Recent Advances in the BiVO4 Photocatalyst for Sun-Driven Water Oxidation: Top-Performing Photoanodes and Scale-Up Challenges
Abstract
:1. Introduction
1.1. BiVO4 as a Photoanode
1.1.1. Advantages of BiVO4 as a Photoanode
1.1.2. Disadvantages of BiVO4 as a Photoanode
2. Top-Performing BiVO4-Based Photoanodes
2.1. Strategies Adopted to Enhance Reaction Kinetics in Solar Water Oxidation
2.2. Strategies Adopted to Enhance the Electron Mobility in Solar Water Oxidation
2.2.1. Morphology Control
Effect of Porosity and the Synthesis Procedure
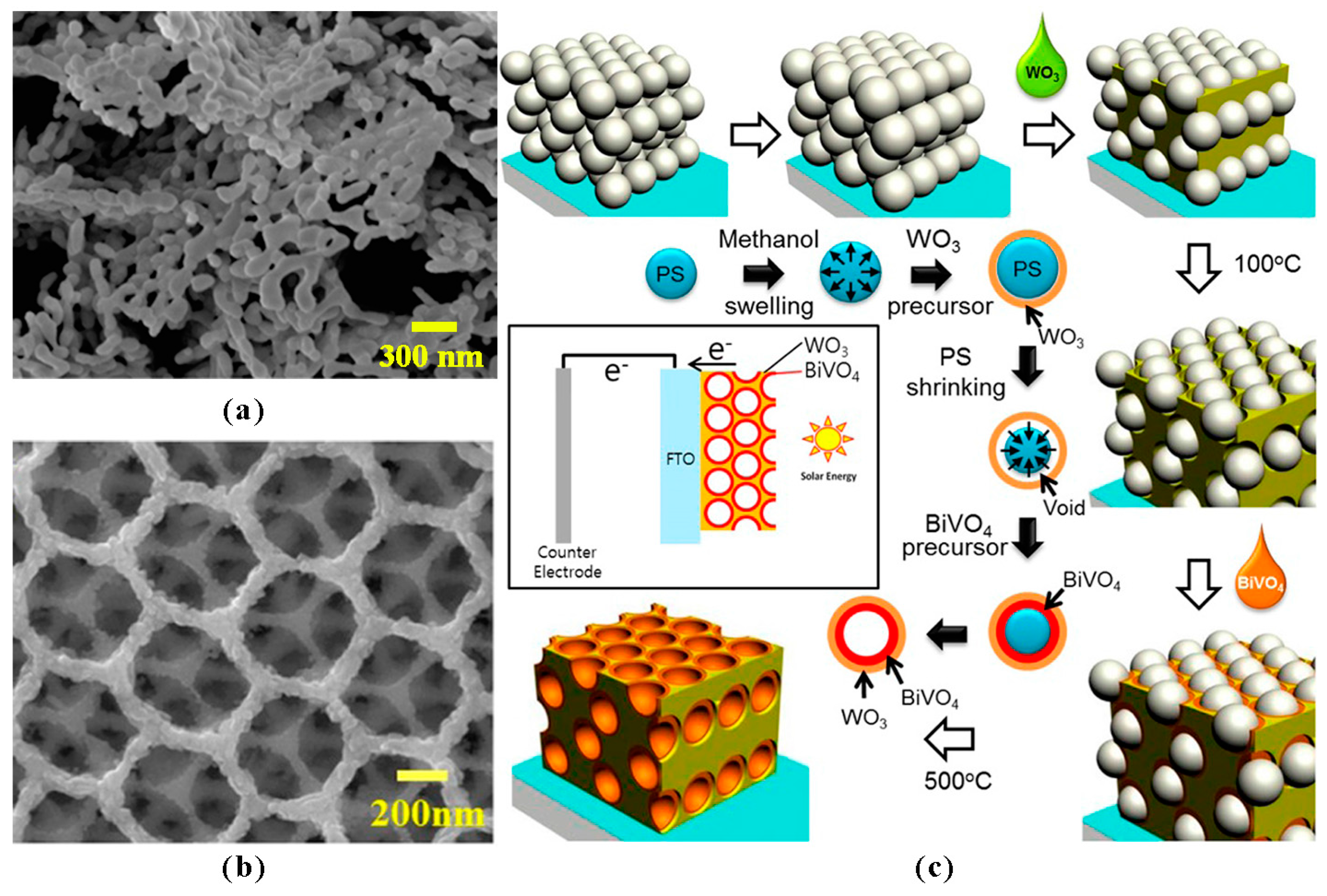
Effect of Ordered Structures/Inverse Opals
2.2.2. Addition of n-Type Conductivity Dopants
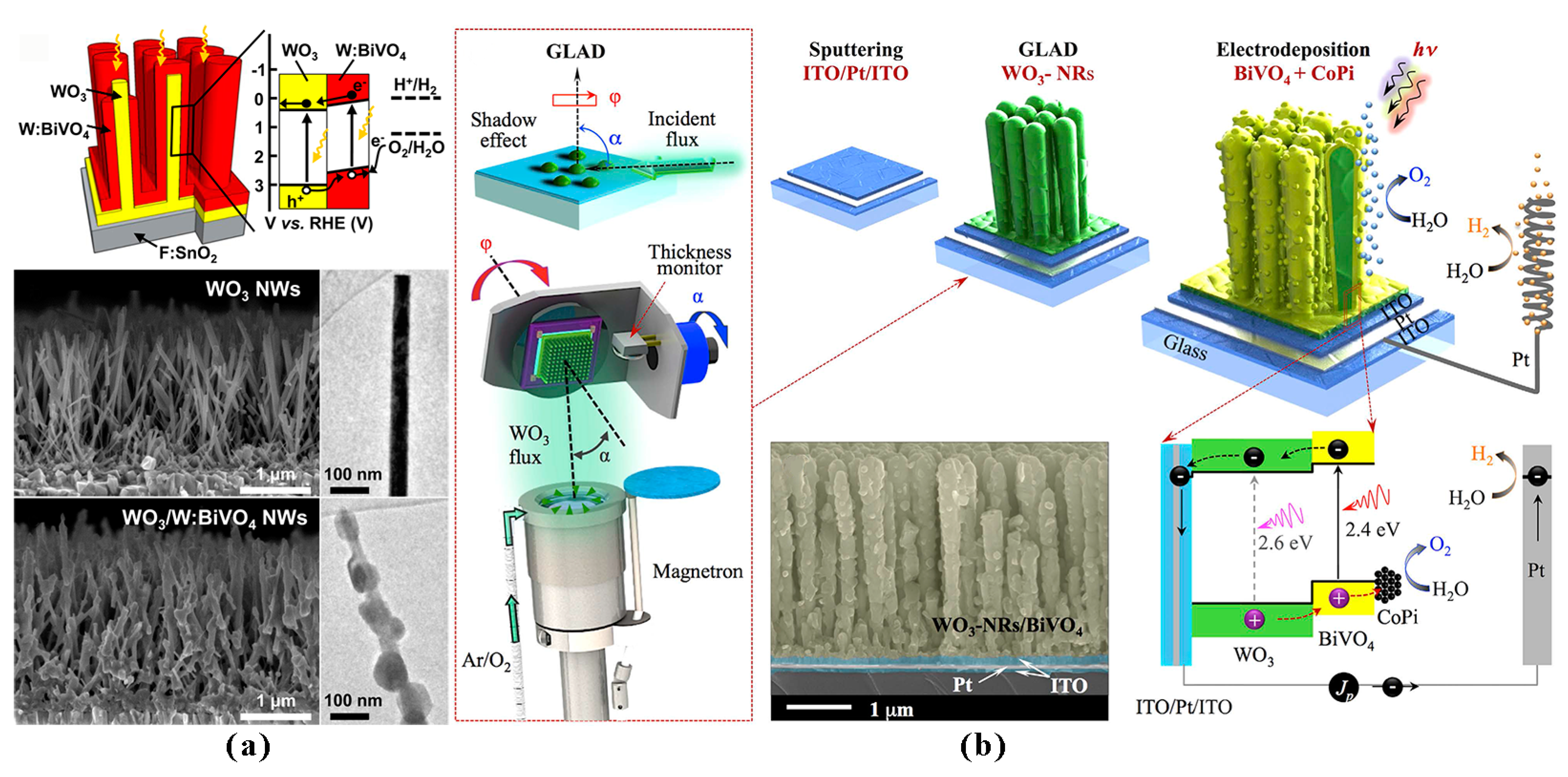
2.2.3. Heterojunction Formation
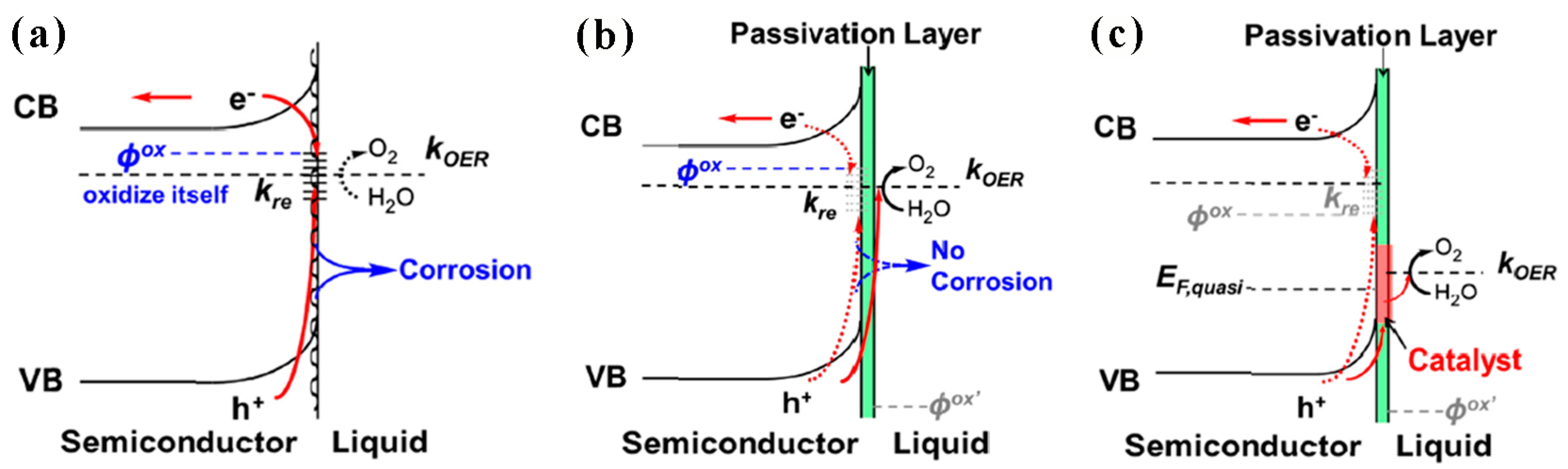
2.2.4. Use of Passivation Layers
2.2.5. Substrate Modification
2.3. Summary of BiVO4-Based Photoanodes with Photocurrent Densities > 3.5 mA/cm2 at 1.23 V vs. RHE
3. Critical Issues Hampering the Scale-Up of BiVO4-Based PECs
3.1. Synthesis Scalability
3.2. Oxygen Evolution Catalysts
3.3. Long-Term Stability
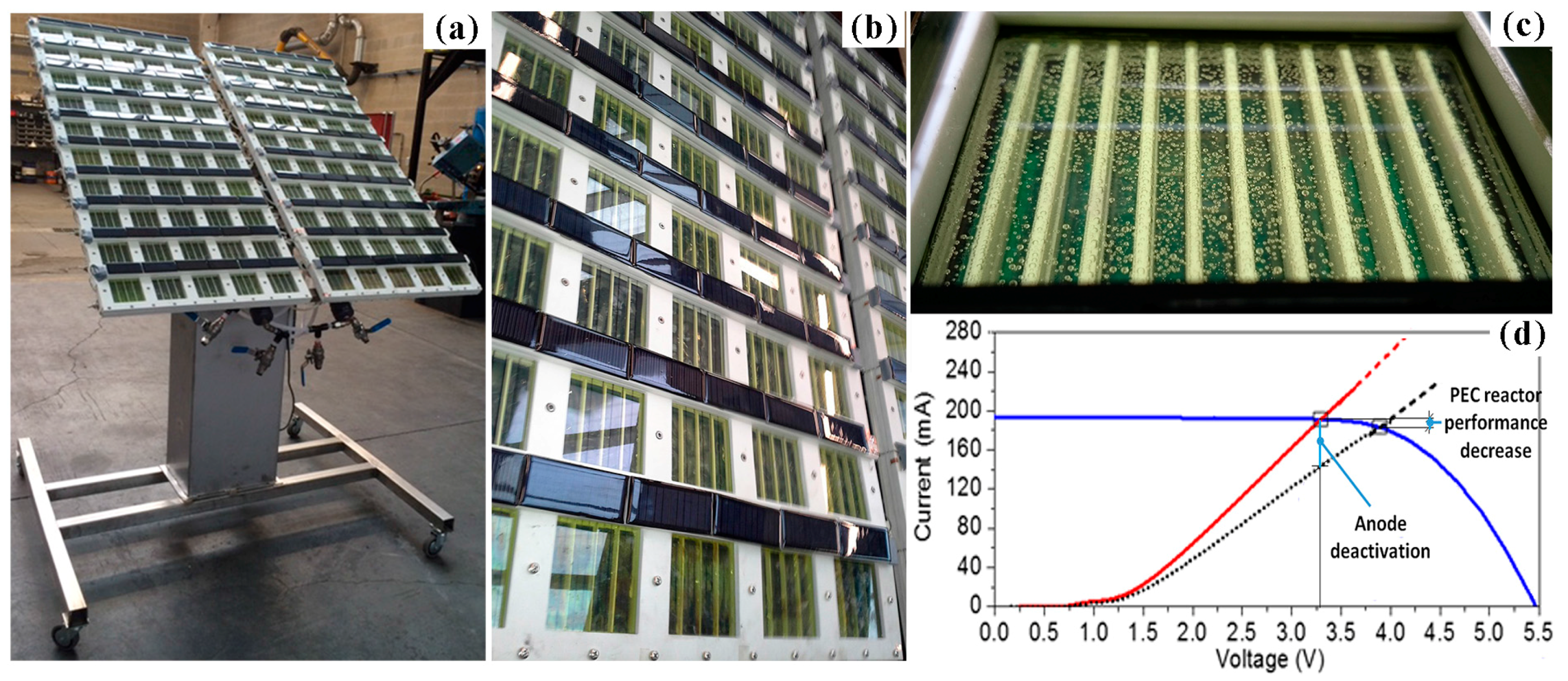
3.4. Large-Scale PEC Prototype Issues: The ARTIPHYCTION Experience
3.5. BiVO4-Photoanode PEC Tandem Device Assemblies
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gray, H.B. Powering the planet with solar fuel. Nat. Chem. 2009, 1, 7. [Google Scholar] [CrossRef] [PubMed]
- Von de Krol, R.; Grätzel, M. Photoelectrochemical Hydrogen Production. Electron. Mater. Sci. Technol. 2012, 102, 13–21. [Google Scholar]
- Chen, Z.; Dinh, H.N.; Miller, E. Introduction. In Photoelectrochemical Water Splitting; Springer: New York, NY, USA, 2013; pp. 1–5. [Google Scholar] [CrossRef]
- Walter, M.G.; Warren, E.L.; McKone, J.R.; Boettcher, S.W.; Mi, Q.; Santori, E.A.; Lewis, N.S. Solar Water Splitting Cells. Chem. Rev. 2010, 110, 6446–6473. [Google Scholar] [CrossRef] [PubMed]
- Martinez Suarez, C.; Hernández, S.; Russo, N. BiVO4 as photocatalyst for solar fuels production through water splitting: A short review. Appl. Catal. A Gen. 2015, 504, 158–170. [Google Scholar] [CrossRef]
- Fujishima, A.; Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef] [PubMed]
- Hurst, J.K. Catalysts for Solar Fuel Production. Science 2010, 328, 315–316. [Google Scholar] [CrossRef] [PubMed]
- Grätzel, M.; Moser, J. Solar Energy Conversion. In Electron Transfer in Chemistry; Wiley-VCH: Weiheim, Germany, 2001; Volume 5, pp. 589–644. [Google Scholar]
- Betley, T.A.; Wu, Q.; Van Voorhis, T.; Nocera, D.G. Electronic design criteria for O–O bond formation via metal-oxo complexes. Inorg. Chem. 2008, 47, 1849–1861. [Google Scholar] [CrossRef] [PubMed]
- Inoue, H.; Shimada, T.; Kou, Y.; Nabetani, Y.; Masui, D.; Takagi, S.; Tachibana, H. The water oxidation bottleneck in artificial photosynthesis: How can we get through it? An alternative route involving a two-electron process. ChemSusChem 2011, 4, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.; Kim, T.W.; Kubota, S.R.; Cardiel, A.C.; Cha, H.G.; Choi, K.S. Electrochemical Synthesis of Photoelectrodes and Catalysts for Use in Solar Water Splitting. Chem. Rev. 2015, 115, 12839–12887. [Google Scholar] [CrossRef] [PubMed]
- Young, K.J.; Martini, L.A.; Milot, R.L.; Snoeberger, R.C.; Batista, V.S.; Schmuttenmaer, C.A.; Crabtree, R.H.; Brudvig, G.W. Light-driven water oxidation for solar fuels. Coord. Chem. Rev. 2012, 256, 2503–2520. [Google Scholar] [CrossRef] [PubMed]
- Tachibana, Y.; Vayssieres, L.; Durrant, J.R. Artificial photosynthesis for solar water-splitting. Nat. Photonics 2012, 6, 511–518. [Google Scholar] [CrossRef]
- Blakemore, J.D.; Crabtree, R.H.; Brudvig, G.W. Molecular Catalysts for Water Oxidation. Chem. Rev. 2015, 115, 12974–13005. [Google Scholar] [CrossRef] [PubMed]
- Harriman, A.; Pickering, I.J.; Thomas, J.M.; Christensen, P.A. Metal Oxides as Heterogeneous Catalysts for Oxygen Evolution under Photochemical Conditions. J. Chem. Soc. Faraday Trans. 1 1988, 84, 2795–2806. [Google Scholar] [CrossRef]
- Kiwi, J.; Grätzel, M. Hydrogen evolution from water induced by visible light mediated by redox catalysis. Nature 1979, 281, 657–658. [Google Scholar] [CrossRef]
