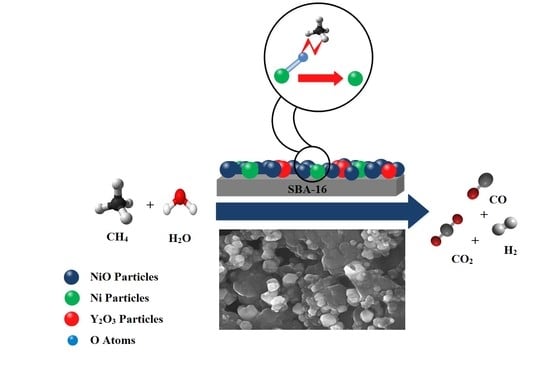
Hydrogen Production from Cyclic Chemical Looping Steam Methane Reforming over Yttrium Promoted Ni/SBA-16 Oxygen Carrier
Abstract
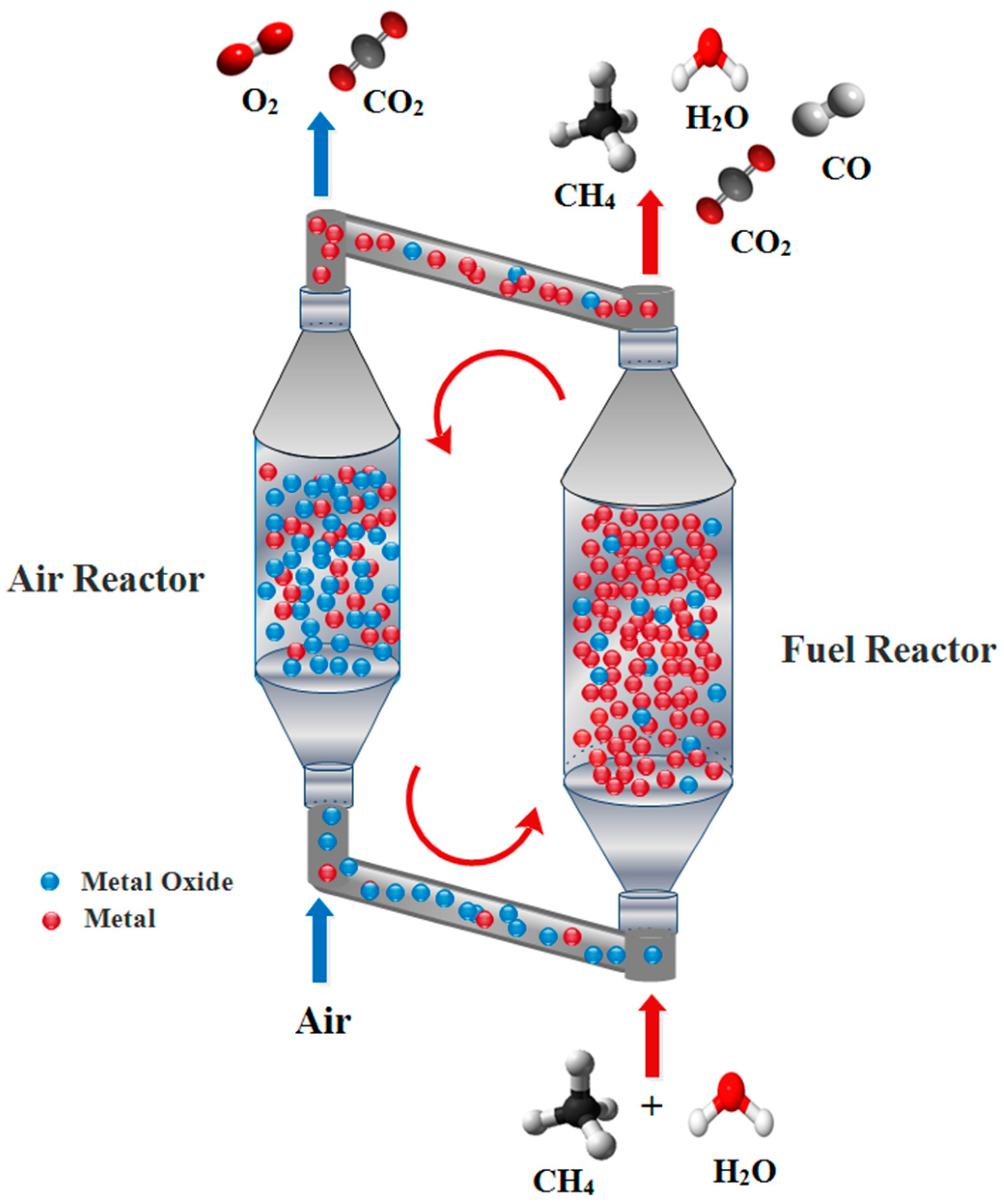
:1. Introduction
2. Results and Discussion
2.1. Sample Characterization
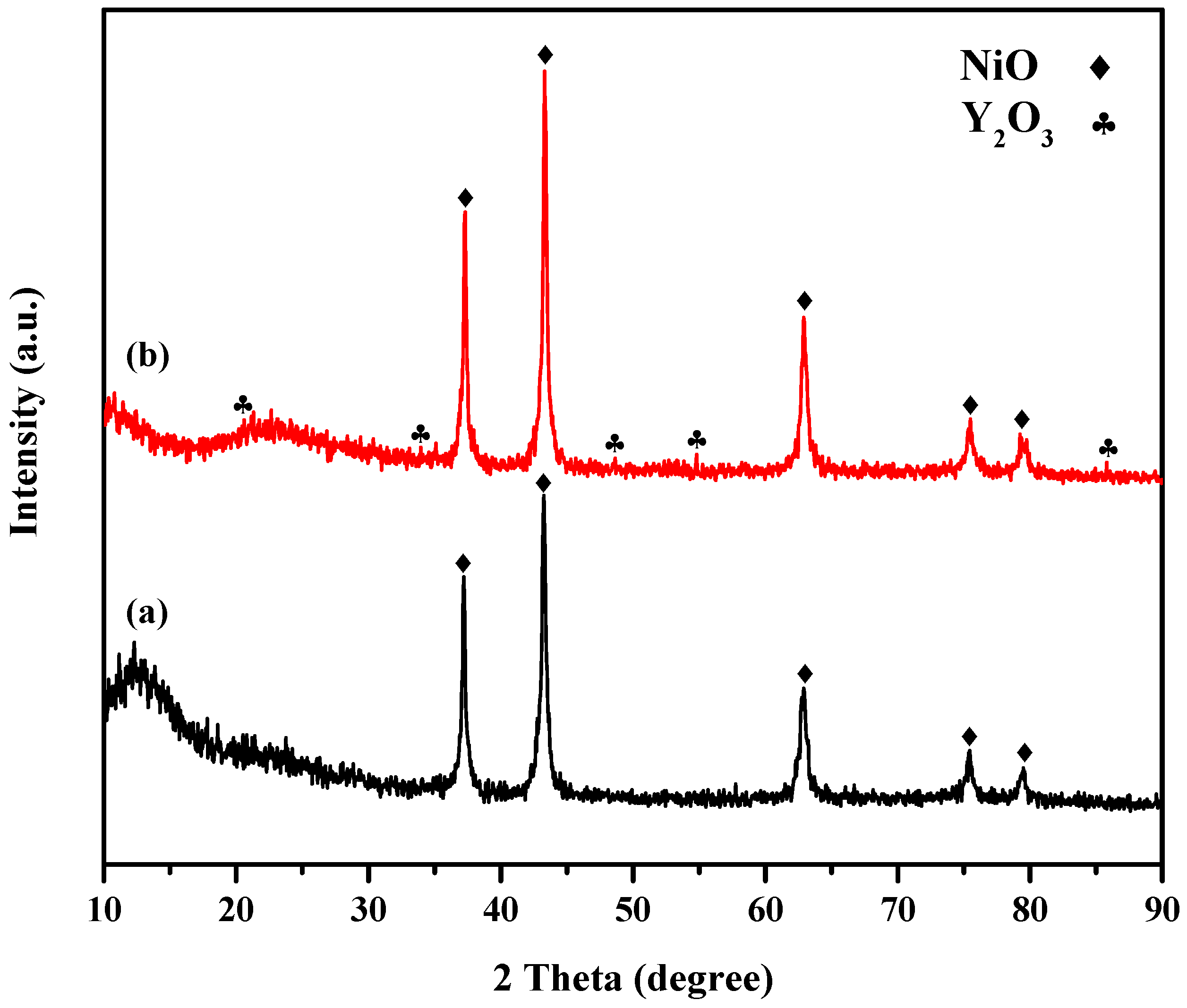
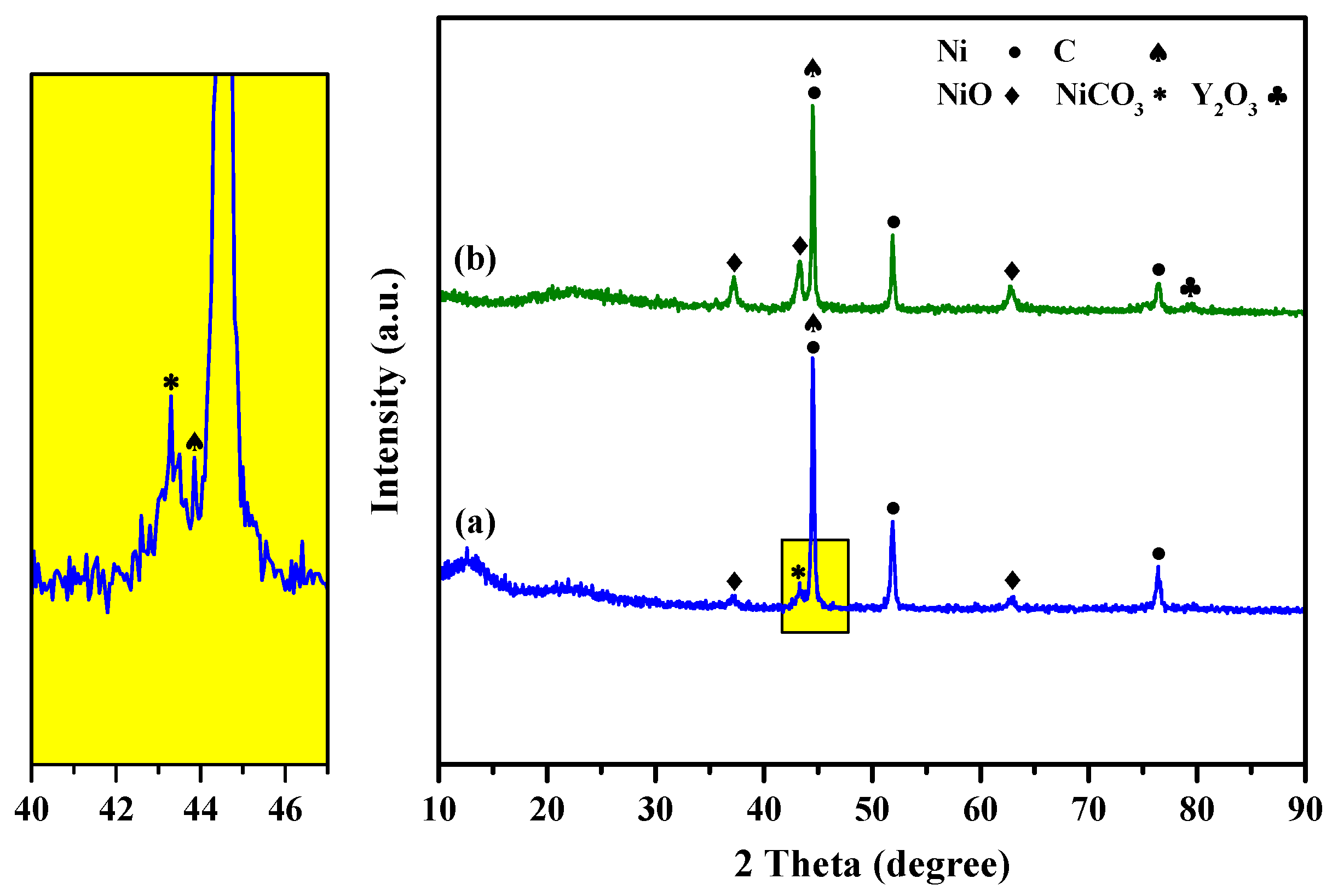
2.1.1. X-ray Powder Diffraction (XRD) Study
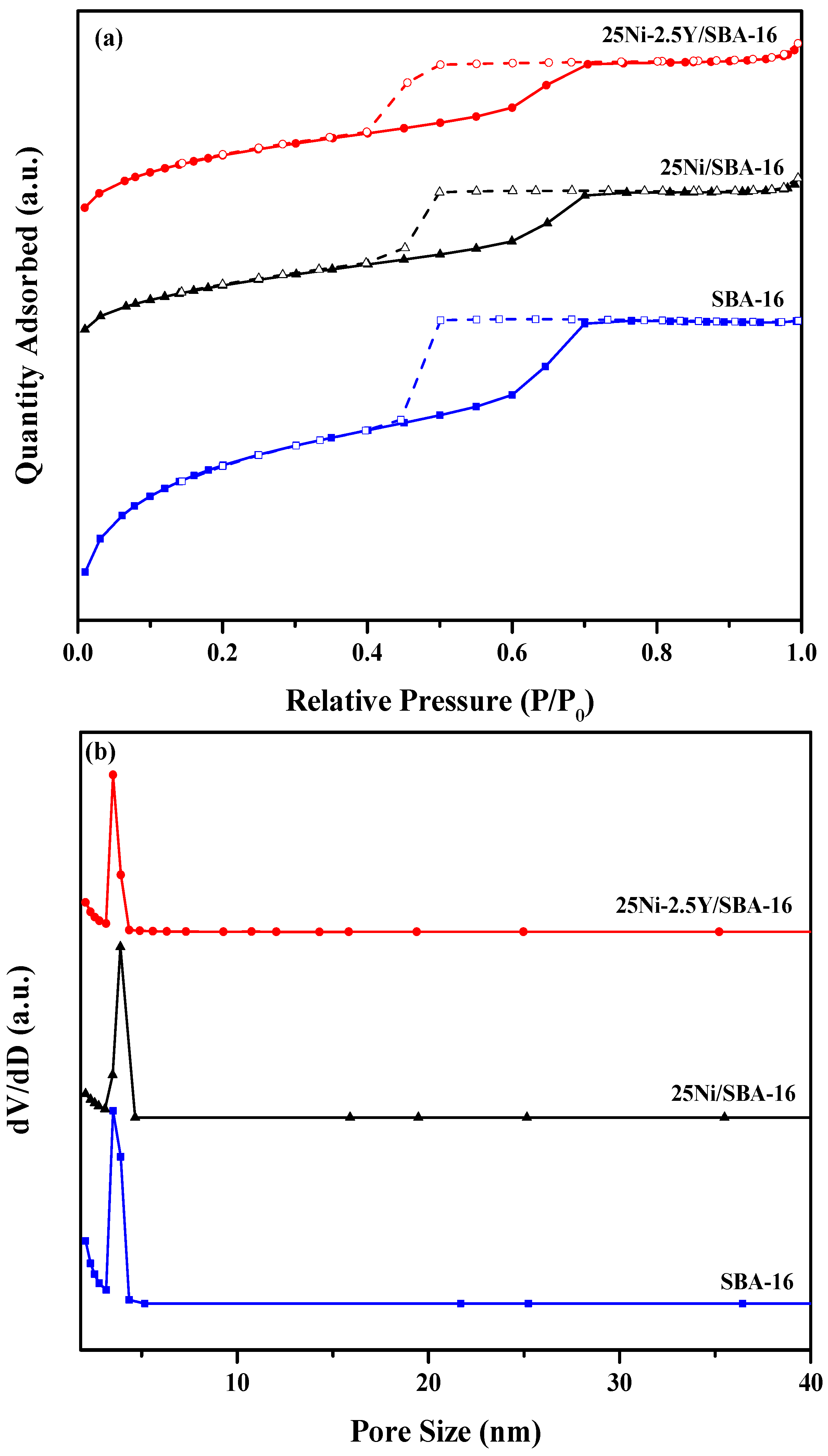
2.1.2. N2 Adsorption-Desorption Isotherms
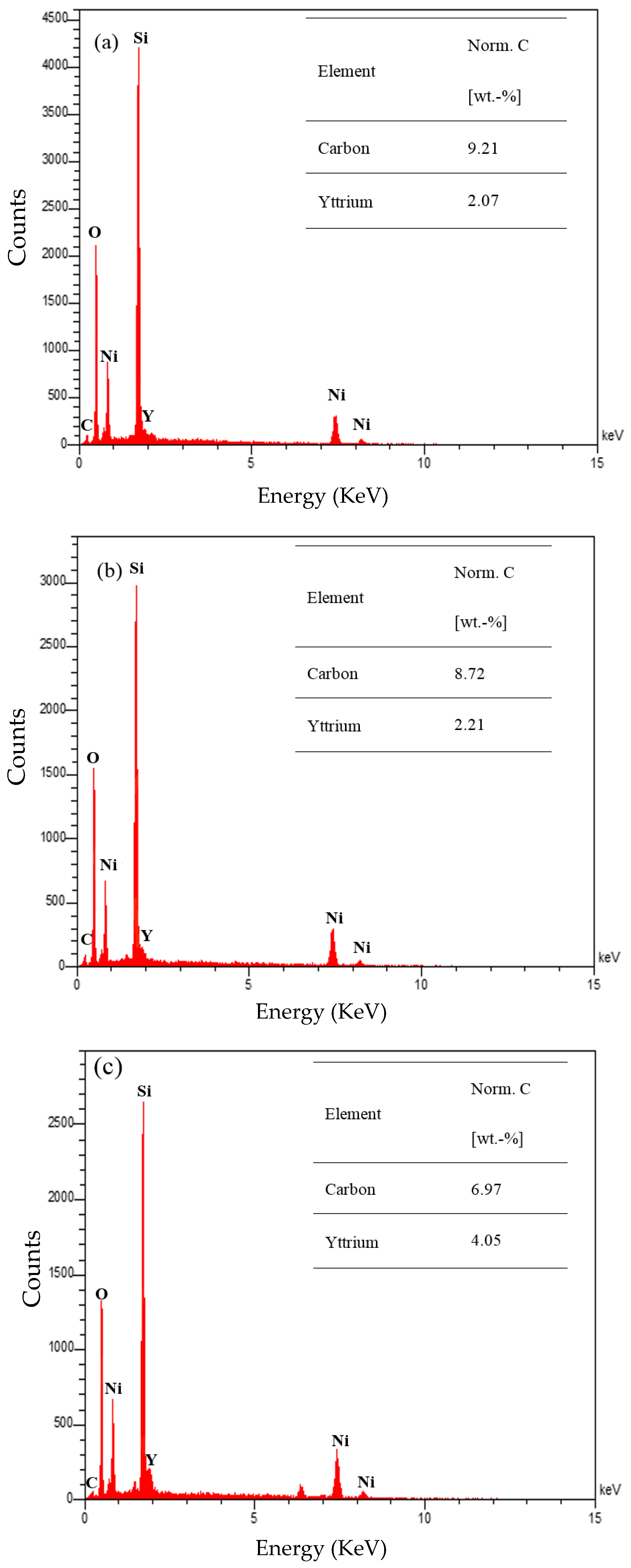
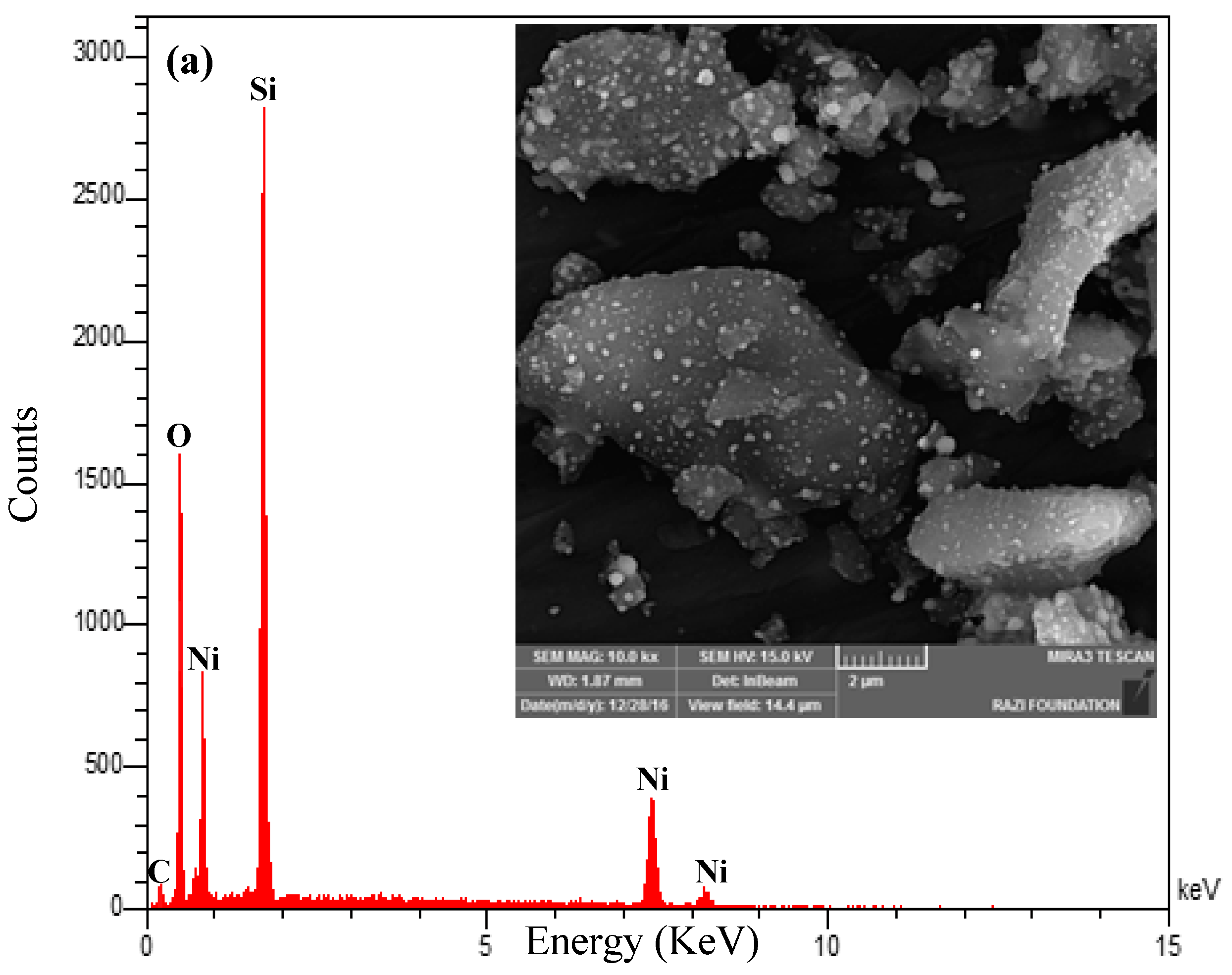
2.1.3. Field Emission Scanning Electron Microscopy (FESEM) Analysis
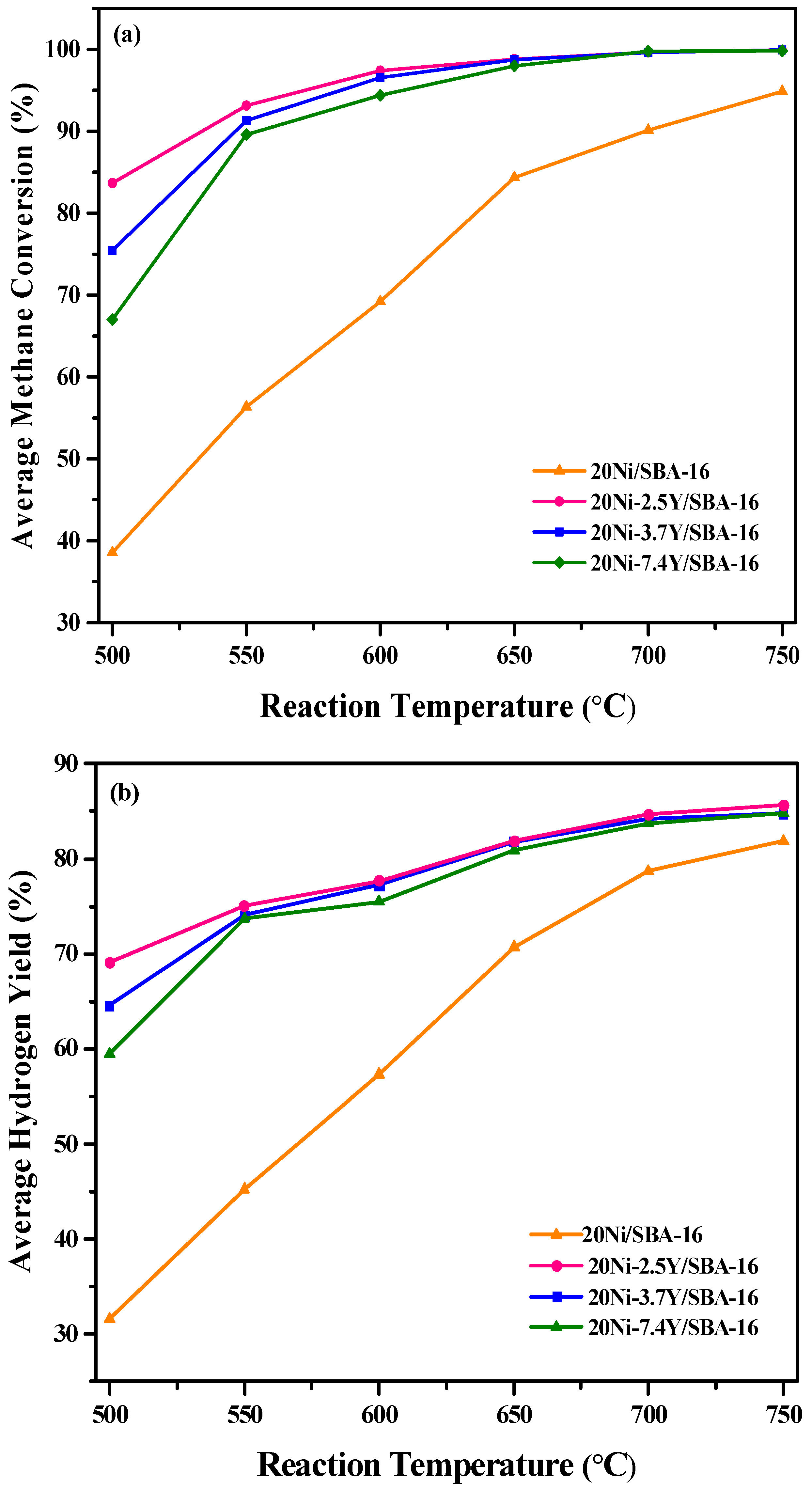
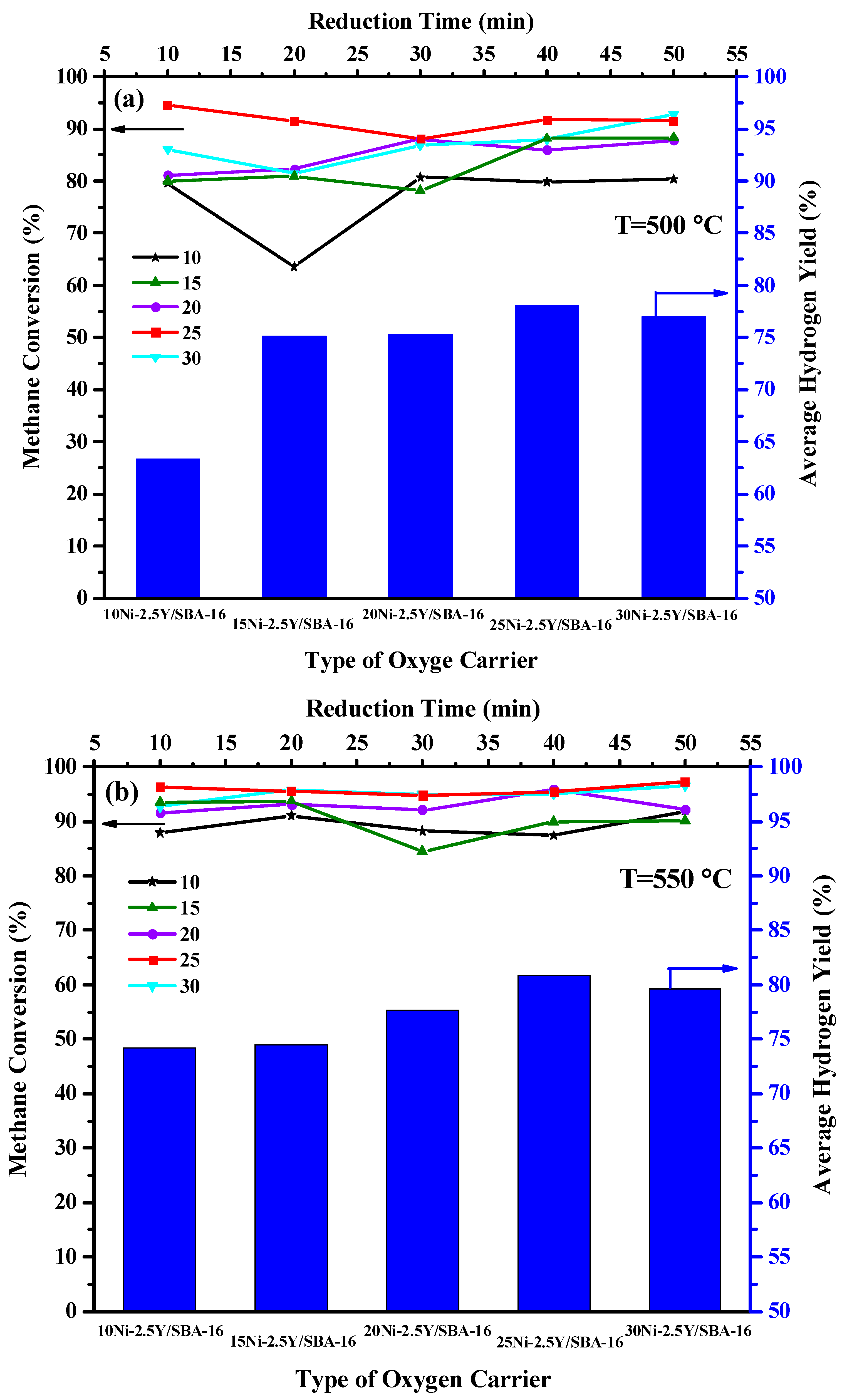
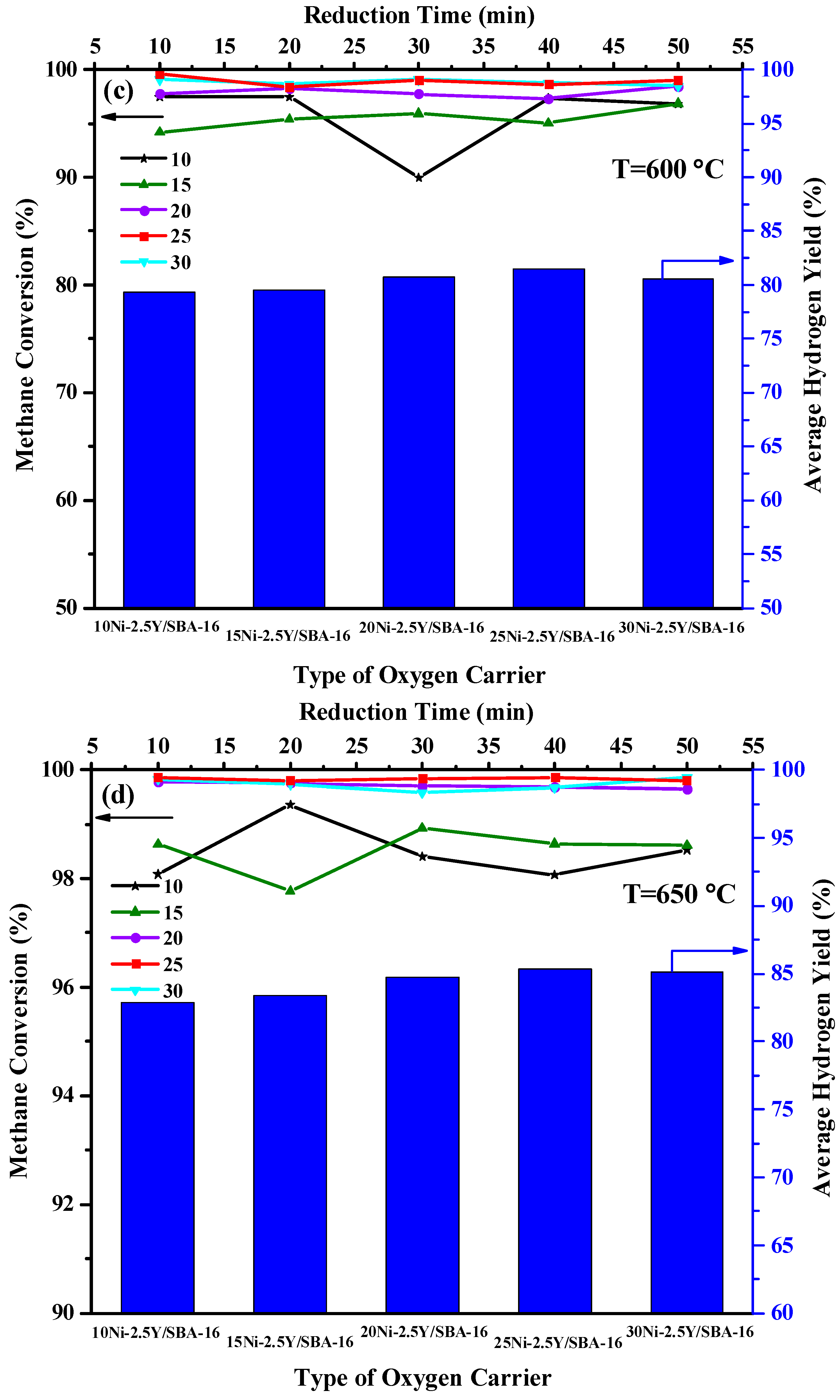
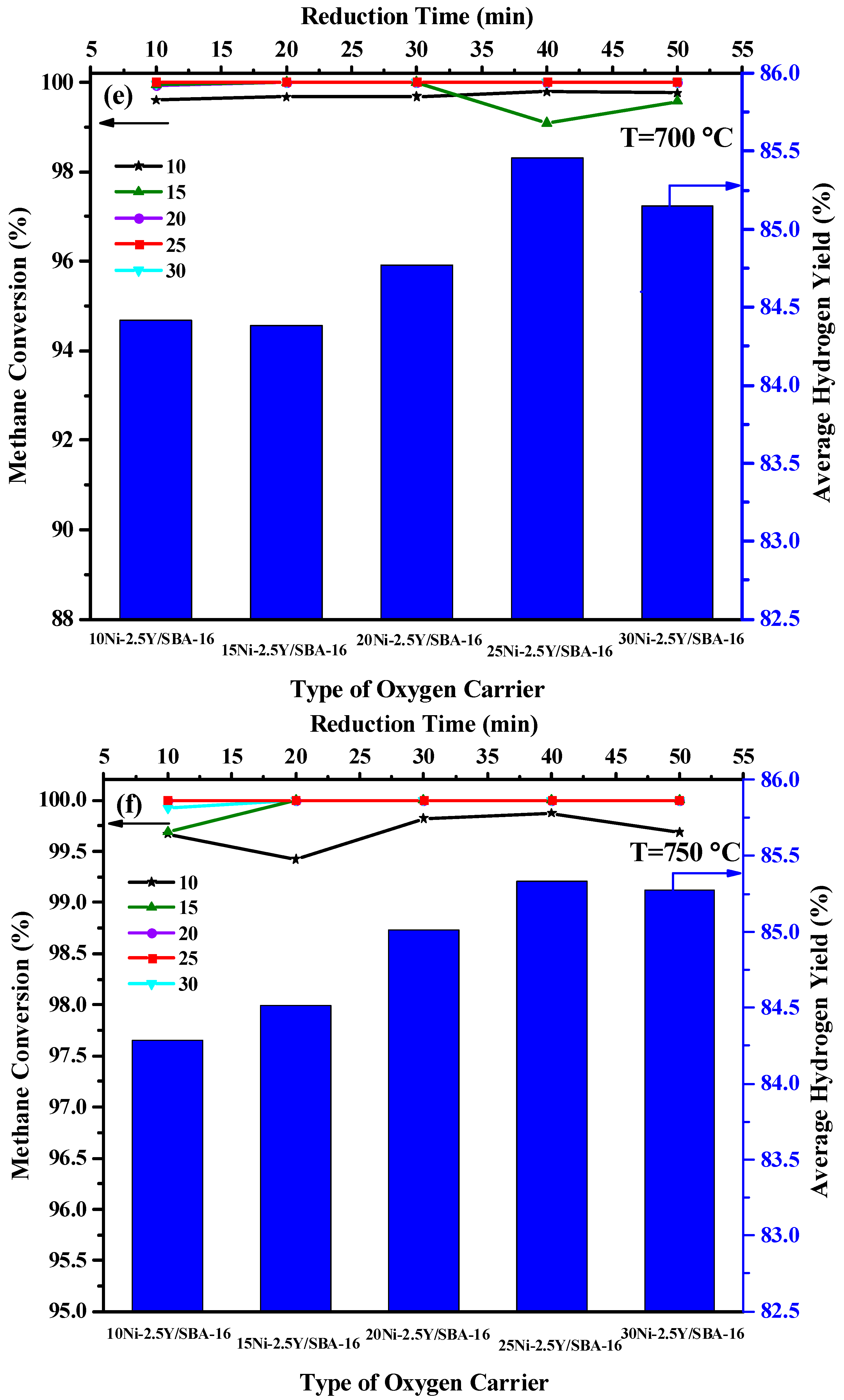
2.2. Effect of Yttrium Weight Percentage and Temperature on the Catalytic Activity
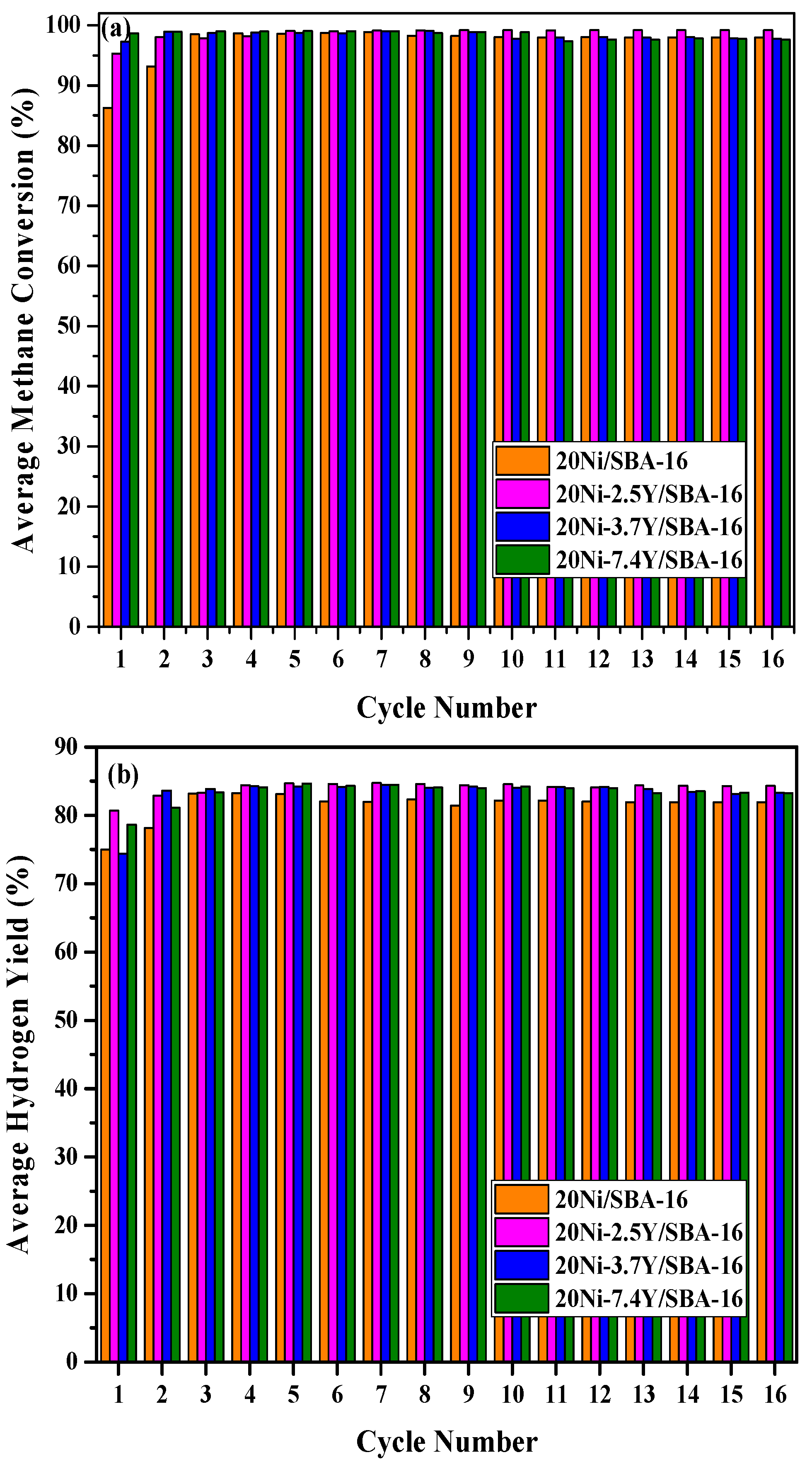
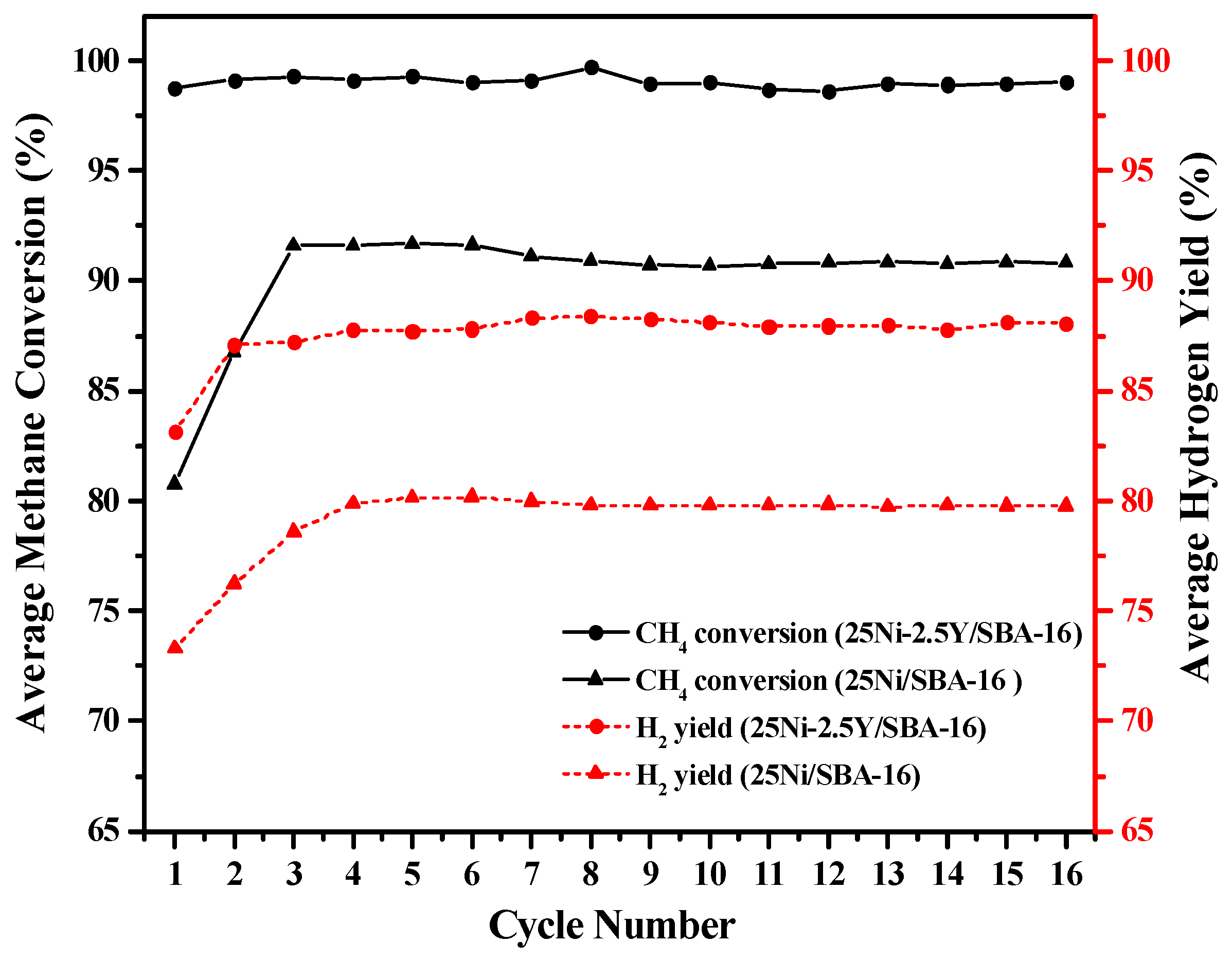
2.3. Life Time Investigation of Different Yttrium Weight Percentage
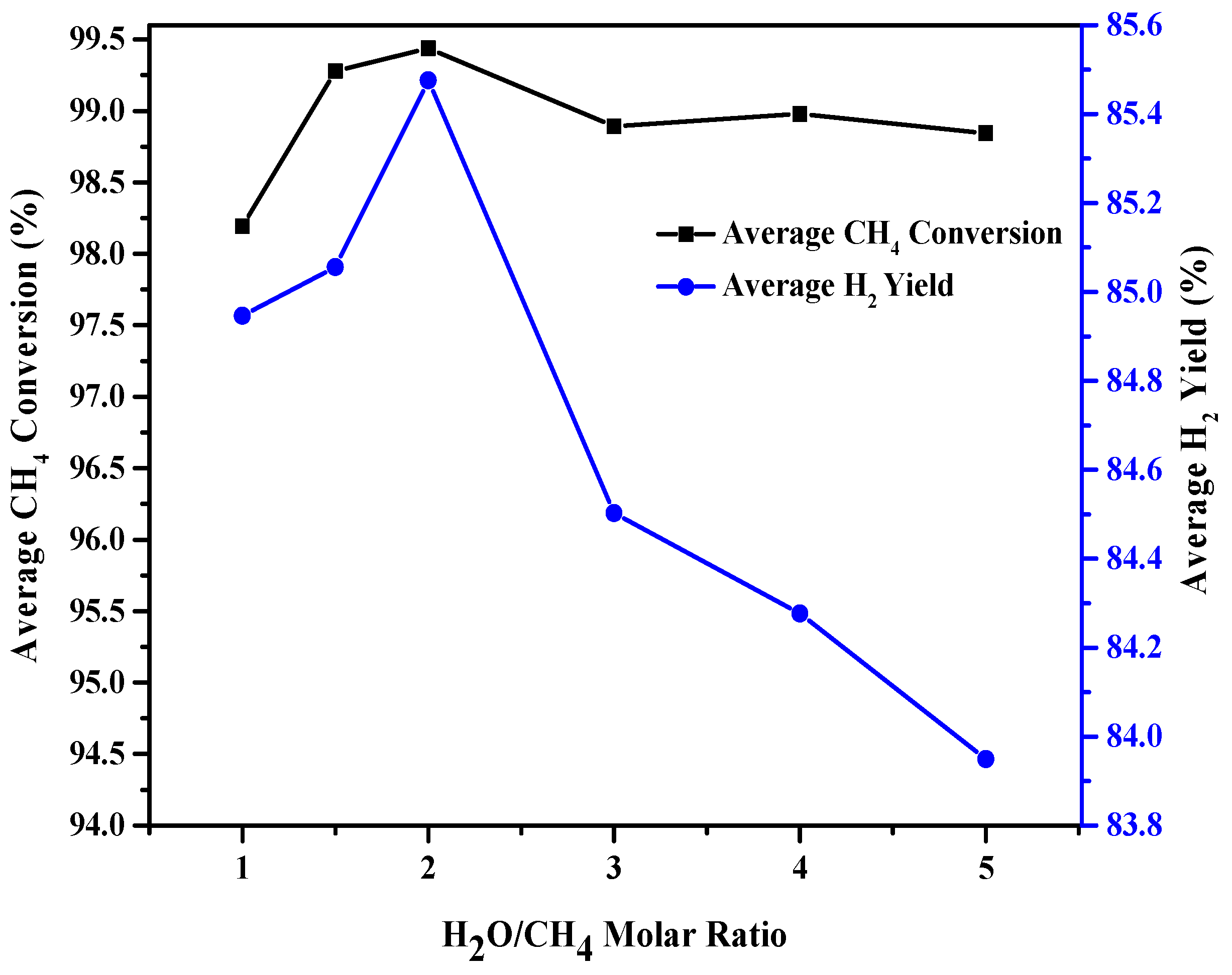
2.4. Effect of Steam/Carbon Molar Ratio on the Catalytic Performance
2.5. Effect of Ni Loading Percentage and Temperature on the Catalytic Activity
2.6. Life Time of 25Ni/SBA-16 and 25Ni-2.5Y/SBA-16 Oxygen Carriers
3. Experimental Methods
3.1. Oxygen Carrier Preparation
3.2. Oxygen Carrier Characterization
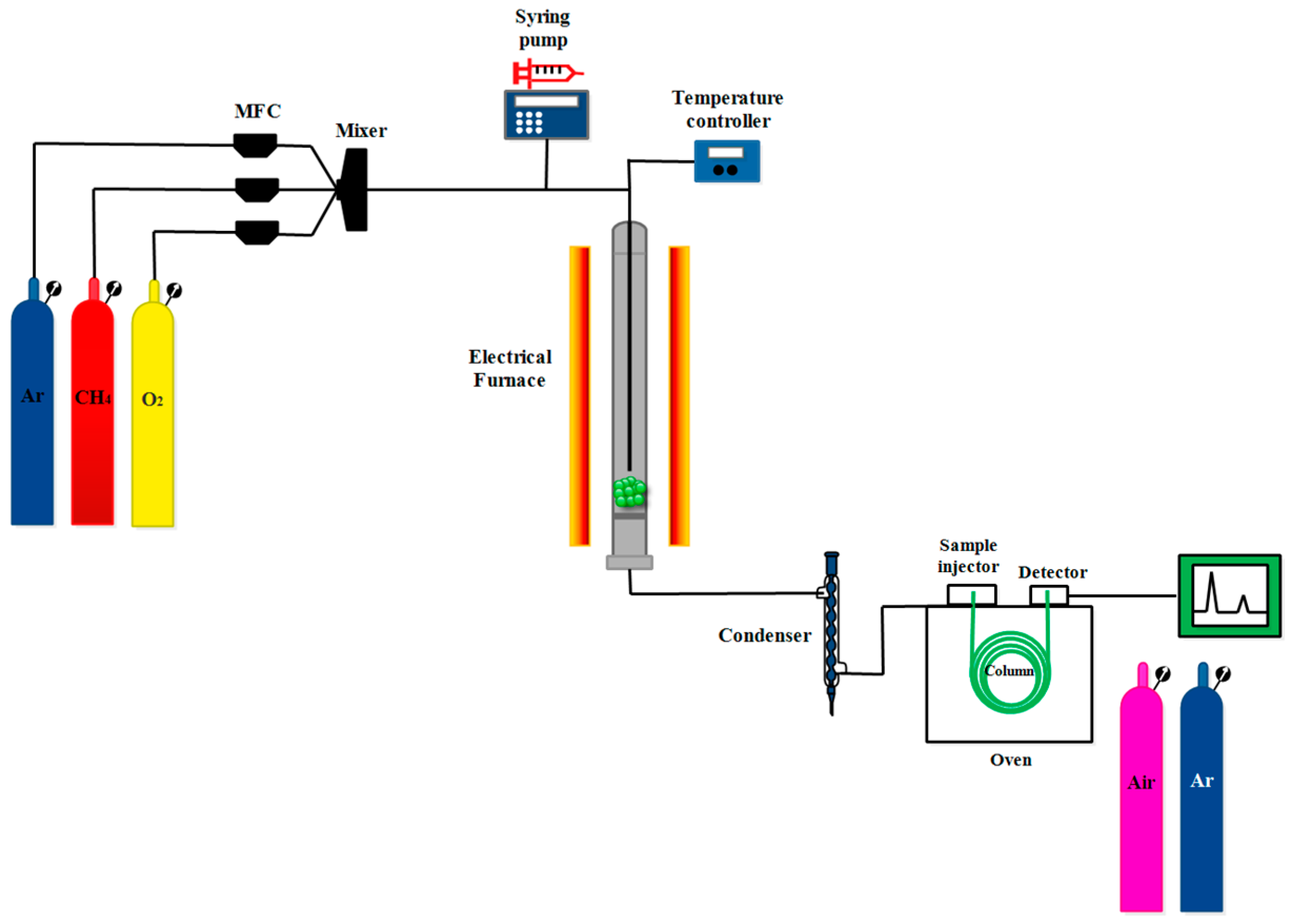
3.3. Process Activity
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Hafizi, A.; Rahimpour, M.R.; Hassanajili, S. Calcium promoted Fe/Al2O3 oxygen carrier for hydrogen production via cyclic chemical looping steam methane reforming process. Int. J. Hydrogen Energy 2015, 40, 16159–16168. [Google Scholar] [CrossRef]
- Hafizi, A.; Rahimpour, M.R.; Hassanajili, S. Hydrogen production by chemical looping steam reforming of methane over Mg promoted iron oxygen carrier: Optimization using design of experiments. J. Taiwan Inst. Chem. Eng. 2016, 62, 140–1409. [Google Scholar] [CrossRef]
- Karimi, E.; Forutan, H.; Saidi, M.; Rahimpour, M.R.; Shariati, A. Experimental study of chemical-looping reforming in a fixed-bed reactor: Performance investigation of different oxygen carriers on Al2O3 and TiO2 support. Energy Fuels 2014, 28, 2811–2820. [Google Scholar] [CrossRef]
