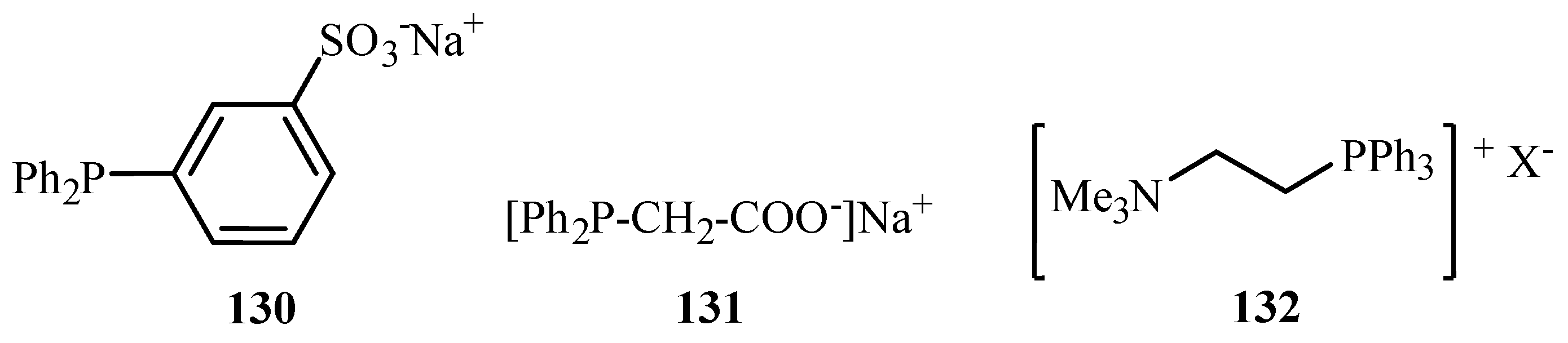1. Introduction
The new era of research started after the introduction of coupling reactions, i.e., carbon–carbon bond forming reactions like Heck [
1], Suzuki [
2], Sonogoshira [
3], Negishi [
4], Kumada [
5], Stille [
6], Tsuji-Trost [
7], etc. These reactions have played an enormously decisive and important role in shaping chemical synthesis and have revolutionized the way one thinks about synthetic organic chemistry. The Heck reaction (Equation (1)) is used extensively in many syntheses, including agrochemical, fine chemicals, pharmaceutical, etc. The reaction was introduced by Mizoroki [
8] and Heck [
9] independently more than four decades ago. It has drawn much attention due to high efficiency and simplicity. Heck methodology is attractive from a synthetic point of view because of its high chemoselectivity and mild reaction conditions along with low toxicity and cost of the reagent if, specifically, the catalyst is recycled.
![Catalysts 07 00267 i001]()
The Heck reaction is described as a vinylation or arylation of olefins where a large variety of olefins can be used, like derivatives of acrylates, styrenes or intramolecular double bonds. The aryl halide variants developed in addition to typical aryl bromides and iodides are aromatic triflates, aroyl chlorides, aryl sulfonyl chlorides, aromatic diazonium salts, aroyl anhydrides, aryl chlorides and arylsilanols. The catalyst is the essential part of a reaction where a variety of metals along with a huge range of ligands is studied. Significant progress for the preparation and characterization of variety of ligands and catalysts has been made for avoiding protection and deprotection procedures, therefore allowing for syntheses to be carried out in fewer steps. Development, novel catalytic properties and extensive mechanistic studies are summarized in several reviews based on seminal work of many researchers and reviewers as described in following sections. Palladium is usually the preferred metal as it tolerates a wide variety of functional groups and it has a remarkable ability to assemble C-C bonds between appropriately functionalized substrates. Most palladium based methodologies proceed with stereo- and regioselectivity and with excellent yields. Generally, the less crowded structure is preferred during Heck reaction, and often favours a trans product. Few mechanisms are also supported by the discussion on the regioselectivity and stereoselectivity of Heck reaction.
Sometimes compounds such as TBAB (Tetra butyl ammonium bromide) are added in the reaction mixture along with organic or inorganic bases needed for the sequestration of acid generated. Typical solvents for the Heck reaction are dipolar aprotic solvents like DMF (dimethyl formamide) and NMP (N-methyl-2-pyrrolidone), however, the reaction is also performed in many other different solvents and in fact there are large numbers of reviews dedicated to the use of various solvents. Besides these, many manuscripts describe the reaction in the absence of one of the component (other than substrates) such as ligand free, organic solvent free and so on. Recent publications also describe how to recover used catalyst specially using aqueous medium which implies the potential for greener approach for organic reactions.
There are a large number of excellent reviews available on Heck reaction and related topics based on applications, mechanism, quest for high turn over numbers (TONs), asymmetric synthesis, separation techniques, etc. In addition to this, there are a few manuscripts that give a general overview about Heck reaction, some of them are detailed and some are not so descriptive. Many reviews discuss various catalysis techniques with certain aspects where along with the Heck reaction, other coupling reactions are also discussed, however for the present review, only the Heck related chemistry is considered. It should be noted that for this review only the reviews and few related articles covering Heck chemistry are considered. Consideration of book articles, although informative, is beyond the scope of this review.
2. Reviews on Catalyst Development
The majority of the work on Heck reaction has been focused on the catalyst development. There are numerous reviews based on this and hence are further categorized for better understanding.
2.1. Overviews
When the mechanism was not fully explored, the reaction and its possible mechanistic pathways are reviewed by one of its pioneers, R.F. Heck [
10,
11,
12] by focusing on possibilities of the intermediate formed during Heck reaction by considering various examples for its support. These reviews mention the working reaction conditions explored for the reaction and focuses on the requirements of the substrates, bases, solvents and the conditions to be used. It was observed that the reaction is usually regioselective and stereospecific and is tolerant of almost every functional group. Data have proved that the direction of addition of the organopalladium complex to the olefin depends upon the steric and electronic influence of the substituent present. The direction of addition is most often dominated by steric effects where the organic group is seen to be added to the least substituted carbon of the double band.
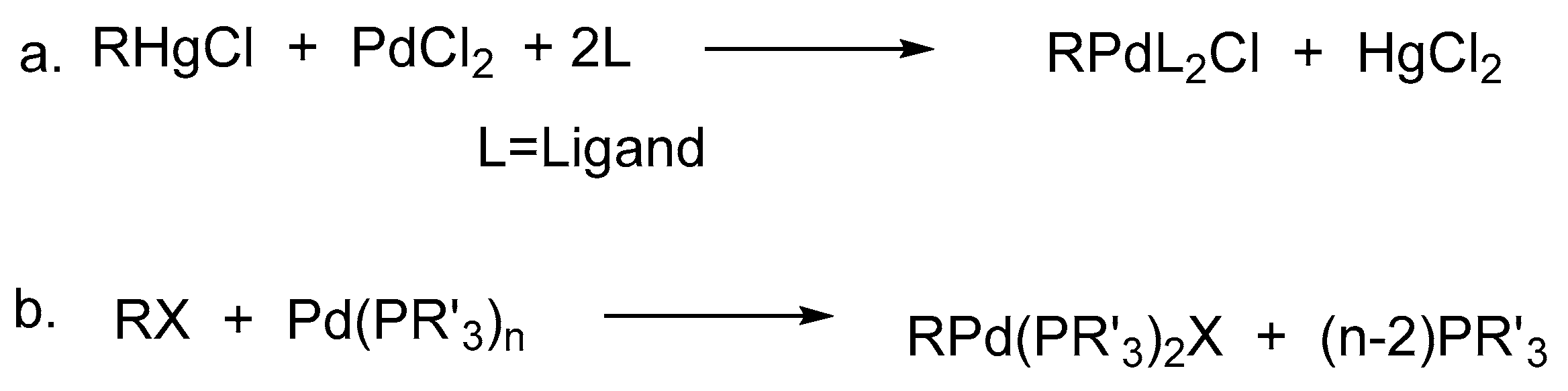
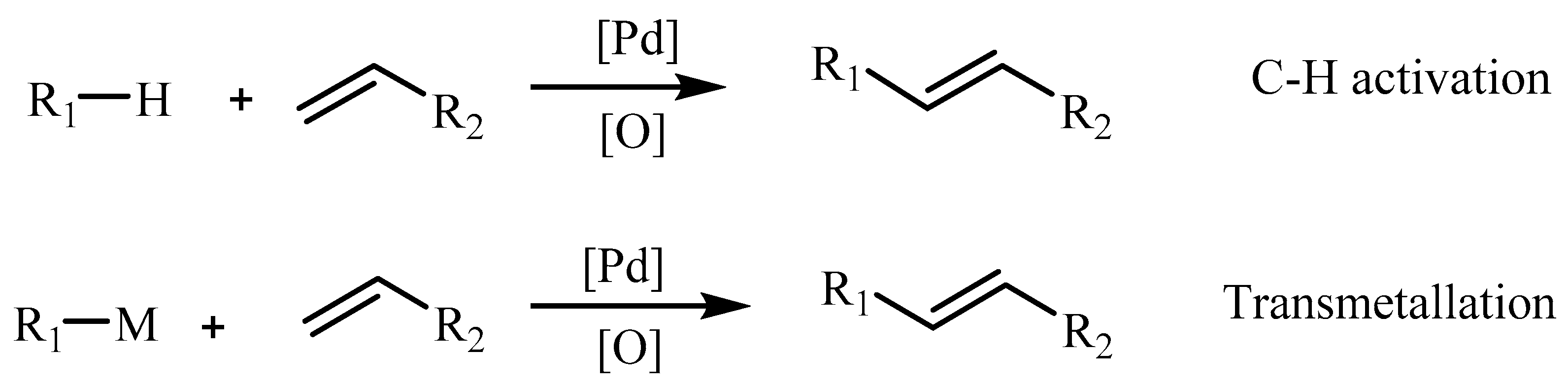
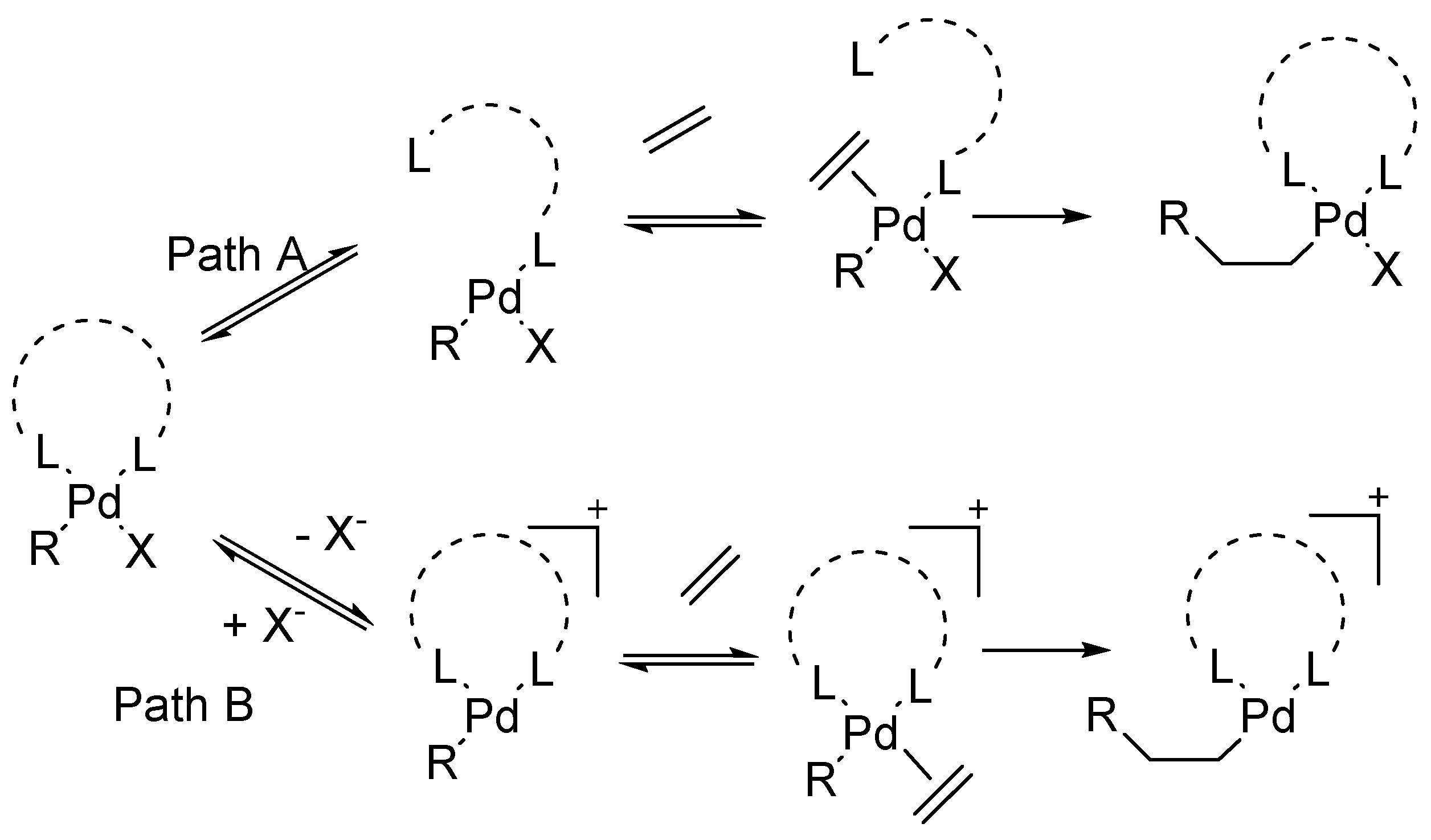
Initially, the intermediate mono-organopalladium(II) species, RPdL
2X, were prepared by exchange reactions of mercurials in general, with palladium(II) salts (
Scheme 1a), however it suffered from the disadvantage that, many such main group organometallics were not easily accessible and moreover they were needed to be used in stoichiometric amounts in the synthesis of organic compounds. Hence, the finely divided palladium metal or palladium(0) phosphine complexes were used for such reactions (
Scheme 1b).
Effects of different triaryl phosphines have also been studied. It is mentioned that, generally the reaction does not require anhydrous or anaerobic conditions although it is advisable to limit access of oxygen when arylphosphines are used as a component of the catalyst. Aryl, heterocyclic, benzyl, or vinyl halides are commonly employed often with bromides and iodides in comparison to halides with an easily eliminated beta-hydrogen atom (i.e., alkyl derivatives) since they form only olefins by elimination under the normal reaction conditions. Generally used bases are secondary or tertiary amine, sodium or potassium acetate, carbonate, or bicarbonates. The catalyst is commonly palladium acetate, although palladium chloride or preformed triarylphosphine palladium complexes, as well as palladium on charcoal, have been used.
Triarylphosphine or a secondary amine is required when organic bromides are used although a reactant, product, or solvent may serve as the ligand for reactions involving organic iodides. Solvents such as acetonitrile, dimethylformamide, hexamethylphosphoramide, N-methylpyrrolidinone, and methanol have been used, but are often not necessary. The procedure is applicable to a very wide range of reactants and yields are generally good to excellent. Temperature used is in the range of 50 to 160 °C, where the reaction proceeds homogeneously. When nucleophilic secondary amines are used as co-reactants with most vinylic halides, a variation occurs that often produces tertiary allylic amines as major products. A comparison of Heck reaction with similar types of reaction where the organic halide is replaced by other reagents such as organometallics, diazonium salts, or aromatic hydrocarbons has also been discussed.
De Meijere and Meyer [
13] have reviewed mainly the Heck couplings with various oligohaloarenes along with an overall discussion on development in mechanism and catalysts until 1994. The review stresses upon the careful choice of substrates and skilful tailoring of reaction conditions leading to impressive sequences.
