[KDy(Hptc)3(H3ptc)]n·2n(Hbipy)·5n(H2O), a Layered Coordination Polymer Containing DyO6N3 Tri-Capped Trigonal Prisms (H3ptc = Pyridine 2,4,6-Tricarboxylic Acid, C8H5NO6; Bipy = 2,2'-Bipyridine, C10H8N2)
1. Introduction
2. Results and Discussion
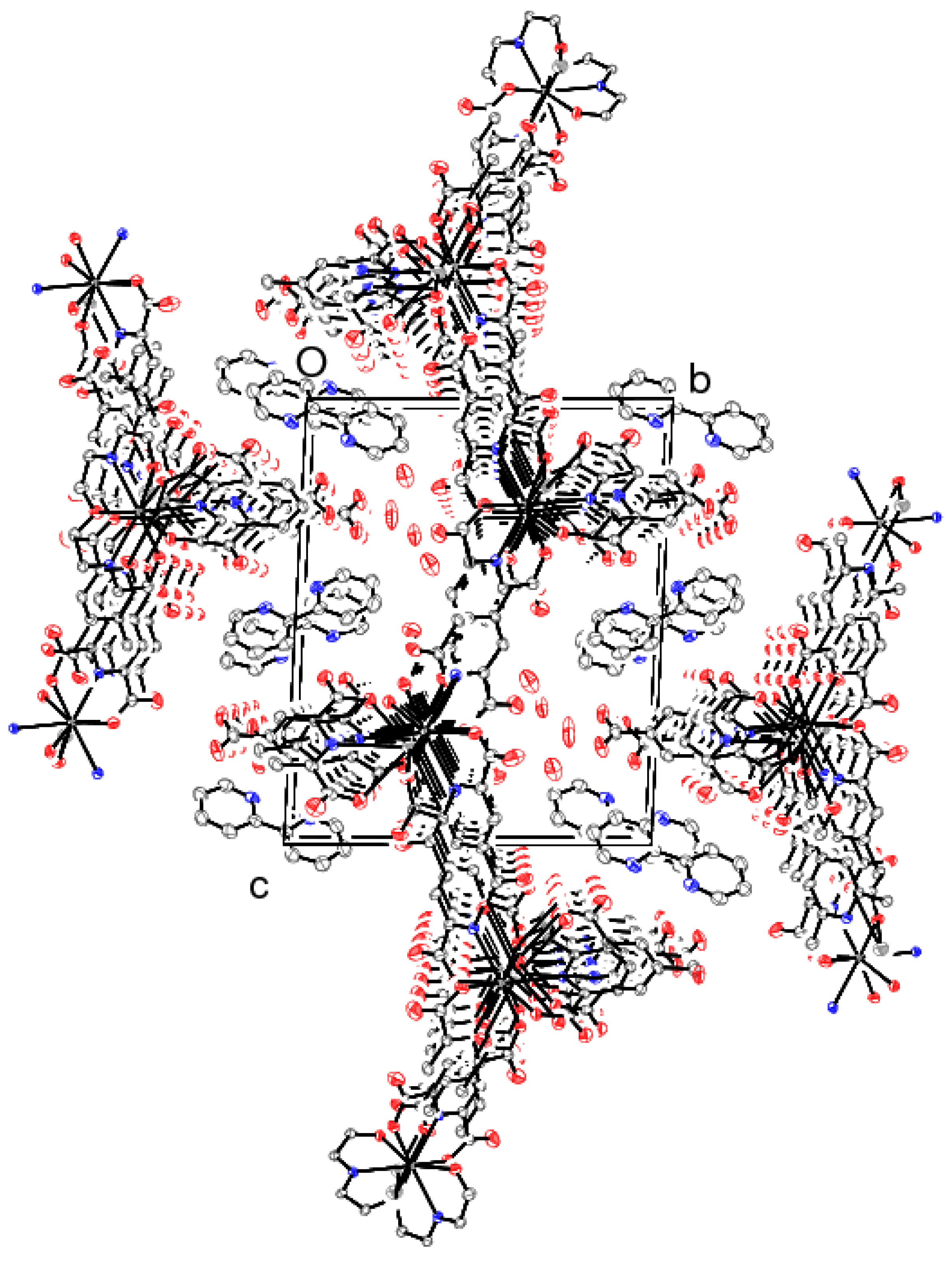
2.1. Crystal Structure of [KDy(C8H3NO6)3(C8H5NO6)]n·2n(C10H9N2)·5n(H2O) (1)
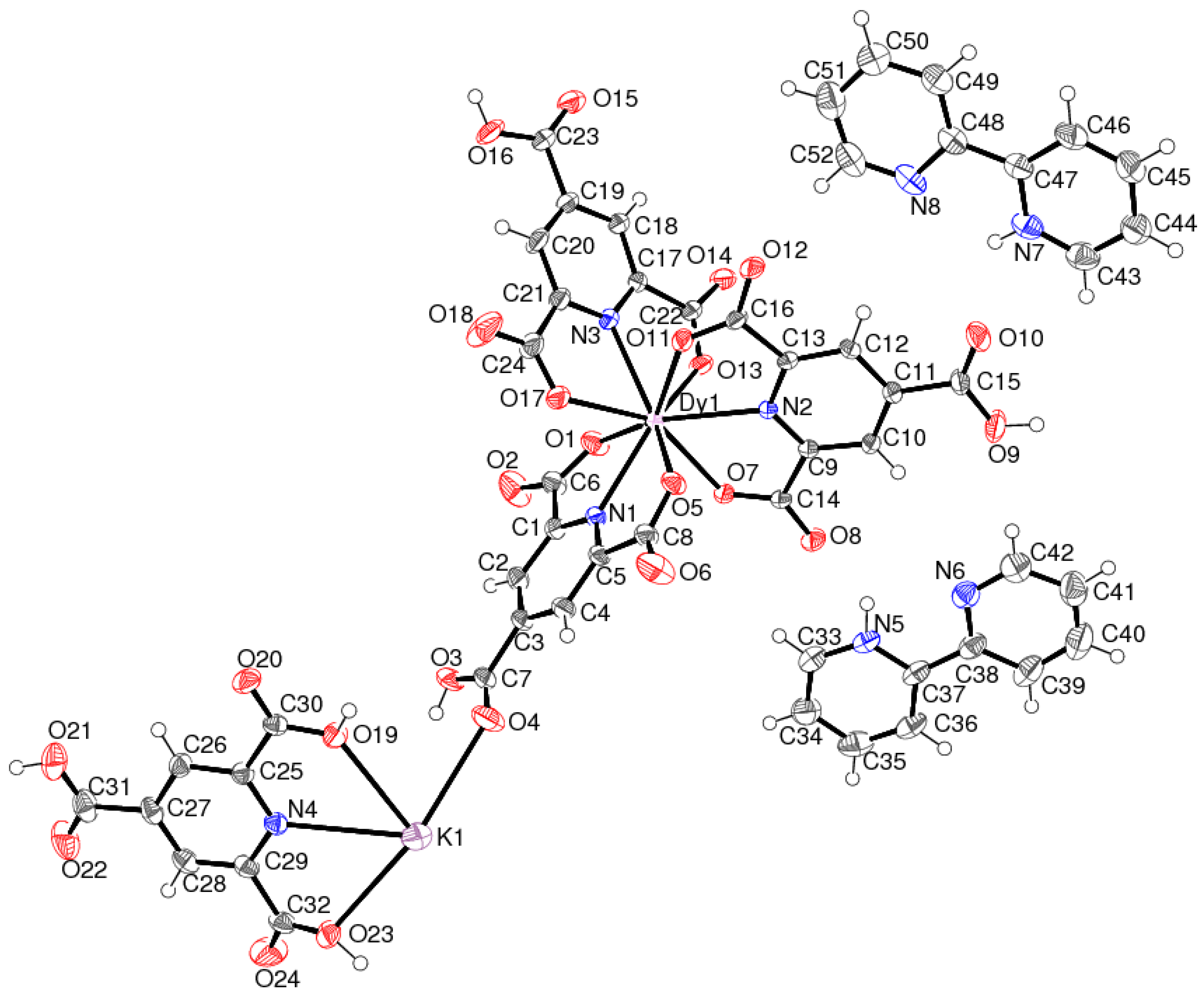
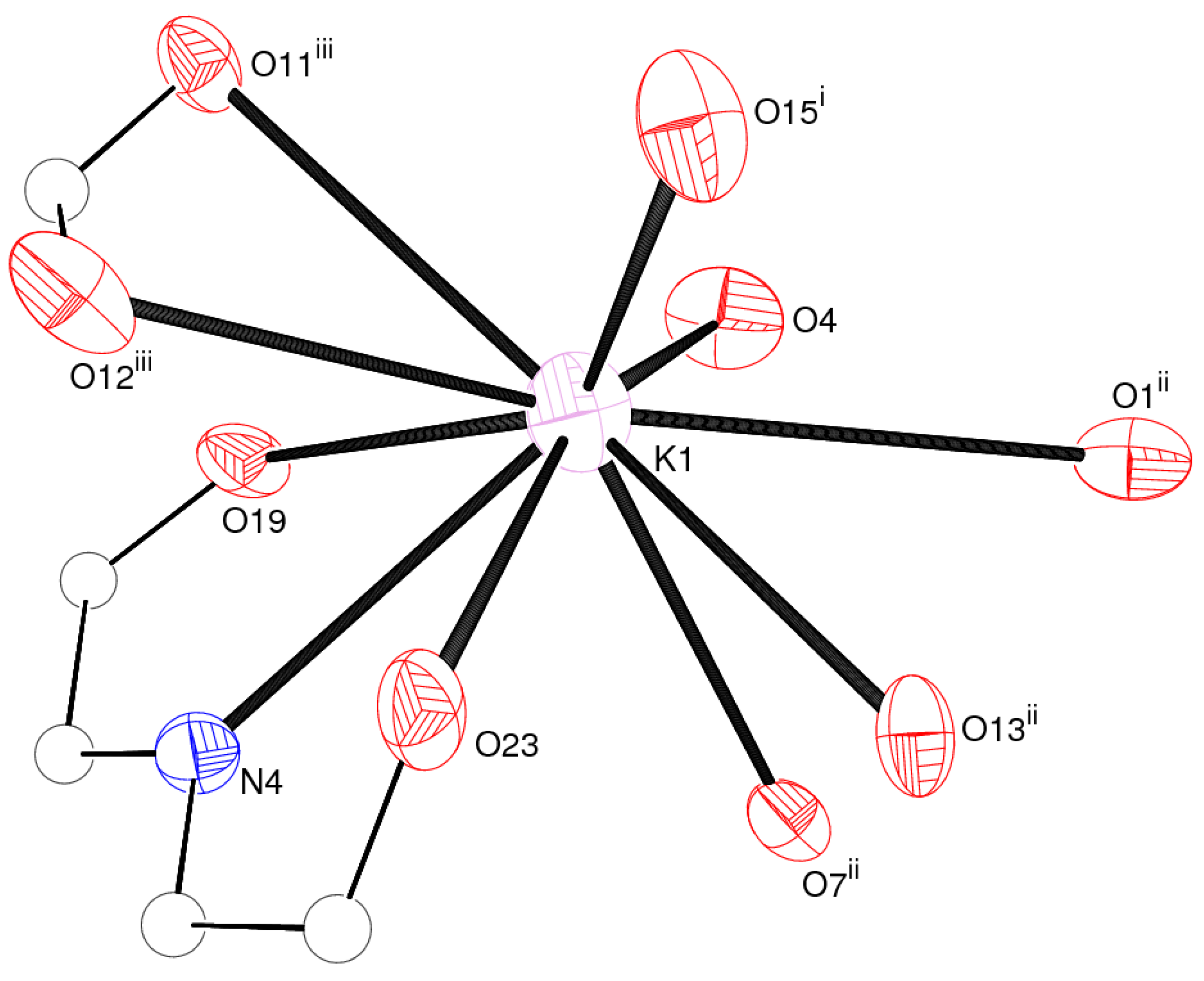
| K1–O19 | 2.885(2) | K1–O23 | 2.946(2) |
| K1–O4 | 2.949(2) | K1–O15 i | 2.958(2) |
| K1–O13 ii | 3.025(2) | K1–O11 iii | 3.073(2) |
| K1–N4 | 3.074(2) | K1–O7 ii | 3.201(2) |
| K1–O12 iii | 3.388(3) | K1–O1 ii | 3.432(3) |
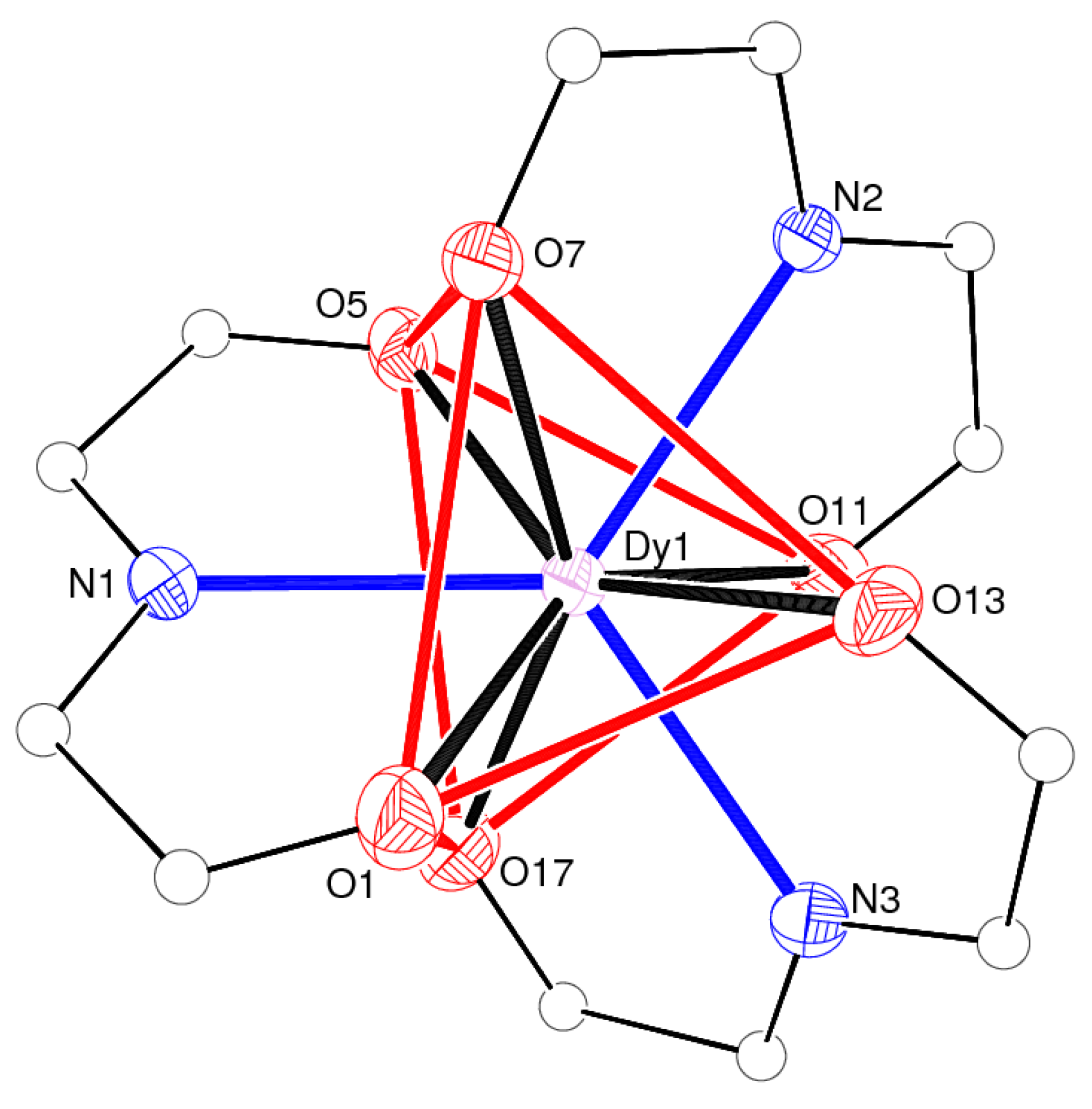
| Dy1–O1 | 2.366(2) | Dy1–O17 | 2.392(2) |
| Dy1–O13 | 2.397(2) | Dy1–O11 | 2.397(2) |
| Dy1–O5 | 2.440(2) | Dy1–N1 | 2.481(2) |
| Dy1–O7 | 2.4838(19) | Dy1–N3 | 2.500(2) |
| Dy1–N2 | 2.523(2) |

| O3–H3o…O7 ii | 0.81 | 1.79 | 2.537(3) | 153 |
| O9–H9o…O4w iv | 0.92 | 1.60 | 2.511(4) | 169 |
| O16–H16o…O12 v | 0.85 | 1.63 | 2.458(3) | 163 |
| O19–H19o…O5 iii | 0.96 | 1.57 | 2.502(3) | 162 |
| O21–H21o…O1w vi | 0.93 | 1.65 | 2.515(3) | 152 |
| O23–H23o…O14 ii | 0.96 | 1.57 | 2.514(3) | 168 |
