An Unusual Bismuth Ethanedisulfonate Network
Abstract
:1. Introduction
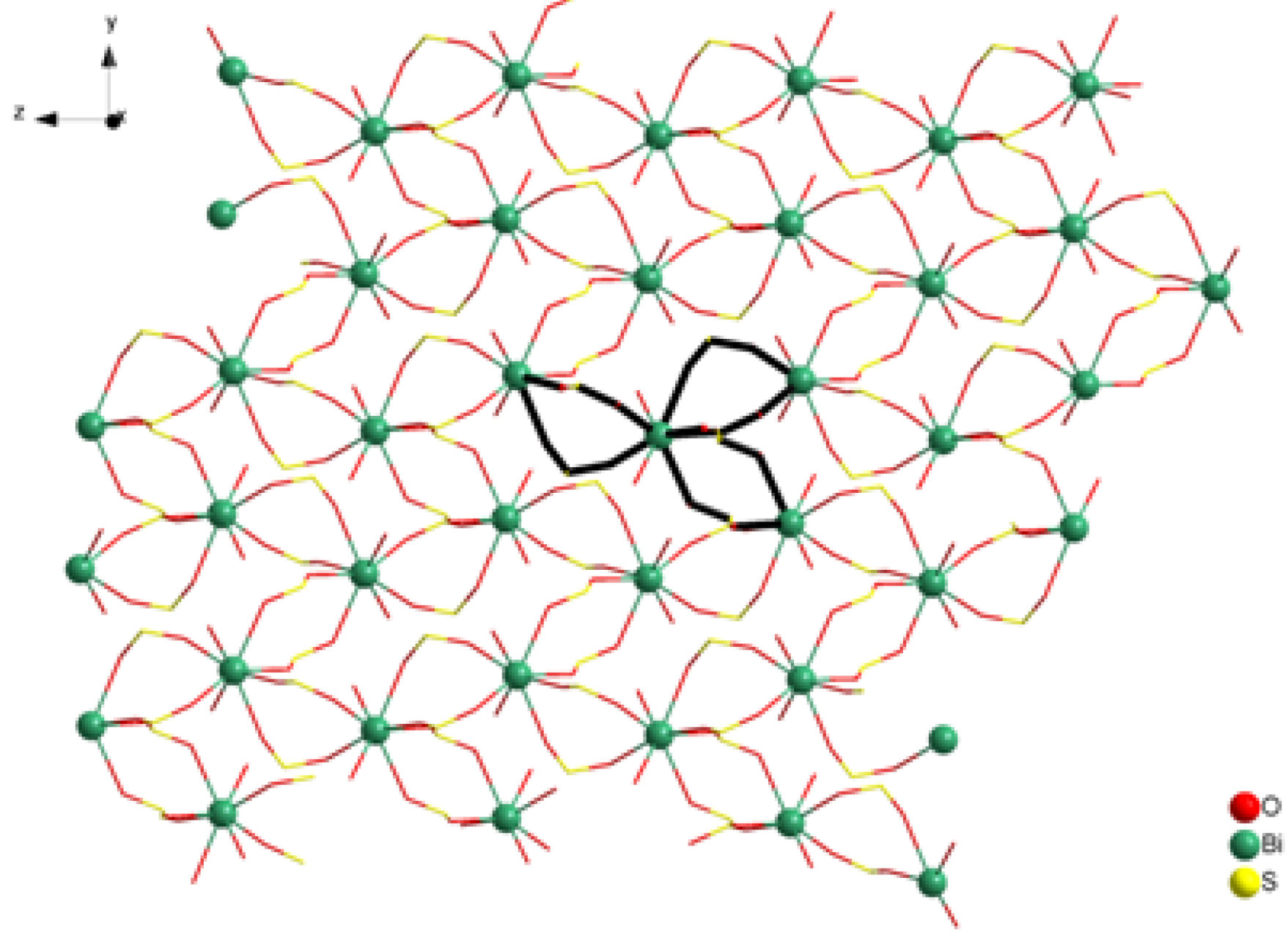
2. Results and Discussion

| Bond | Distance |
|---|---|
| Bi1―O1 | 2.427(9) |
| Bi1―O3 | 2.284(8) |
| Bi1―O4 | 2.590(9) |
| Bi1―O5 | 2.324(8) |
| Bi1―O7 | 2.591(8) |
| Bi1―O9 | 2.510(7) |
| Bi1―O10 | 2.420(8) |
| Bi1―O11 | 2.4249(5) |

| Crystal Data | 1 |
|---|---|
| Formula | C3H10BiO11S3 |
| Mr | 527.27 |
| Cryst. size, mm3 | 0.18 × 0.12 × 0.07 |
| Crystal system | monoclinic |
| Space group | P21/c |
| a, Å | 11.601(2) |
| b, Å | 10.390(2) |
| c, Å | 10.498(2) |
| β, deg | 115.15(3) |
| V, Å3 | 1145.3(4) |
| Z | 4 |
| Dcalcd, g cm–3 | 3.058 |
| μ(MoKα), cm–1 | 15.994 |
| F(000), e | 988 |
| Refl. measd./unique | 12498/10592 |
| Param. refined | 3144 |
| R(F)/wR(F2)a (all refl.) | 0.0475/0.0283 |
| GoF (F2)a | 1.124 |
3. Experimental Section
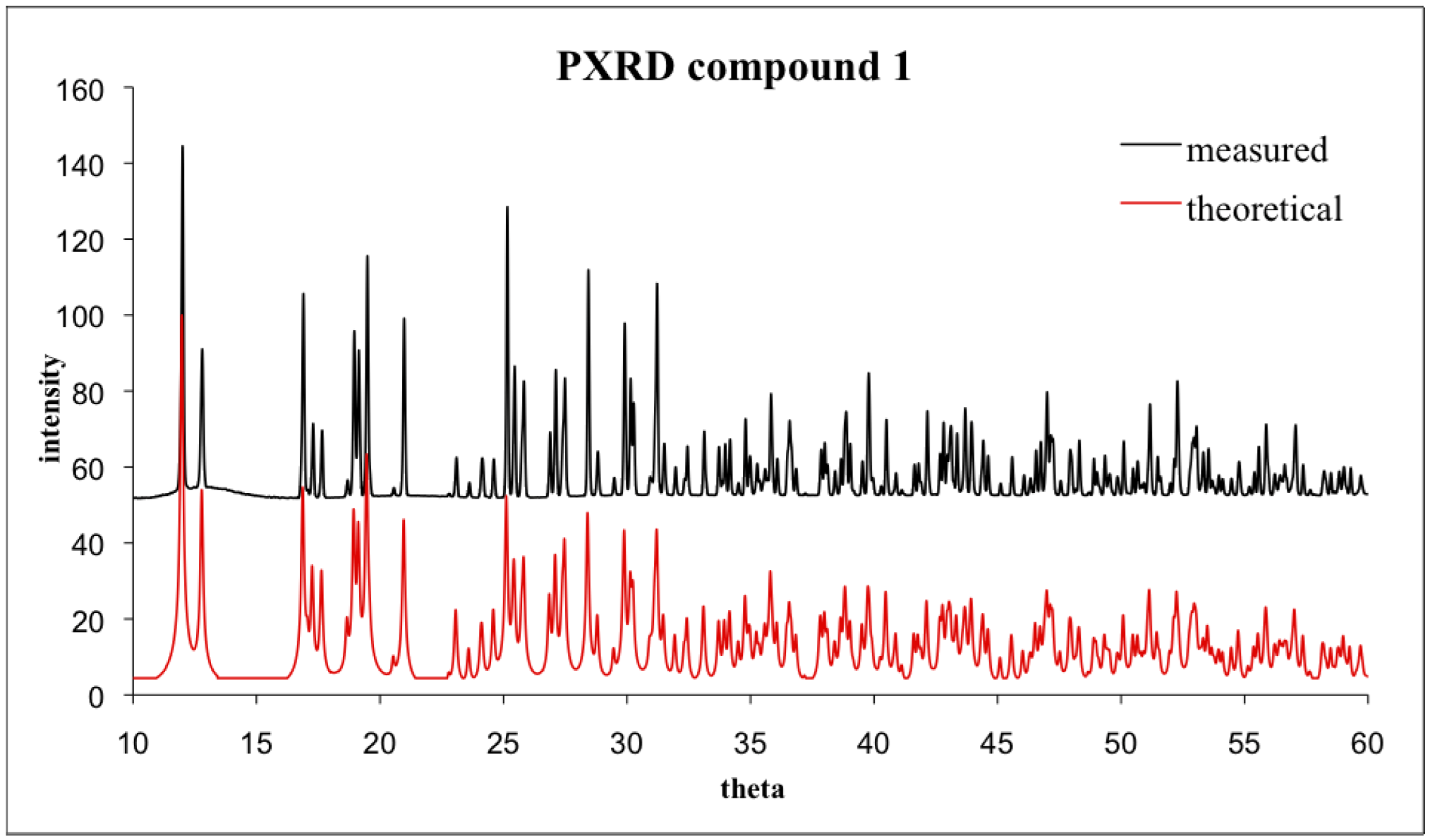
4. Conclusions
Acknowledgment
Conflict of Interest
References
- Briand, G.G.; Burford, N. Bismuth compounds and preparations with biological or medicinal relevance. Chem. Rev. 1999, 99, 2601–2657. [Google Scholar] [CrossRef]
- Csajbok, E.; Baranyai, Z.; Banyai, I.; Brücher, E.; Kiraly, R.; Müller-Fahrnow, A.; Platzek, J.; Radüchel, B.; Schäfer, M. Equilibrium, 1H and 13C NMR Spectroscopy, and X-ray Diffraction Studies on the Complexes Bi(DOTA) and Bi(DO3A-Bu). Inorg. Chem. 2003, 42, 2342–2349. [Google Scholar] [CrossRef]
- Stavila, V.; Davidoch, R.L.; Gulea, A.; Whitmire, K.H. Bismuth (III) complexes with aminopolycarboxylate and polyaminopolycarboxylate ligands: Chemistry and structure. Coord. Chem. Rev. 2006, 250, 2782–2810. [Google Scholar] [CrossRef]
- Garje, S.S.; Jain, V.K. Chemistry of arsenic, antimony and bismuth compounds derived from xanthate, dithiocarbamate and phosphorus based ligands. Coord. Chem. Rev. 2003, 236, 35–56. [Google Scholar]