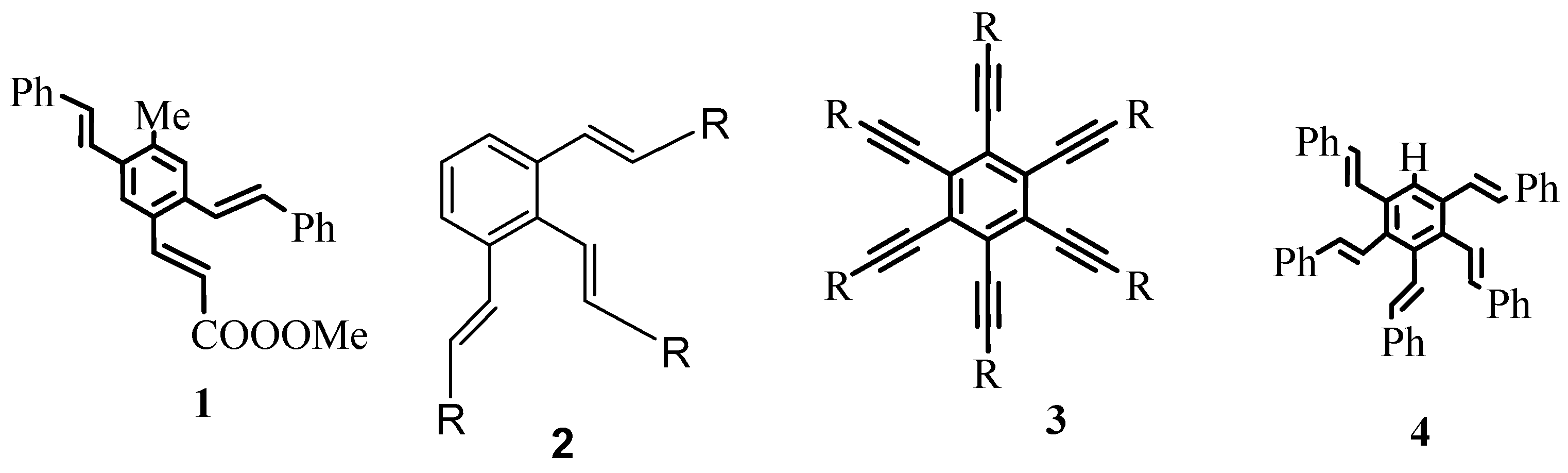
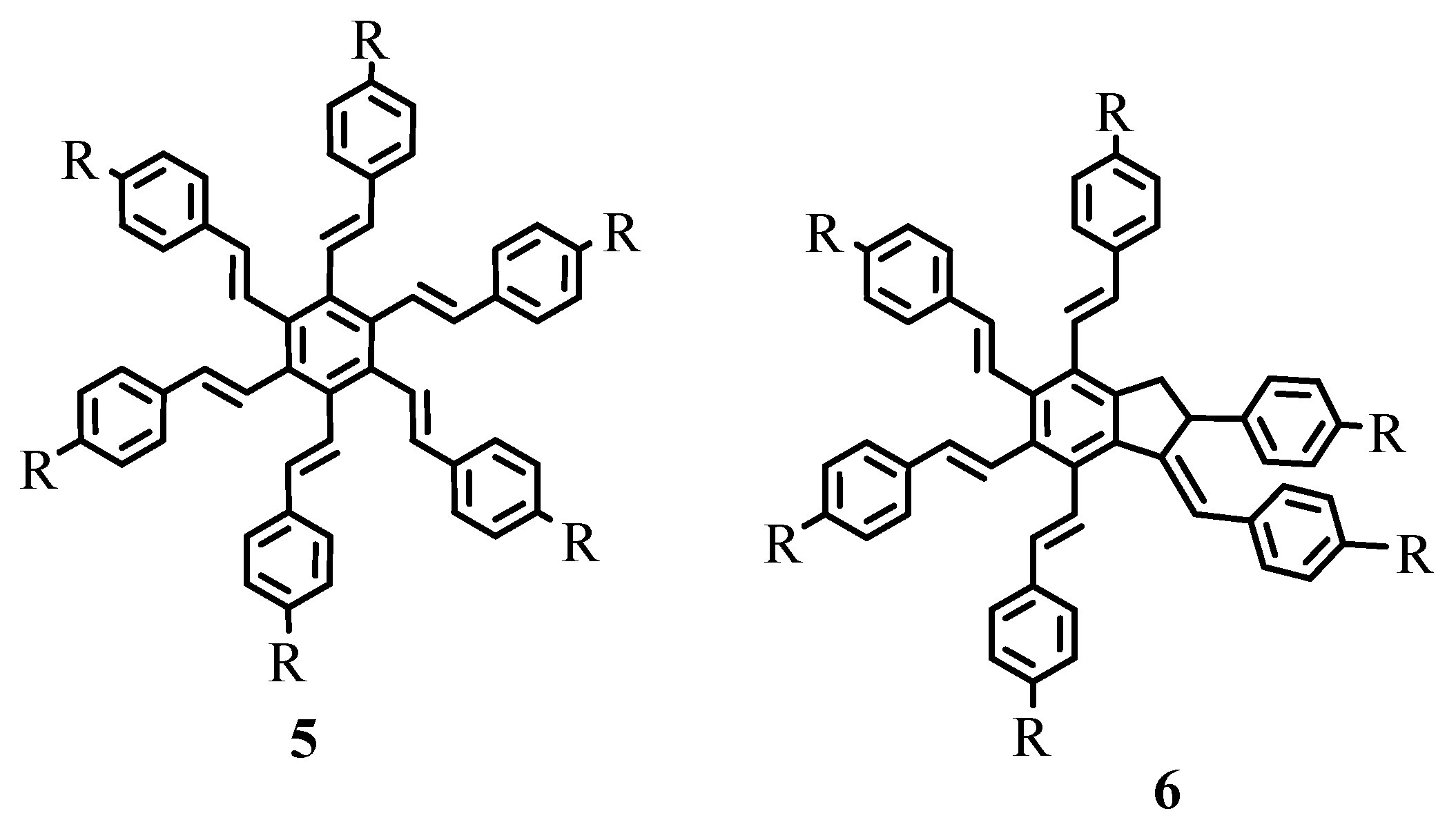
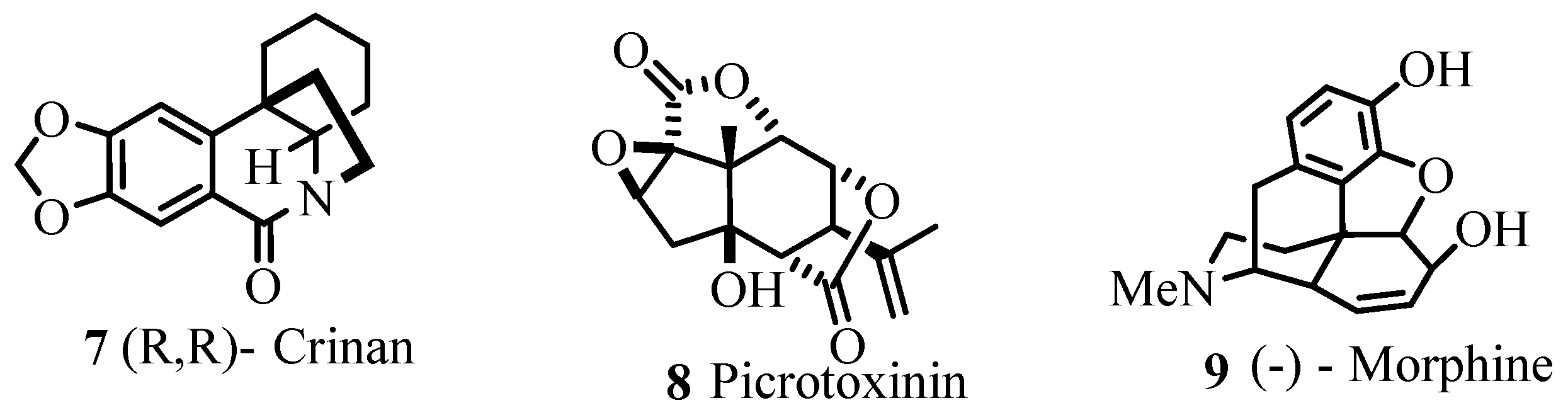
As exemplified in the review, the Heck reaction, together with other mechanistically related palladium catalysed transformations with arene, alkene and alkyne derivatives, opens the door to a tremendous variety of elegant and highly convergent routes to structurally complex molecules. The reaction is not hindered by heteroatoms such as oxygen, nitrogen, sulphur and phosphorus in most of the cases. With Heck reaction, a range of chemoselective and regioselective monocouplings of highly functionalized substrates with unsymmetrical and multisubstituted reaction partners could be achieved. A number of examples are given that demonstrate the cascade reactions in which three, four, five, and even eight new C-C bonds are formed to yield oligofunctional and oligocyclic products like
1–
6 (
Figure 1) with impressive molecular complexity. Cases are presented to establish the reactivity for enantioselective construction of complex natural products with quaternary stereocenters as exemplified by the synthesis of (R,R)-crinan
7, picrotoxinin
8, and morphine
9 (
Figure 2).
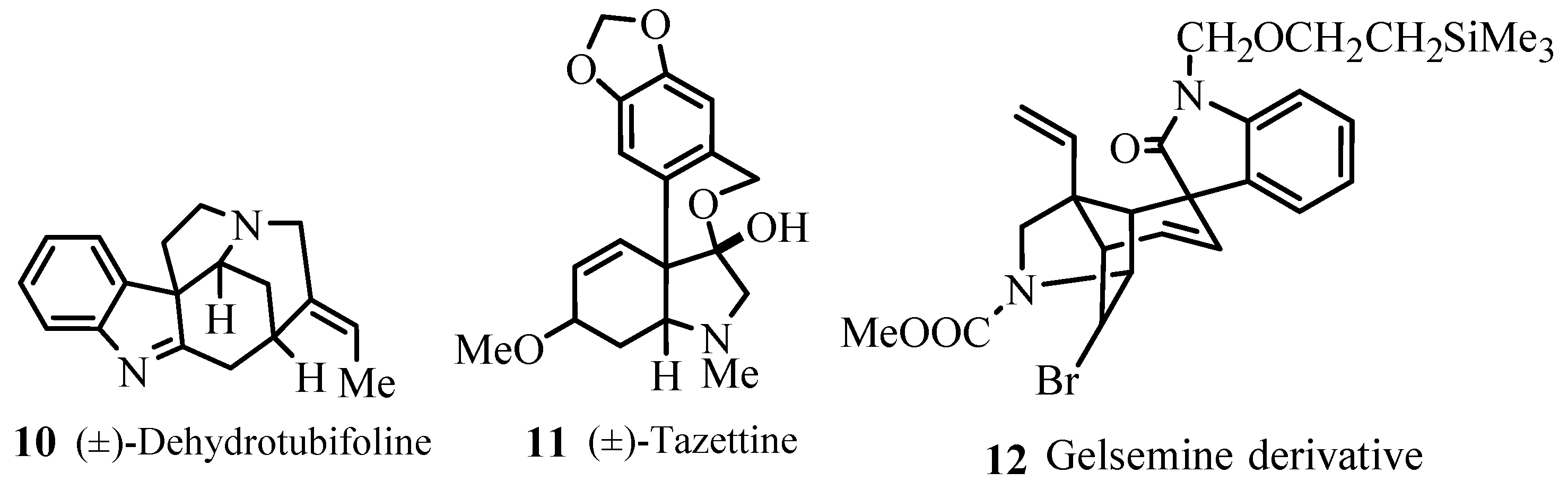
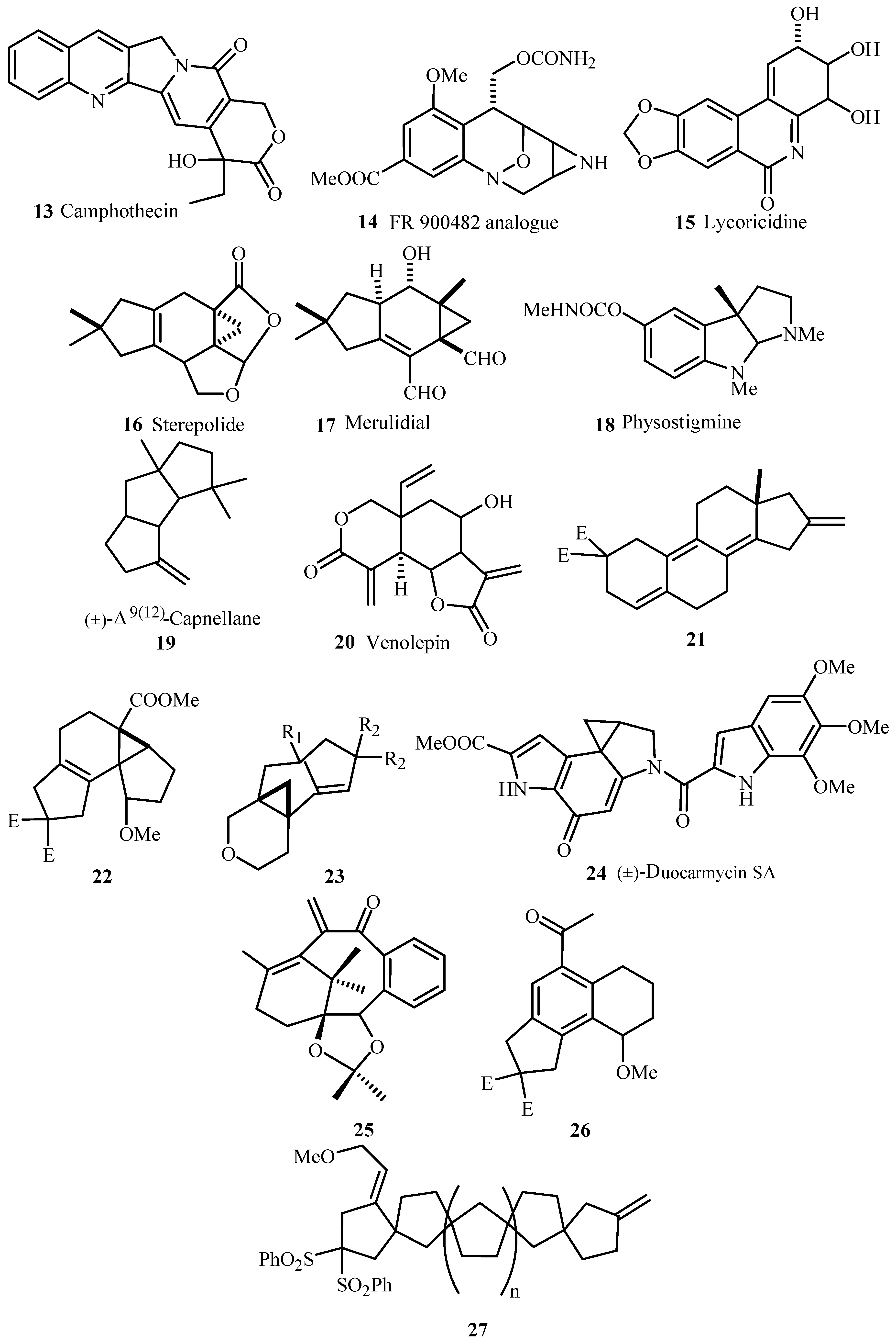
Several examples for syntheses of natural products
10–
27 (
Figure 3) have been provided that involve different types of Heck transformations as one of the important key steps like,
Heck reactions with intermolecular asymmetric induction where compounds with ee (enantiomeric excess) up to 99%.
Multiple component reactions and domino coupling reactions performed with several alkenes and various haloarenes.
Pd-catalysed heteroannelations.
Intramolecular Heck reactions.
Palladium catalysed additions to triple bonds and the coupling products derived from norbornene and dicyclopentadiene that serve as precursors for diverse polycyclic aromatic compound.
Thus, the review gives the expanse of Heck reaction which is one of the important synthetic methods available to organic chemists.
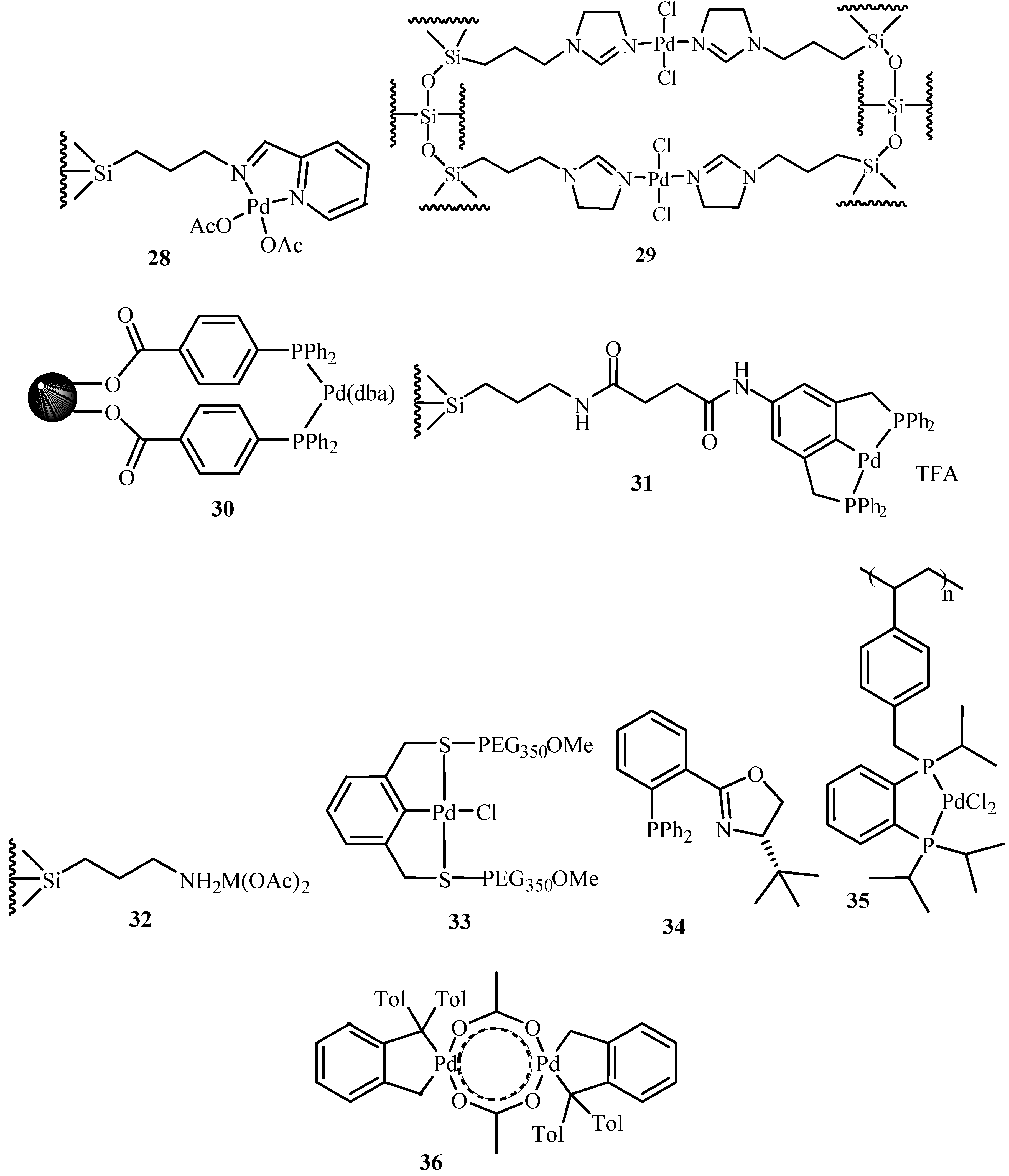
Kobetić and Biliškov [
14] have summarized the basics of Heck reaction mechanism, various compositions of applicable catalyst, their developments and applications of Heck reaction until then. Sequential development of both homogeneous and heterogeneous catalysis has been discussed giving examples. Use of palladium with carbon, metal oxides and their salts, silica, porous glass tube, glass beads and guanidinium phosphane clay, zeolites and molecular sieves, MCM 41, polymer, dendrimer, etc., are elaborated with their reaction conditions and yield. Many of these catalysts shows good activity and few of them have proved to be good in recycle study as well. The review also takes an account of substrates, solvents and reaction conditions used. There are few examples of cascade and multiple coupling, synthesis of natural and biologically active compounds and enantioselective Heck-type reactions. Catalysts
28–
36 (
Figure 4) are seen to give yields in the range of 48 to 93%. Out of all these palladacycles,
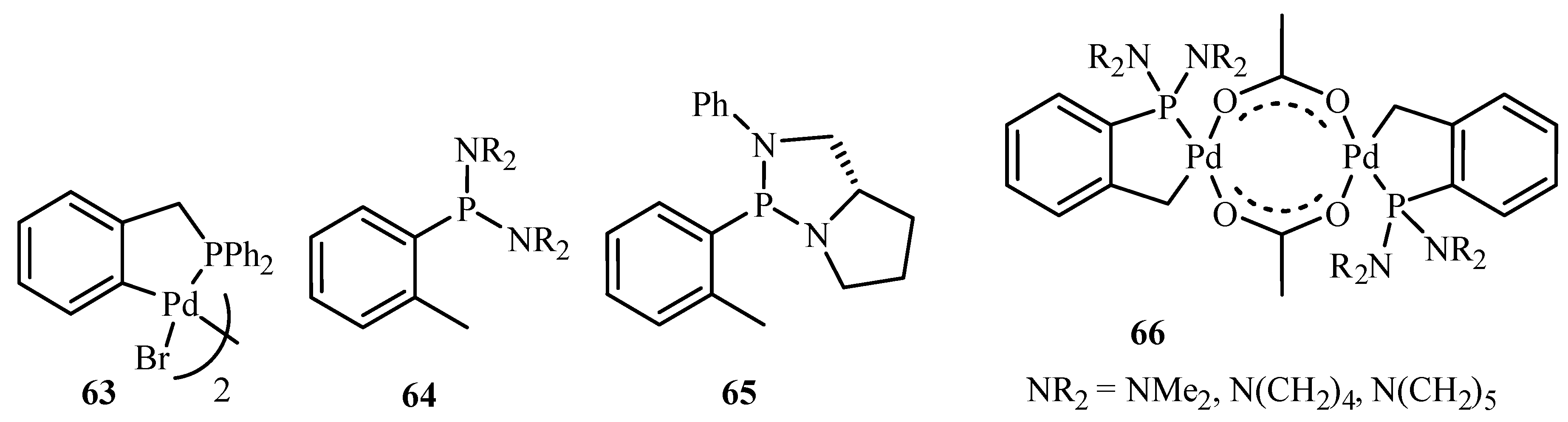
36 is the most studied and most reviewed catalyst. In the review, the use of a phase transition catalyst (quaternary ammonium salts), and the solid base that accelerates the Heck reaction are considered to be big breakthrough discoveries.
