Ethyl 2,6-Dimethoxybenzoate: Synthesis, Spectroscopic and X-ray Crystallographic Analysis
Abstract
:1. Introduction
2. Results and Discussion
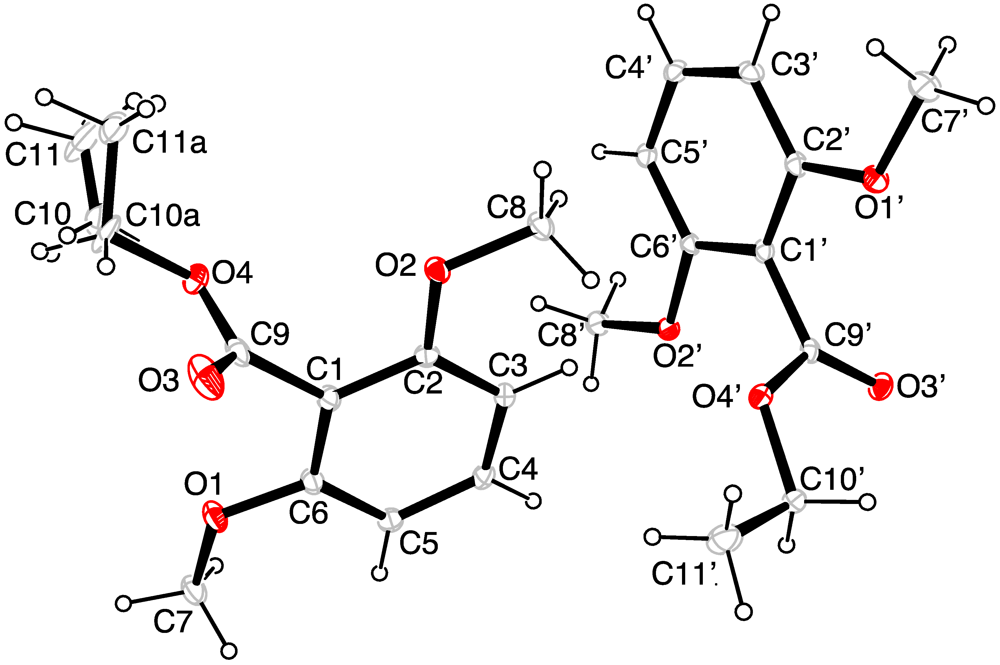
| Atoms | Bond length | Atoms | Bond angle |
|---|---|---|---|
| O(2')-C(6') | 1.367(2) | C(6')-O(2')-C(8') | 116.91(14) |
| O(2')-C(8') | 1.429(2) | C(2')-O(1')-C(7') | 116.82(16) |
| O(3')-C(9') | 1.202(2) | C(9')-O(4')-C(10') | 117.21(14) |
| O(1')-C(2') | 1.362(2) | C(6)-O(2)-C(8) | 117.52(15) |
| O(1')-C(7') | 1.432(2) | O(2')-C(6')-C(1') | 115.06(16) |
| O(4')-C(9') | 1.336(2) | O(1')-C(2')-C(3') | 125.07(17) |
| O(4')-C(10') | 1.456(2) | O(2')-C(6')-C(5') | 124.77(17) |
| O(1)-C(6) | 1.368(2) | O(4')-C(9')-C(1') | 109.96(15) |
| O(4)-C(10A) | 1.504(4) | O(4')-C(10')-C(11') | 106.46(16) |
| C(10')-C(11') | 1.497(3) | O(3)-C(9)-O(4) | 124.5(2) |
| C(10A)-C(11A) | 1.214(5) | C(11A)-C(10A)-O(4) | 120.2(5) |
| C-H…π interactions | C-H…O interactions |
|---|---|
| H7B…C4´ 2.856 (x, y, 1 + z) | H8b…O2 (1-x, -y, 2 − z) 2.620 Å |
| H7B…C5´ 2.862 | H4´…O3´ (1 + x, y, z) 2.680 Å |
| H3…C6´ 2.888 Å | H7´1…O3 (x, y, z − 1) 2.547 Å |
| H4´…C5 (1 − x, 1 − y, 2 − z) 2.881Å | H7´2…O1 (−x, −y, 1 − z) 2.654 Å |
| H5…O3´ (-x, 1 − y, 2 − z) 2.498 Å |
3. Experimental Section
3.1. General
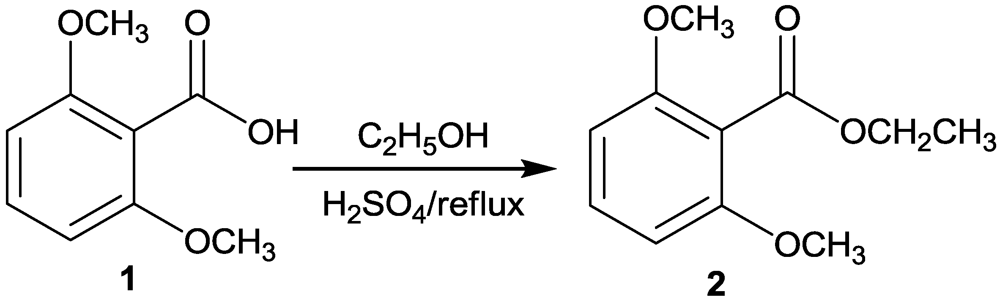
3.2. General Procedure for the Synthesis of Ethyl 2, 6-Dimethoxybenzoate (2)
3.3. Data Collection and Structure Solution
4. Conclusions
Conflict of Interest
Acknowledgments
References
- Manjinder, S.L.; Yeeman, K.R.; Michael, N.G.J.; John, C.V. Serine and Threonine β-Lactones:A New Class of Hepatitis A Virus 3C Cysteine Proteinase Inhibitors. J. Org. Chem. 2002, 67, 1536–1547. [Google Scholar]
- Tandon, V.K.; Yadav, D.B.; Singh, R.V.; Chaturvedi, A.K.; Shukla, P.K. Synthesis and activity of oleanolic acid derivatives, a novel class of inhibitors of osteoclast formation. Bioorg. Med. Chem. Lett. 2005, 15, 5324–5328. [Google Scholar] [CrossRef]
- Kumar, A.P.; Garcia, G.E.; Ghosh, R.; Rajnarayanany, R.V.; Alworth, W.L.; Slaga, T.J. 4-Hydroxy-3-Methoxybenzoic Acid Methyl Ester: A Curcumin Derivative Targets Akt/NFkB Cell Survival Signaling Pathway: Potential for Prostate Cancer Management. Neoplasia 2003, 5, 255–266. [Google Scholar]