- Hanna, T.A. The role of bismuth in the SOHIO process. Coord. Chem. Rev. 2004, 248, 429–440. [Google Scholar] [CrossRef]
- Kumar, R.; Mishra, P. Bi (V) organic framework in an asymmetric system: Synthesis, spectroscopic, XRPD and molecular modeling. Main Group Chem. 2007, 6, 85–95. [Google Scholar] [CrossRef]
- Thirumurugan, A.; Tan, J.C.; Cheetham, A.K. Heterometallic Inorganic-Organic Framework of Sodium-Bismuth Benzenedicarboxylates. Cryst. Growth Des. 2010, 10, 1736–1741. [Google Scholar] [CrossRef]
- Tran, D.T.; Chu, D.; Oliver, A.G.; Oliver, S.R.J. 3-D bismuth-organic framework containing 1-D cationic inorganic [Bi2O2]2+ chains. Inorg. Chem. Commun. 2009, 10, 1081–1084. [Google Scholar]
- Cotton, F.A.; Wilkinson, G.; Gaus, P.L. The GroupVB(15) Elements: Phosphorus, Arsenic, Antimony, and Bismuth. In Basic Inorganic Chemistry, 3rd; Rose, N., Herbert, D., Eds.; Wiley and Sons, Inc.: New York, NY, USA, 1995; pp. 399–435. [Google Scholar]
- Rogow, D.L.; Fei, H.; Brennan, D.P.; Ikehata, M.; Oliver, S.R.J. Hydrothermal synthesis of two cationic bismuthate clusters: An alkylenedisulfonate bridged hexamer, [Bi6O4(OH)4(H2O)2][(CH2)2(SO3)2]3 and a rare nonamer templated by triflate, [Bi9O8(OH)6][CF3SO3]5. Inorg. Chem. 2010, 49, 5619–5624. [Google Scholar] [CrossRef]
- Thurston, J.H.; Trahan, D.; Ould-Ely, T.; Whitmire, K.H. Toward an general strategy for synthesis of heterobimetallic coordination complexes for use as precursors to metal oxide materials: Synthesis, Characterization, and Thermal Decomposition of Bi2(Hsal)6*M(Acac)3 (M = Al, Co, V, Fe, Cr). Inorg. Chem. 2004, 43, 3299–3305. [Google Scholar] [CrossRef]