A review by Sahu and Sapkale [
15] discusses the mechanism of palladium catalysed coupling reactions, details about palladium metal, its reactivity and use for reaction like Heck, Suzuki, Negishi, Hiyama, Stille, Fukuyama, Sonogoshira, Buchwald Hartwig and their variants.
The reactivity of palladium is explained on the basis of few points such as
Electronegativity of palladium develops a polarised Pd-X bond leading to relatively strong Pd-H and Pd-C bonds.
Its variable oxidation states such as Pd(0), Pd(II) and sometimes Pd(IV) are essential for processes such as oxidative addition, transmetalation and reductive elimination.
Oxidation states such as Pd(I), Pd(III) and Pd(IV) are also known, where Pd(IV) species are essential in C-H activation mechanisms.
Although not in detail, references for Heck reaction in ionic liquid and for Amino-Heck are given along with the mention of application of Heck reaction in the synthesis of an alkaloid Rhazinal
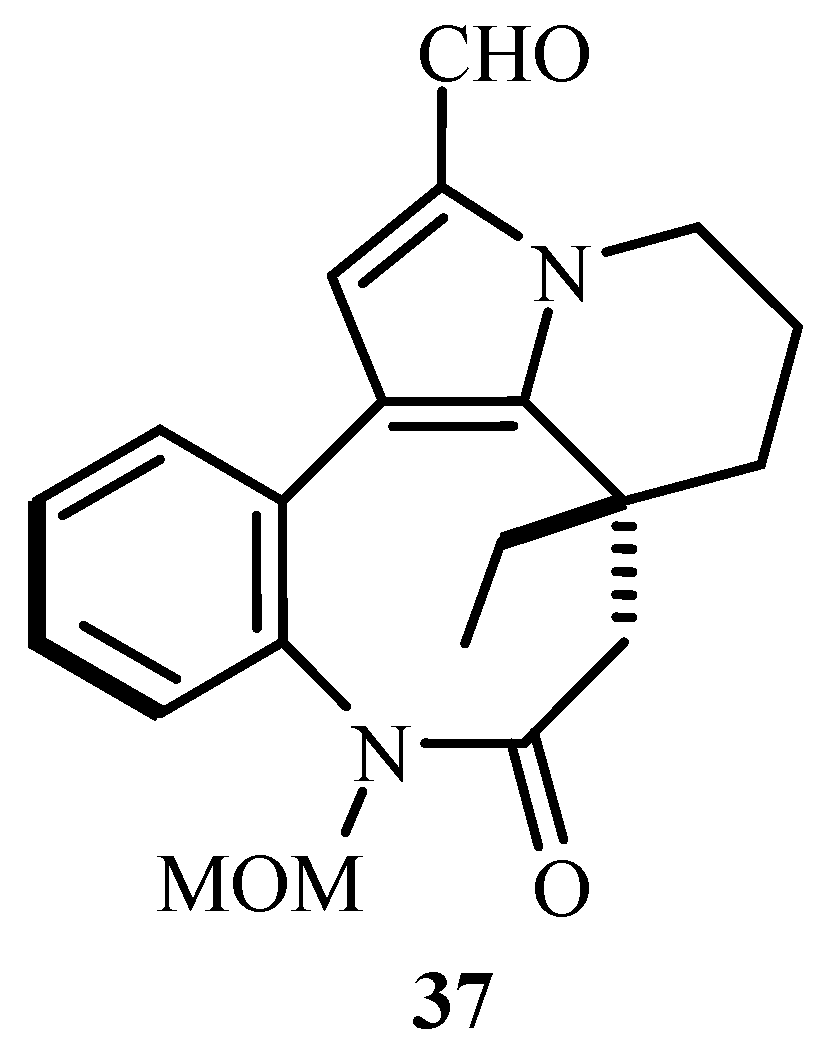
37 (
Figure 5), an antimitotic agent obtained from the stem extract of Kopsia teoi and a promising starting point for anticancer agent. MOM-Rhazinal (MOM = Methoxymethyl ether) derivative is made from the derivative of pyrrol where intramolecular Heck reaction takes place in presence on Pd(OAc)
2.
2.2. Catalysts Variants
Every research in catalysis mostly proceeds with the aim of improving the overall catalytic activity for a particular reaction. A progressive development targeting the increase in activity has been discussed thoroughly for Heck reaction as well. Researchers work hard to find cheaper ligands, catalyst system with higher activity so as to minimize the load of palladium and ligand and to find suitable systems for the processing of aryl chlorides too.
2.2.1. Overall Progress
Beletskaya and Cheprakov [
16] have published a critical and profound review on Heck reaction that systematically gives details of the work done up to 2000. It is still one of the most comprehensive reviews on Heck reactions.
A number of examples are presented to elaborate the use of various catalysts and ligands for Heck reactions like palladacycles, pincers, carbene complexes, etc. The review supports the mechanism involving Pd(0)/Pd(II) cycle by giving many examples, however the possibility of a mechanism involving Pd(IV)/Pd(II) cycle for few ligands has also been considered. For the reaction of styrene and aryl halide with a mechanism involving a Pd(0)/Pd(II) cycle, regiochemistry of the addition to styrene depends on the solvent and the nature of the anionic ligand. At higher solvent polarity and the weaker coordination ability of ligand there is a greater contribution of a truly cationic form of palladium complex, and the higher relative yield of 1,1-diphenylethylene instead of stilbene.
The review elaborates on the necessity of the stability of a catalyst, particularly for recyclable catalytic systems. Use of bidentate phosphines ligands are the better option for this, where use of excess ligand is not necessary to make stable complexes, and a ratio of 1:1 leading to (L-L)Pd complex is effective for more stable catalyst and thus leading to higher TONs.
The problem of catalyst deactivation, various solvent systems such as aqueous, molten salts, fluorous, supercritical, subcritical fluids, Heck reaction under pressure and microwave conditions are also discussed. It has been seen that high pressure has a beneficial effect on Heck reactions where oxidative addition and migratory insertions steps of the Heck cycles have a negative activation volume and thus are likely to be accelerated by pressure. However, the PdH elimination can be retarded by high pressure, leading to a change of the product distribution while potentially extending the lifetime of palladium catalyst. For microwave assisted Heck reaction, very fast heating by means of microwaves lead to shortening of the reaction time, while the yields and selectivity do not greatly differ when compared with the same reactions carried out using conventional heating.
Many examples of recyclable (phase-separation) catalysis of liquid-liquid and solid-liquid systems are discussed in the review. Heck chemistry with less usual leaving groups like diazonium salts, thallium(III) and lead(IV) derivatives and acid chlorides and anhydrides are given that provides an alternative leaving groups so as to have more reactive substrates and milder procedures. A section on reactions using metals other than palladium like Cu, Ni, Co, Rh, Ir is present in review that says ‘though none of them can rival palladium in synthetic versatility, some features may complement Heck chemistry to provide either cheaper catalysts or catalysts capable of effective processing of some specific substrates’.
A number of simple N-heterocyclic carbene (NHC) palladium-based complexes have emerged as effective catalysts for a variety of cross-coupling reactions and have been proved to be excellent substitutes for phosphines ligands in homogeneous catalysis in a wide range of catalytic processes. Their efficiency is not limited to their binding ability to any transition metal as they also bind to main group elements such as beryllium, sulphur, and iodine. Because of their such specific coordination chemistry, they both stabilize and activate metal centres in quite different key catalytic steps of organic syntheses.
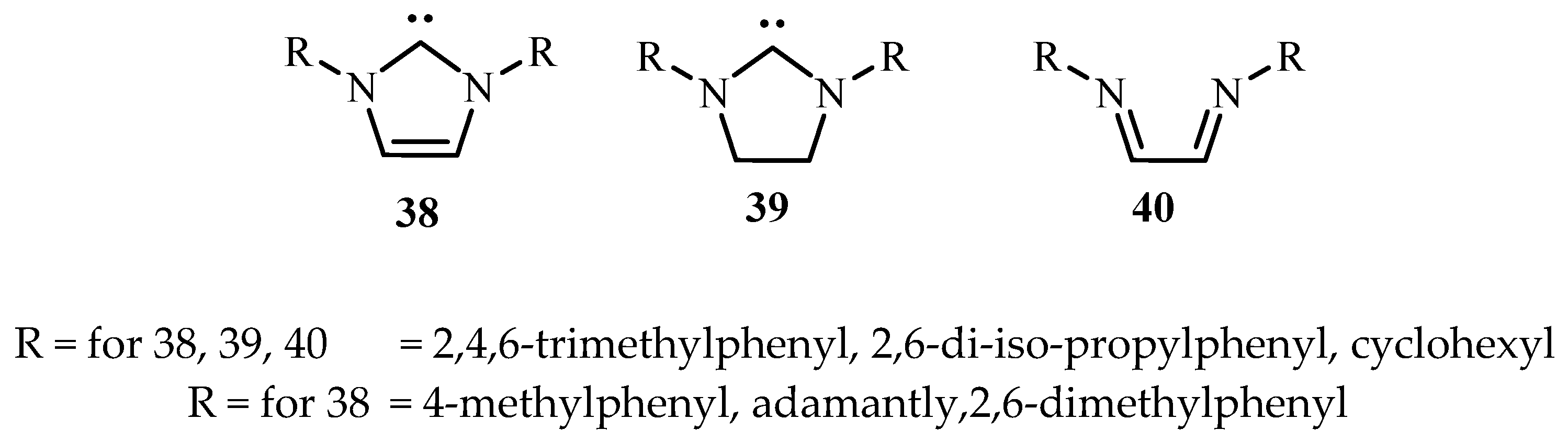
Hillier et al. [
17] have reviewed the catalytic cross-coupling reactions mediated by palladium with nucleophilic NHC as ancillary ligands of diazabutadienes
38–
40 (
Figure 6) mostly based on their own work.
On application of these catalysts to Heck reactions, excellent yields except few were seen. In all cases, the trans products were selectively obtained. However, no activity was observed for aryl chloride substrates. It was also observed that the reactions involving the less reactive aryl halides require bulky electron-donating phosphines. Even excess phosphine and higher Pd loading is required as they are prone to decomposition under Heck conditions increasing the cost of large-scale processes.
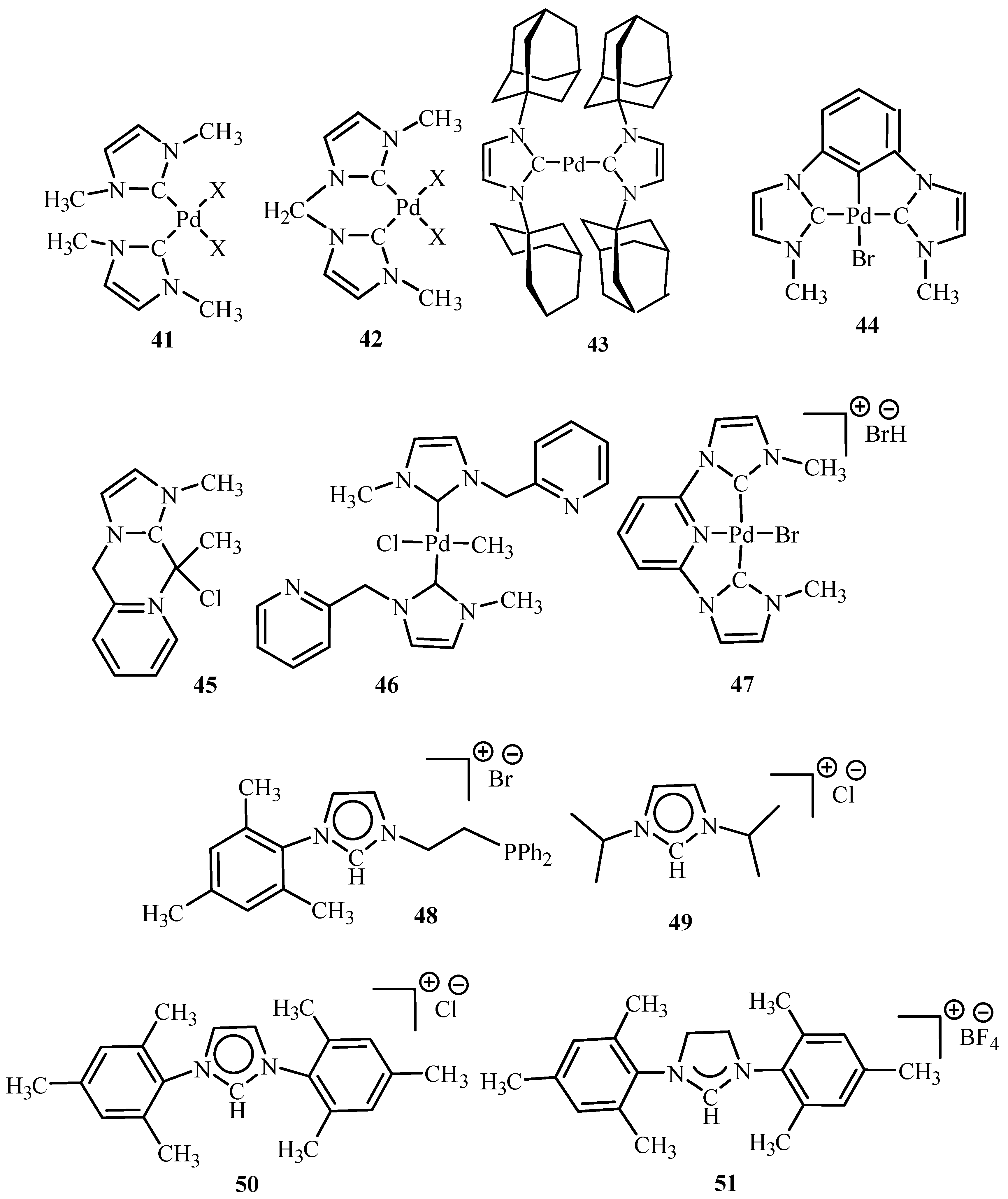
Herrmann [
18] has reviewed N-heterocyclic carbenes as ligands where ligands and metal complexes
41–
51 (
Figure 7) are found to be active towards Heck reaction giving TONs up to 1.7 × 10
6. These ligands are preferred mainly for the reason that they not only bind to transition metal but also to main group elements. They are robust and found to have high thermal and hydrolytic durability, easy accessibility and require lower amount of loading. They could be derivatized in future to have water-soluble catalysts (two-phase catalysis), immobilization, and in chiral modifications. However, it was observed that it is necessary to have bulky NHC ligands for successful reactions with catalysts made of these ligands.
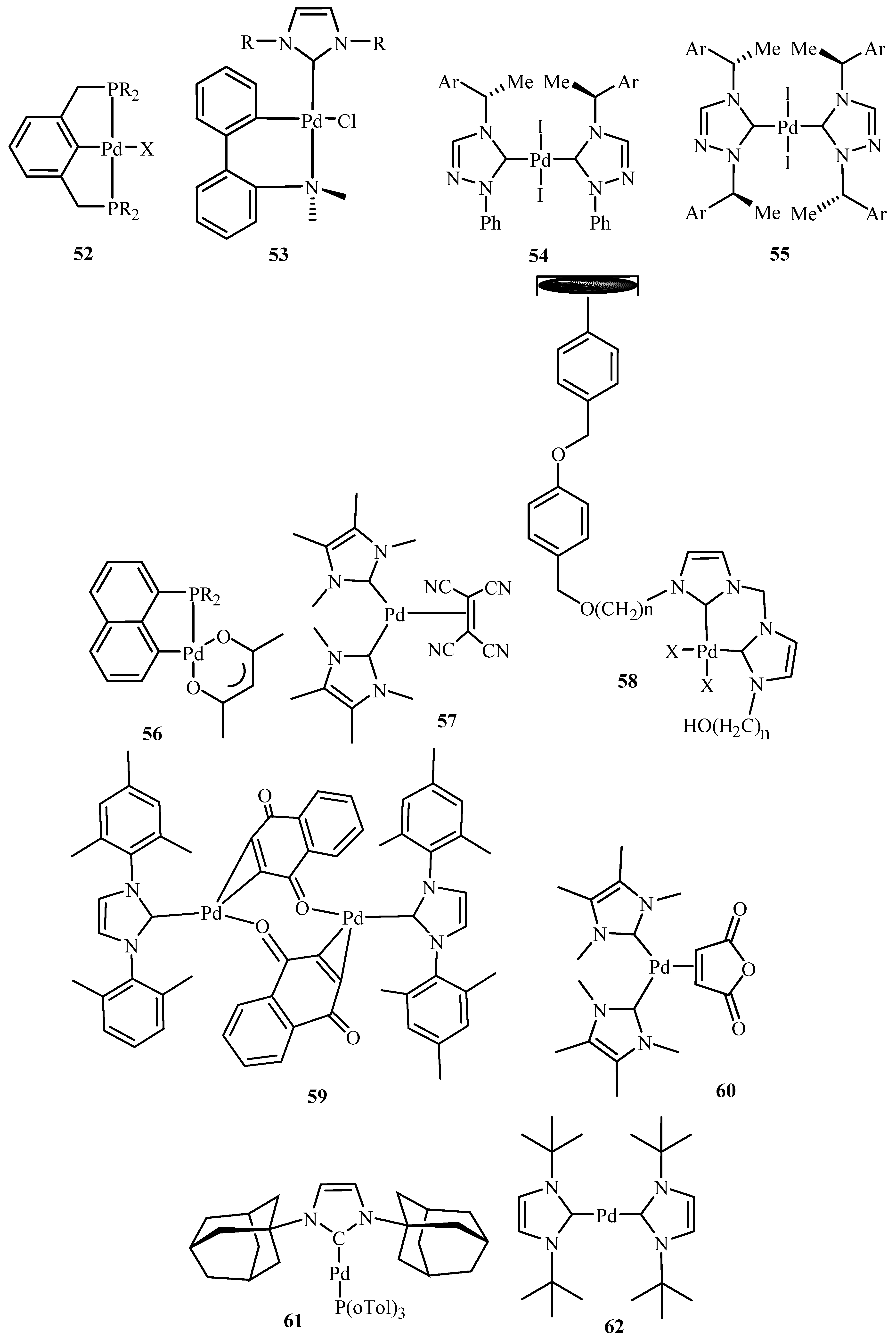
Herrmann et al. [
19] presents an eloquent summary about catalytic applications of palladium complexes with phosphorus ligands containing a metallated sp
3-carbon centre (palladacycles)
36,
52 and with N-heterocyclic carbene ligands
41–
46 and
53–
62 (
Figure 8) for C-C and C-N coupling reactions of aryl halides. The activity of
36 was found to get increased to TONs of up to 4 × 10
4 after addition of tetrabutyl ammonium bromide in the reaction of aryl chlorides like 4-chloroacetophenone with n-butyl acrylate under standard conditions. Recycling of this catalyst could be achieved by using non-aqueous ionic liquids (NAILs) as media.