- Roth, R.; Waring, J. Synthesis and stability of bismutotantalite, stibiotantalite and chemically similar ABO4 compounds. Am. Miner. 1963, 18, 1348–1356. [Google Scholar]
- Lim, A.R.; Choh, S.H.; Jang, M.S. Prominent ferroelastic domain walls in BiVO4 crystal. J. Phys. Condens. Matter 1995, 7, 7309–7323. [Google Scholar] [CrossRef]
- Kudo, A.; Omori, K.; Kato, H. A Novel Aqueous Process for Preparation of Crystal Form-Controlled and Highly Crystalline BiVO4 Powder from Layered Vanadates at Room Temperature and Its Photocatalytic and Photophysical Properties. J. Am. Chem. Soc. 1999, 121, 11459–11467. [Google Scholar] [CrossRef]
- Bhattacharya, A.K. Phase transition in BiVO4. Mater. Lett. 1997, 30, 7–13. [Google Scholar] [CrossRef]
- Walsh, A.; Yan, Y.; Huda, M.N.; Al-Jassim, M.M.; Wei, S.-H. Band Edge Electronic Structure of BiVO4: Elucidating the Role of the Bi s and V d orbitals. Chem. Mater. 2009, 21, 547–551. [Google Scholar] [CrossRef]
- Cooper, J.K.; Gul, S.; Toma, F.M.; Chen, L.; Glans, P.-A.; Guo, J.; Ager, J.W.; Yano, J.; Sharp, I.D. Electronic Structure of Monoclinic BiVO4. Chem. Mater. 2014, 26, 5365–5373. [Google Scholar] [CrossRef]
- Kudo, A.; Ueda, K.; Kato, H.; Mikami, I. Photocatalytic O2 evolution under visible light irradiation on BiVO4 in aqueous AgNO3 solution. Catal. Lett. 1998, 53, 229–230. [Google Scholar] [CrossRef]
- Payne, D.J.; Robinson, M.D.M.; Egdell, R.G.; Walsh, A.; McNulty, J.; Smith, K.E.; Piper, L.F.J. The nature of electron lone pairs in BiVO4. Appl. Phys. Lett. 2011, 98, 46–49. [Google Scholar] [CrossRef] [Green Version]
- Hu, S.; Xiang, C.; Haussener, S.; Berger, A.D.; Lewis, N.S. An analysis of the optimal band gaps of light absorbers in integrated tandem photoelectrochemical water-splitting systems. Energy Environ. Sci. 2013, 6, 2984–2993. [Google Scholar] [CrossRef]
- Abdi, F.F.; Firet, N.; van de Krol, R. Efficient BiVO4 Thin Film Photoanodes Modified with Cobalt Phosphate Catalyst and W-doping. ChemCatChem 2013, 5, 490–496. [Google Scholar] [CrossRef]
- Park, Y.; McDonald, K.J.; Choi, K.S. Progress in bismuth vanadate photoanodes for use in solar water oxidation. Chem. Soc. Rev. 2013, 2321–2337. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.W.; Ping, Y.; Galli, G.A.; Choi, K.S. Simultaneous enhancements in photon absorption and charge transport of bismuth vanadate photoanodes for solar water splitting. Nat. Commun. 2015, 6, 8769. [Google Scholar] [CrossRef] [PubMed]
- McDonald, K.J.; Choi, K.S. A new electrochemical synthesis route for a BiOI electrode and its conversion to a highly efficient porous BiVO4 photoanode for solar water oxidation. Energy Environ. Sci. 2012, 5, 8553–8557. [Google Scholar] [CrossRef]
- Taylor, S.R. Abundance of chemical elements in the continental crust: A new table. Geochim. Cosmochim. Acta 1964, 28, 1273–1285. [Google Scholar] [CrossRef]
- Kronawitter, C.X.; Vayssieres, L.; Shen, S.; Guo, L.; Wheeler, D.A.; Zhang, J.Z.; Antoun, B.R.; Mao, S.S. A perspective on solar-driven water splitting with all-oxide hetero-nanostructures. Energy Environ. Sci. 2011, 4, 3889–3899. [Google Scholar] [CrossRef]
- Cho, S.; Jang, J.W.; Lee, K.H.; Lee, J.S. Research update: Strategies for efficient photoelectrochemical water splitting using metal oxide photoanodes. APL Mater. 2014, 2, 10703. [Google Scholar] [CrossRef]
- Ma, Y.; Pendlebury, S.R.; Reynal, A.; le Formal, F.; Durrant, J.R. Dynamics of photogenerated holes in undoped BiVO4 photoanodes for solar water oxidation. Chem. Sci. 2014, 5, 2964. [Google Scholar] [CrossRef]
- Sinclair, T.S.; Hunter, B.M.; Winkler, J.R.; Gray, H.B.; Astrid, M.M. Materials Horizons Factors affecting bismuth vanadate photoelectrochemical performance. Mater. Horiz. 2015, 22–24. [Google Scholar]
- Zhou, M.; Bao, J.; Xu, Y.; Zhang, J.; Xie, J.; Guan, M.; Wang, C.; Wen, L.; Lei, Y.; Xie, Y. Photoelectrodes based upon Mo:BiVO4 inverse opals for photoelectrochemical water splitting. ACS Nano 2014, 8, 7088–7098. [Google Scholar] [CrossRef] [PubMed]
