithic crystals of zeolite L: Modeling of inhomogeneity. J. Phys. Chem. C 2016, 120, 27192–27199. [Google Scholar] [CrossRef]
- Van Speybroeck, V.; Hemelsoet, K.; Joos, L.; Waroquier, M.; Bell, R.G.; Catlow, C.R.A. Advances in theory and their application within the field of zeolite chemistry. Chem. Soc. Rev. 2015, 44, 7044–7111. [Google Scholar] [CrossRef] [PubMed]
- Calzaferri, G. Entropy in multiple equilibria, theory and applications. Phys. Chem. Chem. Phys. 2017, 19, 10611–10621. [Google Scholar] [CrossRef] [PubMed]
- Tabacchi, G. Supramolecular Organization in Confined Nanospaces. ChemPhysChem 2018. in revision. [Google Scholar]
- Manzano, H.; Gartzia-Rivero, L.; Bañuelos, J.; López-Arbeloa, I. Ultraviolet-visible dual absorption by single BODIPY dye confined in LTL zeolite nanochannels. J. Phys. Chem. C 2013, 117, 13331–13336. [Google Scholar] [CrossRef]
- Cucinotta, F.; Guenet, A.; Bizzarri, C.; Mroz, W.; Botta, C.; Milian-Medina, B.; Gierschner, J.; De Cola, L. Energy transfer at the zeolite l boundaries: Towards photo- and electroresponsive materials. Chempluschem 2014, 79, 45–57. [Google Scholar] [CrossRef]
- Fois, E.; Gamba, A.; Medici, C.; Tabacchi, G. Intermolecular electronic excitation transfer in a confined space: A first-principles study. ChemPhysChem 2005, 6, 1917–1922. [Google Scholar] [CrossRef] [PubMed]
- Insuwan, W.; Rangsriwatananon, K.; Meeprasert, J.; Namuangruk, S.; Surakhot, Y.; Kungwan, N.; Jungsuttiwong, S. Combined experimental and theoretical investigation on photophysical properties of trans-azobenzene confined in LTL zeolite: Effect of cis-isomer forming. Microporous Mesoporous Mater. 2014, 197, 348–357. [Google Scholar] [CrossRef]
- Insuwan, W.; Rangsriwatananon, K.; Meeprasert, J.; Namuangruk, S.; Surakhot, Y.; Kungwan, N.; Jungsuttiwong, S. Combined experimental and theoretical investigation on Fluorescence Resonance Energy Transfer of dye loaded on LTL zeolite. Microporous Mesoporous Mater. 2017, 241, 372–382. [Google Scholar] [CrossRef]
- Gigli, L.; Arletti, R.; Tabacchi, G.; Fois, E.; Vitillo, J.G.; Martra, G.; Agostini, G.; Quartieri, S.; Vezzalini, G. Close-Packed dye molecules in zeolite channels self-assemble into supramolecular nanoladders. J. Phys. Chem. C 2014, 118, 15732–15743. [Google Scholar] [CrossRef]
- Gigli, L.; Arletti, R.; Tabacchi, G.; Fabbiani, M.; Vitillo, J.G.; Martra, G.; Devaux, A.; Miletto, I.; Quartieri, S.; Calzaferri, G.; et al. Structure and host-guest interactions of perylene-diimide dyes in zeolite l nanochannels. J. Phys. Chem. C 2018. accepted. [Google Scholar] [CrossRef]
- Fois, E.; Tabacchi, G.; Devaux, A.; Belser, P.; Brühwiler, D.; Calzaferri, G. Host-guest interactions and orientation of dyes in the one-dimensional channels of zeolite L. Langmuir 2013, 29, 9188–9198. [Google Scholar] [CrossRef] [PubMed]
- Tabacchi, G.; Calzaferri, G.; Fois, E. One-dimensional self-assembly of perylene-diimide dyes by unidirectional transit of zeolite channel openings. Chem. Commun. 2016, 52, 11195–11198. [Google Scholar] [CrossRef] [PubMed]
- Arletti, R.; Ferro, O.; Quartieri, S.; Sani, A.; Tabacchi, G.; Vezzalini, G. Structural deformation mechanisms of zeolites under pressure. Am. Mineral. 2003, 88, 1416–1422. [Google Scholar] [CrossRef]
- Gatta, G.D. A comparative study of fibrous zeolites under pressure. Eur. J. Mineral. 2005, 17, 411–422. [Google Scholar] [CrossRef]
- Gatta, G.D. Does porous mean soft? On the elastic behaviour and structural evolution of zeolites under pressure. Zeitschrift für Kristallographie Cryst. Mater. 2008, 223, 160–170. [Google Scholar] [CrossRef]
- Gatta, G.D.; Lee, Y. Zeolites at high pressure: A review. Mineral. Mag. 2014, 78, 267–291. [Google Scholar] [CrossRef]