| N5–H5n…O8 | 0.95 | 1.96 | 2.772(3) | 142 |
| N7–H7n…O10 | 0.87 | 2.23 | 2.882(3) | 132 |
| O1w–H1w…O2 vii | 0.92 | 1.80 | 2.702(4) | 167 |
| O1w–H2w…O18 | 0.92 | 1.77 | 2.672(4) | 165 |
| O2w–H3w…O1w | 0.92 | 2.37 | 3.044(5) | 130 |
| O2w–H4w…O17 | 0.92 | 2.05 | 2.865(4) | 146 |
| O3w–H5w…O5w | 0.92 | 1.92 | 2.816(5) | 163 |
| O3w–H6w…O22 viii | 0.92 | 1.97 | 2.878(4) | 171 |
| O4w–H7w…O3w | 0.92 | 1.87 | 2.709(6) | 151 |
| O4w–H8w…O2w | 0.85 | 2.06 | 2.909(6) | 180 |
| O5w–H9w…O1 | 0.92 | 1.95 | 2.844(4) | 164 |
| O5w–H10w…O15 ix | 0.92 | 2.01 | 2.891(4) | 161 |
2.2. Bacteriological Tests
| Minimum Inhibitory Concenteration (MIC) | |||||
|---|---|---|---|---|---|
| M. leuteus | S. aureus | Ent. bac | B. step | E. coli | S.typhi |
| 30 ppm | 150 ppm | 200 ppm | 70 ppm | 110 ppm | Nil |
3. Experimental Section
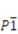 (No. 2), Z = 2, a = 9.188(2) Å, b = 15.7332(17) Å, c = 19.1664(19) Å, α = 92.797(6)°, β = 92.319(7)°, γ = 91.273(9)°, V = 2764.3(7) Å3, F(000) = 1454, T = 296(2) K, ρcalc = 1.736 g·cm−3, μ = 1.532 mm−1, 37880 reflections measured (−7 ≤ h ≤ 12, −20 ≤ k ≤ 20, −25 ≤ l ≤ 25; 4.88° ≤ 2θ ≤ 56.74°), RInt = 0.025, 13445 merged reflections, 12100 with I > 2σ(I), 