- Abdi, F.F.; Savenije, T.J.; May, M.M.; Dam, B.; Van De Krol, R. The Origin of Slow Carrier Transport in BiVO4 Thin Film Photoanodes. J. Phys. Chem. Lett. 2013, 4, 2752–2757. [Google Scholar] [CrossRef]
- Retti, A.J.E.; Lee, H.C.; Marshall, L.G.; Lin, J.-F.; Capan, C.; Lindemuth, J.; McCloy, J.S.; Zhou, J.; Bard, A.J.; Mullins, C.B. Combined Charge Carrier Transport and Photoelectrochemical Characterization of BiVO4 Single Crystals: Intrinsic Behavior of a Complex Metal Oxide. J. Am. Chem. Soc. 2013, 135, 11389–11396. [Google Scholar] [CrossRef] [PubMed]
- Pihosh, Y.; Turkevych, I.; Mawatari, K.; Uemura, J.; Kazoe, Y.; Kosar, S.; Makita, K.; Sugaya, T.; Matsui, T.; Fujita, D.; et al. Photocatalytic generation of hydrogen by core-shell WO3/BiVO4 nanorods with ultimate water splitting efficiency. Sci. Rep. 2015, 5, 11141. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Jang, J.W.; Kang, H.J.; Magesh, G.; Kim, J.Y.; Kim, J.H.; Lee, J.; Lee, J.S. Palladium oxide as a novel oxygen evolution catalyst on BiVO4 photoanode for photoelectrochemical water splitting. J. Catal. 2014, 317, 126–134. [Google Scholar] [CrossRef]
- Wang, D.; Li, R.; Zhu, J.; Shi, J.; Han, J.; Zong, X.; Li, C. Photocatalytic water oxidation on BiVO4 with the electrocatalyst as an oxidation cocatalyst: Essential relations between electrocatalyst and photocatalyst. J. Phys. Chem. C 2012, 116, 5082–5089. [Google Scholar] [CrossRef]
- Seabold, J.A.; Choi, K.S. Efficient and stable photo-oxidation of water by a bismuth vanadate photoanode coupled with an iron oxyhydroxide oxygen evolution catalyst. J. Am. Chem. Soc. 2012, 134, 2186–2192. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Kang, D.; Choi, K.-S. Marked enhancement in electron-hole separation achieved in the low bias region using electrochemically prepared Mo-doped BiVO4 photoanodes. Phys. Chem. Chem. Phys. 2014, 16, 1238–1246. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.W.; Choi, K.-S. Nanoporous BiVO4 Photoanodes with Dual-Layer Oxygen Evolution Catalysts for Solar Water Splitting. Science 2014, 343, 990–994. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Choi, I.Y.; Zhang, K.; Kwon, J.; Kim, D.Y.; Lee, J.K.; Oh, S.H.; Kim, J.K.; Park, J.H. Efficient photoelectrochemical hydrogen production from bismuth vanadate-decorated tungsten trioxide helix nanostructures. Nat. Commun. 2014, 5, 4775. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Jeong, H.; Oh, S.J.; Ma, M.; Zhang, K.; Kwon, J.; Choi, I.T.; Choi, I.Y.; Kim, H.K.; Kim, J.K. Unassisted photoelectrochemical water splitting exceeding 7% solar-to-hydrogen conversion efficiency using photon recycling. Nat. Commun. 2016, 7, 11943. [Google Scholar] [CrossRef] [PubMed]
- Kanan, M.W.; Nocera, D.G. In situ formation of an oxygen-evolving catalyst in neutral water containing phosphate and Co2+. Science 2008, 321, 1072–1075. [Google Scholar] [CrossRef] [PubMed]
- Lutterman, D.A.; Surendranath, Y.; Nocera, D.G. A Self-Healing Oxygen-Evolving Catalyst. J. Am. Chem. Soc. 2009, 131, 3838–3839. [Google Scholar] [CrossRef] [PubMed]
- Abdi, F.F.; Han, L.; Smets, A.H.M.; Zeman, M.; Dam, B.; van de Krol, R. Efficient solar water splitting by enhanced charge separation in a bismuth vanadate-silicon tandem photoelectrode. Nat. Commun. 2013, 4, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zhong, D.K.; Gamelin, D.R. Photoelectrochemical Water Oxidation by Cobalt Catalyst (“Co-Pi”)α-Fe2O3 Composite Photoanodes Oxygen Evolution and Resolution of a Kinetic Bottleneck. J. Am. Chem. Soc. 2010, 132, 4202–4207. [Google Scholar] [CrossRef] [PubMed]
- Pilli, S.K.; Furtak, T.E.; Brown, L.D.; Deutsch, T.G.; Turner, J.A.; Herring, A.M. Cobalt-phosphate (Co-Pi) catalyst modified Mo-doped BiVO4 photoelectrodes for solar water oxidation. Energy Environ. Sci. 2011, 4, 5028. [Google Scholar] [CrossRef]
- Zhong, D.K.; Choi, S.; Gamelin, D.R. Near-Complete Suppression of Surface Recombination in Solar Photoelectrolysis by “Co-Pi” Catalyst-Modified W:BiVO4. J. Am. Chem. Soc. 2011, 133, 18370–18377. [Google Scholar] [CrossRef] [PubMed]