- Vezzalini, G.; Arletti, R.; Quartieri, S. High-pressure-induced structural changes, amorphization and molecule penetration in MFI microporous materials: A review. Acta Crystallogr. Sect. B Struct. Sci. Cryst. Eng. Mater. 2014, 70, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Fraux, G.; Coudert, F.-X.; Boutin, A.; Fuchs, A.H. Forced intrusion of water and aqueous solutions in microporous materials: From fundamental thermodynamics to energy storage devices. Chem. Soc. Rev. 2017, 46, 7421–7437. [Google Scholar] [CrossRef] [PubMed]
- Gatta, G.D.; Lotti, P.; Tabacchi, G. The effect of pressure on open-framework silicates: Elastic behaviour and crystal–fluid interaction. Phys. Chem. Miner. 2017, 1–24. [Google Scholar] [CrossRef]
- Lee, Y.; Vogt, T.; Hriljac, J.A.; Parise, J.B.; Hanson, J.C.; Kim, S.J. Non-framework cation migration and irreversible pressure-induced hydration in a zeolite. Nature 2002, 420, 485–489. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Hriljac, J.A.; Parise, J.B.; Vogt, T. Pressure-induced hydration in zeolite tetranatrolite. Am. Mineral. 2006, 91, 247–251. [Google Scholar] [CrossRef]
- White, C.L.I.M.; Ruiz-Salvador, A.R.; Lewis, D.W. Pressure-Induced Hydration Effects in the Zeolite Laumontite. Angew. Chem. Int. Ed. 2004, 43, 469–472. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Kao, C.C.; Kim, S.J.; Lee, H.H.; Lee, D.R.; Shin, T.J.; Choi, J.Y. Water nanostructures confined inside the quasi-one-dimensional channels of LTL zeolite. Chem. Mater. 2007, 19, 6252–6257. [Google Scholar] [CrossRef]
- Lee, Y.; Kim, S.J.; Ahn, D.C.; Shin, N.S. Confined water clusters in a synthetic rubidium gallosilicate with zeolite LTL topology. Chem. Mater. 2007, 19, 2277–2282. [Google Scholar] [CrossRef]
- Seoung, D.; Lee, Y.; Kao, C.-C.; Vogt, T.; Lee, Y. Two-Step pressure-induced superhydration in small pore natrolite with divalent extra-framework cations. Chem. Mater. 2015, 27, 3874–3880. [Google Scholar] [CrossRef]
- Cailliez, F.; Trzpit, M.; Soulard, M.; Demachy, I.; Boutin, A.; Patarin, J.; Fuchs, A.H. Thermodynamics of water intrusion in nanoporous hydrophobic solids. Phys. Chem. Chem. Phys. 2008, 10, 4817. [Google Scholar] [CrossRef] [PubMed]
- Likhacheva, A.Y.; Seryotkin, Y.V.; Manakov, A.Y.; Goryainov, S.V.; Ancharov, A.I.; Sheromov, M.A. Pressure-induced over-hydration of thomsonite: A synchrotron powder diffraction study. Am. Mineral. 2007, 92, 1610–1615. [Google Scholar] [CrossRef]
- Likhacheva, A.; Seryotkin, Y.; Manakov, A.; Goryainov, S.; Ancharov, A.; Sheromov, M. Anomalous compression of scolecite and thomsonite in aqueous medium to 2 GPa. High Press. Res. 2006, 26, 449–453. [Google Scholar] [CrossRef]
- Arletti, R.; Vezzalini, G.; Quartieri, S.; Di Renzo, F.; Dmitriev, V. Pressure-induced water intrusion in FER-type zeolites and the influence of extraframework species on structural deformations. Microporous Mesoporous Mater. 2014, 191, 27–37. [Google Scholar] [CrossRef]
- Arletti, R.; Vezzalini, G.; Morsli, A.; Di Renzo, F.; Dmitriev, V.; Quartieri, S. Elastic behavior of MFI-type zeolites: 1-Compressibility of Na-ZSM-5 in penetrating and non-penetrating media. Microporous Mesoporous Mater. 2011, 142, 696–707. [Google Scholar] [CrossRef]
- Lotti, P.; Gatta, G.D.; Merlini, M.; Liermann, H.-P. High-pressure behavior of synthetic mordenite-Na: An in situ single-crystal synchrotron X-ray diffraction study. Zeitschrift für Kristallographie Cryst. Mater. 2015, 230, 201–211. [Google Scholar] [CrossRef]
- Comboni, D.; Gatta, G.D.; Lotti, P.; Merlini, M.; Hanfland, M. Crystal-fluid interactions in laumontite. Microporous Mesoporous Mater. 2018, 263, 86–95. [Google Scholar] [CrossRef]
- Lotti, P.; Arletti, R.; Gatta, G.D.; Quartieri, S.; Vezzalini, G.; Merlini, M.; Dmitriev, V.; Hanfland, M. Compressibility and crystal–fluid interactions in all-silica ferrierite at high pressure. Microporous Mesoporous Mater. 2015, 218, 42–54. [Google Scholar] [CrossRef] [Green Version]
- Lotti, P.; Gatta, G.D.; Comboni, D.; Merlini, M.; Pastero, L.; Hanfland, M. AlPO4-5 zeolite at high pressure: Crystal-fluid interaction and elastic behavior. Microporous Mesoporous Mater. 2016, 228, 158–167. [Google Scholar] [CrossRef]