825 variable parameters, R(F) = 0.029, wR(F2) = 0.084, w = 1/[σ2(Fo2) + (0.0387P)2 + 3.7247P], where P = (Fo2 + 2Fc2)/3, min./max. Dρ = −0.74, +1.12 e Å−3. Cambridge Structural Database deposition number: CCDC-879106.
(No. 2), Z = 2, a = 9.188(2) Å, b = 15.7332(17) Å, c = 19.1664(19) Å, α = 92.797(6)°, β = 92.319(7)°, γ = 91.273(9)°, V = 2764.3(7) Å3, F(000) = 1454, T = 296(2) K, ρcalc = 1.736 g·cm−3, μ = 1.532 mm−1, 37880 reflections measured (−7 ≤ h ≤ 12, −20 ≤ k ≤ 20, −25 ≤ l ≤ 25; 4.88° ≤ 2θ ≤ 56.74°), RInt = 0.025, 13445 merged reflections, 12100 with I > 2σ(I), 825 variable parameters, R(F) = 0.029, wR(F2) = 0.084, w = 1/[σ2(Fo2) + (0.0387P)2 + 3.7247P], where P = (Fo2 + 2Fc2)/3, min./max. Dρ = −0.74, +1.12 e Å−3. Cambridge Structural Database deposition number: CCDC-879106.4. Conclusions
References
- Batten, S.R.; Champness, N.R.; Chen, X.-M.; Garcia-Martinez, J.; Kitagawa, S.; Öhrström, L.; O’Keeffe, M.; Suh, M.P.; Reedijk, J. Coordination polymers, metal-organic frameworks and the need for terminology guidelines. CrystEngComm 2012, 14, 3001–3004. [Google Scholar] [CrossRef]