- Zhong, M.; Hisatomi, T.; Kuang, Y.; Zhao, J.; Liu, M.; Iwase, A.; Jia, Q.; Nishiyama, H.; Minegishi, T.; Nakabayashi, M.; et al. Surface Modification of CoOx Loaded BiVO4 Photoanodes with Ultrathin p-Type NiO Layers for Improved Solar Water Oxidation. J. Am. Chem. Soc. 2015, 137, 5053–5060. [Google Scholar] [CrossRef] [PubMed]
- Joya, K.S.; Takanabe, K.; De Groot, H.J.M. Surface generation of a cobalt-derived water oxidation electrocatalyst developed in a neutral HCO3−/CO2 system. Adv. Energy Mater. 2014, 4, 2–7. [Google Scholar]
- Kim, J.H.; Magesh, G.; Kang, H.J.; Banu, M.; Kim, J.H.; Lee, J.; Lee, J.S. Carbonate-coordinated cobalt Co-catalyzed BiVO4/WO3 composite photoanode tailored for CO2 reduction to fuels. Nano Energy 2015, 15, 153–163. [Google Scholar] [CrossRef]
- Kim, J.H.; Jo, Y.; Kim, J.H.; Jang, J.W.; Kang, H.J.; Lee, Y.H.; Kim, D.S.; Jun, Y.; Lee, J.S. Wireless Solar Water Splitting Device with Robust Cobalt-Catalyzed, and Perovskite Solar Cell in Tandem: A Dual Absorber Artificial Leaf. ACS Nano 2015, 9, 11820–11829. [Google Scholar] [CrossRef] [PubMed]
- McCrory, C.C.L.; Jung, S.; Peters, J.C.; Jaramillo, T.F. Benchmarking heterogeneous electrocatalysts for the oxygen evolution reaction. J. Am. Chem. Soc. 2013, 135, 16977–16987. [Google Scholar] [CrossRef] [PubMed]
- McCrory, C.C.L.; Jung, S.; Ferrer, I.M.; Chatman, S.M.; Peters, J.C.; Jaramillo, T.F. Benchmarking Hydrogen Evolving Reaction and Oxygen Evolving Reaction Electrocatalysts for Solar Water Splitting Devices. J. Am. Chem. Soc. 2015, 137, 4347–4357. [Google Scholar] [CrossRef] [PubMed]
- Pickrahn, K.L.; Park, S.W.; Gorlin, Y.; Lee, H.B.R.; Jaramillo, T.F.; Bent, S.F. Active MnOx Electrocatalysts Prepared by Atomic Layer Deposition for Oxygen Evolution and Oxygen Reduction Reactions. Adv. Energy Mater. 2012, 2, 1269–1277. [Google Scholar] [CrossRef]
- Jia, Q.; Iwashina, K.; Kudo, A. Facile fabrication of an efficient BiVO4 thin film electrode for water splitting under visible light irradiation. Proc. Natl. Acad. Sci. USA 2012, 109, 11564–11569. [Google Scholar] [CrossRef] [PubMed]
- Hernández, S.; Gerardi, G.; Bejtka, K.; Fina, A.; Russo, N. Evaluation of the charge transfer kinetics of spin-coated BiVO4 thin films for sun-driven water photoelectrolysis. Appl. Catal. B Environ. 2016, 190, 66–74. [Google Scholar] [CrossRef]
- Thalluri, S.M.; Rojas, R.M.; Rivera, O.D.; Hernandez, S.; Russo, N.; Rodil, S.E. Chemically induced porosity on BiVO4 films produced by double magnetron sputtering to enhance the photo-electrochemical response. Phys. Chem. Chem. Phys. 2015, 17, 17821–17827. [Google Scholar] [CrossRef] [PubMed]
- Thalluri, S.M.; Martinez Suarez, C.; Hussain, M.; Hernandez, S.; Virga, A.; Saracco, G.; Russo, N. Evaluation of the Parameters Affecting the Visible-Light-Induced Photocatalytic Activity of Monoclinic BiVO4 for Water Oxidation. Ind. Eng. Chem. Res. 2013, 52, 17414–17418. [Google Scholar] [CrossRef]
- Thalluri, S.M.; Martinez Suarez, C.; Hernández, S.; Bensaid, S.; Saracco, G.; Russo, N. Elucidation of important parameters of BiVO4 responsible for photo-catalytic O2 evolution and insights about the rate of the catalytic process. Chem. Eng. J. 2014, 245, 124–132. [Google Scholar] [CrossRef]
- Hernández, S.; Thalluri, S.M.; Sacco, A.; Bensaid, S.; Saracco, G.; Russo, N. Photo-catalytic activity of BiVO4 thin-film electrodes for solar-driven water splitting. Appl. Catal. A Gen. 2015, 504, 266–271. [Google Scholar] [CrossRef]
- Ma, M.; Kim, J.K.; Zhang, K.; Shi, X.; Kim, S.J.; Moon, J.H.; Park, J.H. Double-deck Inverse Opal Photoanodes: Efficient Light Absorption and Charge Separation in Heterojunction. Chem. Mater. 2014, 26, 5592–5597. [Google Scholar] [CrossRef]
- Berglund, S.P.; Rettie, A.J.E.; Hoang, S.; Mullins, C.B. Incorporation of Mo and W into nanostructured BiVO4 films for efficient photoelectrochemical water oxidation. Phys. Chem. Chem. Phys. 2012, 14, 7065–7075. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Li, Z.; Yu, T.; Zou, Z. Effects of surface electrochemical pretreatment on the photoelectrochemical performance of Mo-doped BiVO4. J. Phys. Chem. C 2012, 116, 5076–5081. [Google Scholar] [CrossRef]