- Frusteri, F.; Freni, S.; Spadaro, L.; Chiodo, V.; Bonura, G.; Donato, S.; Cavallaro, S. H2 production for MC fuel cell by steam reforming of ethanol over MgO supported Pd, Rh, Ni and Co catalysts. Catal. Commun. 2004, 5, 611–615. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, M.; Liang, T.; Yang, Z.; Yang, J.; Liu, S. Hydrogen generation from catalytic steam reforming of acetic acid by Ni/attapulgite catalysts. Catalysts 2016, 6, 172. [Google Scholar] [CrossRef]
- Du, Y.-L.; Wu, X.; Cheng, Q.; Huang, Y.-L.; Huang, W. Development of Ni-based catalysts derived from hydrotalcite-like compounds precursors for synthesis gas production via methane or ethanol reforming. Catalysts 2017, 7, 70. [Google Scholar] [CrossRef]
- Midilli, A.; Ay, M.; Dincer, I.; Rosen, M. On hydrogen and hydrogen energy strategies: I: Current status and needs. Renew. Sustain. Energy Rev. 2005, 9, 255–271. [Google Scholar] [CrossRef]
- Momirlan, M.; Veziroglu, T.N. The properties of hydrogen as fuel tomorrow in sustainable energy system for a cleaner planet. Int. J. Hydrogen Energy 2005, 30, 795–802. [Google Scholar] [CrossRef]
- Holladay, J.D.; Hu, J.; King, D.L.; Wang, Y. An overview of hydrogen production technologies. Catal. Today 2009, 139, 244–260. [Google Scholar] [CrossRef]
- Abbasi, M.; Farniaei, M.; Rahimpour, M.R.; Shariati, A. Enhancement of Hydrogen Production and Carbon Dioxide Capturing in a Novel Methane Steam Reformer Coupled with Chemical Looping Combustion and Assisted by Hydrogen Perm-Selective Membranes. Energy Fuels 2013, 27, 5359–5372. [Google Scholar] [CrossRef]
- Akbari-Emadabadi, S.; Rahimpour, M.R.; Hafizi, A.; Keshavarz, P. Production of hydrogen-rich syngas using Zr modified Ca-Co bifunctional catalyst-sorbent in chemical looping steam methane reforming. Appl. Energy 2017, 206, 51–62. [Google Scholar] [CrossRef]
- Schädel, B.T.; Duisberg, M.; Deutschmann, O. Steam reforming of methane, ethane, propane, butane, and natural gas over a rhodium-based catalyst. Catal. Today 2009, 142, 42–51. [Google Scholar] [CrossRef]
