Gallium-Promoted Ni Catalyst Supported on MCM-41 for Dry Reforming of Methane
Abstract
1. Introduction
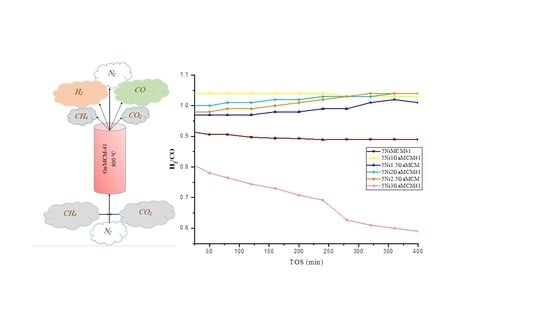
2. Results and Discussion
3. Materials and Methods
3.1. Catalyst Preparation
3.2. Catalytic Activity Test
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Tian, C.L.H.; Ciais, P.; Michalak, A.M.; Canadell, J.G.; Saikawa, E.; Huntzinger, D.N.; Gurney, K.R.; Sitch, S.; Zhang, B.; Yang, J.; et al. The terrestrial biosphere as a net source of greenhouse gases to the atmosphere. Nature 2015, 531, 225–228. [Google Scholar] [CrossRef] [PubMed]
- Lunsford, J.H. Catalytic conversion of methane to more useful chemicals and fuels: A challenge for the 21st century. Catal. Today 2000, 63, 165–174. [Google Scholar] [CrossRef]
- Omae, L. Aspects of carbon dioxide utilization. Catal. Today 2006, 115, 33–52. [Google Scholar] [CrossRef]
- Liu, D.; Cheo, W.N.; Lau, R.; Borgna, A.; Yang, Y.J. MCM-41 supported nickel-based bimetallic catalysts with superior stability during carbon dioxide reforming of methane: Effect of strong metal-support interaction. J. Catal. 2009, 266, 380–390. [Google Scholar] [CrossRef]
- Bradford, M.C.J.; Vannice, M.A. CO2 reforming of methane. Catal. Rev. 1999, 41, 1–42. [Google Scholar] [CrossRef]
- Li, D.L.Y.; Wang, G. Methane decomposition to COx-free hydrogen and nano-carbon material on group 8-10 base metal catalysts: A review. Catal. Today 2011, 162, 1–48. [Google Scholar] [CrossRef]
- Al-Fatesh, A.S.; Amin, A.; Ibrahim, A.A.; Khan, W.U.; Soliman, M.A.; AL-Otaibi, R.L.; Fakeeha, A.H. Effect of Ce and Co Addition to Fe/Al2O3 for Catalytic Methane Decomposition. Catalysts 2016, 6, 40. [Google Scholar] [CrossRef]
- Usman, M.M.; Wan Daud, W.M.A.; Abbas, H.F. Dry reforming of methane: Influence of process parameters—A review. Renew. Sustain. Energy Rev. 2015, 45, 710–744. [Google Scholar] [CrossRef]
- Eltejaei, H.; Bozorgzadeh, J.T.H. R.; Omidkhah, M.R.; Rezaei, M.; Zanganeh, R.; Zamaniyan, A.; Ghalam, A.Z. Methane dry reforming on Ni/Ce0.75Zr0.25O2-MgAl2O4 and Ni/Ce0.75Zr0.25O2-gamma-alumina: Effects of support composition and water addition. Int. J. Hydrogen Energy 2012, 37, 4107–4118. [Google Scholar] [CrossRef]
- Wolfbeisser, O.S.A.; Bernardi, J.; Wittayakun, J.; Föttinger, K.; Rupprechter, G. Methane dry reforming over ceria-zirconia supported Ni catalysts. Catal. Today 2016, 277, 234–245. [Google Scholar] [CrossRef]
- Rahemi, N.; Haghighi, M.; Babaluo, A.A.; Jafari, M.F.; Estifaee, P. Synthesis and physicochemical characterizations of Ni/Al2O3-ZrO2 nanocatalyst prepared via impregnation method and treated with non-thermal plasma for CO2 reforming of CH4. J. Ind. Eng. Chem. 2013, 19, 1566–1576. [Google Scholar] [CrossRef]
- Aghamohammadi, S.; Haghighi, M.; Karimipour, S. A comparative synthesis and physicochemical characterizations of Ni/Al2O3-MgO nanocatalyst via sequential impregnation and sol-gel methods used for CO2 reforming of methane. Nanosci. Nanotechnol. 2013, 13, 4872–4882. [Google Scholar] [CrossRef]