A number of evidences are given for mechanistic discussions and about their role in the catalytic cycle. The catalytic activity of these complexes strongly depends on the steric bulk of the NHC ligand. Structural versatility is a great advantage of N-heterocyclic carbenes where chirality, functionalization, immobilisation and chelate effects can be achieved by easy means. Few complexes contain both NHC and phosphine ligands leading to combine the advantages of stability of bis(carbene) complexes with the good activity of phosphine complexes in C-C coupling reactions.
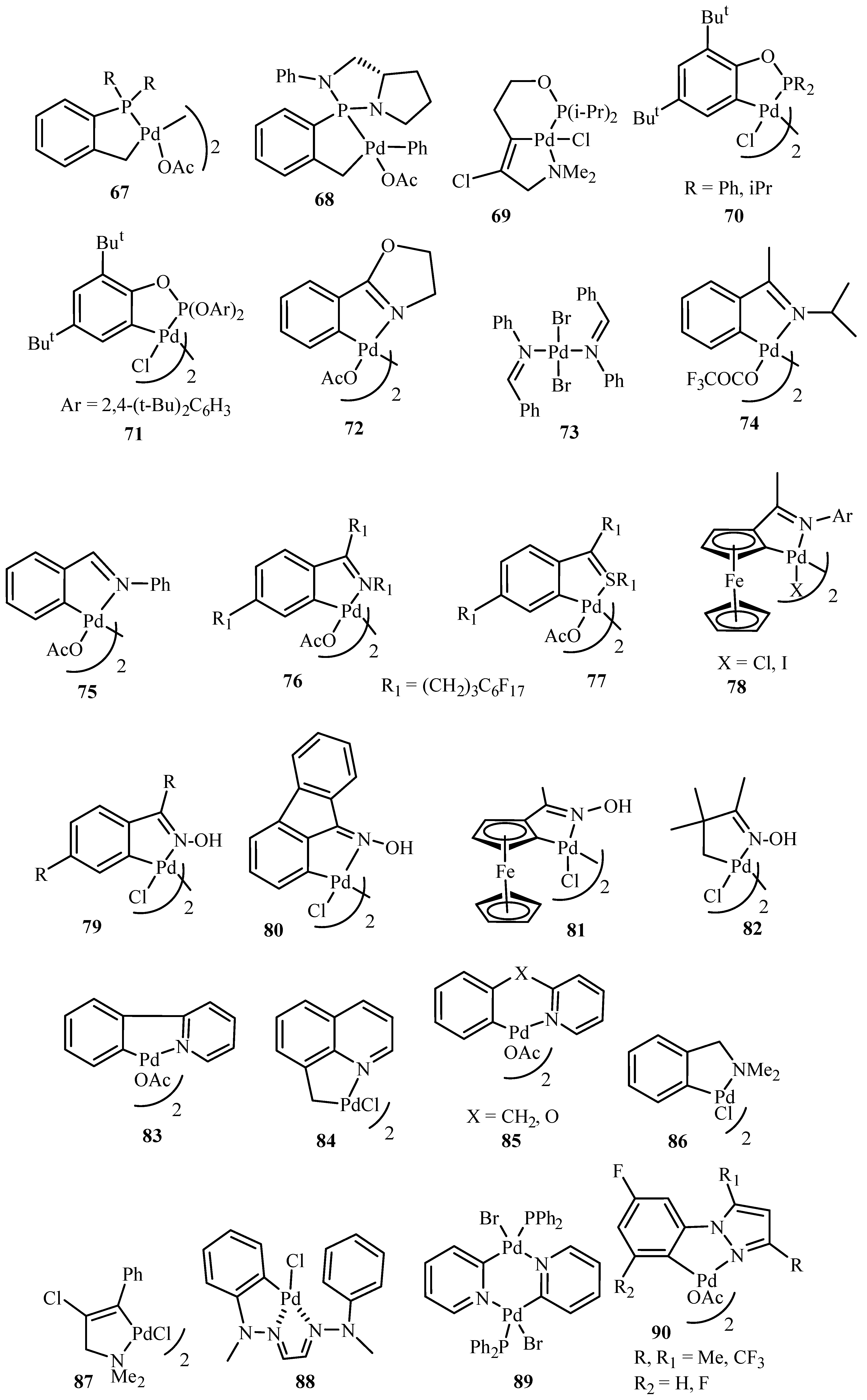
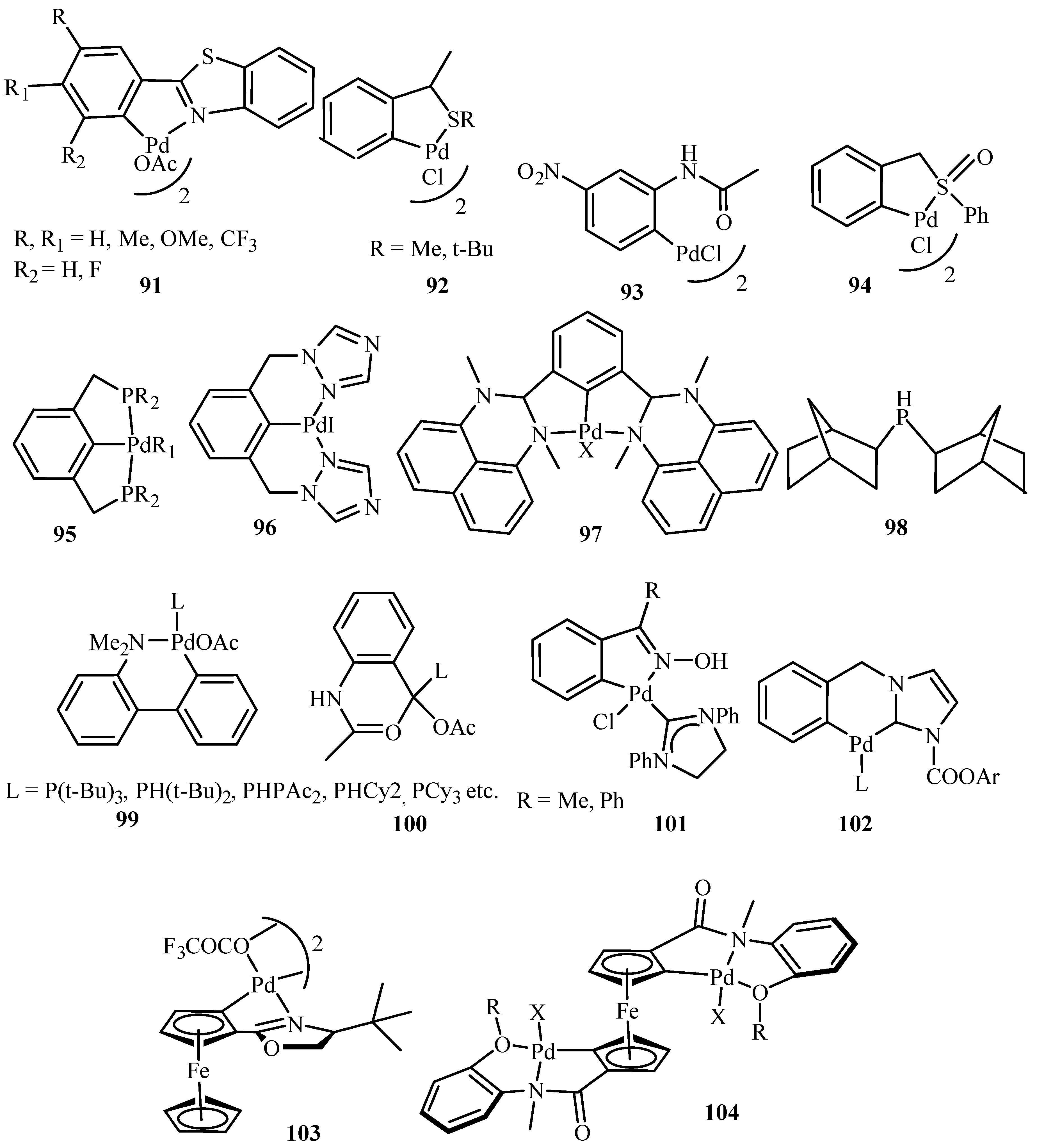
Beletskaya and Cheprakov [
20] have given a critical survey on the application of palladacycles as catalysts for cross-coupling and similar reactions where the advantages and limitations of palladacycle catalysts are discussed. The advantages being the slow release of Pd(0) that helps to suppress unwanted processes of nucleation and growth of large inactive Pd metal particles. Bulky ligands with electron-rich phosphines or heterocyclic carbenes are essential and should be used for desired selectivity. However, the phosphine-free chemistry has limited scope. These ligands can be combined with palladacycles into hybrid catalysts, which retain the advantages of both. Such complexes are usually not more active than non-palladacyclic complexes with the same ligands. A review of various palladacycles with subclass of phosphine-free catalysts such as phosphine-derived palladacycles
36,
56,
63–
68, phosphite palladacycles
69–
71, CN-palladacycles including imine palladacycles
72–
78, oxime palladacycles
79–
82 and some miscellaneous CN-
83–
91, CS- and CO-palladacycles
92–
94, pincer palladacycles
95–
97, hybrid palladacyclic catalysts
98–
102 and couple of palladacycles as structurally defined catalysts
103,
104 (
Figure 9) is taken. It was seen that in most of the cases palladacycles serve as a source of highly active zero-valent palladium species.
Pincer catalysts are bis-chelated palladacycles of XCX (X = P, N, S) type. They are extremely stable. It was observed that using pincer catalysts, electron rich phosphine or NHC ligands, the Heck reaction can be carried out with although cheap but otherwise unreactive, chloroarenes. However, based on a survey over the application of palladacycles in catalysis, it was stated the initial promises have not been fulfilled. The catalysts, often announced as outstanding because of very high catalytic activity in several test reactions, very rarely find applications in preparative chemistry. Neither enantioselectivity nor recyclability has been realized. Dozens of palladacycles of all imaginable classes have been studied in various cross-coupling reactions, but none appeared to be the well-defined catalyst, as was thought earlier.
A review by Zafar et al. [
21] is about the progress of palladium compounds as a catalyst for Heck-Mizoroki and Suzuki-Miyaura coupling reactions. Some synthesized palladium compounds and their progress in terms of ligand modification and other associated parameters up to early 2014 for Heck-Mizoroki and Suzuki-Miyaura coupling reactions are summarized in it. It was observed that the electron donating ligands such as carbenes, phosphines and nitrogen donor ligands can increase the activity, selectivity and stability of its catalysts. Palladium nanoparticles and palladacycle can also act as precatalysts for Heck and Suzuki coupling reactions. The technological hurdles in using homogeneous catalysts are minimized by putting the catalyst on a polymer support.
2.2.2. Homogeneous Catalysis
Quest for High TON
Herrmann et al. [
22] have reviewed applications of metal complexes
36,
45,
52,
56,
105 and
106 (
Figure 10) in Heck type reactions where detailed information about developments in palladium catalytic systems and their successful approach towards activation of less reactive substrates like aryl chlorides is mentioned. Palladacycles are found to be active against a broad spectrum of reactions and have advantages like active towards more economic aryl halides, high activity at low palladium:ligand ratio (1:1) and improved thermal stability and life-time in solution. The possible mechanisms involving Pd(0)/Pd(II) or Pd(II)/Pd(IV) catalytic cycles has also been reviewed for this class of catalyst. The mechanism of Heck type reactions catalysed by palladacycles is thought to involve active Pd(0) species however the possibility of a Pd(II)/Pd(IV) mechanism, working in competition is also not ruled out. In fact, it is stated that, Pd(II):Pd(IV) could only be a side mechanism which cannot solely be responsible for the high turnovers.
Farina [
23] has discussed the problems associated like scope, purity, impurity profile, throughput and time for developing high-turnover catalysts for the cross-coupling and Heck reactions. Numerous examples are given to illustrate the developments in the area of palladacycles and coordinatively unsaturated Pd catalysts featuring bulky phosphanes of high denticity like
107–
109 (
Figure 11) including
36,
52,
70,
71,
76,
78,
79,
90,
91,
95 and few more similar compounds.
These palladacycles are reviewed from a mechanistic and synthetic standpoint, and compared with more traditional catalysts obtained from conventional mono- and polydentate N and P-based ligands, as well as Pd catalysts without strong ligands, such as Pd colloids or heterogeneous catalysts and polymer supported catalysts. Carbene ligands, though less documented until then were believed to have potential for high-TON research because they are more robust than most phosphines ligands.
Reactions Involving Aryl Chlorides and Bromides
For Heck reactions, aryl bromides and chlorides are the most desirable substrates being cheaper and more readily available; however, not many are used as substrates as they are lower in reactivity due to their stronger C-X bonds and by far having lower TONs by several orders of magnitude. This section describes few reviews that draw attention to future challenges in this area by highlighting advances concerning Heck reactions using aryl bromides and chlorides. A review by Whitecombe and others [
24] discuss the development of catalytic systems that can activate unreactive aryl halide towards Heck catalysis. The lesser yields are attributed to the poor reactivity of aryl halides due to their C-X bonds strength as well as the higher temperature used to activate aryl bromides and chlorides resulting in the catalyst decomposition where P-C bond cleavage takes place eventually leading to metal precipitation. This is demonstrated by giving a number of examples using monodentate phosphine ligands, or chelating diphosphine, phosphite, phosphonium salt. However, there are examples of successful reactions taking place under similar conditions with palladacycles, N-heterocyclic carbene complexes and pincers (all previously discussed structures) as they are stable at higher temperature.
The reactivity of ArX is found to be somewhat better if electron withdrawing groups are present in arylhalides; however, reactivity decreases if an electron rich substituent is present typically on aryl halide. The mechanistic study discussed indicates that, the prediction of single mechanism is no longer adequate and there are still unknown factors at play in Heck mechanism. The review exhaustively elaborates the mechanism involving classical Pd(0)/Pd(II) cycles and also Pd(II)/Pd(IV) cycle (this is discussed in detail in present review under mechanism section). In addition to these catalytic systems, heterogeneous catalyst and the catalysts using metals other than palladium, like Ni, Cr, Fe and Co, are described where the yield obtained using these metals are reasonable; however, they are used less than Pd. Use of conditions such as molten salts or phosphine free conditions facilitates the product formation at a higher rate.
A review by Littke and Fu [
25] describes the progressive developments in the area of palladium-catalysed couplings of aryl chlorides. It was always observed that the palladium-catalysed coupling processes show poor reactivity towards aryl chlorides, although they are more attractive substrates than the corresponding bromides, iodides, and triflates in terms of cost and availability. Traditional palladium triarylphosphane catalysts are effective only for the coupling of certain activated aryl chlorides (for example, heteroaryl chlorides and substrates bearing electron-withdrawing groups), but not for aryl chlorides in general. However, catalysts using bulky, electron-rich phosphine and carbene ligands have proved to be mild and versatile for aryl chloride coupling.
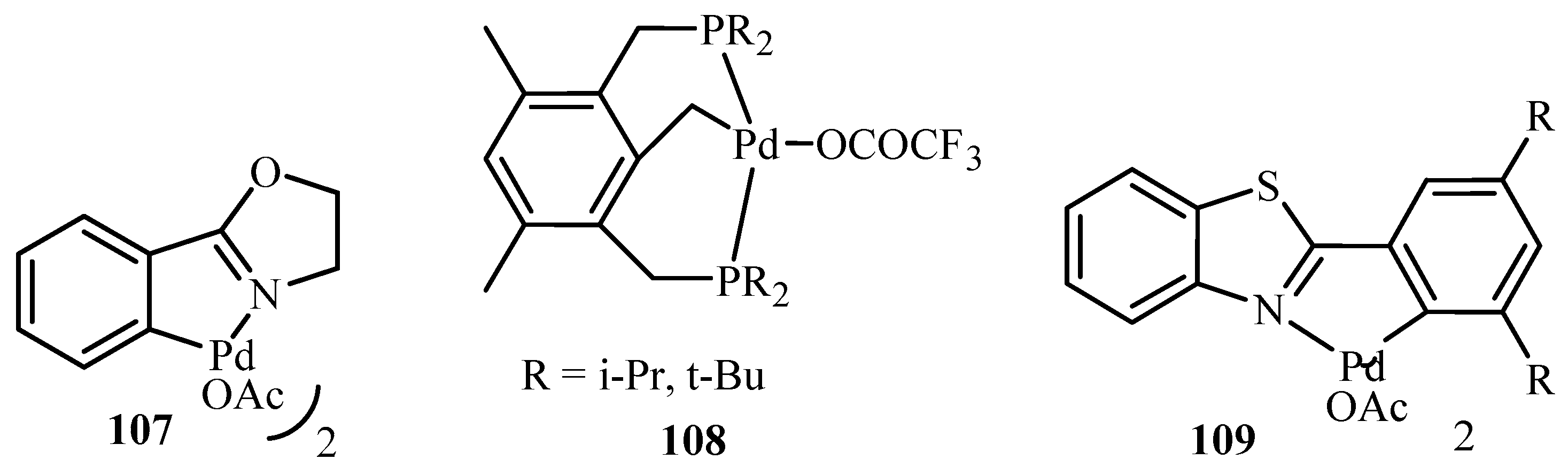
In a similar review by Zapf and Beller [
26], ligands and palladacycles
51,
99 and
110 to
115 (
Figure 12) are reviewed for their activity towards the C-C and C-N coupling reactions of aryl halides, especially aryl chlorides. An important advantage of this class of ligands is their significantly increased stability towards air and moisture. Due to their basicity and steric bulkiness, they constitute excellent ligands for palladium-catalysed coupling reactions. It was found that the palladacycles with high ligand–palladium ratios are suitable for aryl-X activation reactions at elevated temperatures. However, specially designed basic and sterically demanding phosphines show superior performance under milder reaction conditions. In addition, when monocarbene palladium(0) quinone complexes were tested for the Heck reaction using tetra-n-butylammonium bromide as an ionic liquid, a reasonable to good activity for both electron deficient and electron rich aryl chlorides was obtained.