- Basagiannis, A.; Verykios, X. Catalytic steam reforming of acetic acid for hydrogen production. Int. J. Hydrogen Energy 2007, 32, 3343–3355. [Google Scholar] [CrossRef]
- Armor, J. Catalysis and the hydrogen economy. Catal. Lett. 2005, 101, 131–135. [Google Scholar] [CrossRef]
- Chaubey, R.; Sahu, S.; James, O.O.; Maity, S. A review on development of industrial processes and emerging techniques for production of hydrogen from renewable and sustainable sources. Renew. Sustain. Energy Rev. 2013, 23, 443–462. [Google Scholar] [CrossRef]
- Forutan, H.; Karimi, E.; Hafizi, A.; Rahimpour, M.R.; Keshavarz, P. Expert representation chemical looping reforming: A comparative study of Fe, Mn, Co and Cu as oxygen carriers supported on Al2O3. J. Ind. Eng. Chem. 2015, 21, 900–911. [Google Scholar] [CrossRef]
- Rydén, M.; Arjmand, M. Continuous hydrogen production via the steam–iron reaction by chemical looping in a circulating fluidized-bed reactor. Int. J. Hydrogen Energy 2012, 37, 4843–4854. [Google Scholar] [CrossRef]
- Zhu, X.; Wei, Y.; Wang, H.; Li, K. Ce-Fe oxygen carriers for chemical-looping steam methane reforming. Int. J. Hydrogen Energy 2013, 38, 4492–4501. [Google Scholar] [CrossRef]
- Hafizi, A.; Rahimpour, M.R.; Hassanajili, S. Hydrogen production via chemical looping steam methane reforming process: Effect of cerium and calcium promoters on the performance of Fe2O3/Al2O3 oxygen carrier. Appl. Energy 2016, 165, 685–694. [Google Scholar] [CrossRef]
- Moghtaderi, B. Review of the recent chemical looping process developments for novel energy and fuel applications. Energy Fuels 2011, 26, 15–40. [Google Scholar] [CrossRef]
- Solunke, R.D.; Veser, G.T. Hydrogen production via chemical looping steam reforming in a periodically operated fixed-bed reactor. Ind. Eng. Chem. Res. 2010, 49, 11037–11044. [Google Scholar] [CrossRef]
- Hafizi, A.; Rahimpour, M.R.; Hassanajili, S. High purity hydrogen production via sorption enhanced chemical looping reforming: Application of 22Fe2O3/MgAl2O4 and 22Fe2O3/Al2O3 as oxygen carriers and cerium promoted CaO as CO2 sorbent. Appl. Energy 2016, 169, 629–641. [Google Scholar] [CrossRef]
- Rahimpour, M.R.; Hesami, M.; Saidi, M.; Jahanmiri, A.; Farniaei, M.; Abbasi, M. Methane Steam Reforming Thermally Coupled with Fuel Combustion: Application of Chemical Looping Concept as a Novel Technology. Energy Fuels 2013, 27, 2351–2362. [Google Scholar] [CrossRef]