- Sener, T.D.C.; Dogu, G. Effects of synthesis conditions on the structure of Pd incorporated MCM-41 type mesoporous nanocomposite catalytic materials with high Pd/Si ratios. Microporous Mesoporous Mater. 2006, 94, 89–98. [Google Scholar] [CrossRef]
- Amin, M.H.; Tardio, J.; Bhargava, S.K. A comparison study on carbon dioxide reforming of methane over Ni catalysts supported on mesoporous SBA-15, MCM-41, KIT-6 and gamma-Al2O3. In Chemeca 2013: Challenging Tomorrow; Engineers Australia: Brisbane, Australia, 2013; pp. 543–548. [Google Scholar]
- Chen, X.W.C.; Zhang, L.; Zou, X.; Ding, W.; Lu, X. Synthesis of mesoporous Ni-La2O3/SiO2 by ploy(ethylene glycol)-assisted sol-gel route as highly efficient catalysts for dry reforming of methane with a H2/CO ratio of unity. Catal. Commun. 2017, 94, 38–41. [Google Scholar] [CrossRef]
- Deshmane, V.G.; Abrokwah, R.Y.; Kuila, D. Synthesis of stable Cu-MCM-41 nanocatalysts for H2 production with high selectivity via steam reforming of methanol. Int. J. Hydrogen Energy 2015, 40, 10439–10452. [Google Scholar] [CrossRef]
- Abrokwah, R.Y.; Deshmane, V.G.; Kuila, D. Comparative performance of MMCM-41(M: Cu, Co, Ni, Pd, Zn and Sn) catalysts for steam reforming of methanol. J. Mol. Catal. A Chem. 2016, 425, 10–20. [Google Scholar] [CrossRef]
- Damyanova, B.P.S.; Arishtirova, K.; Fierro, J.L.G.; Sener, C.; Dogu, T. MCM-41 supported PdNi catalysts for dry reforming of methane. Appl. Catal. B 2009, 92, 250–261. [Google Scholar] [CrossRef]
- Liu, D.; Quek, X.Y.; Wah, H.H.A.; Zeng, G.; Li, Y.; Yang, Y. Carbon dioxide reforming of methane over nickel-grafted SBA-15 and MCM-41 catalysts. Catal. Today 2009, 148, 243–250. [Google Scholar] [CrossRef]
- Liu, D.; Lau, R.; Borgna, A.; Yang, Y. Carbon dioxide reforming of methane to synthesis gas over Ni-MCM-41 catalysts. Appl. Catal. A 2009, 358, 110–118. [Google Scholar] [CrossRef]
- Lovell, Y.J.E.; Scott, J.; Wang, F.; Suhardja, Y.; Chen, M.; Huang, J.; Amal, R. CO2 reforming of methane over MCM-41-supported nickel catalysts: Altering support acidity by one-pot synthesis at room temperature. Appl. Catal. A 2014, 473, 51–58. [Google Scholar] [CrossRef]
- Xu, L.; Song, H.; Chou, L. Carbon dioxide reforming of methane over ordered mesoporous NiO-MgO-Al2O3 composite oxides. Appl. Catal. B 2011, 108, 177–190. [Google Scholar] [CrossRef]
- Zhang, S.M.S.; Ishiguro, N.; Tada, M. Ceria-Doped Ni/SBA-16 Catalysts for Dry Reforming of Methane. ACS Catal. 2013, 3, 1855–1864. [Google Scholar] [CrossRef]
- Arbag, S.Y.H.; Yasyerli, N.; Dogu, G. Activity and stability enhancement of Ni-MCM-41 catalysts by Rh incorporation for hydrogen from dry reforming of methane. Int. J. Hydrogen Energy 2010, 35, 2296–2304. [Google Scholar] [CrossRef]