2.2.3. Heterogeneous Catalysis
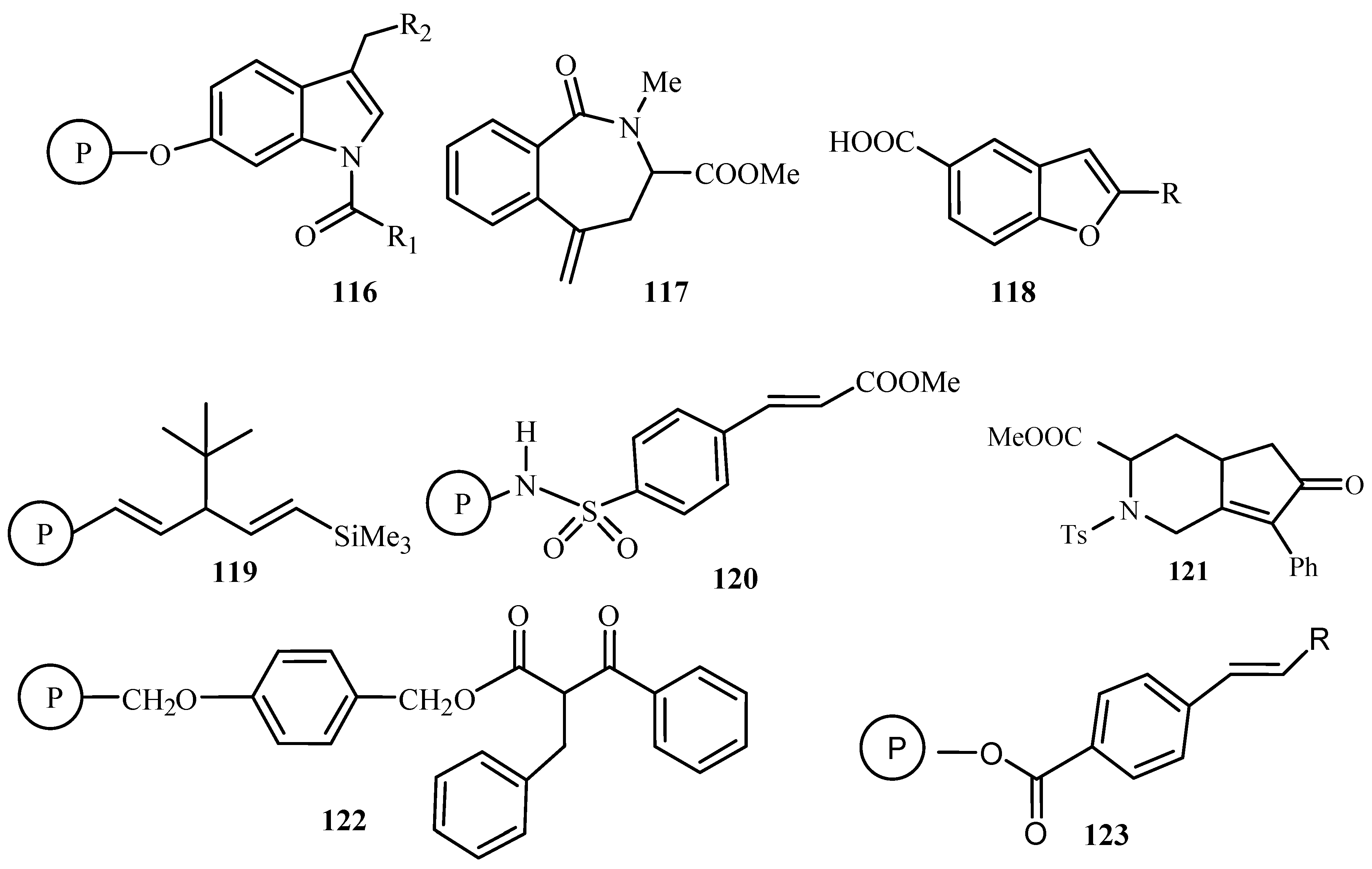
Carbon—carbon bond coupling reactions—Suzuki, Heck and Stille—using catalyst on solid support has been reviewed by Franzén [
27]. Metal-catalysed coupling reactions are reportedly efficient and reliable methods for the introduction of new carbon-carbon bonds onto molecules attached to a solid support. A concise summary of the use of these reactions, in the field of solid phase organic synthesis resulting in small organic molecule libraries is presented. This involves the palladium-catalysed intramolecular Heck reaction for the solid-phase synthesis of indole analogues
116, cyclic tetrapeptide derivative via macrocyclization
117, 2-substituted benzofuran carboxylic acids
118, phenyl acetylene oligomers
119, α,β-unsaturated methyl ester phenyl Sulfonide
120, fused bicyclic amino acid derivatives
121, β-keto esters
122 and in the generation of 1,2-disubstituted olefin libraries
123 (
Figure 13).
A review by Biffis et al. [
28] articulately describes the application of palladium metal catalysts to Heck reaction. The major advantages of supported palladium metal are the simplification of the work-up procedure and the possibility of facile recovery of the precious metal. Initially, the review gives a brief outline of the historical development of heterogeneous catalysis as applied to the Heck reaction followed by the discussion on both supported metal catalysts and stabilized colloidal palladium catalysts. Heterogeneous catalysts supported over different kinds of supports (carbon, inorganic oxides, molecular sieves, polymeric materials, etc.) are reviewed with particular attention to the metal leaching and the nature of catalysis. Ample of examples are given to suggest the presence of leaching, precautions to be taken to prevent leaching or use of methods like re-capture of the leached palladium species with some support. The data provide convincing evidences suggesting the catalytic cycle is sustained mainly by soluble species leached out from the starting solid material after using palladium metal catalysts, either supported or colloidal. The advantage of using heterogeneous catalysis is that it does not necessarily require ligand; the reaction temperature can be high enough for speeding up of the reaction and more of all, the recycling of catalyst is possible although not to a large extent due to extensive metal leaching, metal phase restructuring, structural damage of the support, fouling by carbonaceous deposits, etc., nevertheless systematic tailoring of support can overcome these problems.
Recently, the state of the art, benefits, and challenges of coupling microwave heating with heterogeneous Pd/C catalysis are discussed in the review by Cini et al. [
29]. Microwave dielectric heating allowed a significant acceleration of the C-C coupling reaction rate, shortening the reaction time from hours to minutes. The deactivation of catalyst was not observed when Pd/C-catalysed Mizoroki-Heck reaction was carried out under microwave heating and the recycling of catalyst was also possible. Palladium supported on macroscopic pattern-vertically-aligned carbon nanotubes (Pd/VA-CNTs (carbon nanotubes)) catalyst exhibits higher activity in comparison to Pd supported on simple activated charcoal under the same reaction conditions.
2.2.4. Industrial Catalysis
A review on ‘Palladium-Catalysed C-C Coupling: Then and Now’ by Barnard [
30] focuses on some of the early work in palladium-catalysed C-C bond formation and change in the methodologies during its developments. It talks about how catalyst has been developed from simple Pd compounds (chloride, acetate) with ligands like triphenylphosphine for variety of aryl halides with even sterically restricted partners, up to the development and applicability of stable, bulky and efficient ligands like PCy
3, P(
tBu)
3, biphenyldialkylphosphine, palladacycle, pincer and carbene complexes that can generate highly active species.
The efforts made to develop supported Pd catalysts suitable for recycle, involving both ligandless and anchored ligand systems, the quest for achieving improved conditions allowing high turnover numbers for aryl bromides and reaction of less reactive aryl chlorides are also discussed. It also mentions about two important reviews, i.e., by Corbet and Mignani [
31] on the range of patented cross-coupling technologies and by Yin and Liebscher [
32] on C-C coupling by heterogeneous Pd catalysts.
2.2.5. Nano Catalysis
Narayanan [
33] has highlighted some of the advances in the application of noble metal nanoparticles as catalysts for Suzuki and Heck reactions. Metal nanoparticles suspended in colloidal solutions and those adsorbed onto bulk supports are attractive catalysts for a wide variety of organic and inorganic reactions, compared to bulk catalysts as they have a high surface-to-volume ratio and very active surface atoms. Important aspects such as shape dependence on the catalytic activity, novel types of supported metal nanoparticles as nanocatalysts and the use of bi-metallic, tri-metallic and multi-metallic nanoparticles as catalysts for the Suzuki and Heck cross-coupling reactions are considered.
A review by Cai et al. [
34] provides a summary of bimetallic nanomaterial-catalysed organic transformations and expresses the potential for such bimetallic nanoparticle catalysis to have significant reaction scope, especially with palladium such as magnetically separable “quasi-homogeneous” Pd-Ni nanoalloys, Au-Pd particles confined in silica nanorattles, carbon-supported bimetallic Pd-M (M = Ag, Ni, and Cu) nanoparticles, etc. It was observed that the carbon-supported bimetallic nanoparticles Pd-Cu/C prepared by
γ-irradiation at room temperature exhibits high catalytic efficiency in the Suzuki- and Heck-type coupling reactions.
Baboo [
35] has reviewed the multimetallic nanomaterial based catalysis for the reactions like oxidation, hydrogenation, coupling reactions,
viz. Heck, Suzuki and Sonogoshira, Hydrodechlorination, amidation, reductive amination and hydrogenolysis. Although it is known that nanomaterial based catalysts can easily be separated and reused with same catalytic activities, researchers have paid more attention to the use of multimetallic nano catalysts that show excellent performance than their monometallic nano catalysts. It is mentioned that, despite the great success of bimetallic nanomaterials in terms of their application to oxidation, hydrogenation, and coupling reactions, they have not yet found a wider application in the reactions for the synthesis of complex molecule. The review talks about the use of Pd-based bimetallic nanomaterials and magnetically separable “quasi-homogeneous” Pd-Ni nanoalloys for coupling reactions.
Recently, Labulo et al. [
36] have reviewed the CNTs as efficacious supports for palladium-catalysed carbon–carbon cross-coupling reactions. Such catalysts have shown superior catalytic performance and better recyclability for these reactions as they impart stability to the palladium catalyst. The wide variety of surface functionalization techniques for CNTs that improve their properties as catalyst supports, as well as the methods available for loading the catalyst nanoparticles onto the CNTs with a particular focus on the effect of the solvent, base and catalyst loading has been discussed in detail in this review. It was observed that the yield is largely affected by the choice of solvent and base employed for the catalytic reaction. An improved yield could be achieved with para- and meta-substituted aryl halides and not much improvement is seen with those substituted at the ortho-position. Although this catalyst possesses excellent catalytic activities compared with commercial Pd/C catalysts, it suffers from the problem of leaching.
2.3. Array of Heck Type Reactions
This section deals with the Heck reaction performed with some modifications. Reviews are arranged in the way they fit best into one of the category however there are few with interlinks.
2.3.1. Dehydrogenative/Oxidative Heck Reaction
When Ar-H is used at the place of Ar-X in Heck reaction it is called as dehydrogenative Heck reaction also known as Oxidative Heck reaction or Fujiwara-Moritani [
37] reaction or simply Fujiwara reaction sometimes. The fact is this version of the Mizoroki–Heck reaction was the first catalytic Heck reaction to be discovered in 1968, where the palladium(II)-catalysed arylation of olefins from phenylmercuric chloride, using catalytic amounts of copper(II) chloride assisted by oxygen for the regeneration of palladium(II) was carried out. This reaction is closely related to the classical Mizoroki–Heck reaction, where instead of initiation by oxidative addition process, it follows transmetallation step or a C-H activation step (
Scheme 2).
This is one of the important reactions with respect to atom economy principle since it directly uses the Ar-H compounds thus eliminating additional synthetic step in many total syntheses for halogenations, i.e., in other words, C-H activation of arenes eliminates the need for halogen. The best example of this reaction is the synthesis of benzofuran and dihydrobenzofuran. These structures are important components of numerous biologically active compounds and are preferred to be prepared by dehydrogenative Heck reaction.
Gligorich and Sigman [
38] presented a review on advancements and challenges of palladium(II) catalysed oxidation reactions with molecular oxygen as the sole oxidant, where reviewers have discussed some oxidative Heck reactions. The review paper highlights some of the developments until then in direct molecular oxygen-coupled Pd(II)-catalysed oxidation reactions. Although there are reports with positive results for the development of more efficient ligand-modulated oxidative Heck reactions that can be performed under an air atmosphere at room temperature, the asymmetric oxidative Heck reaction still suffers from many limitations and requires further studies.
Karimi et al. [
39] have reviewed similar types of reactions describing the oxidative Heck reactions of organometallic compounds such as organomercuric acetates, organoboronic acids, organofluorosilicates and arylstannanes. A number of successful examples are given for the intermolecular, intramolecular, nonsymmetrical, asymmetrical oxidative Heck reaction in presence or in absence of air, with ligand-free or ligand based catalysts formed from palladium, polymer supported palladium(II), transition metal and organometallic catalysts other than palladium along with the examples of asymmetric reactions, intermolecular and intramolecular oxidative Heck reactions via C-H activation. However, the substrate scope of these transformations is still extremely limited and there is much room for the development of newer methods that working well under more convenient reaction conditions.
A review by Su and Jiao [
40] reveals the development in the area of palladium catalysed oxidative Heck reactions of alkenes with organometallic compounds, which are effective arylating or akenylating agents. It was observed that the organometallic compounds specially derived from Group III to Group VII like organoboronic. organothallium, organosilicon, organotin, organotin, organophosphorus, organoantimony, organobismuth, organotellurium, hypervalent iodonium salts or simply arenes are efficient substrates for the oxidative Heck reaction. They are found to be remarkably stable and easy to prepare. In addition, various metals other than Pd, including Ru, Rh, Ir and Ni, though less explored for oxidative Heck reaction, are also discussed. The yields of product obtained using them are reasonable up to 72%. The mechanism based on many experimental studies has been reported where the evidence of detection of single charged cationic palladium (II) complexes are given.
A review by Le Bras and Muzart [
41] highlights the same subject particularly with respect to its progress of procedures. Numerous results of stoichiometric as well as catalytic palladium mediated arylations of various arenes and heteroarenes are presented. Most of these reactions use an excess of either the arene or the alkene, often with a relatively high Pd catalyst loading and require a terminal oxidant other than molecular oxygen. This becomes an expensive issue and can be a challenge for applications on large scales and even their compatibility with the atom economy principle. Thus, there is wide scope for researchers to work in this area.
Yet another review by Le Bras and Muzart [
42] covers the palladium-catalysed annelations of internal alkynes through reactions leading to the loss of only two hydrogens from the substrates. This process is explained with the mechanism that involves (i) dual C-H bond activation; (ii) both C-H and N-H bond activation; (iii) successive amino (or oxy) palladation and C-H bond activation; or (iv) C-H bond activation followed by a Heck-type process. Though not much work has been done in this area, such sustainable processes will become valuable tools for the synthesis of diverse carbocycles and heterocycles.
A review by Lee [
43] highlights the use of the oxidative Heck reaction (also referred to as oxidative boron Heck when boron is used in transmetallation reaction (
Scheme 2)) in enantioselective Heck-type couplings. This technique overcomes several limitations of the traditional Pd(0)-catalysed Heck coupling and has subsequently allowed for intermolecular couplings of challenging systems such as cyclic enones, acyclic alkenes, and even site selectively on remote alkenes. This has also enabled enantioselective intermolecular couplings of more challenging systems such as desymmetrisation of quaternary centres, cyclic enones, acyclic alkenes and even site selectively on remote alkenes via a redox–relay coupling. A number of examples have been cited along with probable mechanisms for its justification.
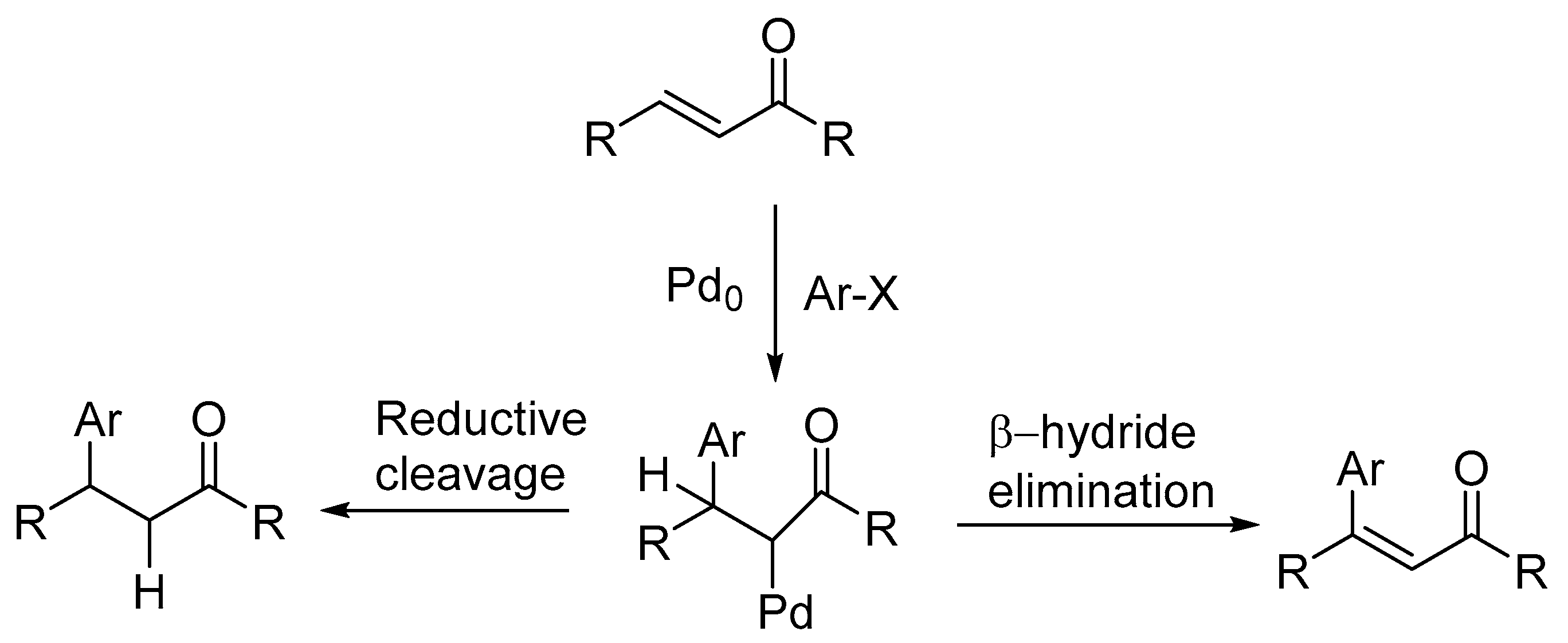
2.3.2. Reductive Heck Reaction
The term reductive Heck or reductive arylation is used when the intermediate forms the conjugate addition product in palladium-catalysed Mizoroki-Heck reactions instead of giving substitution product via β-hydride elimination (
Scheme 3). Reductive Heck product is a regularly observed side product and the extent of its formation in the reaction varies greatly with base, temperature, substrate and solvent.
Xiaomei et al. [
44] have reviewed the progress in reductive Heck reaction, where unsaturated halides react with olefinic compounds at the similar Heck conditions to form the addition products. Discussion on catalytic systems, mechanism, applications and limitation in organic chemistry is given.
2.3.3. Intramolecular Heck Reaction
Intramolecular Heck reaction is generally more efficient than the intermolecular Heck reactions for many reasons like, in the intermolecular Heck reactions, only mono- and disubstituted olefins can participate, while in the intramolecular case tri- and tetrasubstiuted olefins can readily get inserted; regiocontrol of olefin addition is difficult in the intermolecular for electronically neutral olefins whereas the regioselectivity in intramolecular process is governed by ring-size of the newly formed cycle and is generally directed by steric considerations giving highly regioselective couplings. In addition, construction of cyclic compounds containing an endo- or exo-cyclic double bond can efficiently be brought about by using the intramolecular Heck reaction.