- Kim, Y.; Choi, J.; Vogt, T.; Lee, Y. Structuration under pressure: Spatial separation of inserted water during pressure-induced hydration in mesolite. Am. Mineral. 2018, 103, 175–178. [Google Scholar] [CrossRef]
- Goryainov, S.V.; Secco, R.A.; Huang, Y.; Likhacheva, A.Y. Pressure-induced ionic conductivity of overhydrated zeolite NaA at different water/zeolite ratios. Microporous Mesoporous Mater. 2013, 171, 125–130. [Google Scholar] [CrossRef]
- Lee, Y.; Lee, Y.; Seoung, D.; Im, J.H.; Hwang, H.J.; Kim, T.H.; Liu, D.; Liu, Z.; Lee, S.Y.; Kao, C.C.; et al. Immobilization of large, aliovalent cations in the small-pore zeolite K-natrolite by means of pressure. Angew. Chem. Int. Ed. 2012, 51, 4848–4851. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Chen, X.; Ma, Y.; Liu, Z.; Vogt, T.; Lee, Y. Spectroscopic and computational characterizations of alkaline-earth- and heavy-metal-exchanged natrolites. ChemPlusChem 2014, 79, 1096–1102. [Google Scholar] [CrossRef]
- Im, J.; Seoung, D.; Hwang, G.C.; Jun, J.W.; Jhung, S.H.; Kao, C.-C.; Vogt, T.; Lee, Y. Pressure-Dependent structural and chemical changes in a metal–organic framework with one-dimensional pore structure. Chem. Mater. 2016, 28, 5336–5341. [Google Scholar] [CrossRef]
- Kremleva, A.; Vogt, T.; Rösch, N. Monovalent cation-exchanged natrolites and their behavior under pressure. A computational study. J. Phys. Chem. C 2013, 117, 19020–19030. [Google Scholar] [CrossRef]
- Kremleva, A.; Vogt, T.; Rösch, N. Potassium-exchanged natrolite under pressure. computational study vs experiment. J. Phys. Chem. C 2014, 118, 22030–22039. [Google Scholar] [CrossRef]
- Santoro, M.; Gorelli, F.A.; Bini, R.; Haines, J.; van der Lee, A. High-pressure synthesis of a polyethylene/zeolite nano-composite material. Nat. Commun. 2013, 4, 1557. [Google Scholar] [CrossRef] [PubMed]
- Santoro, M.; Dziubek, K.; Scelta, D.; Ceppatelli, M.; Gorelli, F.A.; Bini, R.; Thibaud, J.M.; Di Renzo, F.; Cambon, O.; Rouquette, J.; et al. High pressure synthesis of all-transoid polycarbonyl [-(C=O)-]n in a zeolite. Chem. Mater. 2015, 27, 6486–6489. [Google Scholar] [CrossRef]
- Scelta, D.; Ceppatelli, M.; Santoro, M.; Bini, R.; Gorelli, F.A.; Perucchi, A.; Mezouar, M.; Van Der Lee, A.; Haines, J. High pressure polymerization in a confined space: Conjugated chain/zeolite nanocomposites. Chem. Mater. 2014, 26, 2249–2255. [Google Scholar] [CrossRef]
- Santoro, M.; Scelta, D.; Dziubek, K.; Ceppatelli, M.; Gorelli, F.A.; Bini, R.; Garbarino, G.; Thibaud, J.M.; Di Renzo, F.; Cambon, O.; et al. Synthesis of 1D polymer/zeolite nanocomposites under high pressure. Chem. Mater. 2016, 28, 4065–4071. [Google Scholar] [CrossRef]
- Jordá, J.L.; Rey, F.; Sastre, G.; Valencia, S.; Palomino, M.; Corma, A.; Segura, A.; Errandonea, D.; Lacomba, R.; Manjón, F.J.; et al. Synthesis of a novel zeolite through a pressure-induced reconstructive phase transition process. Angew. Chem. Int. Ed. 2013, 52, 10458–10462. [Google Scholar] [CrossRef] [PubMed]
- Arletti, R.; Ronchi, L.; Quartieri, S.; Vezzalini, G.; Ryzhikov, A.; Nouali, H.; Daou, T.J.; Patarin, J. Intrusion-extrusion experiments of MgCl2 aqueous solution in pure silica ferrierite: Evidence of the nature of intruded liquid by in situ high pressure synchrotron X-ray powder diffraction. Microporous Mesoporous Mater. 2016, 235, 253–260. [Google Scholar] [CrossRef]
- Arletti, R.; Leardini, L.; Vezzalini, G.; Quartieri, S.; Gigli, L.; Santoro, M.; Haines, J.; Rouquette, J.; Konczewicz, L. Pressure-induced penetration of guest molecules in high-silica zeolites: The case of mordenite. Phys. Chem. Chem. Phys. 2015, 17, 24262–24274. [Google Scholar] [CrossRef] [PubMed]