- Park, H.S.; Kweon, K.E.; Ye, H.; Paek, E.; Hwang, G.S. Factors in the Metal Doping of BiVO4 for Improved Photoelectrocatalytic Activity as Studied by Scanning Electrochemical Microscopy (SECM) and First-Principles Density-Functional Calculation. J. Phys. Chem. C 2011, 115, 17870–17879. [Google Scholar] [CrossRef]
- Jo, W.J.; Jang, J.W.; Kong, K.J.; Kang, H.J.; Kim, J.Y.; Jun, H.; Parmar, K.P.S.; Lee, J.S. Phosphate doping into monoclinic BiVO4 for enhanced photoelectrochemical water oxidation activity. Angew. Chem. Int. Ed. 2012, 51, 3147–3151. [Google Scholar] [CrossRef] [PubMed]
- Parmar, K.P.S.; Kang, H.J.; Bist, A.; Dua, P.; Jang, J.S.; Lee, J.S. Photocatalytic and photoelectrochemical water oxidation over metal-doped monoclinic BiVO4 photoanodes. ChemSusChem 2012, 5, 1926–1934. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Yang, Z.; Li, Z.; Zhang, J.; Liu, J.; Zhao, Z.; Wang, Z.; Yan, S.; Yu, T.; Zhou, Z. Solar hydrogen generation from seawater with a modified BiVO4 photoanode. Energy Environ. Sci. 2011, 4, 4046–4051. [Google Scholar] [CrossRef]
- Jeong, H.W.; Jeon, T.H.; Jang, J.S.; Choi, W.; Park, H. Strategic modification of BiVO4 for improving photoelectrochemical water oxidation performance. J. Phys. Chem. C 2013, 117, 9104–9112. [Google Scholar] [CrossRef]
- He, H.; Berglund, S.P.; Rettie, A.J.E.; Chemelewski, W.D.; Xiao, P.; Zhang, Y.; Mullins, C.B. Synthesis of BiVO4 nanoflake array films for photoelectrochemical water oxidation. J. Mater. Chem. A 2014, 2, 9371–9379. [Google Scholar] [CrossRef]
- Yao, W.; Iwai, H.; Ye, J. Effects of molybdenum substitution on the photocatalytic behavior of BiVO4. Dalton Trans. 2008, 1426–1430. [Google Scholar] [CrossRef] [PubMed]
- Thalluri, S.M.; Hernández, S.; Bensaid, S.; Saracco, G.; Russo, N. Green-synthesized W- and Mo-doped BiVO4 oriented along the {040} facet with enhanced activity for the sun-driven water oxidation. Appl. Catal. B Environ. 2016, 180, 630–636. [Google Scholar] [CrossRef]
- Pattengale, B.; Ludwig, J.; Huang, J. Atomic Insight into the W-Doping Effect on Carrier Dynamics and Photoelectrochemical Properties of BiVO4 Photoanodes. J. Phys. Chem. C 2016, 120, 1421–1427. [Google Scholar] [CrossRef]
- Gong, H.; Freudenberg, N.; Nie, M.; Van De Krol, R.; Ellmer, K. BiVO4 photoanodes for water splitting with high injection efficiency, deposited by reactive magnetron co-sputtering. AIP Adv. 2016, 6, 45108. [Google Scholar] [CrossRef]
- Monfort, O.; Sfaelou, S.; Satrapinskyy, L.; Plecenik, T.; Roch, T.; Plesch, G.; Lianos, P. Comparative study between pristine and Nb-modified BiVO4 films employed for photoelectrocatalytic production of H2 by water splitting and for the photocatalytic degradation of organic pollutants under simulated solar light. Catal. Today 2016, 280, 51–57. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, L.; Chen, Z.; Hu, J.; Li, S.; Wang, Z.; Liu, J.; Wang, X. Semiconductor heterojunction photocatalysts: Design, construction, and photocatalytic performances. Chem. Soc. Rev. 2014, 43, 5234–5244. [Google Scholar] [CrossRef] [PubMed]
- Su, J.; Guo, L.; Bao, N.; Grimes, C.A. Nanostructured WO3/BiVO4 heterojunction films for efficient photoelectrochemical water splitting. Nano Lett. 2011, 11, 1928–1933. [Google Scholar] [CrossRef] [PubMed]
- Murcia-López, S.; Fàbrega, C.; Monllor-Satoca, D.; Hernández-Alonso, M.D.; Penelas-Pérez, G.; Morata, A.; Morante, J.R.; Andreu, T. Tailoring Multilayered BiVO4 Photoanodes by Pulsed Laser Deposition for Water Splitting. ACS Appl. Mater. Interfaces 2016, 8, 4076–4085. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Reisner, E.; Baumberg, J.J. Al-doped ZnO inverse opal networks as efficient electron collectors in BiVO4 photoanodes for solar water oxidation. Energy Environ. Sci. 2014, 7, 1402. [Google Scholar] [CrossRef]