A review by Guiry and Kiely [
45] summarizes the development of the intramolecular Heck chemistry and the methodology used for the construction of carbocycles and heterocycles along with the mechanism. The review is mainly focused on the optimization of palladium catalysts derived from various diphosphine and phoshinamine ligands for the preparation of a variety of cyclic products like cis-decalins, hydrindans, indolizidines, diquinanes and the synthesis of quaternary carbon centres. A number of examples of the application of intramolecular Heck cyclization as the key step in the preparation of many complex natural product syntheses are given. Some of their own work on ligand synthesis used for such reactions is also discussed.
A review by Oestreich [
46] summarizes the exciting development of the enantioselective intermolecular Heck reaction (particularly the Heck-Matsuda [
47] reaction that uses arene diazonium salts as an alternative to aryl halides and triflates) of acyclic alkenes and the synthetic utility of enantioselective intermolecular couplings of cyclic alkenes. Many such examples that give very high enantioselectivity for intermolecular Heck reactions are cited.
2.3.4. Asymmetric Heck Reaction
A breakthrough in Heck reaction took place when in 1989 Shibasaki [
48] and Overman [
49] independently reported the first examples of asymmetric Heck reactions. They used chiral ligands BINAP and (
R,
R)-DIOP for such reactions, respectively. A classic example of the intramolecular Heck reaction in action is Overman’s synthesis of (−)-scopadulcic acid A
124 (
Figure 14), where a tandem double Heck cyclisation (6-exo-trig followed by a 5-exo-trig) rapidly accesses the carbon skeleton found in the natural product.
From 2003, the catalytic asymmetric variant of Heck reaction emerged as a reliable method for enantioselective carbon-carbon bond formation. Dounay and Overman [
50] reviewed the application of catalytic asymmetric Heck cyclization in natural product total synthesis for the formation of tertiary and quaternary stereocenters by considering synthesis of some terpenoids (like ernolepin, Oppositol and Prepinnaterpene, Desmethyl-2-methoxycalamenene, Capnellenols, Δ
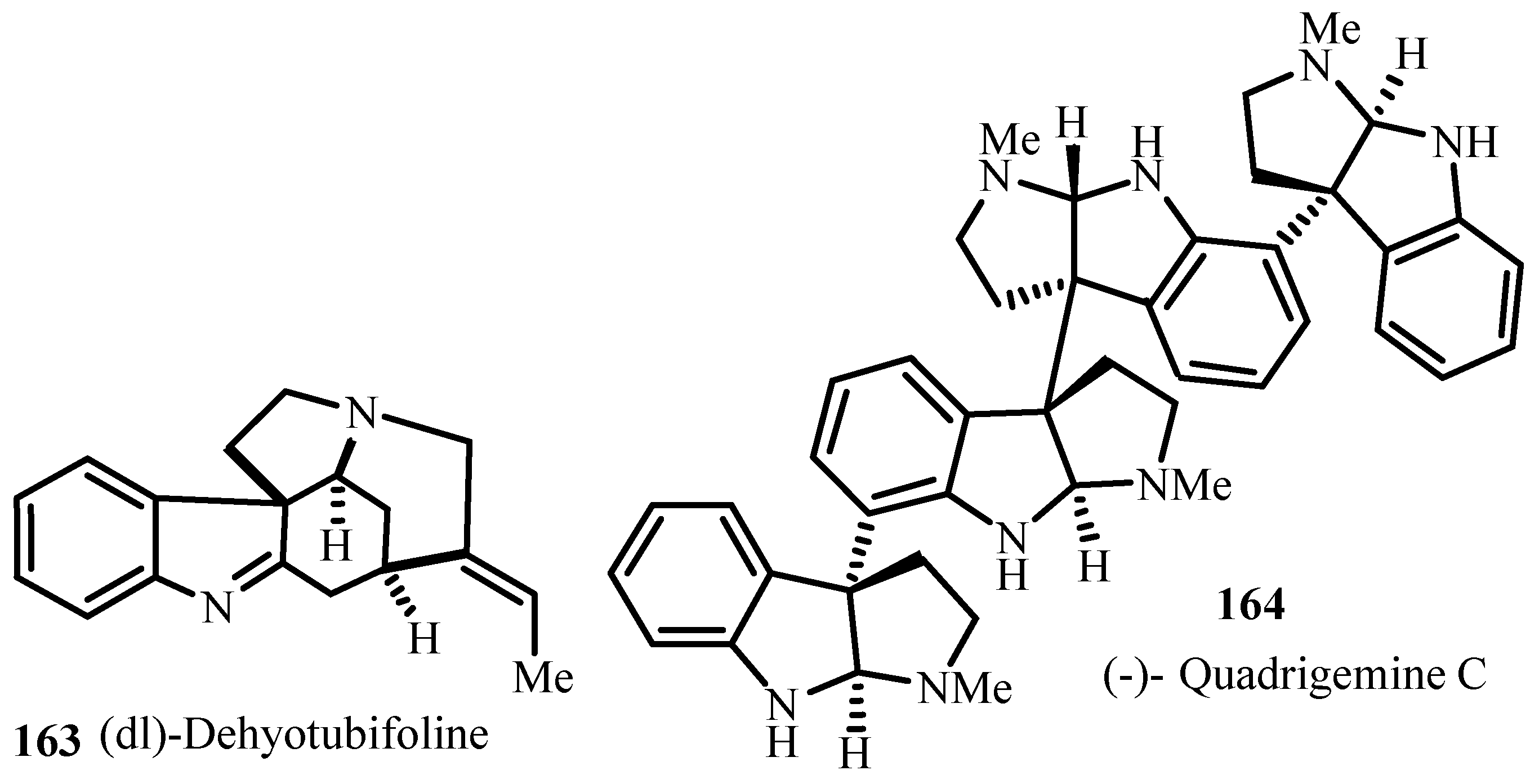
9(12)-Capnellene, Kaurene and Abietic Acid, Retinoids); Alkaloids (like Lentiginosine and Gephyrotoxin 209D, 5-Epiindolizidine 167B and 5E,9Z-indolizidine, 223AB. Physostigmine and Physovenine, Quadrigemine C and Psycholeine, Idiospermuline, Spirotryprostatin, Eptazocine); Polyketides (like Halenaquinone and Halenaquinol, Xestoquinone, Wortmannin). A brief discussion on understanding of the mechanisms of catalytic reactions is provided necessary for the rational development of efficient asymmetric processes.
A review by Diéguez [
51] covers reports on the ligands derived from carbohydrates for asymmetric catalysis until 2003. High enantioselectivities have been observed by using bidentate ligands, usually diphosphines and phosphine-oxazoline ligands. They also have excellent control on selectivity, based on the properties of the ligand. Carbohydrate ligands have proved to be some of the most versatile ligands for enantioselective catalysis. Examples of numerous carbohydrate based ligands are discussed in the review, however, for Pd-catalysed Heck reaction, only the following kinds of carbohydrate derivative ligands
125–
127 (
Figure 15) have been seen to be efficiently applied.
McCartney and Guiry [
52] comprehensively reviewed the asymmetric inter- and intramolecular Heck (
Scheme 4) and related reactions since their original development until 2011 with respect to substrate scope, reactivity, regio- and enantioselectivity. The formation of products is supported by predicting mechanism. The classification is based on the nature of ligands in terms of their denticity, chirality and nature of donor atoms involved so as to understand the continued development of ligand architectural design and their application. The review addresses the significant improvements in reaction times, a disadvantage of Heck reactions performed in classical way, use of microwave-assisted protocols and ligand design. The asymmetric Fujiwara-Moritani and oxidative boron Heck-type reactions and the recent additions to Heck type processes, are also discussed.
2.3.5. Heck Reactions Involving Heteroatom
A review by Daves and Hallberg [
53] highlights typical heteroatom-substituted olefins to organopalladium reagents, where a new carbon-carbon bond is formed via 1,2-addition of an organopalladium species to the often strongly polarized carbon-carbon double bond of a heteroatom substituted olefin. These types of reactions afford versatile and efficient synthetic routes to a wide variety of compounds. Heck and other similar types of reactions involving intermediate palladium complex undergoing 1,2-additions have been discussed. Compounds with heteroatoms O, N, S, P are considered to explore their reactivity towards such addition reaction. A number of examples with cyclic enol ethers, glycals, and cyclic enol ethers derived from carbohydrates, acyclic enol ethers like thioenol ethers, enol carboxylates, enamides and enamines, vinylsilanes and vinylphosphonates are given. Product formation is explained based on well accepted mechanism for Heck reaction involving Pd(0) complex. Increased understanding of the pathways by which these reactions occur, that include delineation of the factors determining reaction regio- and stereochemistry, and control of competing modes of σ-organopalladium adduct decomposition to products, make possible the utilization of these reactions in the synthesis of complex structures.
Bedford [
54] has reviewed the use of palladacyclic catalysts in C-C and C-heteroatom bond-forming reactions. Palladacycles are air and moisture stable inexpensive catalysts, can easily be handled and stored, and act as clean sources of low-coordinate palladium(0). They as pre-catalysts play a significant role in a range of C-C and C-heteroatom bond forming reactions. Activity of the catalysts comprised of P-C palladacyclic, Palladium P,C,P-pincer complexes, L-C palladacyclic, L,L,C- and L,C,L-pincer based (where L = N, S) have been compared for the C-C and C-heteroatom bond-forming reactions including discussion on the likely nature of the true active catalysts produced in situ. These catalysts show the TON ranging from a few thousand up to a few million. Consideration to the Aryl chlorides as substrates and the mechanism involving palladacycle for such reaction is thoroughly discussed. It was found that the complexes
36,
63 and
70 show reasonable activity with some, usually less electronically challenging substrates. The possibility of recyclability of a few catalysts for Heck and other reactions has been discussed. It was predicted previously that when the catalysts are immobilized on solid supports they could work as recyclable catalyst. Polystyrene-immobilized catalyst
74 shows comparable activity to homogeneous analogues in the Heck coupling of iodobenzene with styrene. However, in recycle study the filtrate after reaction and not the recovered catalyst that showed the activity comparable to that obtained in the first run. This activity was concluded to be due to formation of nanoparticulate palladium which is stabilized by an ammonium salt.
2.4. Methodology
Various attempts have been made by researchers to improve the catalyst activity, lowering the cost of catalyst; hence different methodologies are adopted using non-conventional techniques for Heck reactions. Reviews based on these methodologies are described here.
2.4.1. For Improvement in Activity
Use of tetraalkylammonium salts to improve the yields in Heck reactions particularly is considered to be a remarkable contribution in catalysis. The combination of such salts (phase-transfer catalysts) and insoluble bases accelerates the rate of reaction to great extent even at lower reaction temperatures and is commonly known as Jeffery conditions. A review of these salts in Heck type reactions is taken by Jeffery [
55]. It was seen that, under appropriate conditions, tetraalkylammonium hydrogensulfate can just be as efficient as tetraalkylammonium chloride or bromide for facilitating Heck-type reactions. Thus, an appropriate selection of the catalyst system (Pd/Base/QX) can allow this type of reactions to be efficiently realized at will, in a strictly anhydrous medium or in a water-organic solvent mixture or in water alone.
2.4.2. For Lowering the Cost
Tucker and de Vries [
56] describe their own efforts in the area of palladium- and nickel-catalysed aromatic substitution carbon-carbon bond formation reactions in the review titled ‘homogeneous catalysis for the production of fine chemicals’. The main focus of this review is on low cost and low waste production methods. A number of examples are discussed to prove methodology for lowering production cost such as reducing the amount of catalyst, eliminating use of ligands or use of cheaper phosphite or phosphoramidite ligands, carrying out reactions at lower temperatures, replacing palladium by nickel, replacing aryl bromides or iodides with the cheaper chlorides and simplified work-up. For waste free production methods, use of aromatic anhydrides as aryl donor is suggested. Methods for recycling of palladium in ligand-free Heck and Suzuki reactions is described that involves treatment of palladium black, precipitating at the end of the reaction, with a small excess of I
2 prior to its re-use in the next run.
Typically, the Heck reactions are catalysed by palladium, a precious metal. However, in many cases low cost transition metals are found to play a similar role which is reviewed by Wang and Yang [
57]. This review focuses on low-cost transition metal catalysed Heck-type reactions. The cited examples indicate that some low-cost transition metals like Ni, Co, Cu, Fe are active for Heck-type reactions. Ni is found to give best performance among them; in fact, sometimes the results with Ni are comparable to that with Pd. Co and Cu exhibit outstanding activity to alkyl electrophile involved Heck- type reactions. This is attributed to their abilities to generate alkyl radical. It was also observed in a few cases that under certain conditions these low-cost transition metals may show unique catalytic properties, which are absent in Pd-mediated systems. Two mechanisms have been predicted for Heck-type reactions using these transition metals, cationic mechanism and radical mechanism based on the reacting species. Phenyl or benzyl halides predominantly take the cationic mechanism, whereas alkyl halides usually follow the radical mechanism, especially in Co- and Cu- catalysed systems because Co and Cu have good capacity of producing radical from alkyl halides. However, the investigations on low-cost transition metals catalysed Heck-type reactions highlight the extension of the substrate scope and draw a little attention to the development of catalysts, ligands and solvents. Hence, more study is needed in this area.
2.4.3. Non-Conventional Methodologies
Beletskaya et al. [
58] have published a critical overview on unconventional methodologies for transition-metal catalysed Heck reaction highlighting the efforts and interest in developing more efficient processes according to the new requirements of chemistry. A vast array of non-conventional methodologies is described considering different parameters involved in the reaction like substrates, catalytic system, solvent, reaction conditions, or work-up. However, it is also stated that, although large numbers of interesting methodologies are available from an academic point of view, they are useless from a practical point of view. Hence, there is a need for reconsideration of a few points while performing Heck reactions at the industrial level such as:
Most of the methodologies deal with the simpler reactions between the reactive substrates like aryl iodides or activated aryl bromides and acrylates in contrast to the more desirable, but less reactive aryl chlorides or other olefinic substrates such as electron-rich olefins.
More attention should be paid at the workup and separation of by-products formed during achieving a high regio- and stereoselective products.
Whenever possible, commercially available starting materials, reagents, ligands, catalysts, or solvents must be used and proper time should be given to experimental work needed to prepare the different reaction components.
The catalyst must be recyclable and/or display high TONs.
Heterogeneous catalysis or a use of ligandless catalysts, recoverable ligands or stabilized nanoparticles are preferred for better recovery and lower cost provided metal leaching is prevented.
An ideal recyclable catalyst is the one in which filtration of the reaction mixture produces a catalytically active solid and an inactive filtrate.
Use of high temperature with some stable catalysts may prove to be unfavourable for the selectivity of the product.
The expensive equipment or reaction medium utilized in some methodologies cannot compensate for the little or no improvement observed in many cases with respect to the conventional methodologies; in addition, the application of these methodologies is normally restricted to a small scale.
Reproducibility, atom-economy, low-cost, scalable and practical procedures, are needed to extend the methodologies from the academic laboratory to the industrial plant.
Further research must be undertaken in order to clarify the reaction mechanisms involved in the different processes, which remain unclear in most cases; it is crucial to have a better knowledge of the nature and properties of the real catalytic species in order to improve any given reaction.
2.5. Selectivity
Under the section Asymmetric Heck reaction (
Section 2,
Section 3 and
Section 4) reviews dealing with the generation of asymmetric centre by means of Heck reactions were summarized whereas under this section the progress of ligands and catalysts for the development of various regioselective and enantioselective transformations via Heck reactions are discussed.
A review by Tietze et al. [
59] on ‘enantioselective palladium catalysed transformation’ discusses many other important organic reactions including Heck reaction where enantioselectivity in intramolecular and intermolecular Heck reaction is discussed with lot of examples collected right from its first example of such kind by Shibasaki [
48] and Overman [
49]. A number of examples with new improved ligands for development of various enantioselective transformations has been discussed aiming at higher (preferably over 95%) ee values. The review asserts that there is no field where enantioselective Pd catalysis cannot be employed. However, the disadvantage of such catalysis is the high price of Pd and the usually small turnover numbers, making the processes too expensive for industrial use. Nevertheless, novel chiral Pd catalysts resulting in high turnover number can be synthesized to overcome this issue in addition to a broad range of enantioselective transformations suitable for the chemical industry.
Almost at the same time, Shibasaki et al. [
60] reviewed a similar topic particularly aiming at the asymmetric Heck reaction. Since a variety of carbocyclic, heterocyclic and spirocyclic systems can be constructed, the asymmetric Heck reaction becomes a powerful method for the synthesis of both tertiary and quaternary chiral carbon centres, with an enantiomeric excess often in the range of 80% to 99%. The scope of the reaction with respect to the product alkene isomerization was limited due to regioselectivity, and was predicted to be solved by development of new generation of ligands dissociating more rapidly from the products, thus improving both enantio- and regiocontrol.