- Alirezaei, I.; Hafizi, A.; Rahimpour, M.R.; Raeissi, S. Application of zirconium modified Cu-based oxygen carrier in chemical looping reforming. J. CO2 Util. 2016, 14, 112–121. [Google Scholar] [CrossRef]
- Zafar, Q.; Mattisson, T.; Gevert, B. Redox investigation of some oxides of transition-state metals Ni, Cu, Fe, and Mn supported on SiO2 and MgAl2O4. Energy Fuels 2006, 20, 34–44. [Google Scholar] [CrossRef]
- Adanez, J.; Abad, A.; Garcia-Labiano, F.; Gayan, P.; Luis, F. Progress in chemical-looping combustion and reforming technologies. Prog. Energy Comb. Sci. 2012, 38, 215–282. [Google Scholar] [CrossRef] [Green Version]
- Trimm, D.L. Catalysts for the control of coking during steam reforming. Catal. Today 1999, 49, 3–10. [Google Scholar] [CrossRef]
- Van Dillen, A.J.; Terörde, R.J.; Lensveld, D.J.; Geus, J.W.; De Jong, K.P. Synthesis of supported catalysts by impregnation and drying using aqueous chelated metal complexes. J. Catal. 2003, 216, 257–264. [Google Scholar] [CrossRef]
- Zhong, X.; Xie, W.; Wang, N.; Duan, Y.; Shang, R.; Huang, L. Dolomite-Derived Ni-Based Catalysts with Fe Modification for Hydrogen Production via Auto-Thermal Reforming of Acetic Acid. Catalysts 2016, 6, 85. [Google Scholar] [CrossRef]
- Xu, B.-Q.; Wei, J.-M.; Wang, H.-Y.; Sun, K.-Q.; Zhu, Q.-M. Nano-MgO: Novel preparation and application as support of Ni catalyst for CO2 reforming of methane. Catal. Today 2001, 68, 217–225. [Google Scholar] [CrossRef]
- Kambolis, A.; Matralis, H.; Trovarelli, A.; Papadopoulou, C. Ni/CeO2-ZrO2 catalysts for the dry reforming of methane. Appl. Catal. A Gen. 2010, 377, 16–26. [Google Scholar] [CrossRef]
- Kim, J.-H.; Suh, D.J.; Park, T.-J.; Kim, K.-L. Effect of metal particle size on coking during CO2 reforming of CH4 over Ni–alumina aerogel catalysts. Appl. Catal. A Gen. 2000, 197, 191–200. [Google Scholar] [CrossRef]
- Bengaard, H.S.; Nørskov, J.K.; Sehested, J.; Clausen, B.; Nielsen, L.; Molenbroek, A.; Rostrup-Nielsen, J. Steam reforming and graphite formation on Ni catalysts. J. Catal. 2002, 209, 365–384. [Google Scholar] [CrossRef]