- Andrews, P.C.; Deacon, G.B.; Junk, P.C.; Kumar, I.; Silberstein, M. Synthetic and structural comparisons of bismuth(III) carboxylates synthesized under solvent-free and reflux conditions. Dalton Trans. 2006, 40, 4852–4858. [Google Scholar]
- Holmes, R.R.; Schmid, C.G.; Chandrasekhar, V.; Day, R.O.; Holmes, J.M. Oxo carboxylate tin ladder Clusters. A New Structural Class of Organotin Compounds. J. Am. Chem. Soc. 1987, 109, 1408–1414. [Google Scholar]
- Asato, E.; Driessen, W.L.; de Graaff, R.A.G.; Hulsbergen, F.B.; Reedijk, J. Synthesis, Structure, and Spectroscopoc Properties of Bismuth Citrate Compounds. Inorg. Chem. 1991, 30, 4210–4218. [Google Scholar] [CrossRef]
- Adair, B.A.; Neeray, S.; Cheetham, A.K. Role of Chains in the Formation of Extended Framework Tin(II) Phosphates and Related Materials. Chem. Mater. 2003, 15, 1518–1529. [Google Scholar] [CrossRef]
- Bujoli-Doeuff, M.; Evain, M.; Janvier, P.; Massiot, D.; Clearfield, A.; Gan, Z.; Bujoli, B. New pillared layered gallium phosphonates in the gallium/1,2-ethylenediphosphonic acid system. Inorg. Chem. 2001, 40, 6694–6698. [Google Scholar]
- Bruno, I.J.; Cole, J.C.; Edgington, P.R.; Kessler, M.; Macrae, C.F.; McCabe, P.; Pearson, J.; Taylor, R. ConQuest, Version 1.14; Cambridge Crystallographic Data Centre: Cambridge, UK, 2012. [Google Scholar]
- Müller, U.; Schubert, M; Teich, F.; Pütter, H.; Schierle-Arndt, K.; Pastré., J. Metal-organic frameworks-prospective industrial applications. J. Mater. Chem. 2006, 16, 626–636. [Google Scholar]
- Kitagawa, S.; Kitaura, R.; Noro, S.I. Functional Porous Coordination Polymers. Angew. Chem. Int. Ed. 2004, 43, 2334–2375. [Google Scholar] [CrossRef]
- Li, H.; Eddaoudi, M.; O’Keeffe, M.; Yaghi, O.M. Design and synthesis of an exceptionally stable and highly porous metal-organic framework. Nature 1999, 402, 276–279. [Google Scholar]