- Santamaría-Pérez, D.; Marqueño, T.; MacLeod, S.; Ruiz-Fuertes, J.; Daisenberger, D.; Chuliá-Jordan, R.; Errandonea, D.; Jordá, J.L.; Rey, F.; McGuire, C.; et al. Structural evolution of CO2-filled pure silica LTA zeolite under high-pressure high-temperature conditions. Chem. Mater. 2017, 29, 4502–4510. [Google Scholar] [CrossRef]
- Arletti, R.; Fois, E.; Gigli, L.; Vezzalini, G.; Quartieri, S.; Tabacchi, G. Irreversible Conversion of a water–ethanol solution into an organized two-dimensional network of alternating supramolecular units in a hydrophobic zeolite under pressure. Angew. Chem. Int. Ed. 2017, 56, 2105–2109. [Google Scholar] [CrossRef] [PubMed]
- Car, R.; Parrinello, M. Unified approach for molecular dynamics and density-functional theory. Phys. Rev. Lett. 1985, 55, 2471–2474. [Google Scholar] [CrossRef] [PubMed]
- Gigli, L.; Arletti, R.; Quartieri, S.; Di Renzo, F.; Vezzalini, G. The high thermal stability of the synthetic zeolite K–L: Dehydration mechanism by in situ SR-XRPD experiments. Microporous Mesoporous Mater. 2013, 177, 8–16. [Google Scholar] [CrossRef]
- Merrill, L.; Bassett, W.A. Miniature diamond anvil pressure cell for single crystal X-ray diffraction studies. Rev. Sci. Instrum. 1974, 45, 290–294. [Google Scholar] [CrossRef]
- Miletich, R.; Allan, D.R.; Kuhs, W.F. High-Pressure single-crystal techniques. Rev. Mineral. Geochem. 2000, 41, 445–519. [Google Scholar] [CrossRef]
- Angel, R.J.; Bujak, M.; Zhao, J.; Gatta, G.D.; Jacobsen, S.D. Effective hydrostatic limits of pressure media for high-pressure crystallographic studies. J. Appl. Crystallogr. 2007, 40, 26–32. [Google Scholar] [CrossRef]
- Hammersley, A.P.; Svensson, S.O.; Hanfland, M.; Fitch, A.N.; Hausermann, D. Two-dimensional detector software: From real detector to idealised image or two-theta scan. High Press. Res. 1996, 14, 235–248. [Google Scholar] [CrossRef]
- Larson, A.C.; Von Dreele, R.B. General Structure Analysis System (GSAS) Program; Los Alamos National Laboratory: Los Alamos, NM, USA, 1994.
- Toby, B.H. EXPGUI, a graphical user interface for GSAS. J. Appl. Crystallogr. 2001, 34, 210–213. [Google Scholar] [CrossRef]
- Thompson, P.; Cox, D.E.; Hastings, J.B. Rietveld refinement of Debye–Scherrer synchrotron X-ray data from Al2O3. J. Appl. Crystallogr. 1987, 20, 79–83. [Google Scholar] [CrossRef]
- Grimme, S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 2006, 27, 1787–1799. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Suarez, L.; Siemer, N.; Frenzel, J.; Marx, D. Reaction network of methanol synthesis over Cu/ZnO Nanocatalysts. ACS Catal. 2015, 5, 4201–4218. [Google Scholar] [CrossRef]
- Bandoli, G.; Barreca, D.; Gasparotto, A.; Seraglia, R.; Tondello, E.; Devi, A.; Fischer, R.A.; Winter, M.; Fois, E.; Gamba, A.; et al. An integrated experimental and theoretical investigation on Cu(hfa)2·TMEDA: Structure, bonding and reactivity. Phys. Chem. Chem. Phys. 2009, 11, 5998–6007. [Google Scholar] [CrossRef] [PubMed]
- Fois, E.; Tabacchi, G.; Barreca, D.; Gasparotto, A.; Tondello, E. “Hot” Surface activation of molecular complexes: Insight from modeling studies. Angew. Chem. Int. Ed. 2010, 49, 1944–1948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martínez-Suárez, L.; Frenzel, J.; Marx, D. Cu/ZnO nanocatalysts in response to environmental conditions: Surface morphology, electronic structure, redox state and CO2 activation. Phys. Chem. Chem. Phys. 2014, 16, 26119–26136. [Google Scholar] [CrossRef] [PubMed]
- Barreca, D.; Fois, E.; Gasparotto, A.; Seraglia, R.; Tondello, E.; Tabacchi, G. How does CuII convert into CuI? An unexpected ring-mediated single-electron reduction. Chem. Eur. J. 2011, 17, 10864–10870. [Google Scholar] [CrossRef] [PubMed]
- Tabacchi, G.; Fois, E.; Barreca, D.; Gasparotto, A. Opening the Pandora’s jar of molecule-to-material conversion in chemical vapor deposition: Insights from theory. Int. J. Quantum Chem. 2014, 114, 1–7. [Google Scholar] [CrossRef]
- Pietrucci, F.; Andreoni, W. Fate of a graphene flake: A new route toward fullerenes disclosed with ab initio simulations. J. Chem. Theory Comput. 2014, 10, 913–917. [Google Scholar] [CrossRef] [PubMed]