- Tomasz, Z.; Michał, A.; Janusz, J. A simple synthesis of chiral macrocyclic tetraamides derived from α-amino acids. Tetrahedron: Asymmetry 2002, 13, 2053–2059. [Google Scholar] [CrossRef]
- Kim, J.; Kim, J.; Song, S.; Jung, O.; Suh, H. Enantiomeric Recognition of D- and L-Amino acidMethyl Ester Hydrochlorides by New Chiral Bis-pyridino-18-crown-6 Substituted with Urea, and Diphenyl Groups. J. Incl. Phenom.Macrocycl.Chem. 2007, 58, 187–192. [Google Scholar] [CrossRef]
- Somlai, C.; Peter, A.; Forgo, P.; Penke, B. One-Pot Synthesis of N-Protected β-Chiral Amino Alcohols. Syn. Commun. 2003, 33, 1815–1820. [Google Scholar] [CrossRef]
- Pollini, G.; Baricordi, N.; Benetti, S.; De Risi, C.; Zanirato, V. A simple entry to chiralnonracemic 2-piperazinone derivatives. Tetrahedron Lett. 2005, 46, 3699–3701. [Google Scholar]
- Atsushi, N.; Toyoharu, M.; Hiroto, K.; Takeshi, E. Controlled Cationic Ring-Opening Polymerization of 1,3-Oxazolidine-2-thione Derived from L-Serine. Macromolecules 2003, 36, 9335–9339. [Google Scholar] [CrossRef]
- Khan, I.; Ali, S.; Hameed, S.; Rama, N.H.; Hussain, M.T.; Wadood, A.; Uddin, R.; Haq, Z.U.; Khan, A.; Ali, S.; Choudhary, M.I. Synthesis, antioxidant activities and urease inhibition of some new 1,2,4-triazole and 1,3,4-thiadiazole derivatives. Eur. J. Med. Chem. 2010, 45, 5200–5207. [Google Scholar]
- Syed, T.; Akhtar, T.; Al-Masoudi, N.A.; Jones, P.G.; Hameed, S. Synthesis, QSAR and anti- HIV activity of new 5-benzylthio-1,3,4-oxadiazoles derived from α-amino acids. J. Enzym. Inhib. Med. Chem. 2011, 26, 668–680. [Google Scholar] [CrossRef]
- Legrand, S.; Nordlander, G.; Nordenhem, H.; Borg-Karlson, A.K.; Unelius, C.R. Hydroxy-Methoxybenzoic Methyl Esters: Synthesis and Antifeedant Activity on the Pine Weevil, Hylobius abietis. Z. Naturforsch. 2004, 59b, 829–835. [Google Scholar]
- Blustein, G.; Romagnoli, R.; Jaen, J.A.; Di Sarli, A.R.; del Amo, B. Zinc basic benzoate as eco-friendly steel corrosion inhibitor pigment for anticorrosive epoxy-coatings. Colloid. Surface. A 2006, 290, 7–18. [Google Scholar] [CrossRef]
- Yun-Choi, H.S.; Kim, M.H.; Jung, K.H. Esters of Substituted Benzoic Acids as Anti thrombotic Agents. Arch. Pharm. Res. 1996, 19, 66–70. [Google Scholar] [CrossRef]