- Bauer, S.; Müller, H.; Bein, T.; Stock, N. Synthesis and characterization of the open-framework barium bisphosphonate [Ba3(O3PCH2NH2CH2PO3)2(H2O)4]*3H2O. Inorg. Chem. 2005, 44, 9464–9470. [Google Scholar] [CrossRef]
- Serre, C.; Groves, J.A.; Lightfoot, P.; Slawin, A.M.Z.; Wright, P.A.; Stock, N.; Bein, T.; Haouas, M.; Taulelle, F.; Férey, G. Synthesis, Structure and Properties of Related Microporous N,N-Piperazinebismethylenephosphonates of Aluminium and Titanium. Chem. Mater. 2006, 18, 1451–1457. [Google Scholar] [CrossRef]
- Zhang, L.P.; Zhu, L.G. Influence of neutral amine ligands on the network assembly of lead(II) 4-sulfobenzoate complexes. J. Mol. Struct. 2008, 873, 61–68. [Google Scholar] [CrossRef]
- Schreuer, J. Crystal structure of thalium sulfanilate semihydrate, TlH2C6H4SO3·0.5H2O. Z. Kristallogr. New Cryst. Struct. 1999, 214, 313–314. [Google Scholar]
- Miao, X.H.; Zhu, L.G. Influence of secondary bonds on the network assembly and property in lead(II) 3-sulfobenzoate. Z. Anorg. Allg. Chem. 2008, 634, 335–338. [Google Scholar] [CrossRef]
- Chandrasekhar, V.; Singh, P. In Situ Generated Hydrated Diorganotin Cations as Synthons for Hydrogen-Bonded and Coordination-Driven 1D-, 2D, and 3D-Assemblies. Cryst. Growth Des. 2010, 10, 3077–3093. [Google Scholar]
- Charbonnier, F.; Faure, R.; Loiseleur, H. Structure Cristalline de l’Ethanedisulfonate-1,2 de Cuivre Tétrahydraté: Cu[SO3(CH2)2SO3]·4H2O. Acta Cryst. 1977, B33, 3342–3345. [Google Scholar]
- Charbonnier, F.; Faure, R.; Loiseleur, H. Coordination et Disposition de l’Argent dans l’Ethanedisulfonate-1,2 et le Butanedisulfonate-1,4 d’Argent(I) Cristallisés. Acta Cryst. 1981, B37, 822–826. [Google Scholar]
- Forster, P.M.; Tafoya, M.M.; Cheetham, A.K. Synthesis and characterization of Co7(OH)12(C2H4S2O6)(H2O)2—A single crystal structural study of a ferromagnetic layered cobalt hydroxide. J. Phys. Chem. Solids 2004, 65, 11–16. [Google Scholar] [CrossRef]