- Fois, E.; Gamba, A.; Tabacchi, G.; Coluccia, S.; Martra, G. Ab initio study of defect sites at the inner surfaces of mesoporous silicas. J. Phys. Chem. B 2003, 107, 10767–10772. [Google Scholar] [CrossRef]
- Tabacchi, G.; Fois, E.; Barreca, D.; Gasparotto, A. CVD precursors for transition metal oxide nanostructures: Molecular properties, surface behavior and temperature effects. Phys. Status Solidi 2014, 211, 251–259. [Google Scholar] [CrossRef]
- Kraus, P.; Frank, I. On the dynamics of H2 adsorption on the Pt(111) surface. Int. J. Quantum Chem. 2017, 117, e25407. [Google Scholar] [CrossRef]
- Tabacchi, G.; Fois, E.; Barreca, D.; Carraro, G.; Gasparotto, A.; Maccato, C. Advanced Processing and Manufacturing Technologies for Nanostructured and Multifunctional Materials II; Ohji, T., Singh, M., Halbig, M., Eds.; Ceramic Engineering and Science Proceedings; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015; ISBN 9781119211662. [Google Scholar]
- Maccato, C.; Bigiani, L.; Carraro, G.; Gasparotto, A.; Seraglia, R.; Kim, J.; Devi, A.; Tabacchi, G.; Fois, E.; Pace, G.; et al. Molecular engineering of Mn II diamine diketonate precursors for the vapor deposition of manganese oxide nanostructures. Chem. Eur. J. 2017, 23, 17954–17963. [Google Scholar] [CrossRef] [PubMed]
- Koizumi, K.; Nobusada, K.; Boero, M. Reducing the cost and preserving the reactivity in noble-metal-based catalysts: Oxidation of CO by Pt and Al-Pt alloy clusters supported on graphene. Chem. Eur. J. 2016, 22, 5181–5188. [Google Scholar] [CrossRef] [PubMed]
- Deiana, C.; Tabacchi, G.; Maurino, V.; Coluccia, S.; Martra, G.; Fois, E. Surface features of TiO2 nanoparticles: Combination modes of adsorbed CO probe the stepping of (101) facets. Phys. Chem. Chem. Phys. 2013, 15, 13391–13399. [Google Scholar] [CrossRef] [PubMed]
- Deiana, C.; Fois, E.; Martra, G.; Narbey, S.; Pellegrino, F.; Tabacchi, G. On the simple complexity of carbon monoxide on oxide surfaces: Facet-Specific donation and backdonation effects revealed on TiO2 anatase nanoparticles. ChemPhysChem 2016, 17, 1956–1960. [Google Scholar] [CrossRef] [PubMed]
- Fois, E.; Gamba, A.; Tabacchi, G. Electronic spectra of Ti(IV) in zeolites: An ab initio approach. ChemPhysChem 2005, 6, 1237–1239. [Google Scholar] [CrossRef] [PubMed]
- Fois, E.; Gamba, A.; Tabacchi, G. Structure and dynamics of a Brønsted acid site in a zeolite: An ab initio study of hydrogen sodalite. J. Phys. Chem. B 1998, 102, 3974–3979. [Google Scholar] [CrossRef]
- Fois, E.; Gamba, A.; Tabacchi, G. First-principles simulation of the intracage oxidation of nitrite to nitrate sodalite. Chem. Phys. Lett. 2000, 329, 1–6. [Google Scholar] [CrossRef]
- Tabacchi, G.; Silvi, S.; Venturi, M.; Credi, A.; Fois, E. Dethreading of a photoactive azobenzene-containing molecular axle from a crown ether ring: A computational investigation. ChemPhysChem 2016, 17, 1913–1919. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Santiburcio, D.; Marx, D. Chemistry in nanoconfined water. Chem. Sci. 2017, 8, 3444–3452. [Google Scholar] [CrossRef] [PubMed]
- Fois, E.; Gamba, A.; Tabacchi, G. Intracage chemistry: Nitrite to nitrate oxidation via molecular oxygen. A Car Parrinello study. Stud. Surf. Sci. Catal. 2001, 140, 251–268. [Google Scholar] [CrossRef]
- Tabacchi, G.; Gianotti, E.; Fois, E.; Martra, G.; Marchese, L.; Coluccia, S.; Gamba, A. Understanding the vibrational and electronic features of Ti(IV) sites in mesoporous silicas by integrated ab initio and spectroscopic investigations. J. Phys. Chem. C 2007, 111, 4946–4955. [Google Scholar] [CrossRef]
- Fois, E.; Gamba, A.; Spano, E.; Tabacchi, G. Rotation of molecules and ions in confined spaces: A first-principles simulation study. J. Mol. Struct. 2003, 644, 55–66. [Google Scholar] [CrossRef]