- Bradford, M.; Vannice, M. CO2 reforming of CH4. Catal. Rev. 1999, 41, 1–42. [Google Scholar] [CrossRef]
- Takahashi, R.; Sato, S.; Sodesawa, T.; Tomiyama, S. CO2-reforming of methane over Ni/SiO2 catalyst prepared by homogeneous precipitation in sol–gel-derived silica gel. Appl. Catal. A Gen. 2005, 286, 142–147. [Google Scholar] [CrossRef]
- Zhang, M.; Ji, S.; Hu, L.; Yin, F.; Li, C.; Liu, H. Structural characterization of highly stable Ni/SBA-15 catalyst and its catalytic performance for methane reforming with CO2. Chin. J. Catal. 2006, 27, 777–781. [Google Scholar] [CrossRef]
- Liu, D.; Quek, X.Y.; Cheo, W.N.E.; Lau, R.; Borgna, A.; Yang, Y. MCM-41 supported nickel-based bimetallic catalysts with superior stability during carbon dioxide reforming of methane: Effect of strong metal–support interaction. J. Catal. 2009, 266, 380–390. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, J.; Cao, K.; Yang, W.; Gao, H.; Wang, Y.; Li, H. Highly dispersed nickel loaded on mesoporous silica: One-spot synthesis strategy and high performance as catalysts for methane reforming with carbon dioxide. Appl. Catal. B Environ. 2012, 125, 324–330. [Google Scholar] [CrossRef]
- Li, L.; He, S.; Song, Y.; Zhao, J.; Ji, W.; Au, C.-T. Fine-tunable Ni@ porous silica core–shell nanocatalysts: Synthesis, characterization, and catalytic properties in partial oxidation of methane to syngas. J. Catal. 2012, 288, 54–64. [Google Scholar] [CrossRef]
- Carrero, A.; Calles, J.; Vizcaíno, A. Hydrogen production by ethanol steam reforming over Cu-Ni/SBA-15 supported catalysts prepared by direct synthesis and impregnation. Appl. Catal. A Gen. 2007, 327, 82–94. [Google Scholar] [CrossRef]
- Klimova, T.; Calderón, M.; Ramı́rez, J. Ni and Mo interaction with Al-containing MCM-41 support and its effect on the catalytic behavior in DBT hydrodesulfurization. Appl. Catal. A Gen. 2003, 240, 29–40. [Google Scholar] [CrossRef]
- Zhang, S.; Muratsugu, S.; Ishiguro, N.; Tada, M. Ceria-doped Ni/SBA-16 catalysts for dry reforming of methane. ACS Catal. 2013, 3, 1855–1864. [Google Scholar] [CrossRef]
- Kantorovich, D.; Haviv, L.; Vradman, L.; Landau, M. Behaviour of NiO and NiO pohases at high loadings, in SBA-15 and SBA-16 mesoporous silica matrices. Stud. Surf. Sci. Catal. 2005, 156, 147–154. [Google Scholar] [CrossRef]