- Mootz, D.; Wunderlich, H. Kristallstrukturen von säurehydraten und oxoniumsalzen. IV. dioxonium-äthan-1,2-disulfonat, (H3O)2[O3SCH2CH2SO3]. Acta Cryst. 1970, B26, 1820–1825. [Google Scholar]
- Sartori, P.; Juschke, R.; Boese, R.; Blaser, D. Structure of dihydroxononium alkanedisulfonates. Z. Naturforsch. B 1994, 49, 1467–1471. [Google Scholar]
- Fei, H.; Oliver, S.R.J. Two cationic metal–organic frameworks based on cadmium and a,x-alkanedisulfonate anions and their photoluminescent properties. Dalton Trans. 2010, 39, 11193–11200. [Google Scholar] [CrossRef]
- Brese, N.E.; O'Keeffe, M. Bond-valence parameters for solids. Acta Cryst. 1991, B47, 192–197. [Google Scholar]
- Brown, I.D.; Altermatt, D. Bond-Valence Parameters Obtained from a Systematic Analysis of the inorganic crystal structure database. Acta Cryst. 1985, B41, 244–247. [Google Scholar]
- Spek, A.L. Structure validation in chemical crystallography. Acta Cryst. 2009, D65, 148–155. [Google Scholar]
- Sheldrick, G.M. SHELXS-97, Program for Structure Analysis, version 97-2; University of Göttingen: Göttingen, Germany, 1998. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gschwind, F.; Jansen, M. An Unusual Bismuth Ethanedisulfonate Network. Crystals 2012, 2, 1374-1381. https://doi.org/10.3390/cryst2041374
Gschwind F, Jansen M. An Unusual Bismuth Ethanedisulfonate Network. Crystals. 2012; 2(4):1374-1381. https://doi.org/10.3390/cryst2041374
Chicago/Turabian StyleGschwind, Fabienne, and Martin Jansen. 2012. "An Unusual Bismuth Ethanedisulfonate Network" Crystals 2, no. 4: 1374-1381. https://doi.org/10.3390/cryst2041374
APA StyleGschwind, F., & Jansen, M. (2012). An Unusual Bismuth Ethanedisulfonate Network. Crystals, 2(4), 1374-1381. https://doi.org/10.3390/cryst2041374