- Spanó, E.; Tabacchi, G.; Gamba, A.; Fois, E. On the role of Ti(IV) as a Lewis acid in the chemistry of titanium zeolites: Formation, structure, reactivity aging of Ti-peroxo oxidizing intermediates. A first principles study. J. Phys. Chem. B 2006, 110, 21651–21661. [Google Scholar] [CrossRef] [PubMed]
- Gamba, A.; Tabacchi, G.; Fois, E. TS-1 from first principles. J. Phys. Chem. A 2009, 113, 15006–15015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tabacchi, G.; Vanoni, M.A.; Gamba, A.; Fois, E. Does negative hyperconjugation assist enzymatic dehydrogenations? ChemPhysChem 2007, 8, 1283–1288. [Google Scholar] [CrossRef] [PubMed]
- Trudu, F.; Tabacchi, G.; Gamba, A.; Fois, E. First principles studies on boron sites in zeolites. J. Phys. Chem. A 2007, 111, 11626–11637. [Google Scholar] [CrossRef] [PubMed]
- Fois, E.; Gamba, A.; Medici, C.; Tabacchi, G.; Quartieri, S.; Mazzucato, E.; Arletti, R.; Vezzalini, G.; Dmitriev, V. High pressure deformation mechanism of Li-ABW: Synchrotron XRPD study and ab initio molecular dynamics simulations. Microporous Mesoporous Mater. 2008, 115, 267–280. [Google Scholar] [CrossRef]
- Gatta, G.D.; Tabacchi, G.; Fois, E.; Lee, Y. Behaviour at high pressure of Rb7NaGa8Si12O40·3H2O (a zeolite with EDI topology): A combined experimental–computational study. Phys. Chem. Miner. 2016, 43, 209–216. [Google Scholar] [CrossRef]
- Arletti, R.; Fois, E.; Tabacchi, G.; Quartieri, S.; Vezzalini, G. Pressure-Induced penetration of water-ethanol mixtures in all-silica ferrierite. Adv. Sci. Lett. 2017, 23, 5966–5969. [Google Scholar] [CrossRef]
- Fois, E.; Gamba, A.; Tabacchi, G.; Quartieri, S.; Arletti, R.; Vezzalini, G. High-pressure behaviour of yugawaralite at different water content: An ab initio study. Stud. Surf. Sci. Catal. 2005, 155, 271–280. [Google Scholar] [CrossRef]
- Ferro, O.; Quartieri, S.; Vezzalini, G.; Fois, E.; Gamba, A.; Tabacchi, G. High-pressure behavior of bikitaite: An integrated theoretical and experimental approach. Am. Mineral. 2002, 87, 1415–1425. [Google Scholar] [CrossRef]
- Fois, E.; Gamba, A.; Tabacchi, G.; Ferro, O.; Quartieri, S.; Vezzalini, G. A theoretical investigation on pressure-induced changes in the vibrational spectrum of zeolite bikitaite. Stud. Surf. Sci. Catal. 2002, 142, 1877–1884. [Google Scholar] [CrossRef]
- Marx, D.; Hutter, J. Ab Initio Molecular Dynamics: Basic Theory and Advanced Methods; Cambridge University Press: Cambridge, UK, 2009; ISBN 9780511609633. [Google Scholar]
- Copyright IBM Corp. 1990–2017; MPI für Festkörperforschung Stuttgart 1997–2001. CPMD Code: Car Parrinello Molecular Dynamics. Available online: http://www.cpmd.org/.
- Bougeard, D.; Smirnov, K.S. Modelling studies of water in crystalline nanoporous aluminosilicates. Phys. Chem. Chem. Phys. 2007, 9, 226–245. [Google Scholar] [CrossRef] [PubMed]
- Demontis, P.; Gulín-González, J.; Masia, M.; Suffritti, G.B. The behaviour of water confined in zeolites: Molecular dynamics simulations versus experiment. J. Phys. Condens. Matter 2010, 22, 284106. [Google Scholar] [CrossRef] [PubMed]
- Fois, E.; Gamba, A.; Tabacchi, G.; Quartieri, S.; Vezzalini, G. On the collective properties of water molecules in one-dimensional zeolitic channels. Phys. Chem. Chem. Phys. 2001, 3, 4158–4163. [Google Scholar] [CrossRef]
- Quartieri, S.; Sani, A.; Vezzalini, G.; Galli, E.; Fois, E.; Gamba, A.; Tabacchi, G. One-dimensional ice in bikitaite: Single-crystal X-ray diffraction, infra-red spectroscopy and ab initio molecular dynamics studies. Microporous Mesoporous Mater. 1999, 30, 77–87. [Google Scholar] [CrossRef]
- Fois, E.; Tabacchi, G.; Quartieri, S.; Vezzalini, G. Dipolar host/guest interactions and geometrical confinement at the basis of the stability of one-dimensional ice in zeolite bikitaite. J. Chem. Phys. 1999, 111, 355–359. [Google Scholar] [CrossRef]
- Godelitsas, A.; Armbruster, T. HEU-type zeolites modified by transition elements and lead. Microporous Mesoporous Mater. 2003, 61, 3–24. [Google Scholar] [CrossRef]