- Luna, A.E.C.; Iriarte, M.E. Carbon dioxide reforming of methane over a metal modified Ni-Al2O3 catalyst. Appl. Catal. A Gen. 2008, 343, 10–15. [Google Scholar] [CrossRef]
- Hu, Y.H.; Ruckenstein, E. The characterization of a highly effective NiO/MgO solid solution catalyst in the CO2 reforming of CH4. Catal. Lett. 1997, 43, 71–77. [Google Scholar] [CrossRef]
- Li, B.; Xu, X.; Zhang, S. Synthesis gas production in the combined CO2 reforming with partial oxidation of methane over Ce-promoted Ni/SiO2 catalysts. Int. J. Hydrogen Energy 2013, 38, 890–900. [Google Scholar] [CrossRef]
- Qian, L.; Ma, Z.; Ren, Y.; Shi, H.; Yue, B.; Feng, S.; Shen, J.; Xie, S. Investigation of La promotion mechanism on Ni/SBA-15 catalysts in CH4 reforming with CO2. Fuel 2014, 122, 47–53. [Google Scholar] [CrossRef]
- Meshksar, M.; Daneshmand-Jahromi, S.; Rahimpour, M.R. Synthesis and characterization of cerium promoted Ni/SBA-16 oxygen carrier in cyclic chemical looping steam methane reforming. J. Taiwan Inst. Chem. Eng. 2017, 76, 73–82. [Google Scholar] [CrossRef]
- Shi, C.; Zhang, P. Effect of a second metal (Y, K, Ca, Mn or Cu) addition on the carbon dioxide reforming of methane over nanostructured palladium catalysts. Appl. Catal. B Environ. 2012, 115, 190–200. [Google Scholar] [CrossRef]
- Li, J.; Xia, C.; Au, C.; Liu, B. Y2O3-promoted NiO/SBA-15 catalysts highly active for CO2/CH4 reforming. Int. J. Hydrogen Energy 2014, 39, 10927–10940. [Google Scholar] [CrossRef]
- Yogamalar, R.; Srinivasan, R.; Vinu, A.; Ariga, K.; Bose, A.C. X-ray peak broadening analysis in ZnO nanoparticles. Solid State Commun. 2009, 149, 1919–1923. [Google Scholar] [CrossRef]
- Kaneko, K. Determination of pore size and pore size distribution. J. Membr. Sci. 1994, 96, 59–89. [Google Scholar] [CrossRef]
- Cohan, L.H. Hysteresis and the Capillary Theory of Adsorption of Vapors1. J. Am. Chem. Soc. 1944, 66, 98–105. [Google Scholar] [CrossRef]
- Tsoncheva, T.; Linden, M.; Areva, S.; Minchev, C. Copper oxide modified large pore ordered mesoporous silicas for ethyl acetate combustion. Catal. Commun. 2006, 7, 357–361. [Google Scholar] [CrossRef]