- Wang, H.-S.; Zhao, B.; Zhai, B.; Shi, W.; Cheng, P.; Liao, D.-Z.; Yan, S.-P. Syntheses, structures and photoluminescence of one-dimensional lanthanide coordination polymers with 2,4,6-pyridine-tricarboxylic acid. Cryst. Growth Des. 2007, 7, 1851–1857. [Google Scholar] [CrossRef]
- Li, C.-J.; Peng, M.-X.; Leng, J.-D.; Yang, M.-M.; Lin, J.; Tong, M.-L. Synthesis, structure, photoluminescence and magnetic properties of new 3-D lanthanide-pyridine-2,4,6-tricarboxate framework. CrysEngComm 2008, 10, 1645–1652. [Google Scholar] [CrossRef]
- Das, M.C.; Ghosh, S.K.; Sanudo, E.C.; Bharadwaj, P.K. Coordination polymers with pyridine-2,4,6-tricarboxylic acid and alkaline-earth/lanthanide/transition metals: Synthesis and X-ray structures. Dalton Trans. 2009, 1644–1658. [Google Scholar]
- Lin, J.-L.; Xu, W.; Zhao, L.; Zheng, Y.-Q. Synthesis, crystal structure and properties of a new lanthanide pyridine-2,4,6-tricarboxylato coordination polymer. Z. Naturforsch.B 2011, 66, 570–576. [Google Scholar] [CrossRef]
- Holder, A.A.; VanDerveer, D. Potassium (4-carboxypyridine-2,6-dicarboxylato)-dioxido-vanadate(V) monohydrate. Acta Cryst. 2007, E63, m2051–m2052. [Google Scholar]
- Zhang, W.-Z.; Lv, T.-Y.; Wei, D.-Z.; Xu, R.; Xiong, G.; Wang, Y.-Q.; Gao, E.-J.; Sun, Y.-G. Synthesis, crystal structures and luminescence properties of two novel 3D heterometallic coordination polymers. Inorg. Chem. Commun. 2011, 14, 1245–1249. [Google Scholar] [CrossRef]