Oestreich [
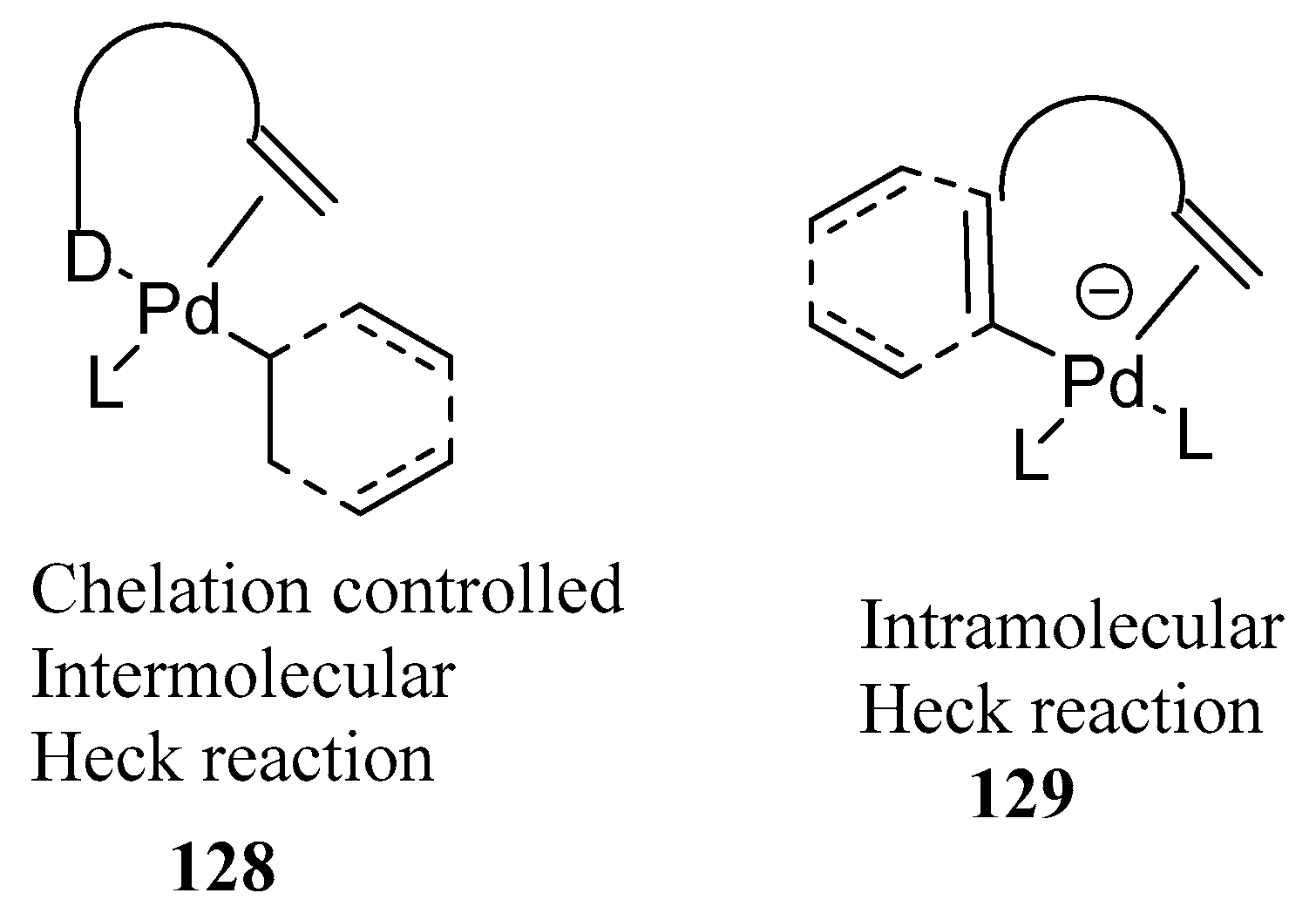
61] describes the evolution of inter- as well as intramolecular Heck reactions from regio- to diastereo- and finally to enantioselective transformations with a special reference to heteroatom-directed Heck reactions in his review. The concept of “Chelation Control”
128,
129 (
Figure 16) controls regio- and stereoselectivity with the aid of attractive interactions between substrate and reagent/catalyst is discussed.
Several examples of removable catalyst-directing groups developed for the preferential regioselective intermolecular arylation of alkenes are given such as amino-directed intermolecular Heck reaction of vinyl ethers. Mono- versus bidentate phosphines influence a regiochemical switch based on the bite angle and the mechanistic rationale for inverted regioselectivity is also discussed. Review covers the syntheses of stereodefined, multi-arylated alkenes, the diastereoselective construction of tertiary and quaternary carbon centres, and also the combination of substrate with catalyst-control in an enantioselective transformation. Many examples have been discussed to show the neighbouring-group effects playing important role in Heck chemistry and expect few more discoveries in this field. The example of a substrate-controlled enantioselective reaction illustrates that the enantioselection is sometimes discriminated not only by the chiral reagent but also by a suitably located donor.
2.6. Aqueous Media
Nowadays, use of water has become increasingly popular for fine synthetic chemistry in industry for the reasons: water is non-toxic, nonflammable, inexpensive, and environmentally friendly solvent and there by use of organic solvent can be avoided. In addition, one of the major drawbacks of homogeneous metal catalysis lies in the separation of the reaction product from the catalyst and requires costly procedures. The concept of transition metal catalysis in water is used where the catalyst is easily recovered by separation of the aqueous and organic phase if a biphasic system is used. Its popularity has been increased since the development of the Ruhrchemie–Rhone Poulenc process using a modified water-soluble rhodium complex in the hydroformylation methodology. However, the limitations of aqueous phase catalysis are:
Stability of substrate or product in water.
Partial solubility of substrate in the aqueous phase to avoid mass transfer limitation.
Necessity of preparation of water soluble ligands or dispersing agents to maintain catalyst in aqueous phase.
Challenges for future developments in this area to develop catalysts with scope and activity comparable to the best organic-phase catalyst systems.
Nevertheless, there is still strong interest in developing efficient and recoverable catalysts for use in pharmaceutical and other fine chemical synthetic processes, as can be seen from following reviews.
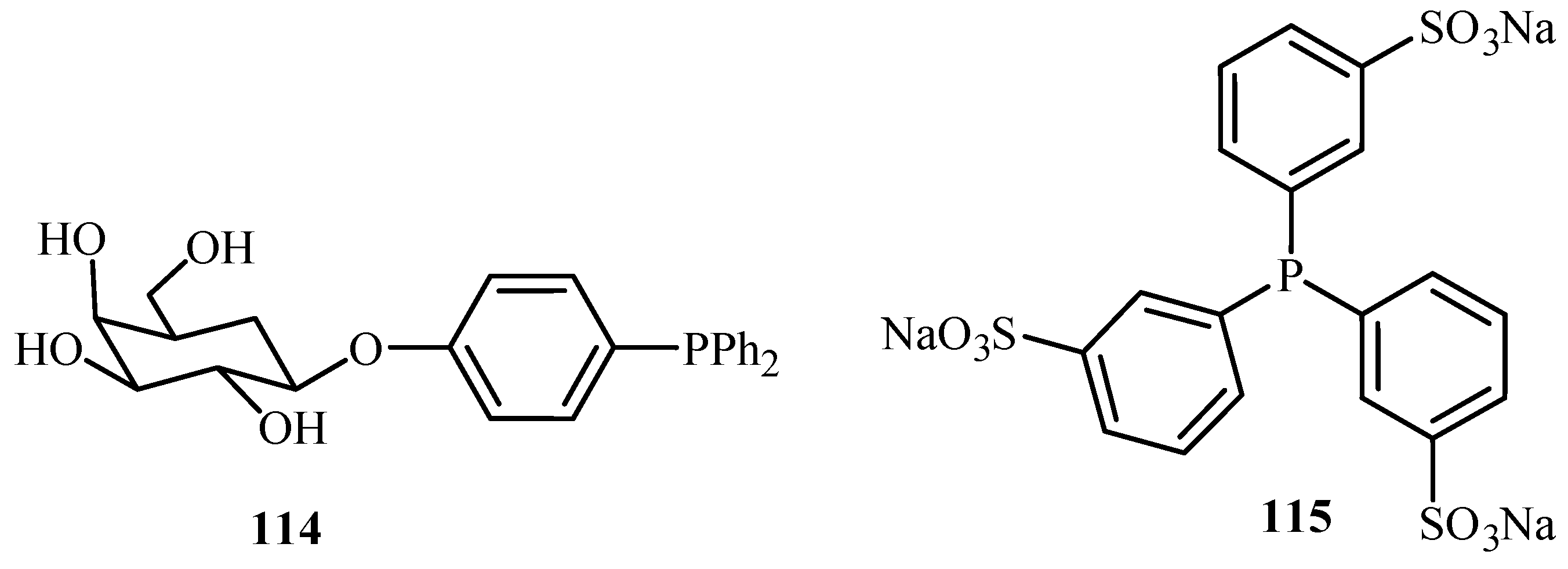
Genet and Savignac [
62] have reviewed the palladium cross-coupling reactions carried out in aqueous medium. Reactions like Heck, Sonogashira, Tsuji-Trost, Suzuki, Stille as well as protecting group chemistry in aqueous media are discussed in the review. Although palladium is known to be unstable in aqueous medium, there are reports of the excellent compatibility of water-soluble palladium catalysts with water- soluble phosphines such as TPPTS (3,3′,3′′-Phosphanetriyltris(benzenesulfonic acid) trisodium salt)
115, TPPMS (Sodium Diphenylphosphinobenzene-3-sulfonate)
130 and salts of acid or amines
131,
132 (
Figure 17), offering new opportunities for such reactions at mild conditions and with new selectivity.
It was seen that the careful selection of reaction conditions, co-solvents and catalysts, is very important for long life catalyst and new selectivities. The advantages of the two-phase aqueous system, i.e., easy separation of the products and recycling the expensive palladium can be obtained by using palladium catalysed reactions with water-soluble phosphines that has increased the potential of modern palladium catalysis.
Li [
63] has reviewed many organic reactions in aqueous media where water serves as a medium for various palladium-catalysed reactions of aryl halides with acrylic acid or acrylonitrile to give the corresponding coupling products in high yields. In addition, reactions at superheated and microwave heating conditions with bulky phosphine ligands along with arenediazonium salts instead of aryl halides in the Heck-type reaction are mentioned. The reason for a high yield of coupling product for reaction involving the use of Pd(OAc)
2 with water-soluble ligand, TPPTS, and formation of complex mixture was attributed to high dielectric constant of water. Examples of many transition metals other than palladium in water have been cited.
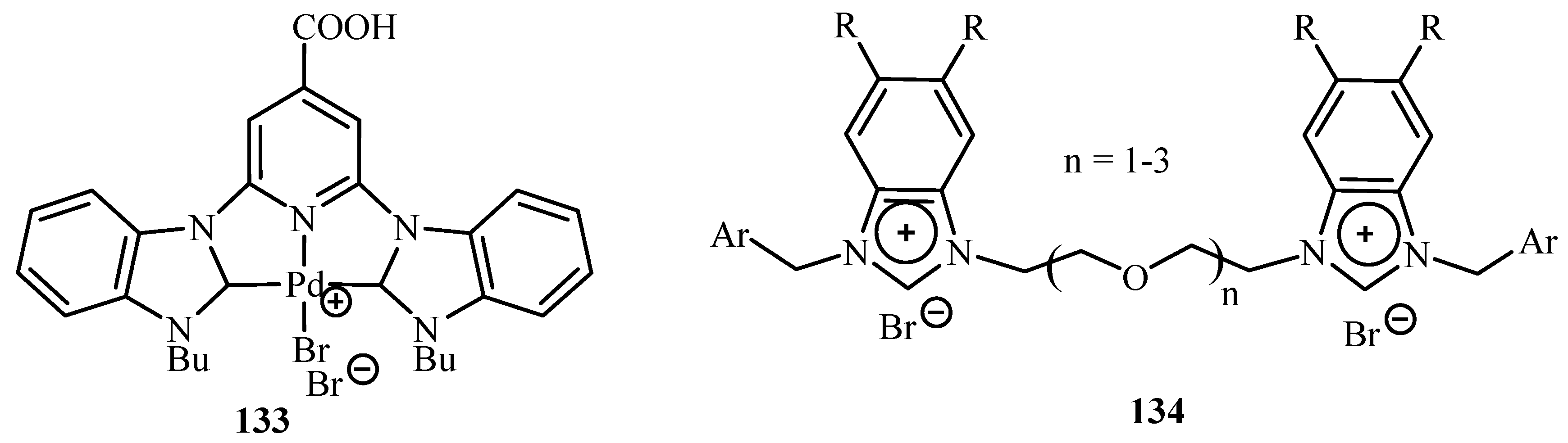
Velazquez and Verpoort [
64] have reviewed the reports on the use of N-heterocyclic carbene transition metal complexes for catalysis in water. The typical phosphine and amine-type ligands can thus be displaced by this type of catalysis because of their higher stability and reactivity. Palladium complex
133 and a complex of ligand
134 (
Figure 18) are used for catalysing the Heck reaction successfully in water.
A review by Hervé and Len [
65] is based on both Heck and Sonogashira cross-coupling of nucleosides following two important aspects of the green chemistry, i.e., use of aqueous medium and no protection/deprotection steps. It focuses on the study of C5-modified pyrimidines and C7-deaza or C8-modified purines where these chemical modifications have been developed using palladium cross-coupling reactions. The review encompasses variations of the starting materials, alkene and alkyne, nature of the solvent, palladium source and ligand at either room temperature or higher temperature. Heck cross-couplings were performed using Na
2PdCl
4 (80 mol %) and Pd(OAc)
2 (5–10 mol %) in the presence of TPPTS as ligand in a mixture of CH
3CN/H
2O and in sole water. Using these procedures, yields up to 98% have been reported.
3. Reviews on Mechanism
The mechanism of Heck reaction is discussed and reviewed by many researchers since 1972 when for the first time it was given in complete detail by Heck and Nolley [
9]. There are articles that argue the case for having a Pd(0)/Pd(II) mechanism while others favour the Pd(II)/Pd(IV) mechanism based on evidential data. Few discuss the specific intermediate formed during a particular mechanism while others demonstrate the presence of different intermediate. This section deals with reviews on such reviews on mechanism of Heck reaction.
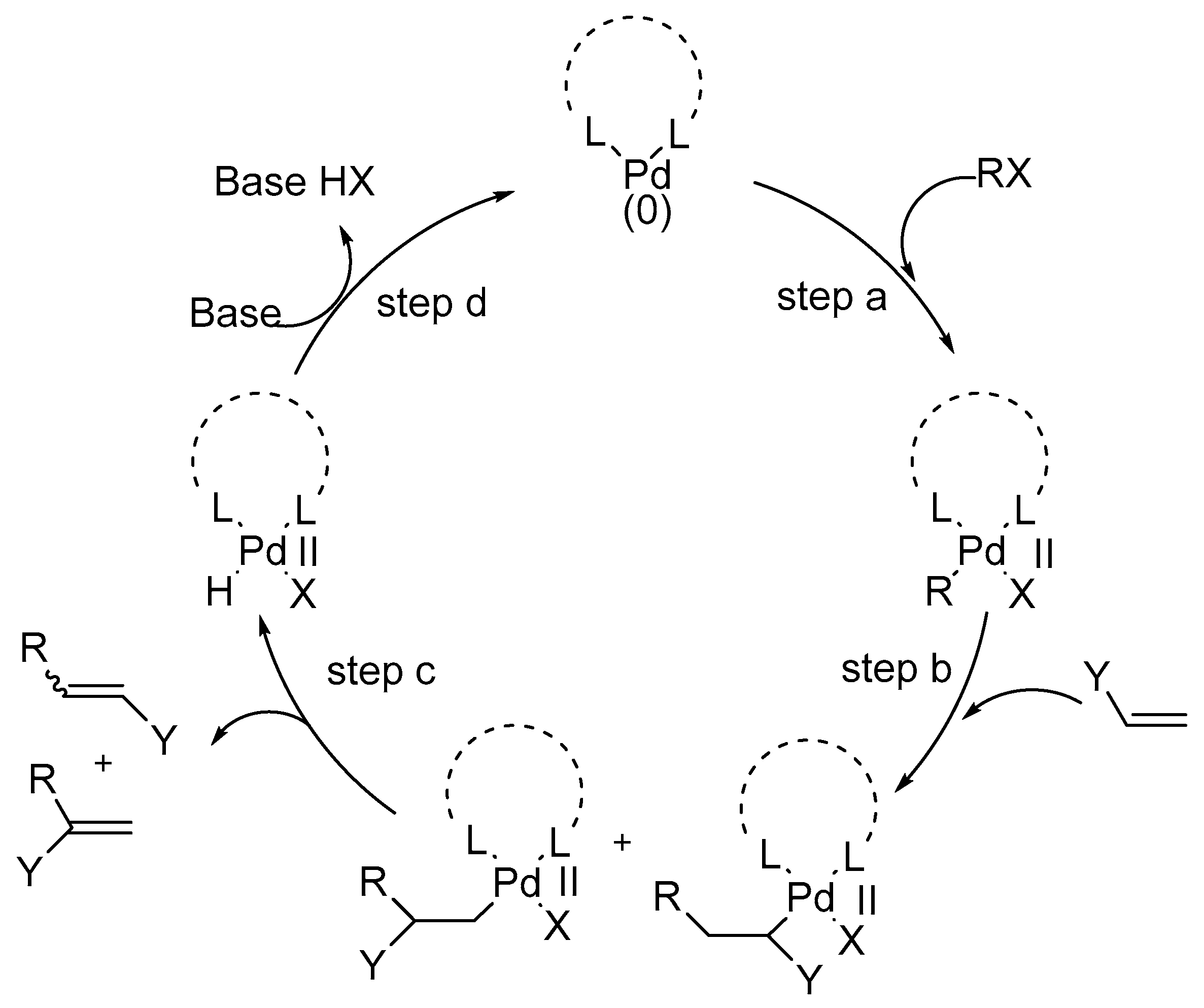
A review by Cabri and Candiani [
66] explains the basis of a common mechanistic hypothesis for the coordination-insertion process of unsaturated systems on palladium(I1) complexes based on the results obtained until then by several research groups on the Heck reaction. All of these results are explained by the commonly accepted mechanism based on Pd(0)/Pd(II) cycle (
Scheme 5).
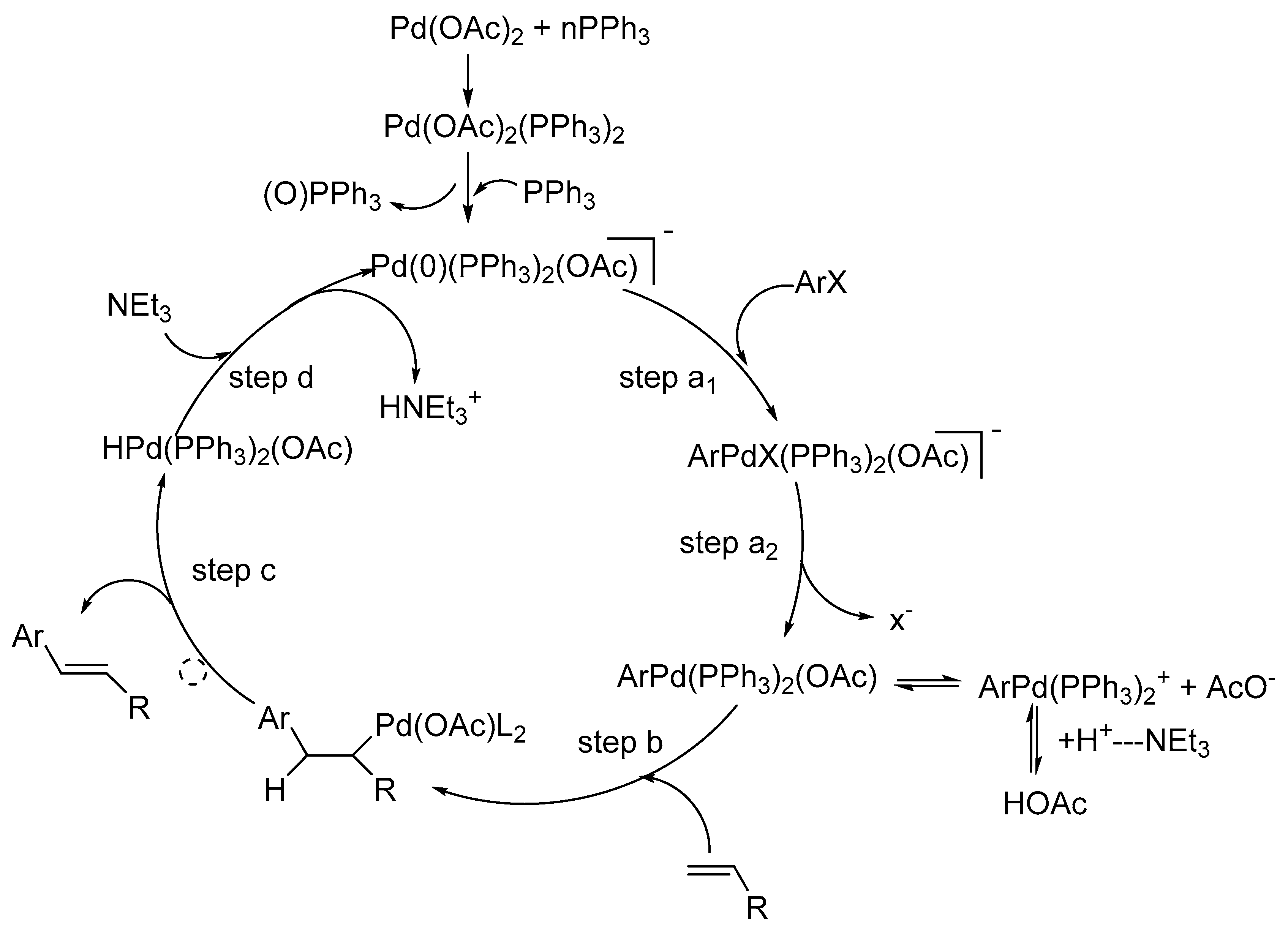
This mechanistic model gives a better understanding of the scope and limitations of the Heck reaction. The steps involved in the mechanism of Heck reaction are:
- step (a)
Oxidative addition
- step (b)
Coordination-insertion
- step (c)
β-Hydride elimination-dissociation
- step (d)
Recycling of the L2Pd(0)
Several factors like leaving groups, neutral ligands, additives (like bases, salts, etc.), and olefin substituents play important roles in overall reaction course via coordination-insertion process as shown in
Scheme 6.