- Rodrıguez-Castellón, E.; Dıaz, L.; Braos-Garcıa, P.; Mérida-Robles, J.; Maireles-Torres, P.; Jiménez-López, A.; Vaccari, A. Nickel-impregnated zirconium-doped mesoporous molecular sieves as catalysts for the hydrogenation and ring-opening of tetralin. Appl. Catal. A Gen. 2003, 240, 83–94. [Google Scholar] [CrossRef]
- Yamazaki, T.; Kikuchi, N.; Katoh, M.; Hirose, T.; Saito, H.; Yoshikawa, T.; Wada, M. Behavior of steam reforming reaction for bio-ethanol over Pt/ZrO2 catalysts. Appl. Catal. B Environ. 2010, 99, 81–88. [Google Scholar] [CrossRef]
- Profeti, L.P.; Dias, J.A.; Assaf, J.M.; Assaf, E.M. Hydrogen production by steam reforming of ethanol over Ni-based catalysts promoted with noble metals. J. Power Sources 2009, 190, 525–533. [Google Scholar] [CrossRef]
- De Diego, LF.; Ortiz, M.; García-Labiano, F.; Adánez, J.; Abad, A.; Gayán, P. Synthesis gas generation by chemical-looping reforming using a Nibased oxygen carrier. Energy Procedia 2009, 1, 3–10. [Google Scholar] [CrossRef]
- Akbari-Emadabadi, S.; Rahimpour, M.R.; Hafizi, A.; Keshavarz, P. Promotion of Ca-Co bifunctional catalyst/sorbent with yttrium for hydrogen production in modified chemical looping steam methane reforming. Catalysts 2017, 7, 270. [Google Scholar] [CrossRef]
- Antzara, A.; Heracleous, E.; Bukur, D.B.; Lemonidou, A.A. Thermodynamic analysis of hydrogen production via chemical looping steam methane reforming coupled with in situ CO2 capture. Int. J. Greenh. Gas Control 2015, 32, 115–128. [Google Scholar] [CrossRef]
- Adiya, Z.I.; Dupont, V.; Mahmud, T. Chemical equilibrium analysis of hydrogen production from shale gas using sorption enhanced chemical looping steam reforming. Fuel Process. Technol. 2017, 159, 128–144. [Google Scholar] [CrossRef]
- Silva, J.M.; Soria, M.A.; Madeira, L.M. Thermodynamic analysis of Glycerol Steam Reforming for hydrogen production with in situ hydrogen and carbon dioxide separation. J. Power Sources 2015, 273, 423–430. [Google Scholar] [CrossRef]
- He, H.; Dai, H.; Wong, K.W.; Au, C. RE0.6 Zr0.4−xYx O2 (RE = Ce, Pr; x = 0, 0.05) solid solutions: An investigation on defective structure, oxygen mobility, oxygen storage capacity, and redox properties. Appl. Catal. A Gen. 2003, 251, 61–74. [Google Scholar] [CrossRef]