- Brown, I.D.; Altermatt, D. Bond-Valence parameters obtained from a systematic analysis of the Inorganic Crystal Structure Database. Acta Cryst. 1985, B41, 244–247. [Google Scholar]
- Nichol, G.S.; Clegg, W. Classical hydrogen bonding and weaker C-H…O interactions in complexes of uracil-5-carboxylic acid with the alkali metals Na-Cs. Polyhedron 2006, 25, 1043–1056. [Google Scholar] [CrossRef]
- Mak, T.C.W.; Wai-Hing, Y.; Smith, G.; O’Reilly, E.J.; Kennard, C.H.L. Metal (phenylthio)acetic acid interactions 3. The crystal structures of anhydrous barium (phenylthio)-acetate and the potassium (phenylthio)acetate (phenylthio)acetic acid adduct. Inorg. Chim. Acta 1984, 88, 35–39. [Google Scholar]
- Syper, L.; Kloc, K.; Młochowski, K. Synthesis of ubiquinone and menaquinone analogues by oxidative demethylation of alkenylhydroquinine ethers with argentic oxide or ceric ammonium nitrate in the presence of 2,4,6-pyridine tricarboxylic aicd. Tetrahedron 1980, 36, 123–129. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Cryst. 2008, A64, 112–122. [Google Scholar]