- Pilli, S.K.; Deutsch, T.G.; Furtak, T.E.; Brown, L.D.; Turner, J.A.; Herring, A.M. BiVO4/CuWO4 heterojunction photoanodes for efficient solar driven water oxidation. Phys. Chem. Chem. Phys. 2013, 15, 3273–3278. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.M.; Cai, L.; Liu, C.; Cho, I.S.; Lee, C.H.; Weisse, J.M.; Yang, P.; Zheng, X. Simultaneously Efficient Light Absorption and Charge Separation in WO3/BiVO4 Core/Shell Nanowire Photoanode for Photoelectrochemical Water Oxidation. Nano Lett. 2014, 14, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Zheng, Z.; Spurgeon, J.; Yang, X. Enhanced photoelectrochemical water-splitting performance of semiconductors by surface passivation layers. Energy Environ. Sci. 2014, 7, 2504–2517. [Google Scholar] [CrossRef]
- Eisenberg, D.; Ahn, H.S.; Bard, A.J. Enhanced Photoelectrochemical Water Oxidation on Bismuth. J. Am. Chem. Soc. 2014, 136, 14011–14014. [Google Scholar] [CrossRef] [PubMed]
- Mcdowell, M.T.; Lichterman, M.F.; Spurgeon, J.M.; Hu, S.; Sharp, I.D.; Brunschwig, B.S.; Lewis, N.S. Improved Stability of Polycrystalline Bismuth Vanadate Photoanodes by Use of Dual-Layer Thin TiO2/Ni Coatings. J. Phys. Chem. C 2014, 118, 19618–19624. [Google Scholar] [CrossRef]
- Kim, T.W.; Choi, K.S. Improving Stability and Photoelectrochemical Performance of BiVO4 Photoanodes in Basic Media by Adding a ZnFe2O4 Layer. J. Phys. Chem. Lett. 2016, 7, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Guo, Y.; Cai, L.; Li, H.; Wang, K.X.; Cho, I.S.; Lee, C.H.; Fan, S.; Zheng, X. High-Performance Ultrathin BiVO4 Photoanode on Textured Polydimethylsiloxane Substrates for Solar Water Splitting. ACS Energy Lett. 2016, 1, 68–75. [Google Scholar] [CrossRef]
- Qiu, Y.; Liu, W.; Chen, W.; Chen, W.; Zhou, G.; Hsu, P.; Zhang, R.; Liang, Z.; Fan, S.; Zhang, Y.; et al. Efficient solar-driven water splitting by nanocone BiVO4-perovskite tandem cells. Sci. Adv. 2016, 2, e1501764. [Google Scholar] [CrossRef] [PubMed]
- Arsenault, E.; Soheilnia, N.; Ozin, G.A. Periodic macroporous nanocrystalline antimony-doped tin oxide electrode. ACS Nano 2011, 5, 2984–2988. [Google Scholar] [CrossRef] [PubMed]
- Long, X.; Li, J.; Xiao, S.; Yan, K.; Wang, Z.; Chen, H.; Yang, S. A strongly coupled graphene and FeNi double hydroxide hybrid as an excellent electrocatalyst for the oxygen evolution reaction. Angew. Chem. Int. Ed. 2014, 53, 7584–7588. [Google Scholar] [CrossRef] [PubMed]
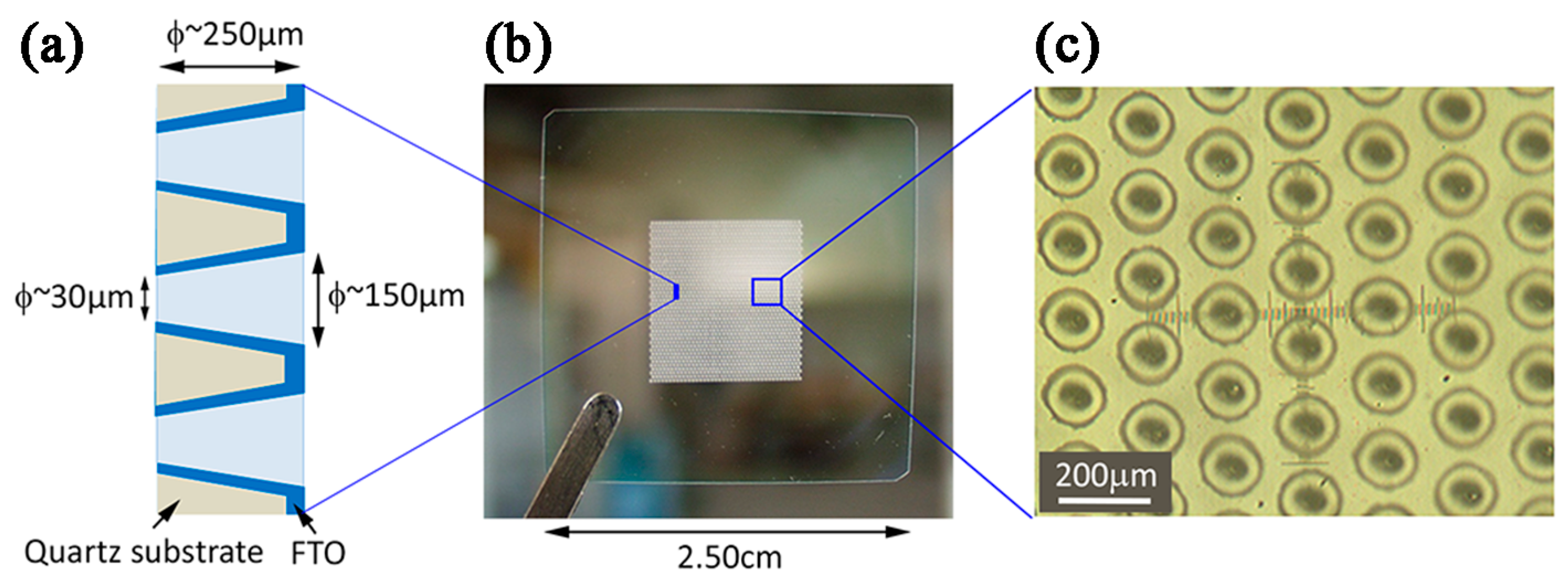
- Hernández, S.; Tortello, M.; Sacco, A.; Quaglio, M.; Meyer, T.; Bianco, S.; Saracco, G.; Pirri, C.F.; Tresso, E. New Transparent Laser-Drilled Fluorine-doped Tin Oxide covered Quartz Electrodes for Photo-Electrochemical Water Splitting. Electrochim. Acta 2014, 131, 184–194. [Google Scholar] [CrossRef]
- Hernández, S.; Saracco, G.; Alexe-Ionescu, A.L.; Barbero, G. Electric investigation of a photo-electrochemical water splitting device based on a proton exchange membrane within drilled FTO-covered quartz electrodes: Under dark and light conditions. Electrochim. Acta 2014, 144, 352–360. [Google Scholar] [CrossRef]
- Pinaud, B.A.; Benck, J.D.; Seitz, L.C.; Forman, A.J.; Chen, Z.; Deutsch, T.G. Technical and economic feasibility of centralized facilities for solar hydrogen production via photocatalysis and photoelectrochemistry. Energy Environ. Sci. 2013, 6, 1983–2002. [Google Scholar] [CrossRef]
- Mckone, J.R.; Lewis, N.S.; Gray, H.B. Will Solar-Driven Water-Splitting Devices See the Light of Day? Chem. Mater. 2013, 26, 407–414. [Google Scholar] [CrossRef]
- Bard, A.J.; Fox, M.A. Artificial Photosynthesis: Solar Splitting of Water to Hydrogen and Oxygen Water Splitting. Acc. Chem. Res. 1995, 28, 141–145. [Google Scholar] [CrossRef]
- Park, H.S.; Lee, H.C.; Leonard, K.C.; Liu, G.; Bard, A.J. Unbiased photoelectrochemical water splitting in Z-scheme device using W/Mo-doped BiVO4 and ZnxCd1−xSe. ChemPhysChem 2013, 14, 2277–2287. [Google Scholar] [CrossRef] [PubMed]
- Toma, F.M.; Cooper, J.K.; Kunzelmann, V.; McDowell, M.T.; Yu, J.; Larson, D.M.; Borys, N.J.; Abelyan, C.; Beeman, J.W.; Yu, K.M.; et al. Mechanistic insights into chemical and photochemical transformations of bismuth vanadate photoanodes. Nat. Commun. 2016, 7, 12012. [Google Scholar] [CrossRef] [PubMed]
- Artiphyction. Available online: http://www.artiphyction.org (accessed on 20 November 2016).