- Oemar, U.; Hidajat, K.; Kawi, S. Role of catalyst support over PdO-NiO catalysts on catalyst activity and stability for oxy-CO2 reforming of methane. Appl. Catal. A Gen. 2011, 402, 176–187. [Google Scholar] [CrossRef]
- Belhadi, A.; Trari, M.; Rabia, C.; Cherifi, O. Methane steam reforming on supported nickel based catalysts. effect of oxide ZrO2, La2O3 and nickel composition. Open J. Phys. Chem. 2013, 3, 89–96. [Google Scholar] [CrossRef]
- Wan, H.; Li, X.; Ji, S.; Huang, B.; Wang, K.; Li, C. Effect of Ni loading and CexZri−xO2 promoter on Ni-based SBA-15 catalysts for steam reforming of methane. J. Nat. Gas Chem. 2007, 16, 139–147. [Google Scholar] [CrossRef]
- Rakass, S.; Oudghiri-Hassani, H.; Rowntree, P.; Abatzoglou, N. Steam reforming of methane over unsupported nickel catalysts. J. Power Sources 2006, 158, 485–496. [Google Scholar] [CrossRef]


| Samples | BET Surface Area (m2/g) | Pore Diameter (nm) | Pore Volume (cm3/g) | Crystal Size a (nm) |
|---|---|---|---|---|
| SBA-16 | 743.93 | 3.30 | 0.38 | - |
| 25Ni/SBA-16 | 321.99 | 3.68 | 0.25 | 42.91 |
| 25Ni-2.5Y/SBA-16 | 363.09 | 3.59 | 0.26 | 34.34 |
| Oxygen Carrier | BET Surface Area (m2/g) | Pore Diameter (nm) | Pore Volume (cm3/g) |
|---|---|---|---|
| 25Ni/SBA-16 | 58.26 | 4.78 | 0.08 |
| 25Ni-2.5Y/SBA-16 | 144.04 | 3.99 | 0.15 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daneshmand-Jahromi, S.; Rahimpour, M.R.; Meshksar, M.; Hafizi, A. Hydrogen Production from Cyclic Chemical Looping Steam Methane Reforming over Yttrium Promoted Ni/SBA-16 Oxygen Carrier. Catalysts 2017, 7, 286. https://doi.org/10.3390/catal7100286
Daneshmand-Jahromi S, Rahimpour MR, Meshksar M, Hafizi A. Hydrogen Production from Cyclic Chemical Looping Steam Methane Reforming over Yttrium Promoted Ni/SBA-16 Oxygen Carrier. Catalysts. 2017; 7(10):286. https://doi.org/10.3390/catal7100286
Chicago/Turabian StyleDaneshmand-Jahromi, Sanaz, Mohammad Reza Rahimpour, Maryam Meshksar, and Ali Hafizi. 2017. "Hydrogen Production from Cyclic Chemical Looping Steam Methane Reforming over Yttrium Promoted Ni/SBA-16 Oxygen Carrier" Catalysts 7, no. 10: 286. https://doi.org/10.3390/catal7100286