- You, Y.J.; Kim, K.; Nam, N.H.; Bang, S.C.; Ahn, B.Z. Alkyl and carboxylalkyl esters of 4′-demethyl-4-deoxypodophyllotoxin: synthesis, cytotoxic, and antitumor activity. Eur. J. Med. Chem. 2004, 39, 189–193. [Google Scholar] [CrossRef]
- Habulin, M.; Sabeder, S.; Knez, Z. Enzymatic synthesis of sugar fatty acid esters in organic solvent and in supercritical carbon dioxide and their antimicrobial activity. J. Supercrit. Fluid. 2008, 45, 338–345. [Google Scholar] [CrossRef]
- Qadeer, G.; Rama, N.H.; Malik, M.A.; Helliwell, M. Methyl 2,6-dimethoxybenzoate. Acta. Cryst. 2007, E63, o3027. [Google Scholar]
- Saleem, R.; Qadeer, G.; Shabir, G.; Hanif, M.; Wong, W.Y. Methyl 2-[2-(2,6-dichloroanilino)phenyl]acetate. Acta Crystallogr. E 2008, E64, o2400. [Google Scholar]
- Abid, O.R.; Babar, T.M.; Ali, F.I.; Ahmed, S.; Wadood, A.; Rama, N.H.; Uddin, R.; Haq, Z.U.; Khan, A.; Choudhary, M.I. Identification of Novel Urease Inhibitors by High-Throughput Virtual and in Vitro Screening. ACS Med. Chem. Lett. 2010, 1, 145–149. [Google Scholar] [CrossRef]
- Oxford Diffraction, CrysAlisPro; Oxford Diffraction Ltd.: Yarnton, England, 2009.
- Allen, F.H. The Cambridge Structural Database: a quarter of a million crystal structures and rising. Acta Cryst. 2008, B58, 380–388. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Cryst. 2008, A64, 112–122. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Khan, I.; Ibrar, A.; White, J.M. Ethyl 2,6-Dimethoxybenzoate: Synthesis, Spectroscopic and X-ray Crystallographic Analysis. Crystals 2012, 2, 521-527. https://doi.org/10.3390/cryst2020521
Khan I, Ibrar A, White JM. Ethyl 2,6-Dimethoxybenzoate: Synthesis, Spectroscopic and X-ray Crystallographic Analysis. Crystals. 2012; 2(2):521-527. https://doi.org/10.3390/cryst2020521
Chicago/Turabian StyleKhan, Imtiaz, Aliya Ibrar, and Jonathan M. White. 2012. "Ethyl 2,6-Dimethoxybenzoate: Synthesis, Spectroscopic and X-ray Crystallographic Analysis" Crystals 2, no. 2: 521-527. https://doi.org/10.3390/cryst2020521
APA StyleKhan, I., Ibrar, A., & White, J. M. (2012). Ethyl 2,6-Dimethoxybenzoate: Synthesis, Spectroscopic and X-ray Crystallographic Analysis. Crystals, 2(2), 521-527. https://doi.org/10.3390/cryst2020521