It is essential that the insertion process requires a coplanar assembly of the metal, ethylene, and the hydride and hence the insertion process is stereoselective and occurs in a syn manner. In addition, the energy barrier for the generation of the reactive configuration in a tetracoordinated complex is low with respect to a pentacoordinated one. Therefore, pentacoordinated species are possibly not involved in the coordination process. The β-hydride elimination is also stereoselective and occurs in a syn manner. Its efficiency is related to the dissociation of the olefin from the palladium(I1)-hydride complex. It was also observed that, the presence of a base is necessary in order to transform the L2Pd(H)X into the starting L2Pd(0) complex and complete the catalytic cycle.
The mechanism is also supported by the discussion on the regioselectivity and stereoselectivity of Heck reaction. It was observed that the preferential formation of the branched products in the arylation of heterosubstituted olefins like enol ethers, enol amides, vinyl acetate, allyl alcohols, and homoallyl alcohols was independent of the substituents on the aromatic ring, the reaction temperature, and the solvent and is related only to the coordination-insertion pathway, though the stereoselectivity was dependent on the added base. In the intramolecular asymmetric Heck reaction, sometimes the enantioselectivity was related to the geometry of the substrate olefin.
A review by Crisp [
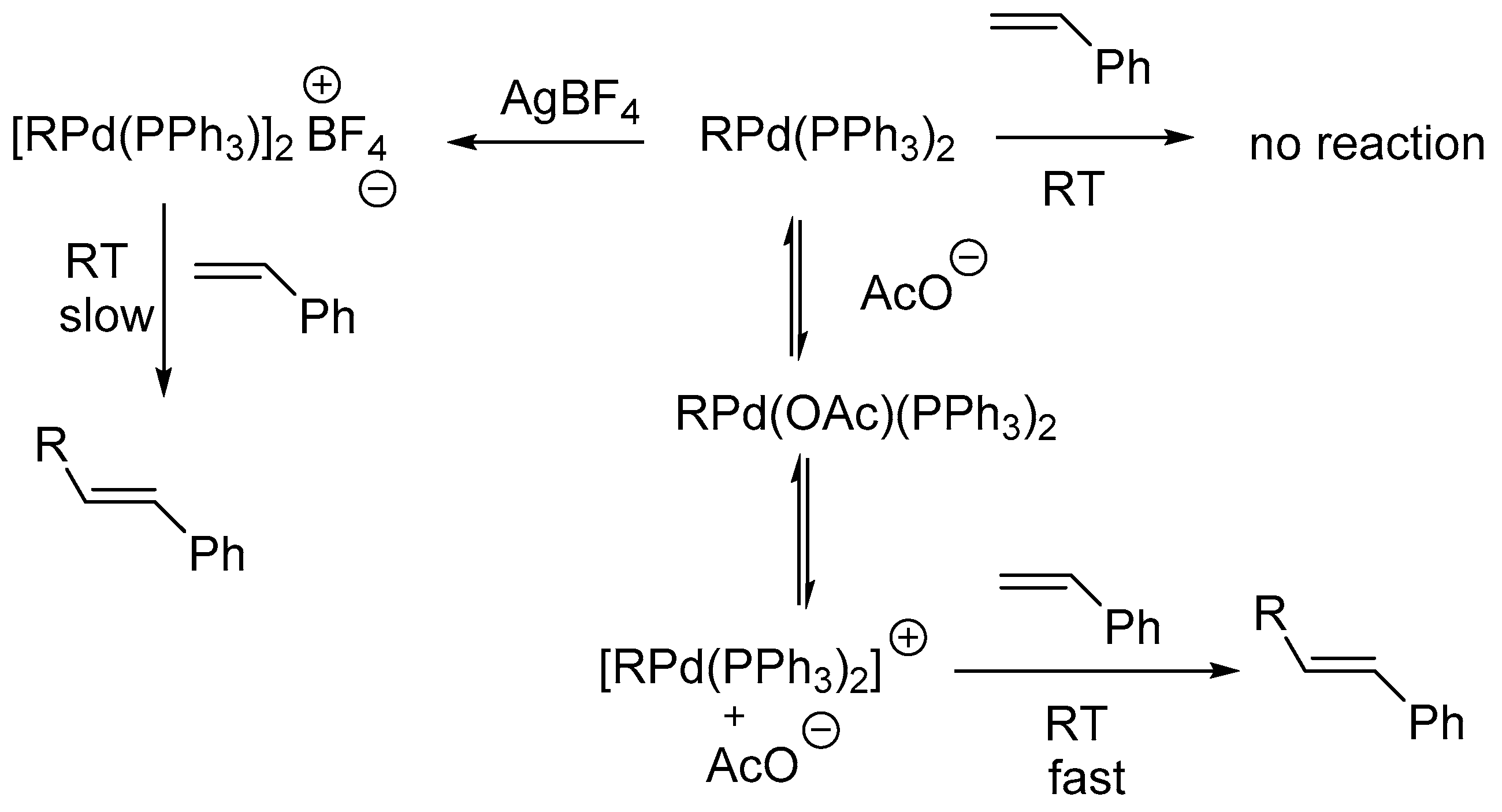
67] examines the implications of details on the mechanism of the Heck reaction using both traditional and non-traditional catalytic systems. It is mentioned that, while studying the intermediate formed during oxidative addition of aryl halide to Pd complex, the conventionally thought intermediate ArPdL
2X is not produced rather an intermediate ArPd(PPh
3)
2(OAc) is formed. The formation of this species is also supported by Amator and co-workers [
68]. It was observed that the styrene on reaction with preformed PhPd(PPh
3)
2(OAc) during the carbon–carbon bond forming step of the Heck reaction in DMF at room temperature forms stilbene whereas on reaction with PhPdI(PPh
3)
2 under similar conditions, stilbene was not formed. Further, when acetate anion was added to a mixture of PhPdI(PPh
3)
2 and styrene, the formation of stilbene was observed at room temperature. These observations are consistent with the dissociation of acetate ion from PhPd(PPh
3)
2(OAc) to form an equilibrium mixture containing the cationic complex [PhPd(PPh
3)
2]
+ (
Scheme 7).
The observations using various reaction conditions for inter and intramolecular Heck couplings, in presence of different ligands (PPh3/P(o-tolyl)3/Chelating phosphine ligands), bases (organic/inorganic), solvent (polar/nonpolar) and/ or TBAB are cited. In addition to this, the recent modifications to traditional reaction conditions are also reviewed and interpreted that, for intermolecular couplings involving reactive electrophiles (aryl or vinyl iodides) and alkenes containing an electron withdrawing group, a traditional catalyst system such as Pd(OAc)2 with 2–4 equivalents of L [L = PPh3 or P(o-tolyl)3] or PdL2Cl2 or PdL4 along with organic or inorganic base should suffice. Such systems require temperatures in the range 50–100 °C. In order to lower the temperature, the most effective protocol is to add R4NX (X = Cl, Br) and use an aqueous solvent with K2CO3 as the base. For aryl or vinyl triflates with alkenes containing an electron withdrawing group, a traditional catalyst system can also be used. For alkenes not containing an electron withdrawing group, a halide free condition, achieved by either using aryl or vinyl triflates as the electrophile or adding a halide sequestering agent (Ag+) for aryl or vinyl halides, will be advantageous. However, for electrophiles (like aryl bromides with electron donating groups or aryl chlorides) undergoing oxidative addition more slowly, high temperatures (above 120 °C) are usually required and a stable metal-ligand system that does not decompose at higher temperature is essential for prolonged life of the catalyst and hence PPh3 will not be suitable ligand in such cases. For intramolecular Heck cyclizations, the reaction conditions appear to vary depending on the ring size, stereochemistry of the alkene and whether a tertiary or quaternary centre is being formed. The presence of halides does not appear to impede the cyclization at elevated temperatures and can be beneficial for high ee’s in asymmetric Heck couplings. The use of halide free conditions can produce rapid Heck couplings but variable ee’s are seen for asymmetric cyclization.
The Amatore group of researchers have invested much effort into investigations of the mechanism of Heck reaction. Kinetic evidences and electrochemical techniques like steady state voltammetry along with spectroscopic techniques like UV (Ultra violet spectroscopy), NMR (Nuclear Magnetic Resonance Spectroscopy) was used for the elucidation of mechanisms of palladium-catalysed reactions [
69]. Their work emphasizes the crucial role played by the anions born by the precursors of palladium(0) complexes and rationalize empirical findings dispersed in literature concerning the specificity of palladium catalytic systems. A new adequate catalytic cycle has been proposed for Heck reactions where the fundamental role of chloride or acetate ions brought by palladium(II) complexes, precursors of palladium(0) complexes, is established (
Scheme 8).
It is proposed that the new reactive anionic palladium(0) complexes species are formed where palladium(0) is ligated by either chloride ions Pd(0)(PPh3)2Cl− or by acetate ions Pd(0)(PPh3)2(OAc)−. The reactivity of such anionic palladium(0) complexes in oxidative addition to aryl iodides strongly depends on the anion ligated to the palladium(0). The structure of the arylpalladium(II) complexes formed in the oxidative addition also depends on the anion.
The existence of intermediate anionic pentacoordinated arylpalladium(II) complexes ArPdI(Cl)(PPh
3)
2− 135 and ArPdI(OAc)(PPh
3)
2− 136 (
Figure 19) are also predicted based on evidences. Their stability and reactivity is discussed depending on the presence of chloride or acetate anion. ArPdI(Cl)(PPh
3)
2− is found to be more stable and it affords trans ArPdI(PPh
3)
2, whereas, ArPdI(OAc)(PPh
3)
2− is quite unstable and rapidly affords the stable trans ArPd(OAc)(PPh
3)
2 complex.
Another review by Amator and Jutand [
70] is the resurgence on the mechanism of Heck reaction. It aims at giving the evidences of anionic Pd(0) and Pd(II) intermediates in palladium-catalysed Heck and cross-coupling reactions. It was proved that the anions of PdCl
2L
2 and Pd(OAc)
2, precursors of palladium(0) play a crucial role during the reaction. Thus, Pd(0)L
2, postulated as the common catalyst in catalytic systems, is not formed as a main intermediate, instead, previously unsuspected reactive tricoordinated anionic complex species Pd
0L
2Cl
−and Pd
0L
2(OAc)
− are found to be the effective catalysts. These anions also affect the kinetics of the oxidative addition to ArI as well as the structure and reactivity of the arylpalladium(II) complexes produced in this reaction. The pentacoordinated anionic complexes ArPdI(Cl)L
2− or ArPdI(OAc)L
2− are formed in the reaction.
An alternative mechanism via Pd(II)/Pd(IV) cycle (
Scheme 9) specially proposed for PCP complexes (pincer palladacycle with Phosphorous as donor ligands) is described in the review by Whitecomb [
24]. The initiation of the catalytic cycle takes place via the oxidative addition of a vinyl C-H of CH
2 = CHR to the palladium (II) complex (step a). This is followed by the reductive elimination of HCl from the Pd(IV) species (step b) and is a rate determining step to generate Pd(II) species. One more oxidative addition of ArCl (step c) generates another Pd(IV) species collapsing further giving out coupled product via reductive elimination to restore the catalyst (step d). The mechanism is different than a classical Pd(0)/Pd(II) as it involves the successive oxidative addition of both aryl halide and alkene substrates and eliminates the need for the migratory insertion step. Formation of intermediate after a reversible step a, has been supported by NMR studies using deuterium.
Cavell and McGuinness [
71] have reviewed the redox process involving N-heterocyclic carbene complexes and associated imidazolium salts in conjunction with Group 10 metals. The mechanistic studies with stoichiometric reaction of the [(tmiy)
2Pd(Ar)]
+ cation (where Ar =
p-nitrophenyl) with butylacrylate was seen to give a complex mixture of products. It was observed that the product distribution was dependent on temperature and other reaction conditions. For example, at −30 °C only the reductive elimination products 2-(4-nitrophenyl)-1,3,4,5-tetramethylimidazolium ion, and traces of the 2-substituted-imidazolium ion were observed. On warming to −20 °C, the Heck coupling product,
n-butyl (
E)-4-nitrocinnamate, and a further product, 1,3,4,5-tetramethyl imidazolium salt, were observed. At room temperature, the main products were the Heck product and the 1,3,4,5-tetramethylimidazolium salt. This indicates that the raise in temperature leads to the Heck coupling product more rapidly, i.e., the rates of migratory insertion plus β-elimination, as required for Heck catalysis, become more competitive with reductive elimination. When the process is run under catalytic conditions, i.e., base with an excess of
p-nitrophenyliodide and butylacrylate at around 120 °C, only the Heck product is observed with high turnover frequencies (TOF) and TON and no products generated from the reductive elimination reaction were observed. This indicates the thermodynamic parameters such as activation barriers and relative exothermicities play a crucial role.
Trzeciak and Ziolkowski [
72] have reviewed the catalytic activity of palladium in C-C bond forming processes in respect to Heck and carbonylation reactions. Catalytic systems based on palladium complexes with phosphorus ligands and phosphine-free systems and based on the Pd(0) colloid are discussed by giving a number of examples. These examples reveal that, both monomolecular Pd(0) phosphine complexes and nanosized Pd(0) colloids can act as active catalysts. This review supports the formation of conventional oxidative addition complex PhPdL
2X on the basis of evidences. The possibility of typical monomolecular Pd(0) complexes (e.g., [Pd(0)P4], P = phosphorus ligand) undergoing partial decomposition during the catalytic process to form nanosized phosphine-free Pd(0) particles is predicted. Palladium in the form of a colloid was observed to be a good catalyst for Heck and carbonylation reactions, especially when applied in an ammonium salt ([R
4N]X) or an ionic liquid (IL) medium. Ammonium salts efficiently contribute to the formation of soluble, monomolecular Pd(0) compounds of probably higher catalytic activity than bigger-sized clusters. The study reveals that carboxylate salts used as bases in the Heck reactions, can act as effective Pd(II) to Pd(0) reducing agents, and therefore they also may initiate the formation of colloidal nanoparticles.
Phan et al. [
73] have a critical review on the nature of the active species in palladium catalysed Mizoroki–Heck and Suzuki–Miyaura couplings using homogeneous or heterogeneous catalysis. Regardless of what palladium precatalyst is used, it is critical for researchers to understand the nature of the true catalytic species generally used in their studies because of palladium deactivation via clustering, leaching and redeposition processes. The review summarizes each type of precatalyst of palladium, proposed to be truly active in the various form of catalysts like discrete soluble palladium complexes, solid-supported metal ligand complexes, supported palladium nano- and macroparticles, soluble palladium nanoparticles, soluble ligandfree palladium and palladium-exchanged oxides. A considerable focus is placed on solid precatalysts and on evidence for and against catalysis by solid surfaces vs. soluble species when starting with a range of precatalysts.
Based on various control experiments and tests to assess the homogeneity or heterogeneity of catalyst systems, it was concluded that, the Heck reactions using NC, SC, PC, SCS and PCP palladacycles (catalysts with N, O, P or S donor ligands) operates by Pd(0)/Pd(II) mechanism only and there is no proven example of catalysts operating by a Pd(II)/Pd(IV) catalytic cycle in Heck or Suzuki coupling reactions.
Knowles and Whiting [
74] have reviewed advances in the mechanistic perspective of the Heck reaction aiming to review and compare work undertaken on the mechanism until 2007. The data provided give the evidence for a mechanism where the oxidative addition strongly depends on the conditions, particularly whether the reaction is saturated with halide. Eventually, the authors concluded that it is not possible to explain all the phenomena observed for the oxidative addition on the basis of single mechanism only and there may be several mechanisms in operation, either independently, depending on the reaction conditions, or in parallel.
Although the electron rich and chelating nature of the phosphines is required to activate aryl chlorides, it disfavors carbometallation or dissociation. It is also assumed that the rate determining step comes after initial oxidative addition; however, the nature of this step is yet unclear and has a strong dependence on the nature of the species produced in the oxidative addition step.
Kumar and co-workers [
75] have reviewed the status regarding in situ generation of palladium chalcogenides phases or Pd(0) protected with organochalcogen fragments when Palladium(II) complexes of organochalcogen ligands (e.g., Pd(II) complex of an (S,C,S) pincer ligand) are used as viable alternatives to complexes of phosphine/carbene ligands for Suzuki−Miyaura and Heck C-C cross coupling reactions. These ligands are thermally stable, easy to handle and air and moisture sensitivity are not impediments with many of them.
Recently, Eremin and Ananikov [
76] summarized the studies mentioning the behaviour of a “Cocktail” of catalysts and mechanistic investigations regarding the evolution of transition metal species during catalytic cycles. They have considered the transformations of molecular catalysts, leaching, aggregation and various interconversions of metal complexes, clusters and nanoparticles that occur during catalytic processes termed as “Cocktail”-type systems. These systems are considered from the perspective of the development of a new generation of efficient, selective and re-usable catalysts for synthetic applications. It also attempts to determine ‘‘What are the true active species?” in mechanism specially in coupling reactions like Heck. It has been concluded that there is nothing static in catalysis and catalytic system is always dynamic where ‘‘Cocktail”-type catalytic systems are formed when the transformation of a metal species occurs during the overall catalytic process. It was observed that the “Cocktail” picture can exist not only in homogeneous catalysis in solution but also in a heterogeneous catalytic system.