- Ambursa, M.M.; Sudarsanam, P.; Voon, L.H.; Hamid, S.B.; Bhargava, S.K. Bimetallic Cu-Ni catalysts supported on MCM-41 and Ti-MCM-41 porous materials for hydrodeoxygenation of lignin model compound into transportation fuels. Fuel Process. Technol. 2017, 162, 87–97. [Google Scholar] [CrossRef]
- Liu, D.; Cheo, W.N.; Lim, Y.W.; Borgna, A.; Lau, R.; Yang, Y. A comparative study on catalyst deactivation of nickel and cobalt incorporated MCM-41 catalysts modified by platinum in methane reforming with carbon dioxide. Catal. Today 2010, 154, 229–236. [Google Scholar] [CrossRef]
- Ochoa, J.V.; Malmusi, A.; Recchi, C.; Cavani, F. Understanding the Role of Gallium as a Promoter of Magnesium Silicate Catalysts for the Conversion of Ethanol into Butadiene. ChemCatChem 2017, 9, 2128–2135. [Google Scholar] [CrossRef]
- Medina, J.C.; Figueroa, M.; Manrique, R.; Pereira, J.R.; Srinivasan, P.D.; Bravo-Suárez, J.J.; Medrano, V.G.; Jiménez, R.; Karelovic, A. Catalytic consequences of Ga promotion on Cu for CO2 hydrogenation to methanol. Catal. Sci. Technol. 2017, 7, 3375–3387. [Google Scholar] [CrossRef]
- Ruff, J.K. Friedel-Crafts and Related Reactions. Volume I: General Aspects. By George Olah. Inorg. Chem. 1964, 3, 1205–1206. [Google Scholar] [CrossRef]
- Häfele, A.R.M.; Roppelt, D.; Emig, G. Hydroxylation of benzene with nitrous oxide on H-Ga-ZSM5 zeolite. Appl. Catal. A 1997, 150, 153–164. [Google Scholar] [CrossRef]
- Ibrahim, A.A.; Fakeeha, A.H. Al-Fatesh, A.S. Enhancing hydrogen production by dry reforming process with strontium promoter. Int. J. Hydrogen Energy 2014, 39, 1680–1687. [Google Scholar] [CrossRef]
- Nguyen, H.K.D.; Dang, T.H.; Nguyen, N.L.T.; Nguyen, H.T.; Dinh, N.T. Novel Ni-Ga alloy based catalyst for converting CO2 to methanol. Can. J. Chem. Eng. 2018, 96, 832–837. [Google Scholar] [CrossRef]
- Studt, I.S.F.; Abild-Pedersen, F.; Elkjær, C.F.; Hummelshøj, J.S.; Dahl, S.; Chorkendorff, S.I.; Nørskov, J.K. Discovery of a Ni-Ga catalyst for carbon dioxide reduction to methanol. Nat. Chem. 2014, 6, 320–324. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.Y.N.; Wang, Y.; Chu, W.; Liu, M. A comparison study on methane dry reforming with carbon dioxide over LaNiO3 perovskite catalysts supported on mesoporous SBA-15, MCM-41 and silica carrier. Catal. Today 2013, 212, 98–107. [Google Scholar] [CrossRef]
- Lin, S.; Yang, L.; Yang, X.; Zhou, R. Redox properties and metal–support interaction of Pd/Ce0.67Zr0.33O2–Al2O3 catalyst for CO, HC and NOx elimination. Appl. Surf. Sci. 2014, 305, 642–649. [Google Scholar] [CrossRef]
- Supamathanon, J.W.N.; Prayoonpokarach, S.; Supronowicz, W.; Rößner, F. Basic properties of potassium oxide supported on zeolite y studied by pyrrole-tpd and catalytic conversion of methylbutynol Quim. Nova 2012, 35, 1719–1723. [Google Scholar]
- Tsoncheva, L.I.T.; Rosenholm, J.; Linden, M. Cobalt oxide species supported on SBA-15, KIT-5 and KIT-6 mesoporous silicas for ethyl acetate total oxidation. Appl. Catal. B 2009, 89, 365–374. [Google Scholar] [CrossRef]