- Gliozzi, A.S.; Hernández, S.; Alexe-Ionescu, A.L.; Saracco, G.; Barbero, G. A model for electrode effects based on adsorption theory. Electrochim. Acta 2015, 178, 280–286. [Google Scholar] [CrossRef]
- Hernández, S.; Barbero, G.; Saracco, G.; Alexe-Ionescu, A.L. Considerations on oxygen bubble formation and evolution on BiVO4 porous anodes used in water splitting photoelectrochemical cells. J. Phys. Chem. C 2015, 119, 9916–9925. [Google Scholar] [CrossRef]
- Olateju, B.; Kumar, A. Techno-economic assessment of hydrogen production from underground coal gasification (UCG) in Western Canada with carbon capture and sequestration (CCS) for upgrading bitumen from oil sands. Appl. Energy 2013, 111, 428–440. [Google Scholar] [CrossRef]
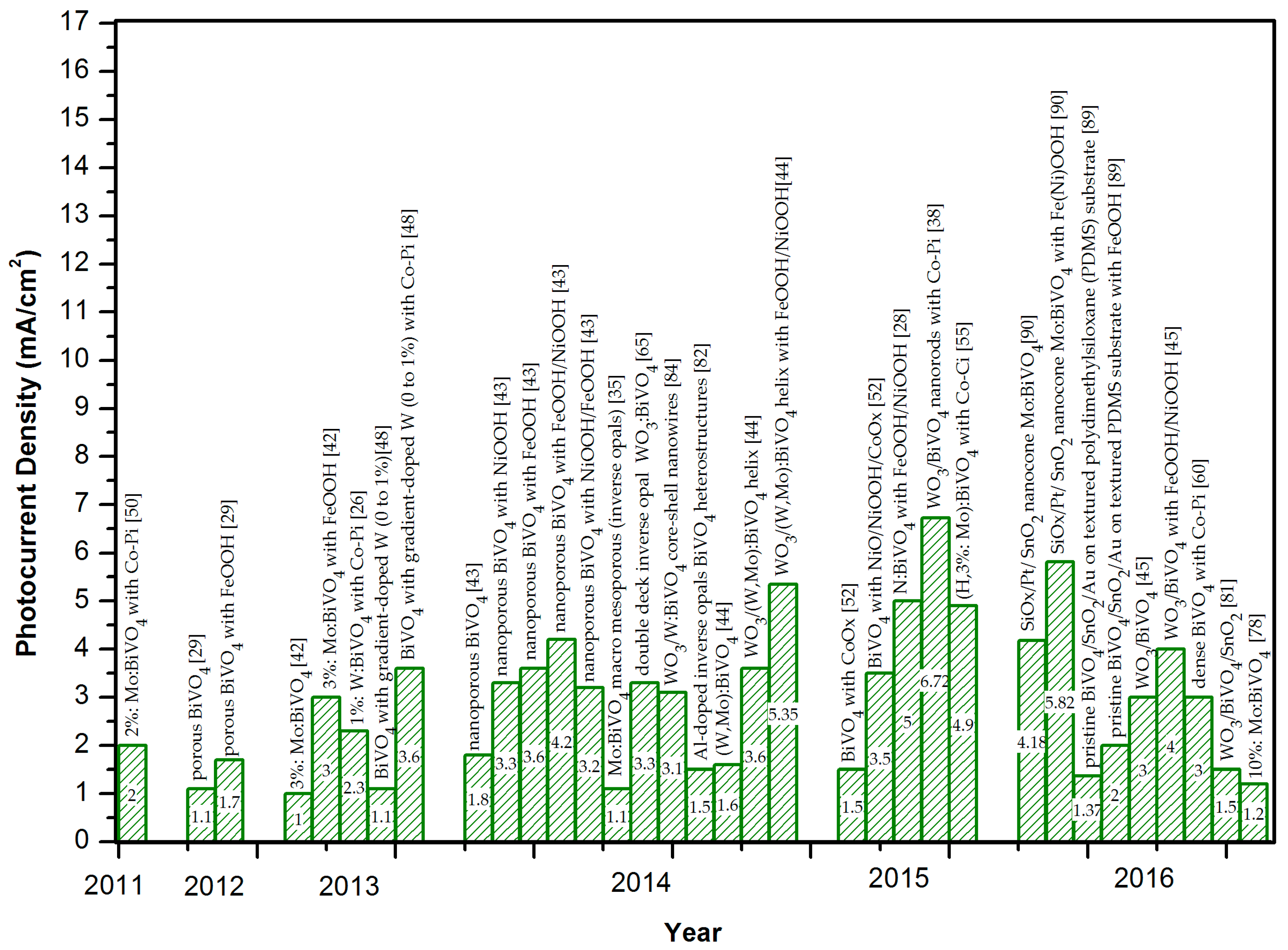

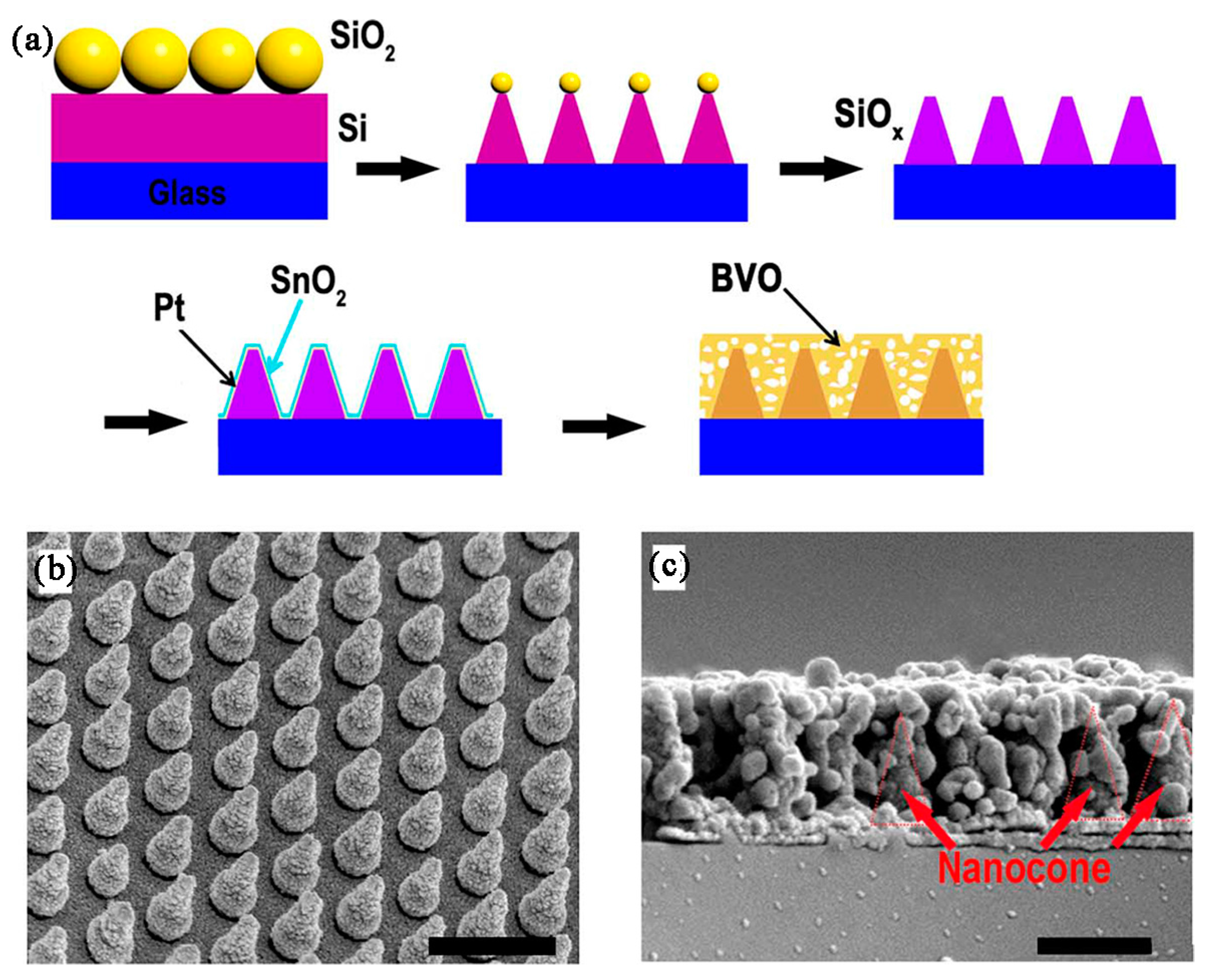
| Photoanode | Photocurrent Density 1 (mA/cm2) | Illuminated Area (cm2) | Efficiency | Stability | Ref. |
|---|---|---|---|---|---|
| WO3/BiVO4 nanorods with Co-Pi | 6.72 | Unspecified. Electrode Area: 0.226 | IPCE 80%–90%, 400–500 nm at 1 V vs. RHE | Not reported | [38] |
| SiOx/Pt/SnO2 Mo:BiVO4 nanocone with Fe(Ni)OOH | 5.82 | 0.25 | IPCE > 75%, <460 nm, at 1.23 V vs. RHE; APBE ~ 2.05% at 0.62 V vs. RHE | About 5.8 mA/cm2 at 1.23 V vs. RHE for 5 h | [90] |
| WO3/(W, Mo):BiVO4 helix nanostructures with FeOOH/NiOOH | 5.35 | Unspecified. Electrode Area: 2.25 | IPCE 90%, 330–450 nm at 1.23 V vs. RHE | About 3 mA/cm2 at 1.23 V vs. RHE for 7 days (8.7% decay) | [44] |
| N:BiVO4 with FeOOH/NiOOH | 5.0 | 0.1–0.2 | IPCE 50%–60%, 400–470 nm at 0.6 V vs. RHE | About 3.2 mA/cm2 at 0.6 V vs. RHE for 30 h | [28] |
| (H, 3% Mo):BiVO4 with Co-Ci | 4.9 | Unspecified | IPCE ~80%, 420 nm at 1.23 V vs. RHE | About 3.5 mA/cm2 at 1.03 V vs. RHE for 12 h | [55] |
| Nanoporous BiVO4 with FeOOH/NiOOH | 4.2 | 0.2 | APBE 2.2% at 0.58 V vs. CE (Pt) | 2.73 mA/cm2 at 0.6 V vs. RHE for 48 h | [43] |