- Spek, A.L. Structure validation in chemical crystallography. Acta Cryst. 2009, D65, 148–155. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Anwar, S.; Mariam, I.; Khan, I.U.; Ejaz; Harrison, W.T.A.; Anwar, S. [KDy(Hptc)3(H3ptc)]n·2n(Hbipy)·5n(H2O), a Layered Coordination Polymer Containing DyO6N3 Tri-Capped Trigonal Prisms (H3ptc = Pyridine 2,4,6-Tricarboxylic Acid, C8H5NO6; Bipy = 2,2'-Bipyridine, C10H8N2). Crystals 2012, 2, 1084-1091. https://doi.org/10.3390/cryst2031084
Anwar S, Mariam I, Khan IU, Ejaz, Harrison WTA, Anwar S. [KDy(Hptc)3(H3ptc)]n·2n(Hbipy)·5n(H2O), a Layered Coordination Polymer Containing DyO6N3 Tri-Capped Trigonal Prisms (H3ptc = Pyridine 2,4,6-Tricarboxylic Acid, C8H5NO6; Bipy = 2,2'-Bipyridine, C10H8N2). Crystals. 2012; 2(3):1084-1091. https://doi.org/10.3390/cryst2031084
Chicago/Turabian StyleAnwar, Shoaib, Irfana Mariam, Islam Ullah Khan, Ejaz, William T. A. Harrison, and Sohail Anwar. 2012. "[KDy(Hptc)3(H3ptc)]n·2n(Hbipy)·5n(H2O), a Layered Coordination Polymer Containing DyO6N3 Tri-Capped Trigonal Prisms (H3ptc = Pyridine 2,4,6-Tricarboxylic Acid, C8H5NO6; Bipy = 2,2'-Bipyridine, C10H8N2)" Crystals 2, no. 3: 1084-1091. https://doi.org/10.3390/cryst2031084