- Samadi-Maybodi, M.T.A.; Vahid, A.; Miranbeigi, A. In situ incorporation of nickel nanoparticles into the mesopores of MCM-41 by manipulation of solvent-solute interaction and its activity toward adsorptive desulfurization of gas oil. J. Hazard. Mater. 2011, 192, 1667–1674. [Google Scholar] [CrossRef] [PubMed]
- Dai, B.W.B.; Zhu, M.; Kang, L.; Yu, F. Nickel catalysts supported on amino-functionalized MCM-41 for syngas methanation. RSC Adv. 2016, 6, 66957–66962. [Google Scholar] [CrossRef]
- Hassan, H.B.; Abdel Rahim, M.A.; Khalil, M.W.; Mohammed, R.F. Ni Modified MCM-41 as a Catalyst for Direct Methanol Fuel Cells. Int. J. Electrochem. Sci 2014, 9, 760–777. [Google Scholar]
- Michorczyk, J.O.P.; Kuśtrowski, P.; Chmielarz, L. Chromium oxide supported on MCM-41 as a highly active and selective catalyst for dehydrogenation of propane with CO2. Appl. Catal. A 2008, 349, 62–69. [Google Scholar] [CrossRef]
- Al–Fatish, A.S.A.; Ibrahim, A.A.; Fakeeha, A.H.; Soliman, M.A.; Siddiqui, M.R.H.; Abasaeed, A.E. Coke formation during CO2 reforming of CH4 over alumina-supported nickel catalysts. Appl. Catal. A Gen. 2009, 364, 150–155. [Google Scholar]












| Catalyst | BET m2 g−1 | P.V cm3 g−1 | P.D nm |
|---|---|---|---|
| MCM-41 | 1132.72 | 0.664 | 2.57 |
| 5%Ni/MCM-41 | 897.85 | 0.485 | 2.48 |
| 1%Ga+5%Ni/MCM-41 | 917.38 | 0.664 | 2.74 |
| 1.5%Ga+5%Ni/MCM-41 | 978.56 | 0.660 | 2.74 |
| 2%Ga+5%Ni/MCM-41 | 983.33 | 0.664 | 2.74 |
| 2.5%Ga+5%Ni/MCM-41 | 1036.02 | 0.593 | 2.57 |
| 3%Ga+5%Ni/MCM-41 | 979.91 | 0.560 | 2.57 |
| Catalyst | 50–170 °C μmol g−1 | 170–500 °C μmol g−1 | 500–800 °C μmol g−1 | Total μmol g−1 |
|---|---|---|---|---|
| 5%Ni/MCM-41 | 8.79 | 43.32 | 182.35 | 234.45 |
| 1.0%Ga5%Ni/MCM-41 | 20.26 | 33.87 | 110.60 | 164.73 |
| 2.0%Ga5%Ni/MCM-41 | 22.74 | 26.30 | 93.65 | 142.69 |
| 2.5%Ga5%Ni/MCM-41 | 23.15 | 42.90 | 79.13 | 145.18 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Fatesh, A.S.; Ibrahim, A.A.; Abu-Dahrieh, J.K.; Al-Awadi, A.S.; El-Toni, A.M.; Fakeeha, A.H.; Abasaeed, A.E. Gallium-Promoted Ni Catalyst Supported on MCM-41 for Dry Reforming of Methane. Catalysts 2018, 8, 229. https://doi.org/10.3390/catal8060229
Al-Fatesh AS, Ibrahim AA, Abu-Dahrieh JK, Al-Awadi AS, El-Toni AM, Fakeeha AH, Abasaeed AE. Gallium-Promoted Ni Catalyst Supported on MCM-41 for Dry Reforming of Methane. Catalysts. 2018; 8(6):229. https://doi.org/10.3390/catal8060229
Chicago/Turabian StyleAl-Fatesh, Ahmed S., Ahmed A. Ibrahim, Jehad K. Abu-Dahrieh, Abdulrahman S. Al-Awadi, Ahmed Mohamed El-Toni, Anis H. Fakeeha, and Ahmed E. Abasaeed. 2018. "Gallium-Promoted Ni Catalyst Supported on MCM-41 for Dry Reforming of Methane" Catalysts 8, no. 6: 229. https://doi.org/10.3390/catal8060229
APA StyleAl-Fatesh, A. S., Ibrahim, A. A., Abu-Dahrieh, J. K., Al-Awadi, A. S., El-Toni, A. M., Fakeeha, A. H., & Abasaeed, A. E. (2018). Gallium-Promoted Ni Catalyst Supported on MCM-41 for Dry Reforming of Methane. Catalysts, 8(6), 229. https://doi.org/10.3390/catal8060229