| SiOx/Pt/SnO2 Mo:BiVO4 nanocone | 4.18 | 0.25 | ABPE about 0.75% at 0.87 V vs. RHE | Not reported | [90] |
| WO3/(W, Mo):BiVO4 with FeOOH/NiOOH | 4.0 | Unspecified. Electrode area: 2.25 | Not reported | Not reported | [45] |
| W(0%–1%) gradient-doped BiVO4 with Co-Pi | 3.6 | Unspecified | Carrier separation efficiency of up to 80% | Not reported | [48] |
| Nanoporous BiVO4 with FeOOH | 3.6 | 0.2 | Not reported | Not reported | [43] |
| BiVO4 Photoanode | In Tandem with | STH Efficiency | Photocurrent Density 1 (mA/cm2) | Stability | Ref. |
|---|---|---|---|---|---|
| WO3/BiVO4 nanorods with Co-Pi | 2-jn GaAs/InGaAsP | 8.1% | 6.56 | Tested for 1 h, sustained for 1 h | [38] |
| WO3/(W, Mo):BiVO4 with FeOOH/NiOOH | Hybrid cDBR | 7.1% | 5.7 | Tested for 10 h, sustained for 10 h | [45] |
| SiOx/Pt/SnO2 Mo:BiVO4 nanocone with Fe(Ni)OOH | perovskite solar cell | 6.2% | 5.82 | 5.8% decay over 10 h | [90] |
| W(0%–1%) gradient-doped BiVO4 with Co-Pi | 2-jn a-Si PV | 4.9% | 4.0 | Tested for 1 h, sustained for 1 h | [48] |
| (H, 3% Mo):BiVO4 with Co-Ci | CH3NH3PbI3 perovskite single jn | Wired: 4%Wireless: 3% | 4.8 | Tested for 10 h, sustained for 10 h | [55] |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tolod, K.R.; Hernández, S.; Russo, N. Recent Advances in the BiVO4 Photocatalyst for Sun-Driven Water Oxidation: Top-Performing Photoanodes and Scale-Up Challenges. Catalysts 2017, 7, 13. https://doi.org/10.3390/catal7010013
Tolod KR, Hernández S, Russo N. Recent Advances in the BiVO4 Photocatalyst for Sun-Driven Water Oxidation: Top-Performing Photoanodes and Scale-Up Challenges. Catalysts. 2017; 7(1):13. https://doi.org/10.3390/catal7010013
Chicago/Turabian StyleTolod, Kristine Rodulfo, Simelys Hernández, and Nunzio Russo. 2017. "Recent Advances in the BiVO4 Photocatalyst for Sun-Driven Water Oxidation: Top-Performing Photoanodes and Scale-Up Challenges" Catalysts 7, no. 1: 13. https://doi.org/10.3390/catal7010013