- Martucci, A.; Alberti, A.; de Lourdes Guzman-Castillo, M.; Di Renzo, F.; Fajula, F. Crystal structure of zeolite omega, the synthetic counterpart of the natural zeolite mazzite. Microporous Mesoporous Mater. 2003, 63, 33–42. [Google Scholar] [CrossRef]
- Fois, E.; Gamba, A.; Tabacchi, G.; Quartieri, S.; Vezzalini, G. Water molecules in single file: First-principles studies of one-dimensional water chains in zeolites. J. Phys. Chem. B 2001, 105, 3012–3016. [Google Scholar] [CrossRef]
- Ceriani, C.; Fois, E.; Gamba, A.; Tabacchi, G.; Ferro, O.; Quartieri, S.; Vezzalini, G. Dehydration dynamics of bikitaite: Part II. Ab initio molecular dynamics study. Am. Mineral. 2004, 89, 102–109. [Google Scholar] [CrossRef]
- Demontis, P.; Gulín-Gonzalez, J.; Suffritti, G.B. Molecular dynamics simulation study of superhydrated perdeuterated natrolite using a new interaction potential model. J. Phys. Chem. B 2006, 110, 7513–7518. [Google Scholar] [CrossRef] [PubMed]
- Demontis, P.; Gulìn-Gonzàlez, J.; Jobic, H.; Masia, M.; Sale, R.; Suffritti, G.B. Dynamical properties of confined water nanoclusters: Simulation study of hydrated zeolite NaA: Structural and vibrational properties. ACS Nano 2008, 2, 1603–1614. [Google Scholar] [CrossRef] [PubMed]
- Trudu, F.; Tabacchi, G.; Gamba, A.; Fois, E. Water in acid boralites: Hydration effects on framework B sites. J. Phys. Chem. C 2008, 112, 15394–15401. [Google Scholar] [CrossRef] [Green Version]
- Fois, E.; Gamba, A.; Trudu, F.; Tabacchi, G. H2O-induced trigonal-to-tetrahedral transition in boron zeolites. Nuovo Cimento della Societa Italiana di Fisica. B Gen. Phys. Relativ. Astron. Math. Phys. Methods 2008, 123, 1567–1574. [Google Scholar] [CrossRef]
- Wang, C.H.; Bai, P.; Siepmann, J.I.; Clark, A.E. Deconstructing hydrogen-bond networks in confined nanoporous materials: Implications for alcohol-water separation. J. Phys. Chem. C 2014, 118, 19723–19732. [Google Scholar] [CrossRef]
- Zhou, T.; Bai, P.; Siepmann, J.I.; Clark, A.E. Deconstructing the confinement effect upon the organization and dynamics of water in hydrophobic nanoporous materials: Lessons Learned from zeolites. J. Phys. Chem. C 2017, 121, 22015–22024. [Google Scholar] [CrossRef]
- Fois, E.; Gamba, A.; Tabacchi, G. Bathochromic effects in electronic excitation spectra of hydrated Ti zeolites: A theoretical characterization. ChemPhysChem 2008, 9, 538–543. [Google Scholar] [CrossRef] [PubMed]
- Balestra, S.R.G.; Hamad, S.; Ruiz-Salvador, A.R.; Domínguez−García, V.; Merkling, P.J.; Dubbeldam, D.; Calero, S. Understanding nanopore window distortions in the reversible molecular valve zeolite RHO. Chem. Mater. 2015, 27, 5657–5667. [Google Scholar] [CrossRef]
- Fischer, M. Water adsorption in SAPO-34: Elucidating the role of local heterogeneities and defects using dispersion-corrected DFT calculations. Phys. Chem. Chem. Phys. 2015, 17, 25260–25271. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M. Structure and bonding of water molecules in zeolite hosts: Benchmarking plane-wave DFT against crystal structure data. Zeitschrift für Kristallographie Cryst. Mater. 2015, 230, 325–336. [Google Scholar] [CrossRef]
- Fischer, M. Interaction of water with (silico)aluminophosphate zeotypes: A comparative investigation using dispersion-corrected DFT. Phys. Chem. Chem. Phys. 2016, 18, 15738–15750. [Google Scholar] [CrossRef] [PubMed]
- Alabarse, F.G.; Haines, J.; Cambon, O.; Levelut, C.; Bourgogne, D.; Haidoux, A.; Granier, D.; Coasne, B. Freezing of water confined at the nanoscale. Phys. Rev. Lett. 2012, 109, 35701. [Google Scholar] [CrossRef] [PubMed]
- Coudert, F.-X.; Vuilleumier, R.; Boutin, A. Dipole moment, hydrogen bonding and IR spectrum of confined water. ChemPhysChem 2006, 7, 2464–2467. [Google Scholar] [CrossRef] [PubMed]
- Coudert, F.-X.; Cailliez, F.; Vuilleumier, R.; Fuchs, A.H.; Boutin, A. Water nanodroplets confined in zeolite pores. Faraday Discuss. 2009, 141, 377–398. [Google Scholar] [CrossRef] [PubMed]
- Fischer, R.X.; Sehovic, M.; Baur, W.H.; Paulmann, C.; Gesing, T.M. Crystal structure and morphology of fully hydrated zeolite Na-A. Zeitschrift für Kristallographie 2012, 227, 438–445. [Google Scholar] [CrossRef]
- Fois, E.; Gamba, A.; Tilocca, A. Structure and dynamics of the flexible triple helix of water inside VPI-5 molecular sieves. J. Phys. Chem. B 2002, 106, 4806–4812. [Google Scholar] [CrossRef]
- Hernandez-Tamargo, C.E.; Roldan, A.; Ngoepe, P.E.; De Leeuw, N.H. Periodic modeling of zeolite Ti-LTA. J. Chem. Phys. 2017, 147, 74701. [Google Scholar] [CrossRef] [PubMed]
- Fois, E.; Gamba, A.; Tabacchi, G.; Arletti, R.; Quartieri, S.; Vezzalini, G. The “template” effect of the extra-framework content on zeolite compression: The case of yugawaralite. Am. Mineral. 2005, 90, 28–35. [Google Scholar] [CrossRef]
- Betti, C.; Fois, E.; Mazzucato, E.; Medici, C.; Quartieri, S.; Tabacchi, G.; Vezzalini, G.; Dmitriev, V. Gismondine under HP: Deformation mechanism and re-organization of the extra-framework species. Microporous Mesoporous Mater. 2007, 103, 190–209. [Google Scholar] [CrossRef]
- Ballone, P.; Quartieri, S.; Sani, A.; Vezzalini, G. High-pressure deformation mechanism in scolecite: A combined computational-experimental study. Am. Mineral. 2002, 87, 1194–1206. [Google Scholar] [CrossRef]



| ZL/0.5FL | |||
| P (GPa) | Pamb 1 | 2 GPa | Pamb (rev) |
| Space group | P6/mmm | P6/mmm | P6/mmm |
| a (Å) | 18.3860 (4) | 18.1788 (9) | 18.4349 (8) |
| c (Å) | 7.5228 (2) | 7.3866 (4) | 7.5498 (6) |
| V (Å) | 2202.4 (1) | 2114.0 (2) | 2222.0 (2) |
| R F**2 (%) | 7.3 | 12.3 | 11.2 |
| No. variables | 73 | 82 | 82 |
| No. obs. | 1319 | 2474 | 2474 |
| No. refl. | 944 | 618 | 618 |
| ZL/1FL | |||
| P (GPa) | Pamb 1 | 2 GPa | Pamb (rev) |
| Space group | P6/mmm | P6/mmm | P6/mmm |
| a (Å) | 18.3940 (6) | 18.250 (1) | 18.3962 (8) |
| c (Å) | 7.5203 (3) | 7.4063 (5) | 7.5189 (4) |
| V (Å) | 2203.5 (1) | 2136.3 (2) | 2203.6 (2) |
| R F**2 (%) | 7.8 | 11.9 | 13.7 |
| No. variables | 81 | 83 | 83 |
| No. obs. | 1319 | 2422 | 2404 |
| No. refl. | 946 | 679 | 639 |
| ZL/0.5FL | ZL/1FL | |||||
|---|---|---|---|---|---|---|
| Pamb 1 | 2 GPa | Pamb (rev) | Pamb 1 | 2 GPa | Pamb (rev) | |
| 12 MR | 12 MR | |||||
| O1–O1 | 10.10 | 9.76 | 9.95 | 10.14 | 10.30 | 10.00 |
| O2–O2 | 10.52 | 10.43 | 10.56 | 10.46 | 10.12 | 10.45 |
| 8 MR ⊥ [001] | 8 MR ⊥ [001] | |||||
| O1–O1 | 8.29 | 8.41 | 8.48 | 8.26 | 7.95 | 8.39 |
| O5–O5 | 4.63 | 4.54 | 4.75 | 4.60 | 4.70 | 5.22 |
| * E | 1.79 | 1.85 | 1.78 | 1.79 | 1.69 | 1.61 |
| 8 MR ‖ [001] | 8 MR ‖ [001] | |||||
| O1–O1 | 7.52 | 7.39 | 7.54 | 7.52 | 7.41 | 7.51 |
| O6–O6 | 4.66 | 4.45 | 4.63 | 4.68 | 4.42 | 4.60 |
| D6R | D6R | |||||
| O5–O3–O5 | 147.67 | 151.82 | 145.63 | 149.18 | 152.6 | 143.00 |
| O3–O5–O3 | 91.68 | 86.51 | 93.23 | 90.09 | 87.7 | 95.97 |
| 12 MR maximum diameter | ||||||
| O6–O6 | 15.60 | 15.44 | 15.69 | 15.67 | 15.67 | 15.91 |
| Distance | Pamb | 1.95 GPa |
|---|---|---|
| O1–O1 | 9.934 | 9.878 |
| O2–O2 | 10.431 | 10.409 |
| O6–O6 | 15.520 | 15.614 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gigli, L.; Arletti, R.; Fois, E.; Tabacchi, G.; Quartieri, S.; Dmitriev, V.; Vezzalini, G. Unravelling the High-Pressure Behaviour of Dye-Zeolite L Hybrid Materials. Crystals 2018, 8, 79. https://doi.org/10.3390/cryst8020079
Gigli L, Arletti R, Fois E, Tabacchi G, Quartieri S, Dmitriev V, Vezzalini G. Unravelling the High-Pressure Behaviour of Dye-Zeolite L Hybrid Materials. Crystals. 2018; 8(2):79. https://doi.org/10.3390/cryst8020079
Chicago/Turabian StyleGigli, Lara, Rossella Arletti, Ettore Fois, Gloria Tabacchi, Simona Quartieri, Vladimir Dmitriev, and Giovanna Vezzalini. 2018. "Unravelling the High-Pressure Behaviour of Dye-Zeolite L Hybrid Materials" Crystals 8, no. 2: 79. https://doi.org/10.3390/cryst8020079





